Inspire Medical Gets FDA Approval for Full-Body MRIs
July 06 2022 - 8:57AM
Dow Jones News
By Dean Seal
Inspire Medical Systems Inc. said the Food and Drug
Administration has granted approval for the use of full-body
magnetic resonance imaging scans in conjunction with its sleep
apnea therapy.
The Golden Valley, Minn.-based company, which develops
treatments for patients with obstructive sleep apnea, previously
had approval to use MRI scans for only the head, neck and
extremities.
The expansion to full-body capabilities is retroactive, meaning
it will apply to all patients that already have the company's
Inspire IV neurostimulator device in place.
"Providing the full range of scan options enables us to better
help all current and future patients with their imaging needs,"
Chief Executive Tim Herbert said.
Write to Dean Seal at dean.seal@wsj.com
(END) Dow Jones Newswires
July 06, 2022 08:42 ET (12:42 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
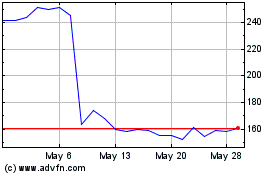
Inspire Medical Systems (NYSE:INSP)
Historical Stock Chart
From Mar 2024 to Apr 2024
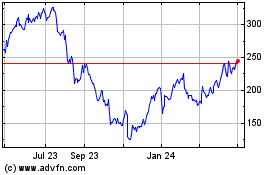
Inspire Medical Systems (NYSE:INSP)
Historical Stock Chart
From Apr 2023 to Apr 2024