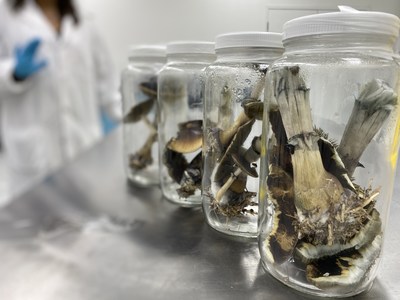
Numinus Bioscience's developments include
harvesting its first flush of Psilocybe mushrooms at its Health
Canada licensed facility.
VANCOUVER, BC, Oct. 22, 2020 /CNW/ - Numinus Wellness Inc.
("Numinus" or the "Company") (TSXV: NUMI), a company creating an
ecosystem of health solutions centred on developing and supporting
the safe, evidence-based, accessible use of psychedelic-assisted
psychotherapies, has harvested the first legal flush of
Psilocybe mushrooms in Canada by a public company under its Health
Canada-issued Controlled Drugs and Substances Dealer's Licence.
This news coincides with other recent developments by Numinus
Bioscience, the Company's 7,000 square foot analytics and research
laboratory, which is focused on handling various psychedelic
substances and developing analytical methods and formulations for
the evolving psychedelics space.
"The first harvest of legally grown
Psilocybe mushrooms is a significant milestone for the
whole industry" said Michael Tan,
Chief Operating Officer. "Now, we can progress with research and
development of standardized cultivation, extraction, and testing
methods and exploring product formulations to support safe,
evidence-based, accessible psychedelic-assisted
psychotherapy, as well as build a sequenced spore
library."
Dennis McKenna, renowned
ethnopharmacologist, Co-founder of the Heffter Research Institute,
Founder of the McKenna Academy of Natural Philosophy, and member of
Numinus' General Advisory Council, adds, "While there is a place
for synthetic psilocybin in clinical practice, well-characterized
and standardized extracts of natural forms of psilocybin i.e.,
mushrooms, cultivated and processed under stringent quality control
protocols, are the wave of the future as therapeutic psilocybin
gains acceptance. Given a choice, many people would prefer the
option to use natural psilocybin. Mushrooms have been used in
traditional medicine for literally thousands of years, and hence,
they are just that much closer to these ancient traditions. Natural
mushroom extracts are also likely to be far more affordable
compared to synthetic psilocybin, and that is an important
consideration when it comes to ensuring accessibility to this
medicine which can be so beneficial to many people. Cost should not
be a barrier to access these medicines, and the use of mushroom
extracts will help ensure those who need them most can benefit from
psilocybin therapies."
Numinus Bioscience is establishing itself at the forefront of
analytical testing for psychedelic substances, as a centre of
excellence for psychedelic product research and development, and as
an ancillary for psychedelic therapy clinical trials. Health
Canada licences, scientific
expertise, and state-of-the-art technologies facilitate ongoing
innovation, while revenue from high-throughput contract cannabis
testing is reinvested to further its psychedelic
initiatives.
In addition to today's announcement, Numinus includes in its
recent developments an amendment application to its Health
Canada-issued Controlled Drugs and Substances Licence. The current
licence permits testing, import and export, storage and
distribution of MDMA, Psilocybin, Psilocin, DMT, and Mescaline as
well as the undertaking of novel mycological research initiatives.
The amendment will additionally permit the sale of Psilocybe
mushroom fruiting bodies and extract for clinical research
protocols and the development of standardized testing for
additional psychedelic substances.
Numinus is also proud to announce the recent appointment of
Sharan Sidhu, regulatory expert and
policy advisor for restrictive high growth industries, as Science
Officer and General Manager. Sidhu brings extensive experience as a
hub between academia, industry, and governing directorates to align
research objectives with regulatory compliance. She has been a key
strategist for research roadmaps, core initiatives, the development
of IP, and the development of compliance requirements including
LIMS, GLP/GMP, GACP, and ISO Standards.
"Sharan's experience and leadership are already proving
instrumental in establishing Numinus Bioscience as a centre of
excellence and major industry player in the delivery of safe,
evidence-based, accessible psychedelic-psychotherapy," said
Payton Nyquvest, CEO, Founder, and
Chairman. "We are honoured to have her join us on our mission."
