October 30, 2023 -- InvestorsHub NewsWire --
via NetworkNewsWire
Editorial Coverage: The prestigious New England
Journal of Medicine recently published results of a large
study of 2,539 adults wherein weight loss of 49 to 52 pounds was a
common outcome. With more than 37 million Americans suffering from
diabetes, and with obesity at epidemic levels, it's little wonder
that millions of people are singing their praises of the new class
of drugs responsible for these outrageously positive outcomes.
GLP-1 agonists are a class of medications that mainly help manage
blood sugar (glucose) levels in people with type 2 diabetes but are
also helping treat obesity. Seems everyone is excited about
controlling blood sugar and easily losing weight. But there may be
an even better avenue to increased effectiveness and better
outcomes. Lexaria Bioscience Corp. (NASDAQ:
LEXX) (Profile), a global innovator in drug-delivery
platforms, began diabetes-related formal studies last year with its
DehydraTECH(TM) platform, which showed reduced blood-sugar levels
and lowered body weight. With such encouraging early results,
Lexaria is planning to launch new human and animal studies to
examine whether DehydraTECH processing can make GLP-1 drugs such as
semaglutide even better. Lexaria's heavily-patented DehydraTECH
drug delivery technology might improve bioavailability, lower
costs, and enhance tolerability, weight-loss potential and
management of diabetes and other health conditions. DehydraTECH may
become a game changer in the multibillion-dollar GLP-1 drug market
and could potentially lead to a collaboration with a dominant
player in the space, such as Novo
Nordisk (NYSE:
NVO), Pfizer Inc. (NYSE:
PFE), Merck & Company Inc.
(NYSE:
MRK), or AstraZeneca PLC (NASDAQ:
AZN).
- Lexaria's DehydraTECH is easily
integrated into oral product manufacturing processes to better
deliver API payloads to improve bioavailability, reduce side
effects and more
- Lexaria is evaluating the impact of
DehydraTECH on oral performance of GLP-1 drugs for diabetes and
weight loss, a market forecast to reach up to an astonishing $200
billion per year
- DehydraTECH is a scalable technology for
a multitude of applications that has separately been licensed by
Altria for oral nicotine products
- DehydraTECH promotes fast-acting, less
expensive and more effective oral drug delivery and has been
thoroughly evaluated through in vivo, in vitro and human clinical
testing
Click here to view the custom infographic of
the Lexaria Bioscience
Corp. editorial.
Diabetes and Obesity Rates Soaring
According to the U.S. Centers for Disease Control and
Prevention, 37.3 million Americans have diabetes, while another 95
million adults, one in three, have prediabetes, of which 80% are
unaware of having it. Globally, the World Health Organization
estimates 422 million people had diabetes in 2014, nearly quadruple
the number in 1980. Because diabetes is often closely connected to
obesity, it is a chronic and growing problem around much of the
world. The CDC says that obesity prevalence in the United States
was a whopping 41.9% in 2017. The WHO shows
that more than
1.9 billion people worldwide were overweight in 2016,
underscoring the spike in diabetes diagnoses.
Against this backdrop, it shouldn't be surprising that analysts
at Markets and
Research estimate the global diabetes drug market at $63.1
billion in 2021 on its way to $82.93 billion in 2027, while the
global market for diabetes devices was valued at another $26.1
billion and growing 7.5% annually. Due to the pain and
unpleasantness of injections, many diabetes sufferers prefer to
treat their condition with drugs rather than devices, if they have
the choice. Some of the most popular type 2 diabetes in recent
years are GLP-1 (glucagon-like peptide-1) receptor agonists. They
work by mimicking the effects of the naturally occurring hormone
GLP-1, which helps to regulate blood sugar levels and appetite.
Lexaria
Bioscience Corp. (NASDAQ:
LEXX) began its DehydraTECH diabetes-related formal
studies in 2022 with compelling data highly relevant to diabetes
showing positive effects of lowering glucose levels, body weight
and triglyceride levels, as well as evidenced increased locomotor
activity. With such positive early results, Lexaria is expanding
its diabetes study program, including using DehydraTECH for GLP-1
drugs alone and in combination with other molecules.
