As
filed with the Securities and Exchange Commission on September 23, 2022
Registration
No.
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
S-3
REGISTRATION
STATEMENT
UNDER
THE
SECURITIES ACT OF 1933
THERAPEUTIC
SOLUTIONS INTERNATIONAL, INC.
(Exact
Name of Registrant as Specified in Its Charter)
| Nevada |
|
45-1226465 |
(State
or Other Jurisdiction of
Incorporation
or Organization) |
|
(I.R.S.
Employer
Identification
Number) |
701
Wild Rose Lane
Elk
City, Idaho 83525
Telephone:
(760) 295-7208
(Address,
Including Zip Code, and Telephone Number, Including Area Code, of Registrant’s Principal Executive Offices)
Timothy
G. Dixon
Chief
Executive Officer
701 Wild Rose Lane
Elk
City, Idaho 83525
Telephone:
(760) 295-7208
(Name,
Address, Including Zip Code, and Telephone Number, Including Area Code, of Agent For Service)
Copies
to:
H.D.
Kelso & Associates
Hugh
D. Kelso III, Esq, Managing Attorney
8799
Balboa Avenue, Suite 155 San Diego, CA 92123
Ph:
619-840-5056
Fax:
760-544-9888
Approximate
date of commencement of proposed sale to the public: From time to time after the effective date of this registration statement.
If
the only securities being registered on this Form are being offered pursuant to dividend or interest reinvestment plans, check the following
box. ☐
If
any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the
Securities Act of 1933, other than securities offered only in connection with dividend or interest reinvestment plans, check the following
box. ☒
If
this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the
following box and list the Securities Act registration statement number of the earlier effective registration statement for the same
offering. ☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the
Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If
this Form is a registration statement pursuant to General Instruction I.D. or a post-effective amendment thereto that shall become effective
upon filing with the Commission pursuant to Rule 462(e) under the Securities Act, check the following box. ☐
If
this Form is a post-effective amendment to a registration statement filed pursuant to General Instruction I.D. filed to register additional
securities or additional classes of securities pursuant to Rule 413(b) under the Securities Act, check the following box. ☐
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting
company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,”
“smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large
accelerated filer |
☐ |
|
Accelerated
filer |
☐ |
| Non-accelerated
filer |
☒ |
|
Smaller
reporting company |
☒ |
| |
|
|
Emerging
growth company |
☐ |
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
The
Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the
Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective
in accordance with Section 8(a) of the Securities Act of 1933 or until this Registration Statement shall become effective on such date
as the Commission, acting pursuant to said Section 8(a), may determine.
The
information in this prospectus is not complete and may be changed. We may not sell these securities or accept an offer to buy these securities
until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell
these securities and it is not soliciting offers to buy these securities in any state where such offer or sale is not permitted.
PRELIMINARY
PROSPECTUS (Subject to Completion) Dated: September 23, 2022
PROSPECTUS
Therapeutic
Solutions International, Inc.
$10,000,000
COMMON
STOCK
PREFERRED
STOCK
WARRANTS
UNITS
●
common stock;
●
preferred stock;
●
warrants to purchase our securities; or
●
units comprised of, or other combinations of, the foregoing securities.
We
(Therapeutic Solutions International, Inc.) may offer and sell these securities separately or together, in one or more series or classes
and in amounts, at prices and on terms described in one or more offerings. We may offer securities through underwriting syndicates managed
or co-managed by one or more underwriters or dealers, through agents or directly to purchasers. The prospectus supplement for each offering
of securities will describe in detail the plan of distribution for that offering. For general information about the distribution of securities
offered, please see “Plan of Distribution” in this prospectus.
Each
time our securities are offered, we will provide a prospectus supplement containing more specific information about the particular offering
and attach it to this prospectus. The prospectus supplements may also add, update or change information contained in this prospectus.
This prospectus may not be used to offer or sell securities without a prospectus supplement which includes a description of the method
and terms of this offering.
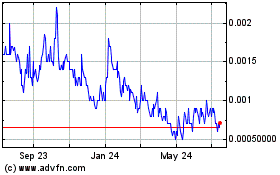
Our
common stock is currently quoted on the OTC Pink Sheets operated by OTC Markets Group, Inc., at the OTC Pink tier under the symbol “TSOI.”
On September 19, 2022, the closing price of our common stock as reported was $0.0101 per share.
If
we decide to seek a listing of any preferred stock, warrants or units offered by this prospectus, the related prospectus supplement will
disclose the exchange or market on which the securities will be listed, if any, or where we have made an application for listing, if
any.
Our
independent registered public accounting firm has included a “going concern” paragraph in the notes to our consolidated financial
statements.
Investing
in our securities involves certain risks. See “Risk Factors” beginning on page 23 and the risk factors in our most recent
Annual Report on Form 10-K, which is incorporated by reference herein, as well as in any other recently filed quarterly or current reports
and, if any, in the relevant prospectus supplement. We urge you to carefully read this prospectus and the accompanying prospectus supplement,
together with the documents we incorporate by reference, describing the terms of these securities before investing.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed
upon the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense. The date of this prospectus
is September 23, 2022.
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS
This
prospectus is part of a registration statement that we filed with the U.S. Securities and Exchange Commission, or the SEC, using a “shelf”
registration process. Under this shelf registration process, we may offer and sell, either individually or in combination, in one or
more offerings, any of the securities described in this prospectus, for total gross proceeds of up to $10,000,000. This prospectus provides
you with a general description of the securities we may offer. Each time that we offer and sell securities under this prospectus, we
will provide a prospectus supplement to this prospectus that contains specific information about the securities being offered and sold
and the specific terms of that offering. The prospectus supplement may also add, update or change information contained in this prospectus
with respect to that offering. If there is any inconsistency between the information in this prospectus and the applicable prospectus
supplement, you should rely on the prospectus supplement. Before purchasing any securities, you should carefully read both this prospectus
and the applicable prospectus supplement, together with the additional information described under the headings “Where You Can
Find More Information” and “Incorporation of Certain Documents by Reference.”
We
have not authorized any other person to provide you with different information. If anyone provides you with different or inconsistent
information, you should not rely on it. We will not make an offer to sell these securities in any jurisdiction where the offer or sale
is not permitted. You should assume that the information appearing in this prospectus and the applicable prospectus supplement to this
prospectus is accurate as of the date on its respective cover, and that any information incorporated by reference is accurate only as
of the date of the document incorporated by reference, unless we indicate otherwise. Our business, financial condition, results of operations
and prospects may have changed since those dates.
SUMMARY
This
summary highlights the information contained elsewhere in or incorporated by reference into this prospectus. Because this is only a summary,
it does not contain all of the information that you should consider before investing in our securities. You should carefully read this
entire prospectus, including the information contained under the heading “Risk Factors,” and all other information included
or incorporated by reference into this prospectus in their entirety before you invest in our securities.
Unless
the context otherwise requires, all references in this prospectus to “TSOI,” “we,” “us,” “our,”
“the Company” or similar words refer to Therapeutic Solutions International, Inc., a Nevada corporation, together with its
consolidated subsidiaries, taken as a whole.
Shelf
Registration
This
prospectus is part of a registration statement that we filed with the Securities and Exchange Commission (the “SEC”) using
a “shelf” registration process. Under this shelf registration process, we may from time to time sell any combination of the
securities described in this prospectus in one or more offerings of up to ten million dollars ($10,000,000). The securities may be shares
of common stock, preferred stock, warrants to purchase common stock or preferred stock, and units or other combinations of, the foregoing
securities. We may offer these securities in amounts, at prices and on terms determined at the time of offering. The securities may be
sold directly to you, through agents, or through underwriters and dealers.
This
prospectus provides you with a general description of the securities we may offer. Each time we sell securities, we will provide one
or more prospectus supplements that will contain specific information about the terms of the offering. The prospectus supplement may
also add, update or change information contained in this prospectus. You should carefully read this prospectus, the accompanying prospectus
supplement, the information incorporated by reference into this prospectus, including our financial statements, and the exhibits to the
registration statement of which this prospectus is a part. You should also read the information discussed under “Risk Factors,”
which describes the risks of investing in our securities.
Neither
we, nor any agent, underwriter or dealer have authorized anyone to provide you with any information other than that contained or incorporated
by reference in this prospectus or any accompanying prospectus supplement or free writing prospectus to which we have referred you. We
and any agent, underwriter or dealer take no responsibility for, and can provide no assurance as to the reliability of, any other information
others may give you. This prospectus and any accompanying prospectus supplement do not constitute an offer to sell or the solicitation
of an offer to buy any securities other than the securities described in this prospectus or any accompanying prospectus supplement or
an offer to sell or the solicitation of an offer to buy such securities in any circumstances in which such offer or solicitation is unlawful.
You should assume that the information appearing in this prospectus, any prospectus supplement, the documents incorporated by reference
and any related free writing prospectus is accurate only as of their respective dates. Our business, financial condition, results of
operations and prospects may have changed materially since those dates.
Company
Overview
Therapeutic
Solutions International, Inc. Therapeutic Solutions International, Inc. (“TSOI” or the “Company”), was organized
August 6, 2007, under the name Friendly Auto Dealers, Inc., under the laws of the State of Nevada. In the first quarter of 2011 the Company
changed its name from Friendly Auto Dealers, Inc. to Therapeutic Solutions International, Inc., and acquired Splint Decisions, Inc.,
a California corporation.
Currently,
the Company is focused on immune modulation for the treatment of several specific diseases. Immune modulation refers to the ability to
upregulate (make more active) or downregulate (make less active) one’s immune system.
Activating
one’s immune system is now an accepted method to treat certain cancers, reduce recovery time from viral or bacterial infections
and to prevent illness. Additionally, inhibiting one’s immune system is vital for reducing inflammation, autoimmune disorders and
allergic reactions.
TSOI
is developing a range of immune-modulatory agents to target certain cancers, schizophrenia, suicidal ideation, traumatic brain injury,
lung pathologies, and for daily health.
Nutraceutical
Division – TSOI has been producing high quality nutraceuticals. Its current flagship product, QuadraMune®, is a multi-patented,
synergistic blend of pterostilbene, sulforaphane, epigallocatechingallate, and thymoquinone. QuadraMune has been shown to increase Natural
Killer Cell activity and healthy Cytokine production. Our synergistic blend of ingredients help the immune system fight off common and
complex ailments and promote healthy T Cell activity. Recently the Company was approved to sell certain nutraceuticals on the Amazon
Platform.
Cellular
Division – TSOI obtained exclusive rights to a patented adult stem cell for development of therapeutics in the area of chronic
traumatic encephalopathy (CTE) and traumatic brain injury (TBI) and Lung Pathology (LP).
The
stem cell licensed, termed “JadiCell” is unique in that it possesses features of mesenchymal stem cells, however, outperforms
these cells in terms of a) enhanced growth factor production; b) augmented ability to secrete exosomes; and c) superior angiogenic and
neurogenic ability.
Chronic
Traumatic Encephalopathy (CTE) is caused by repetitive concussive/sub-concussive hits to the head sustained over a period of years and
is often found in football players. The condition is characterized by memory loss, impulsive/erratic behavior, impaired judgment, aggression,
depression, and dementia. In many patients with CTE, it is anatomically characterized by brain atrophy, reduced mass of frontal and temporal
cortices, and medial temporal lobe. TSOI has previously filed several patents in the area of CTE based on modulating the brain microenvironment
to enhance receptivity of regenerative cells such as stem cells. On March 4, 2021, the Company received an IND Serial # 27377 for a clinical
trial of 10 patients with CTE.
On
August 4th, 2021, the Company announced clearance from the Food and Drug Administration (FDA) to initiate a Phase III pivotal trial for
registration of the Company’s JadiCell™ universal donor stem cell as a treatment for COVID-19 associated lung failure under
IND # 19757. In previous studies the Company has demonstrated the superior activity of JadiCell™ to other types of stem cells including
bone marrow, adipose, cord blood, and placenta. Furthermore, the JadiCell™ was shown to be 100% effective in saving the lives of
COVID-19 patients under the age of 85 in a double-blind placebo controlled clinical trial with patients in the ICU on a ventilator. In
patients over the age of 85 the survival rate was 91%.
In
addition, the Company has filed data with the FDA, as part of IND #17448, which demonstrated that treatment of cancer patients with StemVacs™
resulted in enhanced activity of a type of immunological cell called “natural killer” cells, otherwise known as “NK
cells.”
The
Company has also developed an allogenic version of StemVacs and has filed patents to cover activating universal donor immune system cells
called dendritic cells in a manner so that upon injection they reprogram the body’s NK cells.
On
May 9, 2022, the Company filed an Investigational New Drug Application for Treatment of Chronic Obstructive Pulmonary Disease (COPD)
Using JadiCell™ Universal Donor Adult Stem Cells under IND Serial # 28508.
Investigational
Drug Applications:
Treatment
of Metastatic Breast Cancer by StemVacs-V Cancer Immunotherapeutic IND #
The
Primary Objective is safety and feasibility of StemVacs-V administration at 12 months as assessed by lack of adverse medical events.
The Secondary Objective is efficacy as judged by tumor response, time to progression, and immunological monitoring.
Safety,
Feasibility, and Immunomodulatory Activities of StemVacs in Patients with Advanced Solid Tumors IND # 17448
The
Primary Objective is safety and feasibility of StemVacs administration at 12 months as assessed by lack of adverse medical events. The
Secondary Objective is efficacy as judged by tumor response, time to progression, and immunological monitoring.
Umbilical
Cord-derived Mesenchymal Stem Cells for Patients with COVID-19 (“UC-MSC for COVID-19”) IND # 19757
The
primary objective will be to assess effectiveness of UC-MSC treatment on proportion of patients alive and free of respiratory failure
at Day 60 after randomization. The secondary objectives will be to assess all-cause mortality at Day 60, survival at day 31, number of
subjects experiencing serious adverse events (SAEs) by day 31, SAE-free survival, time to recovery (evaluated until day 60), and time
to oxygen requirement equal or below 40% oxygen.
Investigation
of Umbilical Cord-derived Mesenchymal Stem Cells for the Treatment of Chronic Traumatic Encephalopathy Patients IND # 27377
To
determine safety and efficacy of 100 million intravenously administered JadiCell™ allogeneic umbilical cord mesenchymal stem cells.
Efficacy will be determined by behavioral scores, brain imaging, and reduction in inflammatory markers. Toxicity of treatment was evaluated
for the duration of the study and will be graded according to the criteria of the World Health Organization.
JadiCell
Therapy for COPD IND # 28508
To
determine safety and efficacy of intravenously administered allogeneic JadiCell umbilical cord blood mesenchymal stem cells in patients
with moderate-to-severe COPD. The Primary Endpoint, which is toxicity, will be assessed by number of adverse events (AEs). The Secondary
Endpoint, which is efficacy will be evaluated at baseline and days 30, 60, and 90.
Nutraceutical
Division (TSOI)
●
ProJuvenol® is a patented, (US No.: 9,682,047) and powerful synergistic blend of complex anti-aging ingredients in capsules.
●
NanoStilbene® is an easily absorbed nanoemulsion of nanoparticle pterostilbene derived from the ‘047 patent.
●
DermalStilbene is a topical form of pterostilbene delivered via spray application onto skin, derived from the ‘047 patent.
●
IsoStilbene an injectable formulation of pterostilbene is available by prescription only, derived from the ‘047 patent.
●
NeuroStilbene is an intranasal form of pterostilbene delivered via spray application inside the nostril, derived from the ‘047
patent.
●
NanoPlus is a blend of NanoStilbene and Nano Cannabidiol which are an easily absorbed Nanoparticles formulation of Pterostilbene and
Cannabidiol.
●
Nano Cannabidiol is an easily absorbed Nanoparticle formulation of Cannabidiol Isolate in the range of 75-90 nanometers. This product
is built on the same nano platform as NanoStilbene and is delivered at a concentration of 200mg per milliliter.
●
NanoPSA is a blend of NanoStilbene® and Broccoli Sprout Extract (BSE) providing 74mg of BSE and 125mg of our patented NanoStilbene,
a proprietary formulation of nanoparticle pterostilbene.
●
NLRP3 Trifecta is a two-product combo and consists of one bottle of NanoPSA and one bottle of GTE-50 green tea extract.
●
QuadraMune® is a multi-patented synergistic blend of pterostilbene, sulforaphane, epigallocatechingallate, and thymoquinone.
●
NuBrainer™ HSCP is a nootropic blend of Huperzine-A, Sterubin, Cannabidiol, and Pterostilbene.
Patents:
On
September 19, 2022, the Company filed a patent application titled “Treatment of Bipolar Disorder Using Mesenchymal Stem
Cells and Modification of Mesenchymal Stem Cells” that discloses the utilization of mesenchymal stem cells, exosomes from mesenchymal
stem cells, conditioned media from mesenchymal stem cells, apoptotic bodies from mesenchymal stem cells, and modified mesenchymal stem
cells for treatment of bipolar disorder. In one embodiment mesenchymal stem cells isolated from umbilical cord tissue are treated with
carbon monoxide at a concentration sufficient to induce activation of heme-oxygenase I and infused into a patient at risk or suffering
from bipolar disorder.
On
September 12, 2022, the Company filed a patent application titled “Treatment of COPD by Stimulation of Stem Cell Mobilization”
which discloses means of inducing pulmonary regeneration and/or protection from oxidative stress by stimulation of endogenous stem cell
mobilization together with one or more inhibitors of NF-kappa B and/or one or more inhibitors of oxidative stress. The invention discloses
the unexpected finding that G-CSF administration enhances oxidative stress and pulmonary damage, however, coadministration with pterostilbene,
results in synergistic suppression of COPD pathology.
On
August 29, 2022, the Company filed a patent application titled “Gene Silencing Therapy of Acute Respiratory Disorder”
that teaches treatment means, compositions of matter and protocols useful for suppression of acute respiratory disorder (ARDS) through
induction of RNA interference in the pulmonary microenvironment alone and/or in conjunction with mucolytic and/or DNA disrupting agents.
In one embodiment short interfering RNA (siRNA) is prepared which targets complement receptors C3R and/or C5R together with TNF-receptor,
IL-6 receptor and/or TLR4 and TLR9. In some embodiments NanoStilbene is utilized as a delivery vehicle for siRNA delivery.
On
August 12, 2022, the Company filed a patent application titled “Treatment of Chronic Obstructive Pulmonary Disease by
Mesenchymal Stem Cell Apoptotic Bodies and Compositions Thereof” that discloses means, treatments and compositions of matter
useful for treatment of chronic obstructive pulmonary disease (COPD). In one embodiment the invention provides the administration of
mesenchymal stem cell apoptotic bodies alone or in combination with “regenerative adjuvants” to prevent and/or reverse reduction
in lung function associated with COPD. In other embodiments the invention teaches the utilization of stem cell apoptotic bodies for induction
of pulmonary regeneration directly or indirectly.
On
July 29, 2022, the Company filed a patent application titled “Gene Modified iPSC Derived Cellular Compositions for Regeneration
and Immune Modulation” that disclosed cells and cellular compositions useful for treatment of degenerative and/or autoimmune
diseases derived from gene edited/gene modified pluripotent stem cells. In one embodiment pluripotent stem cell such as inducible pluripotent
stem cells are gene modified to express tissue associated transcription factors such as pdx-1 if endodermal tissue is desired and cells
are differentiated into regenerative-type cells such as along the mesenchymal lineage. In one embodiment the invention teaches transfection
with IL-27 to induce expression of coinhibitory molecules for suppression of autoimmunity. In some embodiments the invention provides
generation of iPSC derived MSC which cannot stimulate inflammation due to gene-editing based removal of inflammatory associated transcription
factors.
