false--12-310001761918Q1http://fasb.org/us-gaap/2023#PrepaidExpenseAndOtherAssetsCurrenthttp://fasb.org/us-gaap/2023#PrepaidExpenseAndOtherAssetsCurrenthttp://fasb.org/us-gaap/2023#OperatingLeaseRightOfUseAsset7220001761918us-gaap:ResearchAndDevelopmentExpenseMember2023-01-012023-03-310001761918eras:TwoThousandAndTwentyFourPrivatePlacementMemberus-gaap:CommonStockMember2024-03-012024-03-310001761918eras:SeriesB1ConvertiblePreferredStockAndSeriesB2ConvertiblePreferredStockMembereras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2020-03-012020-03-310001761918eras:SeriesB2ConvertiblePreferredStockMembereras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2021-01-012021-01-310001761918eras:LicenseAgreementMembereras:NiKangTherapeuticsIncorporationMember2020-02-012020-02-290001761918us-gaap:EmployeeStockOptionMember2023-01-012023-12-310001761918us-gaap:CommercialPaperMember2024-03-310001761918eras:OptionsEarlyExercisedSubjectToFutureVestingMember2023-01-012023-03-310001761918eras:AsanaMergerAgreementMember2020-11-300001761918eras:LifeArcMembereras:LicenseAgreementMember2023-12-310001761918eras:TwoThousandEighteenEquityIncentivePlanMember2021-08-012021-12-310001761918eras:LaboratoryEquipmentMember2023-12-310001761918us-gaap:CashAndCashEquivalentsMemberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918eras:TwoThousandTwentyOneLeaseMember2024-03-310001761918us-gaap:FurnitureAndFixturesMember2024-03-310001761918eras:TwoThousandTwentyOneIncentiveAwardPlanMember2021-07-3100017619182023-12-310001761918us-gaap:AdditionalPaidInCapitalMember2022-12-310001761918eras:TwoThousandTwentyOneIncentiveAwardPlanMember2021-07-012021-07-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USGovernmentCorporationsAndAgenciesSecuritiesMemberus-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918eras:AffiniTTherapeuticsIncMember2024-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918eras:NovartisPharmaAgMembereras:LicenseAgreementMember2023-01-012023-03-310001761918eras:NovartisPharmaAgMembereras:LicenseAgreementMember2022-12-012022-12-310001761918us-gaap:GeneralAndAdministrativeExpenseMember2023-01-012023-03-310001761918srt:MaximumMembereras:UsTreasurySecuritiesShortTermMember2023-12-310001761918eras:EstimatedSharesPurchasableUnderTheEmployeeStockPurchasePlanMember2023-01-012023-03-310001761918srt:MaximumMembereras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2020-03-012020-03-310001761918us-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMembereras:LongTermInvestmentsMember2023-12-310001761918us-gaap:RetainedEarningsMember2022-12-310001761918us-gaap:CorporateDebtSecuritiesMember2023-12-310001761918eras:AwardsAvailableForFutureGrantMember2023-12-310001761918eras:LifeArcMembereras:LicenseAgreementMember2024-03-3100017619182023-01-012023-03-310001761918eras:ErascaFoundationMember2023-04-012023-04-300001761918eras:SharesAvailableForPurchaseUnderEmployeeStockPurchasePlanMember2023-12-3100017619182022-12-310001761918srt:MaximumMembereras:UsTreasurySecuritiesShortTermMember2024-03-310001761918eras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2023-01-012023-03-310001761918eras:LicenseAgreementMembereras:NiKangTherapeuticsIncorporationMember2020-04-012020-04-300001761918us-gaap:ShortTermInvestmentsMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:CommercialPaperMember2023-12-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:CommercialPaperMember2023-12-310001761918us-gaap:AccumulatedOtherComprehensiveIncomeMember2023-01-012023-03-310001761918us-gaap:CashAndCashEquivalentsMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:CommercialPaperMember2024-03-310001761918eras:TwoThousandTwentyFourLeaseMember2024-01-312024-01-310001761918eras:UsGovernmentAgenciesShortTermSecuritiesMembersrt:MaximumMember2023-12-310001761918us-gaap:EmployeeStockOptionMember2023-01-012023-03-310001761918eras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2023-12-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USTreasurySecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USGovernmentCorporationsAndAgenciesSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918eras:AsanaMergerAgreementMember2024-01-012024-03-310001761918eras:EmployeeStockPurchasePlanMembersrt:MinimumMember2021-07-012021-07-310001761918us-gaap:RetainedEarningsMember2024-03-310001761918eras:AsanaMergerAgreementMember2023-12-310001761918eras:EmployeeStockPurchasePlanMember2023-12-310001761918us-gaap:LetterOfCreditMembereras:TwoThousandTwentyLeaseMember2020-09-012020-09-300001761918us-gaap:EmployeeStockOptionMember2024-03-310001761918eras:SeriesB1ConvertiblePreferredStockAndSeriesB2ConvertiblePreferredStockMemberus-gaap:IPOMembereras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2020-03-310001761918us-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918eras:UsTreasurySecuritiesLongTermMember2023-12-310001761918us-gaap:USTreasurySecuritiesMember2024-03-310001761918eras:EmergeLifeSciencesPurchaseAgreementMemberus-gaap:CommonStockMember2021-03-012021-03-310001761918eras:AffiniTTherapeuticsIncMember2024-03-310001761918eras:UsTreasurySecuritiesShortTermMember2024-03-310001761918eras:EmployeeStockPurchasePlanMember2021-07-310001761918eras:AwardsAvailableForFutureGrantMember2024-03-310001761918eras:TwoThousandTwentyFourLeaseMember2024-01-310001761918us-gaap:EmployeeStockOptionMembereras:ForHoldersOfMoreThanTenPercentOfCombinedVotingPowerOfAllClassesOfStockMembersrt:MinimumMember2024-01-012024-03-310001761918eras:NiKangTherapeuticsIncorporationMembereras:LicenseAgreementMember2024-03-310001761918us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918us-gaap:CashAndCashEquivalentsMemberus-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:CommercialPaperMember2024-03-310001761918eras:TwoThousandTwentyLeaseMember2020-09-012020-09-300001761918srt:MaximumMembereras:LifeArcMembereras:LicenseAgreementMember2020-04-012020-04-300001761918eras:NiKangTherapeuticsIncorporationMembereras:LicenseAgreementMember2023-12-310001761918srt:MaximumMembereras:NovartisPharmaAgMembereras:LicenseAgreementMember2022-12-012022-12-310001761918eras:AsanaMergerAgreementMember2023-01-012023-03-310001761918us-gaap:CommercialPaperMember2023-12-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:CommercialPaperMember2024-03-310001761918eras:EmployeeStockPurchasePlanMember2021-07-012021-07-310001761918eras:AsanaMergerAgreementMember2024-03-310001761918us-gaap:AdditionalPaidInCapitalMember2023-03-3100017619182023-01-012023-12-310001761918us-gaap:LetterOfCreditMembereras:TwoThousandTwentyLeaseMember2020-09-300001761918eras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2022-03-012022-03-310001761918us-gaap:CommonStockMember2023-12-310001761918eras:CommercialpapershorttermMember2024-03-310001761918srt:MaximumMembereras:CorporatedebtsecuritiesshorttermMember2023-12-310001761918eras:TwoThousandTwentyOneLeaseMember2024-01-012024-03-310001761918us-gaap:USTreasurySecuritiesMemberus-gaap:FairValueMeasurementsRecurringMembereras:LongTermInvestmentsMember2023-12-310001761918us-gaap:EmployeeStockOptionMembersrt:MinimumMember2023-01-012023-03-310001761918us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918us-gaap:CommonStockMember2024-03-310001761918us-gaap:CommonStockMember2024-01-012024-03-310001761918eras:EmergeLifeSciencesPurchaseAgreementMember2024-01-012024-03-310001761918eras:UsGovernmentAgenciesShortTermSecuritiesMember2024-03-310001761918eras:TwoThousandAndTwentyFourPrivatePlacementMemberus-gaap:SubsequentEventMemberus-gaap:CommonStockMember2024-04-022024-04-020001761918eras:TwoThousandTwentyLeaseMember2020-09-300001761918eras:AffiniTTherapeuticsIncMember2024-01-012024-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USGovernmentCorporationsAndAgenciesSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918us-gaap:CorporateDebtSecuritiesMember2024-03-310001761918srt:MaximumMemberus-gaap:EmployeeStockOptionMember2024-01-012024-03-310001761918us-gaap:AccumulatedOtherComprehensiveIncomeMember2023-03-310001761918eras:UsTreasurySecuritiesShortTermMember2023-12-310001761918us-gaap:EmployeeStockOptionMember2024-01-012024-03-310001761918eras:CorporatedebtsecuritiesshorttermMember2024-03-310001761918us-gaap:CommonStockMember2023-01-012023-03-310001761918eras:EmployeeStockPurchasePlanMember2024-01-012024-03-310001761918us-gaap:AdditionalPaidInCapitalMember2023-01-012023-03-310001761918eras:OptionsEarlyExercisedSubjectToFutureVestingMember2024-01-012024-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918eras:DavidMChackoMember2024-03-310001761918us-gaap:ResearchAndDevelopmentExpenseMember2024-01-012024-03-310001761918eras:LaboratoryEquipmentMember2024-03-310001761918eras:ComputerEquipmentAndSoftwareMember2024-03-310001761918eras:TwoThousandTwentyOneLeaseMemberus-gaap:OtherNoncurrentAssetsMember2021-12-310001761918eras:KatmaiPharmaceuticalsIncorporationMembereras:SeriesB1ConvertiblePreferredStockMembereras:LicenseAgreementMember2020-04-012020-04-300001761918us-gaap:EmployeeStockOptionMembersrt:MinimumMember2024-01-012024-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:CommercialPaperMember2024-03-310001761918eras:UsGovernmentAgenciesShortTermSecuritiesMember2023-12-310001761918us-gaap:LeaseholdImprovementsMember2023-12-310001761918srt:MaximumMembereras:UsTreasurySecuritiesLongTermMember2023-12-310001761918eras:LicenseAgreementMembereras:NiKangTherapeuticsIncorporationMember2023-01-012023-03-310001761918us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918eras:SeriesB2ConvertiblePreferredStockMembereras:AsanaMergerAgreementMemberus-gaap:IPOMemberus-gaap:CommonStockMember2020-11-012020-11-300001761918eras:TwoThousandAndTwentyFourPrivatePlacementMemberus-gaap:SubsequentEventMemberus-gaap:CommonStockMember2024-04-020001761918eras:TwoThousandTwentyOneLeaseMember2021-12-310001761918eras:TwoThousandAndTwentyFourPrivatePlacementMemberus-gaap:CommonStockMember2024-03-310001761918eras:FirstLicensedProductMembereras:LicenseAgreementMembereras:NiKangTherapeuticsIncorporationMember2021-01-012021-01-310001761918us-gaap:AdditionalPaidInCapitalMember2023-12-310001761918eras:AffiniTTherapeuticsIncMember2023-12-310001761918eras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2024-01-012024-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918srt:MaximumMemberus-gaap:EmployeeStockOptionMember2023-01-012023-03-310001761918srt:MaximumMembereras:CommercialpapershorttermMember2024-03-310001761918us-gaap:CommonStockMember2023-03-310001761918eras:FirstLicensedProductMembersrt:MaximumMembereras:NiKangTherapeuticsIncorporationMembereras:LicenseAgreementMember2020-02-012020-02-290001761918eras:TwoThousandTwentyOneIncentiveAwardPlanMember2024-03-310001761918us-gaap:RetainedEarningsMember2024-01-012024-03-310001761918eras:NovartisPharmaAgMembereras:LicenseAgreementMember2024-03-310001761918eras:DavidMChackoMember2024-01-012024-03-310001761918eras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2024-03-310001761918eras:LifeArcMembereras:LicenseAgreementMember2024-01-012024-03-310001761918eras:OptionsToPurchaseCommonStockMember2023-01-012023-03-310001761918srt:MaximumMembereras:CorporatedebtsecuritiesshorttermMember2024-03-310001761918eras:LifeArcMembereras:LicenseAgreementMember2020-04-012020-04-300001761918us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918eras:SeriesB2ConvertiblePreferredStockMembereras:AsanaMergerAgreementMember2020-11-300001761918us-gaap:RetainedEarningsMember2023-12-310001761918eras:FirstAmendmentToTwoThousandAndTwentyLeaseMember2021-03-012021-03-310001761918eras:SeriesB2ConvertiblePreferredStockMembereras:AsanaMergerAgreementMember2020-11-012020-11-300001761918eras:NovartisPharmaAgMembereras:LicenseAgreementMember2023-12-310001761918eras:TwoThousandAndTwentyFourPrivatePlacementMember2024-03-310001761918eras:ComputerEquipmentAndSoftwareMember2023-12-310001761918eras:TwoThousandTwentyOneLeaseMember2021-12-012021-12-3100017619182024-01-012024-03-310001761918eras:AsanaMergerAgreementMember2020-11-012020-11-300001761918us-gaap:ShortTermInvestmentsMemberus-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918eras:EmergeLifeSciencesPurchaseAgreementMember2021-03-012021-03-310001761918us-gaap:AccountsPayableAndAccruedLiabilitiesMember2024-03-310001761918eras:FirstAmendmentToTwoThousandAndTwentyLeaseMember2021-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USGovernmentCorporationsAndAgenciesSecuritiesMemberus-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918eras:KatmaiPharmaceuticalsIncorporationMembereras:LicenseAgreementMember2020-03-012020-03-310001761918us-gaap:FurnitureAndFixturesMember2023-12-310001761918us-gaap:AccountsPayableAndAccruedLiabilitiesMember2023-12-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USTreasurySecuritiesMemberus-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918us-gaap:AccumulatedOtherComprehensiveIncomeMember2024-03-310001761918srt:MinimumMember2024-01-012024-03-310001761918eras:EmployeeStockPurchasePlanMember2023-01-012023-03-310001761918us-gaap:USTreasurySecuritiesMember2023-12-310001761918us-gaap:EmployeeStockOptionMembersrt:MinimumMembereras:AtTimeOptionIsGrantedMember2024-01-012024-03-310001761918eras:NovartisPharmaAgMembereras:LicenseAgreementMember2024-01-012024-03-310001761918us-gaap:LeaseholdImprovementsMember2024-03-310001761918us-gaap:AdditionalPaidInCapitalMember2024-03-310001761918eras:TwoThousandTwentyFourLeaseMember2024-01-012024-03-310001761918us-gaap:RetainedEarningsMember2023-03-310001761918us-gaap:CashAndCashEquivalentsMemberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueMeasurementsRecurringMember2024-03-3100017619182024-03-310001761918us-gaap:AdditionalPaidInCapitalMember2024-01-012024-03-310001761918eras:EmergeLifeSciencesPurchaseAgreementMember2023-01-012023-03-310001761918eras:SecondLicensedProductMembereras:NiKangTherapeuticsIncorporationMembereras:LicenseAgreementMember2020-02-012020-02-290001761918eras:CorporatedebtsecuritiesshorttermMember2023-12-310001761918us-gaap:AccumulatedOtherComprehensiveIncomeMember2023-12-310001761918us-gaap:AccumulatedOtherComprehensiveIncomeMember2022-12-310001761918eras:NiKangTherapeuticsIncorporationMembereras:LicenseAgreementMember2024-01-012024-03-310001761918us-gaap:ShortTermInvestmentsMemberus-gaap:USTreasurySecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918eras:EstimatedSharesPurchasableUnderTheEmployeeStockPurchasePlanMember2024-01-012024-03-3100017619182024-05-020001761918eras:CommercialpapershorttermMember2023-12-310001761918eras:SharesAvailableForPurchaseUnderEmployeeStockPurchasePlanMember2024-03-310001761918us-gaap:CashAndCashEquivalentsMemberus-gaap:FairValueInputsLevel1Memberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueMeasurementsRecurringMember2024-03-310001761918eras:EmployeeStockPurchasePlanMember2024-03-310001761918eras:FirstAmendmentToTwoThousandAndTwentyLeaseMember2022-04-012022-04-300001761918us-gaap:CommonStockMember2022-12-310001761918eras:TwoThousandAndTwentyFourPrivatePlacementMemberus-gaap:SubsequentEventMember2024-04-022024-04-020001761918us-gaap:FairValueMeasurementsRecurringMember2023-12-310001761918us-gaap:AccumulatedOtherComprehensiveIncomeMember2024-01-012024-03-310001761918eras:OptionsToPurchaseCommonStockMember2024-01-012024-03-310001761918eras:UsTreasurySecuritiesLongTermMembersrt:MinimumMember2023-12-310001761918srt:MaximumMembereras:CommercialpapershorttermMember2023-12-310001761918eras:LifeArcMembereras:LicenseAgreementMember2023-01-012023-03-310001761918us-gaap:GeneralAndAdministrativeExpenseMember2024-01-012024-03-3100017619182023-03-310001761918us-gaap:RetainedEarningsMember2023-01-012023-03-310001761918eras:EmergeLifeSciencesPurchaseAgreementMemberus-gaap:CommonStockMember2021-03-310001761918us-gaap:EmployeeStockOptionMember2023-12-310001761918eras:UsGovernmentAgenciesShortTermSecuritiesMembersrt:MaximumMember2024-03-310001761918us-gaap:CashAndCashEquivalentsMemberus-gaap:FairValueInputsLevel1Memberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueMeasurementsRecurringMember2023-12-31eras:Positionxbrli:pureutr:sqftxbrli:sharesiso4217:USD
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
FORM 10-Q
(Mark One)
|
|
☒ |
QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended March 31, 2024
OR
|
|
☐ |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from _____________ to ______________
Commission File Number: 001-40602
ERASCA, INC.
(Exact Name of Registrant as Specified in its Charter)
|
|
Delaware |
83-1217027 |
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer
Identification No.) |
3115 Merryfield Row, Suite 300 San Diego, CA |
92121 |
(Address of principal executive offices) |
(Zip Code) |
Registrant’s telephone number, including area code: (858) 465-6511
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class |
|
Trading Symbol(s) |
|
Name of each exchange on which registered |
Common Stock, $0.0001 par value per share |
|
ERAS |
|
Nasdaq Global Select Market |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☑ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☑ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
|
|
|
|
|
|
|
Large accelerated filer |
|
☐ |
|
Accelerated filer |
|
☐ |
|
|
|
|
Non-accelerated filer |
|
☑ |
|
Smaller reporting company |
|
☑ |
|
|
|
|
|
|
|
Emerging growth company |
|
☑ |
|
|
|
|
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☑
As of May 2, 2024, the registrant had 173,358,966 shares of common stock outstanding.
Table of Contents
PART I—FINANCIAL INFORMATION
Item 1. Financial Statements.
Erasca, Inc.
Condensed Consolidated Balance Sheets
(In thousands, except share and par value amounts)
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Assets |
|
|
|
|
|
|
Current assets: |
|
|
|
Cash and cash equivalents |
|
$ |
76,726 |
|
|
$ |
93,075 |
|
Short-term marketable securities |
|
|
220,959 |
|
|
|
219,275 |
|
Prepaid expenses and other current assets |
|
|
8,938 |
|
|
|
8,326 |
|
Total current assets |
|
|
306,623 |
|
|
|
320,676 |
|
Long-term marketable securities |
|
|
— |
|
|
|
9,642 |
|
Property and equipment, net |
|
|
21,358 |
|
|
|
22,327 |
|
Operating lease assets |
|
|
37,177 |
|
|
|
37,861 |
|
Restricted cash |
|
|
408 |
|
|
|
408 |
|
Other assets |
|
|
4,456 |
|
|
|
4,383 |
|
Total assets |
|
$ |
370,022 |
|
|
$ |
395,297 |
|
Liabilities and Stockholders' Equity |
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
Accounts payable |
|
$ |
2,633 |
|
|
$ |
2,000 |
|
Accrued expenses and other current liabilities |
|
|
16,577 |
|
|
|
20,186 |
|
Proceeds from prepayment of private placement |
|
|
6,907 |
|
|
|
— |
|
Operating lease liabilities |
|
|
4,126 |
|
|
|
3,970 |
|
Total current liabilities |
|
|
30,243 |
|
|
|
26,156 |
|
Operating lease liabilities, net of current portion |
|
|
50,811 |
|
|
|
51,889 |
|
Other liabilities |
|
|
559 |
|
|
|
566 |
|
Total liabilities |
|
|
81,613 |
|
|
|
78,611 |
|
Commitments and contingencies (Note 11) |
|
|
|
|
|
|
Stockholders' equity: |
|
|
|
|
|
|
Preferred stock, $0.0001 par value; 80,000,000 shares authorized at March 31, 2024 and December 31, 2023; no shares issued and outstanding at March 31, 2024 and December 31, 2023 |
|
|
— |
|
|
|
— |
|
Common stock, $0.0001 par value; 800,000,000 shares authorized at March 31, 2024 and December 31, 2023; 151,497,138 and 151,462,103 shares issued at March 31, 2024 and December 31, 2023, respectively; 151,249,219 and 151,090,227 shares outstanding at March 31, 2024 and December 31, 2023, respectively |
|
|
15 |
|
|
|
15 |
|
Additional paid-in capital |
|
|
929,634 |
|
|
|
922,607 |
|
Accumulated other comprehensive (loss) income |
|
|
(210 |
) |
|
|
77 |
|
Accumulated deficit |
|
|
(641,030 |
) |
|
|
(606,013 |
) |
Total stockholders' equity |
|
|
288,409 |
|
|
|
316,686 |
|
Total liabilities and stockholders' equity |
|
$ |
370,022 |
|
|
$ |
395,297 |
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Erasca, Inc.
Condensed Consolidated Statements of Operations and Comprehensive Loss
(In thousands, except share and per share amounts)
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
2024 |
|
|
2023 |
|
Operating expenses: |
|
|
|
|
Research and development |
|
$ |
28,574 |
|
|
$ |
27,585 |
|
General and administrative |
|
|
10,277 |
|
|
|
9,440 |
|
Total operating expenses |
|
|
38,851 |
|
|
|
37,025 |
|
Loss from operations |
|
|
(38,851 |
) |
|
|
(37,025 |
) |
Other income (expense) |
|
|
|
|
|
|
Interest income |
|
|
3,900 |
|
|
|
3,877 |
|
Other expense, net |
|
|
(66 |
) |
|
|
(51 |
) |
Total other income (expense), net |
|
|
3,834 |
|
|
|
3,826 |
|
Net loss |
|
$ |
(35,017 |
) |
|
$ |
(33,199 |
) |
Net loss per share, basic and diluted |
|
$ |
(0.23 |
) |
|
$ |
(0.22 |
) |
Weighted-average shares of common stock used in computing net loss per share, basic and diluted |
|
|
151,161,741 |
|
|
|
149,504,216 |
|
Other comprehensive income (loss): |
|
|
|
|
|
|
Unrealized (loss) gain on marketable securities, net |
|
|
(287 |
) |
|
|
527 |
|
Comprehensive loss |
|
$ |
(35,304 |
) |
|
$ |
(32,672 |
) |
The accompanying notes are an integral part of these condensed consolidated financial statements.
Erasca, Inc.
Condensed Consolidated Statements of Stockholders’ Equity
(In thousands, except share data)
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
Other |
|
|
|
|
|
Total |
|
|
|
Common Stock |
|
|
Paid-in |
|
|
Comprehensive |
|
|
Accumulated |
|
|
Stockholders' |
|
|
|
Shares |
|
|
Amount |
|
|
Capital |
|
|
(Loss) Income |
|
|
Deficit |
|
|
Equity |
|
Balance at December 31, 2023 |
|
|
151,462,103 |
|
|
$ |
15 |
|
|
$ |
922,607 |
|
|
$ |
77 |
|
|
$ |
(606,013 |
) |
|
$ |
316,686 |
|
Exercise of stock options |
|
|
35,035 |
|
|
|
— |
|
|
|
24 |
|
|
|
— |
|
|
|
— |
|
|
|
24 |
|
Vesting of early exercised stock options |
|
|
— |
|
|
|
— |
|
|
|
155 |
|
|
|
— |
|
|
|
— |
|
|
|
155 |
|
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
6,848 |
|
|
|
— |
|
|
|
— |
|
|
|
6,848 |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(35,017 |
) |
|
|
(35,017 |
) |
Unrealized loss on marketable securities, net |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(287 |
) |
|
|
— |
|
|
|
(287 |
) |
Balance at March 31, 2024 |
|
|
151,497,138 |
|
|
$ |
15 |
|
|
$ |
929,634 |
|
|
$ |
(210 |
) |
|
$ |
(641,030 |
) |
|
$ |
288,409 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
Other |
|
|
|
|
|
Total |
|
|
|
Common Stock |
|
|
Paid-in |
|
|
Comprehensive |
|
|
Accumulated |
|
|
Stockholders' |
|
|
|
Shares |
|
|
Amount |
|
|
Capital |
|
|
(Loss) Income |
|
|
Deficit |
|
|
Equity |
|
Balance at December 31, 2022 |
|
|
150,448,363 |
|
|
$ |
15 |
|
|
$ |
893,850 |
|
|
$ |
(1,041 |
) |
|
$ |
(480,971 |
) |
|
$ |
411,853 |
|
Exercise of stock options |
|
|
199,344 |
|
|
|
— |
|
|
|
183 |
|
|
|
— |
|
|
|
— |
|
|
|
183 |
|
Vesting of early exercised stock options |
|
|
— |
|
|
|
— |
|
|
|
243 |
|
|
|
— |
|
|
|
— |
|
|
|
243 |
|
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
6,845 |
|
|
|
— |
|
|
|
— |
|
|
|
6,845 |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(33,199 |
) |
|
|
(33,199 |
) |
Unrealized gain on marketable securities, net |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
527 |
|
|
|
— |
|
|
|
527 |
|
Balance at March 31, 2023 |
|
|
150,647,707 |
|
|
$ |
15 |
|
|
$ |
901,121 |
|
|
$ |
(514 |
) |
|
$ |
(514,170 |
) |
|
$ |
386,452 |
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Erasca, Inc.
Condensed Consolidated Statements of Cash Flows
(In thousands)
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
2024 |
|
|
2023 |
|
Cash flows from operating activities: |
|
|
|
|
|
|
Net loss |
|
$ |
(35,017 |
) |
|
$ |
(33,199 |
) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
Depreciation and amortization |
|
|
998 |
|
|
|
894 |
|
Stock-based compensation expense |
|
|
6,848 |
|
|
|
6,845 |
|
Accretion on marketable securities, net |
|
|
(2,342 |
) |
|
|
(975 |
) |
Changes in operating assets and liabilities: |
|
|
|
|
|
|
Prepaid expenses and other current and long-term assets |
|
|
(507 |
) |
|
|
670 |
|
Accounts payable |
|
|
633 |
|
|
|
446 |
|
Accrued expenses and other current and long-term liabilities |
|
|
(3,626 |
) |
|
|
(4,363 |
) |
Operating lease assets and liabilities, net |
|
|
(238 |
) |
|
|
3,324 |
|
Net cash used in operating activities |
|
|
(33,251 |
) |
|
|
(26,358 |
) |
|
|
|
|
|
|
|
Cash flows from investing activities: |
|
|
|
|
|
|
Purchases of marketable securities |
|
|
(43,987 |
) |
|
|
(24,280 |
) |
Maturities of marketable securities |
|
|
54,000 |
|
|
|
60,200 |
|
In-process research and development |
|
|
— |
|
|
|
(20,000 |
) |
Purchases of property and equipment |
|
|
(42 |
) |
|
|
(1,206 |
) |
Net cash provided by investing activities |
|
|
9,971 |
|
|
|
14,714 |
|
|
|
|
|
|
|
|
Cash flows from financing activities: |
|
|
|
|
|
|
Proceeds from prepayment of private placement |
|
|
6,907 |
|
|
|
— |
|
Proceeds from the exercise of stock options |
|
|
24 |
|
|
|
183 |
|
Net cash provided by financing activities |
|
|
6,931 |
|
|
|
183 |
|
|
|
|
|
|
|
|
Net decrease in cash, cash equivalents and restricted cash |
|
|
(16,349 |
) |
|
|
(11,461 |
) |
Cash, cash equivalents and restricted cash at beginning of the period |
|
|
93,483 |
|
|
|
284,625 |
|
Cash, cash equivalents and restricted cash at end of the period |
|
$ |
77,134 |
|
|
$ |
273,164 |
|
|
|
|
|
|
|
|
Supplemental disclosure of noncash investing and financing activities: |
|
|
|
|
|
|
Amounts accrued for purchases of property and equipment |
|
$ |
— |
|
|
$ |
38 |
|
Amounts accrued for offering costs |
|
$ |
178 |
|
|
$ |
— |
|
Vesting of early exercised options |
|
$ |
155 |
|
|
$ |
243 |
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Erasca, Inc.
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Note 1. Organization and basis of presentation
Organization and nature of operations
Erasca, Inc. (Erasca or the Company) is a clinical-stage precision oncology company singularly focused on discovering, developing, and commercializing therapies for RAS/MAPK pathway-driven cancers. The Company has assembled a wholly-owned or controlled RAS/MAPK pathway-focused pipeline comprising modality-agnostic programs aligned with its three therapeutic strategies of: (1) targeting key upstream and downstream signaling nodes in the RAS/MAPK pathway; (2) targeting RAS directly; and (3) targeting escape routes that emerge in response to treatment. The Company was incorporated under the laws of the State of Delaware on July 2, 2018, as Erasca, Inc., and is headquartered in San Diego, California. In September 2020, the Company established a wholly-owned Australian subsidiary, Erasca Australia Pty Ltd (Erasca Australia), in order to conduct clinical activities in Australia for its development candidates. In November 2020, the Company entered into an agreement and plan of merger with Asana BioSciences, LLC (Asana) and ASN Product Development, Inc. (ASN) (the Asana Merger Agreement), pursuant to which ASN became the Company's wholly-owned subsidiary. In March 2021, the Company established a wholly-owned subsidiary, Erasca Ventures, LLC (Erasca Ventures), to make equity investments in early-stage biotechnology companies that are aligned with the Company’s mission and strategy.
Since inception, the Company has devoted substantially all of its efforts and resources to organizing and staffing the Company, business planning, raising capital, identifying, acquiring and in-licensing the Company’s product candidates, establishing its intellectual property portfolio, conducting research, preclinical studies, and clinical trials, establishing arrangements with third parties for the manufacture of its product candidates and related raw materials, and providing general and administrative support for these operations. As of March 31, 2024, the Company had $297.7 million in cash, cash equivalents, and short-term marketable securities. As of March 31, 2024, the Company had an accumulated deficit of $641.0 million. The Company has incurred significant operating losses and negative cash flows from operations. From its inception through March 31, 2024, the Company’s financial support has primarily been provided from the sale of its convertible preferred stock and the sale of its common stock in its initial public offering (IPO) and underwritten offering (2022 Offering).
The Company expects to use its cash, cash equivalents, and short-term marketable securities to fund research and development, working capital, and other general corporate purposes. The Company does not expect to generate any revenues from product sales unless and until the Company successfully completes development and obtains regulatory approval for any of its product candidates, which will not be for at least the next several years, if ever. Accordingly, until such time as the Company can generate significant revenue from sales of its product candidates, if ever, the Company expects to finance its cash needs through equity offerings, debt financings, or other capital sources, including potential collaborations, licenses or other similar arrangements. However, the Company may not be able to secure additional financing or enter into such other arrangements in a timely manner or on favorable terms, if at all. The Company’s failure to raise capital or enter into such other arrangements when needed would have a negative impact on the Company’s financial condition and could force the Company to delay, limit, reduce or terminate its research and development programs or other operations, or grant rights to develop and market product candidates that the Company would otherwise prefer to develop and market itself. The Company believes its cash, cash equivalents, and short-term marketable securities as of March 31, 2024, plus the net proceeds received in April 2024 from the Company's private placement, which closed on April 2, 2024, will be sufficient for the Company to fund operations for at least one year from the issuance date of these condensed consolidated financial statements.
2024 Private Placement
In March 2024, the Company entered into a stock purchase agreement with the purchasers named therein for the private placement of 21,844,660 shares of its common stock at a price of $2.06 per share (the 2024 Private Placement). On April 2, 2024, the 2024 Private Placement closed and the net proceeds were $43.7 million, after deducting placement agent fees and estimated expenses of approximately $1.3 million. The Company received $6.9 million of the proceeds in March 2024 prior to the closing of the 2024 Private Placement, which is recorded in cash and cash equivalents and as a liability in proceeds from prepayment of private placement on the condensed consolidated balance sheets.
Basis of presentation
The accompanying unaudited condensed consolidated financial statements have been prepared in accordance with US generally accepted accounting principles (US GAAP) for interim financial information and pursuant to Form 10-Q and Article 10 of Regulation S-X of the Securities and Exchange Commission (SEC). Accordingly, they do not include all of the information and notes required by US GAAP for complete financial statements. Any reference in these notes to applicable guidance is meant to refer to US GAAP as found in the Accounting Standards Codification (ASC) and Accounting Standards Updates (ASU) promulgated by the Financial Accounting Standards Board (FASB). The Company’s condensed consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries, Erasca Australia, ASN, and Erasca Ventures. All intercompany balances and transactions have been eliminated.
Note 2. Summary of significant accounting policies
Use of estimates
The preparation of the Company’s condensed consolidated financial statements in conformity with US GAAP requires the Company to make estimates and assumptions that impact the reported amounts of assets, liabilities, expenses, and the disclosure of contingent assets and liabilities in the condensed consolidated financial statements and accompanying notes. Accounting estimates and management judgments reflected in the condensed consolidated financial statements include, but are not limited to, the accrual of research and development expenses, stock-based compensation expense, and the incremental borrowing rate for determining the operating lease asset and liability. Management evaluates its estimates on an ongoing basis. Although estimates are based on the Company’s historical experience, knowledge of current events, and actions it may undertake in the future, actual results may ultimately materially differ from these estimates and assumptions.
Unaudited interim financial information
The accompanying condensed consolidated balance sheet as of March 31, 2024, the condensed consolidated statements of operations and comprehensive loss for the three months ended March 31, 2024 and 2023, the condensed consolidated statements of stockholders’ equity for the three months ended March 31, 2024 and 2023 and the condensed consolidated statements of cash flows for the three months ended March 31, 2024 and 2023 are unaudited. The unaudited condensed consolidated interim financial statements have been prepared on the same basis as the audited annual consolidated financial statements and, in the opinion of management, reflect all adjustments, which include only normal recurring adjustments, necessary for the fair statement of the Company’s condensed consolidated financial position as of March 31, 2024 and the condensed consolidated results of its operations and cash flows for the three months ended March 31, 2024 and 2023. The condensed consolidated financial data and other information disclosed in these notes related to the three months ended March 31, 2024 and 2023 are unaudited. The condensed consolidated results for the three months ended March 31, 2024 are not necessarily indicative of results to be expected for the year ending December 31, 2024, any other interim periods, or any future year or period. These unaudited condensed consolidated financial statements should be read in conjunction with the Company's audited consolidated financial statements included in its Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
Concentration of credit risk and off-balance sheet risk
Financial instruments which potentially subject the Company to significant concentration of credit risk consist of cash and cash equivalents and marketable securities. The Company maintains deposits in federally insured financial institutions in excess of federally insured limits. The Company has not experienced any losses in such accounts, and management believes that the Company is not exposed to significant credit risk due to the financial position of the depository institutions in which those deposits are held. The Company’s investment policy includes guidelines for the quality of the related institutions and financial instruments and defines allowable investments that the Company may invest in, which the Company believes minimizes the exposure to concentration of credit risk.
Cash, cash equivalents and restricted cash
Cash and cash equivalents include cash in readily available checking and savings accounts, money market funds and commercial paper. The Company considers all highly liquid investments with an original maturity of three months or less from the date of purchase to be cash equivalents.
The Company had deposited cash of $408,000 as of March 31, 2024 and December 31, 2023 to secure a letter of credit in connection with the lease of the Company’s facilities (see Note 10). The Company has classified the restricted cash as a noncurrent asset on its condensed consolidated balance sheets.
The following table provides a reconciliation of cash, cash equivalents and restricted cash reported within the condensed consolidated balance sheets that sum to the total of the same amounts shown in the condensed consolidated statements of cash flows (in thousands):
|
|
|
|
|
|
|
|
|
March 31, |
|
|
2024 |
|
|
2023 |
|
Cash and cash equivalents |
$ |
76,726 |
|
|
$ |
272,756 |
|
Restricted cash |
|
408 |
|
|
|
408 |
|
Total cash, cash equivalents and restricted cash |
$ |
77,134 |
|
|
$ |
273,164 |
|
Marketable securities and investments
The Company classifies all marketable securities as available-for-sale, as the sale of such securities may be required prior to maturity. Management determines the appropriate classification of its marketable securities at the time of purchase. Marketable securities with original maturities beyond three months at the date of purchase and which mature at, or less than 12 months from, the balance sheet date are classified as short-term marketable securities. Available-for-sale securities are carried at fair value, with the unrealized gains and losses reported as accumulated other comprehensive income (loss) until realized. The amortized cost of available-for-sale debt securities is adjusted for amortization of premiums and accretion of discounts to maturity. Such amortization and accretion are included in interest income. The Company regularly reviews all of its marketable securities for declines in fair value. The review includes the consideration of the cause of the impairment, including the creditworthiness of the security issuers, the number of securities in an unrealized loss position, the severity of the unrealized loss(es), whether the Company has the intent to sell the securities and whether it is more likely than not that the Company will be required to sell the securities before the recovery of their amortized cost basis. If the decline in fair value is due to credit-related factors, a loss is recognized in net income; whereas, if the decline in fair value is not due to credit-related factors, the loss is recorded in other comprehensive income (loss). Realized gains and losses on available-for-sale securities are included in other income or expense. The cost of securities sold is based on the specific identification method. Interest and dividends on securities classified as available-for-sale are included in interest income.
