NANOBIOTIX PARTNERS WITH THE PROVIDENCE
CANCER INSTITUTE TO RUN IMMUNOTHERAPEUTIC PRECLINICAL RESEARCH IN
PANCREATIC CANCERS
Paris, France and Cambridge, Massachusetts,
USA, January 11, 2018 - NANOBIOTIX (Euronext: NANO - ISIN:
FR0011341205), a late clinical-stage nanomedicine company
pioneering new approaches to the treatment of cancer, today
announced it will begin a pre-clinical collaboration with
Providence Cancer Institute to study Nanobiotix's lead product,
NBTXR3, a first-in-class nanoparticle designed for direct injection
into cancerous tumors and activation by radiotherapy.
The collaboration with Providence Cancer
Institute, located at the Robert W. Franz Cancer Center in
Portland, Ore., one of the world's leading oncological research
centers, will provide essential preclinical data on the ability of
NBTXR3 activated by radiotherapy to induce an antitumoral immune
response. This is an in-depth study into the early immunologic
mechanisms, triggered by nanoparticles activated by radiotherapy
compared to radiotherapy alone, and their impact on tumor control,
survival and metastasis spreading.
Marka R. Crittenden, M.D., Ph.D., radiation
oncologist and director of Translational Radiation Research at the
Robert W. Franz Cancer Center will lead the program. "Promising
pre-clinical data suggests that nanoparticles combined with
radiotherapy enhance tumor-specific immune responses and lead to an
abscopal response, priming a patient's immune system to attack
cancer cells outside of the radiotherapy target area," she said.
"Furthermore, we are absolutely delighted to partner with
Nanobiotix to advance our research on this phenomenon and the role
NBTXR3 can play in immuno-oncology."
The collaboration between Providence and
Nanobiotix will take place over the course of one year, and will
evaluate the use of NBTXR3 activated by radiotherapy in pancreatic
cancer models (in vitro and in vivo). Pancreatic cancer is a
disease with a substantial unmet medical need, poor response to
standard of care and is the third leading cause of cancer mortality
in both men and women in the United State. Pancreatic cancers have
a non-immunogenic tumor microenvironnement, known as "cold tumors",
and often have a poor response rate to immunotherapies.
The results of this joint program will enable
the potential to explore future use of NBTXR3 in immuno-oncology as
well as its potential to control metastatic disease.
In parallel, the Company has received the
approval of its Investigational New Drug (IND) and will launch its
first clinical trial combining NBTXR3 with immune checkpoint
inhibitors in the U.S. with a multi-arm trial targeting a
sub-population of advanced lung cancer patients and head and neck
cancer patients. Nanobiotix's immuno-oncology combination program
opens the door to new developments, potential new indications and
important value creation opportunities.
*** About
Providence Cancer Institute
Providence Cancer Institute, a part of
Providence Health & Services, offers the latest in cancer
services, including diagnostic, treatment, prevention, education,
support and internationally-renowned research. Located within the
Robert W. Franz Cancer Center in Portland, Ore., Providence Cancer
Institute is home to the Earle A. Chiles Research Institute, a
world-class research facility for cancer immunotherapy, a
specialized field of study focused on triggering the immune system
to fight cancer. Visit www.providenceoregon.org/cancer to learn
more.
About NBTXR3
NBTXR3 is an injectable aqueous suspension of
hafnium oxide nanoparticles designed as an innovative therapeutic
agent for the treatment of solid tumors, currently in clinical
development by Nanobiotix.
Once injected intratumorally, NBTXR3 can deposit
high energy within tumors only when activated by an ionizing
radiation source, notably radiotherapy. Upon activation, the high
energy radiation is physically designed to kill the tumor cells by
triggering DNA damage and cell destruction and improve clinical
outcomes.
Promising results indicate that NBTXR3 activity
could be applicable across solid tumors triggering immunogenic cell
death, leading to an immune response, reinforcing a local and
potentially systemic effect, and contributing to transform "cold"
tumors into "hot" tumors. NBTXR3's major characteristics are
represented by a high degree of biocompatibility, one single
administration before and during the whole therapy and the ability
to fit into current standards of radiotherapy care.
