As filed with the Securities and Exchange Commission
on December 19, 2023
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
————————————
FORM S-3
REGISTRATION STATEMENT
UNDER THE SECURITIES ACT OF 1933
————————————
NIGHTHAWK BIOSCIENCES, INC.
(Exact name of registrant as specified in its charter)
| Delaware |
6331 |
26-2844103 |
| (State or other jurisdiction of incorporation or organization) |
(Primary Standard Industrial
Classification Code Number) |
(I.R.S. Employer
Identification No.) |
627 Davis Drive, Suite 300
Morrisville, North Carolina 27560
(919) 240-7133
(Address, including zip code, and telephone number,
including area code, of registrant’s principal executive offices)
————————————
Jeff Wolf
Chief Executive Officer
627 Davis Drive, Suite 300
Morrisville, North Carolina 27560
(919) 240-7133
(Name, address, including zip code, and telephone number,
including area code, of agent for service)
————————————
The Commission is requested to send copies of
all communications to:
|
Leslie Marlow, Esq.
Patrick J. Egan, Esq.
Hank Gracin, Esq.
Melissa Palat Murawsky, Esq.
Blank Rome LLP
1271 Avenue of the Americas
New York, New York 10020
Phone: (212) 885-5000
Fax: (212) 885-5001 |
|
|
————————————
Approximate date of commencement of proposed sale
to the public: From time to time after the effective date of this registration statement, as determined by market conditions.
If the only securities being registered on this Form
are being offered pursuant to dividend or interest reinvestment plans, please check the following box. ☐
If any of the securities being registered on this
Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, other than securities offered
only in connection with dividend or interest reinvestment plans, check the following box. ☒
If this Form is filed to register additional securities
for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration
statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant
to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier
effective registration statement for the same offering. ☐
If this Form is a registration statement pursuant
to General Instruction I.D. or a post-effective amendment thereto that shall become effective upon filing with the Commission pursuant
to Rule 462(e) under the Securities Act, check the following box. ☐
If this Form is a post-effective amendment to a registration
statement filed pursuant to General Instruction I.D. filed to register additional securities or additional classes of securities pursuant
to Rule 413(b) under the Securities Act, check the following box. ☐
Indicate by check mark whether the registrant is a
large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See
the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and
“emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer |
☐ |
Accelerated filer |
☐ |
| |
|
|
|
| Non-accelerated filer |
☒ |
Smaller reporting company |
☒ |
| |
|
|
|
| |
|
Emerging growth company |
☐ |
If an emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided to
Section 7(a)(2)(B) of the Securities Act. ☐
————————————
The Registrant hereby amends this registration
statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment that
specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities
Act of 1933, as amended, or until this registration statement shall become effective on such date as the Securities and Exchange Commission,
acting pursuant to said Section 8(a), may determine.
EXPLANATORY NOTE
This registration statement
is a replacement registration statement being filed pursuant to Rule 415(a)(6) under the Securities Act of 1933, as amended (the “Securities
Act”), with respect to securities which remain unsold under the Registration Statement on Form S-3 (File No. 333-251255), originally
filed on December 10, 2020, amended on December 18, 2020 and declared effective on December 22, 2020 (the “Prior Registration Statement”).
Pursuant to Rule 415(a)(5)(ii) under the Securities Act, by filing this registration statement on Form S-3 (the “Registration Statement”),
the Company may issue and sell securities covered by the Prior Registration Statement until the earlier of (i) the effective date of this
Registration Statement and (ii) June 19, 2024. which is 180 days after the third-year anniversary of the effective date of the Prior
Registration Statement.
This registration statement of
NightHawk Biosciences, Inc. contains two prospectuses:
| |
● |
a base prospectus, which covers the offering, issuance and sale by us of common stock, preferred stock, warrants, and units identified above from time to time in one or more offerings, which together shall have an aggregate initial offering price not to exceed $200,000,000; and |
| |
● |
an “at the market offering” prospectus covering the offer, issuance and sale by us of up to a maximum aggregate offering price of up to $4,037,193 of our common stock that may be issued and sold from time to time under the sales agreement with A.G.P./Alliance Global Partners. |
The base prospectus immediately follows this
explanatory note. The specific terms of any securities to be offered pursuant to the base prospectus will be specified in one or
more prospectus supplements to the base prospectus. The sales agreement prospectus immediately follows the base prospectus. The
$4,037,193 of common stock that may be offered, issued and sold under the sales agreement prospectus is included in the
$200,000,000 of securities that may be offered, issued and sold by us under the base prospectus. Upon termination of the sales
agreement, any portion of the $4,037,193 included in the sales agreement prospectus that is not sold pursuant to the sales agreement
will be available for sale in other offerings pursuant to the base prospectus, and if no shares are sold under the sales agreement,
the full $4,037,193 of securities may be sold in other offerings pursuant to the base prospectus.
The information contained in this prospectus is not complete
and may be changed. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission
is effective. This prospectus is not an offer to sell these securities and is not soliciting an offer to buy these securities in any jurisdiction
where the offer or sale is not permitted.
Subject to Completion, Dated
December 19, 2023
PROSPECTUS
$200,000,000

————————————
Common Stock
Preferred Stock
Warrants
Units
————————————
We may, from time to time, offer and sell up to $200,000,000
of any combination of our common stock, preferred stock warrants, or units described in this prospectus, either individually or in combination
with other securities, at prices and on terms described in one or more supplements to this prospectus. We may also offer common stock
upon conversion of preferred stock, or common stock or preferred stock upon the exercise of warrants or units.
This prospectus provides you with a general description
of the securities that we may offer. Each time we offer and sell securities, we will provide a supplement to this prospectus that contains
specific information about the offering and the amounts, prices and terms of the securities. We may also authorize one or more free writing
prospectuses to be provided to you in connection with these offerings. The prospectus supplement and any related free writing prospectus
may also add, update or change information contained in this prospectus. You should carefully read this prospectus, the applicable prospectus
supplement and any related free writing prospectus, as well as the documents incorporated by reference, before buying any of the securities
being offered.
Securities may be sold by us to or through underwriters
or dealers, directly to purchasers or through agents designated from time to time. For additional information on the methods of sale,
you should refer to the section entitled “Plan of Distribution” in this prospectus and in the applicable prospectus supplement.
If any underwriters, dealers or agents are involved in the sale of any of the securities, their names and any applicable purchase price,
fee, commission or discount arrangement between or among them will be set forth, or will be calculable from the information set forth,
in the applicable prospectus supplement. The price to the public of such securities and the net proceeds we expect to receive from such
sale will also be set forth in a prospectus supplement. No securities may be sold without delivery of this prospectus and the applicable
prospectus supplement describing the method and terms of the offering of such securities.
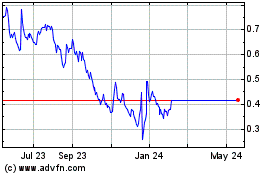
Our common stock is listed on the NYSE American LLC,
or the NYSE American, under the symbol “NHWK.” On December 18, 2023, the last
reported sale price of our common stock on the NYSE American was $0.4457 per share.
The applicable prospectus supplement will contain information, where applicable, as to any other listing, if any, on any securities market
or other exchange of the specific security covered by such prospectus supplement.
As of the date of this prospectus, the aggregate market value
of our outstanding common stock held by non-affiliates is $12,111,581, which is calculated based on 24,742,760 shares of our outstanding
common stock held by non-affiliates and a price of $0.4895 per share, the closing price of our common stock on November 7, 2023, which
is the highest closing sale price of our common stock on the NYSE American within the prior 60 days of this prospectus supplement. During
the prior twelve calendar month period that ends on and includes the date hereof, we have not offered or sold any shares of our common
stock pursuant to General Instruction I.B.6 to Form S-3.
Investing in our securities involves a high degree
of risk. You should review carefully the risks and uncertainties described under the heading “Risk Factors” beginning
on page 5 of this prospectus and contained in the applicable prospectus supplement
and in any free writing prospectuses we have authorized for use in connection with a specific offering, and under similar headings in
the other documents that are incorporated by reference into this prospectus.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete.
Any representation to the contrary is a criminal offense.
The date of this prospectus is
, 2023.
TABLE OF CONTENTS
i
ABOUT THIS PROSPECTUS
This prospectus is part of a registration statement
on Form S-3 that we filed with the Securities and Exchange Commission (the “SEC”) using a “shelf” registration
process. Under this shelf registration statement, we may sell from time to time in one or more offerings up to a total dollar amount of
$200,000,000 of common stock and preferred stock, warrants to purchase any of such securities, either individually or as units in combination
with other securities as described in this prospectus. Each time we sell any type or series of securities under this prospectus, we will
provide a prospectus supplement that will contain more specific information about the terms of that offering. We may also authorize one
or more free writing prospectuses to be provided to you that may contain material information relating to these offerings. The prospectus
supplement and any related free writing prospectus that we may authorize to be provided to you may also add, update or change any of the
information contained in this prospectus or in the documents we have incorporated by reference into this prospectus. To the extent that
any statement that we make in a prospectus supplement is inconsistent with statements made in this prospectus, the statements made in
this prospectus will be deemed modified or superseded by those made in a prospectus supplement. You should carefully read both this prospectus
and the applicable prospectus supplement and any related free writing prospectus, together with the additional information described under
“Where You Can Find More Information,” before buying any of the securities being offered. THIS PROSPECTUS MAY NOT
BE USED TO CONSUMMATE A SALE OF SECURITIES UNLESS IT IS ACCOMPANIED BY A PROSPECTUS SUPPLEMENT
Neither we, nor any agent, underwriter or dealer has
authorized any person to give any information or to make any representation other than those contained or incorporated by reference in
this prospectus, any applicable prospectus supplement or any related free writing prospectus prepared by or on behalf of us or to which
we have referred you. If anyone provides you with different or inconsistent information, you should not rely on it. This prospectus, any
applicable supplement to this prospectus or any related free writing prospectus does not constitute an offer to sell or the solicitation
of an offer to buy any securities other than the registered securities to which they relate, nor does this prospectus, any applicable
supplement to this prospectus or any related free writing prospectus constitute an offer to sell or the solicitation of an offer to buy
securities in any jurisdiction to any person to whom it is unlawful to make such offer or solicitation in such jurisdiction.
You should not assume that the information contained
in this prospectus, any applicable prospectus supplement or any related free writing prospectus is accurate on any date subsequent to
the date set forth on the front of the document or that any information we have incorporated by reference is correct on any date subsequent
to the date of the document incorporated by reference, even though this prospectus, any applicable prospectus supplement or any related
free writing prospectus is delivered, or securities are sold, on a later date.
This prospectus contains summaries of certain provisions
contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the
summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed,
will be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and you
may obtain copies of those documents as described below under the section entitled “Where You Can Find More Information.”
Except as otherwise indicated herein or as the context
otherwise requires, references in this prospectus to “NightHawk,” the “Company,” “we,” “us,”
“our” and similar references refer to NightHawk Biosciences, Inc., an entity
incorporated under the laws of the State of Delaware, and where appropriate our consolidated subsidiaries.
This prospectus and the information incorporated herein
by reference include trademarks, service marks and trade names owned by us or other companies. All trademarks, service marks and trade
names included or incorporated by reference into this prospectus, any applicable prospectus supplement or any related free writing prospectus
are the property of their respective owners.
PROSPECTUS SUMMARY
The following summary highlights information contained
elsewhere in this prospectus or incorporated by reference herein and does not contain all the information that may be important to purchasers
of our securities. Prospective purchasers of our securities should carefully read the entire prospectus, the applicable prospectus supplement
and any related free writing prospectus, including the risks of investing in our securities discussed under the heading “Risk Factors”
contained in this prospectus, the applicable prospectus supplement and any related free writing prospectus, and under similar headings
in the other documents that are incorporated by reference into this prospectus. Prospective purchasers of our securities should also carefully
read the information incorporated by reference into this prospectus, including our financial statements, and the exhibits to the registration
statement of which this prospectus is a part.
Overview
Our current focus is on our contract development and
manufacturing organization (“CDMO”) that provides a comprehensive range of services from process development to Current Good
Manufacturing Practices (“CGMP”) clinical and commercial manufacturing of biologics for the biotechnology and biopharmaceutical
industries through our Scorpius Biomanufacturing, Inc. (“Scorpius”) subsidiary. Scorpius pairs cGMP biomanufacturing and quality
control expertise with cutting edge capabilities in immunoassays, molecular assays, and bioanalytical methods to support cell- and gene-based
therapies as well as large molecule biologics. Our services include clinical and commercial drug substance manufacturing, release and
stability testing and variety of process development services, including upstream and downstream development and optimization, analytical
method development, cell line development, testing and characterization. Our lead facility in San Antonio, Texas commenced operations
in October 2022.
Corporate Information
We are a Delaware corporation. Our principal executive
offices are located at 627 Davis Drive, Suite 300, Morrisville, North Carolina 27560 and our telephone number at this address is (919)
240-7133. Our website is www.nighthawkbio.com. Information contained on, or that may be accessible through, our website is not
a part of, and is not incorporated into, this prospectus.
Smaller Reporting Company
We are a “smaller reporting company” as
defined in the Securities Exchange Act of 1934, as amended (the “Exchange Act”). As a result, we may take advantage of certain
reduced disclosure obligations available to smaller reporting companies, including the exemption from compliance with the auditor attestation
requirements pursuant to the Sarbanes-Oxley Act of 2022, reduced disclosure about our executive compensation arrangements and the requirements
to provide only two years of audited financial statements in our annual reports and registration statements. We will continue to be a
“smaller reporting company” as long as (1) we have a public float (i.e., the market value of our common stock held by non-affiliates)
less than $250 million calculated as of the last business day of our most recently completed second fiscal quarter, or (2) our annual
revenues are less than $100 million for our previous fiscal year and we have either no public float or a public float of less than $700
million as of the end of that fiscal year’s second fiscal quarter. Decreased disclosures in our SEC filings due to our status as
a “smaller reporting company” may make it harder for investors to analyze our results of operations and financial prospects.
Risks Associated with our Business
Our business is subject to numerous risks, as described
under the heading “Risk Factors” contained in the applicable prospectus supplement and in any free writing prospectuses we
have authorized for use in connection with a specific offering, and under similar headings in the documents that are incorporated by reference
into this prospectus.
The Securities We May Offer
We may offer shares of our common stock, preferred
stock and/or warrants to purchase any of such securities, either individually or as units in combination with other securities, with a
total value of up to $200,000,000 from time to time under this prospectus at prices and on terms to be determined at the time of any
offering. This prospectus provides you with a general description of the securities we may offer. Each time we offer a type or series
of securities under this prospectus, we will provide a prospectus supplement that will describe the specific amounts, prices and other
important terms of the securities, including, to the extent applicable:
| |
● |
designation or classification; |
| |
● |
aggregate offering price; |
| |
● |
rates and times of payment of interest or dividends; |
| |
● |
redemption, conversion, exercise, exchange or sinking fund terms; |
| |
● |
ranking; |
| |
● |
restrictive covenants; |
| |
● |
voting or other rights; |
| |
● |
conversion or exchange prices or rates and, if applicable, any provisions for changes to or adjustments in the conversion or exchange prices or rates and in the securities or other property receivable upon conversion or exchange; and |
| |
● |
a discussion of material United States federal income tax considerations, if any. |
The prospectus supplement and any related free writing
prospectus that we may authorize to be provided to you may also add, update or change information contained in this prospectus or in documents
we have incorporated by reference. However, no prospectus supplement or free writing prospectus will offer a security that is not registered
and described in this prospectus at the time of the effectiveness of the registration statement of which this prospectus is a part.
This prospectus may not be used to consummate a
sale of securities unless it is accompanied by a prospectus supplement.
We may sell the securities directly to investors
or to or through agents, underwriters or dealers. We, and our agents, underwriters or dealers reserve the right to accept or reject all
or part of any proposed purchase of securities. If we do offer securities to or through agents, underwriters or dealers, we will include
in the applicable prospectus supplement:
| |
|
|
| |
● |
the names of those agents, underwriters or dealers; |
| |
● |
applicable fees, discounts and commissions to be paid to them; |
| |
● |
details regarding over-allotment options, if any; and |
| |
● |
the net proceeds to us. |
The following is a summary of the securities we may offer with this prospectus.
