Eagle Pharmaceuticals Gets FDA Designation for Infant Apnea Treatment
October 03 2022 - 7:54AM
Dow Jones News
By Dean Seal
Eagle Pharmaceuticals Inc. said that regulators have granted
orphan drug designation to its treatment for a breathing disorder
that affects preterm infants.
The U.S. Food and Drug Administration has given the special
status to ENA-001 for the treatment of apnea of prematurity, a
development disorder characterized by the cessation of breathing
for more than 20 seconds or the cessation of breathing for less
than 20 seconds but which is accompanied by either bradycardia or
hypoxemia.
The treatment is designed to work by inhibiting Big Potassium
ion channels in the carotid bodies located in the neck, which
stimulates breathing across multiple causes of respiratory
depression.
Orphan-drug designation is a special status given to drugs that
show promise for potentially treating rare, or orphan, diseases
that have fewer than 200,000 cases a year in the U.S.
Write to Dean Seal at dean.seal@wsj.com
(END) Dow Jones Newswires
October 03, 2022 07:39 ET (11:39 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
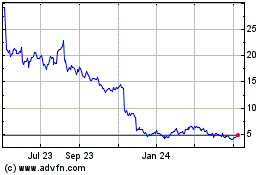
Eagle Pharmaceuticals (NASDAQ:EGRX)
Historical Stock Chart
From Aug 2024 to Sep 2024
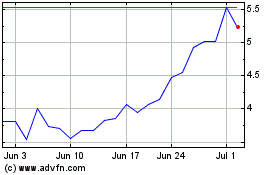
Eagle Pharmaceuticals (NASDAQ:EGRX)
Historical Stock Chart
From Sep 2023 to Sep 2024