GeoVax to Present at the H.C. Wainwright Bioconnect 2021 Virtual Conference
January 05 2021 - 9:00AM

via NewMediaWire -- GeoVax Labs, Inc. (NasdaqCM: GOVX), a
biotechnology company developing vaccines and immunotherapies
against infectious diseases and cancers, announced today that
Chairman, President and CEO David Dodd will present at the H.C.
Wainwright Bioconnect 2021 Virtual Conference taking place January
11-14, 2021.
The presentation will be available for on-demand listening
beginning at 6:00 AM Eastern Time on Monday, January 11, 2021,
through close of business January 14, 2021. For those
interested in viewing Mr. Dodd’s presentation, registration can be
accessed here.
The Company also announced that it will participate in virtual
one-on-one meetings with registered biopharmaceutical companies,
investors and other interested parties through the one-on-one
partnering system hosted by the Biotechnology Innovation
Organization (BIO) during the J.P. Morgan conference week January
11-15, 2021. Registration can be accessed here.
About GeoVax
GeoVax Labs, Inc. is a clinical-stage biotechnology company
developing human vaccines against infectious diseases and cancers
using a novel patented Modified Vaccinia Ankara-Virus Like Particle
(MVA-VLP) based vaccine platform. On this platform, MVA, a large
virus capable of carrying multiple vaccine antigens, expresses
proteins that assemble into VLP immunogens within (in vivo) the
person receiving the vaccine. The production of VLPs in the person
being vaccinated mimics virus production in a natural infection,
stimulating both the humoral and cellular arms of the immune system
to recognize, prevent, and control the target infection. The
MVA-VLP derived vaccines elicit durable immune responses in the
host similar to a live-attenuated virus, while providing the safety
characteristics of a replication-defective vector. The goal of
the GeoVax technology (GV-MVA-VLPTM) is to provide high efficacy in
a single dose, without the need for additional immune response
components (“adjuvants”), strong durability (“minimizing the need
for boosters”), the highly-validated safety of MVA, minimal
refrigeration requirements, and cost-effective manufacturing.
GeoVax’s current development programs are focused on preventive
vaccines against COVID-19, Zika Virus, hemorrhagic fever viruses
(Ebola, Lassa, Marburg and Sudan), HIV and malaria, as well as
therapeutic vaccines against multiple cancers. The Company has
successfully completed six human clinical trials related to HIV,
having designed a preventive HIV vaccine candidate to fight against
the subtype of HIV prevalent in the geographic markets of the
Americas, Western Europe, Japan, and Australia. Human clinical
trials for this program are managed by the HIV Vaccine Trials
Network (HVTN) with the support of the National Institutes of
Health (NIH). GeoVax’s HIV vaccine is also part of two separate
collaborative efforts to apply its innovative approach toward a
functional cure for HIV, addressing those already infected with
HIV.
Forward-Looking Statements
This release contains forward-looking statements regarding the
timing of the release of GeoVax’s financial results and earnings
conference call and the earnings release and call are expected to
discuss forward-looking statements regarding GeoVax’s ability to
implement its business plan, expected revenues and future success.
The words “believe,” “may,” “estimate,” “continue,” “anticipate,”
“intend,” “should,” “plan,” “could,” “target,” “potential,” “is
likely,” “will,” “expect” and similar expressions, as they relate
to us, are intended to identify forward-looking statements. We have
based these forward-looking statements largely on our current
expectations and projections about future events and financial
trends that we believe may affect our financial condition, results
of operations, business strategy and financial needs. Actual
results may differ materially from those included in these
statements due to a variety of factors, including whether: GeoVax
can develop and manufacture its vaccines with the desired
characteristics in a timely manner, GeoVax’s vaccines will be safe
for human use, GeoVax’s vaccines will effectively prevent targeted
infections in humans, GeoVax’s vaccines will receive regulatory
approvals necessary to be licensed and marketed, GeoVax raises
required capital to complete vaccine development, there is
development of competitive products that may be more effective or
easier to use than GeoVax’s products, GeoVax will be able to enter
into favorable manufacturing and distribution agreements, the
impact of the COVID-19 pandemic continues, and other factors, over
which GeoVax has no control.
Further information on our risk factors is contained in our
Quarterly Report on Form 10-Q that we have filed with the
SEC. Any forward-looking statement made by us herein speaks
only as of the date on which it is made. Factors or events that
could cause our actual results to differ may emerge from time to
time, and it is not possible for us to predict all of them. We
undertake no obligation to publicly update any forward-looking
statement, whether as a result of new information, future
developments or otherwise, except as may be required by law.
GeoVax Labs, Inc. 678-384-7220 investor@geovax.com
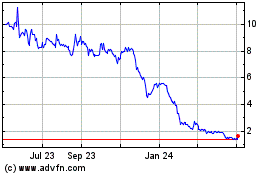
GeoVax Labs (NASDAQ:GOVX)
Historical Stock Chart
From Aug 2024 to Sep 2024
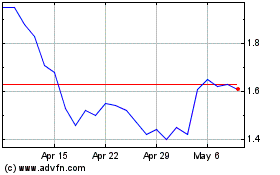
GeoVax Labs (NASDAQ:GOVX)
Historical Stock Chart
From Sep 2023 to Sep 2024