Common Stock
We may issue shares of our common stock from time
to time. Each holder of our common stock is entitled to one vote for each share on all matters submitted to a vote of the stockholders,
including the election of directors. Under our Third Amended and Restated Certificate of Incorporation, as amended as of the date of this
prospectus (the “Certificate of Incorporation”), and our Second Amended and Restated bylaws (the “Bylaws”), our
stockholders do not have cumulative voting rights. Subject to preferences that may be applicable to any then-outstanding preferred stock,
holders of common stock are entitled to receive ratably those dividends, if any, as may be declared from time to time by the board of
directors out of legally available funds. In the event of our liquidation, dissolution or winding up, holders of common stock are entitled
to share ratably in the net assets legally available for distribution to stockholders after the payment of all of our debts and other
liabilities and the satisfaction of any liquidation preference granted to the holders of any then-outstanding shares of preferred stock.
Holders of shares of our common stock do not have preemptive, subscription, redemption, or conversion rights and there are no sinking
fund provisions applicable to the common stock. The rights, preferences and privileges of the holders of common stock are subject to,
and may be adversely affected by, the rights of the holders of shares of any series of preferred stock that we may designate in the future.
In this prospectus, we have summarized certain general features of our
common stock under “Description of Capital Stock—Common Stock.” We urge you, however, to read the applicable prospectus
supplement (and any related free writing prospectus that we may authorize to be provided to you) related to any common stock being offered.
Preferred Stock
We may issue shares of our preferred stock from time
to time, in one or more series. The creation of any series of preferred stock, as of the date of this prospectus, requires the vote of
our stockholders. Our board of directors will determine the designations, voting powers, preferences and rights of the preferred stock,
as well as the qualifications, limitations or restrictions thereof, including dividend rights, conversion rights, preemptive rights, terms
of redemption or repurchase, liquidation preferences, sinking fund terms and the number of shares constituting any series or the designation
of any series, all, as of the date of this prospectus, subject to approval of our stockholders. Convertible preferred stock will be convertible
into our common stock or exchangeable for other securities. Conversion may be mandatory or at the holder’s option and would be at
prescribed conversion rates. If we sell any series of preferred stock under this prospectus, we will fix the designations, voting powers,
preferences and rights of such series of preferred stock, as well as the qualifications, limitations or restrictions thereof, in the certificate
of designation relating to that series. We will file as an exhibit to the registration statement of which this prospectus is a part, or
will incorporate by reference from reports that we file with the Securities and Exchange Commission, or SEC, the form of any certificate
of designation that describes the terms of the series of preferred stock that we are offering before the issuance of the related series
of preferred stock.
In this prospectus, we have summarized certain general
features of the preferred stock under “Description of Capital Stock—Preferred Stock.” We urge you, however, to read
the applicable prospectus supplement (and any free writing prospectus that we may authorize to be provided to you) related to the series
of preferred stock being offered, as well as the complete certificate of designation that contains the terms of the applicable series
of preferred stock.
Warrants
We may issue warrants for the purchase of common stock,
preferred stock and/or debt securities in one or more series. We may issue warrants independently or as units in combination with common
stock, preferred stock and/or debt securities, and the warrants may be attached to or separate from these securities. In this prospectus,
we have summarized certain general features of the warrants under “Description of Warrants.”
We urge you, however, to read the applicable prospectus
supplement (and any free writing prospectus that we may authorize to be provided to you) related to the series of warrants being offered,
as well as any warrant agreements and warrant certificates that contain the terms of the warrants. We will file as exhibits to the registration
statement of which this prospectus is a part, or will incorporate by reference from reports that we file with the SEC, the form of warrant
and/or the warrant agreement and warrant certificate, as applicable, that contain the terms of the particular series of warrants we are
offering, and any supplemental agreements, before the issuance of such warrants.
Any warrants issued under this prospectus may be evidenced
by warrant certificates. Warrants also may be issued under an applicable warrant agreement that we enter into with a warrant agent. We
will indicate the name and address of the warrant agent, if applicable, in the prospectus supplement relating to the particular series
of warrants being offered.
Units
We may issue units consisting of any combination of
the other types of securities offered under this prospectus in one or more series. We may evidence each series of units by unit certificates
that we will issue under a separate agreement. We may enter into unit agreements with a unit agent. Each unit agent will be a bank or
trust company that we select. We will indicate the name and address of the unit agent in the applicable prospectus supplement relating
to a particular series of units.
In this prospectus, we have summarized certain general features of the
units under “Description of Units.” We urge you, however, to read the applicable prospectus supplement (and any related free
writing prospectus that we may authorize to be provided to you) related to the series of units being offered, as well as the complete
unit agreement that contains the terms of the units. We will file as exhibits to the registration statement of which this prospectus is
a part, or will incorporate by reference from reports that we file with the SEC, the specific unit agreement that contains the terms of
the particular series of units we are offering, before the issuance of such units.
RISK FACTORS
Investing in our securities involves a high
degree of risk. Before deciding whether to invest in our securities, you should consider carefully the risks and uncertainties described
under the heading “Risk Factors” contained in the applicable prospectus supplement and any related free writing prospectus,
and discussed under the section entitled “Risk Factors” contained in our most recent Annual Report on Form 10-K, as may be
updated by subsequent annual, quarterly and other reports that are incorporated by reference into this prospectus in their entirety. The
risks described in these documents are not the only ones we face, but those that we consider to be material. There may be other unknown
or unpredictable economic, business, competitive, regulatory or other factors that could have material adverse effects on our future results.
Past financial performance may not be a reliable indicator of future performance, and historical trends should not be used to anticipate
results or trends in future periods. If any of these risks actually occurs, our business, financial condition, results of operations or
cash flow could be seriously harmed. This could cause the trading price of our common stock to decline, resulting in a loss of all or
part of your investment. Please also read carefully the section below entitled “Forward-Looking Statements.”
FORWARD-LOOKING STATEMENTS
This prospectus, including the documents that we incorporate
by reference herein, contains, and any applicable prospectus supplement or free writing prospectus including the documents we incorporate
by reference therein may contain, forward-looking statements within the meaning of Section 27A of the Securities Act and Section 21E of
the Exchange Act, including statements regarding our future financial condition, business strategy and plans and objectives of management
for future operations. Forward-looking statements include all statements that are not historical facts. In some cases, you can identify
forward-looking statements by terminology such as “believe,” “will,” “may,” “estimate,”
“continue,” “anticipate,” “intend,” “should,” “plan,” “might,”
“approximately,” “expect,” “predict,” “could,” “potentially” or the negative
of these terms or other similar expressions. Forward-looking statements include statements regarding our intentions, beliefs, projections,
outlook, analyses or current expectations.
Discussions containing these forward-looking statements
may be found, among other places, in the sections entitled “Business,” “Risk Factors” and “Management’s
Discussion and Analysis of Financial Condition and Results of Operations” contained in the documents incorporated by reference herein,
including our most recent Annual Report on Form 10-K and our Quarterly Reports on Form 10-Q, as well as any amendments thereto.
These statements relate to future events or our future
financial performance and involve known and unknown risks, uncertainties and other factors that could cause our actual results, levels
of activity, performance or achievement to differ materially from those expressed or implied by these forward-looking statements. We discuss
in greater detail, and incorporate by reference into this prospectus in their entirety, many of these risks and uncertainties under the
heading “Risk Factors” contained in the applicable prospectus supplement, in any free writing prospectus we may authorize
for use in connection with a specific offering, and in the documents incorporated by reference herein. These statements reflect our current
views with respect to future events and are based on assumptions and subject to risks and uncertainties. We undertake no obligation to
revise or publicly release the results of any revision to these forward-looking statements, except as required by law. Given these risks
and uncertainties, readers are cautioned not to place undue reliance on such forward-looking statements. All forward-looking statements
are qualified in their entirety by this cautionary statement.
USE OF PROCEEDS
We will retain broad discretion over the use of the
net proceeds from the sale of the securities offered hereby. Except as described in any prospectus supplement or in any related free writing
prospectus that we may authorize to be provided to you, we currently intend to use the net proceeds from the sale of the securities offered
by us hereunder primarily for working capital and general corporate purposes, including the repurchase of securities. We will set forth
in the applicable prospectus supplement or free writing prospectus our intended use for the net proceeds received from the sale of any
securities sold pursuant to the prospectus supplement or free writing prospectus.
DESCRIPTION OF CAPITAL STOCK
The following description of certain terms of our
capital stock does not purport to be complete and is in all respects subject to, and qualified in its entirety by reference to, the relevant
provisions of our Certificate of Incorporation, our Bylaws and Delaware corporate law. You are strongly encouraged to read our Certificate
of Incorporation and our Bylaws in their entirety for a complete description of the rights and preferences of the securities described
below, copies of which have been filed with the SEC. These documents are also incorporated by reference into the registration statement
of which this prospectus forms a part.
Common Stock
We are authorized to issue 250,000,000 shares of common
stock, par value $0.0002 per share, of which 26,081,890 shares are outstanding as of December 13, 2023.
Voting. The holders of our common
stock are entitled to one vote for each share held of record on all matters submitted to a vote of the stockholders, including the election
of directors, and do not have cumulative voting rights.
Dividends. Subject to preferences
that may be applicable to any then outstanding preferred stock, the holders of common stock are entitled to receive dividends, if any,
as may be declared from time to time by our board of directors out of legally available funds.
Liquidation. In the event of our liquidation,
dissolution or winding up, holders of our common stock will be entitled to share ratably in the net assets legally available for distribution
to stockholders after the payment of all of our debts and other liabilities, subject to the satisfaction of any liquidation preference
granted to the holders of any then outstanding shares of preferred stock.
Rights and Preferences. The holders of
our common stock have no preemptive, conversion or subscription rights, and there are no redemption or sinking fund provisions applicable
to our common stock. The rights, preferences and privileges of the holders of our common stock are subject to, and may be adversely affected
by, the rights of the holders of shares of any series of our preferred stock that we may designate and issue in the future.
Preferred Stock
The following description of our preferred stock will
apply generally to any future preferred stock that we may offer, but is not complete. We will describe the particular terms of any class
or series of these securities in more detail in the applicable prospectus supplement.
We are authorized to issue 10,000,000 shares of preferred
stock, par value $0.0001 per share, of which an aggregate of 8,212,500 shares have been designated as of December 13, 2023 as follows:
112,500 shares are designated Series 1 Preferred Stock, 2,000,000 shares are designated Series A Preferred Stock, 4,100,000 are designated
as Series B-1 Preferred Stock and 2,000,000 are designated Series B-2 Preferred Stock. There are currently no shares of preferred stock
outstanding. Any authorized and undesignated shares of preferred stock may be issued from time to time in one or more series pursuant
to a resolution or resolutions providing for such issue duly adopted by our board of directors and, as of the date of this prospectus,
upon stockholder approval.
As of the date of this prospectus, our board of directors
must seek stockholder approval before issuing any shares of preferred stock in order to determine the designations and the powers, preferences
and rights, and the qualifications, limitations and restrictions thereof, including the dividend rights, conversion or exchange rights,
voting rights, redemption rights and terms, liquidation preferences, sinking fund provisions and the number of shares constituting the
series. Our board of directors can, subject to the terms of the Certificate of Incorporation and, as of the date of this prospectus, only
with stockholder approval, issue preferred stock with voting and other rights that could adversely affect the voting power of the holders
of our common stock and which could have certain anti-takeover effects.
The following summary of terms of preferred stock
that we are offering is not complete. We will file as an exhibit to the registration statement of which this prospectus is a part or will
incorporate by reference from reports that we file with the SEC, the form of any certificate of designation that describes the terms of
the series of preferred stock that we are offering before the issuance of the related series of preferred stock. We urge you to read the
applicable prospectus supplement (and any free writing prospectus that we may authorize to be provided to you) related to the series of
preferred stock being offered, as well as the complete certificate of designation that contains the terms of the applicable series of
preferred stock.
Our board of directors will fix the designations,
voting powers, preferences and rights of the preferred stock of each series we issue under this prospectus, as well as the qualifications,
limitations or restrictions thereof, in the certificate of designation relating to that series, all, as of the date of this prospectus,
subject to approval of our stockholders. We will describe in the applicable prospectus supplement the terms of the series of preferred
stock being offered, including, to the extent applicable:
| |
● |
the title and stated value; |
| |
● |
the number of shares we are offering; |
| |
● |
the liquidation preference per share; |
| |
● |
the purchase price; |
| |
● |
the dividend rate, period and payment date and method of calculation for dividends; |
| |
● |
whether dividends will be cumulative or non-cumulative and, if cumulative, the date from which dividends will accumulate; |
| |
● |
the procedures for any auction and remarketing; |
| |
● |
the provisions for a sinking fund; |
| |
● |
the provisions for redemption or repurchase, if applicable, and any restrictions on our ability to exercise those redemption and repurchase rights; |
| |
● |
any listing of the preferred stock on any securities exchange or market; |
| |
● |
whether the preferred stock will be convertible into our common stock, and, if applicable, the conversion price, or how it will be calculated, and the conversion period; |
| |
● |
whether the preferred stock will be exchangeable into debt securities, and, if applicable, the exchange price, or how it will be calculated, and the exchange period; |
| |
● |
voting rights of the preferred stock; |
| |
● |
preemptive rights; |
| |
● |
restrictions on transfer, sale or other assignment; |
| |
● |
whether interests in the preferred stock will be represented by depositary shares; |
| |
● |
a discussion of material United States federal income tax considerations applicable to the preferred stock; |
| |
● |
the relative ranking and preferences of the preferred stock as to dividend rights and rights if we liquidate, dissolve or wind up our affairs; |
| |
● |
any limitations on the issuance of any class or series of preferred stock ranking senior to or on a parity with the series of preferred stock as to dividend rights and rights if we liquidate, dissolve or wind up our affairs; and |
| |
● |
any other specific terms, preferences, rights or limitations of, or restrictions on, the preferred stock. |
Stockholder Rights Plan
On March 11, 2018, we
entered into a Rights Agreement and Continental Stock Transfer & Trust Company, as rights agent
(the “Rights Agreement”) and our board of directors declared a dividend of one Right
for each outstanding share of our common stock, which was amended by Amendment No. 1 thereto dated March 8, 2019, Amendment No. 2 thereto
dated March 10, 2020, Amendment No. 3 thereto dated March 8, 2021, Amendment No. 4 thereto dated March 11, 2022 and Amendment No. 5 thereto
dated March 11, 2023 to, among other things, extend the expiration date of the stockholder’s rights plan to March 11, 2024 and Amendment
No. 6 thereto dated December 11, 2023 which exempted Jeffrey Wolf, the Company’s Chairman, Chief Executive Officer and President,
and Elusys Holdings Inc., a company controlled by Mr. Wolf (the “Buyer”) from
becoming an Acquiring Person ( as such term is defined in the Rights Agreement, as amended) by virtue or as a result of the entry into
the execution and delivery of the Asset and Equity Interests Purchase Agreement (the “Agreement”) Buyer, the execution and
delivery to the Buyer of a convertible promissory note in the aggregate amount of $2,250,000 (the “Note”), the public announcement
of the Agreement, the Note, or any transactions contemplated thereby, the issuance of common stock in connection with the Note, the acquisition
of or right to acquire shares of common stock issuable upon conversion of the Note, and the performance of any other transactions contemplated
by the Agreement or the Note. The dividend was initially authorized and declared on March 23, 2018 (the “Record Date”)
to the stockholders of record at the close of business on that date. Each Right initially entitles the registered holder to purchase
from us one share of common stock at a price set forth therein (the “Purchase Price”), subject to adjustment. The description
and terms of the Rights are set forth in a Rights Agreement, dated as of March 11, 2018, as amended by Amendment No. 1 thereto dated March
8, 2019, Amendment No. 2 thereto dated March 10, 2020, Amendment No. 3 thereto dated March 8, 2021, Amendment No. 4 thereto dated March
11, 2022, Amendment No. 5 thereto dated March 11, 2023 and Amendment No.6 thereto dated December 11, 2023 as the same may be further amended
from time to time (the “Rights Agreement”), between the Company and Continental Stock Transfer & Trust Company, as
Rights Agent (the “Rights Agent”).
The Rights are designed to assure that all
of our stockholders receive fair and equal treatment in the event of a hostile takeover of the Company, to guard against two-tier or partial
tender offers, open market accumulations and other tactics designed to gain control of the Company without paying all stockholders a fair
price, and to enhance the board of director’s ability to negotiate with any prospective acquiror. Until the earlier to occur of
(i) 10 business days following a public announcement that a person or group of affiliated or associated persons has become an Acquiring
Person (as defined below) or (ii) 10 business days (or such later date as may be determined by action of the board of directors prior
to such time as any person or group of affiliated or associated persons becomes an Acquiring Person) following the commencement of, or
public announcement of an intention to make, a tender or exchange offer the consummation of which would result in any person or group
of affiliated or associated persons becoming an Acquiring Person (the earlier of such dates being called the “Distribution Date”),
the Rights will be evidenced, with respect to certificates representing common stock (or book entry shares of common stock) outstanding
as of the Record Date, by such certificates (or such book entry shares) together with a copy of a summary of the Rights (the “Summary
of Rights”). Except in certain situations, a person or group of affiliated or associated persons becomes an “Acquiring
Person” upon acquiring beneficial ownership of 20% or more of the outstanding shares of common stock. Certain synthetic interests
in securities created by derivative positions – whether or not such interests are considered to be ownership of the underlying common
stock or are reportable for purposes of Regulation 13D of the Exchange Act – are treated as beneficial ownership of the number of
shares of the common stock equivalent to the economic exposure created by the derivative security, to the extent actual shares of common
stock are directly or indirectly beneficially owned by a counterparty to such derivative security.