On
May 12, 2022, the Company filed a patent application titled “Inhibition and Reversion of Chronic Obstructive Pulmonary
Disease (COPD) by Endothelial Cell Regeneration” that teaches means, treatment methods, and compositions of matter useful for
prevention and/or reversion of chronic obstructive pulmonary disease (COPD). In one embodiment the invention provides the administration
of mesenchymal stem cells and exosome thereof as a means of augmenting endogenous endothelial regeneration and/or endothelial regeneration
stimulated by exogenous means. In some embodiments the invention provides administration of allogeneic mesenchymal stem cells together
with autologous endothelial progenitor cells and/or mobilization of said autologous endothelial progenitor cells.
On
March 7, 2022, the Company filed a patent application titled “Treatment of Trauma Associated Cognitive Dysfunction Using
Mesenchymal Stem Cell Apoptotic Bodies and Compositions Thereof” which teaches means, treatments and compositions of matter
useful for treatment of chemotherapy/radiotherapy associated cognitive dysfunction. In one embodiment the invention provides the administration
of mesenchymal stem cell apoptotic bodies alone or in combination with “regenerative adjuvants” to prevent and/or reverse
cognitive dysfunction associated with chemotherapy and/or radiation therapy. In other embodiments the invention teaches the utilization
of stem cell apoptotic bodies for induction of neuroregeneration directly or indirectly.
On
February 7, 2022, the Company filed a patent application titled “Treatment of COVID-19 Associated Cognitive Dysfunction
by Nutraceutical Preparations” that teaches means and methods of treating cognitive dysfunction associated with COVID-19 and/or
other associated with inflammatory conditions. In one embodiment treatment of COVID-19 cognitive dysfunction performed by administration
of nutraceutical means, wherein said nutraceuticals are administered at a frequency and/or concentration sufficient to induce proliferation
of endogenous neural progenitor cells and/or protect cells from inflammatory damage. In one embodiment said nutraceuticals are comprised
of green tea extract, and/or nigella sativa, and/or pterostilbene, and/or sulforaphane. In some embodiments nutraceutical compositions
are utilized to overcome treatment resistant of currently used antidepressants.
On
November 1, 2021, the Company filed a patent application titled “Induction of Concurrent Pulmonary Immune Modulation
and Regeneration by Protein Mediated Conjugation of Immune Regulatory Cells with Endogenous Progenitor Cells” that discloses
means, methods and compositions of matter useful for treatment of inflammatory pulmonary diseases such as COVID-19 through administration
of agents that facilitate interaction between immune modulatory cells and endogenous pulmonary progenitor cells. In one embodiment a
bispecific antibody capable of facilitating the interaction between CD25 on T regulatory cells and CD47 on pulmonary epithelial stem
cells is described.
On
October 11, 2021, the Company filed a patent application titled “Umbilical Cord Derived Regenerative and Immune Modulatory
Stem Cell Populations” which provides universal donor cellular populations derived from umbilical cords possessing ability
to elicit immune modulation and evoke regeneration when administered into a mammalian host. Generation of cellular products for clinical
use are provided including methodologies of expansion, characterization, and means of therapeutic implementation.
On
October 4, 2021, the Company filed a patent application titled “Reduction of Neutrophil Extracellular Trap formation
by Mesenchymal Stem Cells and their Exosomes” that disclosed methods of reducing lung inflammation in acute respiratory distress
syndrome elicited by various factors such as COVID-19 infection by reduction of neutrophil extracellular trap formation through administration
of mesenchymal stem cells and/or exosomes thereof. The invention provides means of inhibiting neutrophil release of extracellular traps
by mesenchymal stem cells and/or exosomes derived from said mesenchymal stem cells. Additionally, synergies are provided between mesenchymal
stem cells and/or exosomes derived from mesenchymal stem cells and agents approaches which reduce neutrophil extracellular trap formation.
On
September 22, 2021, the Company filed a patent application titled “Stimulation of Mesenchymal Stem Cell Therapeutic Activities
by T Regulatory Cells” teaches novel means of enhancing mesenchymal stem cell regenerative activities including, intra alia,
production from pulmonary leakage and suppression of scar tissue formation by co-administration with T regulatory cells. In some embodiments
the invention provides an interaction between T regulatory cells and mesenchymal stem cells in which T regulatory cells stimulate upregulation
of mesenchymal stem cell activity in a GITR dependent manner.
On
September 16, 2021, the Company filed a patent application titled “Ivermectin Compositions for Treatment of COVID-19”
that discloses novel mechanisms of action of ivermectin therapy as related to treatment of COVID-19 and means of augmenting therapeutic
activities by co-administration with one or more of the following: pterostilbene, thymoquinone, epigallocatechin-3-gallate, and sulforaphane.
In one embodiment the invention provides enhanced reduction of inflammation induced pulmonary leakage without augmenting immune suppressive
mechanisms.
On
August 23, 2021, the Company filed a patent application titled “Umbilical Cord Mesenchymal Stem Cells for Treatment of
Chronic Obstructive Pulmonary Disease and Lung Degeneration” that discloses means of treating lung degenerative diseases including
chronic obstructive pulmonary disease (CODP) using umbilical cord mesenchymal stem cells such as JadiCells alone, and/or using said cells
under conditions that are activated in order to endow enhanced regenerative activity. In one embodiment said activation of said mesenchymal
stem cells is performed through stimulation with a toll like receptor agonist at a concentration and duration sufficient to induce a
>50% increase in keratinocyte growth factor expression from said stem cells. In another embodiment the invention provides the use
of JadiCells as a means of producing exosomes, wherein said exosomes possess therapeutic properties capable of reducing inflammation,
fibrosis and degeneration associated with COPD, as well as stimulation of regenerative activity. In some JadiCells are activated by a
treatment with Activated Protein C.
On
August 18, 2021, the Company filed a patent application titled “Enhancement of Umbilical Cord Mesenchymal Stem Cell Therapeutic
Activity by Stimulators of T Regulatory Cells and/or Cells Expressing CD73” that teaches compositions of matter and protocols
useful for treatment of COVID-19 and/or other inflammatory pathologies through stimulation of T regulatory cells and/or T cells expressing
CD73 using administration of umbilical cord derived mesenchymal stem cells such as JadiCells. In one embodiment dosage of JadiCells needed
to treat a patient is determined by the increase of T regulatory cells and/or CD73 expressing cells that are increased in number and/or
activity subsequent to a test dose of JadiCells. In another embodiment stimulators of T regulatory cells and/or CD73 expressing T cells
are utilized together with JadiCells in order to augment therapeutic activity. In some embodiments administration of JadiCell is performed
with low dose interleukin-2 as a treatment for COVID-19 or other inflammatory related pathologies.
On
August 11, 2021, the Company filed a patent application titled “Induction of Neurogenesis using Umbilical Cord Derived
Mesenchymal Stem Cells and Derivatives Thereof” that disclosed compositions of matter and protocols useful for treatment of
neurological dysfunctions through stimulation of adult neurogenesis using administration of umbilical cord derived mesenchymal stem cells
such as JadiCells. In one embodiment viral induced neuropathy is reduced by administration of JadiCells to stimulate neurogenesis. In
another embodiment the neurogenic activity of selective serotonin reuptake inhibitors is enhanced by administration of JadiCells. In
some embodiments administration of JadiCell exosomes, conditioned media, microvesicles and/or apoptotic bodies is utilized to stimulate
neurogenesis.
On
July 6, 2021, the Company filed a patent application titled “Treatment of Parkinson’s Disease by Immune Modulation
and Regenerative Means” in which we describe and disclose means, methods and compositions of matter for treatment Parkinson’s
Disease through concurrent immune modulation and regenerative means. In one embodiment Parkinson’s Disease is treated by augmentation
of T regulatory cell numbers and/or activity while concurrently providing regenerative cells such as mesenchymal stem cells, and/or dopamine
secreting cells. In one embodiment administration of immunoglobulins such as IVIG together with low dose interleukin-2 and/or low dose
naltrexone is disclosed as a preparatory means prior to administration of therapeutic cells such as stem cells. Other therapeutic means
utilized in an adjuvant manner are also provided for hormonal rebalancing, transcranial magnetic stimulation, and deep brain stimulation.
On
May 24, 2021, the Company filed a patent application titled “Immunotherapies for Targeting of Tumor Vasculature”
that disclosed novel means, protocols, and compositions of matter for creating targeted immune responses and/or induction of immunological
memory towards the tumor vasculature. In one embodiment pluripotent stem cells are transfected with one or more genes capable of eliciting
immunity, induced to differentiate into endothelial-like cells which resemble the tumor endothelial cells, and utilized as a vaccine.
In some embodiment’s genes are engineered under control of specific promoters to allow for various specificities of activity. In
one specific embodiment pluripotent stem cells engineered to endow properties capable of inducing expression of the α- Gal epitope
(Galα1,3Galα1,4GlcNAc-R). Addition of adjuvants to enhance antigen presentation of the vaccine composition, as well as means
of stimulating systemic enhancement of circulating endothelial specific T cells are also disclosed.
On
May 21, 2021, the Company filed a patent application titled “Lithium as a Monotherapy and/or Stem Cell Adjuvant Therapy
for Pulmonary Fibrosis” that disclosed compositions of matter, therapeutics, and protocols useful for reduction and/or reversion
of pulmonary fibrosis. In one specific embodiment lithium chloride is administered together with a regenerative cell in a patient suffering
from, or at risk of pulmonary fibrosis. In one embodiment said lithium chloride is administered as an adjuvant to a regenerative therapy,
wherein said regenerative therapy is a gene therapy, a protein therapy, a cell therapy, or a tissue transplant. In one embodiment lithium
chloride, or a salt thereof is utilized alone, or with a regenerative means, to evoke preservation and/or elongation of telomere length
in pulmonary tissue. In one embodiment the invention teaches administration of umbilical cord mesenchymal stem cells (MSC) and/or products
derived from said cells in order to induce an inhibition of natural or pathological reduction of telomere length, to preserve telomere
length or to enhance telomere length. In one embodiment the MSC described in the invention as useful are umbilical cord derived MSC.
On
May 17, 2021, the Company filed a patent application titled “Treatment of Major Depressive Disorder by Low Dose Interleukin-2”
which teaches methods, compositions of matter, and protocols useful for treatment of major depressive disorder through administration
of low dose interleukin- 2 at a concentration and/or frequency sufficient to increase expansion of T regulatory cell numbers and/or enhancement
of T regulatory cell activity. In some embodiments administration of interleukin-2 is provided as means of enhancing efficacy of standard
antidepressant therapies. Furthermore, administration of interleukin-2 receptor agonists is also described in the current invention as
a treatment of major depressive disorder.
On
April 13, 2021, the Company filed a patent application titled “Amelioration and Treatment of Opioid Addiction”
that discloses compositions of matter, protocols and treatment means for reducing and/or preventing opioid addiction. In one embodiment
the invention teaches intranasal administration of umbilical cord blood plasma, or extracts thereof, together with pterostilbene or pterostilbene
containing nanoparticles, and/or oxytocin, and/or human chorionic gonadotropin.
On
March 29, 2021, the Company filed a patent application titled “Compositions Capable of Stimulating Immunity Towards Tumor
Blood Vessels” which discloses novel means, protocols, and compositions of matter for eliciting an immune response against
blood vessels supplying neoplastic tissue. In one embodiment pluripotent stem cells are transfected with one or more genes capable of
eliciting immunity. In some embodiments such genes are engineered under control of specific promoters to allow for various specificities
of activity. In one specific embodiment pluripotent stem cells engineered to endow properties capable of inducing expression of the α-Gal
epitope (Galα1,3Galα1,4GlcNAc-R).
On
March 23, 2021, the Company filed a patent application titled “Chimeric Cells Comprising Dendritic Cells and Endothelial
Cells Resembling Tumor Endothelium” which disclosed are means, methods and compositions of matter useful for induction of immunological
responses towards tumor endothelial cells. In one embodiment the invention teaches fusion of dendritic cells and cells resembling tumor
endothelial cells and administration of such chimeric cells as an immunotherapy for stimulation of tumor endothelial cell destruction.
In other embodiments pluripotent stem cells are utilized to generate dendritic cells, wherein said dendritic cells are fused with pluripotent
stem cell derived endothelial cells created in a manner to resemble tumor endothelial cells.
On
March 16, 2021, the Company filed a patent application titled “Pluripotent Stem Cell Derived Dendritic Cells and Engineered
Dendritic Cells for Cancer Immunotherapy” which disclosed are populations of dendritic cells generated from stem cells capable
of inducing immunity towards cancer. In one embodiment said dendritic cells are generated from allogeneic inducible pluripotent stem
cells, for some uses, said pluripotent stem cells are genetically engineered/edited to induce cancer specific immunity and/or resist
immunosuppressive effect of tumor derived microenvironment. In one embodiment pluripotent stem cells are transfected with cancer stem
cell antigens such as BORIS and/or NR2F6.
On
March 4, 2021, the Company filed a patent application titled “Therapeutic Monocytes for Prevention of Suicidal Ideation”
that discloses compositions of matter, protocols, and therapeutic means for treatment of suicidal ideations and/or suppression of suicidal
attempts. In one embodiment the invention provides the use of umbilical cord derived monocytes as a means of treatment. In another embodiment,
monocytes are de-differentiated from adult monocytes using reprogramming means to create monocyte capable of producing anti-inflammatory
as well as regenerative properties useful in reducing suicidal ideations and/or attempts.
Published
on September 8, 2022, https://patents.justia.com/patent/20220280574
On
February 2, 2021, the Company filed a patent application titled “Ex Vivo Generation of Immunocytes Recognizing Brother
Of The Regulator of Imprinted Sites (BORIS) Expressing Cancer Stem Cells” that discusses means, methods and compositions of
matter useful for induction of immunity towards cancer stem cells by providing a dendritic cell, wherein said dendritic cells express
BORIS and/or peptides derived from BORIS, wherein said dendritic cell is cultured in the presence of one or more immunocytes. In one
embodiment said dendritic cells are derived from umbilical cord blood sources and allogeneic to T cells, which are expanded ex vivo and
used for the purposes of immunotherapy.
Published
on August 25, 2022, https://patents.justia.com/patent/20220267730
On
February 8, 2021, the Company filed a patent application titled “Stimulation of Natural Kill Cell Memory by Administration
of Dendritic Cells” which disclosed means, methods and compositions of matter useful for induction of natural killer cell memory
by administration of dendritic cells and/or exosomes thereof. In one embodiment a mammal suffering from cancer is administered allogeneic
cord blood derived dendritic cells that are not pulsed exogenously. In one embodiment the dendritic cells are stimulated to possess chemotactic
activity towards the tumor by culture of dendritic cell progenitors in hypoxia. Natural killer cell memory is induced, in part, by triggering
of upregulation of cytokines associated with homeostatic expansion such as interleukin 7 and interleukin 15.
Published
on August 11, 2022, https://patents.justia.com/patent/20220249551
On
January 26, 2021, the Company filed a patent application titled “Stimulation of Dendritic Cell Activity by Homotaurine
and Analogues Thereof” which discloses means, methods, and compositions of matter useful for enhancement of dendritic cell
activity. In one embodiment the invention provides the use of GABA agonists such as homotaurine for stimulation of dendritic cell activity.
In one embodiment said dendritic cell activity is enhancement of natural killer cell activity and/or of T cell activity. In one embodiment
NK cell activity is ability to induce cytotoxicity in neoplastically transformed cells, whereas T cell activity is either cytokine production
for CD4 cells or cytotoxicity for CD8 cells.
Published
on July 28, 2022, https://patents.justia.com/patent/20220235325
On
December 21, 2020, the Company filed a patent application titled “Immunotherapy for Opioid Addiction” which
teaches means, methods and compositions of matter useful for reduction of brain inflammation and prevention of opioid addiction and/or
tolerance. In one embodiment the invention provides utilization of platelet rich plasma (PRP), alone, or admixed with regenerative/anti-inflammatory
adjuvants, for reduction of neural inflammation. In one embodiments PRP is admixed with oxytocin and administered intranasally in a patient
at risk of opioid addiction. In another embodiment, PRP is admixed with fortified and non-fortified nigella sativa oil, and/or pterostilbene
and administered intranasally. Other embodiments include utilization of autologous stromal vascular fraction cells alone and/or admixed
with regenerative/anti-inflammatory adjuvants.
Published
on June 23, 2022, https://patents.justia.com/patent/20220193127
On
December 8, 2020, the Company filed a patent application titled “Treatment of Major Depressive Disorder and Suicidal
Ideations Through Stimulation of Hippocampal Neurogenesis Utilizing Plant-Based Approaches” that teaches means and methods
of treating major depressive disorder and/or other disorders that predispose to suicide by administration of nutraceutical means, wherein
said nutraceuticals are administered at a frequency and/or concentration sufficient to induce proliferation of endogenous neural progenitor
cells. In one embodiment said nutraceuticals are comprised of green tea extract, and/or nigella sativa, and/or pterostilbene, and/or
sulforaphane. In some embodiment’s nutraceutical compositions are utilized to overcome treatment resistant of currently used antidepressants.
Published
on June 9, 2022, https://patents.justia.com/patent/20220175701
On
November 24, 2020, the Company filed a patent application titled “Stimulation of NK Cell Activity by QuadraMune Alone
and together with Metformin” that disclosed means, compounds, and compositions of matter useful for stimulation of natural
killer cell activity. In some embodiments the invention teaches the administration of a therapeutic combination of ingredients comprising
of metformin, pterostilbene, nigella sativa, sulforaphane, and epigallocatechin-3-gallate (EGCG) to a mammal in need of natural killer
cell immune modulation. In another embodiment, the invention teaches administration of said therapeutic combination to a mammal infected
with said SARS-CoV-2. In some embodiments dosage of said therapeutic combination is based on inflammatory and/or immunological parameters
observed in patients with COVID-19.
Published
on May 26, 2022, https://patents.justia.com/patent/20220160809
On
October 27, 2020, the Company filed a patent application titled “Protection/Regeneration of Neurological Function by
Endothelial Protection/Rejuvenation” using Stem Cells for Treatment of Conditions such as Chronic Traumatic Encephalopathy
and Schizophrenia” which therapeutic compounds, protocols, and compositions of matter useful for treatment of neurological conditions.
In one embodiment the invention teaches the treatment of chronic traumatic encephalopathy (CTE) through protecting/regenerating the endothelial
by administration of cells such as stem cells. In one embodiment stem cells are administered in order to protect the endothelium from
apoptosis and to preserve the blood brain barrier. In another embodiment stem cells are administered together with endothelial progenitor
cells in order to regenerate neural endothelium. In other embodiments preservation of brain integrity in conditions of degeneration is
accomplished by administration of stem cells and/or endothelial cells.
Published
on April 28, 2022, https://patents.justia.com/patent/20220125852
On
October 18, 2020, the Company filed a patent application titled “Nutraceutical Reduction Prevention and/or Reversion
of Multiple Sclerosis” that discloses compositions of matter, protocols, and treatment means for preventing and/or reversing
multiple sclerosis in a mammal. In one embodiment administration of compositions containing pterostilbene, and/or nigella sativa, and/or
sulforaphane, and/or epigallocatechin-3-gallate (EGCG) are provided.
Published
on June 23, 2022, https://patents.justia.com/patent/20220193170
On
September 24, 2020, the Company filed a patent application titled “Personalized Immunotherapies for Reduction of Brain
Inflammation and Suicide Prevention” that discloses means, methods and compositions of matter useful for reduction of brain
inflammation and prevention of suicidal ideations and suicidal attempts. In one embodiment the invention provides utilization of autologous
platelet rich plasma, alone, or admixed with regenerative/anti-inflammatory adjuvants, for reduction of neural inflammation. In one embodiment
autologous PRP is admixed with oxytocin and administered intranasally in a patient at risk of suicidal ideation. In another embodiment,
PRP is admixed with fortified and non-fortified nigella sativa oil and administered intranasally. Other embodiments include utilization
of autologous stromal vascular fraction cells alone and/or admixed with regenerative/anti-inflammatory adjuvants.