More precisely, the company is adding to a growing body of
evidence showing that DehydraTECH improves the way active
pharmaceutical ingredients ("APIs") enter the bloodstream through
oral delivery, the preferred method of drug delivery by patients.
The company has already evidenced the tech to improve the
pharmacokinetics of a variety of drugs, including PDE5 inhibitors
and antiviral drugs, as well as facilitate the transfer of drugs
across the blood-brain barrier, a notoriously challenging component
in drug development and potential key to treating addiction and
neurodegenerative brain disease.
Now Lexaria has its sights set on diabetes and obesity.
Lexaria will launch new human and animal studies to examine
DehydraTECH-processed GLP-1 drugs including but not limited to
semaglutide, which is sold under Novo Nordisk's brand names
Ozempic, Wegovy and Rybelsus. The studies will evaluate its
technology platform for improved bioavailability, cost
effectiveness, tolerability, weight-loss potential, management of
diabetes and more. Per the drug value formulation calculations
of Bay Bridge
Bio, Lexaria is at a launch point in the valuation curve with
the upcoming clinical studies.
DehydraTECH in Four Sentences
DehydraTECH technology is incorporated into the formulation and
manufacturing process of existing or new orally ingestible and
topical products. It involves mixing the API as a delivery payload
with certain fatty acids, infusing the mixture into a substrate
material, and then using controlled dehydration synthesis
processing to associate the payload and fatty acids together at a
molecular level. The newly combined molecules are then integrated
into end-product production across a range of dosage form factors
including pills and capsules.
The output is essentially tasteless and odorless and works
symbiotically with physiological systems to enable improved and
faster absorption of drugs into the bloodstream and brain tissues.
Brilliantly simple from the top level, Lexaria has turned a complex
process into a scalable platform applicable for a litany of both
current and future drugs.
GLP-1 Changing History
Historically, weight-loss drugs have been highly touted and
equally miserable failures (think Fen-phen, meridian, ephedra, to
name a few). Demand is understandable; hundreds of millions of
overweight people are hungry for something that can help them lose
weight and keep it off. To wit, analysts have repeatedly pegged
multibillion sales projections for drugs successfully penetrating
this market.
GLP-1 drugs are changing the course of weight-loss history, and
Lexaria intends to be a leader in the next-generation of these
drugs. The company
said in September that its planned trials of new
formulations of GLP-1 drugs "could enable drug delivery via oral
capsule at lower costs than current injectables, with reduced side
effects and enhanced health benefits." That would be a real game
changer in a burgeoning multibillion-dollar market.
In recent years, GLP-1 drugs realized success by targeting a
novel mechanism of action to activate hormones regulating glucose
while simultaneously decreasing appetite and stomach emptying.
Showing this can be done safely and effectively, several drugs have
earned approvals from the U.S. Food and Drug Administration for
type 2 diabetes and weight-loss management. New research strongly
suggests that GLP-1 can also curb
cravings for drinking, smoking and other addictive
behaviors, indications perfectly aligned for Lexaria's DehydraTECH
platform.
Making Good Even Better
Most approved GLP-1 drugs are injectables, which has started a
race for diabetes/weight-loss drugs in oral form including the
objective to minimize current adverse side effects such as nausea,
vomiting and diarrhea, among others. This is exactly where Lexaria
sees an enormous opportunity for DehydraTECH based upon results
with other types of drugs that lessened negative side effects.
Oral drugs are not only preferred for drug delivery by patients,
but they are also far less expensive than the $900 per month in
costs that is common for a regimen of a GLP-1 injectables. If
successful in developing an oral option, Lexaria could find itself
in a commanding position in a market for GLP-1 drugs that analysts
say could balloon to $150 billion
to $200 billion. That's a lot of headroom for a company with a
market capitalization currently under $15 million.
Patented. Literally.
Lexaria's unique drug-delivery technology is validated by a
wide-ranging patent portfolio. The technology is protected by 37
patents around the world covering antiviral drugs, specific
molecules for hypertension and central nervous system disorders,
nicotine, NSAIDs (non-steroidal anti-inflammatory drugs) and
vitamins. Also in development are patents pending that will further
galvanize DehydraTECH for hypertension, hormone treatments and
other drug formulations.