The Rights Agreement provides that, until
the Distribution Date (or earlier expiration of the Rights), the Rights will be transferred with and only with the common stock. Until
the Distribution Date (or earlier expiration of the Rights), new common stock certificates issued after the Record Date upon transfer
or new issuances of common stock will contain a notation incorporating the Rights Agreement by reference. Until the Distribution Date
(or earlier expiration of the Rights), the surrender for transfer of any certificates for shares of common stock (or book entry shares
of common stock) outstanding as of the Record Date, even without such notation or a copy of the Summary of Rights, will also constitute
the transfer of the Rights associated with the shares of common stock represented thereby. As soon as practicable following the Distribution
Date, separate certificates evidencing the Rights (“Right Certificates”) will be mailed to holders of record of the common
stock as of the close of business on the Distribution Date and such separate Right Certificates alone will evidence the Rights. The Rights
are not exercisable until the Distribution Date. The Rights will expire at the close of business on March 11, 2024, unless the Rights
are earlier redeemed or exchanged by the Company as described below.
The Purchase Price payable, and the number
of shares of common stock (or cash, other assets, debt securities of the Company, or any combination thereof equivalent in value thereto)
issuable, upon exercise of the Rights is subject to adjustment from time to time to prevent dilution (i) in the event of a stock dividend
on, or a subdivision, combination or reclassification of, the common stock, (ii) upon the grant to holders of the common stock of certain
rights or warrants to subscribe for or purchase common stock at a price, or securities convertible into common stock with a conversion
price, less than the then-current market price of the common stock or (iii) upon the distribution to holders of the common stock of evidences
of indebtedness or assets (excluding regular periodic cash dividends or dividends payable in common stock) or of subscription rights or
warrants (other than those referred to above).
The number of outstanding Rights is subject
to adjustment in the event of a stock dividend on the common stock payable in shares of common stock or subdivisions, consolidations or
combinations of the common stock occurring, in any such case, prior to the Distribution Date.
In the event that any person or group of affiliated
or associated persons becomes an Acquiring Person, each holder of a Right, other than Rights beneficially owned by the Acquiring Person
(which will thereupon become void), will thereafter have the right to receive upon exercise of a Right that number of shares of common
stock (or cash, property debt securities of the Company, or any combination thereof) having a market value of two times the exercise price
of the Right.
In the event that, after a person or group has become
an Acquiring Person, the Company is acquired in a merger or other business combination transaction or 50% or more of its consolidated
assets or earning power are sold, proper provisions will be made so that each holder of a Right (other than Rights beneficially owned
by an Acquiring Person which will have become void) will thereafter have the right to receive upon the exercise of a Right that number
of shares of common stock of the person with whom the Company has engaged in the foregoing transaction (or its parent) that at the time
of such transaction have a market value of two times the exercise price of the Right.
At any time after any person or group becomes an Acquiring
Person and prior to the earlier of one of the events described in the previous paragraph or the acquisition by such Acquiring Person of
50% or more of the outstanding shares of common stock, the board of directors may exchange the Rights (other than Rights owned by such
Acquiring Person which will have become void), in whole or in part, for shares of common stock (or cash, other assets, debt securities
of the Company, or any combination thereof with an aggregate value equal to such shares) at an exchange ratio of one share of common stock
(or cash, other assets, debt securities of the Company, or any combination thereof equivalent in value thereto) per Right.
With certain exceptions, no adjustment in the Purchase
Price will be required until cumulative adjustments require an adjustment of at least 1% in such Purchase Price. No fractional shares
of common stock will be issued, and in lieu thereof a cash payment will be made based on then current market price of the common stock.
At any time prior to the time an Acquiring Person
becomes such, the Board may redeem the Rights in whole, but not in part, at a price of $0.001 per Right (the “Redemption Price”)
payable, at the option of the Company, in cash, shares of common stock or such other form of consideration as the board of directors shall
determine. The redemption of the Rights may be made effective at such time, on such basis and with such conditions as the board of directors
in its sole discretion may establish. Immediately upon any redemption of the Rights, the right to exercise the Rights will terminate and
the only right of the holders of Rights will be to receive the Redemption Price.
For so long as the Rights are then redeemable, the
Company may, except with respect to the Redemption Price, amend the Rights Agreement in any manner. After the Rights are no longer
redeemable, the Company may, except with respect to the Redemption Price, amend the Rights Agreement in any manner that does not adversely
affect the interests of holders of the Rights.
Until a Right is exercised or exchanged, the holder
thereof, as such, will have no rights as a stockholder of the Company, including, without limitation, the right to vote or to receive
dividends. For more detailed information, please see the Rights Agreement.
Forum Selection
Our Bylaws provide that, unless we consent to the
selection of an alternative forum, the Court of Chancery of the State of Delaware is the exclusive forum for (i) any derivative action
or proceeding brought on behalf of us, (ii) any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers,
other employee or stockholder to us or our stockholders, (iii) any action asserting a claim pursuant to any provision of the Delaware
General Corporation Law , or (iv) any action asserting a claim governed by the internal affairs doctrine, except, in each case for
claims arising under the Securities Act, the Exchange Act, or other federal securities laws for which there is exclusive federal or concurrent
federal and state jurisdiction.
Anti-Takeover Provisions
Delaware Law
We are subject to Section 203 of the DGCL. Subject
to certain exceptions, Section 203 prevents a publicly held Delaware corporation from engaging in a “business combination”
with any “interested stockholder” for three years following the date that the person became an interested stockholder,
unless the interested stockholder attained such status with the approval of our board of directors or unless the business combination
is approved in a prescribed manner. A “business combination” includes, among other things, a merger or consolidation involving
us and the “interested stockholder” and the sale of more than 10% of our assets. In general, an “interested stockholder”
is any entity or person beneficially owning 15% or more of our outstanding voting stock and any entity or person affiliated with or controlling
or controlled by such entity or person.
Our Certificate of Incorporation and Bylaws
Our Certificate of Incorporation and Bylaws contain
provisions that may delay, defer, or discourage another party from acquiring control of us. We expect that these provisions, which are
summarized below, will discourage coercive takeover practices or inadequate takeover bids. These provisions are also designed to encourage
persons seeking to acquire control of us to first negotiate with our board of directors, which we believe may result in an improvement
of the terms of any such acquisition in favor of our stockholders. However, they also give our board of directors the power to discourage
acquisitions that some stockholders may favor.
Proposals of business and nominations. Stockholders
wishing to nominate persons for election to our Board of Directors or to propose any business to be considered by our stockholders at
an annual meeting must comply with certain advance notice and other requirements which are set forth in our Bylaws. Likewise, if our Board
of Directors has determined that directors shall be elected at a special meeting of stockholders, stockholders wishing to nominate persons
for election to our Board of Directors at such special meeting must comply with certain advance notice and other requirements which are
set forth in our Bylaws. These provisions could have the effect of delaying stockholder actions that are favored by the holders
of a majority of our outstanding voting securities until the next stockholder meeting.
Board Vacancies. Our Bylaws generally provide
that only the board of directors (and not the stockholders) may fill vacancies and newly created directorships.
Special Meeting of Stockholders. Our Bylaws
generally provide that only the board of directors (and no other third party) may call a special meeting of stockholders and that the
board of directors may postpone, reschedule or cancel any special meeting of stockholders that was previously scheduled by the board of
directors.
Stockholder Rights Plan. The Rights issued
pursuant to the Rights Agreement described above, if not redeemed or suspended, could work to dilute the stock ownership of a potential
hostile acquirer, likely preventing acquisitions that have not been approved by our board of directors.
Limitations on Liability and Indemnification of
Officers and Directors
Our Certificate of Incorporation and Bylaws provide
that our directors and officers will be indemnified by us to the fullest extent authorized by Delaware General Corporation Law, as it
now exists or may in the future be amended.
These provisions may be held not to be enforceable
for violations of the federal securities laws of the United States.
Listing of Common Stock
Our common stock is currently listed on the NYSE American
under the trading symbol “NHWK.”
Transfer Agent
The transfer agent and registrar for our common stock
is Continental Stock Transfer & Trust Company. They are located at 1 State Street, 30th Floor, New York, New York
10004. Their telephone number is (212) 509-4000.
DESCRIPTION OF WARRANTS
The following description, together with the additional
information we may include in any applicable prospectus supplement and in any related free writing prospectus, summarizes the material
terms and provisions of the warrants that we may offer under this prospectus, which may consist of warrants to purchase common stock,
preferred stock or debt securities and may be issued in one or more series. Warrants may be offered independently or in combination with
common stock, preferred stock or debt securities offered by any prospectus supplement. While the terms we have summarized below will apply
generally to any warrants that we may offer under this prospectus, we will describe the particular terms of any series of warrants in
more detail in the applicable prospectus supplement. The following description of warrants will apply to the warrants offered by this
prospectus unless we provide otherwise in the applicable prospectus supplement. The applicable prospectus supplement for a particular
series of warrants may specify different or additional terms.
We have filed or will file forms of the warrant agreements
and forms of warrant certificates containing the terms of the warrants that may be offered as exhibits to the registration statement of
which this prospectus is a part. We will file as exhibits to the registration statement of which this prospectus is a part, or will incorporate
by reference from reports that we file with the SEC, the form of warrant and/or the warrant agreement and warrant certificate, as applicable,
that contain the terms of the particular series of warrants we are offering, and any supplemental agreements, before the issuance of such
warrants. The following summaries of material terms and provisions of the warrants are subject to, and qualified in their entirety by
reference to, all the provisions of the form of warrant and/or the warrant agreement and warrant certificate, as applicable, and any supplemental
agreements applicable to a particular series of warrants that we may offer under this prospectus. We urge you to read the applicable prospectus
supplement related to the particular series of warrants that we may offer under this prospectus, as well as any related free writing prospectus,
and the complete form of warrant and/or the warrant agreement and warrant certificate, as applicable, and any supplemental agreements,
which contain the terms of the warrants.
General
We will describe in the applicable prospectus supplement
the terms of the series of warrants being offered, including, to the extent applicable:
| |
● |
the offering price and aggregate number of warrants offered; |
| |
● |
the currency for which the warrants may be purchased; |
| |
● |
the designation and terms of the securities with which the warrants are issued and the number of warrants issued with each such security or each principal amount of such security; |
| |
● |
the date on and after which the warrants and the related securities will be separately transferable; |
| |
● |
in the case of warrants to purchase debt securities, the principal amount of debt securities purchasable upon exercise of one warrant and the price at, and currency in which, this principal amount of debt securities may be purchased upon such exercise; |
| |
● |
in the case of warrants to purchase common stock or preferred stock, the number of shares of common stock or preferred stock, as the case may be, purchasable upon the exercise of one warrant and the price at which these shares may be purchased upon such exercise; |
| |
● |
the effect of any merger, consolidation, sale or other disposition of our business on the warrant agreements and the warrants; |
| |
● |
the terms of any rights to redeem or call the warrants; |
| |
● |
any provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the warrants; |
| |
● |
the dates on which the right to exercise the warrants will commence and expire; |
| |
● |
the manner in which the warrant agreements and warrants may be modified; |
| |
● |
a discussion of material United States federal income tax consequences of holding or exercising the warrants; |
| |
● |
the terms of the securities issuable upon exercise of the warrants; and |
| |
● |
any other specific terms, preferences, rights or limitations of or restrictions on the warrants. |
Before exercising their warrants, holders of warrants
will not have any of the rights of holders of the securities purchasable upon such exercise, including:
| |
● |
in the case of warrants to purchase common stock or preferred stock, the right to receive dividends, if any, or, payments upon our liquidation, dissolution or winding up or to exercise voting rights, if any; or |
| |
● |
in the case of warrants to purchase debt securities, the right to receive payments of principal of, or premium, if any, or interest on, the debt securities purchasable upon exercise or to enforce covenants in the applicable indenture. |
Exercise of Warrants
Each warrant will entitle the holder to purchase the
securities that we specify in the applicable prospectus supplement at the exercise price that we describe in the applicable prospectus
supplement. Unless we otherwise specify in the applicable prospectus supplement, holders of the warrants may exercise the warrants at
any time up to the specified time on the expiration date that we set forth in the applicable prospectus supplement. After the close of
business on the expiration date, unexercised warrants will become void.
Unless we otherwise specify in the applicable prospectus
supplement, holders of the warrants may exercise the warrants by delivering the warrant or warrant certificate representing the warrants
to be exercised together with specified information, and paying the required amount to the warrant agent, if applicable, in immediately
available funds, as provided in the applicable prospectus supplement. We will set forth on the reverse side of any warrant certificate
and in the applicable prospectus supplement the information that the holder of the warrant will be required to deliver to any warrant
agent in connection with the exercise of the warrant.
Upon receipt of payment and the warrant or warrant
certificate, as applicable, properly completed and duly executed at the corporate trust office of the warrant agent, if any, or any other
office, including ours, indicated in the prospectus supplement, we will, as soon as practicable, issue and deliver the securities purchasable
upon such exercise. If fewer than all of the warrants (or the warrants represented by such warrant certificate) are exercised, a new warrant
or a new warrant certificate, as applicable, will be issued for the remaining warrants.
Governing Law
Unless we provide otherwise in the applicable prospectus
supplement, the warrants and warrant agreements, and any claim, controversy or dispute arising under or related to the warrants or warrant
agreements, will be governed by and construed in accordance with the laws of the State of New York.
Enforceability of Rights by Holders of Warrants
Each warrant agent, if any, will act solely as our
agent under the applicable warrant agreement and will not assume any obligation or relationship of agency or trust with any holder of
any warrant. A single bank or trust company may act as warrant agent for more than one issue of warrants. A warrant agent will have no
duty or responsibility in case of any default by us under the applicable warrant agreement or warrant, including any duty or responsibility
to initiate any proceedings at law or otherwise, or to make any demand upon us. Any holder of a warrant may, without the consent of the
related warrant agent or the holder of any other warrant, enforce by appropriate legal action its right to exercise, and receive the securities
purchasable upon exercise of, its warrants.
DESCRIPTION OF UNITS
The following description, together with the additional
information we may include in any applicable prospectus supplement and related free writing prospectus, summarizes the material terms
and provisions of the units that we may offer under this prospectus. We may issue units consisting of any combination of the other types
of securities offered under this prospectus in one or more series. We will issue each unit so that the holder of the unit is also the
holder of each security included in the unit. As a result, the holder of a unit will have the rights and obligations of a holder of each
included security. The unit agreement under which a unit is issued may provide that the securities included in the unit may not be held
or transferred separately, at any time or at any time before a specified date. We may evidence each series of units by unit certificates
that we will issue under a separate agreement. We may enter into unit agreements with a unit agent. Each unit agent will be a bank or
trust company that we select. We will indicate the name and address of any unit agent in the applicable prospectus supplement relating
to a particular series of units. The summary below and that contained in any prospectus supplement is qualified in its entirety by reference
to all of the provisions of the unit agreement and/or unit certificate, and depositary arrangements, if applicable. We urge you to read
the applicable prospectus supplements and any related free writing prospectuses related to the units that we may offer under this prospectus,
as well as the complete unit agreement and/or unit certificate, and depositary arrangements, as applicable, that contain the terms of
the units.
We will file as exhibits to the registration statement
of which this prospectus is a part, or will incorporate by reference from reports that we file with the SEC, the form of unit agreement
and/or unit certificate, and depositary arrangements, as applicable, that contain the terms of the particular series of units we are offering,
and any supplemental agreements, before the issuance of such units.
We will describe in the applicable prospectus supplement
the terms of the series of units being offered, including:
| |
● |
the designation and terms of the units and of the securities comprising the units, including whether and under what circumstances those securities may be held or transferred separately; |
| |
|
|
| |
● |
any provisions for the issuance, payment, settlement, transfer, or exchange of the units or of the securities composing the units; |
| |
|
|
| |
● |
whether the units will be issued in fully registered or global form; and |
| |
|
|
| |
● |
any other terms of the units. |
LEGAL OWNERSHIP OF SECURITIES
We can issue securities in registered form or in the
form of one or more global securities. We describe global securities in greater detail below. We refer to those persons who have securities
registered in their own names on the books that we or any applicable trustee, depositary or warrant agent maintain for this purpose as
the “holders” of those securities. These persons are the legal holders of the securities. We refer to those persons who, indirectly
through others, own beneficial interests in securities that are not registered in their own names, as “indirect holders” of
those securities. As we discuss below, indirect holders are not legal holders, and investors in securities issued in book-entry form or
in street name will be indirect holders.