Published
on March 24, 2022, https://patents.justia.com/patent/20220088086
On
September 14, 2020, the Company filed a patent application titled “Immunotherapy of Schizophrenia and Schizophrenia Associated
Suicidal Ideation/Suicide” Disclosed are methods, means, and protocols of modifying the immune system so as to induce an immunologically
tolerant state insofar as T regulatory cell number and/or activity is augmented in a patient suffering from schizophrenia. In one embodiment
T regulatory cells are administered to the patient from exogenous sources, be they allogeneic or autologous. In other embodiments, T
regulatory cells are generated endogenously through administration of immature dendritic cells, mesenchymal stem cells, and/or pharmaceutical
means.
On
August 28, 2020, the Company filed a patent application titled “Upregulation of Therapeutic T Regulatory Cells and Suppression
of Suicidal Ideations in Response to Inflammation by Administration of Nutraceutical Compositions Alone or Combined with Minocycline”
which discloses compositions of matter, treatments and protocols useful for induction of T regulatory cells in response to inflammation,
as well as inhibition of suicidal ideations and/or neuroinflammation. In some embodiments the invention teaches the administration of
a therapeutic combination of ingredients comprising of minocycline, pterostilbene, nigella sativa, sulforaphane, and epigallocatechin-3-gallate
(EGCG) to a mammal undergoing upregulation of inflammatory mediators.
Published
on March 3, 2022, https://patents.justia.com/patent/20220062367
On
August 21, 2020, the Company filed a patent application titled “Methods of Determining Risk of Suicide and/or Suicidal
Ideation by Immunological Assessment” which discloses means and methods of identifying risk of suicide and/or suicidal ideation
by assessment of immunologically related cytokines and cells. In one embodiment, a score, termed the “Campbell Score” is
devised based on assessment of serum cytokines, ability of immune cells to make cytokines when stimulated ex vivo, and ability of immune
cells to produce neurotransmitters when stimulated ex-vivo. In one embodiment the concentration of interleukin-6 is utilized as a means
of assessing suicidal propensity along, and/or in combination with metabolites of the enzyme indolamine 2,3 deoxygenase.
On
August 05, 2020, the Company filed a patent application titled “Prevention of Neuroinflammation associated Memory Loss
Using Nutraceutical Compositions” which discloses means, methods, and therapeutic compositions for prevention of memory loss
during situations of neuroinflammation. In one embodiment the invention teaches administration of the therapeutic combination of ingredients
comprising of pterostilbene, Nigella sativa, sulforaphane, and epigallocatechin-3-gallate (EGCG) to a mammal suffering from inflammation
in order to preserver memory function.
Published
on February 10, 2022, https://patents.justia.com/patent/20220040248
On
July 28, 2020, the Company filed a patent application titled “Neuroprotection and Neuroregeneration by Pterostilbene
and Compositions Thereof” with new data demonstrating that the blueberry derived compound pterostilbene possesses numerous
brain protective and potentially brain regenerative activities. The data disclosed by the Company indicates: a) pterostilbene suppresses
inflammatory cytokines TNF-alpha, IL-1 beta and IL-6; b) pterostilbene inhibits death of neurons caused by inflammatory mediators; c)
pterostilbene stimulates production of regenerative factors from cells in the brain such as BDNF, NGF, FGF-1, and FGF-2; and d) pterostilbene
allows/enhances proliferation of endogenous brain stem cells.
Published
on February 3, 2022, https://patents.justia.com/patent/20220031793
On
July 22, 2020, the Company filed a patent application titled “Additive and/or Synergistic Combinations of Metformin with
Nutraceuticals for the Prevention, Inhibition and Treatment of SARS-Cov-2 and Associated COVID-19” showing potent synergy between
QuadraMune™ and the antidiabetic drug metformin in treating COVID-19 associated lung damage models. It was discovered that the
ability of QuadraMune™ to protect the lungs from inflammation that resembles coronavirus-induced pathology is markedly amplified
by concurrent administration of metformin. At a mechanistic level, it was shown that metformin increased the ability of QuadraMune™
to a) increase the number of “healing macrophages” (“M2” macrophages); b) augment production of anti-inflammatory
and regenerative proteins; and c) suppress production of pathological inflammatory proteins.
Published
on January 27, 2022, https://patents.justia.com/patent/20220023237
On
July 13, 2020, the Company filed a patent application titled “Prevention of Pathological Coagulation in COVID-19 and
other Inflammatory Conditions” s directed to the utilization of pterostilbene, and/or nigella sativa extract, and/or sulforaphane,
and/or Epigallocatechin gallate (EGCG) alone or in combination, for the prevention of pathological coagulation. In on embodiment a composition
containing all four ingredients is administered to a patient at risk of hypercoagulation in order to prevent aberrant expression of pro-coagulation
molecules and/or induce expression of molecules known to suppress coagulation. In one embodiment the invention teaches administration
of pterostilbene, thymoquinone, sulforaphane, and EGCG as a means of decreasing expression of tissue factor.
Published
on May 12, 2022, https://patents.justia.com/patent/20220143123
On
June 30, 2020, the Company filed a patent application titled “Augmentation of Natural Killer Cell Activity and Induction
of Cytotoxic Immunity Using Leukocyte Lysate Activated Allogeneic Dendritic Cells: StemVacs™” which describes the process
of preparing allogeneic dendritic cells utilizing a leukocyte lysate based approach. These data support development of StemVacs for conditions
that would benefit from NK activation such as cancer and COVID-19.
Published
on March 31, 2022, https://patents.justia.com/patent/20220096542
On
June 22, 2020, the Company filed a patent application titled “Treatment of SARS-CoV-2 with Dendritic Cells for Innate
and/or Adaptive Immunity” that disclosed means, methods, and compositions of matter for prophylaxis and/or treatment of SARS-CoV-2
by administration of dendritic cells in a manner and frequency sufficient to induce activation of innate and/or adaptive immune responses.
In one embodiment the invention teaches administration of dendritic cells pulsed with one or more innate immune stimulants in a manner
endowing said dendritic cell with ability to induce augmentation of natural killer (NK) cell number and/or activity. In another embodiment
the invention teaches the use of dendritic cells stimulated with innate immune activators in a manner to allow for uptake of viral particles
and presentation of viral epitopes to T cells in order to stimulate immunological activation and/or memory responses.
Published
on December 23, 2021, https://patents.justia.com/patent/20210393681
On
June 15, 2020, the Company filed a patent application titled “Nutraceuticals for Suppressing Indolamine 2,3 Deoxygenase”
from new data showing QuadraMune™ significantly inhibited inflammation associated with memory impairment, as well as reduced levels
of kynurenine. Elevation of kynurenine is associated with activation of indolamine 2,3 deoxygenase, an enzyme associated with inflammation
and depression.
Granted
on January 25, 2022, https://patents.justia.com/patent/11229674
On
June 11, 2020, the Company filed a patent application titled “Nutraceuticals for Reducing Myeloid Suppressor Cells”
which disclosed compositions of matter, treatments and protocols useful for reduction of number and/or activity of myeloid suppressor
cells (MSC). In some embodiments the invention teaches the administration of a therapeutic combination of ingredients comprising of pterostilbene,
Nigella sativa, sulforaphane, and epigallocatechin-3-gallate (EGCG) to a mammal at possessing an increased number and/or activity of
said MSC in which reduction of number and/or activity is desired. In another embodiment, the invention teaches administration of said
therapeutic combination to a mammal infected with viral and/or bacterial infections and/or neoplasia. In some embodiments dosage of said
therapeutic combination is based on inflammatory and/or immunological parameters observed in patients.
Published
on December 16, 2021, https://patents.justia.com/patent/20210386815
On
May 11, 2020, the Company filed a patent application titled “Treatment of COVID-19 Lung Injury Using Umbilical Cord Plasma
Based Compositions” which disclosed means, methods, and compositions of matter useful for the treatment of lung inflammation
associated with viral and bacterial infections, as well as with systemic inflammation, through the administration of umbilical cord blood
derived plasma-based compositions. In one embodiment the invention teaches administration of umbilical cord blood plasma together with
pterostilbene, and/or sulforaphane, and/or thymoquinone, and/or Epigallocatechin gallate (EGCG) and/or n-acetylcysteine in an aerosolized
manner to patients suffering from COVID-19 associated pulmonary deficiencies. In another embodiment, umbilical cord blood plasma is administered
with immune-stimulatory agents in order to concurrently inhibit propagation of viral load in the lung while suppressing pulmonary deficiencies.
On
May 4, 2020, the Company filed a patent application titled “Nutraceuticals for the Prevention, Inhibition and Treatment
of SARS-Cov-2 and Associated COVID-19” which teaches compositions of matter, treatments and protocols useful for prevention
of SARS-CoV-2 infection, as well as inhibition of viral propagation and acceleration of viral cure. In some embodiments the invention
teaches the administration of a therapeutic combination of ingredients comprising of pterostilbene, nigella sativa, sulforaphane, and
epigallocatechin-3-gallate (EGCG) to a mammal at risk of infection with SARS-CoV-2. In another embodiment, the invention teaches administration
of said therapeutic combination to a mammal infected with said SARS-CoV-2. In some embodiments dosage of said therapeutic combination
is based on inflammatory and/or immunological parameters observed in patients with COVID-19.
Granted
on March 8, 2022, https://patents.justia.com/patent/11266707
On
November 4, 2019, the Company filed a patent application titled “Cellular, Organ, and Whole-Body Rejuvenation Utilizing
Cord Blood Plasma and Pterostilbene” that disclosed methods, means, and protocols for stimulation of rejuvenation in single
cells, organs, and organisms by administration of cord blood derived plasma, cord blood plasma concentrates, and cord blood derived exosomes
together with pterostilbene. The invention describes the previously unexpected finding that addition of pterostilbene to cord blood enhances
the rejuvenation properties of cord blood. Said rejuvenation properties include telomere preservation, reduction in beta galactosidase,
and retention of cellular activities.
Published
on May 6, 2021, https://patents.justia.com/patent/20210128638
On
September 9, 2019, the Company filed a patent application titled “Pterostilbene and Formulations Thereof for Protection
of Hematopoiesis from Chemotherapy and Radiation” which disclosed compositions of matter useful for treatment and/or prevention
of hematopoietic injury using pterostilbene and formulations thereof. In one embodiment nanoparticle delivered pterostilbene is administered
subsequent to chemotherapy induced neutropenia in order to accelerate recovery of the hematopoietic compartment. In another embodiment,
pterostilbene is provided concurrently with chemotherapy in order to concurrently assist the neoplasia killing action of the chemotherapy
while protecting the bone marrow from suppression. In contrast to conventionally used agents that protect from neutropenia such as G-CSF
and GM-CSF, the products disclosed can be chronically administered, thus allowing for concurrent use with chemotherapeutic or radiotherapeutic
agents.
On
January 21, 2019, the Company filed a patent application titled “Prevention and Reversion of Chronic Traumatic Encephalopathy
through Administration of “Educated” Monocytes and Progenitors Thereof” that provides means of preventing and/or
reversing chronic traumatic encephalopathy in a patient through the modulation of monocytes as well as monocytic progenitors. In one
embodiment the invention teaches administration of monocytes that have been previously “educated” by exposure to mesenchymal
stem cells in order to endow onto said monocytes properties associated with stimulation of neuroregenerative properties. In some embodiments
monocytes are educated by treatment of monocytic progenitors with conditions capable of endowing anti-inflammatory and regenerative conditions,
said conditions include culture with epigenetic modifying agents. In other embodiments, the invention teaches the manipulation of cord
blood derived monocytes as a starting population of cells for education by culture with mesenchymal stem cells.
On
January 21, 2019, the Company filed a patent application titled “Autologous Neurogenic Cells and Uses Thereof for Professional
Athletes at Risk of Chronic Traumatic Encephalopathy” which disclosed are means, compositions of matter and methods of business
for treating Chronic Traumatic Encephalopathy (CTE) using autologous primary cells and modified cells of autologous origin which have
been banked. In one embodiment of the invention autologous dedifferentiation cells are generated and stored for future administration
in patients which have suffered CTE. In other embodiments, dedifferentiated cells are differentiated into neurons or neuronal progenitor
cells and subsequently administered locally or systemically or in a combination. In other embodiments autologous cells are maintained
in an undifferentiated manner and/or neurologically differentiated state and utilized as a conditioning source in an extracorporeal circulatory
system replicating clinical stage extracorporeal liver perfusion (ECLP) with substitution of autologous
dedifferentiated, neurologically differentiated or a combination of said cells instead of hepatic cells.
On
December 18, 2018, the Company filed a patent application titled “Treatment of Chronic Traumatic Encephalopathy via RNA
Administration” which disclosed are protocols, treatment means, and compositions of matter useful for treatment of Chronic
Traumatic Encephalopathy through administration of RNA or modified RNA molecules. In one embodiment said RNA is generated to activate
various toll like receptors (TLR), of which said activation leads to production of cytokines which paradoxically lead to protection from
Chronic Traumatic Encephalopathy, wherein said protection constitutes a) reduction in glial cell activation, b) neuronal apoptosis due
to excitotoxicity; and c) stimulation of endogenous regenerative processes including endothelial progenitor cell mobilization, proliferation
of neuronal progenitor cells in the dentate gyrus and subventricular zones. In one particular embodiment targeting of RNA molecules is
performed to specific brain cells including pyramidal neurons through the use of liposomes, exosomes, apoptotic bodies, nanoparticles
and shark or cameloid antibodies is disclosed.
On
September 25, 2018, the Company filed a patent application titled “Pterostilbene and Formulations Thereof for Treatment
of Pathological Immune Activation” that teaches treatments, protocols, and compositions of matter are described for reduction
of pathological immune system activation. In one embodiment, pterostilbene and/or formulations thereof are administered in a patient
suffering from cytokine release syndrome at a concentration and frequency sufficient to reduce abnormal cytokine production and thus
treat the cause of said cytokine release syndrome. Formulations of pterostilbene are disclosed for rapid release, enhanced biodistribution,
and targeting to cytokine releasing effectors are disclosed for use in the practice of the invention.
On
September 17, 2018, the Company filed a patent application titled “Pterostilbene and Compositions Thereof for Prevention
and Treatment of Chronic Traumatic Encephalopathy” that teaches means, methods, and compositions of matter useful for prevention
of chronic traumatic encephalopathy. In one embodiment of the invention, disclosed is utilization of pterostilbene and/or pterostilbene
based compounds for prevention and/or treatment of chronic traumatic encephalopathy. In one embodiment, the invention teaches administration
of pterostilbene and/or pterostilbene based compounds for reduction of taupathy associated with chronic traumatic encephalopathy.
On
August 13, 2018, the Company filed a patent application titled “Enhancement of Ozone Therapy using Pterostilbene”
that disclosed methods, means and compositions of matter using pterostilbene for enhancing therapeutic efficacy of ozone therapy in the
field of oncology. The invention provides previously unknown synergies between ozone administration together with pterostilbene at inducing
direct and indirect cytotoxicity to cancer cells. The invention provides means of delivery, administration, and therapeutic protocols
for treatment of cancer patients. In one embodiment combination of ozone therapy together with pterostilbene is utilized to overcome
drug resistance.
On
October 08, 2017, the Company filed a patent application titled “Synergistic Inhibition of Glioma Using Pterostilbene
and Analogues Thereof” that teaches methods, means and compositions of matter for utilizing pterostilbene and analogues thereof
for suppression of viability, metastasis and proliferation of glioma cells alone, or together with immunotherapy, chemotherapy, or radiotherapy
means. In one embodiment said pterostilbene augments immunogenicity of glioblastoma cells so as to enhance killing by immune cells or
complement subsequent to damage of said glioblastoma cells by chemotherapy, radiotherapy, or immunotherapy.
On
April 26, 2017, the Company filed a patent application titled “Augmentation of Stem Cell Activity using Pterostilbene
and Compositions Containing Pterostilbene” that disclosed means of augmenting circulating endogenous stem cells through administration
of an effective amount of pterostilbene or derivatives thereof. In one embodiment a patient with reduced levels of circulating endothelial
progenitor cells is treated with pterostilbene at a concentration and frequency sufficient to restore, and/or enhance levels of circulating
endothelial progenitor cells (EPC). In another embodiment endogenous levels of stem cells are restored or enhanced by administration
of pterostilbene, said endogenous stem cells comprising cells of the dentate gyrus, subventricular zone, hepatic stem cells, cardiac
stem cells, and hematopoietic stem cells.
On
March 29, 2017, the Company filed a patent application titled “Stimulation of Immunity to Tumor Stem Cell Specific Proteins
by Peptide Immunization” that discloses treatment of cancer is disclosed through administration of proteins or specific peptides
found on tumor stem cells in vivo, in a matter eliciting monocyte or dendritic cell migration in order to allow uptake of said administrated
proteins or peptides, followed by administration of a maturation signal in vivo. The invention provides for treatment of cancer through
induction of anticancer immunity and/or immunity towards tumor initiating stem cells.
On
March 29, 2017, the Company filed a patent application titled “Targeting the Tumor Microenvironment through Nutraceutical
Based Immunoadjuvants” that disclosed compositions useful for the treatment of cancer which modulate tumor associated immunosuppression,
thus acting as immunoadjuvants. In one embodiment a composition containing apigenin, is provided, said composition useful for inhibition
of tumor associated immune suppression mediated through the molecule indolamine 2,3 deoxygenase (IDO). In another embodiment, liposomal
apigenin is administered as a means of decreasing IDO expression.
On
March 29, 2017, the Company filed a patent application titled “Activated Leukocyte Extract for Repair of Innate Immunity
in Cancer Patients” that disclosed are compositions, methods of use, and pharmaceutical preparations useful for modulation
of immune responses. In one embodiment a composition is extracted polyvalently activated peripheral blood mononuclear cells through dialysis.
Said immune modulator is useful for treatment of cancer and alleviation of cancer associated immune depression. In one embodiment, said
immunomodulator acts as a costimulatory of T cell activation by modulation of cytokine production. In one embodiment said immune modulator
is concentrated for miRNA species capable of activating innate immune cells.
On
March 29, 2017, the Company filed a patent application titled “Augmentation of Anti-Tumor Immunity by Mifepristone and
Analogues Thereof” which relates to compositions of matter and methods useful for improving a treatment outcome and/or an alteration
of immunity in a condition that benefits from immune stimulation. In particular, one embodiment of the invention teaches administration
of sufficient doses of mifepristone or a derivative, alone, or in combination with an immunotherapeutic such as, but not limited to,
an antibody, a vaccine, a cytokine, or a medicament whose therapeutic activity is associated with immune modulation.
On
March 29, 2017, the Company filed a patent application titled “Methods of Re-Activating Dormant Memory Cells with Anticancer
Activity” that disclosed methods, protocols, and compositions of matter useful for stimulation of anticancer immune responses.
In one embodiment of the invention culture of buffy coat cells is performed in an environment resembling non-physiological conditions.
Buffy coat derived products are subsequently harvested, concentrated, and added to a culture of monocytes and lymphocytes. Conditioned
media from said second culture is subsequently utilized as an injectable solution for stimulation of anticancer immunity.
On
March 29, 2017, the Company filed a patent application titled “Modulation of Oral Microbiome for Treatment of Periodontitis”
that disclosed methods, means, and compositions of matter useful for inhibition of, reduction in progression and reversion of periodontitis.