Through its wholly-owned subsidiary, Erasca Ventures, the Company has also invested in equity securities of a company whose securities are not publicly traded and whose fair value is not readily available (see Notes 3 and 14). This investment is recorded using cost minus impairment, plus or minus changes in its estimated fair value resulting from observable price changes in orderly transactions for the identical or a similar investment of the same issuer. Investments in equity securities without readily determinable fair values are assessed for potential impairment on a quarterly basis based on qualitative factors. This investment is included in other assets in the Company's condensed consolidated balance sheets.
Fair value measurements
Certain assets and liabilities are carried at fair value under US GAAP. Fair value is defined as an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. Financial assets and liabilities carried at fair value are classified and disclosed in one of the following three levels of the fair value hierarchy, of which the first two are considered observable and the last is considered unobservable:
Level 1—Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities.
Level 2—Quoted prices for similar assets and liabilities in active markets, quoted prices in markets that are not active, or inputs which are observable, either directly or indirectly, for substantially the full term of the asset or liability.
Level 3—Prices or valuation techniques that require inputs that are both significant to the fair value measurement and unobservable (i.e., supported by little or no market activity).
Recently issued accounting pronouncements not yet adopted
From time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that the Company adopts as of the specified effective date. The Company qualifies as an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012 (JOBS Act) and has elected not to “opt out” of the extended transition related to complying with new or revised accounting standards, which means that when a standard is issued or revised and it has different application dates for public and nonpublic companies, the Company can adopt the new or revised standard at the time nonpublic companies adopt the new or revised standard and can do so until such time that the Company either (i) irrevocably elects to “opt out” of such extended transition period or (ii) no longer qualifies as an emerging growth company.
In November 2023, the FASB issued ASU 2023-07, Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures, which is intended to provide enhancements to segment disclosures, even for entities with only one reportable segment. In particular, the standard will require disclosures of significant segment expenses regularly provided to the chief operating decision maker and included within each reported measure of segment profit and loss. The standard will also require disclosure of all other segment items by reportable segment and a description of its composition. Finally, the standard will require disclosure of the title and position of the chief operating decision maker and an explanation of how the chief operating decision maker uses the reported measure(s) of segment profit or loss in assessing segment performance and deciding how to allocate resources. The standard is effective for annual periods beginning after December 15, 2023, and interim periods within annual periods beginning after December 15, 2024. Early adoption is permitted. Retrospective application to all prior periods presented in the financial statements is required. The Company is currently evaluating the impact of the standard on the presentation of its consolidated financial statements and related disclosures.
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures, which is intended to provide enhancements to annual income tax disclosures. In particular, the standard will require more detailed information in the income tax rate reconciliation, as well as the disclosure of income taxes paid disaggregated by jurisdiction, among other enhancements. The standard is effective for the Company in its annual period beginning after December 15, 2025 and early adoption is permitted. The standard allows for adoption on a prospective basis, with a retrospective option. The Company is currently evaluating the impact of the standard on the presentation of its consolidated financial statements and related disclosures.
Note 3. Fair value measurements
The following tables summarize the Company’s financial assets measured at fair value on a recurring basis and their respective input levels based on the fair value hierarchy (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value measurements as of March 31, 2024 using |
|
|
|
|
|
|
Quoted prices in |
|
|
Significant |
|
|
Significant |
|
|
|
|
|
|
active markets |
|
|
other |
|
|
unobservable |
|
|
|
March 31, |
|
|
for identical |
|
|
observable |
|
|
inputs |
|
|
|
2024 |
|
|
assets (level 1) |
|
|
inputs (level 2) |
|
|
(level 3) |
|
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds(1) |
|
$ |
58,324 |
|
|
$ |
58,324 |
|
|
$ |
— |
|
|
$ |
— |
|
Commercial paper(1) |
|
|
1,492 |
|
|
|
— |
|
|
|
1,492 |
|
|
|
— |
|
US treasury securities(2) |
|
|
73,644 |
|
|
|
73,644 |
|
|
|
— |
|
|
|
— |
|
US government agency securities(2) |
|
|
23,856 |
|
|
|
— |
|
|
|
23,856 |
|
|
|
— |
|
Corporate debt securities(2) |
|
|
22,926 |
|
|
|
— |
|
|
|
22,926 |
|
|
|
— |
|
Commercial paper(2) |
|
|
100,533 |
|
|
|
— |
|
|
|
100,533 |
|
|
|
— |
|
Total fair value of assets |
|
$ |
280,775 |
|
|
$ |
131,968 |
|
|
$ |
148,807 |
|
|
$ |
— |
|
(1)Included as cash and cash equivalents on the condensed consolidated balance sheets.
(2)Included as short-term marketable securities on the condensed consolidated balance sheets.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value measurements as of December 31, 2023 using |
|
|
|
|
|
|
Quoted prices in |
|
|
Significant |
|
|
Significant |
|
|
|
|
|
|
active markets |
|
|
other |
|
|
unobservable |
|
|
|
December 31, |
|
|
for identical |
|
|
observable |
|
|
inputs |
|
|
|
2023 |
|
|
assets (level 1) |
|
|
inputs (level 2) |
|
|
(level 3) |
|
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds(1) |
|
$ |
83,101 |
|
|
$ |
83,101 |
|
|
$ |
— |
|
|
$ |
— |
|
US treasury securities(2) |
|
|
93,303 |
|
|
|
93,303 |
|
|
|
— |
|
|
|
— |
|
US government agency securities(2) |
|
|
26,824 |
|
|
|
— |
|
|
|
26,824 |
|
|
|
— |
|
Corporate debt securities(2) |
|
|
10,734 |
|
|
|
— |
|
|
|
10,734 |
|
|
|
— |
|
Commercial paper(2) |
|
|
88,414 |
|
|
|
— |
|
|
|
88,414 |
|
|
|
— |
|
US treasury securities(3) |
|
|
9,642 |
|
|
|
9,642 |
|
|
|
— |
|
|
|
— |
|
Total fair value of assets |
|
$ |
312,018 |
|
|
$ |
186,046 |
|
|
$ |
125,972 |
|
|
$ |
— |
|
(1)Included as cash and cash equivalents on the condensed consolidated balance sheets.
(2)Included as short-term marketable securities on the condensed consolidated balance sheets.
(3)Included as long-term marketable securities on the condensed consolidated balance sheets.
The carrying amounts of the Company’s financial instruments, including cash, prepaid expenses and other current assets, accounts payable, accrued expenses and other current liabilities and proceeds from prepayment of private placement, approximate fair value due to their short maturities. As of March 31, 2024 and December 31, 2023, the Company held a $2.0 million equity investment in Affini-T Therapeutics, Inc. (Affini-T) at cost. No adjustments have been made to the value of the Company’s investment in Affini-T since its initial measurement either due to impairment or based on observable price changes. None of the Company’s non-financial assets or liabilities are recorded at fair value on a non-recurring basis. No transfers between levels have occurred during the periods presented.
Cash equivalents consist of money market funds and commercial paper, short-term marketable securities consist of US treasury securities, US government agency securities, corporate debt securities and commercial paper, and long-term marketable securities consist of US treasury securities. The Company obtains pricing information from its investment manager and generally determines the fair value of marketable securities using standard observable inputs, including benchmark yields, reported trades, broker/dealer quotes, issuer spreads, two-sided markets, and bid and/or offers.
Note 4. Marketable securities
The following tables summarize the Company’s marketable securities accounted for as available-for-sale securities (in thousands, except years):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2024 |
|
|
|
Maturity |
|
Amortized |
|
|
Unrealized |
|
|
Unrealized |
|
|
Estimated |
|
|
|
(in years) |
|
cost |
|
|
gains |
|
|
losses |
|
|
fair value |
|
US treasury securities |
|
1 or less |
|
$ |
73,753 |
|
|
$ |
8 |
|
|
$ |
(117 |
) |
|
$ |
73,644 |
|
US government agency securities |
|
1 or less |
|
|
23,859 |
|
|
|
11 |
|
|
|
(14 |
) |
|
|
23,856 |
|
Corporate debt securities |
|
1 or less |
|
|
22,967 |
|
|
|
— |
|
|
|
(41 |
) |
|
|
22,926 |
|
Commercial paper |
|
1 or less |
|
|
100,590 |
|
|
|
16 |
|
|
|
(73 |
) |
|
|
100,533 |
|
Total |
|
|
|
$ |
221,169 |
|
|
$ |
35 |
|
|
$ |
(245 |
) |
|
$ |
220,959 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2023 |
|
|
|
Maturity |
|
Amortized |
|
|
Unrealized |
|
|
Unrealized |
|
|
Estimated |
|
|
|
(in years) |
|
cost |
|
|
gains |
|
|
losses |
|
|
fair value |
|
US treasury securities |
|
1 or less |
|
$ |
93,377 |
|
|
$ |
74 |
|
|
$ |
(148 |
) |
|
$ |
93,303 |
|
US government agency securities |
|
1 or less |
|
|
26,783 |
|
|
|
45 |
|
|
|
(4 |
) |
|
|
26,824 |
|
Corporate debt securities |
|
1 or less |
|
|
10,719 |
|
|
|
15 |
|
|
|
— |
|
|
|
10,734 |
|
Commercial paper |
|
1 or less |
|
|
88,356 |
|
|
|
68 |
|
|
|
(10 |
) |
|
|
88,414 |
|
US treasury securities |
|
1-2 |
|
|
9,605 |
|
|
|
37 |
|
|
|
— |
|
|
|
9,642 |
|
Total |
|
|
|
$ |
228,840 |
|
|
$ |
239 |
|
|
$ |
(162 |
) |
|
$ |
228,917 |
|
The following tables present fair values and gross unrealized losses for those available-for-sale securities that were in an unrealized loss position as of March 31, 2024 and December 31, 2023, aggregated by category and the length of time that the securities have been in a continuous loss position (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2024 |
|
|
|
Unrealized losses less than 12 months |
|
|
Unrealized losses 12 months or greater |
|
|
Total |
|
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
US treasury securities |
|
$ |
55,139 |
|
|
$ |
(117 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
55,139 |
|
|
$ |
(117 |
) |
US government agency securities |
|
|
18,845 |
|
|
|
(14 |
) |
|
|
— |
|
|
|
— |
|
|
|
18,845 |
|
|
|
(14 |
) |
Corporate debt securities |
|
|
22,926 |
|
|
|
(41 |
) |
|
|
— |
|
|
|
— |
|
|
|
22,926 |
|
|
|
(41 |
) |
Commercial paper |
|
|
65,574 |
|
|
|
(73 |
) |
|
|
— |
|
|
|
— |
|
|
|
65,574 |
|
|
|
(73 |
) |
Total |
|
$ |
162,484 |
|
|
$ |
(245 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
162,484 |
|
|
$ |
(245 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2023 |
|
|
|
Unrealized losses less than 12 months |
|
|
Unrealized losses 12 months or greater |
|
|
Total |
|
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
US treasury securities |
|
$ |
74,912 |
|
|
$ |
(148 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
74,912 |
|
|
$ |
(148 |
) |
US government agency securities |
|
|
6,950 |
|
|
|
(4 |
) |
|
|
— |
|
|
|
— |
|
|
|
6,950 |
|
|
|
(4 |
) |
Corporate debt securities |
|
|
748 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
748 |
|
|
|
— |
|
Commercial paper |
|
|
22,944 |
|
|
|
(10 |
) |
|
|
— |
|
|
|
— |
|
|
|
22,944 |
|
|
|
(10 |
) |
Total |
|
$ |
105,554 |
|
|
$ |
(162 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
105,554 |
|
|
$ |
(162 |
) |
As of March 31, 2024, there were 28 available-for-sale securities with an estimated fair value of $162.5 million in gross unrealized loss positions, none of which were in an unrealized loss position for more than 12 months. As of December 31, 2023, there were 22 available-for-sale securities with an estimated fair value of $105.6 million in gross unrealized loss positions, none of which were in an unrealized loss position for more than 12 months.
As of March 31, 2024 and December 31, 2023, unrealized losses on available-for-sale securities are not attributed to credit risk. The Company believes that an allowance for credit losses is unnecessary because the unrealized losses on certain of the Company’s available-for-sale securities are due to market factors and interest rate increases. Additionally, the Company does not intend to sell the securities nor is it more likely than not that the Company will be required to sell the securities before recovery of their amortized cost basis.
Accrued interest on the Company’s available-for-sale securities was $871,000 and $1.1 million as of March 31, 2024 and December 31, 2023, respectively, and was included in prepaid expenses and other current assets on the condensed consolidated balance sheets.
Note 5. Property and equipment, net
Property and equipment, net consisted of the following (in thousands):
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Laboratory equipment |
|
$ |
5,621 |
|
|
$ |
5,620 |
|
Furniture and fixtures |
|
|
4,099 |
|
|
|
4,099 |
|
Leasehold improvements |
|
|
18,173 |
|
|
|
18,173 |
|
Computer equipment and software |
|
|
1,695 |
|
|
|
1,667 |
|
Property and equipment |
|
|
29,588 |
|
|
|
29,559 |
|
Less accumulated depreciation and amortization |
|
|
(8,230 |
) |
|
|
(7,232 |
) |
Property and equipment, net |
|
$ |
21,358 |
|
|
$ |
22,327 |
|
Depreciation and amortization expense related to property and equipment was $998,000 and $894,000 for the three months ended March 31, 2024 and 2023, respectively.
Note 6. Accrued expenses and other current liabilities
Accrued expenses and other current liabilities consisted of the following (in thousands):
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Accrued research and development expenses |
|
$ |
9,247 |
|
|
$ |
8,803 |
|
Accrued compensation |
|
|
6,095 |
|
|
|
10,311 |
|
Unvested early exercised stock option liability |
|
|
309 |
|
|
|
464 |
|
Accrued professional services |
|
|
626 |
|
|
|
435 |
|
Accrued offering costs |
|
|
178 |
|
|
|
— |
|
Accrued property and equipment |
|
|
— |
|
|
|
13 |
|
Other accruals |
|
|
122 |
|
|
|
160 |
|
Total |
|
$ |
16,577 |
|
|
$ |
20,186 |
|
Note 7. Asset acquisitions
The following purchased assets were accounted for as asset acquisitions as substantially all of the fair value of the assets acquired were concentrated in a group of similar assets, and the acquired assets did not have outputs or employees. Because the assets had not yet received regulatory approval, the fair value attributable to these assets was recorded as in-process research and development expenses in the Company’s condensed consolidated statements of operations and comprehensive loss.
Asana BioSciences, LLC
In November 2020, the Company entered into the Asana Merger Agreement, pursuant to which ASN became its wholly-owned subsidiary. Asana and ASN had previously entered into a license agreement, which was amended and restated prior to the closing of the merger transaction (the Asana License Agreement, and collectively with the Asana Merger Agreement, the Asana Agreements), pursuant to which ASN acquired an exclusive, worldwide license to certain intellectual property rights relating to inhibitors of ERK1 and ERK2 owned or controlled by Asana to develop and commercialize ERAS-007 and certain other related compounds for all applications.
Under the Asana Merger Agreement, in 2020, the Company made an upfront payment of $20.0 million and issued 4,000,000 shares of its Series B-2 convertible preferred stock to Asana at a value of $7.50 per share or a total fair value of equity of $30.0 million. In connection with the Company's IPO, these shares of Series B-2 convertible preferred stock were converted into 3,333,333 shares of the Company’s common stock. The Company is obligated to make future development and regulatory milestone cash payments for a licensed product in an amount of up to $90.0 million. Additionally, upon achieving a development milestone related to demonstration of successful proof-of-concept in a specified clinical trial, the Company will also be required to issue 3,888,889 shares of its common stock to Asana. The Company is not obligated to pay royalties on the net sales of licensed products. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones had been accrued as the underlying contingencies were not probable or estimable.
Emerge Life Sciences, Pte. Ltd.
In March 2021, the Company entered into an asset purchase agreement (ELS Purchase Agreement) with Emerge Life Sciences, Pte. Ltd. (ELS) wherein it purchased all rights, title, and interest (including all patent and other intellectual property rights) to EGFR antibodies directed against the EGFR domain II (EGFR-D2) and domain III (EGFR-D3) as well as a bispecific antibody where one arm is directed against EGFR-D2 and the other is directed against EGFR-D3 (the Antibodies). Under the terms of the ELS Purchase Agreement, in 2021, the Company made an upfront payment of $2.0 million and issued to ELS 500,000 shares of the Company’s common stock at a value of $3.36 per share or a total fair value of equity of $1.7 million. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023.
Note 8. License agreements
Novartis Pharma AG
In December 2022, the Company entered into an exclusive license agreement (as amended, the Novartis Agreement) with Novartis Pharma AG (Novartis) under which the Company was granted an exclusive, worldwide, royalty-bearing license to certain patent and other intellectual property rights owned or controlled by Novartis to develop, manufacture, use, and commercialize naporafenib in all fields of use. The Company has the right to sublicense (through multiple tiers) its rights under the Novartis Agreement, subject to certain limitations and conditions, and is required to use commercially reasonable efforts to commercialize licensed products in certain geographical markets.
The license granted under the Novartis Agreement is subject to Novartis’ reserved right to: (i) develop, manufacture, use, and commercialize compounds unrelated to naporafenib under the licensed patent rights and know-how, (ii) use the licensed patent rights and know-how for non-clinical research purposes, and (iii) use the licensed patent rights and know-how to the extent necessary to perform ongoing clinical trials and perform its obligations under existing contracts and under the Novartis Agreement.
Under the Novartis Agreement, the Company made an upfront cash payment to Novartis of $20.0 million and issued to Novartis 12,307,692 shares of common stock of the Company having an aggregate value of approximately $80.0 million. The Company is obligated to make future regulatory milestone payments of up to $80.0 million and sales milestone payments of up to $200.0 million. The Company is also obligated to pay royalties on net sales of all licensed products, in the low-single digit percentages, subject to certain reductions. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable.
Katmai Pharmaceuticals, Inc.
In March 2020, the Company entered into a license agreement (the Katmai Agreement) with Katmai Pharmaceuticals, Inc. (Katmai) under which the Company was granted an exclusive, worldwide, royalty-bearing license to certain patent rights and know-how controlled by Katmai related to the development of small molecule therapeutic and diagnostic products that modulate EGFR and enable the identification, diagnosis, selection, treatment, and/or monitoring of patients for neuro-oncological applications to develop, manufacture, use, and commercialize ERAS-801 and certain other related compounds in all fields of use.
Under the Katmai Agreement, the Company made an upfront payment of $5.7 million and Katmai agreed to purchase shares of the Company’s Series B-1 convertible preferred stock and Series B-2 convertible preferred stock having an aggregate value of $2.7 million. In April 2020, Katmai purchased 356,000 shares of the Company’s Series B-1 convertible preferred stock for $1.8 million, and in January 2021, Katmai purchased 118,666 shares of the Company’s Series B-2 convertible preferred stock for $0.9 million. In connection with the Company's IPO, these shares of Series B-1 convertible preferred stock and Series B-2 convertible preferred stock were converted into 395,555 shares of the Company's common stock, in the aggregate. The Company is obligated to make future development and regulatory milestone payments of up to $26.0 million, of which $2.0 million was paid in March 2022, and commercial milestone payments of up to $101.0 million. The Company is also obligated to pay tiered royalties on net sales of each licensed product, at rates ranging from the mid- to high-single digit percentages, subject to a minimum annual royalty payment in the low six figures and certain permitted deductions. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable.
NiKang Therapeutics, Inc.
In February 2020, the Company entered into a license agreement (the NiKang Agreement) with NiKang Therapeutics, Inc. (NiKang) under which the Company was granted an exclusive, worldwide license to certain intellectual property rights owned or controlled by NiKang related to certain SHP2 inhibitors to develop and commercialize ERAS-601 and certain other related compounds for all applications.
Under the NiKang Agreement, in 2020, the Company made an upfront payment of $5.0 million to NiKang and reimbursed NiKang $0.4 million for certain initial manufacturing costs. In addition, the Company paid $7.0 million in 2020 related to the publication of a US patent application that covered the composition of matter of ERAS-601. The Company is also obligated to pay (i) development and regulatory milestone payments in an aggregate amount of up to $16.0 million for the first licensed product, of which $4.0 million was paid in January 2021, and $12.0 million for a second licensed product, and (ii) commercial milestone payments in an aggregate amount of up to $157.0 million for the first licensed product and $151.0 million for a second licensed product. The Company is also obligated to: (i) pay tiered royalties on net sales of all licensed products in the mid-single digit percentages, subject to certain reductions; and (ii) equally split all net sublicensing revenues earned under sublicense agreements that the Company enters into with any third party before commencement of the first Phase I clinical trial for a licensed product. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable.
LifeArc
In April 2020, the Company entered into a license agreement with LifeArc (the LifeArc Agreement) under which the Company was granted an exclusive, worldwide license to certain materials, know-how, and intellectual property rights owned or controlled by LifeArc to develop, manufacture, use, and commercialize certain ULK inhibitors for all applications.
Under the LifeArc Agreement, the Company was granted the license at no upfront cost and a period of three months after the effective date to conduct experiments on LifeArc’s compounds. Upon completion of this initial testing period, the Company had the option to continue the license and make a one-time license payment of $75,000 to LifeArc, which payment was subsequently made in 2020. The Company is obligated to make future development milestone payments for a licensed product of up to $11.0 million and sales milestone payments of up to $50.0 million. The Company is also obligated to pay royalties on net sales of all licensed products, in the low-single digit percentages, subject to certain reductions. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable.
Note 9. Stock-based compensation
In July 2021, the Company’s board of directors adopted and the Company’s stockholders approved the Company’s 2021 Incentive Award Plan (the 2021 Plan), which became effective in connection with the IPO. Upon the adoption of the 2021 Plan, the Company ceased making equity grants under its 2018 Equity Incentive Plan (the 2018 Plan). Under the 2021 Plan, the Company may grant stock options, restricted stock, restricted stock units, stock appreciation rights, and other stock or cash-based awards to individuals who are then employees, officers, directors or non-entity consultants of the Company. A total of 15,150,000 shares of common stock were initially reserved for issuance under the 2021 Plan. In addition, the number of shares of common stock available for issuance under the 2021 Plan may be increased annually on the first day of each calendar year during the term of the 2021 Plan, beginning in 2022, by an amount equal to the lesser of (i) 5% of the shares of common stock outstanding on the final day of the immediately preceding calendar year or (ii) such smaller number of shares as determined by the Company’s board of directors or an authorized committee of the board of directors. As of March 31, 2024, there were 13,204,701 stock-based awards available for future grant under the 2021 Plan.
Subsequent to July 2021, no further awards will be granted under the 2018 Plan and all future stock-based awards will be granted under the 2021 Plan. To the extent outstanding options or restricted stock granted under the 2018 Plan are cancelled, forfeited, repurchased, or otherwise terminated without being exercised or becoming vested, and would otherwise have been returned to the share reserve under the 2018 Plan, the number of shares underlying such awards will be available for future grant under the 2021 Plan.
Options granted are exercisable at various dates as determined upon grant and will expire no more than ten years from their date of grant. Stock options generally vest over a four-year term. The exercise price of each option shall be determined by the Company’s board of directors based on the estimated fair value of the Company’s stock on the date of the option grant. The exercise price shall not be less than 100% of the fair market value of the Company’s common stock at the time the option is granted. For holders of more than 10% of the Company’s total combined voting power of all classes of stock, incentive stock options may not be granted at less than 110% of the fair market value of the Company’s common stock on the date of grant and for a term that exceeds five years. Early exercise was permitted for certain grants under the 2018 Plan.
Stock options
A summary of the Company’s stock option activity under the 2021 Plan and 2018 Plan is as follows (in thousands, except share and per share data and years):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
average remaining |
|
|
Aggregate |
|
|
|
|
|
|
average |
|
|
contractual |
|
|
intrinsic |
|
|
|
Shares |
|
|
exercise price |
|
|
term (years) |
|
|
value |
|
Outstanding at December 31, 2023 |
|
|
24,970,957 |
|
|
$ |
4.98 |
|
|
|
8.12 |
|
|
$ |
4,412 |
|
Granted |
|
|
9,827,650 |
|
|
|
1.71 |
|
|
|
|
|
|
|
Exercised |
|
|
(35,035 |
) |
|
|
0.69 |
|
|
|
|
|
|
|
Canceled |
|
|
(116,449 |
) |
|
|
4.89 |
|
|
|
|
|
|
|
Outstanding at March 31, 2024 |
|
|
34,647,123 |
|
|
$ |
4.06 |
|
|
|
8.44 |
|
|
$ |
7,532 |
|
Options exercisable at March 31, 2024 |
|
|
12,850,952 |
|
|
$ |
4.83 |
|
|
|
7.32 |
|
|
$ |
3,916 |
|
The weighted-average grant date fair value of options granted for the three months ended March 31, 2024 and 2023 was $1.25 and $2.94, respectively. As of March 31, 2024, the unrecognized compensation cost related to unvested stock option grants was $55.9 million and is expected to be recognized as expense over approximately 2.75 years. The intrinsic value of the options exercised for the three months ended March 31, 2024 and 2023 was $47,000 and $517,000, respectively.
Prior to the Company's IPO, certain individuals were granted the ability to early exercise their stock options. The shares of common stock issued from the early exercise of unvested stock options are restricted and continue to vest in accordance with the original vesting schedule. The Company has the option to repurchase any unvested shares at the original purchase price upon any voluntary or involuntary termination. The shares purchased by the employees and non-employees pursuant to the early exercise of stock options are not deemed, for accounting purposes, to be outstanding until those shares vest. The cash received in exchange for exercised and unvested shares related to stock options granted is recorded as a liability for the early exercise of stock options on the accompanying condensed consolidated balance sheets and will be transferred into common stock and additional paid-in capital as the shares vest. As of March 31, 2024 and December 31, 2023, there were 247,919 shares and 371,876 shares subject to repurchase by the Company, respectively. As of March 31, 2024 and December 31, 2023, the Company recorded $309,000 and $464,000 of liabilities associated with shares issued with repurchase rights, respectively, which is recorded in accrued expenses and other current liabilities.
The assumptions used in the Black-Scholes option pricing model to determine the fair value of the employee and nonemployee stock option grants were as follows:
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
2024 |
|
2023 |
Risk-free interest rate |
|
3.84%-4.15% |
|
3.46%-4.22% |
Expected volatility |
|
83.35%-83.91% |
|
85.52%-85.81% |
Expected term (in years) |
|
5.28-6.01 |
|
6.02-6.07 |
Expected dividend yield |
|
--% |
|
--% |
Employee stock purchase plan
In July 2021, the Company’s board of directors adopted and the Company’s stockholders approved the Company's 2021 Employee Stock Purchase Plan (the ESPP), which became effective in connection with the IPO. The ESPP permits participants to contribute up to a specified percentage of their eligible compensation during a series of offering periods of 24 months, each comprised of four six-month purchase periods, to purchase the Company’s common stock. The purchase price of the shares will be 85% of the fair market value of the Company’s common stock on the first day of trading of the applicable offering period or on the applicable purchase date, whichever is lower. A total of 1,260,000 shares of common stock was initially reserved for issuance under the ESPP. In addition, the number of shares of common stock available for issuance under the ESPP may be increased annually on the first day of each calendar year during the term of the ESPP, beginning in 2022, by an amount equal to the lesser of (i) 1% of the shares of common stock outstanding on the final day of the immediately preceding calendar year or (ii) such smaller number of shares as determined by the Company’s board of directors or an authorized committee of the board of directors. The Company recognized stock-based compensation expense related to the ESPP of $342,000 and $707,000 during the three months ended March 31, 2024 and 2023, respectively. As of March 31, 2024, the unrecognized compensation cost related to the ESPP was $1.5 million and is expected to be recognized as expense over approximately 1.69 years. As of March 31, 2024 and December 31, 2023, $317,000 and $46,000 has been withheld on behalf of employees for future purchase under the ESPP, respectively, and is included in accrued expenses and other current liabilities on the condensed consolidated balance sheets. No shares were issued and sold under the ESPP during the three months ended March 31, 2024 and 2023.
Stock-based compensation expense
The allocation of stock-based compensation for all stock awards was as follows (in thousands):
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
2024 |
|
|
2023 |
|
Research and development |
|
$ |
3,484 |
|
|
$ |
3,880 |
|
General and administrative |
|
|
3,364 |
|
|
|
2,965 |
|
Total |
|
$ |
6,848 |
|
|
$ |
6,845 |
|
Common stock reserved for future issuance
Common stock reserved for future issuance consisted of the following as of March 31, 2024 and December 31, 2023:
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Stock options issued and outstanding |
|
|
34,647,123 |
|
|
|
24,970,957 |
|
Awards available for future grant |
|
|
13,204,701 |
|
|
|
15,342,797 |
|
Shares available for purchase under the ESPP |
|
|
2,080,681 |
|
|
|
2,080,681 |
|
Total |
|
|
49,932,505 |
|
|
|
42,394,435 |
|
Note 10. Leases
Operating leases
The Company has facility leases for office space under non-cancellable and cancelable operating leases with various expiration dates through 2032 and equipment under a non-cancellable operating lease with a term expiring in 2026. Total lease costs were approximately $2.7 million and $3.1 million, including operating lease costs of $1.9 million and $1.9 million, variable lease costs of $916,000 and $1.2 million, and sublease income of $84,000 and $0, during the three months ended March 31, 2024 and 2023, respectively. The Company paid $2.1 million and $863,000 in cash for operating leases, net of sublease income, that were included in the operating activities section of the condensed consolidated statements of cash flows for the three months ended March 31, 2024 and 2023, respectively.
The weighted-average remaining lease term and the weighted-average discount rate of the Company’s operating leases were 8.05 years and 8.96%, respectively, at March 31, 2024. The weighted-average remaining lease term and the weighted-average discount rate of the Company’s operating leases were 8.29 years and 8.95%, respectively, at December 31, 2023. The weighted-average remaining lease term does not include any renewal options at the election of the Company.
The Company’s lease agreements do not contain any material residual value guarantees or material restrictive covenants.
Facility leases
In September 2020, the Company entered into a lease agreement for 59,407 square feet of laboratory and office space in San Diego, California, which represented a portion of a new facility that was under construction and which was subsequently amended in March 2021 to expand the rented premises by 18,421 square feet (the 2020 Lease). The construction and design of the asset was the primary responsibility of the lessor. The Company was involved in certain aspects of construction and design for certain interior features and leasehold improvements that is beneficial to the Company to better suit its business needs and intended purpose of the space. The lease is accounted for as an operating lease and commenced in August 2021. In April 2022, the 2020 Lease was modified to amend the rent commencement date from February 2022 to May 2022. The 2020 Lease, as amended, has a term of 10.75 years and includes aggregate monthly payments to the lessor of approximately $51.6 million beginning in May 2023 with a rent escalation clause, and a tenant improvement allowance of approximately $16.8 million. The Company is responsible for its share of operating expenses based on actual operating expenses incurred by the landlord. The 2020 Lease is cancellable at the Company’s request after the 84th month with 12 months written notice and a lump-sum cancellation payment of $2.5 million. The termination option has not been included in the Company's operating lease assets and liabilities. As discussed in Note 2, the Company provided a letter of credit to the lessor for $408,000, which expires July 29, 2032.
In December 2021, the Company entered into a lease agreement for 29,542 square feet of office and laboratory space in South San Francisco, California. The lease is accounted for as an operating lease with the associated operating lease assets and liabilities recorded upon commencement, which occurred in July 2022. The non-cancellable operating lease has an initial term of 124 months with an option to extend the lease term by 5 years at the then-current market rates and includes aggregate monthly payments to the lessor of approximately $34.4 million beginning in November 2022 with a rent escalation clause and a tenant improvement allowance of approximately $8.2 million. The renewal option has not been included in the Company's operating lease assets and liabilities. The Company is responsible for its share of operating expenses based on actual operating expenses incurred by the landlord. The construction and design of the tenant improvements was the primary responsibility of the lessor. While the Company was involved in certain aspects of construction and design for certain interior features and leasehold improvements that is beneficial to the Company to better suit its business needs and intended purpose of the space, all construction was handled directly by the landlord. The Company was not deemed to be the accounting owner of the tenant improvements prior to or after the construction period. All payments made by the Company for landlord-owned tenant improvements were recorded as prepaid rent on the condensed consolidated balance sheets prior to lease commencement and included in the operating lease asset upon lease commencement. In February 2022, the expected project costs exceeded the tenant improvement allowances by $5.1 million, which was paid directly to the landlord by the Company and was recorded as prepaid rent in the condensed consolidated balance sheets and as a cash outflow from operating activities in the condensed consolidated statements of cash flows. Upon lease commencement, the $5.1 million of prepaid rent was included in the operating lease asset. The Company paid a security deposit of $874,000 in December 2021 that was recorded as other assets in the condensed consolidated balance sheets.
In January 2024, the Company entered into an agreement to sublease the second floor of its corporate headquarters in San Diego, California. Pursuant to the agreement, the subleased space is approximately 10,000 square feet of office space with a sublease term of three years which includes an option for the subtenant to renew for an additional year and an early termination clause. The Company determined the sublease to be an operating lease. Therefore, the Company will recognize sublease income on a straight-line basis over the lease term in its condensed consolidated statements of operations and comprehensive loss as a reduction to operating lease costs because the sublease is outside of the Company's normal business operations. The Company will continue to account for the operating lease asset and related liability of the original lease as it did prior to the commencement of the sublease. The Company recorded a reduction to operating lease costs of $84,000 related to income from this sublease during the three months ended March 31, 2024.
Future minimum lease payments under the operating leases with initial lease terms in excess of one year as of March 31, 2024 are as follows (in thousands):
|
|
|
|
Year ending December 31, |
|
|
2024 (remaining nine months) |
$ |
6,602 |
|
2025 |
|
9,024 |
|
2026 |
|
9,170 |
|
2027 |
|
9,199 |
|
2028 |
|
9,277 |
|
Thereafter |
|
35,098 |
|
Total lease payments |
$ |
78,370 |
|
Less: Amount representing interest |
|
(23,433 |
) |
Operating lease liabilities |
$ |
54,937 |
|
Note 11. Commitments and contingencies
Liabilities for loss contingencies arising from claims, assessments, litigation, fines, penalties and other sources are recorded when it is probable that a liability has been incurred and the amount can be reasonably estimated. There are no matters currently outstanding for which any such liabilities have been accrued.
Note 12. Income taxes
No provision for federal, state or foreign income taxes has been recorded for the three months ended March 31, 2024 and 2023. The Company has incurred net operating losses for all the periods presented and has not reflected any benefit of such net operating loss carryforwards in the accompanying condensed consolidated financial statements due to uncertainty around utilizing these tax attributes within their respective carryforward periods. The Company has recorded a full valuation allowance against all of its deferred tax assets as it is not more likely than not that such assets will be realized in the near future. The Company’s policy is to recognize interest expense and penalties related to income tax matters as tax expense. For the three months ended March 31, 2024 and 2023, the Company has not recognized any interest or penalties related to income taxes.
Note 13. Net loss per share
The following table summarizes the computation of basic and diluted net loss per share of the Company (in thousands, except share and per share data):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
|
|
|
|
|
2024 |
|
|
2023 |
|
Net loss |
|
|
|
|
|
|
$ |
(35,017 |
) |
|
$ |
(33,199 |
) |
Weighted-average shares of common stock used in computing net loss per share, basic and diluted |
|
|
|
|
|
|
|
151,161,741 |
|
|
|
149,504,216 |
|
Net loss per share, basic and diluted |
|
|
|
|
|
|
$ |
(0.23 |
) |
|
$ |
(0.22 |
) |
The Company’s potentially dilutive securities, which include options to purchase common stock, shares purchasable under the ESPP and common stock subject to repurchase related to options early exercised, have been excluded from the computation of diluted net loss per share as the effect would be to reduce the net loss per share. Therefore, the weighted-average number of common shares outstanding used to calculate both basic and diluted net loss per share is the same. The Company excluded the following potential common shares, presented as amounts outstanding at each period end, from the computation of diluted net loss per share for the periods indicated because including them would have had an anti-dilutive effect:
|
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
|
March 31, |
|
|
|
2024 |
|
|
|
2023 |
|
Options to purchase common stock |
|
|
34,647,123 |
|
|
|
|
25,892,271 |
|
Options early exercised subject to future vesting |
|
|
247,919 |
|
|
|
|
948,179 |
|
Estimated shares purchasable under the ESPP |
|
|
1,301,205 |
|
|
|
|
755,809 |
|
Total potentially dilutive shares |
|
|
36,196,247 |
|
|
|
|
27,596,259 |
|
Note 14. Related party transactions
Erasca Foundation
In May 2021, the Company established the Erasca Foundation to provide support such as funding research, patient advocacy, patient support and education in underserved populations, and funding for other initiatives to positively impact society that align with the Company’s mission. The Company's chief executive officer and certain board members serve as directors of the Erasca Foundation and the Company's chief executive officer, chief financial officer and chief business officer, and general counsel are also officers of the Erasca Foundation. In April 2024, the Company donated $125,000 to the Erasca Foundation.
Affini-T Therapeutics, Inc.
The Company holds a $2.0 million equity investment in Affini-T. One of the Company’s board members is also a member of the board of Affini-T.
Note 15. Subsequent event
On April 2, 2024, the Company completed the 2024 Private Placement in which the Company issued and sold 21,844,660 shares of common stock at a price of $2.06 per share. Net proceeds from the 2024 Private Placement, after deducting placement agent fees and estimated expenses, were $43.7 million.