Book-Entry Holders
We may issue securities in book-entry form only, as
we will specify in the applicable prospectus supplement. This means securities may be represented by one or more global securities registered
in the name of a financial institution that holds them as depositary on behalf of other financial institutions that participate in the
depositary’s book-entry system. These participating institutions, which are referred to as participants, in turn, hold beneficial
interests in the securities on behalf of themselves or their customers.
Only the person in whose name a security is registered
is recognized as the holder of that security. Global securities will be registered in the name of the depositary or its participants.
Consequently, for global securities, we will recognize only the depositary as the holder of the securities, and we will make all payments
on the securities to the depositary. The depositary passes along the payments it receives to its participants, which in turn pass the
payments along to their customers who are the beneficial owners. The depositary and its participants do so under agreements they have
made with one another or with their customers; they are not obligated to do so under the terms of the securities.
As a result, investors in a global security will not
own securities directly. Instead, they will own beneficial interests in a global security, through a bank, broker or other financial institution
that participates in the depositary’s book-entry system or holds an interest through a participant. As long as the securities are
issued in global form, investors will be indirect holders, and not legal holders, of the securities.
Street Name Holders
We may terminate a global security or issue securities
that are not issued in global form. In these cases, investors may choose to hold their securities in their own names or in “street
name.” Securities held by an investor in street name would be registered in the name of a bank, broker or other financial institution
that the investor chooses, and the investor would hold only a beneficial interest in those securities through an account he or she maintains
at that institution.
For securities held in street name, we or any applicable
trustee or depositary will recognize only the intermediary banks, brokers and other financial institutions in whose names the securities
are registered as the holders of those securities, and we or any such trustee or depositary will make all payments on those securities
to them. These institutions pass along the payments they receive to their customers who are the beneficial owners, but only because they
agree to do so in their customer agreements or because they are legally required to do so. Investors who hold securities in street name
will be indirect holders, not holders, of those securities.
Legal Holders
Our obligations, as well as the obligations of any
applicable trustee or third party employed by us or a trustee, run only to the legal holders of the securities. We do not have obligations
to investors who hold beneficial interests in global securities, in street name or by any other indirect means. This will be the case
whether an investor chooses to be an indirect holder of a security or has no choice because we are issuing the securities only in global
form.
For example, once we make a payment or give a notice
to the holder, we have no further responsibility for the payment or notice even if that holder is required, under agreements with its
participants or customers or by law, to pass it along to the indirect holders but does not do so. Similarly, we may want to obtain the
approval of the holders to amend an indenture, to relieve us of the consequences of a default or of our obligation to comply with a particular
provision of an indenture, or for other purposes. In such an event, we would seek approval only from the legal holders, and not the indirect
holders, of the securities. Whether and how the legal holders contact the indirect holders is up to the legal holders.
Special Considerations for Indirect Holders
If you hold securities through a bank, broker or other
financial institution, either in book-entry form because the securities are represented by one or more global securities or in street
name, you should check with your own institution to find out:
| |
● |
how it handles securities payments and notices; |
| |
● |
whether it imposes fees or charges; |
| |
● |
how it would handle a request for the holders’ consent, if ever required; |
| |
● |
whether and how you can instruct it to send
you securities registered in your own name so you can be a holder, if that is
permitted in the future; |
| |
● |
how it would exercise rights under the securities
if there were a default or other event triggering the need for holders to act
to protect their interests; and |
| |
● |
if the securities are in book-entry form, how the depositary’s rules and procedures will affect these matters. |
Global Securities
A global security is a security that represents one
or any other number of individual securities held by a depositary. Generally, all securities represented by the same global securities
will have the same terms.
Each security issued in book-entry form will be represented
by a global security that we issue to, deposit with and register in the name of a financial institution or its nominee that we select.
The financial institution that we select for this purpose is called the depositary. Unless we specify otherwise in the applicable prospectus
supplement, The Depository Trust Company, New York, New York, known as DTC, will be the depositary for all securities issued in book-entry
form.
A global security may not be transferred to or registered
in the name of anyone other than the depositary, its nominee or a successor depositary, unless special termination situations arise. We
describe those situations below under “—Special Situations When a Global Security Will Be Terminated.” As a result of
these arrangements, the depositary, or its nominee, will be the sole registered owner and legal holder of all securities represented by
a global security, and investors will be permitted to own only beneficial interests in a global security. Beneficial interests must be
held by means of an account with a broker, bank or other financial institution that in turn has an account with the depositary or with
another institution that does. Thus, an investor whose security is represented by a global security will not be a legal holder of the
security, but only an indirect holder of a beneficial interest in the global security.
If the prospectus supplement for a particular security
indicates that the security will be issued in global form only, then the security will be represented by a global security at all times
unless and until the global security is terminated. If termination occurs, we may issue the securities through another book-entry clearing
system or decide that the securities may no longer be held through any book-entry clearing system.
Special Considerations for Global Securities
As an indirect holder, an investor’s rights
relating to a global security will be governed by the account rules of the investor’s financial institution and of the depositary,
as well as general laws relating to securities transfers. We do not recognize an indirect holder as a holder of securities and instead
deal only with the depositary that holds the global security.
If securities are issued only as global securities,
an investor should be aware of the following:
| |
● |
an investor cannot cause the securities to be registered in his or her name, and cannot obtain non-global certificates for his or her interest in the securities, except in the special situations we describe below; |
| |
● |
an investor will be an indirect holder and must look to his or her own bank or broker for payments on the securities and protection of his or her legal rights relating to the securities, as we describe above; |
| |
● |
an investor may not be able to sell interests in the securities to some insurance companies and to other institutions that are required by law to own their securities in non-book-entry form; |
| |
● |
an investor may not be able to pledge his or her interest in the global security in circumstances where certificates representing the securities must be delivered to the lender or other beneficiary of the pledge in order for the pledge to be effective; |
| |
● |
the depositary’s policies, which may change from time to time, will govern payments, transfers, exchanges and other matters relating to an investor’s interest in the global security; |
| |
● |
we and any applicable trustee have no responsibility for any aspect of the depositary’s actions or for its records of ownership interests in the global security, nor will we or any applicable trustee supervise the depositary in any way; |
| |
● |
the depositary may, and we understand that DTC will, require that those who purchase and sell interests in the global security within its book-entry system use immediately available funds, and your broker or bank may require you to do so as well; and |
| |
● |
financial institutions that participate in the depositary’s book-entry system, and through which an investor holds its interest in the global security, may also have their own policies affecting payments, notices and other matters relating to the securities. |
There may be more than one financial intermediary
in the chain of ownership for an investor. We do not monitor and are not responsible for the actions of any of those intermediaries.
Special Situations When a Global Security Will
Be Terminated
In a few special situations described below, a global
security will terminate and interests in it will be exchanged for physical certificates representing those interests. After that exchange,
the choice of whether to hold securities directly or in street name will be up to the investor. Investors must consult their own banks
or brokers to find out how to have their interests in securities transferred to their own names, so that they will be direct holders.
We have described the rights of holders and street name investors above.
Unless we provide otherwise in the applicable prospectus
supplement, a global security will terminate when the following special situations occur:
| |
● |
if the depositary notifies us that it is
unwilling, unable or no longer qualified to continue as depositary for
that global security and we do not appoint
another institution to act as depositary within 90 days; |
| |
● |
if we notify any applicable trustee that we wish to terminate that global security; or |
| |
● |
if an event of default has occurred with
regard to securities represented by that global security and has not
been cured or waived. |
The applicable prospectus supplement may also list
additional situations for terminating a global security that would apply only to the particular series of securities covered by the applicable
prospectus supplement. When a global security terminates, the depositary, and neither we nor any applicable trustee, is responsible for
deciding the names of the institutions that will be the initial direct holders.
PLAN OF DISTRIBUTION
We may sell the securities from time to time pursuant
to underwritten public offerings, best efforts public offerings, direct sales to the public, “at the market” offerings, negotiated
transactions, block trades or a combination of these methods. We may sell the securities to or through one or more underwriters or dealers
(acting as principal or agent), through agents, or directly to one or more purchasers. We may distribute securities from time to time
in one or more transactions:
| |
● |
at a fixed price or prices, which may be changed; |
| |
● |
at market prices prevailing at the time of sale; |
| |
● |
at prices related to such prevailing market prices; or |
| |
● |
at negotiated prices. |
A prospectus supplement or supplements (and any related
free writing prospectus that we may authorize to be provided to you) will describe the terms of the offering of the securities, including,
to the extent applicable:
| |
● |
the name or names of the underwriters, dealers, agents or other purchasers, if any; |
| |
● |
the purchase price of the securities or other consideration therefor, and the proceeds we will receive from the sale; |
| |
● |
any option to purchase additional shares or other options under which underwriters, dealers, agents or other purchasers may purchase additional securities from us; |
| |
● |
any agency fees or underwriting discounts to be allowed or paid to the agent or underwriters and other items constituting agents’ or underwriters’ compensation; |
| |
● |
any public offering price; |
| |
● |
any discounts or concessions allowed or reallowed or paid to dealers; and |
| |
● |
any securities exchange or market on which the securities may be listed. |
Only underwriters named in the prospectus supplement
will be underwriters of the securities offered by the prospectus supplement. Dealers and agents participating in the distribution of the
securities may be deemed to be underwriters, and compensation received by them on resale of the securities may be deemed to be underwriting
discounts. If such dealers or agents were deemed to be underwriters, they may be subject to statutory liabilities under the Securities
Act.
If underwriters are used in the sale, they will acquire
the securities for their own account and may resell the securities from time to time in one or more transactions at a fixed public offering
price or at varying prices determined at the time of sale. The obligations of the underwriters to purchase the securities will be subject
to the conditions set forth in the applicable underwriting agreement. We may offer the securities to the public through underwriting syndicates
represented by managing underwriters or by underwriters without a syndicate. Subject to certain conditions, the underwriters will be obligated
to purchase all of the securities offered by the prospectus supplement other than securities covered by any option to purchase additional
shares or other option. If a dealer is used in the sale of securities, we, or an underwriter, will sell the securities to the dealer,
as principal. The dealer may then resell the securities to the public at varying prices to be determined by the dealer at the time of
resale. To the extent required, we will set forth in the prospectus supplement the name of the dealer and the terms of the transaction.
Any public offering price and any discounts or concessions allowed or reallowed or paid to dealers may change from time to time. We may
use underwriters, dealers or agents with whom we have a material relationship. We will describe in the prospectus supplement, naming the
underwriter, dealer or agent, the nature of any such relationship.
We may sell securities directly or through agents
we designate from time to time. We will name any agent involved in the offering and sale of securities, and we will describe any commissions
we will pay the agent in the prospectus supplement. Unless the prospectus supplement states otherwise, the agent will act on a best-efforts
basis for the period of its appointment.
We may authorize agents or underwriters to solicit
offers by certain types of institutional investors to purchase securities from us at the public offering price set forth in the prospectus
supplement pursuant to delayed delivery contracts providing for payment and delivery on a specified date in the future. We will describe
the conditions to these contracts and the commissions we must pay for solicitation of these contracts in the prospectus supplement.
We may provide agents, dealers and underwriters with
indemnification against civil liabilities, including liabilities under the Securities Act, or contribution with respect to payments that
the agents, dealers or underwriters may make with respect to these liabilities. Agents, dealers and underwriters or their affiliates may
engage in transactions with, or perform services for, us in the ordinary course of business.
All securities we may offer, other than common stock,
will be new issues of securities with no established trading market. Any underwriters may make a market in these securities, but will
not be obligated to do so and may discontinue any market making at any time without notice. We cannot guarantee the liquidity of the trading
markets for any securities.
Any underwriter may engage in overallotment, stabilizing
transactions, short covering transactions and penalty bids. Overallotment involves sales in excess of the offering size, which create
a short position. Stabilizing transactions permit bids to purchase the underlying security so long as the stabilizing bids do not exceed
a specified maximum price. Syndicate covering or other short-covering transactions involve purchases of the securities, either through
exercise of the option to purchase additional shares or in the open market after the distribution is completed, to cover short positions.
Short covering transactions involve purchases of the securities in the open market after the distribution is completed to cover short
positions. Penalty bids permit the underwriters to reclaim a selling concession from a dealer when the securities originally sold by the
dealer are purchased in a stabilizing or covering transaction to cover short positions. Those activities may cause the price of the securities
to be higher than it would otherwise be. If commenced, the underwriters may discontinue any of the activities at any time. These transactions
may be effected on any exchange or over-the-counter market or otherwise.
Any underwriters, dealers or agents who are qualified
market makers may engage on the Nasdaq Capital Market in passive market making transactions in our common stock on the Nasdaq Capital
Market in accordance with Rule 103 of Regulation M under the Exchange Act, during the business day prior to the pricing of the offering,
before the commencement of offers or sales of the securities. Passive market makers must comply with applicable volume and price limitations
and must be identified as passive market makers. In general, a passive market maker must display its bid at a price not in excess of the
highest independent bid for such security; if all independent bids are lowered below the passive market maker’s bid, however, the
passive market maker’s bid must then be lowered when certain purchase limits are exceeded. Passive market making may stabilize the
market price of the securities at a level above that which might otherwise prevail in the open market and, if commenced, may be discontinued
at any time. Agents and underwriters may engage in transactions with, or perform services for, us and our affiliates in the ordinary
course of business.
The specific terms of any lock-up provisions in respect
of any given offering will be described in the applicable prospectus supplement.
The anticipated date of delivery of offered securities
will be set forth in the applicable prospectus supplement relating to each offer.
LEGAL MATTERS
Unless otherwise indicated in the applicable prospectus
supplement, the validity of the securities offered by this prospectus, and any supplement thereto, will be passed upon for us by Blank
Rome LLP, New York, New York. Additional legal matters may be passed upon for us or any underwriters, dealers or agents, by counsel that
we will name in the applicable prospectus supplement.
As of the date of this prospectus, an attorney of
Blank Rome LLP beneficially owns common stock and securities exercisable to purchase shares of our common stock that represent less than
1% of our outstanding shares of common stock.
EXPERTS
The consolidated financial statements as of December
31, 2022 and 2021 and for the years then ended, incorporated by reference in this prospectus and the registration statement have been
so incorporated in reliance on the report of BDO USA, LLP (n/k/a BDO USA, P.C.), an independent registered public accounting firm, incorporated
herein by reference, given on the authority of said firm as experts in accounting and auditing. The report on the consolidated financial
statements contains an explanatory paragraph regarding the Company’s ability to continue as a going concern.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus is part of a registration statement
we filed with the SEC. This prospectus does not contain all of the information set forth in the registration statement and the exhibits
to the registration statement. For further information with respect to us and the securities we are offering under this prospectus, we
refer you to the registration statement and the exhibits and schedules filed as a part of the registration statement. Neither we nor any
agent, underwriter or dealer has authorized any person to provide you with different information. We are not making an offer of these
securities in any state where the offer is not permitted. You should not assume that the information in this prospectus is accurate as
of any date other than the date on the front page of this prospectus, regardless of the time of delivery of this prospectus or any sale
of the securities offered by this prospectus.
We file annual, quarterly and current reports, proxy
statements and other information with the SEC. Our SEC filings are available to the public at the SEC’s website at www.sec.gov.
Our SEC filings are also available on our website, www.nighthawkbio.com under the heading “Investors & News—SEC
Filings.” The reference to our website is an inactive textual reference only, the information contained in, and that can be accessed
through our website, is not incorporated into and is not a part of this prospectus. We make available on our website our SEC filings as
soon as reasonably practicable after those reports are filed with the SEC.
INCORPORATION OF CERTAIN INFORMATION BY
REFERENCE
The SEC allows us to “incorporate by reference”
information into this prospectus, which means that we can disclose important information to you by referring you to another document filed
separately with the SEC. The SEC file number for the documents incorporated by reference in this prospectus is 001-35994. The documents
incorporated by reference into this prospectus contain important information that you should read about us.
