Eagle Pharmaceuticals, Inc. (Nasdaq: EGRX) (“Eagle” or the
“Company”) today announced that the following distinguished Key
Opinion Leaders will be featured at the Company’s Investor Day on
Tuesday, December 6, 2022, at the Lotte New York Palace, at 8:00am
ET:
Key Opinion Leaders
Richard P. Dutton, M.D., M.B.A., is Chief Quality Officer for
USAP. In this role, Dr. Dutton leads the committee responsible for
data analysis and performance measurement using the collective data
and evaluations of all USAP practices to improve patient safety and
clinical outcomes. Dr. Dutton is a widely recognized leader in
anesthesia quality management. Since 2009, he has served in
clinical leadership positions with the American Society of
Anesthesiologists (ASA), including Chief Quality Officer and
Medical Director of the Anesthesia Quality Institute (AQI). Prior
to USAP, he served as Chief Quality Officer for ASA and as the
founding Executive Director of AQI, which maintains the largest
anesthesia registry in the country. Aggregated data in the
registries is used to create educational materials, quality
management benchmarking, academic papers, comparative effectiveness
research and reports for ASA officers and committees to use to
improve the quality of patient care.
Dr. Prem Fort specializes in neonatology with the Johns Hopkins
All Children’s Maternal, Fetal & Neonatal Institute, where he
is Co-chair of its research council. He has ongoing single and
multicenter trials on management of respiratory outcomes, including
an NIH-funded trial for the use of caffeine at home for apnea in
preterm infants. He is also an advocate for research education and
serves as a mentor to residents and fellows. Dr. Fort’s research
focus includes includes respiratory management of premature
infants, control of breathing, and apnea of premature, specifically
as it relates to its management with caffeine.
Dr. TJ Gan is Professor and Head, Division of Anesthesiology,
Critical Care and Pain Medicine, UT Texas MD Anderson Cancer
Center, Houston, Texas. Previously, he was Distinguished Endowed
Chair of the Department of Anesthesiology at Stony Brook University
Renaissance School of Medicine in Stony Brook, New York. Prior to
that, Dr. Gan was Professor of Anesthesiology and Vice Chairman for
Clinical Research and Faculty Development at Duke University. Dr.
Gan is a diplomate of the American Board of Anesthesiology and is a
Fellow of the American Society of Anesthesiologists (FASA). He
serves as the Perioperative Medicine Executive Section Editor of
Anesthesia and Analgesia and is on the Editorial Board of
Perioperative Medicine. He has published over 300 manuscripts in
peer-reviewed journals and numerous books and books chapters.
Dr. Andre Kalil is a professor at the University of Nebraska
Medical Center Division of Infectious Diseases. He was named the
2021 UNMC Scientist Laureate, which is the highest honor UNMC
bestows upon its researchers. For 19 years, Dr. Kalil has been
a practicing physician and conducting clinical research at UNMC.
Dr. Kalil, has studied many challenging infections, including
transplant-related infections, hospital-acquired pneumonia, sepsis,
Ebola, and COVID-19.
Dr. Joseph V. Pergolizzi, Jr., MD is Chief Research and
Development Officer, Board Member and Co-founder of Enalare
Therapeutics. He is an internationally recognized thought leader in
internal, perioperative and pain medicine, as well as critical and
palliative care, pharmacology, drug development and regulatory
affairs. An internationally recognized key opinion leader in pain
medicine, Dr. Pergolizzi is a top development and regulatory
consultant for blockbuster-branded analgesics, such as Zofran,
Nucynta, Cymbalta and Butrans. Dr. Pergolizzi is the co-founder of
NEMA Research, Inc., a specialty CRO. He is a former subcommittee
member of the FDA SUI and SGE VAH Grant Review for Analgesics, and
chairman of PAINWEEK and Consultant to National Pain Foundation
Board of Directors. Dr. Pergolizzi is the former director of
business development and financial affairs for the Johns Hopkins
University School of Medicine Clinical Trials Unit. He also
formerly served as a part-time adjunct assistant
professor in the department of medicine at the Johns Hopkins
University School of Medicine.
Dr. Eugene Vortsman is a practicing Emergency Medicine Physician
working for the largest provider of healthcare in NY State,
Northwell Health, and rotates between large tertiary care academic
hospitals as well as community hospitals. Dr. Vortsman serves as
the Clinical Director for Addiction medicine and Disease Management
while also functioning as Chair for the Long Island Jewish Pain
Committee. His focuses include provider education for alternative
approaches to pain management, creating effective workflows for the
patient’s roads to recovery, as well as broad subject coverage
related to the ongoing opiate epidemic. His research experience
spans multiple institutions, including Northwell Health,
Cornell-Presbyterian Hospital, and University of Medicine and
Dentistry of New Jersey.
Program Schedule:
Date: Tuesday, December 6, 2022Location: Lotte New
York Palace HotelBreakfast/Check-in: 7:30am
ETPresentations/Q&A: 8:00am – 11:45am ETLunch to
follow
Webcast Registration Link: Click Here
Audience members will have the opportunity to ask questions
following the presentations.
Advance registration is required to attend this event in person.
Institutional investors and analysts are kindly requested to RSVP
through this link to attend.
A webcast of this event will be accessible via the Company’s
website at www.eagleus.com, under the Investors section.
About Eagle Pharmaceuticals, Inc.
Eagle is a fully integrated pharmaceutical company with research
and development, clinical, manufacturing and commercial expertise.
Eagle is committed to developing innovative medicines that result
in meaningful improvements in patients’ lives. Eagle’s
commercialized products include vasopressin, PEMFEXY®, RYANODEX®,
BENDEKA®, BELRAPZO®, TREAKISYM® (Japan), and BYFAVO® and BARHEMSYS®
through its wholly owned subsidiary Acacia Pharma Inc. Eagle’s
oncology and CNS/metabolic critical care pipeline includes product
candidates with the potential to address underserved therapeutic
areas across multiple disease states. Additional information is
available on Eagle’s website at www.eagleus.com.
Investor Relations for Eagle Pharmaceuticals,
Inc.: Lisa M. WilsonIn-Site Communications, Inc. T:
212-452-2793 E: lwilson@insitecony.com
Photos accompanying this announcement are available
at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/c47a0179-dd45-4d5b-8a60-c559bf98d784
https://www.globenewswire.com/NewsRoom/AttachmentNg/af95b4b3-5ae8-4894-9372-88217e21774d
https://www.globenewswire.com/NewsRoom/AttachmentNg/a2bca855-957e-4692-b580-60fcec9e0875
https://www.globenewswire.com/NewsRoom/AttachmentNg/473fae9a-8864-4368-83d5-5af2e45f518b
https://www.globenewswire.com/NewsRoom/AttachmentNg/433b9626-ed87-4bef-aff0-44f1fc72a309
https://www.globenewswire.com/NewsRoom/AttachmentNg/ea717543-88cf-40a6-b5fe-da7c47ae846f
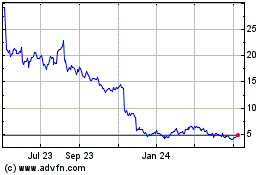
Eagle Pharmaceuticals (NASDAQ:EGRX)
Historical Stock Chart
From Aug 2024 to Sep 2024
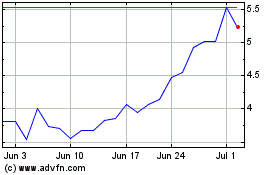
Eagle Pharmaceuticals (NASDAQ:EGRX)
Historical Stock Chart
From Sep 2023 to Sep 2024