Filed
Pursuant to Rule 424(b)(4)
Registration
No. 333-272691
Prospectus
Bioxytran,
Inc.
18,578,982
Shares of Common Stock
This
Prospectus relates to the resale by the investors listed in the section of this Prospectus entitled “Selling Stockholders”
(the “Selling Stockholders”) of up to 18,578,982 shares (the “Shares”) of our Common Stock, par value $0.001
per share (“Common Stock”). The Shares consist of: (i) 11,000,000 shares of Common Stock (the “Offering Shares”)
to be issued as an indirect primary offering to the Selling Stockholder pursuant to a Closing Agreement, dated September 18, 2023, by
TRITON FUNDS LP (the “Closing Agreement”); (ii) 655,905 shares of Common Stock (the “Warrants”) issuable upon
exercise of the Walleye Note (below), to WallachBeth Capital, LLC and/or their affiliates pursuant to the Engagement Letter issued on
July 29, 2022; and (iii) 6,923,077 shares of Common Stock issuable upon the conversion of a $900,000 note with Walleye Opportunities
Master Fund Ltd. (the “Walleye Note”), originally issued on May 3, 2021, and amended on May 5, 2023.
We
are registering the resale of the Offering Shares as required by the Closing Agreement with TRITON FUNDS LP, the resale of the Warrants
pursuant to the terms of the Engagement Letter with WallachBeth Capital LLC, and the Debt Modification Agreement with Walleye Opportunities
Master Fund Ltd.
We
will not receive any of the proceeds from the sale of shares of the Securities by the Selling Stockholders. However, the Company will
receive the proceeds of the sale of the Purchase Shares pursuant to the Closing Agreement.
The
Selling Stockholders identified in this prospectus may offer the shares of the Securities from time to time through public or private
transactions at prevailing market prices or at privately negotiated prices. The Selling Stockholder can offer all, some or none of its
shares of the Securities, thus we have no way of determining the number of shares of Securities it will hold after this offering. See
“Plan of Distribution.”
The
Selling Stockholder is an “underwriter” within the meaning of Section 2(a)(11) of the Securities Act of 1933, as amended
(the “Securities Act”).
Our
registration of the Shares covered by this Prospectus does not mean that the Selling Stockholders will offer or sell any of the Shares.
The Selling Stockholders may sell the Shares covered by this Prospectus in a number of different ways and at varying prices. For additional
information on the possible methods of sale that may be used by the Selling Stockholders, you should refer to the section of this Prospectus
entitled “Plan of Distribution” beginning on page 68 of this Prospectus. We will not receive any of the proceeds from the
Shares sold by the Selling Stockholders, other than any proceeds from any cash exercise of the Warrants.
No
underwriter or other person has been engaged to facilitate the sale of the Shares in this offering. The Selling Stockholders may, individually
but not severally, be deemed to be an “underwriter” within the meaning of the Securities Act of 1933, as amended (the “Securities
Act”), of the Shares that they are offering pursuant to this Prospectus. We will bear all costs, expenses and fees in connection
with the registration of the Shares. The Selling Stockholders will bear all commissions and discounts, if any, attributable to their
respective sales of the Shares.
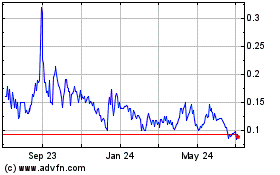
Our
Common Stock is quoted on OTCQB under the symbol BIXT. On September 18, 2023, the last reported sale price of our Common Stock as reported
on the OTCQB was $0.146 per share; however, we have a limited trading market for our stock and there is no assurance that a trading market
will develop, or, if developed, that it will be sustained. Consequently, a purchaser of our Common Stock may find it difficult to resell
the securities offered herein should the purchaser desire to do so.
On
April 15, 2020, the Securities and Exchange Commission (the “SEC”) issued a temporary order of trading suspension pertaining
to the Company’s securities (the “Suspension”); See SEC Release 34-88656. By its terms the Suspension terminated
on April 29, 2020.
The
Company’s stock was, at the time, quoted on the OTCQB. When an SEC trading suspension ends, a broker-dealer generally may not solicit
investors to buy or sell the previously-suspended over-the-counter (“OTC”) stock until certain requirements are met. Before
soliciting quotations or resuming quotations in an OTC stock that has been subject to a trading suspension, a broker-dealer must file
a Form 211 with the Financial Industry Regulatory Authority (“FINRA”) representing that it has satisfied all applicable requirements,
including those of Rule 15c2-11 and FINRA Rule 6432.
Among
other things, Rule 15c2-11 requires broker-dealers to review and maintain certain documents and information about the company, including
in certain cases:
| |
1. |
the
company’s state of organization, business line, and names of certain control affiliates |
| |
2. |
the
title and class of the securities outstanding; and |
| |
3. |
the
company’s most recent balance sheet and its profit and loss and retained earnings statement. |
No
broker-dealer may solicit or recommend that an investor buy an OTC stock that has been subject to a trading suspension unless and until
FINRA has approved a Form 211 relating to the stock, or if there are continuing regulatory concerns about the company, its disclosures,
or other factors, such as a pending regulatory investigation.