In one embodiment the invention provides prebiotic and/or probiotic compositions which modulation the oral microbiome in order to ameliorate,
prevent or reverse periodontitis. In one embodiment a composition is administered into the oral cavity containing Actinomyces naeslundii,
Actinomyces odontolyticus, Streptococcus thermophilius, Lactobaccilus brevis and Lactobacilius plantarum. Administration may be performed
using various means including a mouthwash, a patch, a toothpaste, or in a preferred embodiment said prebiotic and/or probiotic compositions
are delivered via a mouth tray.
On
July 20, 2016, the Company filed a patent application titled “Prevention of Pregnancy Complications by Probiotic Administration”
which disclosed methods, protocols and compositions of matter for the treatment of pregnancy complications through immune modulation
of a mammal in need. In one embodiment the invention provides probiotic compositions for immune modulation to decrease risk of pregnancy
complications. Pregnancy complications include recurrent spontaneous abortions (RSA), preterm birth, pre-eclampsia including hemolysis
elevated liver enzymes low platelets (HELP), premature rupture of the membrane, Antepartum hemorrhage including placental abruption,
chorioamnionitis, Intrauterine growth restriction, placenta pravaevia, sequalae of intraamniotic infection.
Published
on January 26, 2017, https://patents.justia.com/patent/20170020930
On
July 20, 2016, the Company filed a patent application titled “Exosome Mediated Innate and Adaptive Immune Stimulation
for Treatment of Cancer” that teaches means of stimulating innate and/or adaptive immunity to cancer by administration of exosomes.
Stimulation of innate immunity involves modifying exosomes by chemical addition of innate immune stimulators, whereas stimulation of
adaptive immunity involves pulsing dendritic cells generating exosomes with antigens, in some cases, pulsing with Brother of the Regulator
of Imprinted Sites (BORIS) proteins, peptides, or altered peptide ligands thereof.
On
July 8, 2015, the Company filed a patent application titled “Augmentation of Oncology Immunotherapies by Pterostilbene
Containing Compositions” that disclosed compositions of matter and methods useful to augmentation of immune responses to tumors.
In one embodiment, a pterostilbene containing composition is administered to a cancer patient at a sufficient concentration and frequency
to induce de-repression of tumor targeting immune responses. In one specific embodiment of the present invention, pterostilbene enhances
antibody dependent cellular toxicity (ADCC) and in turn augments efficacy of FDA approved antigen specific immunotherapeutics such as
trastuzumab (Herceptin) and other monoclonal antibody therapies used for treating cancer.
Issued
and Granted Patents:
On
June 20, 2017, the US Patent and Trademark Office issued and granted U.S. Patent No.: 9,682,047 titled “Augmentation
of oncology immunotherapies by pterostilbene containing compositions” that discloses compositions and methods useful to enhancing,
improving, or eliciting anti-tumor immune responses are disclosed. A pterostilbene containing composition is administered to a cancer
patient at a sufficient concentration and frequency to induce de-repression of tumor targeting immune responses. The composition enhances
antibody dependent cellular toxicity (ADCC) and augments efficacy of antigen specific immunotherapeutics such as trastuzumab and other
monoclonal antibody therapies useful for treating cancer. See: https://patents.justia.com/patent/9682047.
On
January 25, 2022, the US Patent and Trademark Office issued and granted U.S. Patent No.: 11,229,674 titled “Nutraceuticals
for suppressing indolamine 2,3 deoxygenase” which disclosed are compositions of matter, treatments and protocols useful for
reduction of expression and/or activity of indolamine 2,3 deoxygenase (IDO). In some embodiments the invention teaches the administration
of a therapeutic combination of ingredients comprising of pterostilbene, Nigella sativa, sulforaphane, and epigallocatechin-3-gallate
(EGCG) to a mammal at possessing an increased expression and/or activity of said IDO in which reduction of number and/or activity is
desired. In another embodiment, the invention teaches administration of said therapeutic combination to a mammal infected with viral
and/or bacterial infections and/or neoplasia. In some embodiments dosage of said therapeutic combination is based on inflammatory and/or
immunological parameters observed in patients. See: https://patents.justia.com/patent/11229674.
On
March 08, 2022, the US Patent and Trademark Office issued and granted U.S. Patent No.: 11,266,707 titled “Nutraceuticals
for the prevention, inhibition, and treatment of SARS-CoV-2 and associated COVID-19” that disclosed methods of treating or
preventing complications associated with a SARS-CoV-2 infection, comprising: administration of a combination comprising: a) Green Tea
and/or extract thereof; b) Blueberry and/or extract thereof; c) Nigella sativa and/or extract thereof; and d) broccoli and/or extract
thereof in an amount and frequency sufficient to treat or prevent complications associated with said SARS-CoV-2 infection. See: https://patents.justia.com/patent/11266707.
Immune-Oncology
– Right To Try
In
May of 2018, President Donald J. Trump signed into the law, the Right To Try bill. In 2015/2016 TSOI began and completed a 10 patient
clinical trial of advanced cancer patients in Mexico at the Pan Am Cancer Treatment Center located in Tijuana Mexico using our dendritic
cell vaccine code named StemVacs. TSOI has since generated GCP documentation for the previously treated 10 patients into a Phase I trial,
which will be presented to the FDA by TSOI as part of an Ex-US trial compliant with 21 CFR 312.120 Foreign clinical studies not conducted
under an IND. This is a required step to conform to the new Right To Try law.
StemVacs
is an immunotherapy platform that consists of 5 components. The overarching approach to the StemVacs Immunotherapy Platform is as follows:
| |
1. | Treat innate immune
suppression: Administration of oral apigenin/NanoStilbene (Cancer DeTox Product) to decrease immune suppressive toxic molecules made
by tumor and tumor microenvironment. |
| |
2. | Treat adaptive
immune suppression: Administration of MemoryMune to activate dormant memory cells recognizing the tumor. Administration of LymphoBoost
to repair deficient IL-12 production. |
| |
3. | Stimulation of
immune response to cancer stem cells (StemVacs). |
| |
4. | Consolidation and
maintenance of immunity: Cycles of StemVacs, supported by innaMune and LymphoBoost |
StemVacs
Autologous is a subcutaneously administered vaccine comprised of immune stimulatory peptides resembling cancer stem cell specific proteins.
StemVacs
Allogeneic is a subcutaneously administered vaccine comprised of immune stimulatory peptides resembling cancer stem cell specific proteins.
Cancer
Metabolic DeTox: This is an orally administered agent that is derived from various herbs termed apigenin. The unique property of
apigenin is that it inhibits a cancer associated metabolic pathway that degrades the amino acid tryptophan. Specifically, apigenin inhibits
the enzyme indolamine 2,3 deoxygenase (IDO), which is responsible for breaking down tryptophan in the vicinity of the tumor and generating
by-products such as kynurenine. It is known that immune activation is dependent on tryptophan being present in the tumor environment.
The depletion of tryptophan and generation of kynurenine by tumor cells and tumor associated cells is a major cause of immune suppression
in cancer. By administering Cancer Metabolic DeTox, the innate arm of the immune system has a chance to regenerate. This positions the
patient for better outcome after administration of specific immune stimulating vaccines.
MemoryMune:
This is a product derived from a two-step culture process of donor blood cells. The product MemoryMune reawakens dormant immune memory
cells. It is known that many cancer patients possess memory T cells that enter the tumor, however, once inside the tumor these cells
are inactivated. MemoryMune contains a unique combination of growth factors specific for immune system cells called “cytokines”.
LymphoBoost:
LymphoBoost is a proprietary formulation of Misoprostol, a drug approved for another indication, which we have shown to be capable of
stimulating lymphocytes, particularly NK cells and T cells, both critical in maintaining anti-tumor immunity.
innaMune:
This is a biological product derived from tissue culture of blood cells derived from healthy donors. It is a combination of cytokines
that maintain activity of innate immune system cells, as well as having ability to shift M2 macrophages to M1.
Chronic
Traumatic Encephalopathy (CTE), and Traumatic Brain Injury (TBI) – Right To Try
On
December 10, 2018, Therapeutic Solutions International, Inc., announced the signing of an agreement between TSOI and Jadi Cell LLC for
licensing of the Jadi Cell universal donor adult stem cell, as covered in US Patent No.: 9,803,176 B2 for use in Chronic Traumatic Encephalopathy
(CTE), and Traumatic Brain Injury (TBI).
In
addition, on February 9, 2021, we obtained exclusive rights under the same for use of US Patent No.: 9,803,176 B2 in the treatment of
acute respiratory distress syndrome (ARDS) and other lung pathologies. The JadiCell was reported in a publication from the University
of Miami following a Phase 1/2 clinical trial, demonstrating intravenous administration of JadiCells, resulted in a significant survival
improvement in COVID-19 patients. The Phase 1/2 double blind, placebo-controlled trial treated 12 advanced COVID-19 patients with 100
million JadiCells™ intravenously at days 0 and 3, and 12 patients received placebo control. At 28 days 91% of JadiCell™ treated
patients survived whereas only 42% of patients in the placebo group survived. There were no adverse effects associated with JadiCell™
administration. For those treated with the JadiCell under the age of 85 the survival rate was 100% and in those over 85 the survival
rate was 91% making the JadiCell the most effective therapy to date in the entire world to treat ARDS.
The
Jadi Cell product, which belongs to the mesenchymal stem cell (MSC) family of cells, is a unique adult stem cell, which produces higher
levels of therapeutic factors compared to other stem cells. The cells have demonstrated safety in animal models and pilot human trials.
The Jadi Cell product is generated from umbilical cords, which are a source of medical waste and available in large quantities at inexpensive
prices.
Chronic
Traumatic Encephalopathy (CTE) is caused by repetitive concussive/sub-concussive hits to the head sustained over a period of years and
is often found in football players. The condition is characterized by memory loss, impulsive/erratic behavior, impaired judgment, aggression,
depression, and dementia. In many patients with CTE, it is anatomically characterized by brain atrophy, reduced mass of frontal and temporal
cortices, and medial temporal lobe.
Traumatic
brain injury (TBI) is an insult to the brain, not of a degenerative or congenital nature, but caused by external physical force that
may produce a diminished or altered state of consciousness, which results in an impairment of cognitive abilities or physical functioning.
CTE
represents a significant unmet medical need which we believe is amenable to stem cell intervention. We are eager to accelerate treatments
and potential cures for debilitating conditions such as CTE and traumatic brain injury and plan to leverage New regulatory pathways such
as the recently approved “Right to Try” Law to deliver these medicines as soon as possible to patients which currently have
no other options.
The
Jadi Cell product because of its advanced stage of development in contrast to other stem cell types, which require years, if not decades
of development before entry into American patients, will allow us we believe to be treating patients within 12 months. Currently means
of isolating, producing, scaling up, and delivery of the cells has all been worked out by Jadi Cell and Collaborators.
On
December 17, 2020, the Company filed an Investigational New Drug (IND) application seeking permission from the Food and Drug Administration
(FDA) to initiate a Phase I/II clinical trial assessing safety and signals of efficacy for treatment of Chronic Traumatic Encephalopathy
(CTE) patients with JadiCells™.
On
August 4th, 2021, the Company announced clearance from the Food and Drug Administration (FDA) to initiate a Phase III pivotal trial for
registration of the Company’s JadiCell™ universal donor stem cell as a treatment for COVID-19 associated lung failure under
IND # 19757. In previous studies the Company has demonstrated the superior activity of JadiCell™ to other types of stem cells including
bone marrow, adipose, cord blood, and placenta. Furthermore, the JadiCell™ was shown to be 100% effective in saving the lives of
COVID-19 patients under the age of 85 in a double-blind placebo controlled clinical trial with patients in the ICU on a ventilator. In
patients over the age of 85 the survival rate was 91%.
Sars/CoV2
Clinical Programs
On
June 8, 2020, the Company announced the initiation of a clinical trial aimed at demonstrating safety and efficacy of its immune-boosting
formulation QuadraMune™. The trial is anticipated to recruit 500 subjects at risk of SARS-CoV-2 infection, the type of coronavirus
which causes COVID-19. The new clinical trial has been granted ClinicalTrials.gov Identifier: NCT04421391 and is listed and registered
on the Federal Clinical Trial registry.
The
Company announced recently submission of a publication providing preclinical data which supports repositioning of its Cancer Immunotherapy
StemVacs™ as a candidate for treatment of COVID-19. StemVacs™ is based on activating universal donor immune system cells
called dendritic cells in a manner so that upon injection they reprogram the body’s “Natural Killer” cells.
Natural
killer cells are the most potent cell type in the body in terms of killing viruses. Unfortunately, natural killer cells also produce
chemicals called cytokines which at high concentrations can be lethal. The current data suggests that StemVacs™ can activate natural
killer cells while at the same time suppressing lung inflammation. This dual mechanism of action makes StemVacs™ a promising candidate
for treatment of coronavirus.
Schizophrenia/Suicide
Clinical Programs
On
October 29, 2020, the Company announced publication on the NIH clinical trials website of its newly initiated trial aiming to validate
a blood-based diagnostic for predicting suicide risk and is listed as NCT04606875.
The
Campbell Score™, which is a patent-pending method of quantifying inflammatory-associated biological markers, has previously been
shown in pilot investigator-initiated studies to correlate with propensity for suicide. Based on positive feedback from collaborators,
the Company decided to initiate a formal clinical trial to validate correlations between the Campbell Score™ and established psychiatric
assessment tools of suicidal propensity. Currently the only means of quantifying predisposition to suicide is based on psychological,
question-based techniques.
On
December 31, 2020, the Company signed license agreements with Campbell Neurosciences Inc., a partially owned company, for access to the
9 patents filed related to the previous Campbell Neurosciences Division. The patents are:
| |
1. | 63/128759 Immunotherapy
for Opioid Addiction |
| |
| |
| |
2. | 63/122862 Treatment
of Major Depressive Disorder and Suicidal Ideations Through Stimulation of Hippocampal Neurogenesis Utilizing Plant-Based Approaches |
| |
| |
| |
3. | 63/105964 Protection/Regeneration
of Neurological Function by Endothelial Protection/Rejuvenation using Stem Cells for Treatment of Conditions such as Chronic Traumatic
Encephalopathy and Schizophrenia |
| |
| |
| |
4. | 17/030416 Personalized
Immunotherapies for Reduction of Brain Inflammation and Suicide Prevention |
| |
| |
| |
5. | 63/077723 Immunotherapy
of Schizophrenia and Schizophrenia Associated Suicidal Ideation/Suicide |
| |
| |
| |
6. | 63/071381 Upregulation
of Therapeutic T Regulatory Cells and Suppression of Suicidal Ideations in Response to Inflammation by Administration of Nutraceutical
Compositions Alone or Combined with Minocycline |
| |
| |
| |
7. | 63/068388 Methods
of Determining Risk of Suicide and/or Suicidal Ideation by Immunological Assessment |
| |
8. | 63/061202 Prevention
of Neuroinflammation associated Memory Loss Using Nutraceutical Compositions |
| |
| |
| |
9. | 63/057315 Neuroprotection
and Neuroregeneration by Pterostilbene and Compositions Thereof |
Additionally,
Campbell Neurosciences Inc. has entered into purchase agreements with Therapeutic Solutions International ensuring a continued supply,
at a discounted rate, of nutraceuticals which are being explored for antiinflammation/suicide prevention activity.
Treatment
of Chronic Obstructive Pulmonary Disease (COPD) Using JadiCell™ Universal Donor Adult Stem Cells
COPD
is a consistently progressive, ultimately fatal disease for which no treatment exists capable of either reversing or even interrupting
its course. It afflicts more than 5% of the population in many countries, and it accordingly represents the third most frequent cause
of death in the U.S., where it accounts for more than 600 billion in health care costs, morbidity, and mortality.
JadiCell
UC-MSCs have been utilized to treat patients with severe COVID-19 and have yielded promising results, preventing or attenuating the cytokine
storm. JadiCells have been recently introduced intravenously in patients with a neurodegenerative disorders and have been approved for
testing in patients with Type 1 Diabetes (TlD). We hypothesize that JadiCells will exert beneficial therapeutic effects in COPD.
The
proposed clinical trial will recruit 10 patients of 40-80 years of age with moderate to severe COPD (GOLD [Global Initiative for Chronic
Lung Disease]. The enrolled patients will be treated with 100 million JadiCells intravenously with two injections spaced at 48 hours.
These
two treatments will be performed on days 0 and 2. JadiCell dosing will be performed at approximately 100 million cells/infusion delivered
at a maximum rate of 2 million cells/min per infusion.
Assessment
of lung function and safety will be performed on days 0, 30, 60, and 90. Safety will be assessed by occurrence of adverse events (AEs)
during either study-drug infusion or by physician assessments and laboratory evaluations, a record of COPD exacerbations will be maintained
for each patient.
Efficacy
measures will include improvement from baseline in pulmonary functions (FEV 1 , FVC, FEV 1 /FVC, total lung capacity by plethysmography,
27 single-breath carbon monoxide diffusing capacity, exercise performance [6-min walk test (6MWT)], and dyspnea assessment [Borg scale]),
and QOL (St. George’s Respiratory Questionnaire, and global assessment of patient status). COPD exacerbations were assessed as
the time to the first exacerbation and as the ratio of the rate of exacerbations between MSC- and placebo-treated patients.
Cellular
Manufacturing and Cell Banking
On
October 18, 2021, the Company announced the formation of Allogen Biologics Inc, a wholly owned subsidiary of TSOI. Allogen Biologics
will house intellectual property and Standard Operating Procedures related to generation of the Company’s existing and anticipated
cellular therapeutics. In addition, Allogen will house and maintain all relevant cell banks.
On
May 10, 2022, Allogen Biologic, Inc, and Therapeutic Solutions International Inc, entered into an Exclusive Patent License Agreement
(EPLA) for Patent Application Serial No. 63/254,469, filed by Licensor and titled as: Umbilical Cord Derived Regenerative and Immune
Modulatory Stem Cell Populations.
RISK
FACTORS
Risks
Associated with Our Business and Industry
This
prospectus contains forward-looking statements concerning our future programs and cash needs as well as our plans and strategies. These
forward-looking statements are based on current expectations, and we assume no obligation to update this information, except as required
by applicable laws and regulations. Numerous factors could cause actual results to differ significantly from the results described in
these forward-looking statements, including the following risk factors.
We
have identified material weaknesses in our internal control over financial reporting.
We
are required to comply with the provisions of Section 404 of the Sarbanes-Oxley Act of 2002, which require us to maintain an ongoing
evaluation and integration of the internal controls of our business.
We
evaluated our existing controls as of December 31, 2021. Our Chief Executive Officer and Chief Financial Officer identified material
weaknesses in our internal control over financial reporting. A “material weakness” is a control deficiency, or combination
of control deficiencies in internal control over financial reporting such that there is a reasonable possibility that a material misstatement
of the annual or interim financial statements will not be prevented or detected. Readers are directed to review that portion of this
Form 10-K entitled Item 9A Controls and Procedures for a detailed disclosure.
Under
Section 404 and the SEC’s rules, a company cannot find that its internal control over financial reporting is effective if any “material
weaknesses” exist in its controls over financial reporting.
Our
liquidity and capital resources are very limited.
Our
ability to fund operating activities is also dependent upon our ability to access external sources of financing and our ability to effectively
manage our expenses in relation to revenues. Our ability to fund working capital and anticipated capital expenditures will depend on
our future performance, which is subject to general economic conditions specific to the health, supplements and nutrition products industries,
consumer demand for our products, competition and other factors that are beyond our control. There can be no assurance that our operations
and access to external sources of financing will continue to provide resources sufficient to satisfy our liabilities arising in the ordinary
course of business.
We
will require significant additional external financing to implement our business plan.
