GeoVax Appoints Jayne Morgan, M.D., to its Board of Directors
December 07 2022 - 4:00PM

via NewMediaWire -- GeoVax Labs, Inc. (Nasdaq:
GOVX), a biotechnology company developing immunotherapies and
vaccines against cancers and infectious diseases, announced today
that it has appointed Jayne Morgan, M.D., to its Board of
Directors.
Dr. Morgan is an accomplished and forward-thinking
healthcare management executive and research leader with extensive
expertise in complex hospital and corporation navigation, as well
as leadership in COVID-19 management, community engagement, crisis
mitigation, health equity and clinical trials.
“Dr. Morgan has spearheaded effective outreach
initiatives that have greatly influenced and improved public health
education and sentiment in target communities, particularly
regarding COVID-19,” said David Dodd, Chairman and CEO of GeoVax.
“We are thrilled to welcome her to our Board as we advance our
COVID-19 vaccine programs to provide protection to
immunocompromised patients and individuals underserved by existing
vaccines as well as a booster vaccine for healthy patients who have
already received an mRNA vaccine.”
Dr. Morgan currently serves as Executive Clinical
Director of the Covid Task Force at Piedmont Healthcare, Inc.,
where she develops and implements community outreach strategies in
conjunction with the Division of Diversity and Inclusion to improve
outcomes of disadvantaged populations positive for COVID-19. She
has held several leadership roles at Piedmont including Director of
Innovation, where she set the vision, trajectory and strategic
scaling and partner opportunities, and Director of Cardiovascular
Research and Research Development, where she expanded the
cardiovascular research program with the introduction of
biotechnology, feasibility trials and artificial intelligence
trials.
“Reaching underserved patient communities with
patient care is the driving force behind my work, which has most
recently focused on patients in need of access and options to help
mitigate prevention of and treatment for COVID-19,” said Dr.
Morgan. “I look forward to supporting GeoVax as it continues to
develop vaccines for many of the world’s most threatening
infectious diseases and seeks to address the significant unmet need
for protection against COVID-19 in immunocompromised patients.”
Dr. Morgan currently serves as the President Elect
of the Southeastern Life Sciences Association. She is also a
Founding Board Member of MedtechWomen, Co-Chair of the Bioscience
Leadership Council at the Metro Atlanta Chamber of Commerce, Board
Member at Botanical Sciences and active member of Biomed Investors
Network. Dr. Morgan holds several board positions with the American
Heart Association (AHA) including Board Member of its Atlanta
Chapter, Board Member of the National Diversity and Inclusion team,
National Board Member of the technology team and Regional Board
Member of the AHA Health Equity Committee.
Previously, Dr. Morgan served as Chief Medical
Officer of the American Chemistry Council, where she developed a
new translational research program, and Chief Executive Officer
Forty Million Beats, Inc., a pharmaceutical, biotech and chemical
industry consulting company, supporting clients including Novartis
Pharmaceuticals and Abbott Laboratories.
She completed her B.S. degree at Spelman College,
Medical Degree at Michigan State University, Internal Medicine
Residency at George Washington University and her Cardiology and
Pacemaker Fellowships at Mount Sinai Medical Center.
About GeoVaxGeoVax Labs, Inc. is a
clinical-stage biotechnology company developing novel therapies and
vaccines for cancers and many of the world’s most threatening
infectious diseases. The company’s lead program in oncology is a
novel oncolytic solid tumor gene-directed therapy, Gedeptin®,
presently in a multicenter Phase 1/2 clinical trial for advanced
head and neck cancers. GeoVax’s lead infectious disease candidate
is GEO-CM04S1, a next-generation COVID-19 vaccine targeting
high-risk immunocompromised patient populations. Currently in two
Phase 2 clinical trials, GEO-CM04S1 is being evaluated as a
single-dose, COVID-19 vaccine for immunocompromised patients such
as those suffering from hematologic cancers and other patient
populations for whom the current authorized COVID-19 vaccines are
insufficient. In addition, GEO-CM04S1 is in a Phase 2 clinical
trial evaluating the vaccine as a more robust, durable COVID-19
booster among healthy patients who previously received the mRNA
vaccines. GeoVax has a leadership team who have driven significant
value creation across multiple life science companies over the past
several decades. For more information, visit our
website: www.geovax.com.
Media Relations Contact:Gina Cestari6
Degrees917-797-7904gcestari@6degreespr.com
Investor Relations Contact:Rich CockrellCG
Capital404-736-3838govx@cg.capital
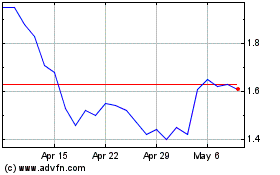
GeoVax Labs (NASDAQ:GOVX)
Historical Stock Chart
From Mar 2024 to Apr 2024
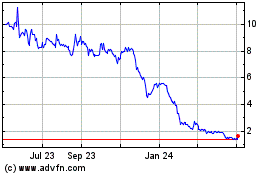
GeoVax Labs (NASDAQ:GOVX)
Historical Stock Chart
From Apr 2023 to Apr 2024