On
March 30, 2022, the Company received the FINRA Clearance Letter and on June 14, 2022, OTC Market Group agreed to the removal of the Caveat
Emptor symbol. The Company was approved for quotation on the OTCQB on September 28, 2022. We cannot assure you that an active public
market for our Common Stock will develop or that the market price of our shares will not decline below the Proposed Maximum Offering
Price. The Proposed Maximum Offering Price of our shares may not be indicative of prices that will prevail in the trading market following
the offering.
The
Common Stock and the Common Stock underlying the Warrants are currently eligible for resale under Rule 144.
The
Selling Stockholders may be deemed to be an “underwriter” within the meaning of the Securities Act of 1933, as amended (the
“Securities Act”), in connection with the resale of the Common Stock offered pursuant to this Prospectus.
This
investment involves a high degree of risk. You should purchase shares of Common Stock only if you can afford a complete loss. See
“Risk Factors” beginning on page 6 to read about factors you should consider before investing in shares of our Common
Stock.
The
information contained in this Prospectus is accurate only as of the date of this Prospectus, regardless of the time of delivery of this
Prospectus or of any sale of our Common Stock. Neither the delivery of this Prospectus nor any distribution of securities in accordance
with this Prospectus shall, under any circumstances, imply that there has been no change in our affairs since the date of this Prospectus.
This Prospectus will be updated and made available for delivery to the extent required by the federal securities laws.
NEITHER
THE SECURITIES AND EXCHANGE COMMISSION NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED OF THESE SECURITIES OR PASSED
UPON THE ACCURACY OR ADEQUACY OF THIS PROSPECTUS. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENSE.
THE
DATE OF THIS PROSPECTUS IS OCTOBER 2, 2023
TABLE
OF CONTENTS
PROSPECTUS
SUMMARY
This
summary highlights selected information contained elsewhere in this Prospectus. To understand this offering fully, you should read the
entire Prospectus carefully, including the “Risk Factors” section, the financial statements and the notes to the financial
statements. Unless the context otherwise requires, references contained in this Prospectus to the “Company,” “we,”
“us,” or “our” refers to Bioxytran, Inc.
Bioxytran,
Inc. (“we”, “us”, or the “Company”) is a clinical stage pharmaceutical company focused on the development,
manufacture and commercialization of therapeutic drugs designed to address hypoxia (a lack of oxygen to tissues) in humans. Hypoxia needs
to be addressed quickly, otherwise it results in necrosis, the death of cells comprising body tissue. Necrosis cannot be reversed. Our
lead drug candidate, a hypoxia platform technology with code-name BXT-25, is planned to be an Acellular Oxygen Carrier (“AOC”)
consisting of bovine hemoglobin stabilized with a co-polymer. BXT-25’s intended application includes the treatment of hypoxic conditions
in the brain resulting from stroke. We believe that our approach is novel when applied to hypoxic conditions in humans. Our drug development
efforts are guided by specialists who work on co-polymer chemistry and other disciplines. We intend to supplement our efforts with input
from a scientific and medical advisory board whose members are leading physicians.
We
plan to initiate pre-clinical studies of BXT-25. However, we cannot provide any assurance that we will successfully initiate or complete
those planned trials and be able to initiate any other clinical trials for BXT-25 or any of our future drug candidates.
At
the issuance of the 11,000,000 shares in the Closing Agreement, and if all of the 655,905 Common Stock underlying the Warrants being
offered for sale in this offering are exercised, and if all the 6,923,077 Common Stock underlying the Walleye Note are converted, we
will have 174,244,388 shares outstanding, which does not include shares of Common Stock issuable under our 2021 Stock Incentive Plan,
but includes the 11,880,421 shares of our Common Stock issuable to our other Note holder (assuming conversion at the end of the holding
period), warrants and options.
This
offering will terminate twelve (12) months from the date of the effective date of this registration statement unless terminated earlier
by the Company.
The
Company was organized on June 9, 2008, as a Nevada corporation.
Company
Overview
Our
former name was U.S. Rare Earth Minerals, Inc. or USREM. On September 21, 2018, the Company was reorganized after reaching a settlement
with a secured creditor with respect to a 6% secured promissory note in the principal amount of $110,000, including all interest due
thereon, which had been in default since August 23, 2013. The note was secured by substantially all of the assets of the Company. As
a condition to the settlement of the outstanding debt, USREM, agreed to acquire Bioxytran, Inc., a Delaware Corporation, or Bioxytran
(Delaware) and divest substantially all of its assets and remaining liabilities to an affiliate of the creditor and former majority stockholder.
The creditor agreed to an accord and satisfaction of the Company’s obligations to the creditor in full and to release all liens
upon the completion of the transaction.