We
will require external financing to sustain our operations, support our expansion, achieve or maintain profitability, or, should we become
subject to unforeseen events or circumstances, continue as a going concern. There can be no assurance that we will be able to secure
any such external financing, or, if we are able to secure such external financing, that it will be on terms favorable, or even acceptable,
to us. Any inability to achieve or sustain profitability or otherwise secure external financing would have a material adverse effect
on our business, financial condition, and results of operations, raising substantial doubts as to our ability to continue as a going
concern, and we may ultimately be forced to seek protection from creditors under the bankruptcy laws or cease operations, which may result
in a substantial or complete loss of your invested capital.
We
may not be able to effectively manage our potential growth and the execution of our business plan.
Our
potential growth and the execution of our business plan together are likely to place significant strain on our managerial, operational
and financial resources. To effectively manage our potential growth and execute our business plan, we will need to, among other things:
| ● | retain additional
personnel across several departments in the Company; |
| ● | develop strong
customer loyalty for new products in a crowded competitive marketplace; |
| ● | continue to establish
and continue to increase awareness of our brands; |
| ● | price our products
and services at points which will allow us to maximize sales while at the same time maximizing gross profit margins; |
| ● | establish, maintain,
expand and manage multiple relationships with various vendors, strategic partners, licensees and other third parties, including suppliers
of the products we sell on our website and elsewhere, warehousing distributors, shipping companies and others; |
| ● | rapidly respond
to competitive developments, particularly when new high-demand products become available; |
| ● | build an operations
structure to support our business and provide efficient and effective customer service and support; |
| ● | expand our IT infrastructure
to respond to increasing customer traffic to our website, demand for content from site users and to manage growing e-commerce transactions; |
| ● | establish and maintain
effective financial and management controls, reporting systems and procedures; |
| ● | provide competitive
employee salaries and benefit packages; and, |
| ● | avoid lawsuits
and other adverse claims. |
There
can be no assurance that we will be able to accomplish any or all of the above goals. If we prove unable to effectively execute our business
plan or manage our growth, it is likely to have a material adverse effect on our business, financial condition, including liquidity and
profitability, and our results of operations.
If
our proposed product sales model does not successfully operate at a profit our growth strategy may be impeded.
To
effectively expand and meet our growth objectives our products sales model must be executed upon in a profitable manner. Profitability
is dependent upon a variety of factors, some beyond our control, including, but not limited to the amount of traffic we can consistently
attract to our brand, to retail sales in “brick and mortar” retailers, to our website, and our ability to stock or otherwise
make available products that our customers purchase, our ability to stock or otherwise make available the best new products as they enter
the market, our ability to provide consistent and superior customer service, the general economic conditions, particularly in the U.S.,
that could impact the amount of money customers spend collectively on the products we sell, and/or that could reduce the amount of money
our average customer spends, and/or could reduce the number or frequency of repeat orders for products, and/or could result in customers
finding products in other venues if they can find those products for a lower price. Other factors that could impact our ability to execute
on our business model in a profitable manner include, but are not limited to, competition in our markets, recruiting, training and retaining
qualified personnel and management, maintenance of required local, state and federal governmental approvals and permits, costs associated
with principal component products and supplies, delivery shortages or interruptions, consumer trends, our ability to finance operations
externally, changes in supply or prices of the products we sell and disruptions or business failures among our product suppliers, distributors,
warehouses or shippers. Any failure to operate in a profitable manner could hurt our ability to meet our growth objectives by attracting
licensees, and our business, financial condition, including liquidity and profitability, and our results of operations would be negatively
affected.
If
we cannot stock, warehouse or otherwise provide product to customers in a consistent, reliable and cost-effective manner our growth strategy
may be impeded.
As
our growth strategy depends to a large extent on our ability to sell various products to consumers on our website and in traditional
“brick and mortar” retailers, if we cannot supply those products in a consistent, reliable and cost-effective manner, we
may lose customers. To accomplish a consistent, reliable and cost-effective method for supplying product to customers, we must successfully
engage with suppliers at a number of levels, including warehousing agreements, stocking agreements and other forms of distribution. Our
ability to conclude such arrangements with specific product suppliers may involve the need for trade finance, purchasing agreement finance
and other capital. In addition, we may encounter problems in fulfilling orders due to business conditions among the products companies
themselves, many of which problems are beyond our control. If we are unable to establish and continue such agreements and structures
with products companies, our growth strategy may be impeded, which could negatively affect our business, financial condition, including
liquidity and profitability, and our results of operations.
We
face significant competition for our products.
The
markets in which we operate are intensely competitive, continually evolving and, in some cases, subject to rapid change. Our competitors
include:
| ● | traditional and
well-established companies with recognized and well patronized brands in the nutritional supplements and health products industry segment; |
| ● | entrenched nutritional
supplements and health products companies with well-known customer on-line services and portals and other high-traffic web sites that
provide sales access to healthcare and nutritional supplements and related products; and |
| ● | companies that
focus on providing on-line and/or off-line healthcare related content, including some that promote competitor brands. |
Many
of our competitors have greater financial, technical, product development, marketing and other resources than we do. These companies
may be better known than we are and have more customers than we do. We cannot provide assurance that we will be able to compete successfully
against these companies or any alliances they have formed or may form. If we are unable to compete with one or more of our competitors,
our growth strategy may be impeded, which could negatively affect our business, financial condition, including liquidity and profitability,
and our results of operations.
Product
revenue.
Although
we intend and continue to develop and introduce new nutraceutical products, we currently market and sell encapsulated ProJuvenol®,
DermalStilbene, IsoStilbene, and NeuroStilbene, NanoStilbene®, NanoPlus, Nano Cannabidiol, NLRP3 Trifecta, and QuadraMune®
are all powerful antioxidants. We currently do not have a broad portfolio of other products completed that we could rely on to
support our operations if we were to experience any difficulty with the manufacture, marketing, sale, or distribution of our current
products.
Government
regulation could adversely affect our business.
Our
products and their associated component ingredients are subject to existing and potential government regulation. Our failure, or the
failure of our business partners or third-party providers, to accurately anticipate the application of laws and regulations affecting
our products and the manner in which we deliver them, or any failure to comply, could create liability for us, result in adverse publicity,
or negatively affect our business. In addition, new laws and regulations, or new interpretations of existing laws and regulations, may
be adopted with respect to consumer protection and other issues, including pricing, products liability, copyrights and patents, distribution
and characteristics and quality of products and services. We cannot predict whether these laws or regulations will change or how such
changes will affect our business. Any of this government regulation could impact our growth strategy, which could negatively affect our
business, financial condition, including liquidity and profitability, and our results of operations.
The
Company’s success may depend upon its ability to protect its patents and proprietary technology.
The
Company owns patents for several of its products and relies upon the protection afforded by its patents and trade secrets to protect
its technology. The Company’s success may depend upon its ability to protect its intellectual property. However, the enforcement
of intellectual property rights can be both expensive and time consuming. Therefore, the Company may not be able to devote the resources
necessary to prevent infringement of its intellectual property. Also, the Company’s competitors may develop or acquire substantially
similar technologies without infringing the Company’s patents or trade secrets. For these reasons, the Company cannot be certain
that its patents and proprietary technology will provide it with a competitive advantage.
Third
parties may claim that we are infringing their intellectual property, and we could suffer significant litigation or licensing expense
or be prevented from providing certain services, and which may otherwise harm our business.
We
could be subject to claims that we are misappropriating or infringing intellectual property, trade secrets or other proprietary rights
of others. These claims, even if not meritorious, could be expensive to defend and divert management’s attention from our operations.
If we become liable to third parties for infringing these rights, we could be required to pay substantial damage awards and to develop
non-infringing products, obtain a license or cease selling the products that use or contain the infringing intellectual property. We
may be unable to develop non-infringing products or obtain a license on commercially reasonable terms, or at all. Any claims against
our company for infringement could impede our growth strategy, which could negatively affect our business, financial condition, including
liquidity and profitability, and our results of operations.
We
may be subject to claims brought against us as a result of product liability claims.
The
Company presently does not carry products liability insurance covering its development, marketing and sale of the products it intends
to sell. However, the Company intends and expects to acquire adequate and reasonable products liability insurance after the business
is funded. There is no guarantee that the amount of funds raised by virtue of this offering will be adequate to acquire or maintain such
insurance. Should the Company not acquire adequate funding to obtain products liability insurance, its uninsured operations would expose
the Company and its shareholders to material risks should products liability claims arise. Any claims can be costly to defend, and any
successful products liability claim against the Company could materially impact the ability of the Company to continue as a going concern
and therefore place your total investment in the Company at risk of being a complete loss.
We
may be subject to claims brought against us as a result of product associated content we provide.
Consumers
are reasonably expected to access health-related information regarding our products through our on-line web site. If our content, or
content we obtain from third parties, contains inaccuracies, it is possible that consumers or others may sue us for various causes of
action. Although our planned web site contains terms and conditions, including disclaimers of liability, that are intended to reduce
or eliminate our liability, the law governing the validity and enforceability of on-line agreements with consumers that provide the terms
and conditions for use of our public or private portals are unenforceable. A finding by a court that these agreements are invalid and
that we are subject to liability could harm our business and require costly changes to our business. We have planned editorial procedures
in place to provide quality control of the information that we publish or provide. However, we cannot assure you that our editorial and
other quality control procedures will be sufficient to ensure that there are no errors or omissions in particular content. Even if potential
claims do not result in liability to us, the fact that we would need to investigate and defend against these claims could be expensive
and time consuming and could divert management’s attention away from our operations. In addition, our business is in part based
on establishing a reputation amongst consumers that our portals as trustworthy and dependable sources of healthcare information. Allegations
of impropriety or inaccuracy, even if unfounded, could therefore harm our reputation and business, which could negatively affect our
business, financial condition, including liquidity and profitability, and our results of operations.
Changes
in commodity and other operating costs or supply chain and business disruptions could adversely affect our results of operations.
Changes
in product costs are a part of our business; any increase in the prices that suppliers charge for their products could adversely affect
our operating results. We remain susceptible to increases in prices as a result of factors beyond our control, such as general economic
conditions, seasonal fluctuations, weather conditions, demand, safety concerns, product recalls, labor disputes and government regulations.
We rely on third-party distribution companies to deliver ingredients to our manufacturers and ultimately our products to customers. Interruption
of distribution services due to financial distress or other issues could adversely affect our operations.
We
face substantial competition in attracting and retaining qualified senior management and key personnel and may be unable to develop and
grow our business if we cannot attract and retain such senior management and key personnel.
As
an early stage company, our ability to develop and grow our business, to a large extent, depends upon our ability to attract, hire and
retain highly qualified and knowledgeable senior management and key personnel who possess the skills and experience necessary to satisfy
our business needs. Our ability to attract and retain such senior management and key personnel will depend on numerous factors, including
our ability to offer salaries, benefits and professional growth opportunities that are comparable with and competitive to those offered
by more established companies operating in our marketplace. We may be required to invest significant time and resources in attracting
and retaining additional senior management and key personnel as needed. Moreover, many of the companies with which we will compete for
any such individuals have greater financial and other resources, affording them the ability to undertake more extensive and aggressive
hiring campaigns, than we can. The normal running of our operations may be interrupted, and our financial condition and results of operations
negatively affected, as a result of any inability on our part to attract or retain the services of qualified and experienced senior management
and key personnel, or should our prospective key personnel refuse to serve, or, once appointed, leave prior to a suitable replacement
being found.
Risks
Associated with This Offering and Our Securities
This
Offering is being placed on a “best efforts” basis and we may not raise the entire $10,000,000.
The
offer and sale of the Restricted Shares pursuant hereto are expected to be made principally through the efforts of our officers, directors
and employees, on a “best efforts” basis, and, as such, there can be no assurance that this Offering will be fully subscribed
or even that a percentage of the Offering will be subscribed. There is no commitment, firm or otherwise, on the part of any one or more
parties to purchase any of the Units in this Offering and there is no minimum number of Units for which we must have subscriptions prior
to our closing on the Offering and we may, therefore, determine to have an initial closing on only a very small portion of the Offering.
As a result of this flexibility, we may not obtain all of the capital required to fund all of our proposed uses of proceeds as identified
in the “Estimated Use of Proceeds” section of this Memorandum, and we may, therefore, find ourselves without sufficient working
capital despite having closed on a portion of this Offering. Moreover, because there is no minimum number of Units for which we must
have subscriptions prior to our closing on the Offering, we may accept subscriptions that are, in the aggregate, insufficient in amount
to allow us to carry out our proposed business plan as set forth in this Memorandum, and, as a result, may cause prospective investors
to lose their entire investment.
Our
management will have substantial discretion over the use of proceeds of this Offering and may not apply them effectively.
Our
management will have significant flexibility in applying the net proceeds of this Offering and may apply the proceeds in ways with which
you do not agree. The failure of our management to apply these funds effectively could have a material adverse effect on our business
and our results of operations. The proposed allocation of the net proceeds of this Offering represents our management’s best estimate
of the expected use of funds to finance our activities in accordance with our management’s current objectives and overall market
conditions. See “Estimated Use of Proceeds”.
The
Units to be issued in this Offering, and the Securities of which they are comprised, will all constitute Restricted Securities.
Neither
the Units nor the Restricted Securities comprising the Units to be issued in this Offering have been registered under the Securities
Act or registered or qualified under any state securities or “Blue Sky” laws and are being sold pursuant to exemptions contained
in and under those laws. Accordingly, such Units and the Securities comprising the Units will constitute “restricted securities”
as defined in Rule 144 promulgated under the Securities Act, and must, therefore, be held indefinitely unless (i) first registered under
applicable federal and state securities laws, or (ii) an exemption from the registration requirements of those laws is available. Otherwise,
the Units and the Securities comprising the Units will not become freely transferable by reason of any change of circumstances whatsoever.
See “Description of Securities” and “Investment Restrictions”.
There
is currently no liquid trading market for our common stock and we cannot ensure that one will ever develop or be sustained.
The
trading market for our common stock is currently not liquid. We cannot predict how liquid the market for our common stock might become.
Our common stock is quoted in OTC Markets under the symbol TSOI.
Our
common stock may be deemed a “penny stock”, which would make it more difficult for investors to sell their shares.
Our
common stock is subject to the “penny stock” rules adopted under the Exchange Act. The penny stock rules apply to companies
whose common stock is not listed on the NASDAQ Stock Market or other national securities exchange and trades at less than $4.00 per share,
other than companies that have had average revenue of at least $6,000,000 for the last three years or that have tangible net worth of
at least $5,000,000 ($2,000,000 if the company has been operating for three or more years). These rules require, among other things,
that brokers who trade penny stock to persons other than “established customers” complete certain documentation, make suitability
inquiries of investors and provide investors with certain information concerning trading in the security, including a risk disclosure
document and quote information under certain circumstances. Many brokers have decided not to trade penny stocks because of the requirements
of the penny stock rules and, as a result, the number of broker-dealers willing to act as market makers in such securities is limited.
If we remain subject to the penny stock rules for any significant period, it could have an adverse effect on the market, if any, for
our securities. If our securities continue to be subject to the penny stock rules, investors will find it more difficult to dispose of
our securities.
Offers
or availability for sale of a substantial number of shares of our common stock may cause the price of our common stock to decline.
If
our stockholders have the right to sell substantial amounts of common stock in the public market, e.g. upon the expiration of any statutory
holding period under Rule 144, it could create a circumstance commonly referred to as an “overhang” and in anticipation of
which the market price of our common stock could fall. The existence of an overhang, whether or not sales have occurred or are occurring,
also could make our ability to raise additional financing through the sale of equity or equity-related securities in the future, at a
time and price that we deem reasonable or appropriate, more difficult.
The
elimination of monetary liability against our directors and officers under the Company’s Articles of Incorporation and Nevada law,
and the existence of indemnification rights to our directors, officers and employees, may result in substantial expenditures by the Company.
Article
6 of our Articles of Incorporation exculpates our directors and officers from certain monetary liabilities. Article 7 of our Articles
of Incorporation provides that we shall indemnify all directors (and all persons serving at our request as a director or officer of another
corporation) to the fullest extent permitted by Nevada law.
Further
pursuant to Article 7, the expenses of the indemnified person incurred in defending a civil suit or proceeding must be paid by us as
incurred and in advance of the final disposition of the action, suit, or proceeding under receipt of an undertaking by or on behalf of
the indemnified person to repay the amount if it is ultimately determined by a court of competent jurisdiction that he or she is not
entitled to be indemnified by us.
The
foregoing indemnification obligations could result in us incurring substantial expenditures, which we may be unable to recoup. These
provisions and resultant costs may also discourage us from bringing a lawsuit against directors and officers for breaches of their fiduciary
duties even though such actions, if successful, might otherwise benefit us and our stockholders.
Public
company compliance may make it more difficult to attract and retain officers and directors.
The
Sarbanes-Oxley Act and related rules implemented by the SEC have required changes in corporate governance practices of public companies.
As a public entity, these rules and regulations increase compliance costs and make certain activities more time consuming and costly.
As a public entity, these rules and regulations also make it more difficult and expensive for us to obtain director and officer liability
insurance and we may be required to accept reduced policy limits and coverage. As a result, it may be more difficult for us to attract
and retain qualified persons to serve as directors or as executive officers.
We
do not plan to pay any cash or stock dividends in the foreseeable future.
The
payment of dividends upon our capital stock is solely within the discretion of our future board of directors and is dependent upon our
financial condition, results of operations, capital requirements, restrictions contained in our future financing instruments and any
other factors our board of directors may deem relevant. We have never declared or paid any cash or stock dividends on our capital stock
and we currently anticipate that we will retain earnings, if any, to finance the development and expansion of our business and, as such,
do not intend on paying any cash or stock dividends in the foreseeable future.
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This
prospectus and the information incorporated by reference in this prospectus contain forward-looking statements within the meaning of
Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange
Act of 1934, as amended (the “Exchange Act”). These statements are therefore entitled to the protection of the safe harbor
provisions of these laws. These statements may be identified by the use of forward-looking terminology such as “anticipate,”
“believe,” “budget,” “contemplate,” “continue,” “could,” “envision,”
“estimate,” “expect,” “forecast,” “guidance,” “indicate,” “intend,”
“may,” “might,” “outlook,” “plan,” “possibly,” “potential,” “predict,”
“probably,” “pro-forma,” “project,” “seek,” “should,” “target,”
“will,” “would,” “will be,” “will continue” or the negative of or other variation on
these words or comparable terminology.
We
have based these forward-looking statements on our current expectations, assumptions, estimates and projections. While we believe these
expectations, assumptions, estimates and projections are reasonable, these forward-looking statements are only predictions and involve
a number of risks and uncertainties, many of which are beyond our control. These and other important factors may cause our actual results,
performance or achievements to differ materially from any future results, performance or achievements expressed or implied by these forward-looking
statements.
Management
cautions that the forward-looking statements contained in this prospectus and the information incorporated by reference are not guarantees
of future performance, and we cannot assume that such statements will be realized, or the forward-looking events and circumstances will
occur.
The
risks, uncertainties and assumptions that could cause actual results to differ materially from those anticipated or implied in our forward-looking
statements include, but are not limited to, those set forth in the “Risk Factors” section below.
Some
of the factors that could cause actual results to differ from our expectations are:
●
the state of the Company’s development;
●
the Company’s ability to continue as a going concern;
●
the Company’s ability to compete in an unproven market;
●
resistance by potential customers to new technologies;
●
performance issues with the Company’s products;
●
uncertainties related to estimates, assumptions and projections relating to unpaid losses and loss adjustment expenses and other accounting
policies;
●
reliance on key personnel;
●
introduction of competing products by other companies;
●
inflation and other changes in economic conditions, including changes in the financial markets;
●
security breaches and other system disruptions;
●
legislative and regulatory developments, especially in the gathering and use of information about private citizens;
●
weather conditions and natural disasters (including, but not limited to, the severity and frequency of storms, hurricanes, tornados and
hail); and
●
acts of war and terrorist activities, among other man-made disasters.