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our unaudited condensed consolidated financial statements and notes thereto included elsewhere in this Quarterly Report on Form 10-Q and with our audited consolidated financial statements and notes thereto and management’s discussion and analysis of financial condition and results of operations for the year ended December 31, 2023, included in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
Cautionary Note Regarding Forward-Looking Statements
This Quarterly Report contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended (the Exchange Act). All statements other than statements of historical facts contained in this Quarterly Report, including statements regarding our future results of operations and financial position, business strategy, research and development plans, the anticipated timing, costs, design and conduct of our ongoing and planned preclinical studies and planned clinical trials for our product candidates, the timing and likelihood of regulatory filings and approvals for our product candidates, our ability to commercialize our product candidates, if approved, the impact of global geopolitical and economic events and war on our business, the pricing and reimbursement of our product candidates, if approved, the potential to develop future product candidates, the potential benefits of current and future licenses, acquisitions, and strategic arrangements with third parties, and our intent to enter into any future strategic arrangements, the timing and likelihood of success, plans and objectives of management for future operations and future results of anticipated product development efforts, are forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “could,” “expect,” “intend,” "target," “plan,” “anticipate,” “believe,” “estimate,” “predict,” “potential,” “continue,” or the negative of these terms or other similar expressions. These forward-looking statements are only predictions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking statements speak only as of the date of this Quarterly Report and are subject to a number of risks, uncertainties and assumptions, including those described in Part II, Item 1A, “Risk Factors.” The events and circumstances reflected in our forward-looking statements may not be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
Overview
We are a clinical-stage precision oncology company singularly focused on discovering, developing, and commercializing therapies for patients with RAS/MAPK pathway-driven cancers. Molecular alterations in RAS, the most frequently mutated oncogene, and the MAPK pathway, one of the most frequently altered signaling pathways in cancer, account for approximately 5.4 million new patients diagnosed with cancer globally each year. Our company was co-founded by leading pioneers in precision oncology and RAS targeting to create novel therapies and combination regimens designed to comprehensively shut down the RAS/MAPK pathway for the treatment of patients with cancer. We have assembled one of the deepest, wholly-owned or controlled RAS/MAPK pathway-focused pipelines in the industry, which comprises modality-agnostic programs aligned with our three therapeutic strategies of: (1) targeting key upstream and downstream signaling nodes in the RAS/MAPK pathway; (2) targeting RAS directly; and (3) targeting escape routes that emerge in response to treatment.
The following figure shows the RAS/MAPK pathway and how the three therapeutic strategies listed above attempt to comprehensively and synergistically shut down the RAS/MAPK pathway.
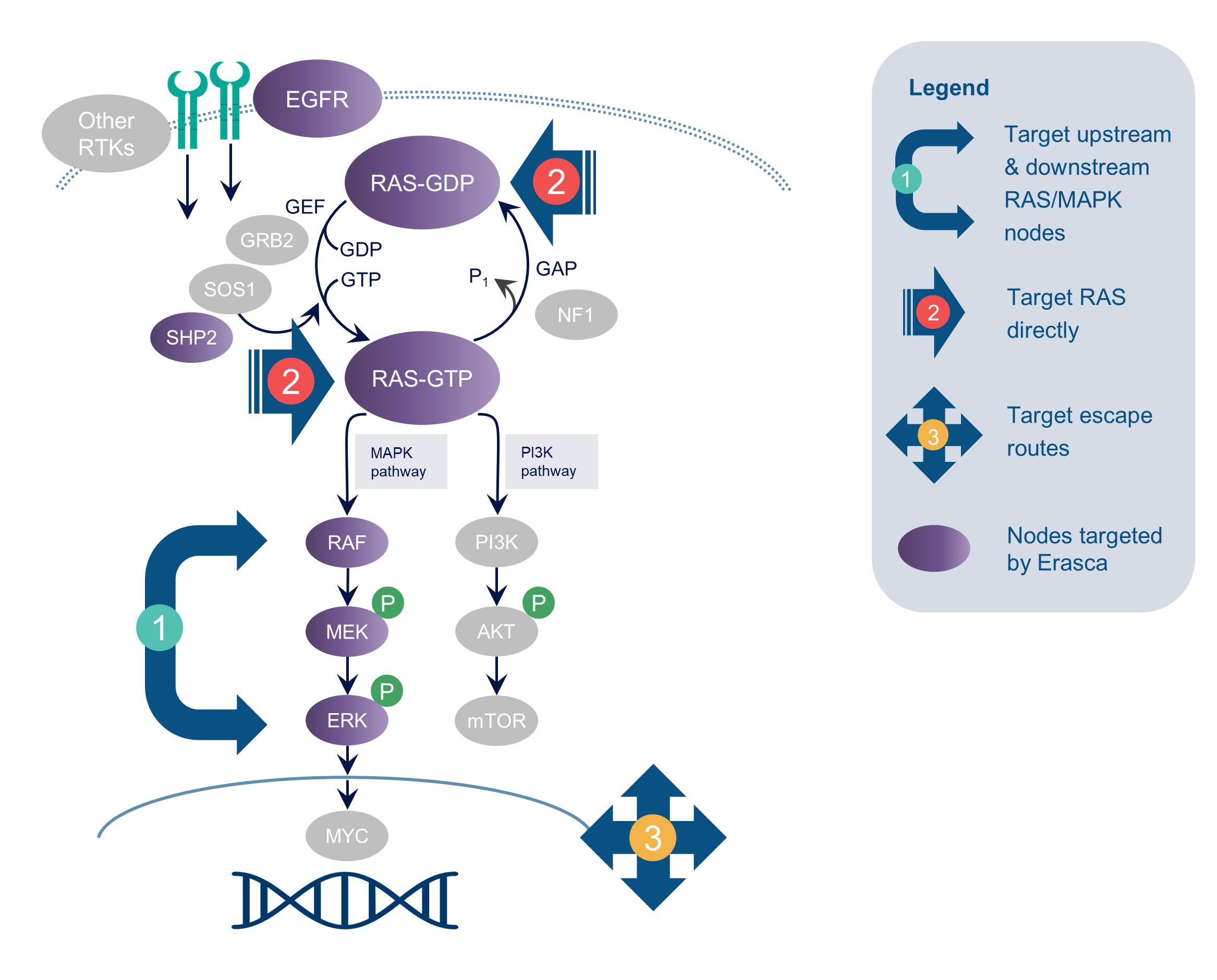
The target breadth and molecular diversity represented in our pipeline enable us to pursue a systematic, data-driven, portfolio-wide clinical development effort to identify single agent and combination approaches with the goal of prolonging survival in numerous patient populations with high unmet medical needs. Our modality-agnostic approach aims to allow us to selectively and potently target critical signaling nodes with the most appropriate modality, including small molecule therapeutics and large molecule therapeutics. Our purpose-built pipeline includes three clinical-stage programs (a pan-RAF inhibitor, an ERK inhibitor, and a central nervous system (CNS)-penetrant EGFR inhibitor), and additional discovery-stage programs targeting other key oncogenic drivers. We believe our world-class team’s capabilities and experience, further guided by our scientific advisory board, which includes the world’s leading experts in the RAS/MAPK pathway, uniquely position us to achieve our bold mission of erasing cancer.
Our lead product candidate is naporafenib, for which we plan to initiate a pivotal Phase 3 trial (SEACRAFT-2) in the second quarter of 2024 for patients with NRAS-mutated (NRASm) melanoma. We dosed the first patient in a Phase 1b trial (SEACRAFT-1) in August 2023 for patients with RAS Q61X solid tumors to inform additional clinical development pathways for naporafenib. Naporafenib is a pan-RAF inhibitor with first-in-class and best-in-class potential for patients with NRASm melanoma, RAS Q61X solid tumors, and other RAS/MAPK pathway-driven tumors. RAF proteins are ubiquitously expressed serine-threonine kinases that constitute a key node of the RAS/MAPK pathway downstream of RAS and upstream of MEK. The RAF protein family consists of ARAF, BRAF, and CRAF (RAF1) that are activated through dimerization. Mutations in RAF proteins have been observed in many cancers, such as melanoma, colorectal cancer (CRC), non-small cell lung cancer (NSCLC), and thyroid cancer. We in-licensed naporafenib from Novartis Pharma AG (Novartis) in December 2022. Naporafenib has been dosed in over
500 patients to date, whereby safety, tolerability, pharmacokinetics (PK), and pharmacodynamics have been established in both monotherapy and select combinations, with clinical proof-of-concept (PoC) data in combination with trametinib (MEKINIST) for patients with NRASm melanoma, which includes NRAS Q61X melanoma, and preliminary clinical PoC data in combination with trametinib for patients with RAS Q61X NSCLC. In December 2023, we announced that the US Food and Drug Administration (FDA) granted Fast Track Designation (FTD) to naporafenib in combination with trametinib for the treatment of adult patients with unresectable or metastatic melanoma who have progressed on, or are intolerant to, an anti‑programmed death-1 (ligand 1) (PD‑(L)1)-based regimen, and whose tumors contain an NRAS mutation (NRASm). Programs that receive FTD may benefit from early and frequent interactions with the FDA during the clinical development process and, if relevant criteria are met, the FDA may consider reviewing portions of a marketing application before the sponsor submits the complete application.
We are pursuing a broad development strategy for naporafenib, which includes our SEACRAFT trials designed to evaluate naporafenib’s development opportunities in combination with other targeted therapies. We are prioritizing rapid development for naporafenib plus trametinib in the Phase 1b SEACRAFT-1 trial for patients with RAS Q61X solid tumors, which dosed its first patient in August 2023, and in the planned Phase 3 SEACRAFT-2 trial for patients with NRASm melanoma. SEACRAFT-1 is supported by clinical PoC data in patients with NRAS Q61X melanoma and preliminary clinical PoC data in patients with KRAS Q61X NSCLC. SEACRAFT-2 is supported by clinical PoC data in patients with NRASm melanoma, as presented by Novartis at the European Society for Medical Oncology Congress 2022 medical conference and as published in March 2023 by de Braud et al. in the Journal of Clinical Oncology. In connection with our SEACRAFT-1 and -2 trials, we have entered into clinical trial collaboration and supply agreements (CTCSAs) with Novartis for its MEK inhibitor, trametinib (MEKINIST). Pursuant to the CTCSAs, we are sponsoring and funding the clinical trials and Novartis is providing its drug to us free of charge.
We anticipate a Phase 1b combination data readout from SEACRAFT-1 between the second and fourth quarters of 2024. We expect to readout randomized dose optimization data from SEACRAFT-2 in 2025.
Our next most-advanced product candidate is ERAS-007 (our oral ERK1/2 inhibitor), which targets the most distal node of the RAS/MAPK pathway. In September 2021, we dosed the first patient in HERKULES-3, a Phase 1b/2 master protocol clinical trial for ERAS-007 in combination with various agents in patients with gastrointestinal (GI) cancers. In connection with our HERKULES-3 trial, we have entered into CTCSAs with Pfizer Inc. for its BRAF inhibitor, encorafenib (BRAFTOVI), Eli Lilly and Company (Lilly) for its EGFR antibody, cetuximab (ERBITUX), and Pierre Fabre for its BRAF inhibitor, encorafenib (BRAFTOVI), in key international territories. Pursuant to all of these CTCSAs, we are sponsoring and funding the clinical trial and the partner is providing its drug to us free of charge.
In May 2023, we announced encouraging preliminary data for the ERAS-007 combination with encorafenib and cetuximab (EC) in patients with EC-naïve BRAFm CRC in a poster presentation that we presented at the American Society of Clinical Oncology Annual Meeting in June 2023.
We anticipate a Phase 1b dose expansion data readout for the HERKULES-3 Phase 1b trial for ERAS-007 plus EC in EC-naïve BRAFm CRC patients in the second quarter of 2024.
Our third clinical program is ERAS-801, a CNS-penetrant EGFR inhibitor. In February 2022, we dosed the first patient in our THUNDERBBOLT-1 Phase 1 clinical trial for ERAS-801 in patients with recurrent glioblastoma (GBM). In May 2023, we announced that the FDA granted FTD to ERAS-801 for the treatment of adult patients with GBM with EGFR gene alterations. In June 2023, we announced that the FDA granted Orphan Drug Designation (ODD) to ERAS-801 for the treatment of patients with malignant glioma, which includes GBM. Provided that the product candidate is approved by the FDA for the orphan-designated disease or condition, ODD entitles a party to the potential for seven years of post-approval marketing exclusivity, subject to certain exemptions, and financial incentives such as tax advantages and user fee waivers. In November 2023, we announced that a maximum tolerated dose (MTD) was identified for ERAS-801.
We anticipate presenting Phase 1 monotherapy data from THUNDERBBOLT-1 in 2024.
We are also advancing additional programs targeting key oncogenic drivers in the RAS/MAPK pathway, which we will need to successfully progress through discovery and investigational new drug application (IND)-enabling activities prior to advancing these programs into clinical development, if at all.
We do not own or operate, and currently have no plans to establish, any manufacturing facilities. We rely, and expect to continue to rely, on third parties for the manufacture of our product candidates for preclinical and clinical testing, as well as for commercial manufacture if any of our product candidates obtain marketing approval. We are working with our current manufacturers to ensure that we will be able to scale up our manufacturing capabilities to support our clinical plans. We are also in the process of
locating and qualifying additional manufacturers to build redundancies into our supply chain. In addition, we rely on third parties to package, label, store, and distribute our product candidates, and we intend to continue to rely on third parties with respect to our commercial products if marketing approval is obtained. We believe that this strategy allows us to maintain a more efficient infrastructure by eliminating the need for us to invest in our own manufacturing facilities, equipment, and personnel while also enabling us to focus our expertise and resources on the design and development of our product candidates.
In August 2022, we entered into an Open Market Sale Agreement (the Sale Agreement) with Jefferies LLC (the Agent), pursuant to which we may offer and sell shares of our common stock having an aggregate offering price of up to $200 million from time to time, in “at the market” offerings through the Agent. Sales of the shares of common stock, if any, will be made at prevailing market prices at the time of sale, or as otherwise agreed with the Agent. The Agent will receive a commission from us of up to 3.0% of the gross proceeds of any shares of common stock sold under the Sale Agreement. There have been no shares of our common stock sold under the Sale Agreement as of March 31, 2024.
In December 2022, we completed the 2022 Offering and issued 15,384,616 shares of our common stock at a price to the public of $6.50 per share. Proceeds from the 2022 Offering were $94.9 million, net of underwriting discounts and commissions and offering costs of $5.1 million.
In March 2024, in connection with the 2024 Private Placement, we entered into a stock purchase agreement with the purchasers named therein for the private placement of 21,844,660 shares of our common stock at a price of $2.06 per share. On April 2, 2024, the 2024 Private Placement closed and the net proceeds were $43.7 million, after deducting placement agent fees and estimated expenses of approximately $1.3 million.
Since our inception in 2018, we have devoted substantially all of our resources to organizing and staffing our company, business planning, raising capital, identifying, acquiring, and in-licensing our product candidates, establishing our intellectual property portfolio, conducting research, preclinical studies and clinical trials, establishing arrangements with third parties for the manufacture of our product candidates and related raw materials, and providing general and administrative support for these operations. We do not have any products approved for sale and have not generated any revenue. As of March 31, 2024, we have raised a total of $765.4 million to fund our operations, comprised primarily of gross proceeds from our IPO and 2022 Offering and the sale and issuance of convertible preferred stock. In April 2024, we raised an additional $45.0 million in gross proceeds from the 2024 Private Placement. As of March 31, 2024, we had cash, cash equivalents and short-term marketable securities of $297.7 million, which includes $6.9 million of net proceeds from the prepayment of the 2024 Private Placement. The remaining $36.8 million of net proceeds were received in April 2024.
We have incurred significant operating losses since inception. Our net losses were $35.0 million and $33.2 million for the three months ended March 31, 2024 and 2023, respectively. As of March 31, 2024, we had an accumulated deficit of $641.0 million. We expect our expenses and operating losses will increase substantially for the foreseeable future, particularly if and as we conduct our ongoing and planned clinical trials and preclinical studies; continue our research and development activities; utilize third parties to manufacture our product candidates and related raw materials; hire additional personnel; acquire, in-license, or develop additional product candidates; expand and protect our intellectual property; and incur additional costs associated with being a public company. If we obtain regulatory approval for any of our product candidates, we expect to incur significant commercialization expenses related to product sales, marketing, manufacturing, and distribution. In addition, as our product candidates progress through development and toward commercialization, we will need to make milestone payments to the licensors and other third parties from whom we have in-licensed or acquired our product candidates. Our net losses may fluctuate significantly from quarter-to-quarter and year-to-year, depending on the timing of our clinical trials and preclinical studies and our expenditures on other research and development activities.
Based upon our current operating plans, we believe that our cash, cash equivalents and short-term marketable securities as of March 31, 2024, together with the net proceeds received in April 2024 from the 2024 Private Placement which closed on April 2, 2024, will be sufficient to fund our operations into the second half of 2026. We do not expect to generate any revenues from product sales until we successfully complete development and obtain regulatory approval for one or more of our product candidates, which we expect will take a number of years and may never occur. Accordingly, until such time as we can generate significant revenue from sales of our product candidates, if ever, we expect to finance our cash needs through equity offerings, debt financings or other capital sources, including potential collaborations, licenses, and other similar arrangements. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. Our failure to raise capital or enter into such other arrangements when needed would have a negative impact on our financial condition and could force us to delay, limit, reduce, or terminate our research and development programs or other operations, or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
Our financial condition and results of operations may also be impacted by other factors we may not be able to control, such as geopolitical and economic events. We do not believe that such factors had a material adverse impact on our results of operations during the three months ended March 31, 2024.
Our acquisition and license agreements
We have entered into in-license and acquisition agreements pursuant to which we in-licensed or acquired certain intellectual property rights related to our product candidates and development programs.
For additional information regarding these agreements, see the section titled “Business—Our acquisition and license agreements” in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
Components of results of operations
Revenue
We do not expect to generate any revenue from the sale of products unless and until such time that our product candidates have advanced through clinical development and obtained regulatory approval, if ever. If we fail to complete preclinical and clinical development of product candidates or obtain regulatory approval for them, our ability to generate future revenues, and our results of operations and financial position would be adversely affected.
Operating expenses
Research and development
Research and development expenses consist of external and internal costs associated with our research and development activities, including our discovery and research efforts and the preclinical and clinical development of our product candidates. Research and development costs are expensed as incurred. Our research and development expenses include:
• external costs, including expenses incurred under arrangements with third parties, such as contract research organizations (CROs), contract manufacturing organizations (CMOs), consultants and our scientific advisors; and
• internal costs, including:
• employee-related expenses, including salaries, benefits, and stock-based compensation for those individuals involved in research and development efforts;
• the costs of laboratory supplies and acquiring, developing and manufacturing preclinical study materials; and
• facilities and depreciation, which include direct and allocated expenses for rent of facilities and depreciation.
The following table summarizes our research and development expenses incurred for the following periods (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
2024 |
|
|
2023 |
|
Naporafenib |
|
|
$ |
12,970 |
|
|
|
$ |
4,199 |
|
ERAS-007 |
|
|
|
3,231 |
|
|
|
|
7,112 |
|
Other clinical programs |
|
|
|
3,816 |
|
|
|
|
7,907 |
|
Other discovery and preclinical programs |
|
|
|
8,557 |
|
|
|
|
8,367 |
|
Total research and development expenses |
|
|
$ |
28,574 |
|
|
|
$ |
27,585 |
|
We expect our research and development expenses to increase substantially for the foreseeable future as we continue to conduct our ongoing research and development activities, conduct clinical trials and advance our preclinical research programs toward clinical development, particularly as more of our product candidates move into later stages of development, which typically cost more. The process of conducting clinical trials and preclinical studies necessary to obtain regulatory approval is costly and time-consuming. We may never succeed in achieving marketing approval for any of our product candidates.
The timelines and costs with research and development activities are uncertain, can vary significantly for each product candidate and program and are difficult to predict. We anticipate we will make determinations as to which product candidates and programs to pursue and how much funding to direct to each product candidate and program on an ongoing basis in response to preclinical and clinical results, regulatory developments, ongoing assessments as to each product candidate’s and program’s commercial potential, and our ability to enter into collaborations, licenses, or other similar agreements to the extent we determine the resources or expertise of a third-party would be beneficial for a given product candidate or program. We will need to raise substantial additional capital in the future. In addition, we cannot forecast which product candidates and programs may be subject to future collaborations, licenses, or other agreements, when such arrangements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements.
Our development costs may vary significantly based on factors such as:
• the number and scope of preclinical and IND-enabling studies and clinical trials;
• per patient trial costs;
• the number of trials required for approval;
• the number of sites included in the trials;
• the countries in which the trials are conducted;
• the length of time required to enroll eligible patients;
• the number of patients that participate in the trials;
• the number of doses that patients receive;
• the drop-out or discontinuation rates of patients;
• potential additional safety monitoring requested by regulatory agencies;
• the duration of patient participation in the trials and follow-up;
• the cost and timing of manufacturing our product candidates;
• the phase of development of our product candidates;
• the efficacy and safety profile of our product candidates;
• the timing, receipt and terms of any approvals from applicable regulatory authorities;
• maintaining a continued acceptable safety profile of our products following approval, if any;
• significant and changing government regulation and regulatory guidance;
• the impact of any interruptions to our operations or to those of third parties with whom we work due to geopolitical and economic events; and
• the extent to which we establish additional collaboration, license or other arrangements.
In-process research and development
In-process research and development expenses include rights acquired as part of asset acquisitions or in-licenses to develop and commercialize product candidates. Upfront payments that relate to the acquisition of a new product candidate, as well as pre-commercial milestone payments, are immediately expensed as in-process research and development in the period in which they are incurred, provided that the new product candidate did not also include processes or activities that would constitute a “business” as defined under US generally accepted accounting principles (US GAAP), the product candidate has not achieved regulatory approval for marketing and, absent obtaining such approval, has no established alternative future use.
In-process research and development expenses consist primarily of our upfront payments, milestone payments, and our stock issuances in connection with our acquisition and in-license agreements.
General and administrative
General and administrative expenses consist primarily of employee-related expenses, including salaries, benefits and stock-based compensation, for employees in our finance, accounting, legal, information technology, business development and support functions. Other general and administrative expenses include allocated facility and depreciation related costs not otherwise included in research and development expenses and professional fees for auditing, tax, intellectual property and legal services. Costs related to filing and pursuing patent applications are recognized as general and administrative expenses as incurred since recoverability of such expenditures is uncertain.
We expect our general and administrative expenses will increase substantially for the foreseeable future as we continue to increase our general and administrative headcount to support our continued research and development activities and, if any product candidates receive marketing approval, commercialization activities, as well as to support our operations generally.
Other income (expense), net
Interest income
Interest income consists primarily of interest earned on our cash, cash equivalents and marketable securities.
Results of operations
Comparison of the three months ended March 31, 2024 and 2023
The following table summarizes our results of operations for the three months ended March 31, 2024 and 2023 (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
|
|
|
2024 |
|
|
2023 |
|
|
Change |
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
Research and development |
|
$ |
28,574 |
|
|
$ |
27,585 |
|
|
$ |
989 |
|
General and administrative |
|
|
10,277 |
|
|
|
9,440 |
|
|
|
837 |
|
Total operating expenses |
|
|
38,851 |
|
|
|
37,025 |
|
|
|
1,826 |
|
Loss from operations |
|
|
(38,851 |
) |
|
|
(37,025 |
) |
|
|
(1,826 |
) |
Total other income (expense), net |
|
|
3,834 |
|
|
|
3,826 |
|
|
|
8 |
|
Net loss |
|
$ |
(35,017 |
) |
|
$ |
(33,199 |
) |
|
$ |
(1,818 |
) |
Research and development expenses
Research and development expenses were $28.6 million for the three months ended March 31, 2024 compared to $27.6 million for the three months ended March 31, 2023. The increase of $1.0 million was primarily driven by an increase of $2.4 million in expenses incurred in connection with clinical trials, preclinical studies and discovery activities, partially offset by a decrease of $1.1 million in outsourced services and consulting fees.
General and administrative expenses
General and administrative expenses were $10.3 million for the three months ended March 31, 2024 compared to $9.4 million for the three months ended March 31, 2023. The increase of $0.8 million was primarily driven by increases of $0.7 million in legal fees and $0.6 million in personnel costs, including stock-based compensation expense, partially offset by a decrease of $0.3 million in insurance costs.
Other income (expense), net
Other income (expense), net was $3.8 million for the three months ended March 31, 2024 and 2023.
Liquidity and capital resources
Sources of liquidity
In July 2021, we completed our IPO and issued 21,562,500 shares of our common stock, including the exercise in full by the underwriters of their option to purchase 2,812,500 shares of our common stock, at a price to the public of $16.00 per share. Our aggregate net proceeds from the offering were $317.0 million, net of underwriting discounts and commissions of $24.2 million and offering costs of $3.8 million. Prior to the IPO, we received aggregate gross proceeds of $320.4 million from the sale of shares of our convertible preferred stock.
In August 2022, we entered into the Sale Agreement with the Agent, pursuant to which we may offer and sell shares of our common stock having an aggregate offering price of up to $200 million from time to time, in “at the market” offerings through the Agent. Sales of the shares of common stock, if any, will be made at prevailing market prices at the time of sale, or as otherwise agreed with the Agent. The Agent will receive a commission from us of up to 3.0% of the gross proceeds of any shares of common stock sold under the Sale Agreement. There have been no shares of our common stock sold under the Sale Agreement as of March 31, 2024.
In December 2022, we completed the 2022 Offering and issued 15,384,616 shares of our common stock at a price to the public of $6.50 per share. Proceeds from the 2022 Offering were $94.9 million, net of underwriting discounts and commissions and offering costs of $5.1 million.
In March 2024, in connection with the 2024 Private Placement, the Company entered into a stock purchase agreement with the purchasers named therein for the private placement of 21,844,660 shares of its common stock at a price of $2.06 per share. On April 2, 2024, the 2024 Private Placement closed and the gross proceeds were $45.0 million, before deducting placement agent fees and estimated expenses of approximately $1.3 million. The Company received $6.9 million of the proceeds in March 2024 prior to the close of the 2024 Private Placement.
Future capital requirements
As of March 31, 2024, we had cash, cash equivalents and short-term marketable securities of $297.7 million, which includes $6.9 million of net proceeds from the prepayment of the 2024 Private Placement, which closed on April 2, 2024. The remaining $36.8 million of net proceeds were received in April 2024. Based upon our current operating plans, we believe that our cash, cash equivalents and short-term marketable securities, together with the net proceeds from the 2024 Private Placement, will be sufficient to fund our operations into the second half of 2026. However, our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement that involves risks and uncertainties, and actual results could vary materially. We have based this estimate on assumptions that may prove to be wrong, and we could deplete our capital resources sooner than we expect. Additionally, the process of conducting preclinical studies and testing product candidates in clinical trials is costly, and the timing of progress and expenses in these studies and trials is uncertain.
Our future capital requirements are difficult to forecast and will depend on many factors, including but not limited to:
• the type, number, scope, progress, expansions, results, costs and timing of discovery, preclinical studies and clinical trials of our product candidates that we are pursuing or may choose to pursue in the future, including the costs of any third-party products used in our combination clinical trials that are not covered by such third party or other sources;
• the costs and timing of manufacturing for our product candidates with CMOs, including commercial manufacturing, if any product candidate is approved;
• the costs, timing and outcome of regulatory review of our product candidates;
• the costs of obtaining, maintaining and enforcing our patents and other intellectual property rights;
• our efforts to enhance operational systems and hire additional personnel to satisfy our obligations as a public company, including enhanced internal controls over financial reporting;
• the costs associated with hiring additional personnel, consultants, and CROs as our preclinical and clinical activities increase;
• the timing and amount of the milestone or other payments we must make to the licensors and other third parties from whom we have in-licensed or acquired our product candidates or technologies;
• the costs and timing of establishing or securing sales and marketing capabilities if any product candidate is approved;
• our ability to achieve sufficient market acceptance, coverage and adequate reimbursement from third-party payors and adequate market share and revenue for any approved products;
• patients’ willingness to pay out-of-pocket for any approved products in the absence of coverage and/or adequate reimbursement from third-party payors;
• the terms and timing of establishing and maintaining collaborations, licenses and other similar arrangements;
• any delays and cost increases that result from geopolitical and economic events; and
• costs associated with any products or technologies that we may in-license or acquire.
We have no other committed sources of capital. Until we can generate a sufficient amount of product revenue to finance our cash requirements, if ever, we expect to finance our future cash needs primarily through equity offerings (including through the Sale Agreement), debt financings or other capital sources, including potential collaborations, licenses and other similar arrangements. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interest of our stockholders will be or could be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our common stockholders. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise additional funds through collaborations, licensing, or other similar arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our research and development programs or other operations, or grant rights to develop and market product candidates to third parties that we would otherwise prefer to develop and market ourselves.
Cash flows
The following table shows a summary of our cash flows for the periods presented (in thousands):
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
2024 |
|
|
2023 |
|
Net cash (used in) provided by: |
|
|
|
|
|
|
Operating activities |
|
$ |
(33,251 |
) |
|
$ |
(26,358 |
) |
Investing activities |
|
|
9,971 |
|
|
|
14,714 |
|
Financing activities |
|
|
6,931 |
|
|
|
183 |
|
Net decrease in cash, cash equivalents and restricted cash |
|
$ |
(16,349 |
) |
|
$ |
(11,461 |
) |
Operating activities
Cash used in operating activities was $33.3 million during the three months ended March 31, 2024, primarily resulting from a net loss of $35.0 million, changes in operating assets and liabilities of $3.7 million and accretion on marketable securities of $2.3 million, partially reduced by stock-based compensation expense of $6.8 million and depreciation and amortization expense of $1.0 million. Net cash used by changes in operating assets and liabilities consisted primarily of a decrease in accrued expenses and other current liabilities of $3.6 million, an increase in prepaid expenses and other current and long-term assets of $0.5 million, and a decrease in operating lease assets and liabilities, net of $0.2 million, offset by an increase in accounts payable of $0.6 million.
Cash used in operating activities was $26.4 million during the three months ended March 31, 2023, primarily resulting from a net loss of $33.2 million and accretion on marketable securities of $1.0 million, partially reduced by stock-based compensation expense of $6.8 million and depreciation and amortization expense of $0.9 million. Net cash provided by changes in operating assets and liabilities consisted primarily of an increase in operating lease assets and liabilities, net of $3.3 million primarily due to the receipt of $2.3 million in reimbursement from our landlord for tenant improvements, an increase in accounts payable of $0.4 million, and a decrease in prepaid expenses and other current and long-term assets of $0.7 million, offset by a decrease in accrued expenses and other current and long-term liabilities of $4.4 million.
Investing activities
Net cash provided by investing activities was $10.0 million during the three months ended March 31, 2024 as compared to $14.7 million during the three months ended March 31, 2023. The decrease in cash provided by investing activities of $4.7 million was primarily the result of an increase in purchases of marketable securities of $19.7 million and a decrease in maturities of marketable securities of $6.2 million, partially offset by decreases in in-process research and development of $20.0 million and purchases of property and equipment of $1.2 million.
Financing activities
Net cash provided by financing activities was $6.9 million during the three months ended March 31, 2024 as compared to $0.2 million during the three months ended March 31, 2023. During the three months ended March 31, 2024, we received $6.9 million of proceeds for the prepayment of the 2024 Private Placement which closed in April 2024. During the three months ended March 31, 2023, we received $0.2 million from the exercise of stock options.
Contractual obligations and commitments
As of March 31, 2024, there have been no material changes outside the ordinary course of our business to the contractual obligations we reported in “Management’s discussion and analysis of financial condition and results of operations – Cash requirements due to contractual obligations and other commitments,” included in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
Critical accounting policies and estimates
This management discussion and analysis of financial condition and results of operations is based on our condensed consolidated financial statements, which have been prepared in accordance with US GAAP. The preparation of these condensed consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenue and expenses. On an ongoing basis, we evaluate these estimates and judgments. We base our estimates on historical experience and on various assumptions that we believe to be reasonable under the circumstances. These estimates and assumptions form the basis for making judgments about the carrying values of assets and liabilities and the recording of revenue and expenses that are not readily apparent from other sources. Actual results may differ materially from these estimates. As of March 31, 2024, there have been no material changes to our critical accounting policies and estimates from those disclosed in “Management’s discussion and analysis of financial condition and results of operations – Critical accounting policies and estimates,” included in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
Recently issued and adopted accounting pronouncements
See Note 2 to our condensed consolidated financial statements included elsewhere in this Quarterly Report on Form 10-Q for recently issued and adopted accounting pronouncements.
Emerging growth company and smaller reporting company status
As an emerging growth company under the Jumpstart Our Business Startups Act of 2012 (the JOBS Act), we can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected to avail ourselves of this exemption from new or revised accounting standards and, therefore, our condensed consolidated financial statements may not be comparable to companies that comply with new or revised accounting pronouncements as of public company effective dates. We also intend to rely on other exemptions provided by the JOBS Act, including without limitation, not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act of 2002 (Sarbanes-Oxley).
We will remain an emerging growth company until the earliest of (i) the last day of the fiscal year following the fifth anniversary of the consummation of our IPO; (ii) the last day of the fiscal year in which we have total annual gross revenue of at least $1.235 billion; (iii) the last day of the fiscal year in which we are deemed to be a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock held by non-affiliates exceeded $700.0 million as of the last business day of the second fiscal quarter of such year; or (iv) the date on which we have issued more than $1.0 billion in nonconvertible debt securities during the prior three-year period.
We are also a smaller reporting company as defined in the Exchange Act. We may continue to be a smaller reporting company even after we are no longer an emerging growth company. We may take advantage of certain of the scaled disclosures available to smaller reporting companies and will be able to take advantage of these scaled disclosures for so long as our voting and non-voting common stock held by non-affiliates is less than $250.0 million measured on the last business day of our second fiscal quarter, or our annual revenue is less than $100.0 million during the most recently completed fiscal year and our voting and non-voting common stock held by non-affiliates is less than $700.0 million measured on the last business day of our second fiscal quarter.
Item 3. Quantitative and Qualitative Disclosures About Market Risk.
As of March 31, 2024, there have been no material changes surrounding our market risk, including interest rate risk, foreign currency exchange risk, and inflation risk, from the discussion provided in “Management’s Discussion and Analysis of Financial Condition and Results of Operations – Quantitative and Qualitative Disclosures About Market Risk” in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
Item 4. Controls and Procedures.
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our periodic and current reports that we file with the SEC is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms, and that such information is accumulated and communicated to our management, including our principal executive officer and principal financial officer, as appropriate, to allow timely decisions regarding required disclosure. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable and not absolute assurance of achieving the desired control objectives. In reaching a reasonable level of assurance, management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures. In addition, the design of any system of controls also is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions; over time, controls may become inadequate because of changes in conditions, or the degree of compliance with policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
Our management, with the participation of our principal executive officer and our principal financial officer, evaluated, as of the end of the period covered by this Quarterly Report on Form 10-Q, the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act). Based on that evaluation, our principal executive officer and principal financial officer have concluded that, as of March 31, 2024, our disclosure controls and procedures were effective at the reasonable assurance level.
Changes in Internal Control Over Financial Reporting
There have been no changes in our internal control over financial reporting during the quarter ended March 31, 2024 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
PART II—OTHER INFORMATION
Item 1. Legal Proceedings.
We are not currently a party to any material proceedings. From time to time, we may become involved in legal proceedings or be subject to claims arising in the ordinary course of our business. Regardless of outcome, such proceedings or claims can have an adverse impact on us because of defense and settlement costs, diversion of resources and other factors, and there can be no assurances that favorable outcomes will be obtained.
Item 1A. Risk Factors.
There have been no material changes to the risk factors set forth in Part I, Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
Item 2. Unregistered Sales of Equity Securities and Use of Proceeds.
Unregistered Sales of Equity Securities
None.
Use of Proceeds
On July 15, 2021, the SEC declared effective our registration statement on Form S-1 (File No. 333-257436), as amended, filed in connection with our IPO. Our IPO closed on July 20, 2021, and we issued and sold 21,562,500 shares of our common stock at a price to the public of $16.00 per share, which included the exercise in full of the underwriters’ option to purchase additional shares. We received gross proceeds from our IPO of $345.0 million, before deducting underwriting discounts and commissions of $24.2 million and offering costs of $3.8 million. The managing underwriters of the offering were J.P. Morgan Securities LLC, Morgan Stanley & Co. LLC, BofA Securities, Inc., Evercore Group L.L.C. and Guggenheim Securities, LLC. No offering costs were paid or are payable, directly or indirectly, to our directors or officers, to persons owning 10% or more of any class of our equity securities or to any of our affiliates.
As of March 31, 2024, we have used all of the proceeds from our IPO for general corporate purposes, including to fund the research and development of naporafenib, ERAS-007, ERAS-801 and our other RAS/MAPK pathway-focused pipeline programs. There has been no material change in the planned use of such proceeds from that described in the prospectus for our IPO.
Item 3. Defaults Upon Senior Securities.
Not applicable.
Item 4. Mine Safety Disclosures.
Not applicable.
Item 5. Other Information.
On January 10, 2024, David M. Chacko, M.D., our Chief Financial Officer and Chief Business Officer, adopted a Rule 10b5-1 trading arrangement that is intended to satisfy the affirmative defense of Rule 10b5-1(c) for the sale of up to 260,000 shares of our common stock until December 31, 2025. During the three months ended March 31, 2024, no other officers (as defined in Rule 16a–1(f)) or directors adopted or terminated any Rule 10b5-1 trading arrangement or non-Rule 10b5-1 trading arrangement, as each such term is defined in Item 408 of Regulation S-K.