Sidhu is leading an accomplished scientific research team
including Dr Bernd O. Keller, PhD
(Advanced Analytical Methods), Dr Kristina
Grotzinger, PhD (Phytochemistry), and Judit Varga, MSc (Advanced Microbiology
Methods).
ON BEHALF OF THE BOARD OF NUMINUS WELLNESS INC
Payton Nyquvest
President, Chief Executive Officer and Chair
About Numinus
Numinus is a Vancouver-based health care company creating
an ecosystem of health solutions centred around developing and
supporting the safe, evidence-based, accessible use of
psychedelic-assisted therapies. Numinus' ecosystem units include
Numinus Bioscience, Numinus R&D, and Numinus Health. Each unit
is undertaking distinct, synergistic initiatives to hasten legal,
for-profit psychedelic-assisted therapy models and position Numinus
as a trusted industry leader for the eventual regulated rollout of
these therapies.
Numinus Bioscience includes a 7,000 square foot
research and testing laboratory as well as numerous Health Canada
licences (through its wholly-owned subsidiary Salvation Botanicals)
including a cannabis testing licence used for sustainable B2B
revenue to support the company's psychedelic efforts. The company
holds a Dealer's Licence which allows it to import, export,
possess, and test MDMA, psilocybin, psilocin, DMT and mescaline as
well as a licence to produce and extract psilocybin from mushrooms.
These licences allow Numinus to support the growing number of
studies on the potential benefits of psychedelic-assisted
psychotherapies through research projects, product development, and
the supply and distribution of these substances.
Numinus R&D is embarking on implementation
science and leveraging established relationships for partnerships
to host studies, develop medical and therapeutic protocols, and
influence regulatory approval.
Numinus Health is dedicated to delivering therapies that
enhance and supplement existing options for people wanting lasting
physical, mental and emotional health — centred around safe,
evidence-based accessible psychedelic-assisted psychotherapy.
Learn more at numinus.ca, and follow us on Facebook, Twitter,
and Instagram.
Forward Looking Statements
This news release
contains forward-looking statements within the meaning of
applicable securities laws. All statements that are not historical
facts, including without limitation, statements regarding future
estimates, plans, programs, forecasts, projections, objectives,
assumptions, expectations or beliefs of future performance, are
"forward-looking statements." Forward-looking statements can be
identified by the use of words such as "plans", "expects" or "does
not expect", "is expected", "estimates", "intends", "anticipates"
or "does not anticipate", or "believes", or variations of such
words and phrases or statements that certain actions, events or
results "may", "could", "would", "might" or "will" be taken, occur
or be achieved. Such forward-looking statements involve known and
unknown risks, uncertainties and other factors that may cause
actual results, events or developments to be materially different
from any future results, events or developments expressed or
implied by such forward looking statements. Such risks and
uncertainties include, among others, dependence on obtaining and
maintaining regulatory approvals, including acquiring and renewing
federal, provincial, municipal, local or other licences and any
inability to obtain all necessary governmental approvals licences
and permits to operate and expand the Company's facilities;
regulatory or political change such as changes in applicable laws
and regulations, including federal and provincial legalization, due
to inconsistent public opinion, perception of the medical-use and
adult-use marijuana industry, bureaucratic delays or inefficiencies
or any other reasons; any other factors or developments which may
hinder market growth; the Company's limited operating history and
lack of historical profits; reliance on management; the Company's
requirements for additional financing, and the effect of capital
market conditions and other factors on capital availability;
competition, including from more established or better financed
competitors; and the need to secure and maintain corporate
alliances and partnerships, including with research and development
institutions, customers and suppliers. These factors should be
considered carefully, and readers are cautioned not to place undue
reliance on such forward-looking statements. Although the Company
has attempted to identify important risk factors that could cause
actual actions, events or results to differ materially from those
described in forward-looking statements, there may be other risk
factors that cause actions, events or results to differ from those
anticipated, estimated or intended. There can be no assurance that
forward-looking statements will prove to be accurate, as actual
results and future events could differ materially from those
anticipated in forward-looking statements. The Company has no
obligation to update any forward-looking statement, even if new
information becomes available as a result of future events, new
information or for any other reason except as required by
law.
SOURCE Numinus Wellness Inc.