Given
these risks and uncertainties, you are cautioned not to place undue reliance on any forward-looking statements. The forward-looking statements
included or incorporated by reference into this prospectus and in the information incorporated by reference are made only as of the date
of this prospectus. Except as required by applicable law, including the securities laws of the United States and the rules and regulations
of the SEC, we do not undertake and specifically decline any obligation to update or revise any forward-looking statements in this prospectus
after we distribute this prospectus, or publicly announce the results of any revisions to any such statements to reflect future events
or developments, whether as a result of any new information, future events or otherwise.
MANAGEMENT
OF THE COMPANY
The
name of the officers and directors of the Company as of September 23, 2022, as well as certain information about them, are set forth
below:
| Name |
|
Position |
|
Age |
| Executive
Officers: |
|
|
|
|
| Timothy
G. Dixon |
|
President
and Chief Executive Officer |
|
64 |
| Famela
Ramos |
|
Vice
President |
|
45 |
| James
Veltmeyer, MD |
|
Chief
Medical Officer |
|
57 |
| Feng
Lin, MD, PhD |
|
Chief
Scientific Officer |
|
52 |
| Directors: |
|
|
|
|
| Thomas
E. Ichim, PhD |
|
Director |
|
45 |
| Timothy
G. Dixon |
|
Chairman |
|
|
Background
of Officers and Directors
Timothy
G. Dixon, CEO, President, and Chairman
Mr.
Dixon currently serves as Chief Executive Officer, President, and Chairman of Therapeutic Solutions International, Inc. Mr. Dixon previously
served as the President of TMD Courses, Inc. from 2006 to 2012 and as the President of Splint Decisions Inc. from 2010 to 2011.
Mr.
Dixon has attended hundreds of hours of continuing medical/dental education throughout the years and has produced many educational DVD’s
used by dental professionals worldwide on the subject of parafunctional control, migraine prevention, therapeutic Botox injections, migraine
pathophysiology, dental sleep medicine, and other therapeutic protocols. Mr. Dixon also has extensive experience in dealing with corporate
compliance matters with the U.S. Food and Drug Administration, (FDA) as well as many international regulatory bodies.
James
Veltmeyer, MD – Chief Medical Officer
Dr.
Veltmeyer is a board-certified family physician in La Mesa, California. A graduate of UC San Diego and the Ross University School of
Medicine, he completed his residency through the UC San Francisco system where he became Chief of Family Medicine Residency, overseeing
36 doctors.
Dr.
Veltmeyer, a member of the San Diego Critical Care Medical Group, has been elected for four years (2012, 2014, 2016, and 2017) by his
colleagues in the San Diego County Medical Society as a “Physician of Exceptional Excellence,” the most prestigious honor
awarded to a “Top Doctor” in San Diego County. He is among a select group of San Diego physicians who was chosen four of
the last fifteen years and he consistently ranks in the top 1% to 2% for patient satisfaction. He is currently the Chief of the Department
of Family Medicine at Sharp Grossmont Hospital where he provides senior leadership to over 200 doctors.
Feng
Lin, MD, Ph.D., – Chief Scientific Officer
Dr.
Lin has a stellar track record of drug development in the area of immunology and immuno-oncology having worked with the public company
Inovio Pharmaceuticals, where he developed technologies for gene delivery and therapeutic DNA vaccines against cancer and infectious
diseases in both R&D and clinical settings. Subsequently, Dr. Lin served as Director of Chinese Operations for MediStem Inc, which
was acquired by Intrexon in May 2014. It was the rapid clinical translation model developed by Dr. Lin at MediStem that resulted in the
company’s accelerated FDA clearance to begin clinical trials, which resulted in the sale of the company.
Dr.
Lin received his postdoctoral training at the Sanford-Burnham Medical Research Institute and his MD and Ph.D. at the Xiangya Medical
School of Central South University, China. He has authored over 20 peer-reviewed scientific publications, including several in top journals
such as Science, Cell, and Cancer Cell. He holds several patents.
Famela
Ramos – Vice President Business Development
Famela
Ramos is a Nurse, a Researcher, and a Politician. Famela was running for Congress in the 53nd Congressional District. Ms. Ramos came
to the United States from the Philippines at the age of two, when her father joined the United States Navy. Her parents worked tirelessly
to support the family of 5 children, all of which became successful entrepreneurs and Government Employees. As a nurse, Famela has experience
from the beginning of life, having practiced in pediatric nursing, to the end of life, having worked as a hospice nurse. Her excellence
in nursing and research is attested by 7 peer reviewed publications that she collaborated with Academy and Industry in advancing cutting
edge research in immunology and regenerative medicine.
The
first paper, was a collaboration with the Moores Cancer Center and several biotechnology companies, describing the state of the art in
cancer immunotherapy, and proposing future directions. The second paper discussed the possibility of stimulating regeneration of injured
lung stem cells using specific types of laser and light based interventions, this was a collaboration between the University of Utah
and the University of California, San Diego. The third paper, a collaboration between a nutraceutical company and Indiana University,
demonstrated the beneficial effects of a nutritional supplement on circulating stem cells in healthy volunteers. The fourth publication
was the first successful use of two different types of stem cells in a patient with heart failure, which resulted in a profound improvement.
The fifth publication is a report of 114 patients that were treated with umbilical cord blood stem cells and demonstrated safety and
signals of efficacy in collaboration with a Chinese Biotech company. The sixth publication was successful treatment of a spinal cord
injury patient with stem cells. The seventh publication was the basis for an investigational new drug (IND) application to the FDA, describing
use of fat stem cells to treat aplastic anemia.
Ms.
Ramos has established the Right to Try Foundation, which assists companies in utilizing this new law that allows for accelerated patient
access to experimental medication. Through this Foundation Ms. Ramos facilitated the first utilization of a cancer vaccine in the United
States and has been assisting both public and private companies. Most recently the Foundation has collaborated on filing new patents
for means of implementing the Right to Try Law. Ms. Ramos is a board member of Silent Voices, a Pregnancy Resource Center that provides
counselling to woman in emergency pregnancies, alternatives to abortion, and for woman that do choose abortion, post abortion support.
Ms. Ramos has been endorsed by business and community leaders as well as nationally known athletes including Dr. Peter Farrell, founder
of Resmed, a $18 billion company, and Wes Chandler, an NFL Hall of Fame San Diego Charger.
Thomas
E. Ichim, Ph.D., Director
Dr.
Ichim was appointed to the Board of Directors on January 22, 2016. Dr. Ichim also served as Chief Executive Officer of Emvolio, Inc.
Dr. Ichim is a seasoned biotechnology entrepreneur with a track record of scientific excellence. He has founded/co-founded several companies
including Batu Biologics, Inc., Medvax Pharma Corp, ToleroTech, Inc, bioRASI, and OncoMune LLC. To date he has published 121 peer-reviewed
articles and is co-editor of the textbooks “RNA Interference: From Bench to Clinical Translation” and “Immuno-Oncology
Text Book.”
Dr.
Ichim is an ad-hoc editor and sits on several editorial boards. Dr. Ichim is inventor on over 135 patents and patent applications. Dr.
Ichim has extensive experience with stem cell therapy and cellular product development through FDA regulatory pathways. Dr. Ichim spent
over 7 years as the President and Chief Scientific Officer of Medistem, developing and commercializing a novel stem cell, the Endometrial
Regenerative Cell, through drug discovery, optimization, preclinical testing, IND filing, and up through Phase II clinical trials with
the FDA. Dr. Ichim has extensive experience in product development, regulatory filings, and business development.
Dr.
Ichim has a BSc in Biology from the University of Waterloo, Waterloo, Ontario, Canada, a MSc in Microbiology and Immunology a University
of Western Ontario, London, Ontario, Canada and a Ph.D. in Immunology from the University of Sciences Arts and Technology, Olveston Monserrat.
Scientific
Advisory Board
Dr.
Santosh Kesari is a board-certified neurologist and neuro-oncologist and is currently Chair, Department of Translational Neuro-Oncology
and Neurotherapeutics, John Wayne Cancer Institute.
He
is also Director of Neuro-Oncology, Providence Saint John’s Health Center and Member, Los Angeles Biomedical Research Institute.
Dr. Kesari is ranked among the top 1% of neuro-oncologists and neurologists in the nation, according to Castle Connolly Medical Ltd and
an internationally recognized scientist and clinician. He is a winner of an Innovation Award by the San Diego Business Journal. He is
on the advisory board of American Brain Tumor Association, San Diego Brain Tumor Foundation, Chris Elliott Fund, Nicolas Conor Institute,
Voices Against Brain Cancer, and Philippine Brain Tumor Alliance. He has been the author of over 250 scientific publications, reviews,
or books. He is the inventor on several patents and patent applications, and founder and advisor to many cancer and neurosciences biotech
startups.
Dr.
Kesari has had a long-standing interest in cancer stem cells and studies their role in the formation of brain tumors and resistance to
treatment. He believes that in order to cure patients with brain tumors we first need to gain a better molecular and biological understanding
of the disease. A physician/scientist, Kesari harnesses his experience in surgery, chemotherapy, immunotherapy, radiation therapy and
novel devices to help develop Precision Therapeutic Strategies that will advance medicine to a new stage in the battle against brain
tumors and eradicate the disease.
Dr.
Francesco Marincola joined Kite in 2021 as Global Head of Cell Therapy Research. Before joining Kite, Francesco was President and
Chief Scientific Officer at Refuge Biotechnologies where responsible for the development and implementation of research and clinical
development strategies for adoptive cell therapy products and lead therapeutic programs based on nuclease deactivated CRISPR circuits.
He is also a National Institutes of Health (NIH) tenured senior investigator in cancer immunotherapy and biomarker research, and spent
23 years at the NIH, including 15 years as the Chief of the Infectious Disease and Immunogenetics Section at the NIH Clinical Center.
Previously, he also served as a distinguished research fellow in immune oncology discovery at AbbVie and as Chief Research Officer at
Sidra Research in Doha, Qatar.
The
former President of the Society for Immunotherapy of Cancer (SITC; 2013-2014), Francesco currently serves as Editor-in-Chief for several
prominent peer-reviewed publications, including Journal of Translational Medicine, Translational Medicine Communications and Immunotherapy,
and is the author of more than 600 peer-reviewed publications. He has edited several books including the SITC-affiliated Cancer Immunotherapy
Principles and Practice Textbook.
The
former President of the Society for Immunotherapy of Cancer (SITC; 2013-2014), Francesco currently serves as Editor-in-Chief for several
prominent peer-reviewed publications, including Journal of Translational Medicine, Translational Medicine Communications and Immunotherapy,
and is the author of more than 600 peer-reviewed publications. He has edited several books including the SITC-affiliated Cancer Immunotherapy
Principles and Practice Textbook.
Dr.
Donald Banerji is a Clinical development professional with 33 years of global clinical research and development experience (Phase
I-IV) in the pharmaceutical industry. Recently retired from Novartis as Global Clinical Development Head of Respiratory and Allergy Medicine.
Recognized by peers and external scientific community as an expert in pulmonary and allergy drug development bringing several iconic
brands to market with millions of patients benefitting from treatment through improving care and outcomes for patients with respiratory
diseases. Managed multidisciplinary teams in the filing of several new drug applications. Responsibilities included strategic and tactical
planning, regulatory interactions with global health authorities, appropriate resource and budgetary management and timely execution,
approval of high-quality large drug development programs and delivery of groundbreaking data. These global programs over a span of 3
decades resulted in the approval and competitive labeling of 14 innovative medicines, including 3 inhaled steroids for asthma, a triple
combination of 2 bronchodilator and an inhaled steroid for asthma, 3 non-steroidal inhaled controller drugs for asthma, 3 intranasal
steroids for allergic rhinitis and 4 bronchodilator drugs for COPD.
Signature
achievements at Novartis included first to market with the development and approval of the first to market inhaled dual combination medicine
in COPD (Ultibro) and the first to market triple combination medicine in asthma (Enerzair). With reimagining medicine as a core driver,
these treatments changed the practice of medicine and were incorporated in global treatment guidelines for COPD and asthma. Recipient
of numerous corporate awards including the highest scientific award of Distinguished Scientist 2016 for pioneering work in COPD. Published
over 400 primary manuscripts and abstracts, including the landmark study FLAME in NEJM.
Dr.
Boris Reznik is the Chairman of Venvalo Group, a venture value optimization firm. During his career, Dr. Reznik founded and built
technology companies into market leaders and successfully dealt with both Fortune 500 and emerging companies as clients and partners.
He has been a lead or co-investor in startups and mid-market firms and has participated in M&A transactions ranging from Millions
to Billions. Dr. Reznik has a depth of experience in processes and systems and a unique perspective in the drug and device development
world. As the Chairman of BioRASI, a global CRO, he led development of novel therapeutics, from filing INDs to getting approved NDAs,
for companies ranging from startups to Big Pharma.
Dr.
Boris Minev is a highly accomplished physician-scientist with extensive industrial and academic experience in Immuno-Oncology, oncolytic
viruses and stem cell biology and applications. He has a significant track record in tumor immunology and cancer vaccine development,
having worked closely on the development of the first cancer vaccine to be approved by a regulatory body (Melacine).
Dr.
Minev has also extensive expertise in immunotherapy clinical trial designs, logistics, and regulatory issues. He has a considerable supervision
& management experience in industrial and academic settings and has excellent skills in biotech business development, communication,
and collaboration.
Previously
he held a position as the Director of Immunotherapy and Translational Oncology at Genelux Corporation, where he was directing several
preclinical and translational projects on oncolytic virotherapy, immunotherapy, and nanotechnology.
Dr.
Minev is also an adjunct professor at the Moores UCSD Cancer Center. There, he served previously as Principal Investigator and Director,
Laboratory of Tumor Immunology and Immunotherapy where, for more than 15 years, his research has been focused on the discovery of new
target antigens for immunotherapy of cancer and the development of optimized cancer vaccines. Prior to that, Dr. Minev worked in Dr.
Steven Rosenberg’s Tumor Immunology Section at the Surgery Branch of the National Cancer Institute.
Dr.
Minev is an Advisory Board Member of the European Society for Translational Medicine (EUSTM). He is a member of the Scientific and Clinical
Advisory Boards of several biotechnology companies and has been an advisor for Amgen, Johnson & Johnson, Geron Corporation, McKinsey
Consulting and Thomson Current Drugs, among others. He is the recipient of the European Association of Cancer Research Fellowship and
the Fogarty International Fellowship.
Dr.
Pablo Guzman is a cardiologist in Fort Lauderdale, Florida where he is on staff at Holy Cross Hospital. He received his medical degree
from University of Puerto Rico School of Medicine and his Cardiology Fellowship at The Johns Hopkins Hospital where he then spent the
first part of his career continuing his basic science and clinical research along with his clinical duties. His CV includes over 25 papers
published in peer-reviewed journals and more than 15 abstracts.
He
is a Fellow of the American College of Cardiology and practiced for more than 30 years. Dr. Guzman is well experienced in basic and clinical
research, having participated in many clinical trials. He is also the acting Chief Medical Officer of Variant Pharmaceuticals, a Specialty
Pharma company developing treatments for kidney diseases.
Dr.
Juergen Winkler is presently practicing at Quantum Functional Medicine in Carlsbad, CA, which he founded in July of 2012. In 2005
he was the co-founder of Genesis Health Systems (Integrative Cancer and Medical Treatment Center) located in Oceanside, CA. He has been
a featured speaker for: the NSCC Women’s Health Seminar, Annual IPT/IPTLD Integrative Cancer Care Conference (Multiple years),
Health Freedom Expo 2011 & 2012, the Japanese Society of Oxidative Medicine in Osaka Japan, ACOSPM 2010 & 2011 conferences, NSCC
Health and Wellness Series 2013, and various other events. He is the physician author of Chapter 5 in the Defeat Cancer book and has
been a featured physician in the Townsend Letter.
Dr.
Nassir Azimi is a cardiologist in La Mesa, California and attended Dartmouth Medical School and completed his residency at the University
of Colorado. He finished his four year fellowship in Cardiovascular and Peripheral Interventions at Yale University in New Haven. Dr.
Azimi has been in private practice for over 13 years establishing a thriving clinical practice for cardiac patients as well as treating
patients for peripheral vascular disease. He is active in Interventional Cardiology and Peripheral Interventions. Dr. Azimi is the director
of La Mesa Cardiac Center’s Nuclear Cardiology Laboratory. He is also an investigator in multiple clinical research studies for
various cardiac and peripheral diseases.
He
has been recognized as San Diego’s Top Interventional Cardiologists by San Diego Magazine 2013,2014,2016, 2017 and also by Castle
Connoly for 2013, 2014, 2015,2016, 2017, and 2018. He is a former chief of biomedical ethics (6 years), former chief of Medicine and
former chief of Endovascular Medicine as well as Vice Chief of Cardiology at SGH. He is on the board of directors of the California ACC
where he serves as chair of the public relations committee. He is on Editorial Review Board for multiple medical journals. He is a national
speaker on various topics in cardiology and internal medicine.
Dr.
James Veltmeyer is a board-certified family physician in La Mesa, California. A graduate of UC San Diego and the Ross University
School of Medicine, he completed his residency through the UC San Francisco system where he became Chief of Family Medicine Residency,
overseeing 36 doctors.
Dr.
Veltmeyer, a member of the San Diego Critical Care Medical Group, has been elected for four years (2012, 2014, 2016, and 2017) by his
colleagues in the San Diego County Medical Society as a “Physician of Exceptional Excellence,” the most prestigious honor
awarded to a “Top Doctor” in San Diego County. He is among a select group of San Diego physicians who was chosen four of
the last fifteen years and he consistently ranks in the top 1% to 2% for patient satisfaction. He is currently the Chief of the Department
of Family Medicine at Sharp Grossmont Hospital where he provides senior leadership to over 200 doctors.
Dr.
Barry Glassman, DMD, DAAPM, DAACP, FICCMO, Diplomate ABDSM, FADI, is a Diplomate of the American Academy of Craniofacial Pain and
the American Academy of Pain Management, as well as a Fellow of the International College of Craniomandibular Orthopedics and the Academy
of Dentistry International, he is also on staff at the Lehigh Valley Hospital where he serves as a resident instructor of Craniofacial
Pain and Dysfunction and Dental Sleep Medicine.
Dr.
Glassman is a Diplomate of the Academy of Dental Sleep Medicine. He is on the staff at the Sacred Heart Hospital Sleep Disorder Center,
as well as serving as the Chief Dental Consultant to three other sleep centers in the Lehigh Valley. A popular and dynamic speaker, Dr.
Glassman lectures internationally, as well as throughout the United States. In addition to his extensive schedule which includes guest
lecture appearances and in-depth courses on joint dysfunction, chronic pain, headache, sleep disorders, and migraine headache, Dr. Glassman
is a frequent speaker at major chronic pain and joint dysfunction professional conferences.
University
of Pittsburgh: Bachelor of Science 1969, Pittsburgh, Pennsylvania University of Pittsburgh School of Dental Medicine; D.M.D. 1973, Pittsburgh
Pennsylvania Post Graduate Hours in Craniomandibular Dysfunction and Sleep Disorders: Over 2500
J.
Christopher Mizer founded Vivaris in June of 1998. Vivaris (formerly Lake Erie Capital) invests in and acquires middle-market businesses
in a broad range of industries that are leaders in their market niches. Mr. Mizer serves as the chairman of each of the portfolio companies
and guides key strategic decisions and their execution. He also serves as the operating president on an interim basis when companies
are going through periods of ownership succession and new management team members are being assembled.
Mr.
Mizer is a former Vice President and Officer of the investment banking division of Key Capital Markets, where he focused on merger, acquisition,
and financing projects for Fortune 500 clients, private companies, and successful entrepreneurs. Prior to joining Key Corp., he was Consultant
in the Capital Markets practice with Ernst & Young.
