Acutus Medical Initiates First IDE Therapy Trial with the AcQBlate® FORCE Sensing Ablation System
April 05 2021 - 8:00AM

Acutus Medical (Nasdaq: AFIB), an arrhythmia management company
focused on improving the way cardiac arrhythmias are diagnosed and
treated, today announced initial US enrollments in the company’s
AcQForce™ Flutter Investigational Device Exemption (IDE) clinical
trial. This trial is expected to enroll up to 150 subjects in
leading centers globally and will evaluate the safety and efficacy
of the AcQBlate FORCE sensing ablation catheter and system in the
treatment of Right Atrial Typical Flutter. In the US, Right Atrial
Typical Flutter ablation procedures currently account for
approximately 30% of cardiac ablations and are expected to reach
200,000 annually by 20251.
In contrast to the most contemporary ablation systems, AcQBlate
FORCE can operate both in a stand-alone manner or in conjunction
with a compatible 3D mapping system. The AcQBlate FORCE sensing
ablation system is comprised of Acutus’ AcQBlate FORCE catheter and
Qubic Force module that seamlessly integrates an RF generator and
irrigation pump. The complete AcQBlate FORCE sensing ablation
catheter and system, which received full CE Mark in late 2020, is
now commercially available in Europe through our Acutus direct
organization and our partner BIOTRONIK.
Designed specifically to provide consistent, effective
therapeutic solutions during cardiac ablation procedures, the
AcQBlate FORCE system shows physicians, in real-time, how much
contact force is being applied to the heart during ablations.
Studies have shown the utility of real-time contact force
information in helping physicians guide safe and effective therapy,
which may improve patient outcomes2.
“The AcQBlate FORCE sensing ablation catheter provides stable
contact force readings with low fluid irrigation requirements. It
displayed both easy maneuverability and excellent stability during
atrial ablation. I look forward to further evaluating this system
and believe this technology has the potential to improve patient
outcomes,” said Dr. Gery Tomassoni of Baptist Health Medical Group
in Lexington, Ky., who performed the first case in the trial.
Dr. Sean Beinart of Adventist Healthcare White Oak Medical
Center in Silver Springs, Md. further commented, “the system was
very easy for our lab staff to set up, and we were able to complete
the case without changing our existing workflow. As the incidence
of atrial arrhythmias increases, we continue to look for safe,
effective and efficient treatment options for patients. The
AcQBlate FORCE sensing ablation catheter incorporates a gold-tipped
electrode that is designed for efficient energy delivery and
effective lesion creation in less time.”
“This trial represents our initial entry into the United States
with a therapeutic technology for the treatment of atrial
arrhythmias,” said Vince Burgess, CEO of Acutus Medical. “The
gold-tipped AcQBlate FORCE catheter is a state-of-the-art ablation
device that is paired with our laser-based contact force console
and further integrated with the smallest and most modern RF
generator and pump available. As we have experienced in our
European centers during our initial market release, when used in
concert with our novel AcQMap 3D mapping system we are seeing
highly efficient workflow and operational efficiency. We look
forward to offering the benefits of a comprehensive, force sensing
ablation system to electrophysiologists and patients in the United
States once we complete clinical trials and gain regulatory
approval.”
Enrollment for the AcQForce Flutter trial is ongoing, and the
company is actively working with selected sites to initiate cases
under the IDE. For more information on this trial, please see
NCT04658940 on https://www.clinicaltrials.gov. AcQBlate FORCE
Sensing Catheter is limited by US Federal Law to investigational
use.
References1. Erik J. Bracciodieta,
Electrophysiology Mapping and Ablation Devices, Market Insights,
US. Decision Resources Group M360EP0059, June 2019.2. Ariyarathna
N. et al., Role of Contact Force Sensing in Catheter Ablation of
Cardiac Arrhythmias: Evolution or History Repeating Itself? JACC:
Clinical Electrophysiology (2018); 707-723
About Acutus Medical Acutus Medical is an
arrhythmia management company focused on improving the way cardiac
arrhythmias are diagnosed and treated. Acutus is committed to
advancing the field of electrophysiology with a unique array of
products and technologies which will enable more physicians to
treat more patients more efficiently and effectively. Through
internal product development, acquisitions and global partnerships,
Acutus has established a global sales presence delivering a broad
portfolio of highly differentiated electrophysiology products that
provide its customers with a complete solution for catheter-based
treatment of cardiac arrhythmias. Founded in 2011, Acutus is based
in Carlsbad, California.
Follow Acutus Medical on:
- Twitter: @AcutusMedical
- LinkedIn: www.linkedin.com/company/acutus-medical-inc-/
- Facebook: @AcutusMedical
Media ContactHolly
Windler619-929-1275holly.windler@acutus.com
Investor ContactCaroline Corner
415-202-5678caroline.corner@westwicke.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/6c455e7c-6377-4f65-b55d-b51acd15a2ea
https://www.globenewswire.com/NewsRoom/AttachmentNg/3f011ba9-59e5-430c-a559-b9d66b6822b8
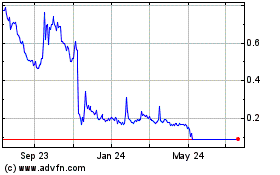
Acutus Medical (NASDAQ:AFIB)
Historical Stock Chart
From Mar 2024 to Apr 2024
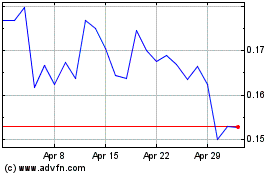
Acutus Medical (NASDAQ:AFIB)
Historical Stock Chart
From Apr 2023 to Apr 2024