Item 6. Exhibits.
|
|
|
|
|
|
|
|
|
|
|
Exhibit Number |
|
Exhibit Description |
|
Incorporated by Reference |
|
Filed Herewith |
|
|
|
|
Form |
|
Date |
|
Number |
|
|
3.1 |
|
Amended and Restated Certificate of Incorporation of Erasca, Inc. |
|
8-K |
|
7/20/2021 |
|
3.1 |
|
|
3.2 |
|
Amended and Restated Bylaws of Erasca, Inc. |
|
8-K |
|
7/20/2021 |
|
3.2 |
|
|
4.1 |
|
Specimen stock certificate evidencing the shares of common stock |
|
S-1 |
|
6/25/2021 |
|
4.1 |
|
|
4.2 |
|
Amended and Restated Stockholders Agreement, dated April 15, 2020, by and among the Registrant and certain of its stockholders |
|
S-1 |
|
6/25/2021 |
|
4.2 |
|
|
10.1 |
|
Stock Purchase Agreement, dated March 27, 2024, by and between Erasca, Inc. and each of the purchasers party thereto |
|
8-K |
|
3/28/2024 |
|
10.1 |
|
|
31.1 |
|
Certification of Chief Executive Officer of Erasca, Inc., as required by Rule 13a-14(a) or Rule 15d-14(a), as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|
|
|
|
|
|
|
X |
31.2 |
|
Certification of Chief Financial Officer of Erasca, Inc., as required by Rule 13a-14(a) or Rule 15d-14(a), as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
|
|
|
|
|
|
|
X |
32.1* |
|
Certification of Chief Executive Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
|
|
|
|
X |
32.2* |
|
Certification of Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
|
|
|
|
|
|
|
X |
101.INS |
|
Inline XBRL Instance Document – the instance document does not appear in the Interactive Data File because XBRL tags are embedded within the Inline XBRL document. |
|
|
|
|
|
|
|
X |
101.SCH |
|
Inline XBRL Taxonomy Extension Schema Document |
|
|
|
|
|
|
|
X |
104 |
|
Cover Page Interactive Data File (embedded within the Inline XBRL document) |
|
|
|
|
|
|
|
X |
* This certification is deemed not filed for purpose of section 18 of the Exchange Act or otherwise subject to the liability of that section, nor shall it be deemed incorporated by reference into any filing under the Securities Act or the Exchange Act.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
|
|
|
|
|
|
Erasca, Inc. |
|
|
|
|
Date: May 8, 2024 |
|
By: |
/s/ Jonathan E. Lim, M.D. |
|
|
|
Jonathan E. Lim, M.D. |
|
|
|
Chairman, Chief Executive Officer and Co-Founder |
|
|
|
(Principal Executive Officer) |
|
|
|
|
Date: May 8, 2024 |
|
By: |
/s/ David M. Chacko, M.D. |
|
|
|
David M. Chacko, M.D. |
|
|
|
Chief Financial Officer and Chief Business Officer |
|
|
|
(Principal Financial and Accounting Officer) |
CERTIFICATION
I, Jonathan E. Lim, M.D., certify that:
1.I have reviewed this Quarterly Report on Form 10-Q of Erasca, Inc.;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) for the registrant and have:
(a)Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b)Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c)Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
(d)Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5.The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
(a)All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
(b)Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
|
|
|
Date: May 8, 2024 |
By: |
/s/ Jonathan E. Lim, M.D. |
|
|
Jonathan E. Lim, M.D. |
|
|
Chairman, Chief Executive Officer and Co-Founder (Principal Executive Officer) |
CERTIFICATION
I, David M. Chacko, M.D., certify that:
1.I have reviewed this Quarterly Report on Form 10-Q of Erasca, Inc.;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) for the registrant and have:
(a)Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b)Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c)Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
(d)Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5.The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
(a)All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
(b)Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
|
|
|
Date: May 8, 2024 |
By: |
/s/ David M. Chacko, M.D. |
|
|
David M. Chacko, M.D. |
|
|
Chief Financial Officer and Chief Business Officer (Principal Financial and Accounting Officer) |
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350, AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
In connection with the Quarterly Report on Form 10-Q of Erasca, Inc. (the “Company”) for the period ended March 31, 2024 as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I certify, pursuant to 18 U.S.C. § 1350, as adopted pursuant to § 906 of the Sarbanes-Oxley Act of 2002, that, to the best of my knowledge:
(1)The Report fully complies with the requirements of section 13(a) or 15(d) of the Securities Exchange Act of 1934; and
(2)The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
|
|
|
Date: May 8, 2024 |
By: |
/s/ Jonathan E. Lim, M.D. |
|
|
Jonathan E. Lim, M.D. |
|
|
Chairman, Chief Executive Officer and Co-Founder (Principal Executive Officer) |
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350, AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
In connection with the Quarterly Report on Form 10-Q of Erasca, Inc. (the “Company”) for the period ended March 31, 2024 as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I certify, pursuant to 18 U.S.C. § 1350, as adopted pursuant to § 906 of the Sarbanes-Oxley Act of 2002, that, to the best of my knowledge:
(1)The Report fully complies with the requirements of section 13(a) or 15(d) of the Securities Exchange Act of 1934; and
(2)The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
|
|
|
Date: May 8, 2024 |
By: |
/s/ David M. Chacko, M.D. |
|
|
David M. Chacko, M.D. |
|
|
Chief Financial Officer and Chief Business Officer (Principal Financial and Accounting Officer) |
v3.24.1.u1
Document and Entity Information - shares
|
3 Months Ended |
|
Mar. 31, 2024 |
May 02, 2024 |
| Cover [Abstract] |
|
|
| Document Type |
10-Q
|
|
| Amendment Flag |
false
|
|
| Document Period End Date |
Mar. 31, 2024
|
|
| Document Fiscal Year Focus |
2024
|
|
| Document Fiscal Period Focus |
Q1
|
|
| Entity Registrant Name |
ERASCA, INC.
|
|
| Entity Central Index Key |
0001761918
|
|
| Current Fiscal Year End Date |
--12-31
|
|
| Entity Filer Category |
Non-accelerated Filer
|
|
| Entity File Number |
001-40602
|
|
| Entity Incorporation, State or Country Code |
DE
|
|
| Entity Tax Identification Number |
83-1217027
|
|
| Entity Address, Address Line One |
3115 Merryfield Row
|
|
| Entity Address, Address Line Two |
Suite 300
|
|
| Entity Address, City or Town |
San Diego
|
|
| Entity Address, State or Province |
CA
|
|
| Entity Address, Postal Zip Code |
92121
|
|
| City Area Code |
858
|
|
| Local Phone Number |
465-6511
|
|
| Entity Current Reporting Status |
Yes
|
|
| Entity Small Business |
true
|
|
| Entity Emerging Growth Company |
true
|
|
| Entity Ex Transition Period |
false
|
|
| Entity Shell Company |
false
|
|
| Entity Interactive Data Current |
Yes
|
|
| Entity Common Stock, Shares Outstanding |
|
173,358,966
|
| Document Quarterly Report |
true
|
|
| Document Transition Report |
false
|
|
| Title of 12(b) Security |
Common Stock, $0.0001 par value per share
|
|
| Trading Symbol |
ERAS
|
|
| Security Exchange Name |
NASDAQ
|
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionEnd date of current fiscal year in the format --MM-DD.
| Name: |
dei_CurrentFiscalYearEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:gMonthDayItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFiscal period values are FY, Q1, Q2, and Q3. 1st, 2nd and 3rd quarter 10-Q or 10-QT statements have value Q1, Q2, and Q3 respectively, with 10-K, 10-KT or other fiscal year statements having FY.
| Name: |
dei_DocumentFiscalPeriodFocus |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fiscalPeriodItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThis is focus fiscal year of the document report in YYYY format. For a 2006 annual report, which may also provide financial information from prior periods, fiscal 2006 should be given as the fiscal year focus. Example: 2006.
| Name: |
dei_DocumentFiscalYearFocus |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:gYearItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true only for a form used as an quarterly report. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Form 10-Q
-Number 240
-Section 308
-Subsection a
| Name: |
dei_DocumentQuarterlyReport |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true only for a form used as a transition report. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Forms 10-K, 10-Q, 20-F
-Number 240
-Section 13
-Subsection a-1
| Name: |
dei_DocumentTransitionReport |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate number of shares or other units outstanding of each of registrant's classes of capital or common stock or other ownership interests, if and as stated on cover of related periodic report. Where multiple classes or units exist define each class/interest by adding class of stock items such as Common Class A [Member], Common Class B [Member] or Partnership Interest [Member] onto the Instrument [Domain] of the Entity Listings, Instrument.
| Name: |
dei_EntityCommonStockSharesOutstanding |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionIndicate 'Yes' or 'No' whether registrants (1) have filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that registrants were required to file such reports), and (2) have been subject to such filing requirements for the past 90 days. This information should be based on the registrant's current or most recent filing containing the related disclosure.
| Name: |
dei_EntityCurrentReportingStatus |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:yesNoItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate whether the registrant is one of the following: Large Accelerated Filer, Accelerated Filer, Non-accelerated Filer. Definitions of these categories are stated in Rule 12b-2 of the Exchange Act. This information should be based on the registrant's current or most recent filing containing the related disclosure. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityFilerCategory |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:filerCategoryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-T
-Number 232
-Section 405
| Name: |
dei_EntityInteractiveDataCurrent |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:yesNoItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the registrant is a shell company as defined in Rule 12b-2 of the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityShellCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicates that the company is a Smaller Reporting Company (SRC). Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntitySmallBusiness |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Condensed Consolidated Balance Sheets (Unaudited) - USD ($)
|
Mar. 31, 2024 |
Dec. 31, 2023 |
| Current assets: |
|
|
| Cash and cash equivalents |
$ 76,726,000
|
$ 93,075,000
|
| Short-term marketable securities |
220,959,000
|
219,275,000
|
| Prepaid expenses and other current assets |
8,938,000
|
8,326,000
|
| Total current assets |
306,623,000
|
320,676,000
|
| Long-term marketable securities |
|
9,642,000
|
| Property and equipment, net |
21,358,000
|
22,327,000
|
| Operating lease assets |
37,177,000
|
37,861,000
|
| Restricted cash |
408,000
|
408,000
|
| Other assets |
4,456,000
|
4,383,000
|
| Total assets |
370,022,000
|
395,297,000
|
| Current liabilities: |
|
|
| Accounts payable |
2,633,000
|
2,000,000
|
| Accrued expenses and other current liabilities |
16,577,000
|
20,186,000
|
| Proceeds from prepayment of private placement |
6,907,000
|
|
| Operating lease liabilities |
4,126,000
|
3,970,000
|
| Total current liabilities |
30,243,000
|
26,156,000
|
| Operating lease liabilities, net of current portion |
50,811,000
|
51,889,000
|
| Other liabilities |
559,000
|
566,000
|
| Total liabilities |
81,613,000
|
78,611,000
|
| Commitments and contingencies (Note 11) |
|
|
| Stockholders' equity: |
|
|
| Preferred stock, $0.0001 par value; 80,000,000 shares authorized at March 31, 2024 and December 31, 2023; no shares issued and outstanding at March 31, 2024 and December 31, 2023 |
|
|
| Common stock, $0.0001 par value; 800,000,000 shares authorized at March 31, 2024 and December 31, 2023; 151,497,138 and 151,462,103 shares issued at March 31, 2024 and December 31, 2023, respectively; 151,249,219 and 151,090,227 shares outstanding at March 31, 2024 and December 31, 2023, respectively |
15,000
|
15,000
|
| Additional paid-in capital |
929,634,000
|
922,607,000
|
| Accumulated other comprehensive (loss) income |
(210,000)
|
77,000
|
| Accumulated deficit |
(641,030,000)
|
(606,013,000)
|
| Total stockholders' equity |
288,409,000
|
316,686,000
|
| Total liabilities and stockholders' equity |
$ 370,022,000
|
$ 395,297,000
|
| X |
- DefinitionLiability from prepayment of private placement.
| Name: |
eras_LiabilityFromPrepaymentOfPrivatePlacementCurrent |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionCarrying value as of the balance sheet date of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer). Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AccountsPayableCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of expenses incurred but not yet paid nor invoiced, and liabilities classified as other.
| Name: |
us-gaap_AccruedLiabilitiesAndOtherLiabilities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount, after tax, of accumulated increase (decrease) in equity from transaction and other event and circumstance from nonowner source. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 220
-SubTopic 10
-Section 45
-Paragraph 14A
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-14A
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-11
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (g)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (h)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30)(a)(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(23)(a)(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 220
-SubTopic 10
-Section 45
-Paragraph 14
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-14
| Name: |
us-gaap_AccumulatedOtherComprehensiveIncomeLossNetOfTax |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionValue received from shareholders in common stock-related transactions that are in excess of par value or stated value and amounts received from other stock-related transactions. Includes only common stock transactions (excludes preferred stock transactions). May be called contributed capital, capital in excess of par, capital surplus, or paid-in capital. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30)(a)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AdditionalPaidInCapitalCommonStock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all assets that are recognized. Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 6: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(12))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 23: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 26: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(11))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
| Name: |
us-gaap_Assets |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all assets that are expected to be realized in cash, sold, or consumed within one year (or the normal operating cycle, if longer). Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 6: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 11: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
| Name: |
us-gaap_AssetsCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_AssetsCurrentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-4
| Name: |
us-gaap_CashAndCashEquivalentsAtCarryingValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionRepresents the caption on the face of the balance sheet to indicate that the entity has entered into (1) purchase or supply arrangements that will require expending a portion of its resources to meet the terms thereof, and (2) is exposed to potential losses or, less frequently, gains, arising from (a) possible claims against a company's resources due to future performance under contract terms, and (b) possible losses or likely gains from uncertainties that will ultimately be resolved when one or more future events that are deemed likely to occur do occur or fail to occur. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(19))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(15))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 942
-SubTopic 210
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03.17)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.25)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommitmentsAndContingencies |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate par or stated value of issued nonredeemable common stock (or common stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable common shares, par value and other disclosure concepts are in another section within stockholders' equity. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all liabilities that are recognized. Liabilities are probable future sacrifices of economic benefits arising from present obligations of an entity to transfer assets or provide services to other entities in the future. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(14))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 21: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 22: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19-26)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_Liabilities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of liabilities and equity items, including the portion of equity attributable to noncontrolling interests, if any. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 4: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(32))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_LiabilitiesAndStockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionTotal obligations incurred as part of normal operations that are expected to be paid during the following twelve months or within one business cycle, if longer. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-25
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (bb)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-5
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 19: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481404/852-10-50-7
Reference 21: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.21)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_LiabilitiesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_LiabilitiesCurrentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of investment in marketable security, classified as current. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_MarketableSecuritiesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of investment in marketable security, classified as noncurrent. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_MarketableSecuritiesNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionPresent value of lessee's discounted obligation for lease payments from operating lease, classified as current. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseLiabilityCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionPresent value of lessee's discounted obligation for lease payments from operating lease, classified as noncurrent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseLiabilityNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's right to use underlying asset under operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseRightOfUseAsset |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of noncurrent assets classified as other. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_OtherAssetsNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of liabilities classified as other, due after one year or the normal operating cycle, if longer. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.24)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_OtherLiabilitiesNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate par or stated value of issued nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable preferred shares, par value and other disclosure concepts are in another section within stockholders' equity. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(21))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PreferredStockValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of asset related to consideration paid in advance for costs that provide economic benefits in future periods, and amount of other assets that are expected to be realized or consumed within one year or the normal operating cycle, if longer. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PrepaidExpenseAndOtherAssetsCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount after accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business to produce goods and services and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 360
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480842/942-360-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of cash restricted as to withdrawal or usage, classified as noncurrent. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 5
-SubTopic 210
-Topic 954
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480632/954-210-45-5
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
| Name: |
us-gaap_RestrictedCashNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of accumulated undistributed earnings (deficit). Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (g)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (h)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-11
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(23)(a)(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30)(a)(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_RetainedEarningsAccumulatedDeficit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of equity (deficit) attributable to parent. Excludes temporary equity and equity attributable to noncontrolling interest. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(19))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(6))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 8: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 9: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 11: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 12: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(31))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 13: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 14: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 310
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SAB Topic 4.E)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480418/310-10-S99-2
| Name: |
us-gaap_StockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_StockholdersEquityAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Condensed Consolidated Balance Sheets (Unaudited) (Parenthetical) - $ / shares
|
Mar. 31, 2024 |
Dec. 31, 2023 |
| Preferred stock, par value |
$ 0.0001
|
$ 0.0001
|
| Preferred stock, shares authorized |
80,000,000
|
80,000,000
|
| Preferred stock, shares issued |
0
|
0
|
| Preferred stock, shares outstanding |
0
|
0
|
| Common stock, par value |
$ 0.0001
|
$ 0.0001
|
| Common stock, shares authorized |
800,000,000
|
800,000,000
|
| Common stock, shares issued |
151,497,138
|
151,462,103
|
| Common stock, shares outstanding |
151,249,219
|
151,090,227
|
| X |
- DefinitionFace amount or stated value per share of common stock. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe maximum number of common shares permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionTotal number of common shares of an entity that have been sold or granted to shareholders (includes common shares that were issued, repurchased and remain in the treasury). These shares represent capital invested by the firm's shareholders and owners, and may be all or only a portion of the number of shares authorized. Shares issued include shares outstanding and shares held in the treasury. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares of common stock outstanding. Common stock represent the ownership interest in a corporation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionFace amount or stated value per share of preferred stock nonredeemable or redeemable solely at the option of the issuer. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PreferredStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe maximum number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PreferredStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionTotal number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) issued to shareholders (includes related preferred shares that were issued, repurchased, and remain in the treasury). May be all or portion of the number of preferred shares authorized. Excludes preferred shares that are classified as debt. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-13
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PreferredStockSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate share number for all nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer) held by stockholders. Does not include preferred shares that have been repurchased. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(16)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 4: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PreferredStockSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
v3.24.1.u1
Condensed Consolidated Statements of Operations and Comprehensive Loss (Unaudited) - USD ($)
$ in Thousands |
3 Months Ended |
Mar. 31, 2024 |
Mar. 31, 2023 |
| Operating expenses: |
|
|
| Research and development |
$ 28,574
|
$ 27,585
|
| General and administrative |
10,277
|
9,440
|
| Total operating expenses |
38,851
|
37,025
|
| Loss from operations |
(38,851)
|
(37,025)
|
| Other income (expense) |
|
|
| Interest income |
3,900
|
3,877
|
| Other expense, net |
(66)
|
(51)
|
| Total other income (expense), net |
3,834
|
3,826
|
| Net loss |
$ (35,017)
|
$ (33,199)
|
| Net loss per share, basic |
$ (0.23)
|
$ (0.22)
|
| Net loss per share, diluted |
$ (0.23)
|
$ (0.22)
|
| Weighted-average shares of common stock used in computing net loss per share, basic |
151,161,741
|
149,504,216
|
| Weighted-average shares of common stock used in computing net loss per share, diluted |
151,161,741
|
149,504,216
|
| Other comprehensive income (loss): |
|
|
| Unrealized (loss) gain on marketable securities, net |
$ (287)
|
$ 527
|
| Comprehensive loss |
$ (35,304)
|
$ (32,672)
|
| X |
- DefinitionAmount after tax of increase (decrease) in equity from transactions and other events and circumstances from net income and other comprehensive income, attributable to parent entity. Excludes changes in equity resulting from investments by owners and distributions to owners. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(24))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(26))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 220
-SubTopic 10
-Section 45
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-5
| Name: |
us-gaap_ComprehensiveIncomeNetOfTax |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of net income (loss) for the period per each share of common stock or unit outstanding during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 15
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-15
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (e)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-7
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-10
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(27))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/exampleRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 52
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-52
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-7
| Name: |
us-gaap_EarningsPerShareBasic |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of net income (loss) for the period available to each share of common stock or common unit outstanding during the reporting period and to each share or unit that would have been outstanding assuming the issuance of common shares or units for all dilutive potential common shares or units outstanding during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 15
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-15
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (e)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-7
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(27))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 15: http://www.xbrl.org/2003/role/exampleRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 52
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-52
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-7
| Name: |
us-gaap_EarningsPerShareDiluted |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate total of expenses of managing and administering the affairs of an entity, including affiliates of the reporting entity, which are not directly or indirectly associated with the manufacture, sale or creation of a product or product line. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_GeneralAndAdministrativeExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe net amount of nonoperating interest income (expense).
| Name: |
us-gaap_InterestIncomeExpenseNonoperatingNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate amount of income or expense from ancillary business-related activities (that is to say, excluding major activities considered part of the normal operations of the business). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.7)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_NonoperatingIncomeExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NonoperatingIncomeExpenseAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionGenerally recurring costs associated with normal operations except for the portion of these expenses which can be clearly related to production and included in cost of sales or services. Includes selling, general and administrative expense.
| Name: |
us-gaap_OperatingExpenses |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_OperatingExpensesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe net result for the period of deducting operating expenses from operating revenues. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 4: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
| Name: |
us-gaap_OperatingIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount, after tax and adjustment, of unrealized gain (loss) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale) and unrealized gain (loss) on investment in debt security measured at amortized cost (held-to-maturity) from transfer to available-for-sale. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10A
-Subparagraph (e)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-10A
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10A
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-10A
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-11
| Name: |
us-gaap_OtherComprehensiveIncomeLossAvailableForSaleSecuritiesAdjustmentNetOfTax |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_OtherComprehensiveIncomeLossTaxAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of expense related to nonoperating activities, classified as other. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.9)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
| Name: |
us-gaap_OtherNonoperatingExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe costs incurred in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process whether intended for sale or the entity's use, during the reporting period charged to research and development projects, excluding in-process research and development acquired in a business combination consummated during the period. Excludes software research and development, which has a separate concept. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 730
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482916/730-10-50-1
| Name: |
us-gaap_ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe average number of shares or units issued and outstanding that are used in calculating diluted EPS or earnings per unit (EPU), determined based on the timing of issuance of shares or units in the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 16
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-16
| Name: |
us-gaap_WeightedAverageNumberOfDilutedSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of [basic] shares or units, after adjustment for contingently issuable shares or units and other shares or units not deemed outstanding, determined by relating the portion of time within a reporting period that common shares or units have been outstanding to the total time in that period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-10
| Name: |
us-gaap_WeightedAverageNumberOfSharesOutstandingBasic |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Condensed Consolidated Statements of Stockholders' Equity (Unaudited) - USD ($)
$ in Thousands |
Total |
Common Stock |
Additional Paid-in Capital |
Accumulated Other Comprehensive (Loss) Income |
Accumulated Deficit |
| Beginning balance, shares at Dec. 31, 2022 |
|
150,448,363
|
|
|
|
| Beginning balance at Dec. 31, 2022 |
$ 411,853
|
$ 15
|
$ 893,850
|
$ (1,041)
|
$ (480,971)
|
| Exercise of stock options, shares |
|
199,344
|
|
|
|
| Exercise of stock options |
183
|
|
183
|
|
|
| Vesting of early exercised stock options |
243
|
|
243
|
|
|
| Stock-based compensation expense |
6,845
|
|
6,845
|
|
|
| Net Income (Loss) |
(33,199)
|
|
|
|
(33,199)
|
| Unrealized gain (loss) on marketable securities, net |
527
|
|
|
527
|
|
| Ending balance, shares at Mar. 31, 2023 |
|
150,647,707
|
|
|
|
| Ending balance at Mar. 31, 2023 |
$ 386,452
|
$ 15
|
901,121
|
(514)
|
(514,170)
|
| Beginning balance, shares at Dec. 31, 2023 |
151,462,103
|
151,462,103
|
|
|
|
| Beginning balance at Dec. 31, 2023 |
$ 316,686
|
$ 15
|
922,607
|
77
|
(606,013)
|
| Exercise of stock options, shares |
|
35,035
|
|
|
|
| Exercise of stock options |
24
|
|
24
|
|
|
| Vesting of early exercised stock options |
155
|
|
155
|
|
|
| Stock-based compensation expense |
6,848
|
|
6,848
|
|
|
| Net Income (Loss) |
(35,017)
|
|
|
|
(35,017)
|
| Unrealized gain (loss) on marketable securities, net |
$ (287)
|
|
|
(287)
|
|
| Ending balance, shares at Mar. 31, 2024 |
151,497,138
|
151,497,138
|
|
|
|
| Ending balance at Mar. 31, 2024 |
$ 288,409
|
$ 15
|
$ 929,634
|
$ (210)
|
$ (641,030)
|
| X |
- DefinitionStock issued during period value, vesting of early exercised stock options.
| Name: |
eras_StockIssuedDuringPeriodValueVestingOfEarlyExercisedStockOptions |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of increase to additional paid-in capital (APIC) for recognition of cost for award under share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 20
-Section 55
-Paragraph 13
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481089/718-20-55-13
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 20
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481089/718-20-55-12
| Name: |
us-gaap_AdjustmentsToAdditionalPaidInCapitalSharebasedCompensationRequisiteServicePeriodRecognitionValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionTotal number of common shares of an entity that have been sold or granted to shareholders (includes common shares that were issued, repurchased and remain in the treasury). These shares represent capital invested by the firm's shareholders and owners, and may be all or only a portion of the number of shares authorized. Shares issued include shares outstanding and shares held in the treasury. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount, after tax and before adjustment, of unrealized holding gain (loss) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale). Excludes unrealized gain (loss) on investment in debt security measured at amortized cost (held-to-maturity) from transfer to available-for-sale. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-9
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10A
-Subparagraph (e)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-10A
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-11
| Name: |
us-gaap_OtherComprehensiveIncomeUnrealizedHoldingGainLossOnSecuritiesArisingDuringPeriodNetOfTax |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of share options (or share units) exercised during the current period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesStockOptionsExercised |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionValue of stock issued as a result of the exercise of stock options. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29-31)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodValueStockOptionsExercised |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of equity (deficit) attributable to parent. Excludes temporary equity and equity attributable to noncontrolling interest. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 12
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-12
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(19))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.6-05(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-2
Reference 5: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(6))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(7))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 8: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 9: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 10: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 11: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 12: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(31))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 13: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 14: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 310
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SAB Topic 4.E)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480418/310-10-S99-2
| Name: |
us-gaap_StockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
Condensed Consolidated Statements of Cash Flows (Unaudited) - USD ($)
|
3 Months Ended |
Mar. 31, 2024 |
Mar. 31, 2023 |
| Cash flows from operating activities: |
|
|
| Net loss |
$ (35,017,000)
|
$ (33,199,000)
|
| Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
| Depreciation and amortization |
998,000
|
894,000
|
| Stock-based compensation expense |
6,848,000
|
6,845,000
|
| Accretion on marketable securities, net |
(2,342,000)
|
(975,000)
|
| Changes in operating assets and liabilities: |
|
|
| Prepaid expenses and other current and long-term assets |
(507,000)
|
670,000
|
| Accounts payable |
633,000
|
446,000
|
| Accrued expenses and other current and long-term liabilities |
(3,626,000)
|
(4,363,000)
|
| Operating lease assets and liabilities, net |
(238,000)
|
3,324,000
|
| Net cash used in operating activities |
(33,251,000)
|
(26,358,000)
|
| Cash flows from investing activities: |
|
|
| Purchases of marketable securities |
(43,987,000)
|
(24,280,000)
|
| Maturities of marketable securities |
54,000,000
|
60,200,000
|
| In-process research and development |
|
(20,000,000)
|
| Purchases of property and equipment |
(42,000)
|
(1,206,000)
|
| Net cash provided by investing activities |
9,971,000
|
14,714,000
|
| Cash flows from financing activities: |
|
|
| Proceeds from prepayment of private placement |
6,907,000
|
|
| Proceeds from the exercise of stock options |
24,000
|
183,000
|
| Net cash provided by financing activities |
6,931,000
|
183,000
|
| Net decrease in cash, cash equivalents and restricted cash |
(16,349,000)
|
(11,461,000)
|
| Cash, cash equivalents and restricted cash at beginning of the period |
93,483,000
|
284,625,000
|
| Cash, cash equivalents and restricted cash at end of the period |
77,134,000
|
273,164,000
|
| Supplemental disclosure of noncash investing and financing activities: |
|
|
| Amounts accrued for purchases of property and equipment |
|
38,000
|
| Amounts accrued for offering costs |
178,000
|
|
| Vesting of early exercised options |
$ 155,000
|
$ 243,000
|
| X |
- DefinitionAmounts accrued for offering costs
| Name: |
eras_AmountsAccruedForOfferingCosts |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmounts accrued for purchase of property and equipment.
| Name: |
eras_AmountsAccruedForPurchaseOfPropertyAndEquipment |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionIncrease decrease in accrued expenses and other current and long-term liabilities.
| Name: |
eras_IncreaseDecreaseInAccruedExpensesAndOtherCurrentAndLongTermLiabilities |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionIncrease (decrease) in operating lease assets and liabilities, net.
| Name: |
eras_IncreaseDecreaseInOperatingLeaseAssetsAndLiabilitiesNet |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionIncrease (decrease) in prepaid expenses and other current and long-term assets.
| Name: |
eras_IncreaseDecreaseInPrepaidExpensesAndOtherCurrentAndLongTermAssets |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionVesting of early exercised options.
| Name: |
eras_VestingOfEarlyExercisedOptions |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe sum of the periodic adjustments of the differences between securities' face values and purchase prices that are charged against earnings. This is called accretion if the security was purchased at a discount and amortization if it was purchased at premium. As a noncash item, this element is an adjustment to net income when calculating cash provided by or used in operations using the indirect method. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_AccretionAmortizationOfDiscountsAndPremiumsInvestments |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_AdjustmentsToReconcileNetIncomeLossToCashProvidedByUsedInOperatingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash and cash equivalents, and cash and cash equivalents restricted to withdrawal or usage; including, but not limited to, disposal group and discontinued operations. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-4
| Name: |
us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsIncludingDisposalGroupAndDiscontinuedOperations |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of increase (decrease) in cash, cash equivalents, and cash and cash equivalents restricted to withdrawal or usage; including effect from exchange rate change. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-SubTopic 230
-Topic 830
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481877/830-230-45-1
| Name: |
us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsPeriodIncreaseDecreaseIncludingExchangeRateEffect |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_CashFlowNoncashInvestingAndFinancingActivitiesDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate expense recognized in the current period that allocates the cost of tangible assets, intangible assets, or depleting assets to periods that benefit from use of the assets. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (b)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Subparagraph (e)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
| Name: |
us-gaap_DepreciationDepletionAndAmortization |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) during the reporting period in the aggregate amount of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_IncreaseDecreaseInAccountsPayable |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_IncreaseDecreaseInOperatingCapitalAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from financing activities, including discontinued operations. Financing activity cash flows include obtaining resources from owners and providing them with a return on, and a return of, their investment; borrowing money and repaying amounts borrowed, or settling the obligation; and obtaining and paying for other resources obtained from creditors on long-term credit. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
| Name: |
us-gaap_NetCashProvidedByUsedInFinancingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInFinancingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from investing activities, including discontinued operations. Investing activity cash flows include making and collecting loans and acquiring and disposing of debt or equity instruments and property, plant, and equipment and other productive assets. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
| Name: |
us-gaap_NetCashProvidedByUsedInInvestingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInInvestingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from operating activities, including discontinued operations. Operating activity cash flows include transactions, adjustments, and changes in value not defined as investing or financing activities. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 25
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-25
| Name: |
us-gaap_NetCashProvidedByUsedInOperatingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInOperatingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflows from the purchase of net carrying value allocated to in-process research and development costs and materials acquired in a business combination. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 13
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-13
| Name: |
us-gaap_PaymentsToAcquireInProcessResearchAndDevelopment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow associated with the acquisition of long-lived, physical assets that are used in the normal conduct of business to produce goods and services and not intended for resale; includes cash outflows to pay for construction of self-constructed assets. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 13
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-13
| Name: |
us-gaap_PaymentsToAcquirePropertyPlantAndEquipment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow associated with the amount received from entity's raising of capital via private rather than public placement. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
| Name: |
us-gaap_ProceedsFromIssuanceOfPrivatePlacement |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow associated with the aggregate amount received by the entity through sale or maturity of marketable securities (held-to-maturity or available-for-sale) during the period.
| Name: |
us-gaap_ProceedsFromSaleAndMaturityOfMarketableSecurities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow from exercise of option under share-based payment arrangement. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 14
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-14
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2A
-Subparagraph (a)
-SubTopic 10
-Topic 718
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2A
| Name: |
us-gaap_ProceedsFromStockOptionsExercised |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe consolidated profit or loss for the period, net of income taxes, including the portion attributable to the noncontrolling interest. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 8: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-11
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 205
-Name Accounting Standards Codification
-Section 45
-Paragraph 3
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480767/946-205-45-3
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(16))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 19
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481231/810-10-45-19
Reference 16: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 18: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 29: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 235
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-05(b)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479557/942-235-S99-1
Reference 32: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 33: http://www.xbrl.org/2003/role/exampleRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 4J
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481175/810-10-55-4J
Reference 34: http://www.xbrl.org/2003/role/exampleRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 4K
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481175/810-10-55-4K
Reference 35: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-2
Reference 38: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1A
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-1A
Reference 39: http://www.xbrl.org/2003/role/disclosureRef
-Topic 810
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1A
-Subparagraph (c)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481203/810-10-50-1A
| Name: |
us-gaap_ProfitLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of noncash expense for share-based payment arrangement. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (a)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
| Name: |
us-gaap_ShareBasedCompensation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
v3.24.1.u1
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 402
-Subsection v
-Paragraph 1
| Name: |
ecd_PvpTable |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
v3.24.1.u1
Insider Trading Arrangements
|
3 Months Ended |
|
Mar. 31, 2024
shares
|
|---|
| Trading Arrangements, by Individual |
|
| Material Terms of Trading Arrangement |
On January 10, 2024, David M. Chacko, M.D., our Chief Financial Officer and Chief Business Officer, adopted a Rule 10b5-1 trading arrangement that is intended to satisfy the affirmative defense of Rule 10b5-1(c) for the sale of up to 260,000 shares of our common stock until December 31, 2025.
|
| Rule 10b5-1 Arrangement Adopted |
false
|
| Non-Rule 10b5-1 Arrangement Adopted |
false
|
| Rule 10b5-1 Arrangement Terminated |
false
|
| Non-Rule 10b5-1 Arrangement Terminated |
false
|
| David M. Chacko [Member] |
|
| Trading Arrangements, by Individual |
|
| Name |
David M. Chacko
|
| Title |
M.D., our Chief Financial Officer and Chief Business Officer
|
| Rule 10b5-1 Arrangement Adopted |
true
|
| Adoption Date |
January 10, 2024
|
| Termination Date |
December 31, 2025
|
| Arrangement Duration |
722 days
|
| Aggregate Available |
260,000
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 1
| Name: |
ecd_MtrlTermsOfTrdArrTextBlock |
| Namespace Prefix: |
ecd_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 1
| Name: |
ecd_NonRule10b51ArrAdoptedFlag |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 1
| Name: |
ecd_NonRule10b51ArrTrmntdFlag |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 1
| Name: |
ecd_Rule10b51ArrAdoptedFlag |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 1
| Name: |
ecd_Rule10b51ArrTrmntdFlag |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 2
-Subparagraph A
| Name: |
ecd_TradingArrByIndTable |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 2
-Subparagraph B
| Name: |
ecd_TrdArrAdoptionDate |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 2
-Subparagraph C
| Name: |
ecd_TrdArrDuration |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 2
-Subparagraph A
| Name: |
ecd_TrdArrIndName |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 2
-Subparagraph A
| Name: |
ecd_TrdArrIndTitle |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 2
-Subparagraph D
| Name: |
ecd_TrdArrSecuritiesAggAvailAmt |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- ReferencesReference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-K
-Number 229
-Section 408
-Subsection a
-Paragraph 2
-Subparagraph B
| Name: |
ecd_TrdArrTerminationDate |
| Namespace Prefix: |
ecd_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ecd_IndividualAxis=eras_DavidMChackoMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Organization and Basis of Presentation
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Organization, Consolidation and Presentation of Financial Statements [Abstract] |
|
| Organization and Basis of Presentation |
Note 1. Organization and basis of presentation Organization and nature of operations Erasca, Inc. (Erasca or the Company) is a clinical-stage precision oncology company singularly focused on discovering, developing, and commercializing therapies for RAS/MAPK pathway-driven cancers. The Company has assembled a wholly-owned or controlled RAS/MAPK pathway-focused pipeline comprising modality-agnostic programs aligned with its three therapeutic strategies of: (1) targeting key upstream and downstream signaling nodes in the RAS/MAPK pathway; (2) targeting RAS directly; and (3) targeting escape routes that emerge in response to treatment. The Company was incorporated under the laws of the State of Delaware on July 2, 2018, as Erasca, Inc., and is headquartered in San Diego, California. In September 2020, the Company established a wholly-owned Australian subsidiary, Erasca Australia Pty Ltd (Erasca Australia), in order to conduct clinical activities in Australia for its development candidates. In November 2020, the Company entered into an agreement and plan of merger with Asana BioSciences, LLC (Asana) and ASN Product Development, Inc. (ASN) (the Asana Merger Agreement), pursuant to which ASN became the Company's wholly-owned subsidiary. In March 2021, the Company established a wholly-owned subsidiary, Erasca Ventures, LLC (Erasca Ventures), to make equity investments in early-stage biotechnology companies that are aligned with the Company’s mission and strategy. Since inception, the Company has devoted substantially all of its efforts and resources to organizing and staffing the Company, business planning, raising capital, identifying, acquiring and in-licensing the Company’s product candidates, establishing its intellectual property portfolio, conducting research, preclinical studies, and clinical trials, establishing arrangements with third parties for the manufacture of its product candidates and related raw materials, and providing general and administrative support for these operations. As of March 31, 2024, the Company had $297.7 million in cash, cash equivalents, and short-term marketable securities. As of March 31, 2024, the Company had an accumulated deficit of $641.0 million. The Company has incurred significant operating losses and negative cash flows from operations. From its inception through March 31, 2024, the Company’s financial support has primarily been provided from the sale of its convertible preferred stock and the sale of its common stock in its initial public offering (IPO) and underwritten offering (2022 Offering). The Company expects to use its cash, cash equivalents, and short-term marketable securities to fund research and development, working capital, and other general corporate purposes. The Company does not expect to generate any revenues from product sales unless and until the Company successfully completes development and obtains regulatory approval for any of its product candidates, which will not be for at least the next several years, if ever. Accordingly, until such time as the Company can generate significant revenue from sales of its product candidates, if ever, the Company expects to finance its cash needs through equity offerings, debt financings, or other capital sources, including potential collaborations, licenses or other similar arrangements. However, the Company may not be able to secure additional financing or enter into such other arrangements in a timely manner or on favorable terms, if at all. The Company’s failure to raise capital or enter into such other arrangements when needed would have a negative impact on the Company’s financial condition and could force the Company to delay, limit, reduce or terminate its research and development programs or other operations, or grant rights to develop and market product candidates that the Company would otherwise prefer to develop and market itself. The Company believes its cash, cash equivalents, and short-term marketable securities as of March 31, 2024, plus the net proceeds received in April 2024 from the Company's private placement, which closed on April 2, 2024, will be sufficient for the Company to fund operations for at least one year from the issuance date of these condensed consolidated financial statements. 2024 Private Placement In March 2024, the Company entered into a stock purchase agreement with the purchasers named therein for the private placement of 21,844,660 shares of its common stock at a price of $2.06 per share (the 2024 Private Placement). On April 2, 2024, the 2024 Private Placement closed and the net proceeds were $43.7 million, after deducting placement agent fees and estimated expenses of approximately $1.3 million. The Company received $6.9 million of the proceeds in March 2024 prior to the closing of the 2024 Private Placement, which is recorded in cash and cash equivalents and as a liability in proceeds from prepayment of private placement on the condensed consolidated balance sheets. Basis of presentation The accompanying unaudited condensed consolidated financial statements have been prepared in accordance with US generally accepted accounting principles (US GAAP) for interim financial information and pursuant to Form 10-Q and Article 10 of Regulation S-X of the Securities and Exchange Commission (SEC). Accordingly, they do not include all of the information and notes required by US GAAP for complete financial statements. Any reference in these notes to applicable guidance is meant to refer to US GAAP as found in the Accounting Standards Codification (ASC) and Accounting Standards Updates (ASU) promulgated by the Financial Accounting Standards Board (FASB). The Company’s condensed consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries, Erasca Australia, ASN, and Erasca Ventures. All intercompany balances and transactions have been eliminated.