The
Agreement and Plan of Merger and Reorganization by and among USREM, Bioxy Acquisition Corp., a Wyoming corporation and wholly owned subsidiary
of USREM, and Bioxytran (Delaware) was entered into contemporaneously with the settlement and all of the transactions contemplated by
the settlement were consummated on September 21, 2018. Our operations are conducted within Bioxytran (Delaware).
On
November 7, 2018, U.S. Rare Earth Minerals, Inc. changed its name to Bioxytran, Inc.
We
are a clinical stage pharmaceutical company focusing on the development, manufacture and commercialization of therapeutic drugs designed
to address hypoxia (a lack of oxygen to tissues) in humans. Our initial focus is the treatment of hypoxic conditions in the brain resulting
from stroke. The Company’s Subsidiary, Pharmalectin, Inc. (“Pharmalectin”), is developing a glyco-viral pharmaceutical,
with emphasis on treating Covid-19.
Currently,
our lead pharmaceutical drug candidate in Bioxytran code named BXT-25, is planned to be an Acellular Oxygen Carrier (“AOC”)
consisting of bovine hemoglobin stabilized with a co-polymer. This modified hemoglobin will be designed to be an injectable intravenous
drug and we plan to begin pre-clinical studies and apply to the Food and Drug Administration (FDA) for approval to use BXT-25 to prevent
necrosis, by carrying oxygen to human tissue when blood flow to the brain is blocked during the initial stages of stroke in adults.
If
we successfully complete Phase I testing with the FDA, we plan to explore the use of additional drug candidates using chemical structures
that are a sub-class of BXT-25 that share the same physical properties, to treat wound healing due to hypoxia, cardiovascular ischemia,
anemia, cancer conditions and trauma, subject to FDA approval.
BXT-25
is based in part on a technology developed by the Biopure Corporation which separates the hemoglobin molecule from red blood cells. Biopure
filed for bankruptcy in 2009 and the technology we use from Biopure is in the public domain. We plan to apply our proprietary chemistry
to enhance the hemoglobin molecule to produce BXT-25 which is hemoglobin and co-polymer based. BXT-25 is a novel, unproven technology.
We may be unsuccessful in developing this technology into drugs ultimately dependent on FDA approval.
On
April 19, 2023, the Company announced that its long-awaited AOC BXT-25 has been successfully tested in animals. The initial results are
very encouraging because they show the non-toxicity of the experimental drug, along with the corresponding full recovery in Swiss Albino
mice, in an experiment carried out in a joint venture with NDPD Pharma, Inc. As a next step, the Company intends to proceed with a 14-day
repeated dose toxicity study using New Zealand Rabbits and Wistar Rats as funding permits.
If
successful in our initial trials, we will need to raise additional funds in order to expand the use to BXT-25 and new indications.
The
lead pharmaceutical drug candidate of Pharmalectin, code named ProLectin, is a complex galectin antagonist that binds to, and blocks
the activity of galectin-3, a type of galectin. Galectins are a member of a family of proteins in the body called lectins. Lectins interact
with carbohydrate sugars located in, on the surface of, and in between cells. This interaction causes the cells to change behavior, including
cell movement, multiplication, and other cellular functions. The interactions between lectins and their target carbohydrate sugars occur
via a carbohydrate recognition domain, or CRD, within the lectin. Galectins are a subfamily of lectins that have a CRD that bind specifically
to ß-galactoside proteins. Galectins have a broad range of functions, including regulation of cell survival and adhesion, promotion
of cell-to-cell interactions, growth of blood vessels, regulation of the immune response and inflammation. During viral infections galectins
are upregulated and downregulated based on the type of virus.
Pharmalectin
has an exclusive license for an earlier developed molecule, for treatment of mild to moderate COVID-19. The molecule, code name ProLectin-M,
is currently in the process of being cleared for clinical trials in India and the U.S. Based on the results of the initial human trials,
the technology, developed by NDPD Pharma, Inc., indicates the possibility for development of a new molecule that serves as a proof of
concept in the form of an oral treatment for mild to moderate conditions of the disease. This potential treatment complements the Company’s
intravenous drug candidates, currently under development, for treatment of more severe conditions for treatment of the SARS-Cov-2 virus
and developed from carbohydrate technologies.
In
the past, galectin antagonists have been used as a fibrosis drug and a cancer drug. It is currently being reformulated to treat viral
infections. We believe that we have a novel approach in treating viral infections in humans. Our drug development efforts are guided
by specialists on carbohydrate chemistry and other disciplines, and we intend to supplement our efforts with input from a scientific
and medical advisory board whose members are leading physicians.
A
Proof-of-Concept trial was approved by the IRB at Mazumdar Shaw Medical Center, Narayana Health in Bangalore, India. The results of the
trial are described in our three peer-reviewed articles Galectin antagonist use in mild cases of SARS-CoV-2; pilot feasibility randomised,
open label, controlled trial, published in Journal of Vaccines & Vaccination on December 30, 2020, Carbohydrate ProLectin-M,
a Galectin-3 Antagonist, Blocks SARS-CoV-2 Activity published in the International Journal of Health Sciences on June 30, 2022 and
PLG-007 and Its Active Component Galactomannan-α Competitively Inhibit Enzymes That Hydrolyze Glucose Polymers published
in the International Journal of Molecular Science on July 13, 2022.