Lexaria has already caught the attention of tobacco juggernaut
Altria, which has licensed Lexaria's DehydraTECH and agreed to pay
royalties on any oral nicotine product sales. This alone could be a
company maker as DehydraTECH makes reduced risk noncombustible
nicotine possible, which could contribute to decreasing the more
than 7 million deaths attributed to smoking every year and help
Altria with its mission to move "beyond smoking."
This Ball Is Just Getting Rolling
The opportunity for Lexaria is undergirded by recent results and
the progress of majors actively in the diabetes and weight-loss
markets. In fact, it is arguable that the fervor for these types of
drugs is only in the early stages as drug makers parlay diabetes
drugs into weight-loss medications and likely much more,
effectively expanding markets and revenue while greatly slashing
development costs.
Novo
Nordisk (NYSE:
NVO) is a leader in diabetes and obesity
medicines. In June, Novo Nordisk said that it
is taking
actions to protect U.S. patients from the unlawful
marketing and sales of non-FDA approved counterfeit and compounded
semaglutide products claiming to contain semaglutide, while
reinforcing the responsible use of Novo Nordisk's FDA-approved
medicines. Novo Nordisk's prescription-only, FDA-approved
semaglutide medicines are Wegovy(R) for chronic weight management
along with Ozempic(R) and Rybelsus(R) for type 2 diabetes.
Pfizer Inc. (NYSE:
PFE) is leveraging its small molecule design
expertise, continuing advancement of the clinical program for
danuglipron (PF-06882961), subject to results from the ongoing
phase 2 trial. Danuglipron is the largest oral, small molecule
GLP-1-RA clinical development program underway with more than 1,400 participants
enrolled for the treatment of obesity and type 2 diabetes. The
company expects to finalize plans for the danuglipron late-stage
program by the end of 2023 and also is developing a once-daily
modified release version.
Merck & Company Inc.'s
(NYSE:
MRK) oral GLP-1
drug MK-6024 (efinopegdutide) activates glucagon receptors
to increase energy expenditure and reduce food intake. Recently,
the drug, which isn't specifically being developed as a weight-loss
substance, was granted Fast Track Designation from the FDA as a
potential treatment for patients with nonalcoholic steatohepatitis
("NASH"), a more severe form of nonalcoholic fatty liver disease
("NAFLD") that includes inflammation and damage to the liver.
MK-6024 is an investigational peptide and dual agonist at GLP-1 and
glucagon receptors.
AstraZeneca PLC (NASDAQ:
AZN) has successfully developed and
commercialized Farxiga (dapagliflozin) as a treatment for Type 2
diabetes, chronic kidney disease and heart failure. Farxiga is a
first-in-class, oral, once-daily SGLT2 inhibitor. Research has
shown Farxiga's efficacy in preventing and delaying cardiorenal
disease, while also protecting the organs — important findings
given the underlying links between the heart, kidneys and pancreas.
In May, Farxiga was
approved in the United States to reduce the risk of
cardiovascular death, hospitalization for heart failure and urgent
heart failure ("HF") visits in adults with HF.
The combined demographic for diabetes and weight loss might be
the largest in the world. Recent treatment successes have set the
market ablaze, creating enormous opportunity both now and well into
the future.
For more information about Lexaria Bioscience
Corp., please visit Lexaria
Bioscience Corp.
About NetworkNewsWire
NetworkNewsWire ("NNW") is a specialized
communications platform with a focus on financial news and content
distribution for private and public companies and the investment
community. It is one of 60+ brands
within the Dynamic Brand
Portfolio @ IBN that
delivers: (1) access to a vast network of
wire solutions via InvestorWire to efficiently and effectively reach
a myriad of target markets, demographics and diverse
industries; (2) article
and editorial
syndication to 5,000+ outlets; (3)
enhanced press
release enhancement to ensure maximum
impact; (4) social media
distribution via IBN to millions of social media
followers; and (5) a full array of
tailored corporate
communications solutions. With broad reach and a seasoned team
of contributing journalists and writers, NNW is uniquely positioned
to best serve private and public companies that want to reach a
wide audience of investors, influencers, consumers, journalists and
the general public. By cutting through the overload of information
in today's market, NNW brings its clients unparalleled recognition
and brand awareness. NNW is where breaking news, insightful content
and actionable information converge.