|
| X |
- References
+ Details
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for organization, consolidation and basis of presentation of financial statements disclosure. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480424/946-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480424/946-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 810
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//810/tableOfContent
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 205
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//205/tableOfContent
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Summary of Significant Accounting Policies
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Accounting Policies [Abstract] |
|
| Summary of Significant Accounting Policies |
Note 2. Summary of significant accounting policies Use of estimates The preparation of the Company’s condensed consolidated financial statements in conformity with US GAAP requires the Company to make estimates and assumptions that impact the reported amounts of assets, liabilities, expenses, and the disclosure of contingent assets and liabilities in the condensed consolidated financial statements and accompanying notes. Accounting estimates and management judgments reflected in the condensed consolidated financial statements include, but are not limited to, the accrual of research and development expenses, stock-based compensation expense, and the incremental borrowing rate for determining the operating lease asset and liability. Management evaluates its estimates on an ongoing basis. Although estimates are based on the Company’s historical experience, knowledge of current events, and actions it may undertake in the future, actual results may ultimately materially differ from these estimates and assumptions. Unaudited interim financial information The accompanying condensed consolidated balance sheet as of March 31, 2024, the condensed consolidated statements of operations and comprehensive loss for the three months ended March 31, 2024 and 2023, the condensed consolidated statements of stockholders’ equity for the three months ended March 31, 2024 and 2023 and the condensed consolidated statements of cash flows for the three months ended March 31, 2024 and 2023 are unaudited. The unaudited condensed consolidated interim financial statements have been prepared on the same basis as the audited annual consolidated financial statements and, in the opinion of management, reflect all adjustments, which include only normal recurring adjustments, necessary for the fair statement of the Company’s condensed consolidated financial position as of March 31, 2024 and the condensed consolidated results of its operations and cash flows for the three months ended March 31, 2024 and 2023. The condensed consolidated financial data and other information disclosed in these notes related to the three months ended March 31, 2024 and 2023 are unaudited. The condensed consolidated results for the three months ended March 31, 2024 are not necessarily indicative of results to be expected for the year ending December 31, 2024, any other interim periods, or any future year or period. These unaudited condensed consolidated financial statements should be read in conjunction with the Company's audited consolidated financial statements included in its Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024. Concentration of credit risk and off-balance sheet risk Financial instruments which potentially subject the Company to significant concentration of credit risk consist of cash and cash equivalents and marketable securities. The Company maintains deposits in federally insured financial institutions in excess of federally insured limits. The Company has not experienced any losses in such accounts, and management believes that the Company is not exposed to significant credit risk due to the financial position of the depository institutions in which those deposits are held. The Company’s investment policy includes guidelines for the quality of the related institutions and financial instruments and defines allowable investments that the Company may invest in, which the Company believes minimizes the exposure to concentration of credit risk. Cash, cash equivalents and restricted cash Cash and cash equivalents include cash in readily available checking and savings accounts, money market funds and commercial paper. The Company considers all highly liquid investments with an original maturity of three months or less from the date of purchase to be cash equivalents. The Company had deposited cash of $408,000 as of March 31, 2024 and December 31, 2023 to secure a letter of credit in connection with the lease of the Company’s facilities (see Note 10). The Company has classified the restricted cash as a noncurrent asset on its condensed consolidated balance sheets. The following table provides a reconciliation of cash, cash equivalents and restricted cash reported within the condensed consolidated balance sheets that sum to the total of the same amounts shown in the condensed consolidated statements of cash flows (in thousands):
|
|
|
|
|
|
|
|
|
March 31, |
|
|
2024 |
|
|
2023 |
|
Cash and cash equivalents |
$ |
76,726 |
|
|
$ |
272,756 |
|
Restricted cash |
|
408 |
|
|
|
408 |
|
Total cash, cash equivalents and restricted cash |
$ |
77,134 |
|
|
$ |
273,164 |
|
Marketable securities and investments The Company classifies all marketable securities as available-for-sale, as the sale of such securities may be required prior to maturity. Management determines the appropriate classification of its marketable securities at the time of purchase. Marketable securities with original maturities beyond three months at the date of purchase and which mature at, or less than 12 months from, the balance sheet date are classified as short-term marketable securities. Available-for-sale securities are carried at fair value, with the unrealized gains and losses reported as accumulated other comprehensive income (loss) until realized. The amortized cost of available-for-sale debt securities is adjusted for amortization of premiums and accretion of discounts to maturity. Such amortization and accretion are included in interest income. The Company regularly reviews all of its marketable securities for declines in fair value. The review includes the consideration of the cause of the impairment, including the creditworthiness of the security issuers, the number of securities in an unrealized loss position, the severity of the unrealized loss(es), whether the Company has the intent to sell the securities and whether it is more likely than not that the Company will be required to sell the securities before the recovery of their amortized cost basis. If the decline in fair value is due to credit-related factors, a loss is recognized in net income; whereas, if the decline in fair value is not due to credit-related factors, the loss is recorded in other comprehensive income (loss). Realized gains and losses on available-for-sale securities are included in other income or expense. The cost of securities sold is based on the specific identification method. Interest and dividends on securities classified as available-for-sale are included in interest income. Through its wholly-owned subsidiary, Erasca Ventures, the Company has also invested in equity securities of a company whose securities are not publicly traded and whose fair value is not readily available (see Notes 3 and 14). This investment is recorded using cost minus impairment, plus or minus changes in its estimated fair value resulting from observable price changes in orderly transactions for the identical or a similar investment of the same issuer. Investments in equity securities without readily determinable fair values are assessed for potential impairment on a quarterly basis based on qualitative factors. This investment is included in other assets in the Company's condensed consolidated balance sheets. Fair value measurements Certain assets and liabilities are carried at fair value under US GAAP. Fair value is defined as an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. Financial assets and liabilities carried at fair value are classified and disclosed in one of the following three levels of the fair value hierarchy, of which the first two are considered observable and the last is considered unobservable: Level 1—Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities. Level 2—Quoted prices for similar assets and liabilities in active markets, quoted prices in markets that are not active, or inputs which are observable, either directly or indirectly, for substantially the full term of the asset or liability. Level 3—Prices or valuation techniques that require inputs that are both significant to the fair value measurement and unobservable (i.e., supported by little or no market activity). Recently issued accounting pronouncements not yet adopted From time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that the Company adopts as of the specified effective date. The Company qualifies as an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012 (JOBS Act) and has elected not to “opt out” of the extended transition related to complying with new or revised accounting standards, which means that when a standard is issued or revised and it has different application dates for public and nonpublic companies, the Company can adopt the new or revised standard at the time nonpublic companies adopt the new or revised standard and can do so until such time that the Company either (i) irrevocably elects to “opt out” of such extended transition period or (ii) no longer qualifies as an emerging growth company. In November 2023, the FASB issued ASU 2023-07, Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures, which is intended to provide enhancements to segment disclosures, even for entities with only one reportable segment. In particular, the standard will require disclosures of significant segment expenses regularly provided to the chief operating decision maker and included within each reported measure of segment profit and loss. The standard will also require disclosure of all other segment items by reportable segment and a description of its composition. Finally, the standard will require disclosure of the title and position of the chief operating decision maker and an explanation of how the chief operating decision maker uses the reported measure(s) of segment profit or loss in assessing segment performance and deciding how to allocate resources. The standard is effective for annual periods beginning after December 15, 2023, and interim periods within annual periods beginning after December 15, 2024. Early adoption is permitted. Retrospective application to all prior periods presented in the financial statements is required. The Company is currently evaluating the impact of the standard on the presentation of its consolidated financial statements and related disclosures. In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures, which is intended to provide enhancements to annual income tax disclosures. In particular, the standard will require more detailed information in the income tax rate reconciliation, as well as the disclosure of income taxes paid disaggregated by jurisdiction, among other enhancements. The standard is effective for the Company in its annual period beginning after December 15, 2025 and early adoption is permitted. The standard allows for adoption on a prospective basis, with a retrospective option. The Company is currently evaluating the impact of the standard on the presentation of its consolidated financial statements and related disclosures. |
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for all significant accounting policies of the reporting entity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483426/235-10-50-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 235
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//235/tableOfContent
| Name: |
us-gaap_SignificantAccountingPoliciesTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Fair Value Measurements
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Fair Value Disclosures [Abstract] |
|
| Fair Value Measurements |
Note 3. Fair value measurements The following tables summarize the Company’s financial assets measured at fair value on a recurring basis and their respective input levels based on the fair value hierarchy (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value measurements as of March 31, 2024 using |
|
|
|
|
|
|
Quoted prices in |
|
|
Significant |
|
|
Significant |
|
|
|
|
|
|
active markets |
|
|
other |
|
|
unobservable |
|
|
|
March 31, |
|
|
for identical |
|
|
observable |
|
|
inputs |
|
|
|
2024 |
|
|
assets (level 1) |
|
|
inputs (level 2) |
|
|
(level 3) |
|
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds(1) |
|
$ |
58,324 |
|
|
$ |
58,324 |
|
|
$ |
— |
|
|
$ |
— |
|
Commercial paper(1) |
|
|
1,492 |
|
|
|
— |
|
|
|
1,492 |
|
|
|
— |
|
US treasury securities(2) |
|
|
73,644 |
|
|
|
73,644 |
|
|
|
— |
|
|
|
— |
|
US government agency securities(2) |
|
|
23,856 |
|
|
|
— |
|
|
|
23,856 |
|
|
|
— |
|
Corporate debt securities(2) |
|
|
22,926 |
|
|
|
— |
|
|
|
22,926 |
|
|
|
— |
|
Commercial paper(2) |
|
|
100,533 |
|
|
|
— |
|
|
|
100,533 |
|
|
|
— |
|
Total fair value of assets |
|
$ |
280,775 |
|
|
$ |
131,968 |
|
|
$ |
148,807 |
|
|
$ |
— |
|
(1)Included as cash and cash equivalents on the condensed consolidated balance sheets. (2)Included as short-term marketable securities on the condensed consolidated balance sheets.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value measurements as of December 31, 2023 using |
|
|
|
|
|
|
Quoted prices in |
|
|
Significant |
|
|
Significant |
|
|
|
|
|
|
active markets |
|
|
other |
|
|
unobservable |
|
|
|
December 31, |
|
|
for identical |
|
|
observable |
|
|
inputs |
|
|
|
2023 |
|
|
assets (level 1) |
|
|
inputs (level 2) |
|
|
(level 3) |
|
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds(1) |
|
$ |
83,101 |
|
|
$ |
83,101 |
|
|
$ |
— |
|
|
$ |
— |
|
US treasury securities(2) |
|
|
93,303 |
|
|
|
93,303 |
|
|
|
— |
|
|
|
— |
|
US government agency securities(2) |
|
|
26,824 |
|
|
|
— |
|
|
|
26,824 |
|
|
|
— |
|
Corporate debt securities(2) |
|
|
10,734 |
|
|
|
— |
|
|
|
10,734 |
|
|
|
— |
|
Commercial paper(2) |
|
|
88,414 |
|
|
|
— |
|
|
|
88,414 |
|
|
|
— |
|
US treasury securities(3) |
|
|
9,642 |
|
|
|
9,642 |
|
|
|
— |
|
|
|
— |
|
Total fair value of assets |
|
$ |
312,018 |
|
|
$ |
186,046 |
|
|
$ |
125,972 |
|
|
$ |
— |
|
(1)Included as cash and cash equivalents on the condensed consolidated balance sheets. (2)Included as short-term marketable securities on the condensed consolidated balance sheets. (3)Included as long-term marketable securities on the condensed consolidated balance sheets. The carrying amounts of the Company’s financial instruments, including cash, prepaid expenses and other current assets, accounts payable, accrued expenses and other current liabilities and proceeds from prepayment of private placement, approximate fair value due to their short maturities. As of March 31, 2024 and December 31, 2023, the Company held a $2.0 million equity investment in Affini-T Therapeutics, Inc. (Affini-T) at cost. No adjustments have been made to the value of the Company’s investment in Affini-T since its initial measurement either due to impairment or based on observable price changes. None of the Company’s non-financial assets or liabilities are recorded at fair value on a non-recurring basis. No transfers between levels have occurred during the periods presented. Cash equivalents consist of money market funds and commercial paper, short-term marketable securities consist of US treasury securities, US government agency securities, corporate debt securities and commercial paper, and long-term marketable securities consist of US treasury securities. The Company obtains pricing information from its investment manager and generally determines the fair value of marketable securities using standard observable inputs, including benchmark yields, reported trades, broker/dealer quotes, issuer spreads, two-sided markets, and bid and/or offers.
|
| X |
- References
+ Details
| Name: |
us-gaap_FairValueDisclosuresAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for the fair value of financial instruments (as defined), including financial assets and financial liabilities (collectively, as defined), and the measurements of those instruments as well as disclosures related to the fair value of non-financial assets and liabilities. Such disclosures about the financial instruments, assets, and liabilities would include: (1) the fair value of the required items together with their carrying amounts (as appropriate); (2) for items for which it is not practicable to estimate fair value, disclosure would include: (a) information pertinent to estimating fair value (including, carrying amount, effective interest rate, and maturity, and (b) the reasons why it is not practicable to estimate fair value; (3) significant concentrations of credit risk including: (a) information about the activity, region, or economic characteristics identifying a concentration, (b) the maximum amount of loss the entity is exposed to based on the gross fair value of the related item, (c) policy for requiring collateral or other security and information as to accessing such collateral or security, and (d) the nature and brief description of such collateral or security; (4) quantitative information about market risks and how such risks are managed; (5) for items measured on both a recurring and nonrecurring basis information regarding the inputs used to develop the fair value measurement; and (6) for items presented in the financial statement for which fair value measurement is elected: (a) information necessary to understand the reasons for the election, (b) discussion of the effect of fair value changes on earnings, (c) a description of [similar groups] items for which the election is made and the relation thereof to the balance sheet, the aggregate carrying value of items included in the balance sheet that are not eligible for the election; (7) all other required (as defined) and desired information. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 820
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
| Name: |
us-gaap_FairValueDisclosuresTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Marketable Securities
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Investments, Debt and Equity Securities [Abstract] |
|
| Marketable Securities |
Note 4. Marketable securities The following tables summarize the Company’s marketable securities accounted for as available-for-sale securities (in thousands, except years):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2024 |
|
|
|
Maturity |
|
Amortized |
|
|
Unrealized |
|
|
Unrealized |
|
|
Estimated |
|
|
|
(in years) |
|
cost |
|
|
gains |
|
|
losses |
|
|
fair value |
|
US treasury securities |
|
1 or less |
|
$ |
73,753 |
|
|
$ |
8 |
|
|
$ |
(117 |
) |
|
$ |
73,644 |
|
US government agency securities |
|
1 or less |
|
|
23,859 |
|
|
|
11 |
|
|
|
(14 |
) |
|
|
23,856 |
|
Corporate debt securities |
|
1 or less |
|
|
22,967 |
|
|
|
— |
|
|
|
(41 |
) |
|
|
22,926 |
|
Commercial paper |
|
1 or less |
|
|
100,590 |
|
|
|
16 |
|
|
|
(73 |
) |
|
|
100,533 |
|
Total |
|
|
|
$ |
221,169 |
|
|
$ |
35 |
|
|
$ |
(245 |
) |
|
$ |
220,959 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2023 |
|
|
|
Maturity |
|
Amortized |
|
|
Unrealized |
|
|
Unrealized |
|
|
Estimated |
|
|
|
(in years) |
|
cost |
|
|
gains |
|
|
losses |
|
|
fair value |
|
US treasury securities |
|
1 or less |
|
$ |
93,377 |
|
|
$ |
74 |
|
|
$ |
(148 |
) |
|
$ |
93,303 |
|
US government agency securities |
|
1 or less |
|
|
26,783 |
|
|
|
45 |
|
|
|
(4 |
) |
|
|
26,824 |
|
Corporate debt securities |
|
1 or less |
|
|
10,719 |
|
|
|
15 |
|
|
|
— |
|
|
|
10,734 |
|
Commercial paper |
|
1 or less |
|
|
88,356 |
|
|
|
68 |
|
|
|
(10 |
) |
|
|
88,414 |
|
US treasury securities |
|
1-2 |
|
|
9,605 |
|
|
|
37 |
|
|
|
— |
|
|
|
9,642 |
|
Total |
|
|
|
$ |
228,840 |
|
|
$ |
239 |
|
|
$ |
(162 |
) |
|
$ |
228,917 |
|
The following tables present fair values and gross unrealized losses for those available-for-sale securities that were in an unrealized loss position as of March 31, 2024 and December 31, 2023, aggregated by category and the length of time that the securities have been in a continuous loss position (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2024 |
|
|
|
Unrealized losses less than 12 months |
|
|
Unrealized losses 12 months or greater |
|
|
Total |
|
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
US treasury securities |
|
$ |
55,139 |
|
|
$ |
(117 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
55,139 |
|
|
$ |
(117 |
) |
US government agency securities |
|
|
18,845 |
|
|
|
(14 |
) |
|
|
— |
|
|
|
— |
|
|
|
18,845 |
|
|
|
(14 |
) |
Corporate debt securities |
|
|
22,926 |
|
|
|
(41 |
) |
|
|
— |
|
|
|
— |
|
|
|
22,926 |
|
|
|
(41 |
) |
Commercial paper |
|
|
65,574 |
|
|
|
(73 |
) |
|
|
— |
|
|
|
— |
|
|
|
65,574 |
|
|
|
(73 |
) |
Total |
|
$ |
162,484 |
|
|
$ |
(245 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
162,484 |
|
|
$ |
(245 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2023 |
|
|
|
Unrealized losses less than 12 months |
|
|
Unrealized losses 12 months or greater |
|
|
Total |
|
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
US treasury securities |
|
$ |
74,912 |
|
|
$ |
(148 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
74,912 |
|
|
$ |
(148 |
) |
US government agency securities |
|
|
6,950 |
|
|
|
(4 |
) |
|
|
— |
|
|
|
— |
|
|
|
6,950 |
|
|
|
(4 |
) |
Corporate debt securities |
|
|
748 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
748 |
|
|
|
— |
|
Commercial paper |
|
|
22,944 |
|
|
|
(10 |
) |
|
|
— |
|
|
|
— |
|
|
|
22,944 |
|
|
|
(10 |
) |
Total |
|
$ |
105,554 |
|
|
$ |
(162 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
105,554 |
|
|
$ |
(162 |
) |
As of March 31, 2024, there were 28 available-for-sale securities with an estimated fair value of $162.5 million in gross unrealized loss positions, none of which were in an unrealized loss position for more than 12 months. As of December 31, 2023, there were 22 available-for-sale securities with an estimated fair value of $105.6 million in gross unrealized loss positions, none of which were in an unrealized loss position for more than 12 months. As of March 31, 2024 and December 31, 2023, unrealized losses on available-for-sale securities are not attributed to credit risk. The Company believes that an allowance for credit losses is unnecessary because the unrealized losses on certain of the Company’s available-for-sale securities are due to market factors and interest rate increases. Additionally, the Company does not intend to sell the securities nor is it more likely than not that the Company will be required to sell the securities before recovery of their amortized cost basis. Accrued interest on the Company’s available-for-sale securities was $871,000 and $1.1 million as of March 31, 2024 and December 31, 2023, respectively, and was included in prepaid expenses and other current assets on the condensed consolidated balance sheets.
|
| X |
- References
+ Details
| Name: |
us-gaap_InvestmentsDebtAndEquitySecuritiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for investments in certain debt and equity securities. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//320/tableOfContent
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 10
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-10
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 820
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6B
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-6B
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 820
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6B
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-6B
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name Regulation S-K (SK)
-Number 229
-Section 1403
-Paragraph (b)
-Publisher SEC
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 946
-SubTopic 320
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//946-320/tableOfContent
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 940
-SubTopic 320
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//940-320/tableOfContent
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 320
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//942-320/tableOfContent
| Name: |
us-gaap_InvestmentsInDebtAndMarketableEquitySecuritiesAndCertainTradingAssetsDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Property and Equipment, Net
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Property, Plant and Equipment [Abstract] |
|
| Property and Equipment, Net |
Note 5. Property and equipment, net Property and equipment, net consisted of the following (in thousands):
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Laboratory equipment |
|
$ |
5,621 |
|
|
$ |
5,620 |
|
Furniture and fixtures |
|
|
4,099 |
|
|
|
4,099 |
|
Leasehold improvements |
|
|
18,173 |
|
|
|
18,173 |
|
Computer equipment and software |
|
|
1,695 |
|
|
|
1,667 |
|
Property and equipment |
|
|
29,588 |
|
|
|
29,559 |
|
Less accumulated depreciation and amortization |
|
|
(8,230 |
) |
|
|
(7,232 |
) |
Property and equipment, net |
|
$ |
21,358 |
|
|
$ |
22,327 |
|
Depreciation and amortization expense related to property and equipment was $998,000 and $894,000 for the three months ended March 31, 2024 and 2023, respectively.
|
| X |
- References
+ Details
| Name: |
us-gaap_PropertyPlantAndEquipmentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for long-lived, physical asset used in normal conduct of business and not intended for resale. Includes, but is not limited to, work of art, historical treasure, and similar asset classified as collections. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 360
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//360/tableOfContent
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-6
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (d)
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-SubTopic 360
-Topic 958
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480321/958-360-50-7
| Name: |
us-gaap_PropertyPlantAndEquipmentDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Accrued Expenses and Other Current Liabilities
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Payables and Accruals [Abstract] |
|
| Accrued Expenses and Other Current Liabilities |
Note 6. Accrued expenses and other current liabilities Accrued expenses and other current liabilities consisted of the following (in thousands):
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Accrued research and development expenses |
|
$ |
9,247 |
|
|
$ |
8,803 |
|
Accrued compensation |
|
|
6,095 |
|
|
|
10,311 |
|
Unvested early exercised stock option liability |
|
|
309 |
|
|
|
464 |
|
Accrued professional services |
|
|
626 |
|
|
|
435 |
|
Accrued offering costs |
|
|
178 |
|
|
|
— |
|
Accrued property and equipment |
|
|
— |
|
|
|
13 |
|
Other accruals |
|
|
122 |
|
|
|
160 |
|
Total |
|
$ |
16,577 |
|
|
$ |
20,186 |
|
|
| X |
- DefinitionThe entire disclosure for accounts payable, accrued expenses, and other liabilities that are classified as current at the end of the reporting period.
| Name: |
us-gaap_AccountsPayableAccruedLiabilitiesAndOtherLiabilitiesDisclosureCurrentTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_PayablesAndAccrualsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Asset Acquisitions
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Asset Acquisition [Abstract] |
|
| Asset Acquisitions |
Note 7. Asset acquisitions The following purchased assets were accounted for as asset acquisitions as substantially all of the fair value of the assets acquired were concentrated in a group of similar assets, and the acquired assets did not have outputs or employees. Because the assets had not yet received regulatory approval, the fair value attributable to these assets was recorded as in-process research and development expenses in the Company’s condensed consolidated statements of operations and comprehensive loss. Asana BioSciences, LLC In November 2020, the Company entered into the Asana Merger Agreement, pursuant to which ASN became its wholly-owned subsidiary. Asana and ASN had previously entered into a license agreement, which was amended and restated prior to the closing of the merger transaction (the Asana License Agreement, and collectively with the Asana Merger Agreement, the Asana Agreements), pursuant to which ASN acquired an exclusive, worldwide license to certain intellectual property rights relating to inhibitors of ERK1 and ERK2 owned or controlled by Asana to develop and commercialize ERAS-007 and certain other related compounds for all applications. Under the Asana Merger Agreement, in 2020, the Company made an upfront payment of $20.0 million and issued 4,000,000 shares of its Series B-2 convertible preferred stock to Asana at a value of $7.50 per share or a total fair value of equity of $30.0 million. In connection with the Company's IPO, these shares of Series B-2 convertible preferred stock were converted into 3,333,333 shares of the Company’s common stock. The Company is obligated to make future development and regulatory milestone cash payments for a licensed product in an amount of up to $90.0 million. Additionally, upon achieving a development milestone related to demonstration of successful proof-of-concept in a specified clinical trial, the Company will also be required to issue 3,888,889 shares of its common stock to Asana. The Company is not obligated to pay royalties on the net sales of licensed products. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones had been accrued as the underlying contingencies were not probable or estimable. Emerge Life Sciences, Pte. Ltd. In March 2021, the Company entered into an asset purchase agreement (ELS Purchase Agreement) with Emerge Life Sciences, Pte. Ltd. (ELS) wherein it purchased all rights, title, and interest (including all patent and other intellectual property rights) to EGFR antibodies directed against the EGFR domain II (EGFR-D2) and domain III (EGFR-D3) as well as a bispecific antibody where one arm is directed against EGFR-D2 and the other is directed against EGFR-D3 (the Antibodies). Under the terms of the ELS Purchase Agreement, in 2021, the Company made an upfront payment of $2.0 million and issued to ELS 500,000 shares of the Company’s common stock at a value of $3.36 per share or a total fair value of equity of $1.7 million. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023.
|
| X |
- References
+ Details
| Name: |
us-gaap_AssetAcquisitionAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for asset acquisition. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 805
-SubTopic 50
-Name Accounting Standards Codification
-Section 15
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480123/805-50-15-3
| Name: |
us-gaap_AssetAcquisitionTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
License Agreements
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| License Agreements [Abstract] |
|
| License Agreements |
Note 8. License agreements Novartis Pharma AG In December 2022, the Company entered into an exclusive license agreement (as amended, the Novartis Agreement) with Novartis Pharma AG (Novartis) under which the Company was granted an exclusive, worldwide, royalty-bearing license to certain patent and other intellectual property rights owned or controlled by Novartis to develop, manufacture, use, and commercialize naporafenib in all fields of use. The Company has the right to sublicense (through multiple tiers) its rights under the Novartis Agreement, subject to certain limitations and conditions, and is required to use commercially reasonable efforts to commercialize licensed products in certain geographical markets. The license granted under the Novartis Agreement is subject to Novartis’ reserved right to: (i) develop, manufacture, use, and commercialize compounds unrelated to naporafenib under the licensed patent rights and know-how, (ii) use the licensed patent rights and know-how for non-clinical research purposes, and (iii) use the licensed patent rights and know-how to the extent necessary to perform ongoing clinical trials and perform its obligations under existing contracts and under the Novartis Agreement. Under the Novartis Agreement, the Company made an upfront cash payment to Novartis of $20.0 million and issued to Novartis 12,307,692 shares of common stock of the Company having an aggregate value of approximately $80.0 million. The Company is obligated to make future regulatory milestone payments of up to $80.0 million and sales milestone payments of up to $200.0 million. The Company is also obligated to pay royalties on net sales of all licensed products, in the low-single digit percentages, subject to certain reductions. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable. Katmai Pharmaceuticals, Inc. In March 2020, the Company entered into a license agreement (the Katmai Agreement) with Katmai Pharmaceuticals, Inc. (Katmai) under which the Company was granted an exclusive, worldwide, royalty-bearing license to certain patent rights and know-how controlled by Katmai related to the development of small molecule therapeutic and diagnostic products that modulate EGFR and enable the identification, diagnosis, selection, treatment, and/or monitoring of patients for neuro-oncological applications to develop, manufacture, use, and commercialize ERAS-801 and certain other related compounds in all fields of use. Under the Katmai Agreement, the Company made an upfront payment of $5.7 million and Katmai agreed to purchase shares of the Company’s Series B-1 convertible preferred stock and Series B-2 convertible preferred stock having an aggregate value of $2.7 million. In April 2020, Katmai purchased 356,000 shares of the Company’s Series B-1 convertible preferred stock for $1.8 million, and in January 2021, Katmai purchased 118,666 shares of the Company’s Series B-2 convertible preferred stock for $0.9 million. In connection with the Company's IPO, these shares of Series B-1 convertible preferred stock and Series B-2 convertible preferred stock were converted into 395,555 shares of the Company's common stock, in the aggregate. The Company is obligated to make future development and regulatory milestone payments of up to $26.0 million, of which $2.0 million was paid in March 2022, and commercial milestone payments of up to $101.0 million. The Company is also obligated to pay tiered royalties on net sales of each licensed product, at rates ranging from the mid- to high-single digit percentages, subject to a minimum annual royalty payment in the low six figures and certain permitted deductions. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable. NiKang Therapeutics, Inc. In February 2020, the Company entered into a license agreement (the NiKang Agreement) with NiKang Therapeutics, Inc. (NiKang) under which the Company was granted an exclusive, worldwide license to certain intellectual property rights owned or controlled by NiKang related to certain SHP2 inhibitors to develop and commercialize ERAS-601 and certain other related compounds for all applications. Under the NiKang Agreement, in 2020, the Company made an upfront payment of $5.0 million to NiKang and reimbursed NiKang $0.4 million for certain initial manufacturing costs. In addition, the Company paid $7.0 million in 2020 related to the publication of a US patent application that covered the composition of matter of ERAS-601. The Company is also obligated to pay (i) development and regulatory milestone payments in an aggregate amount of up to $16.0 million for the first licensed product, of which $4.0 million was paid in January 2021, and $12.0 million for a second licensed product, and (ii) commercial milestone payments in an aggregate amount of up to $157.0 million for the first licensed product and $151.0 million for a second licensed product. The Company is also obligated to: (i) pay tiered royalties on net sales of all licensed products in the mid-single digit percentages, subject to certain reductions; and (ii) equally split all net sublicensing revenues earned under sublicense agreements that the Company enters into with any third party before commencement of the first Phase I clinical trial for a licensed product. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable. LifeArc In April 2020, the Company entered into a license agreement with LifeArc (the LifeArc Agreement) under which the Company was granted an exclusive, worldwide license to certain materials, know-how, and intellectual property rights owned or controlled by LifeArc to develop, manufacture, use, and commercialize certain ULK inhibitors for all applications. Under the LifeArc Agreement, the Company was granted the license at no upfront cost and a period of three months after the effective date to conduct experiments on LifeArc’s compounds. Upon completion of this initial testing period, the Company had the option to continue the license and make a one-time license payment of $75,000 to LifeArc, which payment was subsequently made in 2020. The Company is obligated to make future development milestone payments for a licensed product of up to $11.0 million and sales milestone payments of up to $50.0 million. The Company is also obligated to pay royalties on net sales of all licensed products, in the low-single digit percentages, subject to certain reductions. No IPR&D expense was recorded during the three months ended March 31, 2024 and 2023. As of March 31, 2024 and December 31, 2023, no milestones are accrued as the underlying contingencies are not probable or estimable.