The following documents are incorporated by reference
into this prospectus:
| |
• |
Our Annual Report on Form
10-K for the fiscal year ended December 31, 2022 filed with the SEC on March 31, 2023; |
| |
• |
Our Quarterly Report on Form 10-Q for the fiscal quarter ended March 31,
2023 filed with the SEC on May 15, 2023, our Quarterly Report on Form 10-Q for the quarter ended June 30, 2023 filed with the SEC
on August 14, 2023, and our Quarterly Report on Form 10-Q for the quarter ended September 30, 2023 filed with the SEC on November
20, 2023; |
| |
• |
Our Current Report on Form 8-K filed with the SEC on January
5, 2023, February 2, 2023, March
13, 2023, March 23, 2023, August
21, 2023, September 19,
2023, as amended on September
20, 2023, September 28,
2023, December
8, 2023,
December 12, 2023, and December 19, 2023; and |
| |
• |
The description of our common stock and our common stock purchase rights
is set forth in our registration statement on Form
8-A filed with the SEC on February 4, 2022, Form
8-A/A (Amendment No. 1) filed on March 11, 2022 and Form
8-A/A (Amendment No. 2) filed on March 13, 2023, as updated by the description of our common stock filed as Exhibit 4.18
to our Annual Report on Form 10-K for the year ended December 31, 2022 filed with the SEC on March 31, 2023, including any
amendments or reports filed for the purpose of updating such description. |
We also incorporate by reference into this prospectus
all documents (other than Current Reports furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits filed on such form that are
related to such items or other information “furnished” to the SEC, which is not deemed filed and not incorporated in this
prospectus) that are filed by us with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act (i) after the date of
the initial filing of the registration statement of which this prospectus forms a part and prior to effectiveness of the registration
statement, or (ii) after the date of this prospectus and before the completion of the offering of the securities included in this prospectus.
These documents include periodic reports, such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form
8-K, as well as proxy statements.
We will provide to each person, including any beneficial
owner, to whom a prospectus is delivered, without charge upon written or oral request, a copy of any or all of the documents that are
incorporated by reference into this prospectus but not delivered with the prospectus, including exhibits that are specifically incorporated
by reference into such documents. You can request a copy of these filings, at no cost, by writing or telephoning us at the following address
or telephone number:
NightHawk Biosciences, Inc.
627 Davis Drive, Suite 300
Morrisville, North Carolina 27560
Telephone: (919) 240-7133
Attn: Investor Relations
Any statement contained in this prospectus or contained
in a document incorporated or deemed to be incorporated by reference into this prospectus will be deemed to be modified or superseded
to the extent that a statement contained in this prospectus or any subsequently filed supplement to this prospectus, or document deemed
to be incorporated by reference into this prospectus, modifies or supersedes such statement.
The information contained in this prospectus is not complete
and may be changed. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission
is effective. This prospectus is not an offer to sell these securities and is not soliciting an offer to buy these securities in any jurisdiction
where the offer or sale is not permitted.
Subject to Completion, Dated December
19, 2023
PROSPECTUS

Up to $4,037,193 of Shares
of Common Stock
We have entered into a Sales Agreement, dated December
8, 2023 (the “sales agreement”), with A.G.P./Alliance Global Partners (“A.G.P.” or the “sales agent”)
relating to the sale of shares of our common stock, par value $0.0002 per share, having an aggregate offering price of up to $4,037,193
from time to time through or to A.G.P., acting as sales agent or principal.
Sales of shares of our common stock, if any, under
this prospectus will be made in sales deemed to be an “at the market offering” as defined in Rule 415(a)(4) promulgated
under the Securities Act of 1933, as amended (the “Securities Act”). The sales agent is not required to sell any specific
amount, but will act as sales agent on a commercially reasonable efforts basis consistent with its normal trading and sales practices,
on mutually agreed terms between the sales agent and us. There is no arrangement for funds to be received in any escrow, trust or similar
arrangement.
As sales agent, A.G.P. is entitled to compensation
at a fixed commission rate equal to 3.0% of the gross proceeds of each sale of shares of our common stock. In connection with the sale
of our shares of common stock on our behalf, the sales agent will be deemed to be an “underwriter” within the meaning of the
Securities Act and the compensation of the sales agent will be deemed to be underwriting commissions or discounts. We have also agreed
to provide indemnification and contribution to the sales agent with respect to certain liabilities, including liabilities under the Securities
Act.
Our common stock is listed on the NYSE American under
the symbol “NHWK.” On December 18, 2023, the last reported sale price of our common stock on the NYSE American was $0.4457
per share.
As of the date of this prospectus, the aggregate market
value of our outstanding common stock held by non-affiliates is $12,111,581, which is calculated based on 24,742,760 shares of our outstanding
common stock held by non-affiliates and a price of $0.4895 per share, the closing price of our common stock on November 7, 2023, which
is the highest closing sale price of our common stock on the NYSE American within the prior 60 days of this prospectus. During
the prior twelve calendar month period that ends on and includes the date hereof, we have not offered or sold any shares of our common
stock pursuant to General Instruction I.B.6 to Form S-3.
Investing in our common stock involves a high degree
of risk. Before making an investment decision, please read the information under the heading “Risk Factors” beginning on page S-3 of this prospectus supplement and in the documents incorporated by reference into
this prospectus.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
A.G.P.
The date of this prospectus is , 2023
TABLE OF CONTENTS
i
ABOUT THIS PROSPECTUS
This prospectus relates to part of a registration statement on Form
S-3 that we have filed with the Securities and Exchange Commission (the “SEC”) utilizing a “shelf” registration
process. Under this shelf registration process, we may sell any combination of the securities described in our base prospectus included
in the shelf registration statement in one or more offerings up to a total aggregate offering price of $200,000,000. The $4,037,193 of
shares of common stock that may be offered, issued and sold under this prospectus is included in the $200,000,000 of securities that may
be offered, issued and sold by us pursuant to our shelf registration statement. In connection with such offers and when accompanied by
the base prospectus included in the registration statement of which this prospectus is a part, this prospectus will be deemed a prospectus
supplement to such base prospectus
This prospectus relates to the offering of
our shares of common stock. Before buying any of our shares of common stock that we are offering, we urge you to carefully read this prospectus,
together with the information incorporated by reference as described under the headings “Where You Can Find More Information”
and “Incorporation of Certain Information by Reference” in this prospectus. These documents contain important information
that you should consider when making your investment decision.
This prospectus describes the terms of this
offering of our shares of common stock and also adds to and updates information contained in the documents incorporated by reference into
this prospectus. To the extent there is a conflict between the information contained in this prospectus, on the one hand, and the information
contained in any document incorporated by reference into this prospectus that was filed with the SEC before the date of this prospectus,
on the other hand, you should rely on the information in this prospectus. If any statement in one of these documents is inconsistent with
a statement in another document having a later date (for example, a document incorporated by reference into this prospectus) the statement
in the document having the later date modifies or supersedes the earlier statement.
You should rely only on the information contained
in or incorporated by reference in this prospectus and in any free writing prospectus that we have authorized for use in connection with
this offering. We have not, and A.G.P. has not, authorized anyone to provide you with different information. If anyone provides you with
different or inconsistent information, you should not rely on it. We are not, and A.G.P. is not, making an offer to sell these securities
in any jurisdiction where the offer or sale is not permitted. You should assume that the information appearing in this prospectus, the
documents incorporated by reference in this prospectus, and in any free writing prospectus that we have authorized for use in connection
with this offering, is accurate only as of the date of those respective documents. Our business, financial condition, results of operations
and prospects may have changed since those dates. You should carefully read this prospectus, the documents incorporated by reference in
this prospectus, and any free writing prospectus that we have authorized for use in connection with this offering, in their entirety before
making an investment decision.
We further note that the representations, warranties
and covenants made by us in any agreement that is filed as an exhibit to any document that is incorporated by reference in this prospectus
were made solely for the benefit of the parties to such agreement, including, in some cases, for the purpose of allocating risk among
the parties to such agreements, and should not be deemed to be a representation, warranty or covenant to you. Moreover, such representations,
warranties or covenants were accurate only as of the date when made. Accordingly, such representations, warranties and covenants should
not be relied on as accurately representing the current state of our affairs.
This prospectus contains and incorporates
by reference market data and industry statistics and forecasts that are based on independent industry publications and other publicly
available information. Although we believe that these sources are reliable, we do not guarantee the accuracy or completeness of this information
and we have not independently verified this information. Although we are not aware of any misstatements regarding the market and industry
data presented in this prospectus and the documents incorporated herein by reference, these estimates involve risks and uncertainties
and are subject to change based on various factors, including those discussed under the heading “Risk Factors” in this prospectus
and under similar headings in the other documents that are incorporated by reference into this prospectus. Accordingly, investors should
not place undue reliance on this information.
Except as otherwise indicated herein or as
the context otherwise requires, references in this prospectus to “NightHawk,” the “Company,” “we,”
“us,” “our” and similar references refer to NightHawk Biosciences,
Inc., an entity incorporated under the laws of the State of Delaware, and where appropriate our consolidated subsidiaries.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus, including the documents that we incorporate
by reference herein, may contain, forward-looking statements within the meaning of Section 27A of the Securities Act and Section 21E
of the Securities Exchange Act of 1934, as amended (“the Exchange Act”), including statements regarding our future financial
condition, business strategy and plans and objectives of management for future operations. Forward-looking statements include all statements
that are not historical facts. In some cases, you can identify forward-looking statements by terminology such as “believe,”
“will,” “may,” “estimate,” “continue,” “anticipate,” “intend,”
“should,” “plan,” “might,” “approximately,” “expect,” “predict,”
“could,” “potentially” or the negative of these terms or other similar expressions. Forward-looking statements
include statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations.
Discussions containing these forward-looking statements
may be found, among other places, in the sections entitled “Business,” “Risk Factors” and “Management’s
Discussion and Analysis of Financial Condition and Results of Operations” contained in the documents incorporated by reference in
this prospectus, including our most recent Annual Report on Form 10-K and our Quarterly Reports on Form 10-Q, as well as any
amendments thereto.
These statements relate to future events or our future
financial performance and involve known and unknown risks, uncertainties and other factors that could cause our actual results, levels
of activity, performance or achievement to differ materially from those expressed or implied by these forward-looking statements. We discuss
in greater detail, and incorporate by reference into this prospectus in their entirety, many of these risks and uncertainties under the
section entitled “Risk Factors” included in our most recent Annual Report on Form 10-K and our Quarterly Reports on Form 10-Q,
as well as any amendments thereto, filed with the SEC, which are incorporated by reference into this prospectus. Additional factors are
discussed under the section entitled “Risk Factors” in this prospectus and any free writing prospectus and under similar headings
in the other documents that are incorporated by reference into this prospectus. These statements reflect our current views with respect
to future events and are based on assumptions and subject to risks and uncertainties. New risks and uncertainties arise from time to time,
and it is impossible for us to predict these events or how they may affect us. We undertake no obligation to revise or publicly release
the results of any revision to these forward-looking statements, except as required by law. Given these risks and uncertainties, readers
are cautioned not to place undue reliance on such forward-looking statements. All forward-looking statements are qualified in their entirety
by this cautionary statement.
| |
|
|
| |
|
|
| |
|
|
| |
PROSPECTUS SUMMARY
The items in the following summary are described
in more detail elsewhere in this prospectus and in the documents incorporated by reference herein. This summary highlights selected information
contained elsewhere in this prospectus. This summary is not intended to be complete and does not contain all of the information that you
should consider before deciding to invest in our securities. You should read this entire prospectus carefully, especially the “Risk
Factors” section beginning on page S-3 and other documents or information included or incorporated by reference in this prospectus
before making an investment decision.
Overview
Our current focus is on our contract development
and manufacturing organization (“CDMO”) that provides a comprehensive range of services from process development to Current
Good Manufacturing Practices (“CGMP”) clinical and commercial manufacturing of biologics for the biotechnology and biopharmaceutical
industries through our Scorpius Biomanufacturing, Inc. (“Scorpius”) subsidiary. Scorpius pairs cGMP biomanufacturing and quality
control expertise with cutting edge capabilities in immunoassays, molecular assays, and bioanalytical methods to support cell- and gene-based
therapies as well as large molecule biologics. Our services include clinical and commercial drug substance manufacturing, release and
stability testing and variety of process development services, including upstream and downstream development and optimization, analytical
method development, cell line development, testing and characterization. Our lead facility in San Antonio, Texas commenced operations
in October 2022.
Corporate Information
We are a Delaware corporation. Our principal executive
offices are located at 627 Davis Drive, Suite 300, Morrisville, North Carolina 27560 and our telephone number at this address is (919)
240-7133. Our website is www.nighthawkbio.com. Information contained on, or that may be accessible through, our website is not
a part of, and is not incorporated into, this prospectus.
Smaller Reporting Company
We are a “smaller reporting company” as
defined in the Exchange Act. As a result, we may take advantage of certain reduced disclosure obligations available to smaller reporting
companies, including the exemption from compliance with the auditor attestation requirements pursuant to the Sarbanes-Oxley Act of 2022,
reduced disclosure about our executive compensation arrangements and the requirements to provide only two years of audited financial statements
in our annual reports and registration statements. We will continue to be a “smaller reporting company” as long as (1) we
have a public float (i.e., the market value of our common stock held by non-affiliates) less than $250 million calculated as of the last
business day of our most recently completed second fiscal quarter, or (2) our annual revenues are less than $100 million for our previous
fiscal year and we have either no public float or a public float of less than $700 million as of the end of that fiscal year’s second
fiscal quarter. Decreased disclosures in our SEC filings due to our status as a “smaller reporting company” may make it harder
for investors to analyze our results of operations and financial prospects.
|
|
| |
|
|
| |
|
|
|
|
| |
|
|
|
| |
|
|
| |
THE
OFFERING |
|
| |
|
|
|
| |
Issuer |
NightHawk Biosciences, Inc., |
|
| |
|
|
|
| |
Common stock to be offered by us
pursuant to this prospectus |
12,522,310 shares of our common stock having an aggregate offering
price of up to $4,037,193 at an assumed offering price of $0.3224 per share, which was the approximate last reported sale price of our
common stock on the NYSE American on December 13, 2023. |
|
| |
|
|
|
| |
Common stock to be outstanding
after the offering |
Up to 38,604,200 (as more fully described in the notes following
this table), assuming sales of $4,037,193 shares of our common stock, or 12,522,310 shares of our common stock in this offering at
an assumed offering price of $0.3224 per share, which was the approximate last reported sale price of our common stock on the NYSE
American on December 13, 2023. The actual number of shares issued will vary depending on the sales price under this offering.(1) |
|
| |
|
|
|
| |
Manner of offering |
“At the market offering” that may be made from time to time for our common
stock in the United States through or to A.G.P., as sales agent or principal. See the section entitled
“Plan of Distribution.” |
|
| |
|
|
|
| |
Use of proceeds |
We currently intend to use the net proceeds from this offering
for working capital and general corporate purposes, including, including, to acquire, license or invest in assets, businesses, technologies,
product candidates or other intellectual property and to repurchase securities. See “Use of Proceeds.” |
|
| |
|
|
|
| |
Risk factors |
You should
read the “Risk Factors” section of this prospectus and in the documents incorporated by reference in this prospectus
for a discussion of factors to consider before deciding to purchase shares of our common stock. |
|
| |
|
|
|
| |
NYSE American Trading Symbol |
NHWK |
|
| |
Unless we indicate otherwise, all
information in this prospectus is based on 26,081,890 shares outstanding as of December 1, 2023. The number of shares
outstanding as used throughout this prospectus, unless otherwise indicated, excludes:
·
6,719,943 shares of our common stock issuable upon the exercise of outstanding stock options as of December 1, 2023, with a weighted
average exercise price of $3.61 per share;
·
280,000 shares of our common stock issuable upon vesting of restricted stock units as of December 1, 2023, with an average fair
value of $1.18 per share; and
·
535,159 additional shares of our common stock reserved for future issuance under our equity incentive plans as of December 1, 2023. |
|
| |
|
|
RISK FACTORS
Investing in our common stock involves
a high degree of risk, and you should be able to bear the complete loss of your investment. You should consider carefully the risks described
below and those described under the section captioned “Risk Factors” contained in our Annual Report on Form 10-K for the year
ended December 31, 2022, any subsequent Annual Reports on Form 10-K, any subsequent Quarterly Reports on Form 10-Q or Current Reports
on Form 8-K, and all other information contained or incorporated by reference into this prospectus and documents incorporated by reference
into this prospectus before deciding whether to purchase any of the common stock being offered under this prospectus. If any of the risks
actually occur, our business, consolidated financial condition or results of operations could be adversely affected. In such case, the
trading price of our common stock could decline and you could lose all or part of your investment. Our actual results could differ materially
from those anticipated in the forward-looking statements made throughout this prospectus as a result of different factors, including the
risks we face described below. Unless we have indicated otherwise or the context otherwise requires, references in this prospectus or
the documents incorporated by reference herein and therein to the “Company,” “NightHawk Biosciences,” “we,”
“us” and “our” refer to NightHawk Biosciences, Inc.