Results
from our latest Phase 2 trial on COVID-19 patients conducted at ESIS Medical College and Hospital, Sanath Nagar, Hyderabad, India were
published in in the peer-reviewed journal Virus: An Oral Galectin Antagonist in COVID-19—A Phase II Randomized Controlled Trial
on February 23, 2023, show positive results of its randomized, placebo-controlled Phase 2 clinical trial in thirty-four (34) patients
with mild-to-moderate COVID-19. During the seven (7) days of treatment, an orally administered Galectin Antagonist in the form of a chewable
tablet was administered eight (8) times per day on an hourly basis. The endpoint was a statistically significant reduction in viral load
measured by the number of patients reaching a below threshold PCR value (Ct value ≥ 29) by day 7. The trial met its endpoint with
a one hundred percent (100%) response rate by day 7 versus six percent (6%) in placebo, which was statistically significant (p-value
= .001). Our analysis also revealed an 82% response rate by day 3, which was statistically significant (p-value = .001). There were no
drug-related serious adverse events (SAE’s) in the patient population or viral rebounds by day fourteen (14) in the patient population.
On
December 2, 2022, India’s Central Drugs Standard Control Organisation (CDSCO) issued an IND with permission to conduct: “A
Phase 1b/2a Randomized, Blinded, placebo-controlled Study in Participants with Mild to Moderate COVID-19 to Evaluate the Safety, Efficacy,
and Pharmacokinetics of Orally Administered ProLectin-M”. The trial is planned to start on, or around, October 1, 2023.
On
August 21, 2023, the Company’s IND #153742 under the title “PROTECT: ProLectin-M, a nucleocapsid TErminal GaleCTin antagonist
for COVID-19 (PROTECT), a Randomized, Double-blinded Clinical Trial to Evaluate the Efficacy and Safety in Non-Hospitalized Adult Participants
with COVID-19” was approved by the FDA. The trial is expected to start in the first quarter of 2024, provided we obtain adequate
funding.
On
January 27, 2023, an additional IND with the CDSCO was issued for ProLectin-I for an “IV treatment of SARS-CoV-2 in hospitalized
patients with moderate Covid-19 infections and for Long Covid”, and for ProLectin-F for “treatment of lung-fibrosis as a
result of use of ventilator”.
On
April 19, 2023, the Company announced that its long awaited Acelluar Oxygen Carrier (“AOC”) BXT-25 had been successfully
tested in animals. The initial results are very encouraging because they show the non-toxicity of the experimental drug, along with the
corresponding full recovery in Swiss Albino mice, in an experiment carried out in a joint venture with NDPD Pharma, Inc. As a next step,
the Company intends to proceed with a 14-day repeated dose toxicity study using New Zealand Rabbits and Wistar Rats as funding permits.
BXT-25
and ProLectin are novel, unproven technologies. We may be unsuccessful in developing this technology into drugs which the FDA ultimately
will approve.
Our
foreign subsidiary, Pharmalectin (BVI), Inc. (“Pharmalectin BVI” or the “Foreign Subsidiary”) is the owner and
custodian of the Company’s Copyrights, Trademarks, Patents and Licenses.
Our
subsidiary, Pharmalectin India Pvt Ltd. (“Pharmalectin India”) is managing the Company’s local clinical research and
trials, and holds the local commercialization rights.
Our
independent registered accounting firm noted in its report accompanying our financial statements for the period ending December 31, 2022,
that the Company’s limited resources and operating history, as well as operating losses raise substantial doubt about the Company’s
ability to continue as a going concern. As of June 30, 2023, we had a cumulative net loss of thirteen million four hundred thirteen thousand
nine hundred one dollars ($13,413,901), while the Company had forty-five thousand one hundred eighty-one dollars ($45,181) cash on hand.
We
do not currently have sufficient capital resources to fund operations. To stay in business and to continue the development of our products,
we will need to raise additional capital through public or private sales of our securities, debt financing or short-term bank loans,
or a combination of the foregoing. We believe that we must raise not less than three million seven hundred thousand dollars ($3,700,000)
in the current offering in addition to current cash on hand to be able to continue our business operations for approximately the next
fifteen (15) months; however, funding at any level lower than five million three hundred thousand dollars ($5,300,000) will delay the
development of our technology and business.
We
have not applied to register the shares in any state. An exemption from registration will be relied upon in the states where the shares
are distributed and may only be traded in such jurisdictions after compliance with applicable securities laws. There can be no assurances
that the shares will be eligible for sale or resale in such jurisdictions. We may apply to register the shares in several states for
secondary trading; however, we are under no requirement to do so.
Our
current officers are David Platt, Mike Sheikh and Ola Soderquist. We are dependent upon these officers for implementation and execution
of our business plan. The loss of any of them could have a material adverse effect upon our results of operations and financial position
and could delay or prevent the achievement of our business objectives.
