Alkermes Gets Fast Track Designation for Ovarian Cancer Treatment
October 25 2021 - 7:44AM
Dow Jones News
By Chris Wack
Alkermes Plc said the U.S. Food and Drug Administration granted
fast track designation to nemvaleukin alfa, the company's
investigational interleukin-2 variant immunotherapy, in combination
with anti-PD-1 antibody pembrolizumab for the treatment of
platinum-resistant ovarian cancer.
The FDA previously granted fast track designation and orphan
drug designation to nemvaleukin for the treatment of mucosal
melanoma.
Fast Track is an FDA process designed to facilitate the
development, and expedite the review, of potential therapies that
seek to treat serious conditions and fill an unmet medical need. A
drug candidate that receives fast track designation is eligible for
more frequent communication with the FDA throughout the drug
development process and a rolling and/or priority review of its
marketing application if relevant criteria are met.
Alkermes shares were up 4% to $31.90 in premarket trading.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
October 25, 2021 07:29 ET (11:29 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
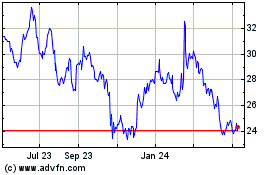
Alkermes (NASDAQ:ALKS)
Historical Stock Chart
From Apr 2024 to May 2024
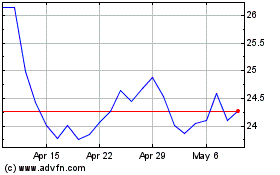
Alkermes (NASDAQ:ALKS)
Historical Stock Chart
From May 2023 to May 2024