Risks Related to this Offering
Our management will have broad discretion over
the use of proceeds from this offering and may not use the proceeds effectively.
Our management will have broad discretion over the
use of proceeds from this offering. We intend to use the net proceeds from this offering, if any, for working capital and general corporate
purposes, including, to acquire, license or invest in assets, businesses, technologies, product candidates or other intellectual property
and to repurchase securities. Our management will have considerable discretion in the application of the net proceeds, and you will not
have the opportunity, as part of your investment decision, to assess whether the proceeds are being used appropriately. The net proceeds,
if any, may be used for corporate purposes that do not improve our operating results or enhance the value of our common stock. The failure
of our management to use these funds effectively could have a material adverse effect on our business and cause the market price of our
common stock to decline. Pending their use, we may invest the net proceeds from this offering in short-term, investment-grade, interest-bearing
instruments and U.S. government securities. These investments may not yield a favorable return to our stockholders.
Our need for future financing may result in
the issuance of additional securities which will cause investors to experience dilution.
Our cash requirements may vary from those now planned
depending upon numerous factors. We expect to require additional capital until our operations generate sufficient revenue to cover our
expenses. Accordingly, we will need to obtain substantial additional funding in connection with our continuing operations. There are no
other commitments by any person for future financing. Our securities may be offered to other investors at a price lower than the price
per share offered to current stockholders, or upon terms which may be deemed more favorable than those offered to current stockholders.
In addition, the issuance of securities in any future financing may dilute an investor’s equity ownership and have the effect of
depressing the market price for our securities. Moreover, we may issue derivative securities, including options and/or warrants, from
time to time, to procure qualified personnel or for other business reasons. The issuance of any such derivative securities, which is at
the discretion of our Board of Directors, may further dilute the equity ownership of our stockholders.
We may sell shares or other securities in any other
offering at a price per share that is less than the price per share paid by investors in this offering, and investors purchasing shares
or other securities in the future could have rights superior to existing stockholders. The price per share at which we sell additional
shares of our common stock, or securities convertible or exchangeable into common stock, in future transactions may be higher or lower
than the price per share paid by investors in this offering. No assurance can be given as to our ability to procure additional financing,
if required, and on terms deemed favorable to us. To the extent additional capital is required and cannot be raised successfully, we may
then have to limit our then current operations and/or may have to curtail certain, if not all, of our business objectives and plans.
We have additional securities available for
issuance, which, if issued, could adversely affect the rights of the holders of our common stock.
Our Third Amended and Restated Certificate of Incorporation,
as amended (the “Certificate of Incorporation”), authorizes the issuance of 250,000,000 shares of our common stock and 10,000,000
shares of preferred stock. In certain circumstances, shares of our common stock, as well as the awards available for issuance under our
equity incentive plans, can be issued by our Board of Directors, without stockholder approval. Any future issuances of such stock would
further dilute the percentage ownership of us held by holders of our common stock and preferred stock. In addition, the issuance of certain
securities, including pursuant to the terms of our stockholder rights plan, may be used as an “anti-takeover” device without
further action on the part of our stockholders, and may adversely affect the holders of the common stock.
Because we do not intend to declare cash dividends
on our shares of common stock in the foreseeable future, stockholders must rely on appreciation of the value of our common stock for any
return on their investment.
As stated above, we have never declared or paid cash
dividends on our common stock. We currently anticipate that we will retain future earnings for the development, operation and expansion
of our business and do not anticipate declaring or paying any cash dividends in the foreseeable future. As a result, we expect that only
appreciation of the price of our common stock, if any, will provide a return to investors in this offering for the foreseeable future.
Resales of our common stock in the public market
during this offering by our stockholders may cause the market price of our common stock to fall.
We may issue shares of our common stock from time
to time in connection with this offering. This issuance from time to time of these new shares of our common stock, or our ability to issue
these shares of our common stock in this offering, could result in resales of our common stock by our current stockholders concerned about
the potential dilution of their holdings. In turn, these resales could have the effect of depressing the market price for our common stock.
The actual number of shares we will issue under
the sales agreement, at any one time or in total, is uncertain.
Subject to certain limitations in the sales agreement
with the sales agent and compliance with applicable law, we have the discretion to deliver placement notices to the sales agent at any
time throughout the term of the sales agreement. The number of shares that are sold by the sales agent after delivering a placement notice
will fluctuate based on the market price of the common stock during the sales period and limits we set with the sales agent.
The shares of common stock offered under this
prospectus and documents incorporated by reference into this prospectus may be sold in “at the market offerings,” and investors
who buy shares at different times will likely pay different prices.
Investors who purchase shares under this prospectus
and documents incorporated by reference into this prospectus at different times will likely pay different prices, and so may experience
different outcomes in their investment results. We will have discretion, subject to market demand, to vary the timing, prices, and numbers
of shares sold, and there is no minimum or maximum sales price. Investors may experience declines in the value of their shares as a result
of share sales made at prices lower than the prices they paid.
USE OF PROCEEDS
We may issue and sell shares of our common stock
having aggregate gross proceeds of up to $4,037,193 from time to time under this prospectus and the documents incorporated by reference
herein and therein. Because there is no minimum offering amount required as a condition to close this offering, the actual total offering
amount, commissions and proceeds to us, if any, are not determinable at this time. The amount of proceeds from this offering will depend
upon the number of shares of our common stock sold and the market price at which they are sold. There can be no assurance that we will
be able to sell any shares of our common stock under or fully utilize the sales agreement with A.G.P. as a source of financing.
We intend to use the net proceeds, if any, from
the sales of shares of our common stock offered by this prospectus for working capital and general corporate purposes, including, to acquire,
license or invest in assets, businesses, technologies, product candidates or other intellectual property and to repurchase securities.
We have broad discretion in determining how the proceeds of this offering will be used, and our discretion is not limited by the aforementioned
possible uses. Our Board of Directors believes the flexibility in application of the net proceeds is prudent.
As of the date of this prospectus, we cannot
specify with certainty all of the particular uses for the net proceeds to be received from this offering. The amounts and timing of our
actual expenditures will depend on numerous factors including our operating costs and the amount of funding, if any, received from grants.
Accordingly, our management will have broad discretion in the application of the net proceeds, and investors will be relying on the judgment
of management regarding the application of the net proceeds from the offering. We may find it necessary or advisable to reallocate the
net proceeds of this offering; however, any such reallocation would be substantially limited to the categories set forth above as we do
not intend to use the net proceeds for other purposes. Pending such uses set forth above, we plan to invest the net proceeds in government
securities and other short-term investment grade, marketable securities.
DIVIDEND POLICY
We have never declared or paid any cash dividends
on our capital stock and we do not currently intend to pay any cash dividends on our common stock in the foreseeable future. We expect
to retain all available funds and future earnings, if any, to fund the development and growth of our business. Any future determination
to pay dividends, if any, on our common stock will be at the discretion of our Board of Directors and will depend on, among other factors,
our results of operations, financial condition, capital requirements and contractual restrictions.
PLAN OF DISTRIBUTION
We entered into the sales agreement with A.G.P. under
which we may issue and sell from time to time shares of our common stock having an aggregate offering price of up to $4,037,193 to or
through A.G.P., acting as our sales agent or principal. The sales of shares of our common stock, if any, under this prospectus supplement
will be made at market prices by any method deemed to be an “at the market offering” as defined in Rule 415(a)(4) under the
Securities Act.
We entered into the sales agreement with A.G.P. under
which we may issue and sell from time to time shares of our common stock having an aggregate offering price of up to $4,037,193 to or
through A.G.P., acting as our sales agent or principal. The sales of shares of our common stock, if any, under this prospectus will be
made at market prices by any method deemed to be an “at the market offering” as defined in Rule 415(a)(4) under the Securities
Act.
Each time that we wish to issue and sell shares of
our common stock under the sales agreement, we will provide A.G.P. with a placement notice describing the amount of shares to be sold,
the time period during which sales are requested to be made, any limitation on the amount of shares of our common stock that may be sold
in any single day, any minimum price below which sales may not be made or any minimum price requested for sales in a given time period
and any other instructions relevant to such requested sales. Upon receipt of a placement notice, A.G.P., acting as our sales agent, will
use commercially reasonable efforts, consistent with its normal trading and sales practices and applicable state and federal laws, rules
and regulations and the rules of the NYSE American, to sell shares of our common stock under the terms and subject to the conditions of
the placement notice and the sales agreement. We or A.G.P. may suspend the offering of shares of our common stock pursuant to a placement
notice upon notice.
Settlement for sales of shares of common stock, unless
the parties agree otherwise, will occur on the second trading day following the date on which any sales are made in return for payment
of the net proceeds to us. There are no arrangements to place any of the proceeds of this offering in an escrow, trust or similar account.
Sales of shares of our common stock as contemplated in this prospectus will be settled through the facilities of The Depository Trust
Company or by such other means as we and A.G.P. may agree upon.
A.G.P. will be entitled to compensation at a fixed
commission rate of 3.0% of the gross proceeds from the sale of shares of our common stock on our behalf pursuant to the sales agreement.
Pursuant to the terms of the sales agreement, we agreed to reimburse A.G.P. for the documented fees and costs of its legal counsel reasonably
incurred in connection with entering into the transactions contemplated by the sales agreement in an amount not to exceed $50,000 in the
aggregate, in addition to up to $10,000 annually for A.G.P.’s counsel’s fees and any incidental expenses to be reimbursed
by us. We will report at least quarterly the number of shares of common stock sold through A.G.P. under the sales agreement, the net proceeds
to us and the compensation paid by us to A.G.P. in connection with the sales of common stock.
We estimate that the total expenses for this offering,
excluding compensation payable to A.G.P. and certain expenses reimbursable to A.G.P. under the terms of the sales agreement, will be approximately
$125,000. The remaining sales proceeds, after deducting any expenses payable by us and any transaction fees imposed by any governmental,
regulatory, or self-regulatory organization in connection with the sales, will equal our net proceeds for the sale of such shares of common
stock.
Because there are no minimum sale requirements as
a condition to this offering, the actual total public offering price, commissions and net proceeds to us, if any, are not determinable
at this time. The actual dollar amount and number of shares of common stock we sell through this prospectus will be dependent, among other
things, on market conditions and our capital raising requirements.
In connection with the sale of shares of common stock
on our behalf, A.G.P. will be deemed to be an “underwriter” within the meaning of the Securities Act, and the compensation
of A.G.P. will be deemed to be underwriting commissions or discounts. We have agreed to provide indemnification and contribution to A.G.P.
against certain civil liabilities, including liabilities under the Securities Act.
A.G.P. will not engage in any market making activities
involving shares of our common stock while the offering is ongoing under this prospectus if such activity would be prohibited under Regulation
M or other anti-manipulation rules under the Securities Act. As our sales agent, A.G.P. will not engage in any transactions that stabilizes
shares of our common stock.
The offering pursuant to the sales agreement will
terminate upon the earlier of (i) the sale of all shares of common stock subject to the sales agreement and (ii) termination of the sales
agreement as permitted therein. We may terminate the sales agreement in our sole discretion at any time by giving one (1) day’s
prior notice to A.G.P. A.G.P. may terminate the sales agreement under the circumstances specified in the sales agreement and in its sole
discretion at any time by giving five (5) days’ prior notice to us.
A.G.P. and/or its affiliates have provided, and may
in the future provide, various investment banking and other financial services for us, for which services they have received and may in
the future receive customary fees. In addition, A.G.P. served as a financial advisor with respect to our search for strategic alternatives.
This prospectus may be made available in electronic
format on a website maintained by A.G.P., and A.G.P. may distribute this prospectus electronically.
LEGAL MATTERS
The validity of the shares of common stock offered
hereby will be passed upon for us by Blank Rome LLP, New York, New York. Duane Morris LLP, New York, New York, is acting as counsel to
the sales agent in this offering.
As of the date of this prospectus, an attorney of
Blank Rome LLP beneficially owns common stock and securities exercisable to purchase shares of our common stock that represent less than
1% of our outstanding shares of common stock.
EXPERTS
The consolidated financial statements as of December
31, 2022 and 2021 and for the years then ended, incorporated by reference in this prospectus and the registration statement have been
so incorporated in reliance on the report of BDO USA, LLP (n/k/a BDO USA, P.C.), an independent registered public accounting firm, incorporated
herein by reference, given on the authority of said firm as experts in accounting and auditing. The report on the consolidated financial
statements contains an explanatory paragraph regarding the Company’s ability to continue as a going concern.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus is part of a registration statement
we filed with the SEC. This prospectus does not contain all of the information set forth in the registration statement and the exhibits
to the registration statement. For further information with respect to us and the Common Stock we are offering under this prospectus,
we refer you to the registration statement and the exhibits and schedules filed as a part of the registration statement. Neither we nor
any agent, underwriter or dealer has authorized any person to provide you with different information. We are not making an offer of the
Common Stock in any state where the offer is not permitted. You should not assume that the information in this prospectus is accurate
as of any date other than the date on the front page of this prospectus, regardless of the time of delivery of this prospectus or any
sale of the shares of our Common Stock offered by this prospectus.
We file annual, quarterly and current reports, proxy
statements and other information with the SEC. Our SEC filings are available to the public at the SEC’s website at www.sec.gov.
Our SEC filings are also available on our website, www.nighthawkbio.com under the heading “Investors & News—SEC
Filings.” The reference to our website is an inactive textual reference only, the information contained in, and that can be accessed
through our website, is not incorporated into and is not a part of this prospectus. We make available on our website our SEC filings as
soon as reasonably practicable after those reports are filed with the SEC.
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to “incorporate by reference”
information into this prospectus, which means that we can disclose important information to you by referring you to another document filed
separately with the SEC. The SEC file number for the documents incorporated by reference in this prospectus is 001-35994. The documents
incorporated by reference into this prospectus contain important information that you should read about us.
The following documents are incorporated by reference
into this prospectus:
| |
• |
Our Annual Report on Form
10-K for the fiscal year ended December 31, 2022 filed with the SEC on March 31, 2023; |
| |
• |
Our Quarterly Report on Form
10-Q for the fiscal quarter ended March 31, 2023 filed with the SEC on May 15, 2023, our Quarterly Report on Form
10-Q for the quarter ended June 30, 2023 filed with the SEC on August 14, 2023, and our Quarterly Report on Form
10-Q for the quarter ended September 30, 2023 filed with the SEC on November 20, 2023; |
| |
• |
Our Current Report on Form 8-K filed with the SEC on January
5, 2023, February 2, 2023, March
13, 2023, March 23, 2023,
August 21, 2023, September
19, 2023, as amended on September
20, 2023, September 28, 2023,
December 8, 2023, December 12, 2023, and December 19, 2023; and |
| |
• |
The description of our common stock and our common stock purchase rights
is set forth in our registration statement on Form
8-A filed with the SEC on February 4, 2022, Form
8-A/A (Amendment No. 1) filed on March 11, 2022 and Form
8-A/A (Amendment No. 2) filed on March 13, 2023, as updated by the description of our common stock filed as Exhibit 4.18
to our Annual Report on Form 10-K for the year ended December 31, 2022 filed with the SEC on March 31, 2023, including any
amendments or reports filed for the purpose of updating such description. |
We also incorporate by reference into this prospectus
all documents (other than Current Reports furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits filed on such form that are
related to such items or other information “furnished” to the SEC, which is not deemed filed and not incorporated in this
prospectus) that are filed by us with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of this
prospectus and before the completion of the offering of the securities included in this prospectus. These documents include periodic reports,
such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K.
We will provide to each person, including any beneficial
owner, to whom a prospectus is delivered, without charge upon written or oral request, a copy of any or all of the documents that are
incorporated by reference into this prospectus but not delivered with the prospectus, including exhibits that are specifically incorporated
by reference into such documents. You can request a copy of these filings, at no cost, by writing or telephoning us at the following address
or telephone number:
NightHawk Biosciences, Inc.
627 Davis Drive, Suite 300
Morrisville, North Carolina 27560
Telephone: (919) 240-7133
Attn: Investor Relations
Any statement contained in this prospectus or contained
in a document incorporated or deemed to be incorporated by reference into this prospectus will be deemed to be modified or superseded
to the extent that a statement contained in this prospectus or any subsequently filed supplement to this prospectus, or document deemed
to be incorporated by reference into this prospectus, modifies or supersedes such statement.

Up to $4,037,193
Shares of Common Stock
Prospectus
A.G.P.
, 2023
Item 14. Other Expenses of Issuance and Distribution.