NBTXR3 entered clinical development in 2011 in a
Phase I/II with patients suffering from advanced soft tissue
sarcoma of the extremities and is currently in the final stages of
its subsequent phase II/III. In parallel, it is currently being
tested in numerous Phase I/II clinical trials with patients
suffering from locally advanced squamous cell carcinoma of the oral
cavity or oropharynx (head and neck), liver cancer (hepatocellular
carcinoma and liver metastasis), locally advanced or unresectable
rectal cancer in combination with chemotherapy, head and neck
cancer in combination with concurrent chemotherapy, and prostate
adenocarcinoma.
About NANOBIOTIX: www.nanobiotix.com
Nanobiotix (Euronext: NANO / ISIN: FR0011341205)
is a late clinical-stage nanomedicine company pioneering novel
approaches for the treatment of cancer. The Company's
first-in-class, proprietary technology, NanoXray, enhances
radiotherapy energy with a view to providing a new, more efficient
treatment for cancer patients.
NanoXray products are compatible with current
radiotherapy treatments and are meant to treat potentially a wide
variety of solid tumors including soft tissue sarcoma, head and
neck cancers, liver cancers, prostate cancer, breast cancer,
glioblastoma, etc., via multiple routes of administration.
NBTXR3 is being evaluated in: soft tissue
sarcoma (STS), head and neck cancers, prostate cancer, and liver
cancers (primary and metastases). Additionally, head and neck
cancer and rectal cancer trials led by Nanobiotix's Taiwanese
partner, PharmaEngine, are underway in the Asia Pacific region.
The Company started a new preclinical research
program in Immuno-oncology with its lead product NBTXR3, which
could have the potential to bring a new dimension to cancer
immunotherapies.
Nanobiotix is listed on the regulated market of
Euronext in Paris (ISIN: FR0011341205, Euronext ticker: NANO,
Bloomberg: NANO: FP). The Company's Headquarters is based in Paris,
France, with a U.S. affiliate in Cambridge, MA.
Contact
| Nanobiotix |
|
Sarah GaubertDirector, Communications & Public
Affairs+33 (0)1 40 26 07 55sarah.gaubert@nanobiotix.com
/contact@nanobiotix.com |
Noël Kurdi Director, Investor
Relations +1 (646) 241-4400 noel.kurdi@nanobiotix.com /
investors@nanobiotix.com |
| Media relations |
| NanobiotixFrance -
Springbok ConsultantsMarina Rosoff+33 (0)6 71 58 00
34marina@springbok.fr |
|
United States -
RooneyPartners Marion Janic +1 (212)
223-4017mjanic@rooneyco.com |
|
Providence Cancer InstituteJean Powell Marks+1 (503)
215-6433jean.marks@providence.org
Disclaimer
This press release contains certain
forward-looking statements concerning Nanobiotix and its business.
Such forward-looking statements are based on assumptions that
Nanobiotix considers to be reasonable. However, there can be no
assurance that the estimates contained in such forward-looking
statements will be verified, which estimates are subject to
numerous risks including the risks set forth in the reference
document of Nanobiotix filed with the French Financial Markets
Authority (Autorité des Marchés Financiers) under number D.17-0470
on April 28, 2017 (a copy of which is available on
www.nanobiotix.com) and to the development of economic conditions,
financial markets and the markets in which Nanobiotix operates. The
forward-looking statements contained in this press release are also
subject to risks not yet known to Nanobiotix or not currently
considered material by Nanobiotix. The occurrence of all or part of
such risks could cause actual results, financial conditions,
performance or achievements of Nanobiotix to be materially
different from such forward-looking statements.
This press release and the information that it
contains do not constitute an offer to sell or subscribe for, or a
solicitation of an offer to purchase or subscribe for, Nanobiotix
shares in any country. At the moment NBTXR3 does not bear a CE mark
and is not permitted to be placed on the market or put into service
until NBTXR3 has obtained a CE mark.
Nanobiotix (EU:NANO)
Historical Stock Chart
From Jun 2024 to Jul 2024
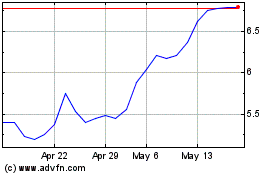
Nanobiotix (EU:NANO)
Historical Stock Chart
From Jul 2023 to Jul 2024