|
| X |
- Definition
+ References
+ Details
| Name: |
eras_LicenseAgreementsDisclosureTextBlock |
| Namespace Prefix: |
eras_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Stock-based Compensation
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Share-Based Payment Arrangement [Abstract] |
|
| Stock-based Compensation |
Note 9. Stock-based compensation In July 2021, the Company’s board of directors adopted and the Company’s stockholders approved the Company’s 2021 Incentive Award Plan (the 2021 Plan), which became effective in connection with the IPO. Upon the adoption of the 2021 Plan, the Company ceased making equity grants under its 2018 Equity Incentive Plan (the 2018 Plan). Under the 2021 Plan, the Company may grant stock options, restricted stock, restricted stock units, stock appreciation rights, and other stock or cash-based awards to individuals who are then employees, officers, directors or non-entity consultants of the Company. A total of 15,150,000 shares of common stock were initially reserved for issuance under the 2021 Plan. In addition, the number of shares of common stock available for issuance under the 2021 Plan may be increased annually on the first day of each calendar year during the term of the 2021 Plan, beginning in 2022, by an amount equal to the lesser of (i) 5% of the shares of common stock outstanding on the final day of the immediately preceding calendar year or (ii) such smaller number of shares as determined by the Company’s board of directors or an authorized committee of the board of directors. As of March 31, 2024, there were 13,204,701 stock-based awards available for future grant under the 2021 Plan. Subsequent to July 2021, no further awards will be granted under the 2018 Plan and all future stock-based awards will be granted under the 2021 Plan. To the extent outstanding options or restricted stock granted under the 2018 Plan are cancelled, forfeited, repurchased, or otherwise terminated without being exercised or becoming vested, and would otherwise have been returned to the share reserve under the 2018 Plan, the number of shares underlying such awards will be available for future grant under the 2021 Plan. Options granted are exercisable at various dates as determined upon grant and will expire no more than ten years from their date of grant. Stock options generally vest over a four-year term. The exercise price of each option shall be determined by the Company’s board of directors based on the estimated fair value of the Company’s stock on the date of the option grant. The exercise price shall not be less than 100% of the fair market value of the Company’s common stock at the time the option is granted. For holders of more than 10% of the Company’s total combined voting power of all classes of stock, incentive stock options may not be granted at less than 110% of the fair market value of the Company’s common stock on the date of grant and for a term that exceeds five years. Early exercise was permitted for certain grants under the 2018 Plan. Stock options A summary of the Company’s stock option activity under the 2021 Plan and 2018 Plan is as follows (in thousands, except share and per share data and years):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
average remaining |
|
|
Aggregate |
|
|
|
|
|
|
average |
|
|
contractual |
|
|
intrinsic |
|
|
|
Shares |
|
|
exercise price |
|
|
term (years) |
|
|
value |
|
Outstanding at December 31, 2023 |
|
|
24,970,957 |
|
|
$ |
4.98 |
|
|
|
8.12 |
|
|
$ |
4,412 |
|
Granted |
|
|
9,827,650 |
|
|
|
1.71 |
|
|
|
|
|
|
|
Exercised |
|
|
(35,035 |
) |
|
|
0.69 |
|
|
|
|
|
|
|
Canceled |
|
|
(116,449 |
) |
|
|
4.89 |
|
|
|
|
|
|
|
Outstanding at March 31, 2024 |
|
|
34,647,123 |
|
|
$ |
4.06 |
|
|
|
8.44 |
|
|
$ |
7,532 |
|
Options exercisable at March 31, 2024 |
|
|
12,850,952 |
|
|
$ |
4.83 |
|
|
|
7.32 |
|
|
$ |
3,916 |
|
The weighted-average grant date fair value of options granted for the three months ended March 31, 2024 and 2023 was $1.25 and $2.94, respectively. As of March 31, 2024, the unrecognized compensation cost related to unvested stock option grants was $55.9 million and is expected to be recognized as expense over approximately 2.75 years. The intrinsic value of the options exercised for the three months ended March 31, 2024 and 2023 was $47,000 and $517,000, respectively. Prior to the Company's IPO, certain individuals were granted the ability to early exercise their stock options. The shares of common stock issued from the early exercise of unvested stock options are restricted and continue to vest in accordance with the original vesting schedule. The Company has the option to repurchase any unvested shares at the original purchase price upon any voluntary or involuntary termination. The shares purchased by the employees and non-employees pursuant to the early exercise of stock options are not deemed, for accounting purposes, to be outstanding until those shares vest. The cash received in exchange for exercised and unvested shares related to stock options granted is recorded as a liability for the early exercise of stock options on the accompanying condensed consolidated balance sheets and will be transferred into common stock and additional paid-in capital as the shares vest. As of March 31, 2024 and December 31, 2023, there were 247,919 shares and 371,876 shares subject to repurchase by the Company, respectively. As of March 31, 2024 and December 31, 2023, the Company recorded $309,000 and $464,000 of liabilities associated with shares issued with repurchase rights, respectively, which is recorded in accrued expenses and other current liabilities. The assumptions used in the Black-Scholes option pricing model to determine the fair value of the employee and nonemployee stock option grants were as follows:
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
2024 |
|
2023 |
Risk-free interest rate |
|
3.84%-4.15% |
|
3.46%-4.22% |
Expected volatility |
|
83.35%-83.91% |
|
85.52%-85.81% |
Expected term (in years) |
|
5.28-6.01 |
|
6.02-6.07 |
Expected dividend yield |
|
--% |
|
--% |
Employee stock purchase plan In July 2021, the Company’s board of directors adopted and the Company’s stockholders approved the Company's 2021 Employee Stock Purchase Plan (the ESPP), which became effective in connection with the IPO. The ESPP permits participants to contribute up to a specified percentage of their eligible compensation during a series of offering periods of 24 months, each comprised of four six-month purchase periods, to purchase the Company’s common stock. The purchase price of the shares will be 85% of the fair market value of the Company’s common stock on the first day of trading of the applicable offering period or on the applicable purchase date, whichever is lower. A total of 1,260,000 shares of common stock was initially reserved for issuance under the ESPP. In addition, the number of shares of common stock available for issuance under the ESPP may be increased annually on the first day of each calendar year during the term of the ESPP, beginning in 2022, by an amount equal to the lesser of (i) 1% of the shares of common stock outstanding on the final day of the immediately preceding calendar year or (ii) such smaller number of shares as determined by the Company’s board of directors or an authorized committee of the board of directors. The Company recognized stock-based compensation expense related to the ESPP of $342,000 and $707,000 during the three months ended March 31, 2024 and 2023, respectively. As of March 31, 2024, the unrecognized compensation cost related to the ESPP was $1.5 million and is expected to be recognized as expense over approximately 1.69 years. As of March 31, 2024 and December 31, 2023, $317,000 and $46,000 has been withheld on behalf of employees for future purchase under the ESPP, respectively, and is included in accrued expenses and other current liabilities on the condensed consolidated balance sheets. No shares were issued and sold under the ESPP during the three months ended March 31, 2024 and 2023. Stock-based compensation expense The allocation of stock-based compensation for all stock awards was as follows (in thousands):
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
2024 |
|
|
2023 |
|
Research and development |
|
$ |
3,484 |
|
|
$ |
3,880 |
|
General and administrative |
|
|
3,364 |
|
|
|
2,965 |
|
Total |
|
$ |
6,848 |
|
|
$ |
6,845 |
|
Common stock reserved for future issuance Common stock reserved for future issuance consisted of the following as of March 31, 2024 and December 31, 2023:
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Stock options issued and outstanding |
|
|
34,647,123 |
|
|
|
24,970,957 |
|
Awards available for future grant |
|
|
13,204,701 |
|
|
|
15,342,797 |
|
Shares available for purchase under the ESPP |
|
|
2,080,681 |
|
|
|
2,080,681 |
|
Total |
|
|
49,932,505 |
|
|
|
42,394,435 |
|
|
| X |
- DefinitionThe entire disclosure for share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//718/tableOfContent
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (h)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (h)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (l)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_DisclosureOfCompensationRelatedCostsShareBasedPaymentsTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Leases
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Leases [Abstract] |
|
| Leases |
Note 10. Leases Operating leases The Company has facility leases for office space under non-cancellable and cancelable operating leases with various expiration dates through 2032 and equipment under a non-cancellable operating lease with a term expiring in 2026. Total lease costs were approximately $2.7 million and $3.1 million, including operating lease costs of $1.9 million and $1.9 million, variable lease costs of $916,000 and $1.2 million, and sublease income of $84,000 and $0, during the three months ended March 31, 2024 and 2023, respectively. The Company paid $2.1 million and $863,000 in cash for operating leases, net of sublease income, that were included in the operating activities section of the condensed consolidated statements of cash flows for the three months ended March 31, 2024 and 2023, respectively. The weighted-average remaining lease term and the weighted-average discount rate of the Company’s operating leases were 8.05 years and 8.96%, respectively, at March 31, 2024. The weighted-average remaining lease term and the weighted-average discount rate of the Company’s operating leases were 8.29 years and 8.95%, respectively, at December 31, 2023. The weighted-average remaining lease term does not include any renewal options at the election of the Company. The Company’s lease agreements do not contain any material residual value guarantees or material restrictive covenants. Facility leases In September 2020, the Company entered into a lease agreement for 59,407 square feet of laboratory and office space in San Diego, California, which represented a portion of a new facility that was under construction and which was subsequently amended in March 2021 to expand the rented premises by 18,421 square feet (the 2020 Lease). The construction and design of the asset was the primary responsibility of the lessor. The Company was involved in certain aspects of construction and design for certain interior features and leasehold improvements that is beneficial to the Company to better suit its business needs and intended purpose of the space. The lease is accounted for as an operating lease and commenced in August 2021. In April 2022, the 2020 Lease was modified to amend the rent commencement date from February 2022 to May 2022. The 2020 Lease, as amended, has a term of 10.75 years and includes aggregate monthly payments to the lessor of approximately $51.6 million beginning in May 2023 with a rent escalation clause, and a tenant improvement allowance of approximately $16.8 million. The Company is responsible for its share of operating expenses based on actual operating expenses incurred by the landlord. The 2020 Lease is cancellable at the Company’s request after the 84th month with 12 months written notice and a lump-sum cancellation payment of $2.5 million. The termination option has not been included in the Company's operating lease assets and liabilities. As discussed in Note 2, the Company provided a letter of credit to the lessor for $408,000, which expires July 29, 2032. In December 2021, the Company entered into a lease agreement for 29,542 square feet of office and laboratory space in South San Francisco, California. The lease is accounted for as an operating lease with the associated operating lease assets and liabilities recorded upon commencement, which occurred in July 2022. The non-cancellable operating lease has an initial term of 124 months with an option to extend the lease term by 5 years at the then-current market rates and includes aggregate monthly payments to the lessor of approximately $34.4 million beginning in November 2022 with a rent escalation clause and a tenant improvement allowance of approximately $8.2 million. The renewal option has not been included in the Company's operating lease assets and liabilities. The Company is responsible for its share of operating expenses based on actual operating expenses incurred by the landlord. The construction and design of the tenant improvements was the primary responsibility of the lessor. While the Company was involved in certain aspects of construction and design for certain interior features and leasehold improvements that is beneficial to the Company to better suit its business needs and intended purpose of the space, all construction was handled directly by the landlord. The Company was not deemed to be the accounting owner of the tenant improvements prior to or after the construction period. All payments made by the Company for landlord-owned tenant improvements were recorded as prepaid rent on the condensed consolidated balance sheets prior to lease commencement and included in the operating lease asset upon lease commencement. In February 2022, the expected project costs exceeded the tenant improvement allowances by $5.1 million, which was paid directly to the landlord by the Company and was recorded as prepaid rent in the condensed consolidated balance sheets and as a cash outflow from operating activities in the condensed consolidated statements of cash flows. Upon lease commencement, the $5.1 million of prepaid rent was included in the operating lease asset. The Company paid a security deposit of $874,000 in December 2021 that was recorded as other assets in the condensed consolidated balance sheets. In January 2024, the Company entered into an agreement to sublease the second floor of its corporate headquarters in San Diego, California. Pursuant to the agreement, the subleased space is approximately 10,000 square feet of office space with a sublease term of three years which includes an option for the subtenant to renew for an additional year and an early termination clause. The Company determined the sublease to be an operating lease. Therefore, the Company will recognize sublease income on a straight-line basis over the lease term in its condensed consolidated statements of operations and comprehensive loss as a reduction to operating lease costs because the sublease is outside of the Company's normal business operations. The Company will continue to account for the operating lease asset and related liability of the original lease as it did prior to the commencement of the sublease. The Company recorded a reduction to operating lease costs of $84,000 related to income from this sublease during the three months ended March 31, 2024. Future minimum lease payments under the operating leases with initial lease terms in excess of one year as of March 31, 2024 are as follows (in thousands):
|
|
|
|
Year ending December 31, |
|
|
2024 (remaining nine months) |
$ |
6,602 |
|
2025 |
|
9,024 |
|
2026 |
|
9,170 |
|
2027 |
|
9,199 |
|
2028 |
|
9,277 |
|
Thereafter |
|
35,098 |
|
Total lease payments |
$ |
78,370 |
|
Less: Amount representing interest |
|
(23,433 |
) |
Operating lease liabilities |
$ |
54,937 |
|
|
| X |
- References
+ Details
| Name: |
us-gaap_LeasesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for operating leases of lessee. Includes, but is not limited to, description of operating lease and maturity analysis of operating lease liability. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//842-20/tableOfContent
| Name: |
us-gaap_LesseeOperatingLeasesTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Commitments and Contingencies
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Commitments and Contingencies Disclosure [Abstract] |
|
| Commitments and Contingencies |
Note 11. Commitments and contingencies Liabilities for loss contingencies arising from claims, assessments, litigation, fines, penalties and other sources are recorded when it is probable that a liability has been incurred and the amount can be reasonably estimated. There are no matters currently outstanding for which any such liabilities have been accrued.
|
| X |
- References
+ Details
| Name: |
us-gaap_CommitmentsAndContingenciesDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for commitments and contingencies. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 440
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482648/440-10-50-4
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 450
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//450/tableOfContent
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 954
-SubTopic 440
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480327/954-440-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 440
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482648/440-10-50-4
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 440
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//440/tableOfContent
| Name: |
us-gaap_CommitmentsAndContingenciesDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Income Taxes
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Income Tax Disclosure [Abstract] |
|
| Income Taxes |
Note 12. Income taxes No provision for federal, state or foreign income taxes has been recorded for the three months ended March 31, 2024 and 2023. The Company has incurred net operating losses for all the periods presented and has not reflected any benefit of such net operating loss carryforwards in the accompanying condensed consolidated financial statements due to uncertainty around utilizing these tax attributes within their respective carryforward periods. The Company has recorded a full valuation allowance against all of its deferred tax assets as it is not more likely than not that such assets will be realized in the near future. The Company’s policy is to recognize interest expense and penalties related to income tax matters as tax expense. For the three months ended March 31, 2024 and 2023, the Company has not recognized any interest or penalties related to income taxes.
|
| X |
- DefinitionThe entire disclosure for income taxes. Disclosures may include net deferred tax liability or asset recognized in an enterprise's statement of financial position, net change during the year in the total valuation allowance, approximate tax effect of each type of temporary difference and carryforward that gives rise to a significant portion of deferred tax liabilities and deferred tax assets, utilization of a tax carryback, and tax uncertainties information. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 13
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-13
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(h)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//740/tableOfContent
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 14
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-14
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 21
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-21
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 270
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482526/740-270-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 17
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482685/740-10-50-17
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB TOPIC 6.I.5.Q1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479360/740-10-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SAB Topic 11.C)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479360/740-10-S99-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 740
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482603/740-30-50-2
| Name: |
us-gaap_IncomeTaxDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Net Loss Per Share
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Earnings Per Share [Abstract] |
|
| Net Loss Per Share |
Note 13. Net loss per share The following table summarizes the computation of basic and diluted net loss per share of the Company (in thousands, except share and per share data):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
|
|
|
|
|
2024 |
|
|
2023 |
|
Net loss |
|
|
|
|
|
|
$ |
(35,017 |
) |
|
$ |
(33,199 |
) |
Weighted-average shares of common stock used in computing net loss per share, basic and diluted |
|
|
|
|
|
|
|
151,161,741 |
|
|
|
149,504,216 |
|
Net loss per share, basic and diluted |
|
|
|
|
|
|
$ |
(0.23 |
) |
|
$ |
(0.22 |
) |
The Company’s potentially dilutive securities, which include options to purchase common stock, shares purchasable under the ESPP and common stock subject to repurchase related to options early exercised, have been excluded from the computation of diluted net loss per share as the effect would be to reduce the net loss per share. Therefore, the weighted-average number of common shares outstanding used to calculate both basic and diluted net loss per share is the same. The Company excluded the following potential common shares, presented as amounts outstanding at each period end, from the computation of diluted net loss per share for the periods indicated because including them would have had an anti-dilutive effect:
|
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
|
March 31, |
|
|
|
2024 |
|
|
|
2023 |
|
Options to purchase common stock |
|
|
34,647,123 |
|
|
|
|
25,892,271 |
|
Options early exercised subject to future vesting |
|
|
247,919 |
|
|
|
|
948,179 |
|
Estimated shares purchasable under the ESPP |
|
|
1,301,205 |
|
|
|
|
755,809 |
|
Total potentially dilutive shares |
|
|
36,196,247 |
|
|
|
|
27,596,259 |
|
|
| X |
- References
+ Details
| Name: |
us-gaap_EarningsPerShareAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for earnings per share. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//260/tableOfContent
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-3
| Name: |
us-gaap_EarningsPerShareTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Related Party Transactions
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Related Party Transactions [Abstract] |
|
| Related Party Transactions |
Note 14. Related party transactions Erasca Foundation In May 2021, the Company established the Erasca Foundation to provide support such as funding research, patient advocacy, patient support and education in underserved populations, and funding for other initiatives to positively impact society that align with the Company’s mission. The Company's chief executive officer and certain board members serve as directors of the Erasca Foundation and the Company's chief executive officer, chief financial officer and chief business officer, and general counsel are also officers of the Erasca Foundation. In April 2024, the Company donated $125,000 to the Erasca Foundation. Affini-T Therapeutics, Inc. The Company holds a $2.0 million equity investment in Affini-T. One of the Company’s board members is also a member of the board of Affini-T.
|
| X |
- DefinitionThe entire disclosure for related party transactions. Examples of related party transactions include transactions between (a) a parent company and its subsidiary; (b) subsidiaries of a common parent; (c) and entity and its principal owners; and (d) affiliates. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-5
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-6
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 235
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481062/946-235-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 235
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481062/946-235-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(g)(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(c))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(2)(e))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//850/tableOfContent
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-6
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-1
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 850
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483326/850-10-50-1
| Name: |
us-gaap_RelatedPartyTransactionsDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Subsequent Event
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Subsequent Events [Abstract] |
|
| Subsequent Event |
Note 15. Subsequent event On April 2, 2024, the Company completed the 2024 Private Placement in which the Company issued and sold 21,844,660 shares of common stock at a price of $2.06 per share. Net proceeds from the 2024 Private Placement, after deducting placement agent fees and estimated expenses, were $43.7 million.
|
| X |
- References
+ Details
| Name: |
us-gaap_SubsequentEventsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for significant events or transactions that occurred after the balance sheet date through the date the financial statements were issued or the date the financial statements were available to be issued. Examples include: the sale of a capital stock issue, purchase of a business, settlement of litigation, catastrophic loss, significant foreign exchange rate changes, loans to insiders or affiliates, and transactions not in the ordinary course of business. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 855
-Name Accounting Standards Codification
-Publisher FASB
-URI https://asc.fasb.org//855/tableOfContent
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 855
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483399/855-10-50-2
| Name: |
us-gaap_SubsequentEventsTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Summary of Significant Accounting Policies (Policies)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Accounting Policies [Abstract] |
|
| Use of Estimates |
Use of estimates The preparation of the Company’s condensed consolidated financial statements in conformity with US GAAP requires the Company to make estimates and assumptions that impact the reported amounts of assets, liabilities, expenses, and the disclosure of contingent assets and liabilities in the condensed consolidated financial statements and accompanying notes. Accounting estimates and management judgments reflected in the condensed consolidated financial statements include, but are not limited to, the accrual of research and development expenses, stock-based compensation expense, and the incremental borrowing rate for determining the operating lease asset and liability. Management evaluates its estimates on an ongoing basis. Although estimates are based on the Company’s historical experience, knowledge of current events, and actions it may undertake in the future, actual results may ultimately materially differ from these estimates and assumptions.
|
| Unaudited Interim Financial Information |
Unaudited interim financial information The accompanying condensed consolidated balance sheet as of March 31, 2024, the condensed consolidated statements of operations and comprehensive loss for the three months ended March 31, 2024 and 2023, the condensed consolidated statements of stockholders’ equity for the three months ended March 31, 2024 and 2023 and the condensed consolidated statements of cash flows for the three months ended March 31, 2024 and 2023 are unaudited. The unaudited condensed consolidated interim financial statements have been prepared on the same basis as the audited annual consolidated financial statements and, in the opinion of management, reflect all adjustments, which include only normal recurring adjustments, necessary for the fair statement of the Company’s condensed consolidated financial position as of March 31, 2024 and the condensed consolidated results of its operations and cash flows for the three months ended March 31, 2024 and 2023. The condensed consolidated financial data and other information disclosed in these notes related to the three months ended March 31, 2024 and 2023 are unaudited. The condensed consolidated results for the three months ended March 31, 2024 are not necessarily indicative of results to be expected for the year ending December 31, 2024, any other interim periods, or any future year or period. These unaudited condensed consolidated financial statements should be read in conjunction with the Company's audited consolidated financial statements included in its Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC on March 27, 2024.
|
| Concentration of Credit Risk and Off-Balance Sheet Risk |
Concentration of credit risk and off-balance sheet risk Financial instruments which potentially subject the Company to significant concentration of credit risk consist of cash and cash equivalents and marketable securities. The Company maintains deposits in federally insured financial institutions in excess of federally insured limits. The Company has not experienced any losses in such accounts, and management believes that the Company is not exposed to significant credit risk due to the financial position of the depository institutions in which those deposits are held. The Company’s investment policy includes guidelines for the quality of the related institutions and financial instruments and defines allowable investments that the Company may invest in, which the Company believes minimizes the exposure to concentration of credit risk.
|
| Cash, Cash Equivalents and Restricted Cash |
Cash, cash equivalents and restricted cash Cash and cash equivalents include cash in readily available checking and savings accounts, money market funds and commercial paper. The Company considers all highly liquid investments with an original maturity of three months or less from the date of purchase to be cash equivalents. The Company had deposited cash of $408,000 as of March 31, 2024 and December 31, 2023 to secure a letter of credit in connection with the lease of the Company’s facilities (see Note 10). The Company has classified the restricted cash as a noncurrent asset on its condensed consolidated balance sheets. The following table provides a reconciliation of cash, cash equivalents and restricted cash reported within the condensed consolidated balance sheets that sum to the total of the same amounts shown in the condensed consolidated statements of cash flows (in thousands):
|
|
|
|
|
|
|
|
|
March 31, |
|
|
2024 |
|
|
2023 |
|
Cash and cash equivalents |
$ |
76,726 |
|
|
$ |
272,756 |
|
Restricted cash |
|
408 |
|
|
|
408 |
|
Total cash, cash equivalents and restricted cash |
$ |
77,134 |
|
|
$ |
273,164 |
|
|
| Marketable Securities and Investments |
Marketable securities and investments The Company classifies all marketable securities as available-for-sale, as the sale of such securities may be required prior to maturity. Management determines the appropriate classification of its marketable securities at the time of purchase. Marketable securities with original maturities beyond three months at the date of purchase and which mature at, or less than 12 months from, the balance sheet date are classified as short-term marketable securities. Available-for-sale securities are carried at fair value, with the unrealized gains and losses reported as accumulated other comprehensive income (loss) until realized. The amortized cost of available-for-sale debt securities is adjusted for amortization of premiums and accretion of discounts to maturity. Such amortization and accretion are included in interest income. The Company regularly reviews all of its marketable securities for declines in fair value. The review includes the consideration of the cause of the impairment, including the creditworthiness of the security issuers, the number of securities in an unrealized loss position, the severity of the unrealized loss(es), whether the Company has the intent to sell the securities and whether it is more likely than not that the Company will be required to sell the securities before the recovery of their amortized cost basis. If the decline in fair value is due to credit-related factors, a loss is recognized in net income; whereas, if the decline in fair value is not due to credit-related factors, the loss is recorded in other comprehensive income (loss). Realized gains and losses on available-for-sale securities are included in other income or expense. The cost of securities sold is based on the specific identification method. Interest and dividends on securities classified as available-for-sale are included in interest income. Through its wholly-owned subsidiary, Erasca Ventures, the Company has also invested in equity securities of a company whose securities are not publicly traded and whose fair value is not readily available (see Notes 3 and 14). This investment is recorded using cost minus impairment, plus or minus changes in its estimated fair value resulting from observable price changes in orderly transactions for the identical or a similar investment of the same issuer. Investments in equity securities without readily determinable fair values are assessed for potential impairment on a quarterly basis based on qualitative factors. This investment is included in other assets in the Company's condensed consolidated balance sheets.
|
| Fair Value Measurements |
Fair value measurements Certain assets and liabilities are carried at fair value under US GAAP. Fair value is defined as an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. Financial assets and liabilities carried at fair value are classified and disclosed in one of the following three levels of the fair value hierarchy, of which the first two are considered observable and the last is considered unobservable: Level 1—Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities. Level 2—Quoted prices for similar assets and liabilities in active markets, quoted prices in markets that are not active, or inputs which are observable, either directly or indirectly, for substantially the full term of the asset or liability. Level 3—Prices or valuation techniques that require inputs that are both significant to the fair value measurement and unobservable (i.e., supported by little or no market activity).
|
| Recently Issued Accounting Pronouncements Not Yet Adopted |
Recently issued accounting pronouncements not yet adopted From time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that the Company adopts as of the specified effective date. The Company qualifies as an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012 (JOBS Act) and has elected not to “opt out” of the extended transition related to complying with new or revised accounting standards, which means that when a standard is issued or revised and it has different application dates for public and nonpublic companies, the Company can adopt the new or revised standard at the time nonpublic companies adopt the new or revised standard and can do so until such time that the Company either (i) irrevocably elects to “opt out” of such extended transition period or (ii) no longer qualifies as an emerging growth company. In November 2023, the FASB issued ASU 2023-07, Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures, which is intended to provide enhancements to segment disclosures, even for entities with only one reportable segment. In particular, the standard will require disclosures of significant segment expenses regularly provided to the chief operating decision maker and included within each reported measure of segment profit and loss. The standard will also require disclosure of all other segment items by reportable segment and a description of its composition. Finally, the standard will require disclosure of the title and position of the chief operating decision maker and an explanation of how the chief operating decision maker uses the reported measure(s) of segment profit or loss in assessing segment performance and deciding how to allocate resources. The standard is effective for annual periods beginning after December 15, 2023, and interim periods within annual periods beginning after December 15, 2024. Early adoption is permitted. Retrospective application to all prior periods presented in the financial statements is required. The Company is currently evaluating the impact of the standard on the presentation of its consolidated financial statements and related disclosures. In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures, which is intended to provide enhancements to annual income tax disclosures. In particular, the standard will require more detailed information in the income tax rate reconciliation, as well as the disclosure of income taxes paid disaggregated by jurisdiction, among other enhancements. The standard is effective for the Company in its annual period beginning after December 15, 2025 and early adoption is permitted. The standard allows for adoption on a prospective basis, with a retrospective option. The Company is currently evaluating the impact of the standard on the presentation of its consolidated financial statements and related disclosures.
|
| X |
- DefinitionUnaudited interim financial information.
| Name: |
eras_UnauditedInterimFinancialInformationPolicyTextBlock |
| Namespace Prefix: |
eras_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionEntity's cash and cash equivalents accounting policy with respect to restricted balances. Restrictions may include legally restricted deposits held as compensating balances against short-term borrowing arrangements, contracts entered into with others, or company statements of intention with regard to particular deposits; however, time deposits and short-term certificates of deposit are not generally included in legally restricted deposits. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(1)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-1
| Name: |
us-gaap_CashAndCashEquivalentsRestrictedCashAndCashEquivalentsPolicy |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for fair value measurements of financial and non-financial assets, liabilities and instruments classified in shareholders' equity. Disclosures include, but are not limited to, how an entity that manages a group of financial assets and liabilities on the basis of its net exposure measures the fair value of those assets and liabilities.
| Name: |
us-gaap_FairValueMeasurementPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for investment classified as marketable security. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 942
-SubTopic 320
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480832/942-320-50-5
| Name: |
us-gaap_MarketableSecuritiesPolicy |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy pertaining to new accounting pronouncements that may impact the entity's financial reporting. Includes, but is not limited to, quantification of the expected or actual impact.
| Name: |
us-gaap_NewAccountingPronouncementsPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDescription of accounting policies and methodologies used to estimate the entity's liability for off-balance sheet credit exposures and related charges for those credit exposures. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 310
-SubTopic 10
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481962/310-10-50-9
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 21
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479319/326-20-50-21
| Name: |
us-gaap_OffBalanceSheetCreditExposurePolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for the use of estimates in the preparation of financial statements in conformity with generally accepted accounting principles. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-9
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-4
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (c)
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 12
-SubTopic 10
-Topic 275
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-12
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482861/275-10-50-8
| Name: |
us-gaap_UseOfEstimates |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Summary of Significant Accounting Policies (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Accounting Policies [Abstract] |
|
| Reconciliation of Cash, Cash Equivalents and Restricted Cash |
The following table provides a reconciliation of cash, cash equivalents and restricted cash reported within the condensed consolidated balance sheets that sum to the total of the same amounts shown in the condensed consolidated statements of cash flows (in thousands):
|
|
|
|
|
|
|
|
|
March 31, |
|
|
2024 |
|
|
2023 |
|
Cash and cash equivalents |
$ |
76,726 |
|
|
$ |
272,756 |
|
Restricted cash |
|
408 |
|
|
|
408 |
|
Total cash, cash equivalents and restricted cash |
$ |
77,134 |
|
|
$ |
273,164 |
|
|
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of cash and cash equivalents restricted as to withdrawal or usage. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(1)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
| Name: |
us-gaap_ScheduleOfRestrictedCashAndCashEquivalentsTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Fair Value Measurements (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Fair Value Disclosures [Abstract] |
|
| Summary of Financial Assets Measured at Fair Value on Recurring Basis |
The following tables summarize the Company’s financial assets measured at fair value on a recurring basis and their respective input levels based on the fair value hierarchy (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value measurements as of March 31, 2024 using |
|
|
|
|
|
|
Quoted prices in |
|
|
Significant |
|
|
Significant |
|
|
|
|
|
|
active markets |
|
|
other |
|
|
unobservable |
|
|
|
March 31, |
|
|
for identical |
|
|
observable |
|
|
inputs |
|
|
|
2024 |
|
|
assets (level 1) |
|
|
inputs (level 2) |
|
|
(level 3) |
|
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds(1) |
|
$ |
58,324 |
|
|
$ |
58,324 |
|
|
$ |
— |
|
|
$ |
— |
|
Commercial paper(1) |
|
|
1,492 |
|
|
|
— |
|
|
|
1,492 |
|
|
|
— |
|
US treasury securities(2) |
|
|
73,644 |
|
|
|
73,644 |
|
|
|
— |
|
|
|
— |
|
US government agency securities(2) |
|
|
23,856 |
|
|
|
— |
|
|
|
23,856 |
|
|
|
— |
|
Corporate debt securities(2) |
|
|
22,926 |
|
|
|
— |
|
|
|
22,926 |
|
|
|
— |
|
Commercial paper(2) |
|
|
100,533 |
|
|
|
— |
|
|
|
100,533 |
|
|
|
— |
|
Total fair value of assets |
|
$ |
280,775 |
|
|
$ |
131,968 |
|
|
$ |
148,807 |
|
|
$ |
— |
|
(1)Included as cash and cash equivalents on the condensed consolidated balance sheets. (2)Included as short-term marketable securities on the condensed consolidated balance sheets.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value measurements as of December 31, 2023 using |
|
|
|
|
|
|
Quoted prices in |
|
|
Significant |
|
|
Significant |
|
|
|
|
|
|
active markets |
|
|
other |
|
|
unobservable |
|
|
|
December 31, |
|
|
for identical |
|
|
observable |
|
|
inputs |
|
|
|
2023 |
|
|
assets (level 1) |
|
|
inputs (level 2) |
|
|
(level 3) |
|
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds(1) |
|
$ |
83,101 |
|
|
$ |
83,101 |
|
|
$ |
— |
|
|
$ |
— |
|
US treasury securities(2) |
|
|
93,303 |
|
|
|
93,303 |
|
|
|
— |
|
|
|
— |
|
US government agency securities(2) |
|
|
26,824 |
|
|
|
— |
|
|
|
26,824 |
|
|
|
— |
|
Corporate debt securities(2) |
|
|
10,734 |
|
|
|
— |
|
|
|
10,734 |
|
|
|
— |
|
Commercial paper(2) |
|
|
88,414 |
|
|
|
— |
|
|
|
88,414 |
|
|
|
— |
|
US treasury securities(3) |
|
|
9,642 |
|
|
|
9,642 |
|
|
|
— |
|
|
|
— |
|
Total fair value of assets |
|
$ |
312,018 |
|
|
$ |
186,046 |
|
|
$ |
125,972 |
|
|
$ |
— |
|
(1)Included as cash and cash equivalents on the condensed consolidated balance sheets. (2)Included as short-term marketable securities on the condensed consolidated balance sheets. (3)Included as long-term marketable securities on the condensed consolidated balance sheets.
|
| X |
- DefinitionTabular disclosure of assets, including [financial] instruments measured at fair value that are classified in stockholders' equity, if any, by class that are measured at fair value on a recurring basis. The disclosures contemplated herein include the fair value measurements at the reporting date by the level within the fair value hierarchy in which the fair value measurements in their entirety fall, segregating fair value measurements using quoted prices in active markets for identical assets (Level 1), significant other observable inputs (Level 2), and significant unobservable inputs (Level 3). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 820
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 820
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
| Name: |
us-gaap_FairValueAssetsMeasuredOnRecurringBasisTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_FairValueDisclosuresAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Marketable Securities (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Investments, Debt and Equity Securities [Abstract] |
|
| Summary of Marketable Securities Accounted for Available-for-Sale Securities |
The following tables summarize the Company’s marketable securities accounted for as available-for-sale securities (in thousands, except years):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2024 |
|
|
|
Maturity |
|
Amortized |
|
|
Unrealized |
|
|
Unrealized |
|
|
Estimated |
|
|
|
(in years) |
|
cost |
|
|
gains |
|
|
losses |
|
|
fair value |
|
US treasury securities |
|
1 or less |
|
$ |
73,753 |
|
|
$ |
8 |
|
|
$ |
(117 |
) |
|
$ |
73,644 |
|
US government agency securities |
|
1 or less |
|
|
23,859 |
|
|
|
11 |
|
|
|
(14 |
) |
|
|
23,856 |
|
Corporate debt securities |
|
1 or less |
|
|
22,967 |
|
|
|
— |
|
|
|
(41 |
) |
|
|
22,926 |
|
Commercial paper |
|
1 or less |
|
|
100,590 |
|
|
|
16 |
|
|
|
(73 |
) |
|
|
100,533 |
|
Total |
|
|
|
$ |
221,169 |
|
|
$ |
35 |
|
|
$ |
(245 |
) |
|
$ |
220,959 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2023 |
|
|
|
Maturity |
|
Amortized |
|
|
Unrealized |
|
|
Unrealized |
|
|
Estimated |
|
|
|
(in years) |
|
cost |
|
|
gains |
|
|
losses |
|
|
fair value |
|
US treasury securities |
|
1 or less |
|
$ |
93,377 |
|
|
$ |
74 |
|
|
$ |
(148 |
) |
|
$ |
93,303 |
|
US government agency securities |
|
1 or less |
|
|
26,783 |
|
|
|
45 |
|
|
|
(4 |
) |
|
|
26,824 |
|
Corporate debt securities |
|
1 or less |
|
|
10,719 |
|
|
|
15 |
|
|
|
— |
|
|
|
10,734 |
|
Commercial paper |
|
1 or less |
|
|
88,356 |
|
|
|
68 |
|
|
|
(10 |
) |
|
|
88,414 |
|
US treasury securities |
|
1-2 |
|
|
9,605 |
|
|
|
37 |
|
|
|
— |
|
|
|
9,642 |
|
Total |
|
|
|
$ |
228,840 |
|
|
$ |
239 |
|
|
$ |
(162 |
) |
|
$ |
228,917 |
|
|
| Summary of Fair Values and Gross Unrealized Losses for Available-for-sale Securities |
The following tables present fair values and gross unrealized losses for those available-for-sale securities that were in an unrealized loss position as of March 31, 2024 and December 31, 2023, aggregated by category and the length of time that the securities have been in a continuous loss position (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2024 |
|
|
|
Unrealized losses less than 12 months |
|
|
Unrealized losses 12 months or greater |
|
|
Total |
|
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
US treasury securities |
|
$ |
55,139 |
|
|
$ |
(117 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
55,139 |
|
|
$ |
(117 |
) |
US government agency securities |
|
|
18,845 |
|
|
|
(14 |
) |
|
|
— |
|
|
|
— |
|
|
|
18,845 |
|
|
|
(14 |
) |
Corporate debt securities |
|
|
22,926 |
|
|
|
(41 |
) |
|
|
— |
|
|
|
— |
|
|
|
22,926 |
|
|
|
(41 |
) |
Commercial paper |
|
|
65,574 |
|
|
|
(73 |
) |
|
|
— |
|
|
|
— |
|
|
|
65,574 |
|
|
|
(73 |
) |
Total |
|
$ |
162,484 |
|
|
$ |
(245 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
162,484 |
|
|
$ |
(245 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2023 |
|
|
|
Unrealized losses less than 12 months |
|
|
Unrealized losses 12 months or greater |
|
|
Total |
|
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
|
Fair value |
|
|
Unrealized losses |
|
US treasury securities |
|
$ |
74,912 |
|
|
$ |
(148 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
74,912 |
|
|
$ |
(148 |
) |
US government agency securities |
|
|
6,950 |
|
|
|
(4 |
) |
|
|
— |
|
|
|
— |
|
|
|
6,950 |
|
|
|
(4 |
) |
Corporate debt securities |
|
|
748 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
748 |
|
|
|
— |
|
Commercial paper |
|
|
22,944 |
|
|
|
(10 |
) |
|
|
— |
|
|
|
— |
|
|
|
22,944 |
|
|
|
(10 |
) |
Total |
|
$ |
105,554 |
|
|
$ |
(162 |
) |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
105,554 |
|
|
$ |
(162 |
) |
|
| X |
- References
+ Details
| Name: |
us-gaap_InvestmentsDebtAndEquitySecuritiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of marketable securities. This may consist of investments in certain debt and equity securities, short-term investments and other assets.
| Name: |
us-gaap_MarketableSecuritiesTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor all investments in an unrealized loss position, including those for which other-than-temporary impairments have not been recognized in earnings (including investments for which a portion of an other-than-temporary impairment has been recognized in other comprehensive income), a tabular disclosure of the aggregate related fair value of investments with unrealized losses and the aggregate amount of unrealized losses (that is, the amount by which amortized cost basis exceeds fair value). Reference 1: http://fasb.org/us-gaap/role/ref/otherTransitionRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-6
| Name: |
us-gaap_ScheduleOfUnrealizedLossOnInvestmentsTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Property and Equipment, Net (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Property, Plant and Equipment [Abstract] |
|
| Schedule of Property and Equipment, Net |
Property and equipment, net consisted of the following (in thousands):
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Laboratory equipment |
|
$ |
5,621 |
|
|
$ |
5,620 |
|
Furniture and fixtures |
|
|
4,099 |
|
|
|
4,099 |
|
Leasehold improvements |
|
|
18,173 |
|
|
|
18,173 |
|
Computer equipment and software |
|
|
1,695 |
|
|
|
1,667 |
|
Property and equipment |
|
|
29,588 |
|
|
|
29,559 |
|
Less accumulated depreciation and amortization |
|
|
(8,230 |
) |
|
|
(7,232 |
) |
Property and equipment, net |
|
$ |
21,358 |
|
|
$ |
22,327 |
|
|
| X |
- References
+ Details
| Name: |
us-gaap_PropertyPlantAndEquipmentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of physical assets used in the normal conduct of business and not intended for resale. Includes, but is not limited to, balances by class of assets, depreciation and depletion expense and method used, including composite depreciation, and accumulated deprecation. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Accrued Expenses and Other Current Liabilities (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Payables and Accruals [Abstract] |
|
| Schedule of Accrued Expenses and Other Current Liabilities |
Accrued expenses and other current liabilities consisted of the following (in thousands):
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Accrued research and development expenses |
|
$ |
9,247 |
|
|
$ |
8,803 |
|
Accrued compensation |
|
|
6,095 |
|
|
|
10,311 |
|
Unvested early exercised stock option liability |
|
|
309 |
|
|
|
464 |
|
Accrued professional services |
|
|
626 |
|
|
|
435 |
|
Accrued offering costs |
|
|
178 |
|
|
|
— |
|
Accrued property and equipment |
|
|
— |
|
|
|
13 |
|
Other accruals |
|
|
122 |
|
|
|
160 |
|
Total |
|
$ |
16,577 |
|
|
$ |
20,186 |
|
|
| X |
- References
+ Details
| Name: |
us-gaap_PayablesAndAccrualsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of the (a) carrying value as of the balance sheet date of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business (accounts payable); (b) other payables; and (c) accrued liabilities. Examples include taxes, interest, rent and utilities. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer). An alternative caption includes accrued expenses.