He
began his career as a Research Assistant with The Center for Economic Issues, a think-tank focused on economic development. He earned
the B.S. (biology, applied math), B.A. (economics), M.S. degrees (biology – neurogenetics), and MBA (finance, accounting) degrees
from Case Western Reserve University.
Christopher
has taught business strategy, finance and entrepreneurship at the graduate level at Case Western Reserve University, John Carroll University,
and the University of California, San Diego and at the undergraduate at San Diego State University.
Howard
Leonhardt is an inventor and serial entrepreneur. He has 21 U.S. patents with over 100 patent claims for products for treating cardiovascular
disease and has over 40 new patent claims pending. His TALENT (Taheri-Leonhardt) stent graft developed in the early 1990′s holds
a leading world market share for repairing aortic aneurysms without surgery.
His
inventions have treated over 500,000 patients in 60 countries. In early 1999 Leonhardt founded Bioheart, Inc. www.bioheartinc.com a leader
in applying adult muscle stem cells to treat heart failure.. Leonhardt holds a Diploma in International Trade from Anoka Technical College.
He attended the University of Minnesota,Anoka Ramsey College and UCLA Extension. He holds an honorary Doctorate in Biomedical Engineering
from the University of Northern California and is an honorary alumnus of the University of Florida and Florida International University.
He is co-leader of Startup California and Founder and Chairman of The California Stock Exchange TM (Cal-X) preparing to be the first
social good impact stock exchange currently operating the Cal-X 30 Social Good Impact fund powered by Motif Investing- www.calstockexchange.com
– He founded Cal-X Crowdfund Connect www.calxcrowdfund.com a crowdfunding campaign management co. and Cal-X Stars Business Accelerator,
Inc.www.calxstars.com a business incubator and accelerator focused on cardiovascular life sciences and social good impact innovations.
There
are 30 regenerative medtech and regenerative economy startups in the current portfolio class. His Leonhardt Ventures angels network has
raised and put to work over $145 million in 32 companies to date, most of them founded by Leonhardt. BioLeonhardt www.bioleonhardt.com
is developing the first implantable programmable and re-fillable stem cell pump. He leads CerebraCell for brain regeneration. EyeCell
for eye regeneration and AortaCell for aorta regeneration and number of other organ regeneration spin offs from his patented core technologies.
. Leonhardt serves as state spokesperson in California for the JOBS ACT and Crowdfunding for Startup California and has given over 40
speeches on the subject. He has operated Leonhardt’s Launchpads NorCal at the University of Northern California Science & Technology
Innovation Center in Rohnert Park, CA since 2008 and recently opened Leonhardt’s Launchpads Utah in Salt Lake City just off the
campus of the University of Utah. He has served on the Board of Directors of the University of Northern California, a private biomedical
engineering school, since 1999.
WHERE
YOU CAN FIND MORE INFORMATION
We
are subject to the informational requirements of the Exchange Act and, accordingly, file periodic reports, proxy statements and other
information with the SEC. You can obtain these reports, proxy statements and other information that we file electronically with the SEC
on the SEC’s website at www.sec.gov. Our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K
and amendments to these reports that are filed or furnished pursuant to Section 13 of the Exchange Act are available on our website at
www.vikingenergygroup.com, as soon as reasonably practicable after they are electronically filed with the SEC. The information on our
website is not part of this prospectus, except to the extent filed with the SEC and specifically incorporated into this prospectus by
reference.
This
prospectus is part of a registration statement that we filed with the SEC under the Securities Act. This prospectus does not contain
all of the information presented in the registration statement and its exhibits in accordance with SEC rules. Our descriptions in this
prospectus of the provisions of documents filed as exhibits to the registration statement or otherwise filed with the SEC are only summaries
of the terms of those documents and are not intended to be comprehensive. For a complete description of the content of the documents,
you should obtain copies of the full document.
INCORPORATION
OF CERTAIN DOCUMENTS BY REFERENCE
The
SEC allows us to incorporate by reference much of the information we file with the SEC, which means that we can disclose important information
to you by referring you to those publicly available documents. The information that we incorporate by reference in this prospectus is
considered to be part of this prospectus. Because we are incorporating by reference future filings with the SEC, this prospectus is continually
updated and those future filings may modify or supersede some of the information included or incorporated in this prospectus. This means
that you must look at all of the SEC filings that we incorporate by reference to determine if any of the statements in this prospectus
or in any document previously incorporated by reference have been modified or superseded.
We
incorporate by reference the following:
●
Our Annual Report on Form 10-K for the year ended December 31, 2021, filed on March 11, 2022;
●
Our Quarterly Report on Form 10-Q for the quarter ended March 31, 2022 filed with the SEC on May 23, 2022;
●
Our Quarterly Report on Form 10-Q for the quarter ended June 30, 2022, filed with the SEC on August 19, 2022
In
addition, TSOI further incorporate by reference into this prospectus all documents (other than current reports furnished under Item 2.02
or Item 7.01 of Form 8-K and exhibits filed on that form which are related to those items) that are filed by us with the SEC pursuant
to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act (i) on or after the date of the initial filing of the registration statement
of which this prospectus forms a part and before the effectiveness of the registration statement and (ii) following the effectiveness
of the registration statement until the offering of the securities under the registration statement is terminated or completed. These
documents include annual, quarterly and other periodic reports, such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q,
and Current Reports on Form 8-K, as well as proxy statements.
You
may request, at no cost, a copy of the documents that are incorporated by reference into this prospectus but not delivered with the prospectus,
including exhibits that are specifically incorporated by reference into such documents, by writing or calling us at the following address
or telephone number:
Therapeutic
Solutions International, Inc.
Attention:
Investor Relations
4093
Oceanside Blvd. Suite B, Oceanside CA
Ph:
(760) 295-7208
USE
OF PROCEEDS
The
Company will retain broad discretion over the use of the net proceeds from the sale of the securities. We currently intend to use the
net proceeds for working capital, capital expenditures, extinguishment of debt or other liabilities, and general corporate purposes.
We may also use a portion of the net proceeds to invest in or acquire businesses or technologies that we believe are complementary to
our own, although we have no current commitments or binding agreements with respect to any acquisitions as of the date of this prospectus.
DESCRIPTION
OF SECURITIES WE MAY OFFER
General
This
prospectus describes the general terms of our capital stock. The following description is not complete and may not contain all the information
you should consider before investing in our capital stock. For a more detailed description of these securities, you should read the applicable
provisions of Nevada law and our amended and restated certificate of incorporation, as amended, referred to herein as our certificate
of incorporation, and our amended and restated bylaws, referred to herein as our bylaws. When we offer to sell a particular series of
these securities, we will describe the specific terms of the series in a supplement to this prospectus. Accordingly, for a description
of the terms of any series of securities, you must refer to both the prospectus supplement relating to that series and the description
of the securities described in this prospectus. To the extent the information contained in the prospectus supplement differs from this
summary description, you should rely on the information in the prospectus supplement.
The
total number of shares of capital stock we are authorized to issue is 3,505,000,000 shares, of which (a) 3, 500,000,000 are common stock,
par value $0.001 per share, and (b) 5,000,000 are preferred stock, par value $0.001 per share.
Although
anticipated to be common stock, TSOI, directly or through agents, dealers or underwriters designated from time to time, may offer, issue
and sell, together or separately, up to $10,000,000 in the aggregate of:
●
common stock;
●
preferred stock;
●
warrants to purchase our securities; or
●
units comprised of, or other combinations of, the foregoing securities.
The
preferred stock may also be exchangeable for and/or convertible into shares of common stock, another series of preferred stock or other
securities that may be sold by us pursuant to this prospectus or any combination of the foregoing. When a particular series of securities
is offered, a supplement to this prospectus will be delivered with this prospectus, which will set forth the terms of the offering and
sale of the offered securities.
Common
Stock
As
of August 31, 2022, there were 2,583,073,458 shares of common stock issued and outstanding, held of record by approximately 212 stockholders.
Subject to preferential rights with respect to any outstanding preferred stock, all outstanding shares of common stock are of the same
class and have equal rights and attributes.
Subject
to the rights of the preferred stock, holders of common stock are entitled to receive such dividends as are declared by our board of
directors out of funds legally available for the payment of dividends. We presently intend to retain any earnings to fund the development
of our business.
Accordingly,
we do not anticipate paying any dividends on our common stock for the foreseeable future. Any future determination as to declaration
and payment of dividends will be made at the discretion of our board of directors.
In
the event of the liquidation, dissolution, or winding up of the Company, each outstanding share of our common stock will be entitled
to share equally in any of our assets remaining after payment of or provision for our debts and other liabilities.
Holders
of common stock are entitled to one vote per share on matters to be voted upon by stockholders. There is no cumulative voting for the
election of directors, which means that the holders of shares entitled to exercise more than fifty percent (50%) of the voting rights
in the election of directors are able to elect all of the directors.
Holders
of common stock have no preemptive rights to subscribe for or to purchase any additional shares of common stock or other obligations
convertible into shares of common stock which we may issue after the date of this prospectus.
All
of the outstanding shares of common stock are fully paid and non-assessable. Holders of our common stock are not liable for further calls
or assessments.
The
rights, preferences and privileges of the holders of common stock are subject to, and may be adversely affected by, the rights of the
holders of shares of any series of preferred stock that we may designate in the future.
Transfer
Agent and Registrar
The
transfer agent and registrar for our common stock is New Horizon Transfer, Inc., located at 202-515 West Pender Street, Vancouver, BC
V6B 6H5, (604) 876-5526.
Listing
Our
common stock is not listed on a national securities exchange but is quoted for trading on the quoted on the OTC Pink Sheets operated
by OTC Markets Group, Inc., at the OTCPK tier under the symbol “TSOI.” We have not applied to list our common stock on any
other exchange or quotation system.
Limitations
on Directors’ Liability
Our
articles of incorporation and bylaws contain provisions indemnifying our directors and officers to the fullest extent permitted by Nevada
law. Section 78.7502 of the Nevada Revised Statutes provides in part that a corporation shall have the power to indemnify any person
who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding (other than
an action by or in the right of the corporation) by reason of the fact that such person is or was a director, officer, employee or agent
of another corporation or other enterprise, against expenses (including attorneys’ fees), judgments, fines and amounts paid in
settlement actually and reasonably incurred by him in connection with such action, suit or proceeding if he acted in good faith and in
a manner he reasonably believed to be in or not opposed to the best interests of the corporation, and with respect to any criminal action
or proceeding, had no reasonable cause to believe her conduct was unlawful.
The
effect of these provisions is to restrict our rights and the rights of our stockholders in derivative suits to recover monetary damages
against a director for breach of the director’s fiduciary duty as a director, except that a director will be personally liable
for:
●
any breach of his or her duty of loyalty to us or our stockholders;
●
acts or omissions not in good faith which involve intentional misconduct or a knowing violation of law;
●
the payment of dividends or the redemption or purchase of stock in violation of state or federal law; or
●
any transaction from which the director derived an improper personal benefit.
This
provision does not affect a director’s liability under the federal securities laws.
To
the extent that our directors, officers and controlling persons are indemnified under the provisions contained in our articles of incorporation,
bylaws or Nevada law against liabilities arising under the Securities Act, we have been advised that in the opinion of the SEC, such
indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
Provisions
of our Certificate of Incorporation, Bylaws and Nevada Law that May Have an Anti-Takeover Effect
Certain
provisions set forth in our articles of incorporation and bylaws, as well as Nevada statutes could have the effect of discouraging potential
acquisition proposals or making a tender offer or delaying or preventing a change in control, including changes a stockholder might consider
favorable. Such provisions may also prevent or frustrate attempts by our stockholders to replace or remove our management.
Articles
of Incorporation and Bylaws
In
particular, articles certificate of incorporation and bylaws, among other things:
●
authorize our board of directors to designate and issue, without further action by the stockholders, up to 5,000,000 shares of undesignated
preferred stock;
●
provide that vacancies on the board of directors may be filled by a majority of directors in office, although less than a quorum, or
by the sole remaining director; and,
●
provide the board of directors with the ability to alter the bylaws without stockholder approval.
Nevada
Law
Nevada
law contains provisions that govern an “acquisition of controlling interest” in a Nevada corporation. The control share provisions
generally provide that any person or entity that acquires 20% or more of the outstanding voting shares of a publicly held Nevada corporation
in the secondary public or private market may be denied voting rights with respect to the acquired shares, unless a majority of the disinterested
shareholders of the corporation elects to restore those voting rights in whole or in part. However, the Company’s securities are
not subject to these control share provisions because the Company’s articles of incorporation, as permitted by Nevada law, specifically
exempt the Company from the control share provisions.
In
addition, Nevada law contains a provision that prevents an “interested stockholder” and a resident domestic Nevada corporation
from entering into a business “combination,” unless certain conditions are met. Nevertheless, the Company’s articles
of incorporation, as permitted by Nevada law, specifically exempt the Company from these “interested stockholder” provisions.
Preferred
Stock
Our
articles of incorporation, and its amendments, empowers our board of directors, without action by our shareholders, to designate and
issue up to 5,000,000 shares of preferred stock from time to time in one or more series, which preferred stock may be offered by this
prospectus and supplements thereto.
Nevada
law provides that the holders of preferred stock will have the right to vote separately as a class on any proposal involving fundamental
changes in the rights of holders of that preferred stock. This right is in addition to any voting rights provided for in the applicable
certificate of designation.
We
will fix the rights, preferences, privileges and restrictions of the preferred stock of each series in a certificate of designation relating
to that series filed with the State of Nevada. We will file as an exhibit to the registration statement of which this prospectus is a
part or will incorporate by reference from a current report on Form 8-K that we file with the SEC, the form of any certificate of designation
that describes the terms of the series of preferred stock we are offering before the issuance of the related series of preferred stock.
This description will include any or all of the following, as required:
●
the title and stated value;
●
the number of shares we are offering;
●
the liquidation preference per share;
●
the purchase price;
●
the dividend rate, period and payment date and method of calculation for dividends;
●
whether dividends will be cumulative or non-cumulative and, if cumulative, the date from which dividends will accumulate;
●
any contractual limitations on our ability to declare, set aside or pay any dividends;
●
the procedures for any auction and remarketing, if any;
●
the provisions for a sinking fund, if any;
●
the provisions for redemption or repurchase, if applicable, and any restrictions on our ability to exercise those redemption and repurchase
rights;
●
any listing of the preferred stock on any securities exchange or market;
●
whether the preferred stock will be convertible into our common stock, and, if applicable, the conversion price, or how it will be calculated,
and the conversion period;
●
whether the preferred stock will be exchangeable into debt securities, and, if applicable, the exchange price, or how it will be calculated,
and the exchange period;
●
voting rights, if any, of the preferred stock;
●
preemptive rights, if any;
●
restrictions on transfer, sale or other assignment, if any;
●
whether interests in the preferred stock will be represented by depositary shares;
●
a discussion of any material or special United States federal income tax considerations applicable to the preferred stock;
●
the relative ranking and preferences of the preferred stock as to dividend rights and rights if we liquidate, dissolve or wind up our
affairs;
●
any limitations on issuance of any class or series of preferred stock ranking senior to or on a parity with the series of preferred stock
as to dividend rights and rights if we liquidate, dissolve or wind up our affairs; and
●
any other specific terms, preferences, rights or limitations of, or restrictions on, the preferred stock.
If
we issue shares of preferred stock under this prospectus, after receipt of payment therefor, the shares will be fully paid and non-assessable.
Our
board of directors may authorize the issuance of preferred stock with voting or conversion rights that could adversely affect the voting
power or other rights of the holders of our common stock. Preferred stock could be issued quickly with terms designed to delay or prevent
a change in control of our Company or make removal of management more difficult. Additionally, the issuance of preferred stock could
have the effect of decreasing the market price of our common stock.
Existing
Preferred Stock
The
Company currently has one classes or series of preferred stock designated: the Series A Preferred Stock. As of September 13, 2022, two
shares of Series A Preferred Stock are outstanding leaving 4,998,000 treasury preferred. No other shares of preferred stock are issued
or outstanding.
Series
A Preferred Stock
On
August 4, 2022, the Board of Directors designated “Series A Preferred Stock” and caused to be filed a Certificate of Designation
pursuant to NRS 78.1955 with the State of Nevada, and upon approval the Board has issued One (1) share of Series A Preferred Stock to
Thomas E. Ichim, and One (1) share of Series A Preferred Stock to Timothy G. Dixon. The Holder of the Series A Preferred Stock shall
be entitled to vote on all matters subject to a vote or written consent of the holders of the Corporation’s Common Stock, and on
all such matters, the share of Series A Preferred Stock shall be entitled to that number of votes equal to the number of votes that all
issued and outstanding shares of Common Stock and all other securities of the Corporation are entitled to, as of any such date of determination,
on a fully diluted basis, plus One Million (1,000,000) votes, it being the intention that the Holder(s) of the Series A Preferred Stock
shall have effective voting control of the Corporation, on a fully diluted basis. The Holder(s) of the Series A Preferred Stock shall
vote together with the holders of Common Stock as a single class.
On
August 9, 2022, TSOI issued 2 shares of preferred stock valued at 0.001 per share.
Warrants
We
may issue warrants to purchase our securities or other rights, including rights to receive payment in cash or securities based on the
value, rate or price of one or more specified commodities, currencies, securities or indices, or any combination of the foregoing. Warrants
may be issued independently or together with any other securities that may be sold by us pursuant to this prospectus or any combination
of the foregoing and may be attached to, or separate from, such securities. To the extent warrants that we issue are to be publicly-traded,
each series of such warrants will be issued under a separate warrant agreement to be entered into between us and a warrant agent.
We
will file as exhibits to the registration statement of which this prospectus is a part, or will incorporate by reference from a current
report on Form 8-K that we file with the SEC, forms of the warrant and warrant agreement, if any. The prospectus supplement relating
to any warrants that we may offer will contain the specific terms of the warrants and a description of the material provisions of the
applicable warrant agreement, if any. These terms may include the following:
●
the title of the warrants;
●
the price or prices at which the warrants will be issued;
●
the designation, amount and terms of the securities or other rights for which the warrants are exercisable;
●
the designation and terms of the other securities, if any, with which the warrants are to be issued and the number of warrants issued
with each other security;
●
the aggregate number of warrants;
●
any provisions for adjustment of the number or amount of securities receivable upon exercise of the warrants or the exercise price of
the warrants;
●
the price or prices at which the securities or other rights purchasable upon exercise of the warrants may be purchased;
●
if applicable, the date on and after which the warrants and the securities or other rights purchasable upon exercise of the warrants
will be separately transferable;
●
a discussion of any material U.S. federal income tax considerations applicable to the exercise of the warrants;
●
the date on which the right to exercise the warrants will commence, and the date on which the right will expire;
●
the maximum or minimum number of warrants that may be exercised at any time;
●
information with respect to book-entry procedures, if any; and
●
any other terms of the warrants, including terms, procedures and limitations relating to the exchange and exercise of the warrants.
Exercise
of Warrants. Each warrant will entitle the holder of warrants to purchase the amount of securities or other rights, at the exercise
price stated or determinable in the prospectus supplement for the warrants. Warrants may be exercised at any time up to the close of
business on the expiration date shown in the applicable prospectus supplement, unless otherwise specified in such prospectus supplement.
After the close of business on the expiration date, if applicable, unexercised warrants will become void. Warrants may be exercised in
the manner described in the applicable prospectus supplement. When the warrant holder makes the payment and properly completes and signs
the warrant certificate at the corporate trust office of the warrant agent, if any, or any other office indicated in the prospectus supplement,
we will, as soon as possible, forward the securities or other rights that the warrant holder has purchased. If the warrant holder exercises
less than all of the warrants represented by the warrant certificate, we will issue a new warrant certificate for the remaining warrants.