Recent
Equity Financing
On
November 28, 2022, one hundred fifty-six thousand two hundred fifty (156,250) shares of Common Stock were sold in a private placement
for the amount of fifty thousand and No/100 dollars ($50,000.00), or $0.32/share
On
December 29, 2022, ninety-three thousand seven hundred fifty (93,750) shares of Common Stock were sold in a private placement for the
amount of thirty thousand and No/100 dollars ($30,000.00), or $0.32/share
On
February 10, 2023, one hundred fifty-six thousand two hundred fifty (156,250) shares of Common Stock were sold in a private placement
for the amount of fifty thousand and No/100 dollars ($50,000.00), or $0.32/share
On
April 14, 2023, one hundred thirty-seven thousand six hundred fifty-six (137,656) shares of Common Stock were issued against supplier
invoices amounting to forty-four thousand fifty and No/100 dollars ($44,050.00), or $0.32/share
On
April 14, 2023, six million seven hundred sixty-three thousand five hundred sixty-two (6,763,562) shares of Common Stock were issued
to offset an affiliate against invoices paid on behalf of the Company and accrued salaries to our Officers, for a total value of two
million one hundred sixty-four thousand three hundred forty and No/100 dollars ($2,164,340.00), or $0.32/share
On
May 12, 2023, one hundred fourteen thousand two hundred eighty-six (114,286) shares of Common Stock were sold in a private placement
for the amount of forty thousand and No/100 dollars ($40,000.00), or $0.32/share
On
July 24, 2023, five hundred thousand (500,000) shares of Common Stock were sold in a private placement for the amount of one hundred
thousand and No/100 dollars ($100,000.00), or $0.20/share
On
August 21, 2023, one million six hundred twelve nine hundred three (1,612,903) shares of Common Stock were sold on a prior S-1 for the
amount of one hundred forty-five thousand one hundred sixty-one and 27/100 dollars ($145,161.27), or $0.09/share
On
August 21, 2023, one million six hundred thousand (1,600,000) shares of Common Stock were exchanged for invoices in the amount of one
hundred forty-five thousand and No/100 dollars ($145,000.00), or $0.09/share
On
August 25, 2023, five hundred thousand one hundred eighty-six (505,186) shares of Common Stock were sold in a private placement for the
amount of sixty-eight thousand two hundred and No/100 dollars ($68,200.00), or $0.135/share
On
September 14, 2023 five million eight hundred twenty-four thousand seven hundred forty-one (5,824,741) shares of Common Stock were exchanged
by the Company’s officers for invoices and salary past due in the amount of seven hundred eighty-six thousand three hundred forty
and No/100 dollars ($786,340.00), or $0.135/share
The
Company claims an exemption from the registration requirements of the Securities Act of 1933 (the “Securities Act”) for the
private placement of these securities pursuant to Section 4(a)(2) of the Securities Act and/or Rule 506 of Regulation D promulgated under
the Securities Act.
Note
Financing
A
summary of the outstanding convertible notes at September 18, 2023, are as follows:
| Debtor | |
Date of Issuance | | |
Principal Amount | | |
Interest Rate | | |
Accrued Interest | | |
Total Amount | | |
Maturity Date |
| Private Placement, 2021 Note 1 | |
| 5/3/2021 | | |
$ | 1,000,000 | | |
| 6 | % | |
$ | 142,356 | | |
$ | 1,142,356 | | |
8/31/2023 |
| Private Placement, 2021 Note 2 | |
| 5/3/2021 | | |
| 900,000 | | |
| 10 | % | |
| 37,430 | | |
| 937,430 | | |
4/30/2024 |
| Total Notes Due | |
| | | |
$ | 1,900,000 | | |
| | | |
$ | 179,786 | | |
$ | 2,079,786 | | |
|
Note
Holders
Around
April 29, 2021, we entered into four (4) Securities Purchase Agreements, or “the 2021 SPA’s”, under which we agreed
to sell convertible promissory notes, “the Notes”, in an aggregate principal amount of two million one hundred sixty-five
thousand and No/100 dollars ($2,165,000.00); one million ($1,000,000.00) with six percent (6%) interest and million one hundred sixty-five
thousand and No/100 dollars ($1,165,000.00) with ten percent (10%) interest to the debtors, as shown in the table above. A note of sixty-five
thousand and No/100 dollars ($65,000.00) was converted on May 17, 2023, and a note of one hundred thousand and No/100 dollars ($100,000.00)
was converted on June 28, 2023. An additional $100,000 was drawn from a $1,000,000 note was drawn on August 30, 2023.
At
any time after the issue date of the Notes, the holders of the Notes, (“the Holders”), have the option to convert all or
any part of the outstanding and unpaid principal amount and accrued and unpaid interest of the Notes into shares of our Common Stock
at the Conversion Price. The “Conversion Price” will be the lesser of (i) thirteen cents ($0.13) per share or (ii) eighty-five
percent (85%) of the closing price of Any Qualified Financing, which consists of any fundraising receiving gross proceeds of not less
than five hundred thousand dollars ($500,000).