| Name: |
us-gaap_ScheduleOfAccountsPayableAndAccruedLiabilitiesTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Stock-based Compensation (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Share-Based Compensation Arrangement by Share-Based Payment Award [Line Items] |
|
| Summary of Stock Option Activity |
A summary of the Company’s stock option activity under the 2021 Plan and 2018 Plan is as follows (in thousands, except share and per share data and years):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
average remaining |
|
|
Aggregate |
|
|
|
|
|
|
average |
|
|
contractual |
|
|
intrinsic |
|
|
|
Shares |
|
|
exercise price |
|
|
term (years) |
|
|
value |
|
Outstanding at December 31, 2023 |
|
|
24,970,957 |
|
|
$ |
4.98 |
|
|
|
8.12 |
|
|
$ |
4,412 |
|
Granted |
|
|
9,827,650 |
|
|
|
1.71 |
|
|
|
|
|
|
|
Exercised |
|
|
(35,035 |
) |
|
|
0.69 |
|
|
|
|
|
|
|
Canceled |
|
|
(116,449 |
) |
|
|
4.89 |
|
|
|
|
|
|
|
Outstanding at March 31, 2024 |
|
|
34,647,123 |
|
|
$ |
4.06 |
|
|
|
8.44 |
|
|
$ |
7,532 |
|
Options exercisable at March 31, 2024 |
|
|
12,850,952 |
|
|
$ |
4.83 |
|
|
|
7.32 |
|
|
$ |
3,916 |
|
|
| Schedule of Stock-based Compensation Expense |
The allocation of stock-based compensation for all stock awards was as follows (in thousands):
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
2024 |
|
|
2023 |
|
Research and development |
|
$ |
3,484 |
|
|
$ |
3,880 |
|
General and administrative |
|
|
3,364 |
|
|
|
2,965 |
|
Total |
|
$ |
6,848 |
|
|
$ |
6,845 |
|
|
| Summary of Common Stock Reserved for Future Issuance |
Common stock reserved for future issuance consisted of the following as of March 31, 2024 and December 31, 2023:
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
|
|
2024 |
|
|
2023 |
|
Stock options issued and outstanding |
|
|
34,647,123 |
|
|
|
24,970,957 |
|
Awards available for future grant |
|
|
13,204,701 |
|
|
|
15,342,797 |
|
Shares available for purchase under the ESPP |
|
|
2,080,681 |
|
|
|
2,080,681 |
|
Total |
|
|
49,932,505 |
|
|
|
42,394,435 |
|
|
| Employee Stock Option |
|
| Share-Based Compensation Arrangement by Share-Based Payment Award [Line Items] |
|
| Assumptions used to Determine Fair Value of Stock Option Grants |
The assumptions used in the Black-Scholes option pricing model to determine the fair value of the employee and nonemployee stock option grants were as follows:
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
2024 |
|
2023 |
Risk-free interest rate |
|
3.84%-4.15% |
|
3.46%-4.22% |
Expected volatility |
|
83.35%-83.91% |
|
85.52%-85.81% |
Expected term (in years) |
|
5.28-6.01 |
|
6.02-6.07 |
Expected dividend yield |
|
--% |
|
--% |
|
| X |
- DefinitionCommon stock reserved for future issuance.
| Name: |
eras_CommonStockReservedForFutureIssuanceTableTextBlock |
| Namespace Prefix: |
eras_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of allocation of amount expensed and capitalized for award under share-based payment arrangement to statement of income or comprehensive income and statement of financial position. Includes, but is not limited to, corresponding line item in financial statement. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (h)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ScheduleOfEmployeeServiceShareBasedCompensationAllocationOfRecognizedPeriodCostsTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure for stock option plans. Includes, but is not limited to, outstanding awards at beginning and end of year, grants, exercises, forfeitures, and weighted-average grant date fair value. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (c)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)
-SubTopic 10
-Topic 718
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)
-SubTopic 10
-Topic 718
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ScheduleOfShareBasedCompensationStockOptionsActivityTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of the significant assumptions used during the year to estimate the fair value of stock options, including, but not limited to: (a) expected term of share options and similar instruments, (b) expected volatility of the entity's shares, (c) expected dividends, (d) risk-free rate(s), and (e) discount for post-vesting restrictions. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 718
-SubTopic 10
-Subparagraph (f)(2)
-Name Accounting Standards Codification
-Paragraph 2
-Section 50
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ScheduleOfShareBasedPaymentAwardStockOptionsValuationAssumptionsTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 1D
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-1D
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=us-gaap_EmployeeStockOptionMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Leases (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Leases [Abstract] |
|
| Schedule of Future Minimum Lease Payments under Operating Leases |
Future minimum lease payments under the operating leases with initial lease terms in excess of one year as of March 31, 2024 are as follows (in thousands):
|
|
|
|
Year ending December 31, |
|
|
2024 (remaining nine months) |
$ |
6,602 |
|
2025 |
|
9,024 |
|
2026 |
|
9,170 |
|
2027 |
|
9,199 |
|
2028 |
|
9,277 |
|
Thereafter |
|
35,098 |
|
Total lease payments |
$ |
78,370 |
|
Less: Amount representing interest |
|
(23,433 |
) |
Operating lease liabilities |
$ |
54,937 |
|
|
| X |
- References
+ Details
| Name: |
us-gaap_LeasesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of undiscounted cash flows of lessee's operating lease liability. Includes, but is not limited to, reconciliation of undiscounted cash flows to operating lease liability recognized in statement of financial position. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityMaturityTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Net Loss Per Share (Tables)
|
3 Months Ended |
Mar. 31, 2024 |
|---|
| Earnings Per Share [Abstract] |
|
| Summary of Computation of Basic and Diluted Net Loss Per Share |
The following table summarizes the computation of basic and diluted net loss per share of the Company (in thousands, except share and per share data):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
|
|
|
|
|
2024 |
|
|
2023 |
|
Net loss |
|
|
|
|
|
|
$ |
(35,017 |
) |
|
$ |
(33,199 |
) |
Weighted-average shares of common stock used in computing net loss per share, basic and diluted |
|
|
|
|
|
|
|
151,161,741 |
|
|
|
149,504,216 |
|
Net loss per share, basic and diluted |
|
|
|
|
|
|
$ |
(0.23 |
) |
|
$ |
(0.22 |
) |
|
| Anti-dilutive Securities Excluded from Calculation of Diluted Net Loss Per Share |
The Company’s potentially dilutive securities, which include options to purchase common stock, shares purchasable under the ESPP and common stock subject to repurchase related to options early exercised, have been excluded from the computation of diluted net loss per share as the effect would be to reduce the net loss per share. Therefore, the weighted-average number of common shares outstanding used to calculate both basic and diluted net loss per share is the same. The Company excluded the following potential common shares, presented as amounts outstanding at each period end, from the computation of diluted net loss per share for the periods indicated because including them would have had an anti-dilutive effect:
|
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
|
March 31, |
|
|
|
2024 |
|
|
|
2023 |
|
Options to purchase common stock |
|
|
34,647,123 |
|
|
|
|
25,892,271 |
|
Options early exercised subject to future vesting |
|
|
247,919 |
|
|
|
|
948,179 |
|
Estimated shares purchasable under the ESPP |
|
|
1,301,205 |
|
|
|
|
755,809 |
|
Total potentially dilutive shares |
|
|
36,196,247 |
|
|
|
|
27,596,259 |
|
|
| X |
- References
+ Details
| Name: |
us-gaap_EarningsPerShareAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of securities (including those issuable pursuant to contingent stock agreements) that could potentially dilute basic earnings per share (EPS) in the future that were not included in the computation of diluted EPS because to do so would increase EPS amounts or decrease loss per share amounts for the period presented, by antidilutive securities. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
| Name: |
us-gaap_ScheduleOfAntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTabular disclosure of an entity's basic and diluted earnings per share calculations, including a reconciliation of numerators and denominators of the basic and diluted per-share computations for income from continuing operations. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
| Name: |
us-gaap_ScheduleOfEarningsPerShareBasicAndDilutedTableTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Organization and Basis of Presentation - Additional Information (Details) - USD ($)
$ / shares in Units, $ in Thousands |
|
1 Months Ended |
|
Apr. 02, 2024 |
Mar. 31, 2024 |
Dec. 31, 2023 |
| Organization And Basis Of Presentation [Line Items] |
|
|
|
| Cash, cash equivalents, and short-term marketable securities |
|
$ 297,700
|
|
| Accumulated deficit |
|
(641,030)
|
$ (606,013)
|
| Proceeds from prepayment of private placement |
|
6,907
|
|
| 2024 Private Placement |
|
|
|
| Organization And Basis Of Presentation [Line Items] |
|
|
|
| Proceeds from prepayment of private placement |
|
$ 6,900
|
|
| 2024 Private Placement | Subsequent Event |
|
|
|
| Organization And Basis Of Presentation [Line Items] |
|
|
|
| Net proceeds from private placement, after deducting placement agent fees and estimated expenses |
$ 43,700
|
|
|
| Placement agent fees and estimated expenses |
$ 1,300
|
|
|
| Common Stock | 2024 Private Placement |
|
|
|
| Organization And Basis Of Presentation [Line Items] |
|
|
|
| Stock issued and sold |
|
21,844,660
|
|
| Price per share |
|
$ 2.06
|
|
| Common Stock | 2024 Private Placement | Subsequent Event |
|
|
|
| Organization And Basis Of Presentation [Line Items] |
|
|
|
| Stock issued and sold |
21,844,660
|
|
|
| Price per share |
$ 2.06
|
|
|
| X |
- DefinitionLiability from prepayment of private placement.
| Name: |
eras_LiabilityFromPrepaymentOfPrivatePlacementCurrent |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionOrganization and basis of presentation.
| Name: |
eras_OrganizationAndBasisOfPresentationLineItems |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionProceeds from issuance of private placement after deducting placement agent fees and estimated expenses.
| Name: |
eras_ProceedsFromIssuanceOfPrivatePlacementAfterDeductingPlacementAgentFeesAndEstimatedExpenses |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionCash includes currency on hand as well as demand deposits with banks or financial institutions. It also includes other kinds of accounts that have the general characteristics of demand deposits in that the customer may deposit additional funds at any time and effectively may withdraw funds at any time without prior notice or penalty. Cash equivalents, excluding items classified as marketable securities, include short-term, highly liquid Investments that are both readily convertible to known amounts of cash, and so near their maturity that they present minimal risk of changes in value because of changes in interest rates. Generally, only investments with original maturities of three months or less qualify under that definition. Original maturity means original maturity to the entity holding the investment. For example, both a three-month US Treasury bill and a three-year Treasury note purchased three months from maturity qualify as cash equivalents. However, a Treasury note purchased three years ago does not become a cash equivalent when its remaining maturity is three months. Short-term investments, exclusive of cash equivalents, generally consist of marketable securities intended to be sold within one year (or the normal operating cycle if longer) and may include trading securities, available-for-sale securities, or held-to-maturity securities (if maturing within one year), as applicable. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CashCashEquivalentsAndShortTermInvestments |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash outflow for cost incurred directly with the issuance of an equity security. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 15
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-15
| Name: |
us-gaap_PaymentsOfStockIssuanceCosts |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of accumulated undistributed earnings (deficit). Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (g)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 2
-Subparagraph (h)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480016/944-40-65-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480990/946-20-50-11
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(23)(a)(4))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-04(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479617/946-210-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(30)(a)(3))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_RetainedEarningsAccumulatedDeficit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionPer share amount received by subsidiary or equity investee for each share of common stock issued or sold in the stock transaction.
| Name: |
us-gaap_SaleOfStockPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=eras_TwoThousandAndTwentyFourPrivatePlacementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsequentEventTypeAxis=us-gaap_SubsequentEventMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash restricted as to withdrawal or usage, classified as noncurrent. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 5
-SubTopic 210
-Topic 954
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480632/954-210-45-5
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
| Name: |
us-gaap_RestrictedCashNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
v3.24.1.u1
Summary of Significant Accounting Policies - Reconciliation of Cash, Cash Equivalents and Restricted Cash (Details) - USD ($)
$ in Thousands |
Mar. 31, 2024 |
Dec. 31, 2023 |
Mar. 31, 2023 |
| Cash, Cash Equivalents, Restricted Cash, and Restricted Cash Equivalents [Abstract] |
|
|
|
| Cash and cash equivalents |
$ 76,726
|
$ 93,075
|
$ 272,756
|
| Restricted cash |
408
|
|
408
|
| Total cash, cash equivalents and restricted cash |
$ 77,134
|
|
$ 273,164
|
| X |
- DefinitionAmount of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-4
| Name: |
us-gaap_CashAndCashEquivalentsAtCarryingValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of cash and cash equivalents, and cash and cash equivalents restricted to withdrawal or usage. Excludes amount for disposal group and discontinued operations. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 24
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-24
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-4
| Name: |
us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalents |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_CashCashEquivalentsRestrictedCashAndRestrictedCashEquivalentsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash and cash equivalents restricted as to withdrawal or usage. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Cash equivalents include, but are not limited to, short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(2))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(1)(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-4
| Name: |
us-gaap_RestrictedCashAndCashEquivalents |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
v3.24.1.u1
Fair Value Measurements - Summary of Financial Assets Measured at Fair Value on Recurring Basis (Details) - Fair Value, Recurring - USD ($)
$ in Thousands |
Mar. 31, 2024 |
Dec. 31, 2023 |
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
$ 280,775
|
|
$ 312,018
|
|
| Money Market Funds | Cash and Cash Equivalents |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
58,324
|
[1] |
83,101
|
[2] |
| Commercial Paper | Cash and Cash Equivalents |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
[1] |
1,492
|
|
|
|
| Commercial Paper | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
100,533
|
[3] |
88,414
|
[4] |
| US Treasury Securities | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
73,644
|
[3] |
93,303
|
[4] |
| US Treasury Securities | Long-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
[5] |
|
|
9,642
|
|
| US Government Agency Securities | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
23,856
|
[3] |
26,824
|
[4] |
| Corporate Debt Securities | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
22,926
|
[3] |
10,734
|
[4] |
| Quoted Prices in Active Markets for Identical Assets (Level 1) |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
131,968
|
|
186,046
|
|
| Quoted Prices in Active Markets for Identical Assets (Level 1) | Money Market Funds | Cash and Cash Equivalents |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
58,324
|
[1] |
83,101
|
[2] |
| Quoted Prices in Active Markets for Identical Assets (Level 1) | US Treasury Securities | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
73,644
|
[3] |
93,303
|
[4] |
| Quoted Prices in Active Markets for Identical Assets (Level 1) | US Treasury Securities | Long-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
[5] |
|
|
9,642
|
|
| Significant Other Observable Inputs (Level 2) |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
148,807
|
|
125,972
|
|
| Significant Other Observable Inputs (Level 2) | Commercial Paper | Cash and Cash Equivalents |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
[1] |
1,492
|
|
|
|
| Significant Other Observable Inputs (Level 2) | Commercial Paper | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
100,533
|
[3] |
88,414
|
[4] |
| Significant Other Observable Inputs (Level 2) | US Government Agency Securities | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
23,856
|
[3] |
26,824
|
[4] |
| Significant Other Observable Inputs (Level 2) | Corporate Debt Securities | Short-Term Investments |
|
|
|
|
|
| Assets: |
|
|
|
|
|
| Total fair value of assets |
|
$ 22,926
|
[3] |
$ 10,734
|
[4] |
|
|
| X |
- DefinitionFair value portion of probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 820
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
| Name: |
us-gaap_AssetsFairValueDisclosure |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_AssetsFairValueDisclosureAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_FairValueByMeasurementFrequencyAxis=us-gaap_FairValueMeasurementsRecurringMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FairValueByAssetClassAxis=us-gaap_MoneyMarketFundsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_InvestmentTypeAxis=us-gaap_CashAndCashEquivalentsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FairValueByAssetClassAxis=us-gaap_CommercialPaperMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_InvestmentTypeAxis=us-gaap_ShortTermInvestmentsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FairValueByAssetClassAxis=us-gaap_USTreasurySecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_InvestmentTypeAxis=eras_LongTermInvestmentsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FairValueByAssetClassAxis=us-gaap_USGovernmentCorporationsAndAgenciesSecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FairValueByAssetClassAxis=us-gaap_CorporateDebtSecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionEquity securities without readily determinable fair value, impairment or price adjustment amount.
| Name: |
eras_EquitySecuritiesWithoutReadilyDeterminableFairValueImpairmentOrPriceAdjustmentAmount |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of investment in equity security without readily determinable fair value. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 321
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479536/321-10-50-3
| Name: |
us-gaap_EquitySecuritiesWithoutReadilyDeterminableFairValueAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
v3.24.1.u1
Marketable Securities - Summary of Marketable Securities Accounted for Available-for-Sale-Securities (Details) - USD ($)
$ in Thousands |
Mar. 31, 2024 |
Dec. 31, 2023 |
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Available for sale securities, Amortized cost |
$ 221,169
|
$ 228,840
|
| Available for sale securities, Unrealized gains |
35
|
239
|
| Available for sale securities, Unrealized losses |
(245)
|
(162)
|
| Available for sale securities, Estimated fair value |
220,959
|
228,917
|
| US Treasury Securities |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Available for sale securities, Amortized cost |
73,753
|
93,377
|
| Available for sale securities, Unrealized gains |
8
|
74
|
| Available for sale securities, Unrealized losses |
(117)
|
(148)
|
| Available for sale securities, Estimated fair value |
$ 73,644
|
$ 93,303
|
| US Treasury Securities | Maximum |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Maturity |
1 year
|
1 year
|
| US Government Agency Securities |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Available for sale securities, Amortized cost |
$ 23,859
|
$ 26,783
|
| Available for sale securities, Unrealized gains |
11
|
45
|
| Available for sale securities, Unrealized losses |
(14)
|
(4)
|
| Available for sale securities, Estimated fair value |
$ 23,856
|
$ 26,824
|
| US Government Agency Securities | Maximum |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Maturity |
1 year
|
1 year
|
| Corporate Debt Securities |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Available for sale securities, Amortized cost |
$ 22,967
|
$ 10,719
|
| Available for sale securities, Unrealized gains |
|
15
|
| Available for sale securities, Unrealized losses |
(41)
|
|
| Available for sale securities, Estimated fair value |
$ 22,926
|
$ 10,734
|
| Corporate Debt Securities | Maximum |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Maturity |
1 year
|
1 year
|
| Commercial Paper |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Available for sale securities, Amortized cost |
$ 100,590
|
$ 88,356
|
| Available for sale securities, Unrealized gains |
16
|
68
|
| Available for sale securities, Unrealized losses |
(73)
|
(10)
|
| Available for sale securities, Estimated fair value |
$ 100,533
|
$ 88,414
|
| Commercial Paper | Maximum |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Maturity |
1 year
|
1 year
|
| US Treasury Securities |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Available for sale securities, Amortized cost |
|
$ 9,605
|
| Available for sale securities, Unrealized gains |
|
37
|
| Available for sale securities, Estimated fair value |
|
$ 9,642
|
| US Treasury Securities | Minimum |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Maturity |
|
1 year
|
| US Treasury Securities | Maximum |
|
|
| Schedule Of Available For Sale Securities [Line Items] |
|
|
| Maturity |
|
2 years
|
| X |
- DefinitionAmount, before tax, of unrealized gain in accumulated other comprehensive income (AOCI) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
| Name: |
us-gaap_AvailableForSaleDebtSecuritiesAccumulatedGrossUnrealizedGainBeforeTax |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount, before tax, of unrealized loss in accumulated other comprehensive income (AOCI) on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
| Name: |
us-gaap_AvailableForSaleDebtSecuritiesAccumulatedGrossUnrealizedLossBeforeTax |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmortized cost of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479130/326-30-45-1
| Name: |
us-gaap_AvailableForSaleDebtSecuritiesAmortizedCostBasis |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (aa)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481830/320-10-45-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-03(6))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479853/942-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479130/326-30-45-1
| Name: |
us-gaap_AvailableForSaleSecuritiesDebtSecurities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionPeriod between issuance and maturity of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 820
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (bbb)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482106/820-10-50-2
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleTerm |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (aa)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (aaa)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
| Name: |
us-gaap_ScheduleOfAvailableForSaleSecuritiesLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=eras_UsTreasurySecuritiesShortTermMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MaximumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=eras_UsGovernmentAgenciesShortTermSecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=eras_CorporatedebtsecuritiesshorttermMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=eras_CommercialpapershorttermMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=eras_UsTreasurySecuritiesLongTermMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MinimumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Marketable Securities - Summary of Fair Values and Gross Unrealized Losses for Available-for-sale Securities (Details) - USD ($)
$ in Thousands |
Mar. 31, 2024 |
Dec. 31, 2023 |
| Debt Securities, Available-for-Sale [Line Items] |
|
|
| Unrealized losses less than 12 months, Fair value |
$ 162,484
|
$ 105,554
|
| Unrealized losses less than 12 months, Unrealized losses |
(245)
|
(162)
|
| Fair value, Total |
162,484
|
105,554
|
| Unrealized losses, Total |
(245)
|
(162)
|
| US Treasury Securities |
|
|
| Debt Securities, Available-for-Sale [Line Items] |
|
|
| Unrealized losses less than 12 months, Fair value |
55,139
|
74,912
|
| Unrealized losses less than 12 months, Unrealized losses |
(117)
|
(148)
|
| Fair value, Total |
55,139
|
74,912
|
| Unrealized losses, Total |
(117)
|
(148)
|
| US Government Agency Securities |
|
|
| Debt Securities, Available-for-Sale [Line Items] |
|
|
| Unrealized losses less than 12 months, Fair value |
18,845
|
6,950
|
| Unrealized losses less than 12 months, Unrealized losses |
(14)
|
(4)
|
| Fair value, Total |
18,845
|
6,950
|
| Unrealized losses, Total |
(14)
|
(4)
|
| Corporate Debt Securities |
|
|
| Debt Securities, Available-for-Sale [Line Items] |
|
|
| Unrealized losses less than 12 months, Fair value |
22,926
|
748
|
| Unrealized losses less than 12 months, Unrealized losses |
(41)
|
|
| Fair value, Total |
22,926
|
748
|
| Unrealized losses, Total |
(41)
|
|
| Commercial Paper |
|
|
| Debt Securities, Available-for-Sale [Line Items] |
|
|
| Unrealized losses less than 12 months, Fair value |
65,574
|
22,944
|
| Unrealized losses less than 12 months, Unrealized losses |
(73)
|
(10)
|
| Fair value, Total |
65,574
|
22,944
|
| Unrealized losses, Total |
$ (73)
|
$ (10)
|
| X |
- DefinitionAmount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in continuous unrealized loss position for less than 12 months, without allowance for credit loss. Includes beneficial interest in securitized financial asset. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 55
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479081/326-30-55-8
Reference 2: http://fasb.org/us-gaap/role/ref/otherTransitionRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-7
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-5
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleContinuousUnrealizedLossPositionLessThan12Months |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of accumulated unrealized loss on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in continuous unrealized loss position for less than 12 months, without allowance for credit loss. Includes beneficial interest in securitized financial asset. Reference 1: http://fasb.org/us-gaap/role/ref/otherTransitionRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-7
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 5
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-5
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleContinuousUnrealizedLossPositionLessThan12MonthsAccumulatedLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in unrealized loss position without allowance for credit loss. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 55
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479081/326-30-55-8
Reference 2: http://fasb.org/us-gaap/role/ref/otherTransitionRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-6
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-4
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleUnrealizedLossPosition |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of accumulated unrealized loss on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in unrealized loss position, without allowance for credit loss. Includes beneficial interest in securitized financial asset. Reference 1: http://fasb.org/us-gaap/role/ref/otherTransitionRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-6
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-4
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleUnrealizedLossPositionAccumulatedLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (aa)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (aaa)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-3
| Name: |
us-gaap_ScheduleOfAvailableForSaleSecuritiesLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=us-gaap_USTreasurySecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=eras_UsGovernmentAgenciesShortTermSecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=us-gaap_CorporateDebtSecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_FinancialInstrumentAxis=us-gaap_CommercialPaperMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Marketable Securities - Additional Information (Details)
|
Mar. 31, 2024
USD ($)
Position
|
Dec. 31, 2023
USD ($)
Position
|
| Investments, Debt and Equity Securities [Abstract] |
|
|
| Available of sale securities, unrealized loss position, Number of positions | Position |
28
|
22
|
| Available for sale securities, gross unrealized loss position | $ |
$ 162,484,000
|
$ 105,554,000
|
| Available of sale securities, unrealized loss position, greater than 12 months, Number of positions | Position |
0
|
0
|
| Available-for-sale securities, accrued interest | $ |
$ 871,000
|
$ 1,100,000
|
| Debt Securities, Available-for-Sale, Accrued Interest, after Allowance for Credit Loss, Current, Statement of Financial Position [Extensible Enumeration] |
Prepaid expenses and other current assets
|
Prepaid expenses and other current assets
|
| X |
- DefinitionAmount, after allowance for credit loss, of accrued interest on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), classified as current. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2A
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-2A
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479130/326-30-45-1
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 3A
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-3A
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleAccruedInterestAfterAllowanceForCreditLossCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionIndicates line item in statement of financial position that includes accrued interest, after allowance for credit loss, on investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), classified as current. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 3A
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-3A
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleAccruedInterestAfterAllowanceForCreditLossCurrentStatementOfFinancialPositionExtensibleList |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
enum2:enumerationSetItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of investments in debt securities measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in continuous unrealized loss position for 12 months or longer, without an allowance for credit loss. Includes beneficial interest in securitized financial asset. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (b)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-4
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleContinuousUnrealizedLossPosition12MonthsOrLongerNumberOfPositions |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:integerItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of investment in debt security measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in unrealized loss position without allowance for credit loss. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 55
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479081/326-30-55-8
Reference 2: http://fasb.org/us-gaap/role/ref/otherTransitionRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-6
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-4
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleUnrealizedLossPosition |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of investments in debt securities measured at fair value with change in fair value recognized in other comprehensive income (available-for-sale), in unrealized loss position, without allowance for credit loss. Includes beneficial interest in securitized financial asset. Reference 1: http://fasb.org/us-gaap/role/ref/otherTransitionRef
-Topic 320
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (b)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481800/320-10-50-6
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 326
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (b)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479106/326-30-50-4
| Name: |
us-gaap_DebtSecuritiesAvailableForSaleUnrealizedLossPositionNumberOfPositions |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:integerItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_InvestmentsDebtAndEquitySecuritiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Property and Equipment, Net - Schedule of Property and Equipment, Net (Details) - USD ($)
$ in Thousands |
Mar. 31, 2024 |
Dec. 31, 2023 |
| Property Plant And Equipment [Line Items] |
|
|
| Property and equipment |
$ 29,588
|
$ 29,559
|
| Less accumulated depreciation and amortization |
(8,230)
|
(7,232)
|
| Property and equipment, net |
21,358
|
22,327
|
| Laboratory Equipment |
|
|
| Property Plant And Equipment [Line Items] |
|
|
| Property and equipment |
5,621
|
5,620
|
| Furniture and Fixtures |
|
|
| Property Plant And Equipment [Line Items] |
|
|
| Property and equipment |
4,099
|
4,099
|
| Leasehold Improvements |
|
|
| Property Plant And Equipment [Line Items] |
|
|
| Property and equipment |
18,173
|
18,173
|
| Computer Equipment and Software |
|
|
| Property Plant And Equipment [Line Items] |
|
|
| Property and equipment |
$ 1,695
|
$ 1,667
|
| X |
- DefinitionAmount of accumulated depreciation, depletion and amortization for physical assets used in the normal conduct of business to produce goods and services. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(14))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_AccumulatedDepreciationDepletionAndAmortizationPropertyPlantAndEquipment |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount before accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(13))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 360
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
us-gaap_PropertyPlantAndEquipmentLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount after accumulated depreciation, depletion and amortization of physical assets used in the normal conduct of business to produce goods and services and not intended for resale. Examples include, but are not limited to, land, buildings, machinery and equipment, office equipment, and furniture and fixtures. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-SubTopic 10
-Topic 360
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482099/360-10-50-1
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 210
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-03(a)(8))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479440/944-210-S99-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 360
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480842/942-360-50-1
| Name: |
us-gaap_PropertyPlantAndEquipmentNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
us-gaap_PropertyPlantAndEquipmentByTypeAxis=eras_LaboratoryEquipmentMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PropertyPlantAndEquipmentByTypeAxis=us-gaap_FurnitureAndFixturesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PropertyPlantAndEquipmentByTypeAxis=us-gaap_LeaseholdImprovementsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PropertyPlantAndEquipmentByTypeAxis=eras_ComputerEquipmentAndSoftwareMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionThe aggregate expense recognized in the current period that allocates the cost of tangible assets, intangible assets, or depleting assets to periods that benefit from use of the assets. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Subparagraph (b)
-SubTopic 10
-Topic 230
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Subparagraph (e)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
| Name: |
us-gaap_DepreciationDepletionAndAmortization |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_PropertyPlantAndEquipmentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
| X |
- DefinitionAccrued offering costs, current.
| Name: |
eras_AccruedOfferingCostsCurrent |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAccrued property and equipment current.
| Name: |
eras_AccruedPropertyAndEquipmentCurrent |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAccrued research and development expenses current.
| Name: |
eras_AccruedResearchAndDevelopmentExpensesCurrent |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionUnvested early exercised stock option liability.
| Name: |
eras_UnvestedEarlyExercisedStockOptionLiability |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionCarrying value as of the balance sheet date of obligations, excluding pension and other postretirement benefits, incurred through that date and payable for perquisites provided to employees pertaining to services received from them. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.20)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AccruedEmployeeBenefitsCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of expenses incurred but not yet paid nor invoiced, and liabilities classified as other.
| Name: |
us-gaap_AccruedLiabilitiesAndOtherLiabilities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionCarrying value as of the balance sheet date of obligations incurred through that date and payable for professional fees, such as for legal and accounting services received. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.20)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AccruedProfessionalFeesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of expenses incurred but not yet paid classified as other, due within one year or the normal operating cycle, if longer. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.20)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_OtherAccruedLiabilitiesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_PayablesAndAccrualsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Asset Acquisitions - Additional Information (Details) - USD ($)
|
1 Months Ended |
3 Months Ended |
|
Mar. 31, 2021 |
Nov. 30, 2020 |
Mar. 31, 2024 |
Mar. 31, 2023 |
Dec. 31, 2023 |
| Asana Merger Agreement |
|
|
|
|
|
| Asset Acquisition [Line Items] |
|
|
|
|
|
| Upfront payment made |
|
$ 20,000,000
|
|
|
|
| Future development and regulatory milestone cash payments maximum |
|
$ 90,000,000
|
|
|
|
| Additional shares required to issue upon achieving development milestone |
|
3,888,889
|
|
|
|
| In-process research and development |
|
|
$ 0
|
$ 0
|
|
| Milestones accrued |
|
|
0
|
|
$ 0
|
| Asana Merger Agreement | Series B-2 Convertible Preferred Stock |
|
|
|
|
|
| Asset Acquisition [Line Items] |
|
|
|
|
|
| Shares issued |
|
4,000,000
|
|
|
|
| Value per share |
|
$ 7.5
|
|
|
|
| Total fair value of equity |
|
$ 30,000,000
|
|
|
|
| Asana Merger Agreement | Series B-2 Convertible Preferred Stock | Common Stock | IPO |
|
|
|
|
|
| Asset Acquisition [Line Items] |
|
|
|
|
|
| Convertible preferred stock converted into shares of common stock |
|
3,333,333
|
|
|
|
| ELS Purchase Agreement |
|
|
|
|
|
| Asset Acquisition [Line Items] |
|
|
|
|
|
| Upfront payment made |
$ 2,000,000
|
|
|
|
|
| In-process research and development |
|
|
$ 0
|
$ 0
|
|
| ELS Purchase Agreement | Common Stock |
|
|
|
|
|
| Asset Acquisition [Line Items] |
|
|
|
|
|
| Shares issued |
500,000
|
|
|
|
|
| Value per share |
$ 3.36
|
|
|
|
|
| Total fair value of equity |
$ 1,700,000
|
|
|
|
|
| X |
- DefinitionAdditional shares required to issue upon achieving development milestone.
| Name: |
eras_AdditionalSharesRequiredToIssueUponAchievingDevelopmentMilestone |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFuture development and regulatory milestone cash payments maximum.
| Name: |
eras_FutureDevelopmentAndRegulatoryMilestoneCashPaymentsMaximum |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionIn process research and development expense.
| Name: |
eras_InProcessResearchAndDevelopmentExpense |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 805
-SubTopic 50
-Name Accounting Standards Codification
-Section 15
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480123/805-50-15-3
| Name: |
us-gaap_AssetAcquisitionLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionValue of equity interests (such as common shares, preferred shares, or partnership interest) issued or issuable to acquire the entity. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 805
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479581/805-30-50-1
| Name: |
us-gaap_BusinessAcquisitionEquityInterestIssuedOrIssuableValueAssigned |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares of equity interests issued or issuable to acquire entity. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 805
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479581/805-30-50-1
| Name: |
us-gaap_BusinessAcquisitionEquityInterestsIssuedOrIssuableNumberOfSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPrice of a single share of a number of saleable stocks paid or offered to be paid in a business combination.
| Name: |
us-gaap_BusinessAcquisitionSharePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares issued during the period as a result of the conversion of convertible securities. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 1E
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481139/470-20-50-1E
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29-30)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesConversionOfConvertibleSecurities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_BusinessAcquisitionAxis=eras_AsanaMergerAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=eras_SeriesB2ConvertiblePreferredStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=us-gaap_IPOMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_BusinessAcquisitionAxis=eras_EmergeLifeSciencesPurchaseAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
License Agreements - Additional Information (Details) - USD ($)
|
1 Months Ended |
3 Months Ended |
|
Dec. 31, 2022 |
Mar. 31, 2022 |
Jan. 31, 2021 |
Apr. 30, 2020 |
Mar. 31, 2020 |
Feb. 29, 2020 |
Mar. 31, 2024 |
Mar. 31, 2023 |
Dec. 31, 2023 |
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Accounts payable |
|
|
|
|
|
|
$ 2,633,000
|
|
$ 2,000,000
|
| License Agreement | Novartis Pharma AG |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Upfront payment |
$ 20,000,000
|
|
|
|
|
|
|
|
|
| In-process research and development |
|
|
|
|
|
|
0
|
$ 0
|
|
| Accrued milestone payment as underlying contingencies |
|
|
|
|
|
|
0
|
|
0
|
| Stock issued, value |
$ 80,000,000
|
|
|
|
|
|
|
|
|
| Stock issued and sold |
12,307,692
|
|
|
|
|
|
|
|
|
| License Agreement | Novartis Pharma AG | Maximum |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Development and regulatory milestone payments obligation |
$ 80,000,000
|
|
|
|
|
|
|
|
|
| Sales milestone payments obligation |
$ 200,000,000
|
|
|
|
|
|
|
|
|
| License Agreement | NiKang Therapeutics, Inc. |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Upfront payment |
|
|
|
|
|
$ 5,000,000
|
|
|
|
| Reimbursement of certain initial manufacturing costs |
|
|
|
|
|
400,000
|
|
|
|
| Payment for US patent application |
|
|
|
$ 7,000,000
|
|
|
|
|
|
| In-process research and development |
|
|
|
|
|
|
0
|
0
|
|
| Accrued milestone payment as underlying contingencies |
|
|
|
|
|
|
0
|
|
0
|
| License Agreement | NiKang Therapeutics, Inc. | First Licensed Product |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Payment of development and regulatory milestone obligation |
|
|
$ 4,000,000
|
|
|
|
|
|
|
| License Agreement | NiKang Therapeutics, Inc. | First Licensed Product | Maximum |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Development and regulatory milestone payments obligation |
|
|
|
|
|
16,000,000
|
|
|
|
| Commercial milestone payments obligation |
|
|
|
|
|
157,000,000
|
|
|
|
| License Agreement | NiKang Therapeutics, Inc. | Second Licensed Product |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Development and regulatory milestone payments obligation |
|
|
|
|
|
12,000,000
|
|
|
|
| Commercial milestone payments obligation |
|
|
|
|
|
$ 151,000,000
|
|
|
|
| License Agreement | Katmai Pharmaceuticals, Inc. |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Upfront payment |
|
|
|
|
$ 5,700,000
|
|
|
|
|
| Payment of development and regulatory milestone obligation |
|
$ 2,000,000
|
|
|
|
|
|
|
|
| In-process research and development |
|
|
|
|
|
|
0
|
0
|
|
| Accrued milestone payment as underlying contingencies |
|
|
|
|
|
|
0
|
|
0
|
| License Agreement | Katmai Pharmaceuticals, Inc. | Series B-1 Convertible Preferred Stock and Series B-2 Convertible Preferred Stock |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Stock issued, value |
|
|
|
|
$ 2,700,000
|
|
|
|
|
| License Agreement | Katmai Pharmaceuticals, Inc. | Series B-1 Convertible Preferred Stock and Series B-2 Convertible Preferred Stock | IPO |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Conversion of convertible preferred stock into common stock |
|
|
|
|
395,555
|
|
|
|
|
| License Agreement | Katmai Pharmaceuticals, Inc. | Series B-1 Convertible Preferred Stock |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Stock issued, value |
|
|
|
$ 1,800,000
|
|
|
|
|
|
| Stock issued and sold |
|
|
|
356,000
|
|
|
|
|
|
| License Agreement | Katmai Pharmaceuticals, Inc. | Series B-2 Convertible Preferred Stock |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Stock issued, value |
|
|
$ 900,000
|
|
|
|
|
|
|
| Stock issued and sold |
|
|
118,666
|
|
|
|
|
|
|
| License Agreement | Katmai Pharmaceuticals, Inc. | Maximum |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Development and regulatory milestone payments obligation |
|
|
|
|
$ 26,000,000
|
|
|
|
|
| Commercial milestone payments obligation |
|
|
|
|
$ 101,000,000
|
|
|
|
|
| License Agreement | LifeArc |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Upfront payment |
|
|
|
$ 0
|
|
|
|
|
|
| In-process research and development |
|
|
|
|
|
|
0
|
$ 0
|
|
| Accrued milestone payment as underlying contingencies |
|
|
|
|
|
|
$ 0
|
|
$ 0
|
| One-time license payment |
|
|
|
75,000
|
|
|
|
|
|
| License Agreement | LifeArc | Maximum |
|
|
|
|
|
|
|
|
|
| License Agreements [Line Items] |
|
|
|
|
|
|
|
|
|
| Development and regulatory milestone payments obligation |
|
|
|
11,000,000
|
|
|
|
|
|
| Sales milestone payments obligation |
|
|
|
$ 50,000,000
|
|
|
|
|
|
| X |
- DefinitionAccrued milestone payment as underlying contingencies.