The following sets forth
the various costs and expenses, all of which shall be borne by NightHawk Biosciences, Inc. (the “Registrant”), in connection
with the offering of the securities pursuant to this registration statement. All amounts shown are estimates, except for the registration
fee required by the Securities and Exchange Commission (“SEC”) and the Financial Industry Regulatory Authority, Inc. (“FINRA”).
| SEC registration fee |
|
$ |
— |
* |
| FINRA filing fee |
|
|
— |
|
| Accounting fees and expenses |
|
|
(1) |
|
| Legal fees and expenses |
|
|
(1) |
|
| Transfer agent fees and expenses |
|
|
(1) |
|
| Printing fees and expenses |
|
|
(1) |
|
| Miscellaneous |
|
|
(1) |
|
| Total |
|
$ |
(1) |
|
| (1) | These fees are calculated based on the securities offered and the number
of issuances and, accordingly, cannot be estimated at this time. An estimate of the aggregate expenses in connection with the sale and
distribution of securities being offered will be included in the applicable prospectus supplement. |
| * |
Excludes the registration fee previously paid in connection with unsold securities pursuant to Rule 415(a)(6). |
Item 15. Indemnification of Directors and Officers.
Section 145 of the Delaware General Corporation Law
empowers a corporation to indemnify its directors and officers and to purchase insurance with respect to liability arising out of their
capacity or status as directors and officers, provided that the person acted in good faith and in a manner the person reasonably believed
to be in our best interests, and, with respect to any criminal action, had no reasonable cause to believe the person’s actions were
unlawful. The Delaware General Corporation Law further provides that the indemnification permitted thereunder shall not be deemed exclusive
of any other rights to which the directors and officers may be entitled under the corporation’s bylaws, any agreement, a vote of
stockholders or otherwise. Our Third Amended and Restated Certificate of Incorporation, as amended (the “Certificate of Incorporation”),
and our Second Amended and Restated Bylaws (the “Bylaws”) provide that our directors and officers will be indemnified by us
to the fullest extent authorized by Delaware General Corporation Law, as it now exists or may in the future be amended.
Section 102(b)(7) of the Delaware General Corporation
Law permits a corporation to provide in its certificate of incorporation that a director of the corporation shall not be personally liable
to the corporation or its stockholders for monetary damages for breach of fiduciary duty as a director, except (i) for any breach of the
director’s duty of loyalty to the corporation or its stockholders; (ii) for acts or omissions not in good faith or which involve
intentional misconduct or a knowing violation of law; (iii) for payments of unlawful dividends or unlawful stock repurchases or redemptions;
or (iv) for any transaction from which the director derived an improper personal benefit. Our Certificate of Incorporation provides that
the liability of the registrant’s directors for monetary damages shall be eliminated to the fullest extent under applicable law.
As permitted by the Delaware General Corporation
Law, we have entered into separate indemnification agreements with each of our directors and certain of our officers which require the
registrant, among other things, to indemnify them against certain liabilities which may arise by reason of their status as directors or
officers.
We have an insurance policy in place that covers our
officers and directors with respect to certain liabilities, including liabilities arising under the Securities Act or otherwise.
In any underwriting agreement, securities purchase
agreement, placement agency agreement or similar agreement that we may enter into in connection with the sale of certain securities being
registered hereby, the underwriter, purchase, or placement agent will agree to indemnify, under certain conditions, us, our directors,
our officers and persons who control us, within the meaning of the Securities Act, against certain liabilities.
Item 16. Exhibits
The exhibits to this registration
statement are listed in the Exhibit Index to this registration statement, which immediately precedes the Signature Page and which Exhibit
Index is hereby incorporated by reference.
Item 17. Undertakings
The undersigned registrant hereby undertakes:
(1) To file, during any period in which offers or
sales are being made, a post-effective amendment to this registration statement:
| (i) |
To include any prospectus required by Section 10(a)(3) of the Securities Act; |
| |
|
| (ii) |
To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the SEC pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20 percent change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and |
| |
|
| (iii) |
To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement; |
provided, however, that the undertakings set
forth in paragraphs (1)(i), (1)(ii) and (1)(iii) above do not apply if the information required to be included in a post-effective amendment
by those paragraphs is contained in reports filed with or furnished to the SEC by the registrant pursuant to Section 13 or Section 15(d)
of the Securities Exchange Act of 1934, as amended, or the Exchange Act, that are incorporated by reference in this registration statement
or are contained in a form of prospectus filed pursuant to Rule 424(b) that is part of this registration statement.
(2) That, for the purpose of determining any liability
under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities
offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
(3) To remove from registration by means of a post-effective
amendment any of the securities being registered which remain unsold at the termination of the offering.
(4) That, for the purpose of determining liability
under the Securities Act to any purchaser:
| |
(i) |
Each prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the date the filed prospectus was deemed part of and included in the registration statement; and |
| |
|
|
| |
(ii) |
Each prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) as part of a registration statement in reliance on Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii), or (x) for the purpose of providing the information required by Section 10(a) of the Securities Act shall be deemed to be part of and included in the registration statement as of the earlier of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale of securities in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities in the registration statement to which that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such effective date, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such effective date. |
(5) That, for the purpose of determining liability
of the registrant under the Securities Act to any purchaser in the initial distribution of the securities, the undersigned registrant
undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless
of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means
of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or
sell such securities to such purchaser:
| (i) |
any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424; |
| (ii) |
any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant; |
| (iii) |
the portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and |
| (iv) |
any other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
(6) That, for purposes of determining any liability
under the Securities Act, each filing of the registrant’s annual report pursuant to Section 13(a) or 15(d) of the Exchange Act (and,
where applicable, each filing of an employee benefit plan’s annual report pursuant to Section 15(d) of the Exchange Act) that is
incorporated by reference in the registration statement shall be deemed to be a new registration statement relating to the securities
offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
(7) To file
an application for the purpose of determining the eligibility of the trustee to act under subsection (a) of Section 310 of the
Trust Indenture Act of 1939, as amended (the “Trust Indenture Act”) in accordance with the rules and regulations prescribed
by the SEC under Section 305(b)(2) of the Trust Indenture Act.
Insofar as indemnification for liabilities arising
under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions,
or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification is against public policy as expressed
in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other
than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the
successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the
securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent,
submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in
the Securities Act and will be governed by the final adjudication of such issue.
EXHIBIT INDEX
| Exhibit No. Description |
|
1.1**
|
|
Form of Underwriting Agreement
|
| 1.2** |
|
Form of Placement Agency Agreement |
| 1.3 |
|
Sales Agreement, dated December 8, 2023, by and between NightHawk Biosciences, Inc. and A.G.P./Alliance Global Partners (incorporated by reference to Exhibit 1.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission December 8, 2023 (File No. 001 35994)) |
| 3.1 |
|
Third Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.5 to the Registration Statement on Form S-1 with the Securities and Exchange Commission on May 6, 2013 (File No. 333-188365)) |
| 3.2 |
|
Certificate of Amendment to the Third Amended and Restated Certificate of Incorporation filed on May 29, 2013 (incorporated by reference to Exhibit 3.6 to the Registration Statement on Form S-1 with the Securities and Exchange Commission on May 30, 2013 (File No. 333-188365)) |
| 3.3 |
|
Certificate of Amendment to the Third Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on July 17, 2017 (File No. 001-35994)) |
| 3.4 |
|
Certificate of Amendment to the Third Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on January 19, 2018 (File No. 001-35994)) |
| 3.5 |
|
Certificate of Amendment to the Third Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission March 23, 2020 (File No. 001 35994)) |
| 3.6 |
|
Amended and Restated Bylaws, dated October 17, 2019 (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on October 18, 2019 (File No. 001-35994)) |
| 3.7 |
|
Certificate of Amendment to the Third Amended and Restated Certificate of Incorporation of Heat Biologics, Inc. (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on December 10, 2020 (File No. 001 35994)) |
| 3.8 |
|
Certificate of Amendment to the Third Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on May 3, 2022 (File No. 001 35994)) |
| 3.9 |
|
Second Amended and Restated Bylaws, dated May 3, 2022 (incorporated by reference to Exhibit 3.2 to the Current Report on Form 8-K filed with the Securities and Exchange Commission on May 3, 2022 (File No. 001-35994)) |
| 4.1 |
|
Specimen stock certificate evidencing the shares of common stock (Incorporated by reference to Exhibit 4.8 of the Registrant’s Form S-1 (File No. 333-188365) filed with the Securities and Exchange Commission on May 6, 2013) |
| 4.2** |
|
Specimen Preferred Stock Certificate |
| 4.3** |
|
Form of Certificate of Designation of Preferred Stock |
| 4.4** |
|
Form of Common Stock Warrant Agreement and Warrant Certificate |
| 4.5** |
|
Form of Preferred Stock Warrant Agreement and Warrant Certificate |
| 4.6** |
|
Form of Debt Securities Warrant Agreement and Warrant Certificate |
| 4.7** |
|
Form of Unit Agreement |
| 5.1* |
|
Legal opinion of Blank Rome LLP |
| 23.1* |
|
Consent of Independent Registered Public Accounting Firm (BDO USA, P.C.) |
| 23.2** |
|
Consent of Blank Rome LLP (included in Exhibit 5.1) |
| 24.1* |
|
Power of Attorney (included on the signature page of this Registration Statement) |
| 25.1*** |
|
Statement of Eligibility of Trustee under the Indenture |
| 107* |
|
Filing fee table |
_________
| * |
Filed herewith. |
| ** |
To be filed, if applicable, by amendment or by a report filed under the Exchange Act and incorporated herein by reference. |
| *** |
To be filed separately, if applicable, in accordance with the requirements of Section 305(b)(2) of the Trust Indenture Act of 1939 as amended, and the appropriate rules and regulations thereunder. |
SIGNATURES
Pursuant to the requirements of the Securities Act
of 1933, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized
in the City of Morrisville, State of North Carolina, on December 19, 2023.
| |
|
NightHawk Biosciences, Inc. |
| |
|
|
|
| |
|
By: |
/s/ Jeffrey Wolf |
| |
|
|
Jeffrey Wolf, Chief Executive Officer |
| |
|
|
(Principal Executive Officer) |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person
whose signature appears below hereby constitutes and appoints Jeffrey Wolf and William Ostrander, and each of them singly (with full power
to each of them to act alone), our true and lawful attorneys-in-fact and agents, with full power of substitution and re-substitution in
each of them for him and in his name, place and stead, and in any and all capacities, to sign any and all amendments (including post-effective
amendments) to this Registration Statement, and to file the same, with all exhibits thereto and other documents in connection therewith,
with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority
to do and perform each and every act and thing requisite or necessary to be done in and about the premises, as fully to all intents and
purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or any of them,
or their substitutes, may lawfully do or cause to be done by virtue hereof. This power of attorney may be executed in counterparts.
Pursuant to the requirements of the Securities Act
of 1933, this registration statement has been signed by the following persons in the capacities and on the dates indicated:
| |
|
|
|
|
|
| Signature |
|
Title |
|
Date |
|
| |
|
|
|
|
|
| /s/ Jeffrey Wolf |
|
Chief Executive Officer,
President and Chairman
(Principal Executive Officer) |
|
December 19, 2023 |
|
Jeffrey Wolf
|
|
|
|
|
|
| /s/ William Ostrander |
|
Chief Financial Officer
(Principal Financial Officer and
Principal Accounting Officer) |
|
December 19, 2023 |
|
William Ostrander
|
|
|
|
|
| /s/ John Monahan |
|
Director |
|
December 19, 2023 |
|
John Monahan, Ph.D.
|
|
|
|
|
| /s/ John Prendergast |
|
Director |
|
December 19, 2023 |
|
John Prendergast, Ph.D.
|
|
|
|
|
| /s/ Edward B. Smith, III |
|
Director |
|
December 19, 2023 |
| Edward B. Smith, III |
|
|
|
|
Exhibit 5.1

1271 Avenue of the Americas | New York,
New York 10020
Blankrome.com
December 19,
2023
NightHawk Biosciences, Inc.
627 Davis Drive, Suite 300
Morrisville, North Carolina 27560
Re: NightHawk Biosciences,
Inc.
Registration Statement
on Form S-3
Ladies and Gentlemen:
We
have acted as counsel to NightHawk Biosciences, Inc., a Delaware corporation (the “Company”), in connection with the
preparation and filing by the Company with the Securities and Exchange Commission (the “Commission”) of a Registration
Statement on Form S-3 (the “Registration Statement”) under the Securities Act of 1933, as amended (the “Securities
Act”), with respect to the contemplated issuance from time to time of up to $200,000,000 of any combination of the following
securities (the “Registered Securities”): (i) shares of common stock of the Company, par value $0.0002 per share (the
“Common Stock”); (ii) shares of preferred stock of the Company, par value $ 0.0001 per share (the “Preferred
Stock”); (iii) warrants to purchase Common Stock, Preferred Stock or Debt Securities (the “Warrants”), which
may be issued under one or more warrant agreements, to be dated on or about the date of the first issuance of the Warrants thereunder,
by and between a warrant agent to be selected by the Company (the “Warrant Agent”) and the Company (each, a “Warrant
Agreement”); and (iv) units consisting of one or more of the other securities described in (i) through (iii) above in any combination
(the “Units”). This opinion is being furnished in accordance with the requirements of Item 601(b)(5) of Regulation
S-K under the Securities Act.
The
Registration Statement also relates to the Common Stock purchase rights (the “Rights”) issuable in accordance with
the rights agreement, dated as of March 11, 2018, as amended by Amendment No. 1 thereto, dated March 8, 2019, Amendment No. 2 thereto,
dated March 10, 2020, by and Amendment No. 3 thereto dated March 8, 2021, Amendment No. 4 thereto dated March 11, 2022, Amendment No.
5 thereto dated March 11, 2023 and Amendment No. 6 thereto dated December 11, 2023 (collectively, the “Rights Agreement”)
between the Company and Continental Stock Transfer & Trust Company, as Rights Agent, which Rights are presently attached to and trade
with the Common Stock.
We
are also acting as counsel for the Company in connection with the Sales Agreement prospectus included in the Registration Statement (the
“Sales Agreement ATM Prospectus”) relating to the issuance and sale of shares of Common Stock having an aggregate
offering price of up to $4,037,193 (the “Sales Agreement Shares”) under the Sales Agreement, dated December 8, 2023,
by and between the Company and A.G.P./Alliance Global Partners, as the sales agents (the “Sales Agreement”).
In
rendering the opinions set forth herein, we have examined originals or copies, certified or otherwise identified to our satisfaction,
of (i) the Registration Statement, (ii) the Sales Agreement ATM Prospectus, (iii) the Sales Agreement, (iv) resolutions adopted by the
Board of Directors of the Company (the “Board”), (v) the Third Amended and Restated Certificate of Incorporation,
as amended (the “Certificate of Incorporation”), (vi) the Second Amended and Restated bylaws, (vii) the Rights Agreement
and (viii) such other corporate records, agreements, certificates, including, but not limited to, certificates or comparable documents
of public officials and of officers and representatives of the Company, statutes and other instruments and documents as we considered
relevant and necessary as a basis for the opinions hereinafter expressed.

NightHawk Biosciences,
Inc.
December 19, 2023
Page 2
In
rendering this opinion, we have assumed, without inquiry, (i) the authenticity of all documents submitted to us as originals; (ii) the
conformity to the original documents of all documents submitted to us as facsimile, electronic, certified or photostatic copies, and
the authenticity of the originals of such copies; (iii) the legal capacity of all natural persons and the genuineness of all signatures
on the Registration Statement and all documents submitted to us; and (iv) that the books and records of the Company are maintained in
accordance with proper corporate procedures.