If
the Notes are converted prior to us paying off such Note, it would lead to substantial dilution to our Shareholders as a result of the
conversion discounted for the Notes. There can be no assurance that there will be any funds available to pay of the Notes, or if available,
on terms that will be acceptable to us or our Shareholders. If we fail to obtain such additional financing on a timely basis, the Holders
may convert the Notes and sell the underlying shares, which may result in significant dilution to Shareholders due to the conversion
discount, as well as a significant decrease in our stock price.
As
at September 18, 2023, the principal amount of debt to the Holders of the 2021 Notes is one million nine hundred thousand and No/100
dollars ($1,900,000.00) and the interest mounts to one hundred seventy-nine thousand seven hundred eighty-six and No/100 dollars ($179,786.00).
The interest mounts to $274/day.
Recent
Conversions of Notes and Warrants
A
note of sixty-five thousand and No/100 dollars ($65,000.00) and two thousand eight hundred and seventy-nine and No/100 dollars ($2,879.00)
in interest was on May 17, 2023, converted into five hundred thirty-tree thousand one hundred thirty-eight (533,138) shares and registered
as subject to Rule 144.
A
note of one hundred thousand and No/100 dollars ($100,000.00) and four thousand four hundred and twenty-eight and No/100 dollars ($4,428.00)
in interest was on June 28, 2023, converted into eight hundred three thousand two hundred ninety-two (803,292) shares and registered
as subject to Rule 144.
A
note of one hundred thousand and No/100 dollars ($100,000.00) and forty-four thousand two hundred and eighty-two and No/100 dollars ($44,282.00)
in interest was on August 30, 2023, converted into one million one hundred nine thousand eight hundred sixty-one (1,109,861) shares and
registered as subject to Rule 144.
RISK
FACTORS
An
investment in our Common Stock involves substantial risks, including the risks described below. You should carefully consider the risks
described below before purchasing our Common Stock. The risks highlighted here are not the only ones that we may face. For example, additional
risks presently unknown to us or that we currently consider immaterial or unlikely to occur could also impair our operations. If any
of the risks or uncertainties described below or any such additional risks and uncertainties actually occur, our business, prospects,
financial condition or results of operations could be negatively affected, and you might lose all or part of your investment.
Risks
Related to Our Business
Our
plan relies upon our ability to obtain additional sources of capital and financing. If the amount of capital we are able to raise from
financing activities, together with our revenues from operations, is not sufficient to satisfy our capital needs, we may be required
to cease operations.
To
become and remain profitable, we must succeed in developing and commercializing products that generate significant income. This will
require us to be successful in a range of challenging activities, including completing preclinical testing and clinical trials of our
drug candidates, discovering additional drug candidates, obtaining regulatory approval for these drug candidates, manufacturing, marketing
and selling any products for which we may obtain regulatory approval, and establishing and managing our collaborations at various stages
of each candidate’s development. We are only in the preliminary stages of these activities. We may never succeed in these activities
and, even if we do, may never generate income that is significant enough to achieve profitability.
Because
of the numerous risks and uncertainties associated with pharmaceutical product development, we are unable to accurately predict the timing
or amount of increased expenses or when, or if, we will be able to achieve profitability. If we are required by the U.S. Food and Drug
Administration, or FDA, or the European Medicines Agency, or EMA, to perform studies in addition to those currently expected, or if there
are any delays in completing our clinical trials or the development of any of our drug candidates, our expenses could increase, and revenue
could be further delayed.
Even
if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to
become and remain profitable would depress the value of our Company and could impair our ability to raise capital, expand our business,
maintain the research and development efforts that will be initially funded by the proceeds of this offering, diversify our product offerings
or even continue our operations. A decline in the value of our Company could also cause you to lose all or part of your investment.
On
April 15, 2020 the SEC ordered a 10-day trading suspension of the Company’s Common Stock. A repeat suspension could occur.
The
SEC suspended trading of the Company’s Common Stock due to “questions that have been raised about the accuracy and adequacy
of information in the marketplace relating to BIXT common stock [including] statements that BIXT made in press releases, blog posts,
and a podcast about BIXT’s present ability to develop a drug to mitigate or treat COVID-19.” Because the Company was not
in direct communication with SEC Bioxytran was unable to ascertain the specifics related to “blog posts” and “podcasts”
that contributed to the suspension being ordered. In order to avoid receiving an additional suspension the Company has taken remediate
steps by developing written policies for;
| |
● |
external
communications, |
| |
● |
social
media and |
| |
● |
networking |
Additionally,
the Company has since completely changed its website, hired new SEC counsel and instituted a simple process requiring multiple levels
of review (including by counsel) before any press release is published.
The
suspension of trading of the Company’s stock was part of a larger Securities and Exchange Commission (“SEC”) crackdown
on companies making “COVID-19 claims”. In the 2020 timeframe there were thirty-eight (38) COVID-19 related suspensions.