| Name: |
eras_AccruedMilestonePaymentAsUnderlyingContingencies |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionCommercial milestone payments obligation.
| Name: |
eras_CommercialMilestonePaymentsObligation |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionDevelopment and regulatory milestone payments obligation.
| Name: |
eras_DevelopmentAndRegulatoryMilestonePaymentsObligation |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionIn process research and development expense.
| Name: |
eras_InProcessResearchAndDevelopmentExpense |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
eras_LicenseAgreementsLineItems |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionOne-time license payment.
| Name: |
eras_OneTimeLicensePayment |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionPayment of Development and Regulatory Milestone Obligation
| Name: |
eras_PaymentOfDevelopmentAndRegulatoryMilestoneObligation |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionReimbursement of certain initial manufacturing costs.
| Name: |
eras_ReimbursementOfCertainInitialManufacturingCosts |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionSales milestone payments obligation.
| Name: |
eras_SalesMilestonePaymentsObligation |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionCarrying value as of the balance sheet date of liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received that are used in an entity's business. Used to reflect the current portion of the liabilities (due within one year or within the normal operating cycle if longer). Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 852
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481372/852-10-55-10
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19(a))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_AccountsPayableCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares issued for each share of convertible preferred stock that is converted. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(27))
-SubTopic 10
-Topic 210
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-3
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 16
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-16
| Name: |
us-gaap_ConvertiblePreferredStockSharesIssuedUponConversion |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionEquity impact of the value of new stock issued during the period. Includes shares issued in an initial public offering or a secondary public offering. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 11
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-11
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 205
-Name Accounting Standards Codification
-Section 45
-Paragraph 4
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480767/946-205-45-4
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 8: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodValueNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_TypeOfArrangementAxis=eras_LicenseAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_CounterpartyNameAxis=eras_NovartisPharmaAgMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MaximumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_CounterpartyNameAxis=eras_NiKangTherapeuticsIncorporationMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_ProductOrServiceAxis=eras_FirstLicensedProductMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_ProductOrServiceAxis=eras_SecondLicensedProductMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_CounterpartyNameAxis=eras_KatmaiPharmaceuticalsIncorporationMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=eras_SeriesB1ConvertiblePreferredStockAndSeriesB2ConvertiblePreferredStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=us-gaap_IPOMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=eras_SeriesB1ConvertiblePreferredStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=eras_SeriesB2ConvertiblePreferredStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_CounterpartyNameAxis=eras_LifeArcMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Stock-based Compensation - Additional Information (Details) - USD ($)
|
1 Months Ended |
3 Months Ended |
5 Months Ended |
|
Jul. 31, 2021 |
Mar. 31, 2024 |
Mar. 31, 2023 |
Dec. 31, 2021 |
Dec. 31, 2023 |
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Common stock reserved for future issuance |
|
49,932,505
|
|
|
42,394,435
|
| Share-based compensation, shares subject to repurchase |
|
247,919
|
|
|
371,876
|
| Stock-based compensation |
|
$ 6,848,000
|
$ 6,845,000
|
|
|
| Accrued Expenses and Other Current Liabilities |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Share-based compensation, liabilities associated with shares issued with repurchase rights |
|
$ 309,000
|
|
|
$ 464,000
|
| Minimum |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Share-based compensation, common stock grant in period, term |
|
5 years
|
|
|
|
| Employee Stock Option |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Common stock reserved for future issuance |
|
34,647,123
|
|
|
24,970,957
|
| Stock vesting period |
|
4 years
|
|
|
|
| Weighted-average grant date fair value of options granted |
|
$ 1.25
|
$ 2.94
|
|
|
| Unrecognized compensation cost related to unvested stock option grants |
|
$ 55,900,000
|
|
|
|
| Unrecognized compensation cost expected to be recognized as expense, period |
|
2 years 9 months
|
|
|
|
| Intrinsic value of options exercised |
|
$ 47,000
|
$ 517,000
|
|
|
| Share-based compensation, options granted that vest based on performance milestone |
|
9,827,650
|
|
|
|
| Employee Stock Option | Minimum | At the Time Option is Granted |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Percentage of fair market value of common stock |
|
100.00%
|
|
|
|
| Employee Stock Option | Minimum | For Holders of More Than 10% of Company's Total Combined Voting Power of All Classes of Stock |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Percentage of fair market value of common stock |
|
110.00%
|
|
|
|
| Employee Stock Option | Maximum |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Share-based compensation, plan expiration period |
|
10 years
|
|
|
|
| 2018 Equity Incentive Plan |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Share-based compensation, options granted that vest based on performance milestone |
|
|
|
0
|
|
| 2021 Incentive Award Plan |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Common stock reserved for future issuance |
|
13,204,701
|
|
|
|
| Shares of common stock, authorized for issuance |
15,150,000
|
|
|
|
|
| Percentage of outstanding common stock maximum |
5.00%
|
|
|
|
|
| ESPP |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Common stock reserved for future issuance |
1,260,000
|
|
|
|
|
| Unrecognized compensation cost related to unvested stock option grants |
|
$ 1,500,000
|
|
|
|
| Unrecognized compensation cost expected to be recognized as expense, period |
|
1 year 8 months 8 days
|
|
|
|
| Stock-based compensation |
|
$ 342,000
|
$ 707,000
|
|
|
| Share-based payment arrangement, withheld on behalf of employees for future purchase |
|
$ 317,000
|
|
|
$ 46,000
|
| Offering period |
24 months
|
|
|
|
|
| Purchase period |
6 months
|
|
|
|
|
| Shares issued under ESPP |
|
0
|
0
|
|
|
| ESPP | Minimum |
|
|
|
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
|
|
|
| Percentage of fair market value of common stock |
85.00%
|
|
|
|
|
| X |
- DefinitionShare-based compensation arrangement by share-based payment award offering period.
| Name: |
eras_ShareBasedCompensationArrangementByShareBasedPaymentAwardOfferingPeriod |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionShare-based compensation arrangement by share-based payment award purchase period.
| Name: |
eras_ShareBasedCompensationArrangementByShareBasedPaymentAwardPurchasePeriod |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionShare-based compensation arrangement by share-based payment award, shares subject to repurchase.
| Name: |
eras_ShareBasedCompensationArrangementByShareBasedPaymentAwardSharesSubjectToRepurchase |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionShare based compensation by share based payment award common stock granted in period, term.
| Name: |
eras_ShareBasedCompensationByShareBasedPaymentAwardOptionsGrantedInPeriodTerm |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionShare-based payment arrangement, withheld on behalf of employees for future purchase.
| Name: |
eras_ShareBasedPaymentArrangementWithheldOnBehalfOfEmployeesForFuturePurchase |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of expense for award under share-based payment arrangement. Excludes amount capitalized. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB Topic 14.F)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479830/718-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (h)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_AllocatedShareBasedCompensationExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAggregate number of common shares reserved for future issuance. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockCapitalSharesReservedForFutureIssuance |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate carrying value as of the balance sheet date of the liabilities for stock option plans and other equity-based compensation arrangements payable within one year (or the operating cycle, if longer). Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.20)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_DeferredCompensationShareBasedArrangementsLiabilityCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted-average period over which cost not yet recognized is expected to be recognized for award under share-based payment arrangement, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedPeriodForRecognition1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cost to be recognized for option under share-based payment arrangement. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_EmployeeServiceShareBasedCompensationNonvestedAwardsTotalCompensationCostNotYetRecognizedStockOptions |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionPeriod over which grantee's right to exercise award under share-based payment arrangement is no longer contingent on satisfaction of service or performance condition, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Includes, but is not limited to, combination of market, performance or service condition. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardAwardVestingPeriod1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 1D
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-1D
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares authorized for issuance under share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardNumberOfSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of accumulated difference between fair value of underlying shares on dates of exercise and exercise price on options exercised (or share units converted) into shares. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisesInPeriodTotalIntrinsicValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionGross number of share options (or share units) granted during the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average grant-date fair value of options granted during the reporting period as calculated by applying the disclosed option pricing methodology. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageGrantDateFairValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionMaximum number of shares that may be issued in accordance with the plan as a proportion of outstanding capital stock.
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardPercentageOfOutstandingStockMaximum |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPeriod from grant date that an equity-based award expires, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardExpirationPeriod |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPurchase price of common stock expressed as a percentage of its fair value.
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardPurchasePriceOfCommonStockPercent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares issued during the period as a result of an employee stock purchase plan. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesEmployeeStockPurchasePlans |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_BalanceSheetLocationAxis=us-gaap_AccountsPayableAndAccruedLiabilitiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MinimumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=us-gaap_EmployeeStockOptionMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_StatementScenarioAxis=eras_AtTimeOptionIsGrantedMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_StatementScenarioAxis=eras_ForHoldersOfMoreThanTenPercentOfCombinedVotingPowerOfAllClassesOfStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MaximumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PlanNameAxis=eras_TwoThousandEighteenEquityIncentivePlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PlanNameAxis=eras_TwoThousandTwentyOneIncentiveAwardPlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_PlanNameAxis=eras_EmployeeStockPurchasePlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Stock-based Compensation - Summary of Stock Option Activity (Details) - Employee Stock Option - USD ($)
$ / shares in Units, $ in Thousands |
3 Months Ended |
12 Months Ended |
Mar. 31, 2024 |
Dec. 31, 2023 |
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
| Shares Outstanding at December 31, 2023 |
24,970,957
|
|
| Shares Granted |
9,827,650
|
|
| Shares Exercised |
(35,035)
|
|
| Shares Canceled |
(116,449)
|
|
| Shares Outstanding at March 31, 2024 |
34,647,123
|
24,970,957
|
| Shares Options exercisable at March 31, 2024 |
12,850,952
|
|
| Weighted-average exercise price, Outstanding at December 31, 2023 |
$ 4.98
|
|
| Weighted-average exercise price, Granted |
1.71
|
|
| Weighted-average exercise price, Exercised |
0.69
|
|
| Weighted-average exercise price, Canceled |
4.89
|
|
| Weighted-average exercise price, Outstanding at March 31, 2024 |
4.06
|
$ 4.98
|
| Weighted-average exercise price, Options exercisable at March 31, 2024 |
$ 4.83
|
|
| Weighted-average remaining contractual term (years), Outstanding |
8 years 5 months 8 days
|
8 years 1 month 13 days
|
| Weighted-average remaining contractual term (years), Options exercisable at March 31, 2024 |
7 years 3 months 25 days
|
|
| Aggregate intrinsic value, Outstanding |
$ 7,532
|
$ 4,412
|
| Aggregate intrinsic value, Options exercisable at March 31, 2024 |
$ 3,916
|
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 1D
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-1D
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of shares into which fully or partially vested stock options outstanding as of the balance sheet date can be currently converted under the option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableNumber |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe weighted-average price as of the balance sheet date at which grantees can acquire the shares reserved for issuance on vested portions of options outstanding and currently exercisable under the stock option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsExercisableWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe number of shares under options that were cancelled during the reporting period as a result of occurrence of a terminating event specified in contractual agreements pertaining to the stock option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsForfeituresInPeriod |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionGross number of share options (or share units) granted during the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsGrantsInPeriodGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount by which the current fair value of the underlying stock exceeds the exercise price of options outstanding. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingIntrinsicValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of options outstanding, including both vested and non-vested options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingNumber |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average price at which grantees can acquire the shares reserved for issuance under the stock option plan. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardOptionsOutstandingWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average price at which option holders acquired shares when converting their stock options into shares. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsExercisesInPeriodWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionWeighted average price at which grantees could have acquired the underlying shares with respect to stock options that were terminated. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsForfeituresInPeriodWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionWeighted average per share amount at which grantees can acquire shares of common stock by exercise of options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementsByShareBasedPaymentAwardOptionsGrantsInPeriodWeightedAverageExercisePrice |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of difference between fair value of the underlying shares reserved for issuance and exercise price of vested portions of options outstanding and currently exercisable. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsExercisableIntrinsicValue1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average remaining contractual term for vested portions of options outstanding and currently exercisable or convertible, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsExercisableWeightedAverageRemainingContractualTerm1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionWeighted average remaining contractual term for option awards outstanding, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents the reported fact of one year, five months, and thirteen days. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 718
-SubTopic 10
-Subparagraph (e)(1)
-Name Accounting Standards Codification
-Paragraph 2
-Section 50
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardOptionsOutstandingWeightedAverageRemainingContractualTerm2 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of share options (or share units) exercised during the current period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 3: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 4: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesStockOptionsExercised |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=us-gaap_EmployeeStockOptionMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionThe estimated dividend rate (a percentage of the share price) to be paid (expected dividends) to holders of the underlying shares over the option's term. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedDividendRate |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe estimated measure of the maximum percentage by which a share price is expected to fluctuate during a period. Volatility also may be defined as a probability-weighted measure of the dispersion of returns about the mean. The volatility of a share price is the standard deviation of the continuously compounded rates of return on the share over a specified period. That is the same as the standard deviation of the differences in the natural logarithms of the stock prices plus dividends, if any, over the period.
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedVolatilityRateMaximum |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe estimated measure of the minimum percentage by which a share price is expected to fluctuate during a period. Volatility also may be defined as a probability-weighted measure of the dispersion of returns about the mean. The volatility of a share price is the standard deviation of the continuously compounded rates of return on the share over a specified period. That is the same as the standard deviation of the differences in the natural logarithms of the stock prices plus dividends, if any, over the period.
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsExpectedVolatilityRateMinimum |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe maximum risk-free interest rate assumption that is used in valuing an option on its own shares.
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateMaximum |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe minimum risk-free interest rate assumption that is used in valuing an option on its own shares.
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardFairValueAssumptionsRiskFreeInterestRateMinimum |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 1D
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-1D
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionExpected term of award under share-based payment arrangement, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_SharebasedCompensationArrangementBySharebasedPaymentAwardFairValueAssumptionsExpectedTerm1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=us-gaap_EmployeeStockOptionMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MinimumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RangeAxis=srt_MaximumMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionAmount of expense for award under share-based payment arrangement. Excludes amount capitalized. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SAB Topic 14.F)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479830/718-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (h)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_AllocatedShareBasedCompensationExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 1D
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-1D
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_IncomeStatementLocationAxis=us-gaap_ResearchAndDevelopmentExpenseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_IncomeStatementLocationAxis=us-gaap_GeneralAndAdministrativeExpenseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Stock-based Compensation - Summary of Common Stock Reserved for Future Issuance (Details) - shares
|
Mar. 31, 2024 |
Dec. 31, 2023 |
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
| Common stock reserved for future issuance |
49,932,505
|
42,394,435
|
| Stock Options Issued and Outstanding |
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
| Common stock reserved for future issuance |
34,647,123
|
24,970,957
|
| Awards Available for Future Grant |
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
| Common stock reserved for future issuance |
13,204,701
|
15,342,797
|
| Shares Available for Purchase Under ESPP |
|
|
| Share Based Compensation Arrangement By Share Based Payment Award [Line Items] |
|
|
| Common stock reserved for future issuance |
2,080,681
|
2,080,681
|
| X |
- DefinitionAggregate number of common shares reserved for future issuance. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_CommonStockCapitalSharesReservedForFutureIssuance |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 1D
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-1D
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 35
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480483/718-10-35-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(1)(iv)(04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(01)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(02)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (c)(2)(iii)(03)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 20: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (d)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (e)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(i)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(ii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iii)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(iv)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 718
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (f)(2)(v)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480429/718-10-50-2
| Name: |
us-gaap_ShareBasedCompensationArrangementByShareBasedPaymentAwardLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=us-gaap_EmployeeStockOptionMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=eras_AwardsAvailableForFutureGrantMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AwardTypeAxis=eras_SharesAvailableForPurchaseUnderEmployeeStockPurchasePlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Leases - Additional Information (Details)
|
|
1 Months Ended |
3 Months Ended |
|
|
Jan. 31, 2024
ft²
|
Apr. 30, 2022 |
Dec. 31, 2021
USD ($)
ft²
|
Mar. 31, 2021
ft²
|
Sep. 30, 2020
USD ($)
ft²
|
Mar. 31, 2024
USD ($)
|
Mar. 31, 2023
USD ($)
|
Dec. 31, 2023
USD ($)
|
| Lessee Lease Description [Line Items] |
|
|
|
|
|
|
|
|
| Operating leases cancelable and non-cancellable expiration dates |
|
|
|
|
|
2032
|
|
|
| Operating leases Noncancelable expiration dates |
|
|
|
|
|
2026
|
|
|
| Total lease cost |
|
|
|
|
|
$ 2,700,000
|
$ 3,100,000
|
|
| Operating lease cost |
|
|
|
|
|
1,900,000
|
1,900,000
|
|
| Variable lease costs |
|
|
|
|
|
916,000
|
1,200,000
|
|
| Cash paid for operating leases |
|
|
|
|
|
2,100,000
|
863,000
|
|
| Sublease income |
|
|
|
|
|
$ 84,000
|
$ 0
|
|
| Operating lease, weighted-average remaining lease term |
|
|
|
|
|
8 years 18 days
|
|
8 years 3 months 14 days
|
| Operating lease, weighted-average discount rate |
|
|
|
|
|
8.96%
|
|
8.95%
|
| Operating lease, aggregate monthly payments to lessor |
|
|
|
|
|
$ 78,370,000
|
|
|
| Letter of credit provided to lessor |
|
|
|
|
|
$ 408,000
|
|
$ 408,000
|
| 2020 Lease |
|
|
|
|
|
|
|
|
| Lessee Lease Description [Line Items] |
|
|
|
|
|
|
|
|
| Office space leases | ft² |
|
|
|
|
59,407
|
|
|
|
| Lease commencement month and year |
|
|
|
|
2021-08
|
|
|
|
| Operating lease initial term |
|
|
|
|
10 years 9 months
|
|
|
|
| Operating lease, aggregate monthly payments to lessor |
|
|
|
|
$ 51,600,000
|
|
|
|
| Tenant improvement allowance |
|
|
|
|
$ 16,800,000
|
|
|
|
| Term for cancellation of lease |
|
|
|
|
84 months
|
|
|
|
| Notice period for cancellation of lease |
|
|
|
|
12 months
|
|
|
|
| Operating lease cancellation payment |
|
|
|
|
$ 2,500,000
|
|
|
|
| 2020 Lease | Letter of Credit |
|
|
|
|
|
|
|
|
| Lessee Lease Description [Line Items] |
|
|
|
|
|
|
|
|
| Letter of credit provided to lessor |
|
|
|
|
$ 408,000
|
|
|
|
| Line of credit facility, expiration date |
|
|
|
|
Jul. 29, 2032
|
|
|
|
| First Amendment to 2020 Lease |
|
|
|
|
|
|
|
|
| Lessee Lease Description [Line Items] |
|
|
|
|
|
|
|
|
| Office space leases | ft² |
|
|
|
18,421
|
|
|
|
|
| Lease commencement month and year |
|
2022-05
|
|
2022-02
|
|
|
|
|
| 2021 Lease |
|
|
|
|
|
|
|
|
| Lessee Lease Description [Line Items] |
|
|
|
|
|
|
|
|
| Office space leases | ft² |
|
|
29,542
|
|
|
|
|
|
| Lease commencement month and year |
|
|
2022-07
|
|
|
|
|
|
| Operating lease initial term |
|
|
124 months
|
|
|
|
|
|
| Operating lease option to extend |
|
|
|
|
|
5 years
|
|
|
| Operating lease existence of option to extend [true false] |
|
|
|
|
|
true
|
|
|
| Operating lease, aggregate monthly payments to lessor |
|
|
$ 34,400,000
|
|
|
|
|
|
| Tenant improvement allowance |
|
|
8,200,000
|
|
|
|
|
|
| Prepaid rent |
|
|
|
|
|
$ 5,100,000
|
|
|
| Operating Lease, Right-of-Use Asset, Statement of Financial Position [Extensible Enumeration] |
|
|
|
|
|
Operating Lease, Right-of-Use Asset
|
|
|
| 2021 Lease | Other Assets |
|
|
|
|
|
|
|
|
| Lessee Lease Description [Line Items] |
|
|
|
|
|
|
|
|
| Security deposit paid |
|
|
$ 874,000
|
|
|
|
|
|
| 2024 Lease |
|
|
|
|
|
|
|
|
| Lessee Lease Description [Line Items] |
|
|
|
|
|
|
|
|
| Sublease income |
|
|
|
|
|
$ 84,000
|
|
|
| Office space leases | ft² |
10,000
|
|
|
|
|
|
|
|
| Operating lease initial term |
3 years
|
|
|
|
|
|
|
|
| Operating lease existence of option to extend [true false] |
true
|
|
|
|
|
|
|
|
| Operating sublease existence of option to terminate [true false] |
true
|
|
|
|
|
|
|
|
| X |
- DefinitionBuilding subject to ground leases.
| Name: |
eras_BuildingSubjectToGroundLeases |
| Namespace Prefix: |
eras_ |
| Data Type: |
dtr-types:areaItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionLease commencement month and year.
| Name: |
eras_LeaseCommencementMonthAndYear |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:gYearMonthItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNotice period for cancellation of lease.
| Name: |
eras_NoticePeriodForCancellationOfLease |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionOperating lease cancellation payment.
| Name: |
eras_OperatingLeaseCancellationPayment |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionOperating leases cancelable and non-cancellable expiration dates.
| Name: |
eras_OperatingLeasesCancelableAndNonCancellableExpirationDates |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:gYearItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionOperating leases Noncancelable expiration dates.
| Name: |
eras_OperatingLeasesNonCancelableExpirationDates |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:gYearItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTenant improvement allowance.
| Name: |
eras_TenantImprovementAllowance |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionTerm for cancellation of lease.
| Name: |
eras_TermForCancellationOfLease |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of lease cost recognized by lessee for lease contract. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_LeaseCost |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-3
| Name: |
us-gaap_LesseeLeaseDescriptionLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicates (true false) whether lessee has option to extend operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-3
| Name: |
us-gaap_LesseeOperatingLeaseExistenceOfOptionToExtend |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionDescription of terms and conditions of option to extend lessee's operating lease. Includes, but is not limited to, information about option recognized as part of right-of-use asset and lease liability. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-3
| Name: |
us-gaap_LesseeOperatingLeaseOptionToExtend |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTerm of lessee's operating lease, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-3
| Name: |
us-gaap_LesseeOperatingLeaseTermOfContract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionIndicates (true false) whether sublessee has option to terminate operating sublease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-3
| Name: |
us-gaap_LesseeOperatingSubleaseExistenceOfOptionToTerminate |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDate the credit facility terminates, in YYYY-MM-DD format. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19(b),22(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_LineOfCreditFacilityExpirationDate1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of single lease cost, calculated by allocation of remaining cost of lease over remaining lease term. Includes, but is not limited to, single lease cost, after impairment of right-of-use asset, calculated by amortization of remaining right-of-use asset and accretion of lease liability. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_OperatingLeaseCost |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash outflow from operating lease, excluding payments to bring another asset to condition and location necessary for its intended use. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 5
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-5
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (g)(1)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_OperatingLeasePayments |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionIndicates line item in statement of financial position that includes operating lease right-of-use asset. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-2
| Name: |
us-gaap_OperatingLeaseRightOfUseAssetStatementOfFinancialPositionExtensibleList |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
enum2:enumerationSetItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average discount rate for operating lease calculated at point in time. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (g)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_OperatingLeaseWeightedAverageDiscountRatePercent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionWeighted average remaining lease term for operating lease, in 'PnYnMnDTnHnMnS' format, for example, 'P1Y5M13D' represents reported fact of one year, five months, and thirteen days. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (g)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_OperatingLeaseWeightedAverageRemainingLeaseTerm1 |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of asset related to consideration paid in advance for rent that provides economic benefits within a future period of one year or the normal operating cycle, if longer. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (g)(3)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483467/210-10-45-1
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 340
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483032/340-10-45-1
Reference 3: http://www.xbrl.org/2003/role/exampleRef
-Topic 340
-SubTopic 10
-Name Accounting Standards Codification
-Section 05
-Paragraph 5
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482955/340-10-05-5
| Name: |
us-gaap_PrepaidRent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of cash restricted as to withdrawal or usage, classified as noncurrent. Cash includes, but is not limited to, currency on hand, demand deposits with banks or financial institutions, and other accounts with general characteristics of demand deposits. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name Accounting Standards Codification
-Section 45
-Paragraph 5
-SubTopic 210
-Topic 954
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480632/954-210-45-5
Reference 3: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482913/230-10-50-8
| Name: |
us-gaap_RestrictedCashNoncurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of an asset, typically cash, provided to a counterparty to provide certain assurance of performance by the entity pursuant to the terms of a written or oral agreement, such as a lease. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(17))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_SecurityDeposit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of sublease income excluding finance and operating lease expense. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (e)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_SubleaseIncome |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of variable lease cost, excluded from lease liability, recognized when obligation for payment is incurred for finance and operating leases. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 55
-Paragraph 53
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479589/842-20-55-53
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-4
| Name: |
us-gaap_VariableLeaseCost |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_LeaseContractualTermAxis=eras_TwoThousandTwentyLeaseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_CreditFacilityAxis=us-gaap_LetterOfCreditMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_LeaseContractualTermAxis=eras_FirstAmendmentToTwoThousandAndTwentyLeaseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_LeaseContractualTermAxis=eras_TwoThousandTwentyOneLeaseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_BalanceSheetLocationAxis=us-gaap_OtherNoncurrentAssetsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_LeaseContractualTermAxis=eras_TwoThousandTwentyFourLeaseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Leases - Schedule of Future Minimum Lease Payments under Operating Leases (Details)
$ in Thousands |
Mar. 31, 2024
USD ($)
|
| Leases [Abstract] |
|
| 2024 (remaining nine months) |
$ 6,602
|
| 2025 |
9,024
|
| 2026 |
9,170
|
| 2027 |
9,199
|
| 2028 |
9,277
|
| Thereafter |
35,098
|
| Total lease payments |
78,370
|
| Less: Amount representing interest |
(23,433)
|
| Operating lease liabilities |
$ 54,937
|
| X |
- DefinitionLessee operating lease liability payments due after Year four.
| Name: |
eras_LesseeOperatingLeaseLiabilityPaymentsDueAfterYearFour |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_LeasesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease to be paid in next fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueNextTwelveMonths |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease to be paid in fourth fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearFour |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease to be paid in third fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearThree |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease to be paid in second fiscal year following current fiscal year. Excludes interim and annual periods when interim periods are reported from current statement of financial position date (rolling approach). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsDueYearTwo |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payment for operating lease having initial or remaining lease term in excess of one year to be paid in remainder of current fiscal year. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityPaymentsRemainderOfFiscalYear |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of lessee's undiscounted obligation for lease payments in excess of discounted obligation for lease payments for operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147478964/842-20-50-6
| Name: |
us-gaap_LesseeOperatingLeaseLiabilityUndiscountedExcessAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionPresent value of lessee's discounted obligation for lease payments from operating lease. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 842
-SubTopic 20
-Name Accounting Standards Codification
-Section 45
-Paragraph 1
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479041/842-20-45-1
| Name: |
us-gaap_OperatingLeaseLiability |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.24.1.u1
v3.24.1.u1
Net Loss Per Share - Summary of Computation of Basic and Diluted Net Loss Per Share (Details) - USD ($)
$ / shares in Units, $ in Thousands |
3 Months Ended |
Mar. 31, 2024 |
Mar. 31, 2023 |
| Earnings Per Share [Abstract] |
|
|
| Net Income (Loss) |
$ (35,017)
|
$ (33,199)
|
| Weighted-average shares of common stock used in computing net loss per share, basic |
151,161,741
|
149,504,216
|
| Weighted-average shares of common stock used in computing net loss per share, diluted |
151,161,741
|
149,504,216
|
| Net loss per share, basic |
$ (0.23)
|
$ (0.22)
|
| Net loss per share, diluted |
$ (0.23)
|
$ (0.22)
|
| X |
- References
+ Details
| Name: |
us-gaap_EarningsPerShareAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of net income (loss) for the period per each share of common stock or unit outstanding during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 15
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-15
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (e)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-7
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-10
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(27))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/exampleRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 52
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-52
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-7
| Name: |
us-gaap_EarningsPerShareBasic |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of net income (loss) for the period available to each share of common stock or common unit outstanding during the reporting period and to each share or unit that would have been outstanding assuming the issuance of common shares or units for all dilutive potential common shares or units outstanding during the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 15
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-15
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (e)(4)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-7
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-2
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (d)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(25))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 13: http://www.xbrl.org/2003/role/disclosureRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(27))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(23))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 15: http://www.xbrl.org/2003/role/exampleRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 55
-Paragraph 52
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482635/260-10-55-52
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-7
| Name: |
us-gaap_EarningsPerShareDiluted |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 235
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08(g)(1)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480678/235-10-S99-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 323
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481687/323-10-50-3
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 825
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 28
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482907/825-10-50-28
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 6
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482765/220-10-50-6
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-3
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (b)(2)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-1
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Topic 815
-SubTopic 40
-Name Accounting Standards Codification
-Section 65
-Paragraph 1
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480175/815-40-65-1
Reference 8: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 8
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-8
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 9
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-9
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 11: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 11
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-11
Reference 12: http://www.xbrl.org/2003/role/disclosureRef
-Topic 250
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 4
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483443/250-10-50-4
Reference 13: http://www.xbrl.org/2003/role/exampleRef
-Topic 946
-SubTopic 830
-Name Accounting Standards Codification
-Section 55
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480167/946-830-55-10
Reference 14: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section 45
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483581/946-220-45-7
Reference 15: http://www.xbrl.org/2003/role/disclosureRef
-Topic 944
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04(18))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483586/944-220-S99-1
Reference 16: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 22
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-22
Reference 17: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.6-07(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-1
Reference 18: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(1)(d))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 19: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 20: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(ii))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 21: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 22: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 23: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1A
-Subparagraph (SX 210.13-01(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1A
Reference 24: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(i))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 25: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(A))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 26: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iii)(B))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 27: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(4)(iv))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 28: http://www.xbrl.org/2003/role/disclosureRef
-Topic 470
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1B
-Subparagraph (SX 210.13-02(a)(5))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480097/470-10-S99-1B
Reference 29: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 30
-Subparagraph (b)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-30
Reference 30: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (f)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 31: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 60B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-60B
Reference 32: http://www.xbrl.org/2003/role/exampleRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 31
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-31
Reference 33: http://www.xbrl.org/2003/role/disclosureRef
-Topic 280
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 32
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482810/280-10-50-32
Reference 34: http://www.xbrl.org/2003/role/disclosureRef
-Topic 205
-SubTopic 20
-Name Accounting Standards Codification
-Section 50
-Paragraph 7
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483499/205-20-50-7
Reference 35: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 230
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 28
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482740/230-10-45-28
Reference 36: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1A
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1A
Reference 37: http://www.xbrl.org/2003/role/disclosureRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 1B
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482790/220-10-45-1B
Reference 38: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 220
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03(20))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483621/220-10-S99-2
Reference 39: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 942
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.9-04(22))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483589/942-220-S99-1
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe average number of shares or units issued and outstanding that are used in calculating diluted EPS or earnings per unit (EPU), determined based on the timing of issuance of shares or units in the period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 16
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-16
| Name: |
us-gaap_WeightedAverageNumberOfDilutedSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of [basic] shares or units, after adjustment for contingently issuable shares or units and other shares or units not deemed outstanding, determined by relating the portion of time within a reporting period that common shares or units have been outstanding to the total time in that period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 45
-Paragraph 10
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482689/260-10-45-10
| Name: |
us-gaap_WeightedAverageNumberOfSharesOutstandingBasic |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.24.1.u1
Net Loss Per Share - Anti-dilutive Securities Excluded from Calculation of Diluted Net Loss Per Share (Details) - shares
|
3 Months Ended |
Mar. 31, 2024 |
Mar. 31, 2023 |
| Antidilutive Securities Excluded From Computation Of Earnings Per Share [Line Items] |
|
|
| Anti-dilutive securities excluded from calculation of diluted net loss per share |
36,196,247
|
27,596,259
|
| Options to Purchase Common Stock |
|
|
| Antidilutive Securities Excluded From Computation Of Earnings Per Share [Line Items] |
|
|
| Anti-dilutive securities excluded from calculation of diluted net loss per share |
34,647,123
|
25,892,271
|
| Options Early Exercised Subject to Future Vesting |
|
|
| Antidilutive Securities Excluded From Computation Of Earnings Per Share [Line Items] |
|
|
| Anti-dilutive securities excluded from calculation of diluted net loss per share |
247,919
|
948,179
|
| Estimated Shares Purchasable Under the ESPP |
|
|
| Antidilutive Securities Excluded From Computation Of Earnings Per Share [Line Items] |
|
|
| Anti-dilutive securities excluded from calculation of diluted net loss per share |
1,301,205
|
755,809
|
| X |
- DefinitionSecurities (including those issuable pursuant to contingent stock agreements) that could potentially dilute basic earnings per share (EPS) or earnings per unit (EPU) in the future that were not included in the computation of diluted EPS or EPU because to do so would increase EPS or EPU amounts or decrease loss per share or unit amounts for the period presented. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 260
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 1
-Subparagraph (c)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147482662/260-10-50-1
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent financial concepts included in a table. These concepts are used to disclose reportable information associated with domain members defined in one or many axes to the table.
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis=eras_OptionsToPurchaseCommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis=eras_OptionsEarlyExercisedSubjectToFutureVestingMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_AntidilutiveSecuritiesExcludedFromComputationOfEarningsPerShareByAntidilutiveSecuritiesAxis=eras_EstimatedSharesPurchasableUnderTheEmployeeStockPurchasePlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
Related Party Transactions - Additional Information (Details) - USD ($)
|
1 Months Ended |
|
|
Apr. 30, 2023 |
Mar. 31, 2024 |
Dec. 31, 2023 |
| Related Party Transaction [Line Items] |
|
|
|
| Prepaid expenses and other current assets |
|
$ 8,938,000
|
$ 8,326,000
|
| Affini T Therapeutics, Inc. |
|
|
|
| Related Party Transaction [Line Items] |
|
|
|
| Investment in equity securities |
|
$ 2,000,000
|
|
| Erasca Foundation |
|
|
|
| Related Party Transaction [Line Items] |
|
|
|
| Amount donated to related party |
$ 125,000
|
|
|
| X |
- DefinitionAmount of investment in equity security without readily determinable fair value. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 321
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 3
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479536/321-10-50-3
| Name: |
us-gaap_EquitySecuritiesWithoutReadilyDeterminableFairValueAmount |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of asset related to consideration paid in advance for costs that provide economic benefits in future periods, and amount of other assets that are expected to be realized or consumed within one year or the normal operating cycle, if longer. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(9))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_PrepaidExpenseAndOtherAssetsCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
us-gaap_EquityMethodInvestmentNonconsolidatedInvesteeAxis=eras_AffiniTTherapeuticsIncMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_LegalEntityAxis=eras_ErascaFoundationMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.24.1.u1
| X |
- DefinitionProceeds from issuance of private placement after deducting placement agent fees and estimated expenses.
| Name: |
eras_ProceedsFromIssuanceOfPrivatePlacementAfterDeductingPlacementAgentFeesAndEstimatedExpenses |
| Namespace Prefix: |
eras_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionPer share amount received by subsidiary or equity investee for each share of common stock issued or sold in the stock transaction.
| Name: |
us-gaap_SaleOfStockPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://fasb.org/us-gaap/role/ref/legacyRef
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-SubTopic 10
-Topic 505
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481112/505-10-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 505
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Subparagraph (a)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481004/946-505-50-2
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 220
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-09(4)(b))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483575/946-220-S99-3
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Topic 946
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 3
-Subparagraph (SX 210.6-03(i)(1))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147479886/946-10-S99-3
Reference 5: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(28))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
Reference 6: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 505
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480008/505-10-S99-1
Reference 7: http://fasb.org/us-gaap/role/ref/legacyRef
-Topic 210
-SubTopic 10
-Name Accounting Standards Codification
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02(29))
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147480566/210-10-S99-1
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDetail information of subsequent event by type. User is expected to use existing line items from elsewhere in the taxonomy as the primary line items for this disclosure, which is further associated with dimension and member elements pertaining to a subsequent event. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Topic 830
-SubTopic 30
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147481674/830-30-50-2
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Topic 855
-SubTopic 10
-Name Accounting Standards Codification
-Section 50
-Paragraph 2
-Publisher FASB
-URI https://asc.fasb.org//1943274/2147483399/855-10-50-2
| Name: |
us-gaap_SubsequentEventLineItems |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_SubsidiarySaleOfStockAxis=eras_TwoThousandAndTwentyFourPrivatePlacementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_SubsequentEventTypeAxis=us-gaap_SubsequentEventMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
Erasca (NASDAQ:ERAS)
Historical Stock Chart
From May 2024 to Jun 2024
Erasca (NASDAQ:ERAS)
Historical Stock Chart
From Jun 2023 to Jun 2024