In
making our examination of executed documents or documents to be executed, we have assumed that the parties thereto had or will have the
power, corporate or other, to enter into and perform all obligations thereunder and we have also assumed the due authorization by all
requisite action, corporate or other, and execution and delivery by such parties of such documents and (except to the extent we have
opined on such matters below) the validity and binding effect on such parties. In addition, we have assumed that (i) a purchase, underwriting
or similar agreement with respect to any Registered Securities will have been duly authorized and validly executed and delivered by the
Company and the other parties thereto; (ii) the Registration Statement and any amendments thereto will have become effective and comply
with all applicable laws at the time the Registered Securities are offered or issued as contemplated by the Registration Statement and
no stop order suspending its effectiveness will have been issued and remain in effect; (iii) all Registered Securities will be issued
and sold in compliance with applicable federal and state securities laws and in the manner stated in the Registration Statement and the
applicable prospectus supplement; (iv) a prospectus supplement or term sheet will have been prepared and filed with the Commission describing
the Registered Securities offered thereby and will comply at all relevant times with all applicable laws; (v) the Company will have obtained
any legally required consents, approvals, authorizations and other orders of the Commission and any other regulatory authorities necessary
(x) to issue and sell the Registered Securities being offered, and (y) to execute and deliver the applicable purchase, underwriting or
similar agreement, or other applicable operative document; (vi) any securities issuable upon conversion, exchange, redemption or exercise
of any Registered Securities being offered will be duly authorized, created and, if appropriate, reserved for issuance upon such conversion,
exchange, redemption or exercise and, with respect to shares of Common Stock or Preferred Stock offered, there will be sufficient shares
of Common Stock or Preferred Stock, as applicable, authorized under the Certificate of Incorporation and not otherwise reserved for issuance;
(vii) at the time of issuance of the Registered Securities, the Company validly exists and is duly qualified and in good standing under
the laws of its jurisdiction of incorporation, and has the necessary corporate power for such issuance; (viii) at the time of issuance
of the Registered Securities, the Certificate of Incorporation and then-operative by-laws of the Company are in full force and effect
and have not been amended, restated, supplemented or otherwise altered, and there has been no authorization of any such amendment, restatement,
supplement or other alteration, in either case since the date hereof; and (ix) the terms, execution and delivery of the Registered Securities
(x) do not result in breaches of, or defaults under, agreements or instruments to which the Company is bound or violations of applicable
statutes, rules, regulations or court or governmental orders, and (y) comply with any applicable requirement or restriction imposed by
any court or governmental body having jurisdiction over the Company. As to any facts material to the opinions expressed herein that we
have not independently established or verified, we have relied upon, and assumed the accuracy of, statements and representations of officers
and other representatives of the Company and others.
In connection with the Sales Agreement Shares, we
have assumed (i) that each sale of Sales Agreement Shares will be duly authorized by the Board of Directors of the Company, a duly authorized
committee thereof or a person or body pursuant to an authorization granted in accordance with Section 152 of the Delaware General Corporation
Law, (ii) at the time of issuance of any Sales Agreement Shares, there shall be a sufficient number of duly authorized and unissued shares
of Common Stock to accommodate the issuance of the Sales Agreement Shares, and (iii) that the price at which the Sales Agreement Shares
are sold will equal or exceed the par value of the Common Stock. We express no opinion to the extent that future issuances of securities
of the Company, anti-dilution adjustments to outstanding securities of the Company and/or other matters cause the number of shares of
the Common Stock available for issuance under the Sales Agreement to exceed the number of shares of the Common Stock available for issuance
by the Company.

NightHawk Biosciences,
Inc.
December 19, 2023
Page 3
Based on the foregoing,
and subject to the qualifications, exceptions and assumptions stated herein, we are of the opinion that:
1.
With respect to shares of Common Stock, when (i) the Board or a committee thereof has taken all necessary corporate action to approve
the issuance and terms of the offering thereof and related matters; and (ii) certificates representing the shares of Common Stock have
been duly executed, countersigned, registered and delivered, or if uncertificated, valid book-entry notations have been made in the share
register of the Company, in each case in accordance with the Certificate of Incorporation and then-operative by-laws, either (A) in accordance
with the applicable purchase, underwriting or similar agreement approved by the Board or a committee thereof, then upon payment of the
consideration therefor (not less than the par value of the Common Stock) provided for therein; or (B) upon conversion, exchange or exercise
of any other security in accordance with the terms of the security or the instrument governing the security providing for the conversion,
exchange or exercise as approved by the Board or a committee thereof, for the consideration approved by the Board or a committee thereof
(not less than the par value of the Common Stock), such shares of Common Stock will be validly issued, fully paid and non-assessable.
2.
With respect to shares of any class or series of Preferred Stock, when (i) the Board or a committee thereof has taken all necessary corporate
action to approve the issuance and terms of the shares of the class or series, the terms of the offering thereof and related matters,
including the adoption of a resolution establishing and designating the series and fixing and determining the preferences, limitations
and relative rights thereof and the filing of a certificate of designation with respect to the class or series with the Secretary of
State of the State of Delaware as required by applicable law (the “Certificate of Designation”); and (ii) certificates
representing the shares of the series of Preferred Stock have been duly executed, countersigned, registered and delivered, or if uncertificated,
valid book-entry notations have been made in the share register of the Company, in each case in accordance with the Certificate of Incorporation,
Certificate of Designations and then-operative by-laws, either (A) in accordance with the applicable purchase, underwriting or similar
agreement approved by the Board or a committee thereof, then upon payment of the consideration therefor (not less than the par value
of the Preferred Stock) provided for therein; or (B) upon conversion, exchange or exercise of any other security in accordance with the
terms of the security or the instrument governing the security providing for the conversion, exchange or exercise as approved by the
Board or a committee thereof, for the consideration approved by the Board or a committee thereof (not less than the par value of the
Preferred Stock), such shares of such series of Preferred Stock will be validly issued, fully paid and non-assessable.
3.
With respect to Warrants, when (i) the Board or a committee thereof has taken all necessary corporate action to approve the issuance
and terms of the Warrants, the terms of the offering thereof and related matters; (ii) the Warrant Agreement relating to the Warrants
has been duly authorized and validly executed and delivered by the Company and the Warrant Agent appointed by the Company; and (iii)
the Warrants or certificates representing the Warrants have been duly executed, countersigned, registered and delivered in accordance
with the appropriate Warrant Agreement and the applicable purchase, underwriting or similar agreement approved by the Board or a committee
thereof, then upon payment of the consideration provided for therein, the Warrants will constitute valid and binding obligations of the
Company.

NightHawk Biosciences,
Inc.
December 19, 2023
Page 4
4.
With respect to Units, when (i) the Board or a committee thereof has taken all necessary corporate action to approve the terms of the
Units, the terms of the offering thereof and related matters; and (ii) the Units have been duly executed and delivered in accordance
with the applicable purchase, underwriting or similar agreement approved by or on behalf of the Board or a committee thereof, then upon
payment of the consideration therefor provided therein, the Units will constitute valid and binding obligations of the Company.
5. With respect to the Sales Agreement
Shares, when the Sales Agreement Shares have been issued and paid for in accordance with the terms and conditions of the Sales Agreement,
the Sales Agreement Shares will be validly issued, fully paid and nonassessable.
6. With respect to each of the
Rights attached to shares of Common Stock, each of such Rights when issued in accordance with the terms of the Rights Agreement, will
constitute a valid and binding obligation of the Company.
In addition to the assumptions,
comments, qualifications, limitations and exceptions set forth above, the opinions set forth herein are further limited by, subject to
and based upon the following:
| |
a. |
Our opinions herein are expressed solely with
respect to the Delaware General Corporation Law, and, as to the Warrants and Units constituting valid and binding obligations of
the Company, the applicable laws of the State of New York that, in our experience, are normally applicable to transactions of the
type contemplated by the Registration Statement. The opinion set forth herein related to the Units assumes that such Units are governed
by New York law. The opinions set forth herein are made as of the date hereof and are subject to, and may be limited by, future changes
in the factual matters set forth herein, and we undertake no duty to advise you of the same that may occur after the Registration
Statement becomes effective. The opinions expressed herein are based upon the law in effect (and published or otherwise generally
available) on the date hereof, which laws are subject to change with possible retroactive effect, and we assume no obligation to
revise or supplement these opinions should such law be changed by legislative action, judicial decision or otherwise after the Registration
Statement becomes effective. In rendering our opinions, we have not considered, and hereby disclaim any opinion as to, the application
or impact of any laws, cases, decisions, rules or regulations of any other jurisdiction, court or administrative agency. |
| |
b. |
Our opinions set forth above are subject to and
may be limited by (i) applicable bankruptcy, reorganization, insolvency, conservatorship, moratorium, fraudulent conveyance, debtor
and creditor, and similar laws which relate to or affect creditors’ rights generally, and (ii) general principles of equity
(including, without limitation, concepts of materiality, reasonableness, impossibility of performance, good faith and fair dealing)
regardless of whether considered in a proceeding in equity or at law. |
| |
c. |
Our opinions are subject to the qualification
that the availability of specific performance, an injunction or other equitable remedies is subject to the discretion of the court
before which the request is brought. |

NightHawk Biosciences,
Inc.
December 19, 2023
Page 5
| |
d. |
In addition, in rendering the opinion in paragraph
6 above with respect to the Rights, (1) we do not address the determination a court of competent jurisdiction may make regarding whether
the Board would be required to redeem or terminate, or take other action with respect to, the Rights at some future time based on the
facts and circumstances existing at that time, (2) we assume that the members of the Board acted in a manner consistent with their fiduciary
duties as required under applicable law in adopting the Rights Agreement, and (3) we address the Rights and the Rights Agreement in their
entirety, and it is not settled whether the invalidity of any particular provision of the Rights Agreement or of the Rights issued thereunder
would result in invalidating the Rights Agreement or the Rights in their entirety.
|
| |
e. |
You have informed us that you intend to issue
the Registered Securities from time to time on a delayed or continuous basis, and this opinion is limited to the laws, including
the rules and regulations, as in effect on the date hereof. We understand that prior to issuing any Registered Securities you will
afford us an opportunity to review the operative documents pursuant to which such Registered Securities are to be issued (including
the applicable prospectus supplement) and will file such supplement or amendment to this opinion (if any) as we may reasonably consider
necessary or appropriate by reason of the terms of such Registered Securities. |
We
consent to the use of this opinion as an exhibit to the Registration Statement. We also consent to any and all references to us in the
prospectus which is part of said Registration Statement. In giving this consent, we do not thereby admit that we are within the category
of persons whose consent is required under Section 7 of the Securities Act or the rules and regulations of the Commission promulgated
thereunder. This opinion is strictly limited to the matters stated herein and no other or more extensive opinion is intended, implied
or to be inferred beyond the matters expressly stated herein. This opinion letter is not a guaranty nor may one be inferred or implied.
| |
Very truly yours, |
| |
|
| |
/s/ Blank Rome LLP |
| |
BLANK ROME LLP |
Exhibit 23.1
Consent of Independent Registered Public
Accounting Firm
We hereby consent to the incorporation by reference
in the Prospectus constituting a part of this Registration Statement of our report dated March 31, 2023, except for the effects of discontinued
operations discussed in Note 18, as to which the date is December 19, 2023, relating to the consolidated financial statements of NightHawk
Biosciences, Inc. (the Company), which appears in the Company’s Form 8-K filed on December 19, 2023. Our report contains an explanatory
paragraph regarding the Company’s ability to continue as a going concern.
We also consent to the reference to us under
the caption “Experts” in the Prospectus.
/s/ BDO USA, P.C
Raleigh, North Carolina
December 19, 2023
Exhibit 107
Calculation of Filing Fee Tables
Form S-3
(Form Type)
NightHawk
Biosciences, Inc.
(Exact name of Registrant as specified in its charter)
Table 1: Newly Registered and Carry Forward Securities
| |
|
Security
Type |
|
Security Class
Title |
|
Fee
Calculation
or Carry
Forward
Rule |
|
Amount
Registered |
|
|
Proposed
Maximum
Offering
Price Per
Unit |
|
|
Maximum
Aggregate
Offering Price |
|
|
Fee
Rate |
|
|
Amount
of
Registration
Fee |
|
|
Carry
Forward
Form
Type |
|
Carry
Forward
File Number |
|
Carry
Forward
Initial
Effective
Date |
|
Filing
Fee
Previously
Paid In Connection
with Unsold
Securities to
be Carried
Forward |
|
| Newly
Registered Securities |
|
|
| Fees to Be Paid |
|
Equity |
|
Common
Stock (1) (6) |
|
Rule
457(o) |
|
|
(2) |
|
|
|
(3) |
|
|
|
(3) |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
| |
|
Equity |
|
Preferred
Stock (1) |
|
Rule
457(o) |
|
|
(2) |
|
|
|
(3) |
|
|
|
(3) |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
| |
|
Other |
|
Warrants
(1) |
|
Rule
457(o) |
|
|
(2) |
|
|
|
(3) |
|
|
|
(3) |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
| |
|
Other |
|
Units
(1) |
|
Rule
457(o) |
|
|
(2) |
|
|
|
(3) |
|
|
|
(3) |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Unallocated (Universal)
Shelf |
|
— |
|
Rule
457(o) |
|
|
(2) |
|
|
|
(3) |
|
|
$ |
0 (5)
|
|
|
|
0.0001476 |
|
|
|
$
0.00 |
|
|
|
|
|
|
|
|
|
|
|
| Carry
Forward Securities |
|
|
| Carry Forward Securities |
|
Equity |
|
Common
Stock (6) |
|
Rule
415 (a)(6) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Equity |
|
Preferred
Stock |
|
Rule
415 (a)(6) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Other |
|
Warrants |
|
Rule
415 (a)(6) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Other |
|
Units |
|
Rule
415 (a)(6) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Unallocated
(Universal) Shelf |
|
— |
|
Rule
415 (a)(6) |
|
|
(5) |
|
|
|
|
|
|
$ |
200,000,000 |
|
|
|
|
|
|
|
|
|
|
S-3 |
|
333-251255 |
|
12/22/2020 |
|
$ |
21,820 |
|
| |
|
Total
Offering Amounts |
|
|
|
|
|
|
$ |
200,000,000 |
|
|
|
|
|
|
$ |
0 |
|
|
(5) |
|
|
|
|
|
|
|
|
| |
|
Total
Fees Previously Paid |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ |
21,820 |
|
|
|
|
|
|
|
|
|
|
|
| |
|
Total
Fee Offsets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N/A |
|
|
|
|
|
|
|
|
|
|
|
| |
|
Net
Fee Due |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$ |
0 |
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (1) |
Separate consideration may or may not be received for securities that are issuable upon the conversion or exercise of, or in exchange for, other securities offered hereby. |
| (2) |
There are being registered hereunder such indeterminate number of the securities of each identified class being registered as may be sold by the registrant from time to time at indeterminate prices, with the maximum aggregate public offering price not to exceed $200 million. Any securities registered hereunder may be sold separately or together with other securities registered hereunder. |
| (3) |
The proposed maximum per security and aggregate offering prices per class of securities will be determined from time to time by the Registrant in connection with the issuance by the Registrant of the securities registered hereunder and is not specified as to each class of security pursuant to Instruction 2.A.iii.b of the Instructions to the Calculation of Filing Fee Tables and related disclosure on Form S-3. |
|
(4)
|
Calculated pursuant to Rule 457(o) under the Securities
Act. |
|
(5) |
Pursuant
to Rule 415(a)(6) under the Securities
Act, the securities registered pursuant to this registration statement include $200,000,000 of unsold securities (the “Unsold Securities”)
previously registered pursuant to the Registration Statement on Form S-3 (File No. 333-251255), which initially
became effective upon filing on December 22, 2020 (the “Prior Registration Statement”). In connection with the filing
of the Prior Registration Statement, the registrant paid a filing fee of $21,820 associated with the offering of the Unsold Securities
(based on the filing fee rate in effect at the time of the filing of the Prior Registration Statement). The filing fee associated with
the offering of the Unsold Securities is hereby carried forward to be applied to the Unsold Securities registered hereunder, and no additional
filing fee is due with respect to the Unsold Securities in connection with the filing of this Registration Statement. The registrant is
not registering new securities (“New Securities”) on this registration statement Therefore no filing fee with respect to the
New Securities is being paid in connection with the filing of this registration statement. To the extent that, after the filing date hereof
and prior to the effectiveness of this registration statement, the registrant sells any Unsold Securities pursuant to the Prior Registration
Statement, the registrant will identify in a pre-effective amendment to this registration statement the updated number of Unsold
Securities from the Prior Registration Statement to be included in this registration statement pursuant to Rule 415(a)(6) and the updated
amount of New Securities, if any, to be registered on this registration statement. Pursuant to Rule 415(a)(6), the offering of securities
under the Prior Registration Statement will be deemed terminated as of the date of effectiveness of this registration statement. |
| (6) | This registration statement also covers the common stock purchase rights issuable in accordance with
the rights agreement, dated as of March 11, 2018, as amended by Amendment No. 1 thereto, dated March 8, 2019, Amendment No. 2
thereto, dated March 10, 2020, by and Amendment No. 3 thereto dated March 8, 2021, Amendment No. 4 thereto dated March 11, 2022,
Amendment No. 5 thereto dated March 11, 2023, and Amendment No. 6 thereto dated December 11, 2023 between the Company and Continental
Stock Transfer & Trust Company, as Rights Agent, which are presently attached to and trade with the Registrant’s common
stock. |
Nighthawk Biosciences (AMEX:NHWK)
Historical Stock Chart
From Mar 2024 to Apr 2024
Nighthawk Biosciences (AMEX:NHWK)
Historical Stock Chart
From Apr 2023 to Apr 2024