The
Company believes that the completion of two human clinical trials in COVID-19 which were published in peer-reviewed journals, indicating
Bioxytran’s ability to potentially develop COVID-19 anti-viral compounds:
●
Galectin antagonist use in mild cases of SARS-CoV-2; pilot feasibility randomised, open label, controlled trial, published in Journal
of Vaccines & Vaccination on December 30, 2020; and
●
An Oral Galectin Antagonist in COVID-19—A Phase II Randomized Controlled Trial published in the journal Virus on February 23, 2023.
Currently,
the Company has received three approvals to conduct clinical human trials:
●
On December 2, 2022, India’s Central Drugs Standard Control Organisation (CDSCO) issued an IND with permission to conduct: “A
Phase 1b/2a Randomized, Blinded, placebo-controlled Study in Participants with Mild to Moderate COVID-19 to Evaluate the Safety, Efficacy,
and Pharmacokinetics of Orally Administered ProLectin-M”;
●
On August 21, 2023 the Company’s IND #153742 under the title “PROTECT: ProLectin-M, a nucleocapsid TErminal GaleCTin antagonist
for COVID-19 (PROTECT), a Randomized, Double-blinded Clinical Trial to Evaluate the Efficacy and Safety in Non-Hospitalized Adult Participants
with COVID-19” was approved by the FDA, the trial is expected to start in the first quarter of 2024, provided we obtain adequate
funding; and
●
On January 27, 2023, an additional IND with the CDSCO was issued for ProLectin-I for an “IV treatment of SARS-CoV-2 in hospitalized
patients with moderate Covid-19 infections and for Long Covid”, and for ProLectin-F “for treatment of lung-fibrosis as a
result of use of ventilator”.
We
have incurred losses since our inception and expect to incur losses for the foreseeable future and may never achieve or maintain profitability.
As
at June 30, 2023, we have incurred losses since inception and have an accumulated deficit of thirteen million four hundred thirteen thousand
nine hundred one dollars ($13,413,901) and, we had approximately forty-five thousand one hundred eighty-one dollars ($45,181) of cash
on hand. The report of our independent registered public accountants as of and for years ending December 31, 2022 and 2021, contained
an explanatory paragraph regarding substantial doubt about our ability to continue as a going concern. Our ability to continue as a going
concern is dependent upon our ability to generate revenue and raise capital from financing transactions. Management anticipates that
our cash resources are not sufficient to continue operations until additional cash investments are secured. The future of the Company
is dependent upon its ability to obtain financing and upon future profitable operations from the development of its new business opportunities.
There can be no assurance that we will be successful in accomplishing its objectives. Without such additional capital, we may be required
to curtail or cease operations.
We
have a limited operating history, which makes it difficult to evaluate our current business and future prospects.
We
are a company with limited operating history, and our operations are subject to all of the risks inherent in establishing a new business
enterprise. The likelihood of our success must be considered in light of the problems, expenses, difficulties, complications and delays
frequently encountered in connection with the formation of a new business, the development of new technologies or those subject to clinical
testing, and the competitive and regulatory environment in which we will operate. We may never obtain FDA or EMA approval of our products
in development and, even if we do so and are also able to commercialize our products, we may never generate revenue sufficient to become
profitable. Our failure to generate revenue and profit would likely cause our securities to decrease in value or become worthless.
We
will require additional financing to implement our business plan, which may not be available on favorable terms or at all, and we may
have to accept financing terms that would place restrictions on us.
We
believe that we must raise not less than three million seven hundred thousand and No/100 Dollars ($3,700,000) in the current offering
in addition to current cash on hand to be able to continue our business operations for approximately the next fifteen (15) months; however,
funding at any level lower than five million three hundred thousand and No/100 Dollars ($5,300,000) over the next year will delay the
development of our technology and business. We will need to continue to conduct significant research, development, testing and regulatory
compliance activities for BXT-25, together with projected general and administrative expenses, we expect will result in operating losses
for the foreseeable future. We may not be able to obtain equity or debt financing on acceptable terms or at all to implement our growth
strategy. As a result, adequate capital may not be available to finance our current development plan, take advantage of business opportunities
or respond to competitive pressures. If we are unable to raise additional funds, we may be forced to curtail or even abandon our business
plan.
Until
such time, if ever, as we can generate substantial product income, we expect to finance our cash needs through a combination of equity
offerings, debt financings and license and collaboration agreements. To the extent that we raise additional capital through the sale
of equity or convertible debt securities, the ownership interest of existing stockholders will be diluted, and the terms of these securities
may include liquidation or other preferences that adversely affect the rights of Common Stockholders. In addition, the terms of any future
financings may impose restrictions on our right to declare dividends or on the manner in which we conduct our business. Debt financing
and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take
specific actions, such as incurring additional debt, making capital expenditures, declaring dividends, or making acquisitions or significant
asset sales.