Units
We
may issue units consisting of any combination of the other types of securities offered under this prospectus in one or more series. We
may evidence each series of units by unit certificates that we may issue under a separate agreement. We may enter into unit agreements
with a unit agent.
Each
unit agent, if any, may be a bank or trust company that we select. We will indicate the name and address of the unit agent, if any, in
the applicable prospectus supplement relating to a particular series of units. Specific unit agreements, if any, will contain additional
important terms and provisions. We will file as an exhibit to the registration statement of which this prospectus is a part, or will
incorporate by reference from a current report that we file with the SEC, the form of unit and the form of each unit agreement, if any,
relating to units offered under this prospectus.
If
we offer any units, certain terms of that series of units will be described in the applicable prospectus supplement, including, without
limitation, the following, as applicable:
●
the title of the series of units;
●
identification and description of the separate constituent securities comprising the units;
●
the price or prices at which the units will be issued;
●
the date, if any, on and after which the constituent securities comprising the units will be separately transferable;
●
a discussion of certain United States federal income tax considerations applicable to the units; and
●
any other material terms of the units and their constituent securities.
Forms
of Securities
Each
security may be represented either by a certificate issued in definitive form to a particular investor or by one or more global securities
representing the entire issuance of securities. Certificated securities in definitive form and global securities will be issued in registered
form. Definitive securities name you or your nominee as the owner of the security, and in order to transfer or exchange these securities
or to receive payments other than interest or other interim payments, you or your nominee must physically deliver the securities to the
trustee, registrar, paying agent or other agent, as applicable. Global securities name a depositary or its nominee as the owner of the
debt securities, warrants or units represented by these global securities. The depositary maintains a computerized system that will reflect
each investor’s beneficial ownership of the securities through an account maintained by the investor with its broker/dealer, bank,
trust company or other representative, as we explain more fully below.
Registered
Global Securities
We
may issue the securities in the form of one or more fully registered global securities that will be deposited with a depositary or its
nominee identified in the applicable prospectus supplement and registered in the name of that depositary or nominee. In those cases,
one or more registered global securities will be issued in a denomination or aggregate denominations equal to the portion of the aggregate
principal or face amount of the securities to be represented by registered global securities. Unless and until it is exchanged in whole
for securities in definitive registered form, a registered global security may not be transferred except as a whole by and among the
depositary for the registered global security, the nominees of the depositary or any successors of the depositary or those nominees.
The
specific terms of the depositary arrangement with respect to any securities to be represented by a registered global security will be
described in the prospectus supplement relating to those securities. We anticipate that the following provisions will apply to all depositary
arrangements.
Ownership
of beneficial interests in a registered global security will be limited to persons, called participants, that have accounts with the
depositary or persons that may hold interests through participants. Upon the issuance of a registered global security, the depositary
will credit, on its book entry registration and transfer system, the participants’ accounts with the respective principal or face
amounts of the securities beneficially owned by the participants. Any dealers, underwriters or agents participating in the distribution
of the securities will designate the accounts to be credited.
Ownership
of beneficial interests in a registered global security will be shown on, and the transfer of ownership interests will be effected only
through, records maintained by the depositary, with respect to interests of participants, and on the records of participants, with respect
to interests of persons holding through participants. The laws of some states may require that some purchasers of securities take physical
delivery of these securities in definitive form. These laws may impair your ability to own, transfer or pledge beneficial interests in
registered global securities.
So
long as the depositary, or its nominee, is the registered owner of a registered global security, that depositary or its nominee, as the
case may be, will be considered the sole owner or holder of the securities represented by the registered global security for all purposes
under the applicable indenture, warrant agreement or unit agreement.
Except
as described below, owners of beneficial interests in a registered global security will not be entitled to have the securities represented
by the registered global security registered in their names, will not receive or be entitled to receive physical delivery of the securities
in definitive form and will not be considered the owners or holders of the securities under the applicable indenture, warrant agreement
or unit agreement. Accordingly, each person owning a beneficial interest in a registered global security must rely on the procedures
of the depositary for that registered global security and, if that person is not a participant, on the procedures of the participant
through which the person owns its interest, to exercise any rights of a holder under the applicable indenture, warrant agreement or unit
agreement. We understand that under existing industry practices, if we request any action of holders or if an owner of a beneficial interest
in a registered global security desires to give or take any action that a holder is entitled to give or take under the applicable indenture,
warrant agreement or unit agreement, the depositary for the registered global security would authorize the participants holding the relevant
beneficial interests to give or take that action, and the participants would authorize beneficial owners owning through them to give
or take that action or would otherwise act upon the instructions of beneficial owners holding through them.
Payments
to holders with respect to securities represented by a registered global security registered in the name of a depositary or its nominee
will be made to the depositary or its nominee, as the case may be, as the registered owner of the registered global security. None of
the Company, the trustees, the warrant agents, the unit agents or any other agent of the Company, agent of the trustees, the warrant
agents or unit agents will have any responsibility or liability for any aspect of the records relating to payments made on account of
beneficial ownership interests in the registered global security or for maintaining, supervising or reviewing any records relating to
those beneficial ownership interests.
We
expect that the depositary for any of the securities represented by a registered global security, upon receipt of any payment of principal,
premium, interest or other payment or distribution to holders of that registered global security, will immediately credit participants’
accounts in amounts proportionate to their respective beneficial interests in that registered global security as shown on the records
of the depositary. We also expect that payments by participants to owners of beneficial interests in a registered global security held
through participants will be governed by standing customer instructions and customary practices, as is now the case with the securities
held for the accounts of customers or registered in “street name,” and will be the responsibility of those participants.
If
the depositary for any of these securities represented by a registered global security is at any time unwilling or unable to continue
as depositary or ceases to be a clearing agency registered under the Exchange Act and a successor depositary registered as a clearing
agency under the Exchange Act is not appointed by us within 90 days, we will issue securities in definitive form in exchange for the
registered global security that had been held by the depositary. Any securities issued in definitive form in exchange for a registered
global security will be registered in the name or names that the depositary gives to the relevant trustee, warrant agent, unit agent
or other relevant agent of ours or theirs. It is expected that the depositary’s instructions will be based upon directions received
by the depositary from participants with respect to ownership of beneficial interests in the registered global security that had been
held by the depositary.
PLAN
OF DISTRIBUTION
We
may sell securities directly to purchasers, through underwriters, dealers or agents, or through a combination of these methods of sale.
We may directly solicit offers to purchase securities, or agents may be designated to solicit such offers. In the prospectus supplement
relating to such offering, we will name any agent that could be viewed as an underwriter under the Securities Act and describe any commissions
that we must pay. Any such agent will be acting on a best-efforts basis for the period of its appointment or, if indicated in the related
prospectus supplement, on a firm commitment basis.
Each
prospectus supplement will describe the method of distribution of the securities and any applicable restrictions. The securities may
be distributed from time to time in one or more transactions at a fixed price, at a price that may be changed from time to time, at the
market price prevailing at the time of sale, at prices related to the prevailing market prices, or at negotiated prices.
The
prospectus supplement with respect to the securities of a particular series will describe the terms of the offering of the securities,
including the public offering price and the proceeds we will receive from the sale; the name of any selling agent or underwriters; any
discounts or commissions to be allowed or re-allowed or paid to any agent, underwriter, or dealers; any discounts or commissions to be
allowed or re-allowed or paid to any agent or underwriters; and all other items constituting underwriting or selling compensation.
If
the Company uses any underwriters or agents are utilized in the sale of the securities in respect of which this prospectus is delivered,
they will acquire the securities for their own account and may resell the securities from time to time in one or more transactions at
a fixed public offering price or at varying prices determined at the time of sale. The obligations of the underwriters to purchase the
securities will be subject to the conditions set forth in the applicable underwriting agreement. We will set forth in the prospectus
supplement relating to such offering the names of the underwriters or agents and the terms of the related agreement with them.
We
may sell securities directly or through dealers or agents we designate from time to time. We will name any dealer or agent involved in
the offering and sale of securities, and we will describe any commissions we will pay the dealer or agent in the prospectus supplement.
Unless the prospectus supplement states otherwise, our agent will act on a best-efforts basis for the period of its appointment.
We
may authorize agents or underwriters to solicit offers by certain types of institutional investors to purchase securities from us at
the public offering price set forth in the prospectus supplement pursuant to delayed delivery contracts providing for payment and delivery
on a specified date in the future. We will describe the conditions to these contracts and the commissions we must pay for solicitation
of these contracts in the prospectus supplement.
We
may provide agents and underwriters with indemnification against civil liabilities related to this offering, including liabilities under
the Securities Act, or contribution with respect to payments that the agents or underwriters may make with respect to these liabilities.
Agents and underwriters may engage in transactions with, or perform services for, us in the ordinary course of business.
We
will disclose in the related prospectus supplement for an offering if any persons participating in the offering, in order to facilitate
the offering of the offered securities, may engage in transactions that stabilize, maintain or otherwise affect the price of the securities.
We
may engage in at the market offerings into an existing trading market in accordance with Rule 415(a)(4) under the Securities Act. In
addition, we may enter into derivative transactions with third parties, or sell securities not covered by this prospectus to third parties
in privately negotiated transactions. If the related prospectus supplement so indicates, in connection with those derivatives, the third
parties may sell securities covered by this prospectus and the related prospectus supplement, including in short sale transactions. If
so, the third party may use securities pledged by us or borrowed from us or others to settle those sales or to close out any related
open borrowings of stock and may use securities received from us in settlement of those derivatives to close out any related open borrowings
of stock. The third party in these sale transactions will be an underwriter and will be named in the related prospectus supplement (or
a post-effective amendment). In addition, we may otherwise lend or pledge securities to a financial institution or other third party
that in turn may sell the securities short using this prospectus and a related prospectus supplement. The financial institution or other
third party may transfer its economic short position to investors in our securities or in connection with a concurrent offering of other
securities.
The
specific terms of any lock-up provisions in respect of any given offering will be described in the related prospectus supplement.
Without
limiting the generality of the foregoing, we may enter into a continuous offering program equity distribution agreement with a broker-dealer,
under which we may offer and sell shares of our common stock from time to time through a broker-dealer as our sales agent. If we enter
into such a program, sales of the shares of common stock, if any, will be made by means of ordinary brokers’ transactions on the
OTCPK or other market on which are shares may then trade at market prices, block transactions and such other transactions as agreed upon
by us and the broker-dealer. Under the terms of such a program, we also may sell shares of common stock to the broker-dealer, as principal
for its own account at a price agreed upon at the time of sale. If we sell shares of common stock to such broker-dealer as principal,
we will enter into a separate terms agreement with such broker-dealer, and we will describe this agreement in a separate prospectus supplement
or pricing supplement.
LEGAL
MATTERS
Certain
legal matters in connection with the offering and the validity of the securities offered by this prospectus will be passed upon by Hugh
D. Kelso III, Esq, Managing Attorney with H.D. Kelso & Associates, office located at 8799 Balboa Avenue, Suite 155 San Diego, CA
92123.
EXPERTS
The
consolidated financial statements of the Company as of December 31, 2021, and December 31, 2020, and for the two years then ended have
been incorporated by reference into this prospectus from the Company’s Annual Report on Form 10-K upon the report of Fruci &
Associates II, PLLC 802 N. Washington St. Spokane, WA 99201, an independent registered public accounting firm and PCAOB registered accounting
firm, and upon the authority of that firm as an expert in accounting and auditing. The report thereon contains an explanatory paragraph
which describes the conditions that raise substantial doubt about the ability of the Company to continue as a going concern and are contained
in Footnote 2 to the consolidated financial statements.
DISCLOSURE
OF COMMISSION POSITION ON INDEMNIFICATION
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers or persons controlling
the Company pursuant to the provisions of the Company’s charter documents or bylaws, the Company has been informed that in the
opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Act and is therefore
unenforceable.
[The
balance of page intentionally left blank]
$10,000,000
THERAPEUTIC
SOLUTIONS INTERNATIONAL, INC.
COMMON
STOCK
PREFERRED
STOCK
WARRANTS
UNITS
Prospectus
Dated
September 23, 2022
PART
II
INFORMATION
NOT REQUIRED IN PROSPECTUS
Item
14. Other Expenses of Issuance and Distribution
The
following is an estimate of the fees and expenses, other than underwriting discounts or commissions, payable by the Registrant in connection
with the issuance and distribution of the securities being registered. All the amounts shown are estimates except for the registration
fee. All of the expenses below will be paid by the Registrant.
| SEC Registration Fee | |
$ | 927.00 | |
| Transfer Agent’s Fees and Expenses | |
| $0.00 | (1) |
| Legal Fees and Expenses | |
| 10000 | |
| Accounting Fees and Expenses | |
| | (1) |
| Printing Fees and Expenses | |
| | (1) |
| Miscellaneous Fees and Expenses | |
| | (1) |
(1)
These fees are calculated based on the securities offered and the number of issuances and, accordingly, cannot be estimated at this time.
Item
15. Indemnification of Directors and Officers
The
Company’s Bylaws and Articles of Incorporation provide that we shall, to the full extent permitted by Nevada law, indemnify all
of our directors and officers. Section 78.7502 of the Nevada Revised Statutes provides in part that a corporation shall have the power
to indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit
or proceeding (other than an action by or in the right of the corporation) by reason of the fact that such person is or was a director,
officer, employee or agent of another corporation or other enterprise, against expenses (including attorneys’ fees), judgments,
fines and amounts paid in settlement actually and reasonably incurred by him in connection with such action, suit or proceeding if he
acted in good faith and in a manner he reasonably believed to be in or not opposed to the best interests of the corporation, and with
respect to any criminal action or proceeding, had no reasonable cause to believe her conduct was unlawful.
Similar
indemnity is authorized for such persons against expenses (including attorneys’ fees) actually and reasonably incurred in defense
or settlement of any threatened, pending or completed action or suit by or in the right of the corporation, if such person acted in good
faith and in a manner he reasonably believed to be in or not opposed to the best interests of the corporation, and provided further that
(unless a court of competent jurisdiction otherwise provides) such person shall not have been adjudged liable to the corporation. Any
such indemnification may be made only as authorized in each specific case upon a determination by the stockholders or disinterested directors
that indemnification is proper because the indemnity has met the applicable standard of conduct. Under our Bylaws and Articles of Incorporation,
the indemnity is presumed to be entitled to indemnification and we have the burden of proof to overcome that presumption. Where an officer
or a director is successful on the merits or otherwise in the defense of any action referred to above, we must indemnify him against
the expenses which such officer or director actually or reasonably incurred. Insofar as indemnification for liabilities arising under
the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing
provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification
is against public policy as expressed in the Act and is, therefore, unenforceable. In the event that a claim for indemnification against
such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person
of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person
in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled
by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public
policy as expressed in the Act and will be governed by the final adjudication of such issue.
Number/Description
of Exhibits
Item
17. Undertakings
(a)
The undersigned registrant hereby undertakes:
(1)
To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
(i)
to include any prospectus required by Section 10(a)(3) of the Securities Act of 1933;
(ii)
to reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective
amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration
statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities
offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range
may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume
and price represent no more than a 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration
Fee” table in the effective registration statement; and
(iii)
to include any material information with respect to the plan of distribution not previously disclosed in the registration statement or
any material change to such information in the registration statement; provided, however, that subparagraphs (1)(i), (1)(ii) and (1)(iii)
do not apply if the information required to be included in a post-effective amendment by those paragraphs is contained in reports filed
with or furnished to the Commission by the registrant pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 that are
incorporated by reference in the registration statement, or is contained in a form of prospectus filed pursuant to Rule 424(b) that is
part of the registration statement.
(2)
That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed
to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall
be deemed to be the initial bona fide offering thereof.
(3)
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the
termination of the offering.
(4)
That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser:
(i)
Each prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the
date the filed prospectus was deemed part of and included in the registration statement; and
(ii)
Each prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) as part of a registration statement in reliance on
Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii), or (x) for the purpose of providing the information required
by Section 10(a) of the Securities Act of 1933 shall be deemed to be part of and included in the registration statement as of the earlier
of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale of securities in the
offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date
an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities in the
registration statement to which that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial
bona fide offering thereof. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration
statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is
part of the registration statement will, as to a purchaser with a time of contract of sale prior to such effective date, supersede or
modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in
any such document immediately prior to such effective date; or
(5)
That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution
of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant
to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities
are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to
the purchaser and will be considered to offer or sell such securities to such purchaser:
(i)
Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule
424;
(ii)
Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by
the undersigned registrant;
(iii)
The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant
or its securities provided by or on behalf of the undersigned registrant; and
(iv)
Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
(b)
The undersigned registrant hereby undertakes that, for purposes of determining any liability of the registrant under the Securities Act
of 1933, each filing of the registrant’s annual report pursuant to Section 13(a) or Section 15(d) of the Securities Exchange Act
of 1934 (and, where applicable, each filing of an employee benefit plan’s annual report pursuant to section 15(d) of the Securities
Exchange Act of 1934) that is incorporated by reference in the registration statement shall be deemed to be a new registration statement
relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide
offering thereof.
(c)
The undersigned registrant hereby undertakes to supplement the prospectus, after the expiration of the subscription period, to set
forth the results of the subscription offer, the transactions by the underwriters during the subscription period, the amount of
unsubscribed securities to be purchased by the underwriters, and the terms of any subsequent reoffering thereof. If any
public offering by the underwriters is to be made on terms differing from those set forth on the cover page of the prospectus, a
post-effective amendment will be filed to set forth the terms of such offering.
(d)
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling
persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of
the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore,
unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses
incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding)
is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will,
unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction
the question whether such indemnification by it is against public policy as expressed in the Securities Act of 1933 and will be governed
by the final adjudication of such issue.
(e)
The registrant hereby undertakes that:
(1)
For purposes of determining any liability under the Securities Act, the information omitted from the form of prospectus filed as part
of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule
424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared
effective.
(2)
For the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus
shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at
that time shall be deemed to be the initial bona fide offering thereof.
Each
person whose signature appears below constitutes and appoints Hugh D. Kelso III, Esq., as his true and lawful attorney in fact and agent,
with full powers of substitution and re-substitution, for him and in his name, place and stead, in any and all capacities, to sign any
or all amendments (including post effective amendments) to the Registration Statement, and to sign any registration statement for the
same offering covered by this Registration Statement that is to be effective upon filing pursuant to Rule 462(b) under the Securities
Act of 1933, as amended, and all post effective amendments thereto, and to file the same, with all exhibits thereto, and all documents
in connection therewith, with the Securities and Exchange Commission, granting unto said attorney-in-fact and agent, each acting alone,
full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about the premises,
as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorney-in-fact
and agent, each acting alone, or his or her substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant
to the requirements of the Securities Act of 1933, this Registration Statement has been signed by the following persons in the capacities
indicated below as of September 23, 2022.
SIGNATURES
Pursuant
to the requirements of the Securities Act of 1933, the registrant certifies that it has reasonable grounds to believe that it meets all
of the requirements for filing on Form S-3 and has duly caused this registration statement to be signed on its behalf by the undersigned,
thereunto duly authorized, in the City of Oceanside, County of San Diego, United States, on September 26, 2022.
THERAPEUTIC
SOLUTIONS INTERNATIONAL, INC.
| By: |
/s/
Timothy G. Dixon |
|
| |
Timothy
G. Dixon, CEO and Director |
|
| |
|
|
Therapeutic Solutions (PK) (USOTC:TSOI)
Historical Stock Chart
From Mar 2024 to Apr 2024
Therapeutic Solutions (PK) (USOTC:TSOI)
Historical Stock Chart
From Apr 2023 to Apr 2024