To receive SMS text alerts from NetworkNewsWire, text
"STOCKS" to 77948 (U.S. Mobile Phones Only)
For more information, please visit https://www.NetworkNewsWire.com
Please see full terms of use and disclaimers on the
NetworkNewsWire website applicable to all content provided by NNW,
wherever published or re-published: https://www.NetworkNewsWire.com/Disclaimer
NetworkNewsWire
New York, NY
www.NetworkNewsWire.com
212.418.1217 Office
Editor@NetworkNewsWire.com
NetworkNewsWire is powered by IBN
DISCLAIMER: NetworkNewsWire (NNW) is the source of the Article
and content set forth above. References to any issuer other than
the profiled issuer are intended solely to identify industry
participants and do not constitute an endorsement of any issuer and
do not constitute a comparison to the profiled issuer. The
commentary, views and opinions expressed in this release by NNW are
solely those of NNW. Readers of this Article and content agree that
they cannot and will not seek to hold liable NNW for any investment
decisions by their readers or subscribers. NNW is a news
dissemination and financial marketing solutions provider and are
NOT registered broker-dealers/analysts/investment advisers, hold no
investment licenses and may NOT sell, offer to sell or offer to buy
any security.
The Article and content related to the profiled company
represent the personal and subjective views of the Author, and are
subject to change at any time without notice. The information
provided in the Article and the content has been obtained from
sources which the Author believes to be reliable. However, the
Author has not independently verified or otherwise investigated all
such information. None of the Author, NNW, or any of their
respective affiliates, guarantee the accuracy or completeness of
any such information. This Article and content are not, and should
not be regarded as investment advice or as a recommendation
regarding any particular security or course of action; readers are
strongly urged to speak with their own investment advisor and
review all of the profiled issuer's filings made with the
Securities and Exchange Commission before making any investment
decisions and should understand the risks associated with an
investment in the profiled issuer's securities, including, but not
limited to, the complete loss of your investment.
NNW HOLDS NO SHARES OF ANY COMPANY NAMED IN THIS RELEASE.
This release contains "forward-looking statements" within the
meaning of Section 27A of the Securities Act of 1933, as amended,
and Section 21E the Securities Exchange Act of 1934, as amended and
such forward-looking statements are made pursuant to the safe
harbor provisions of the Private Securities Litigation Reform Act
of 1995. "Forward-looking statements" describe future expectations,
plans, results, or strategies and are generally preceded by words
such as "may", "future", "plan" or "planned", "will" or "should",
"expected," "anticipates", "draft", "eventually" or "projected".
You are cautioned that such statements are subject to a multitude
of risks and uncertainties that could cause future circumstances,
events, or results to differ materially from those projected in the
forward-looking statements, including the risks that actual results
may differ materially from those projected in the forward-looking
statements as a result of various factors, and other risks
identified in a company's annual report on Form 10-K or 10-KSB and
other filings made by such company with the Securities and Exchange
Commission. You should consider these factors in evaluating the
forward-looking statements included herein, and not place undue
reliance on such statements. The forward-looking statements in this
release are made as of the date hereof and NNW undertakes no
obligation to update such statements.
SOURCE: NetworkNewsWire
Editorial Coverage
Lexaria Bioscience (NASDAQ:LEXX)
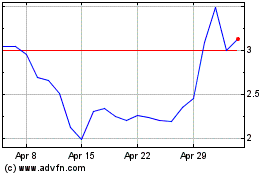
Historical Stock Chart
From Apr 2024 to May 2024
Lexaria Bioscience (NASDAQ:LEXX)
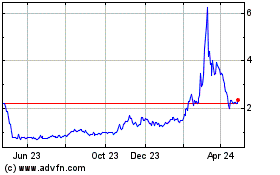
Historical Stock Chart
From May 2023 to May 2024