Erasca's Eras-801 for Treatment of Brain Tumors Gets FDA Orphan Drug Designation
June 22 2023 - 4:56PM
Dow Jones News
By Sabela Ojea
Erasca said Thursday that its Eras-801 medication aimed at
treating brain tumors was granted Orphan Drug Designation by the
U.S. Food and Drug Administration.
Shares rise 3.4% to $2.75 in after-hours trading.
The clinical-stage oncology company said Eras-801 has been
designated to treat aggressive brain tumors such as
Glioblastoma.
While orphan-drug status doesn't guarantee FDA approval as a
treatment for a rare disease, it is a prerequisite for getting
incentives such as tax breaks and marketing exclusivity if the
agency approves the drug for that purpose.
Write to Sabela Ojea at sabela.ojea@wsj.com; @sabelaojeaguix
(END) Dow Jones Newswires
June 22, 2023 16:41 ET (20:41 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
Erasca (NASDAQ:ERAS)
Historical Stock Chart
From Apr 2024 to May 2024
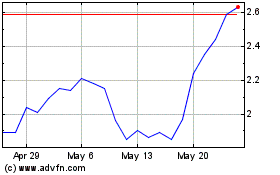
Erasca (NASDAQ:ERAS)
Historical Stock Chart
From May 2023 to May 2024