If
we raise additional funds through collaborations, strategic alliances, marketing, distribution or licensing arrangements with third parties,
we may have to relinquish valuable rights to our technologies, future revenue streams, research programs, drug candidates or grant licenses
on terms that may not be favorable to us and/or that may reduce the value of our Common Stock.
Our
products are based on novel, unproven technologies.
Our
drug candidates in development are based on novel, unproven technologies using proprietary co-polymer compounds in combination with similar
FDA approved drug for veterinary use. Co-polymers are difficult to synthesize, and we may not be able to synthesize co-polymer that will
be usable as delivery vehicles for the anti-hypoxia drugs we are working with or other therapeutics we intend to develop. Clinical trials
are expensive, time-consuming and may not be successful. They involve the testing of potential therapeutic agents, or effective treatments,
in humans, typically in three phases, to determine the safety and efficacy of the products necessary for an approved drug. Many products
in human clinical trials fail to demonstrate the desired safety and efficacy characteristics. Even if our products progress successfully
through initial or subsequent human testing, they may fail in later stages of development. We may engage others to conduct our clinical
trials, including clinical research organizations and, possibly, government-sponsored agencies. These trials may not start or be completed
as we forecast or may not achieve desired results.
Clinical
drug development involves a lengthy and expensive process, with an uncertain outcome. We may incur additional costs or experience delays
in completing, or ultimately be unable to complete, the development and commercialization of our drug candidates.
Our
drug candidate is unproven, and its risk of failure is high. It is impossible to predict when or if our current or any future drug candidates
will receive regulatory approval or prove effective and safe in humans. Before obtaining marketing approval from regulatory authorities
for the sale of any drug candidate, we must conduct extensive clinical trials and, in the case of BXT-25, first complete preclinical
development, to demonstrate the safety and efficacy of our drug candidates in humans. Clinical testing is expensive, difficult to design
and implement, can take many years to complete and is uncertain as to outcome. A failed clinical trial can occur at any stage of testing.
The outcome of preclinical testing and early clinical trials may not be predictive of the success of later clinical trials, and interim
results of a clinical trial do not necessarily predict final results. Moreover, preclinical and clinical data are often susceptible to
varying interpretations and analyses, and many companies that have believed their drug candidates performed satisfactorily in preclinical
studies and clinical trials have nonetheless failed to obtain marketing approval of their products.
We
may experience numerous unforeseen events during, or as a result of, clinical trials that could delay or prevent our ability to receive
marketing approval or commercialize our drug candidates, including:
| |
● |
regulators
or institutional review boards may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial
at a prospective trial site; |
| |
|
|
| |
● |
we
may experience delays in reaching, or fail to reach, agreement on acceptable clinical trial contracts or clinical trial protocols
with prospective trial sites; |
| |
|
|
| |
● |
clinical
trials of our drug candidates may produce negative or inconclusive results, and we may decide, or regulators may require us, to conduct
additional clinical trials or abandon product development programs; |
| |
|
|
| |
● |
the
number of patients required for clinical trials of our drug candidates may be larger than we anticipate, enrollment in these clinical
trials may be slower than we anticipate, or participants may drop out of these clinical trials at a higher rate than we anticipate; |
| |
|
|
| |
● |
our
third-party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner,
or at all; |
| |
|
|
| |
● |
we
may have to suspend or terminate clinical trials of our drug candidates for various reasons, including a finding that the participants
are being exposed to unacceptable health risks; |
| |
|
|
| |
● |
regulators
or institutional review boards may require that we or our investigators suspend or terminate clinical research for various reasons,
including noncompliance with regulatory requirements or a finding that the participants are being exposed to unacceptable health
risks; |
| |
|
|
| |
● |
the
cost of clinical trials of our drug candidates may be greater than we anticipate; |
| |
|
|
| |
● |
the
supply or quality of our drug candidates or other materials necessary to conduct clinical trials of our drug candidates may be insufficient
or inadequate; |
| |
|
|
| |
● |
our
drug candidates may have undesirable side effects or other unexpected characteristics, causing us or our investigators, regulators
or institutional review boards to suspend or terminate the trials; and |
| |
|
|
| |
● |
regulators
may revise the requirements for approving our drug candidates, or such requirements may not be as we anticipate. |
If
we are required to conduct additional clinical trials or other testing of our drug candidates beyond those that we currently contemplate,
if we are unable to successfully complete clinical trials of our drug candidates or other testing, if the results of these trials or
tests are not positive or are only modestly positive or if there are safety concerns, we may:
| |
● |
be
delayed in obtaining marketing approval for our drug candidates; |
| |
|
|
| |
● |
not
obtain marketing approval at all, which would seriously impair our viability; |
| |
|
|
| |
● |
obtain
marketing approval in some countries and not in others; |
| |
|
|
| |
● |
obtain
approval for indications or patient populations that are not as broad as we intend or desire; |
| |
|
|
| |
● |
obtain
approval with labeling that includes significant use or distribution restrictions or safety warnings; |
| |
|
|
| |
● |
be
subject to additional post-marketing testing requirements; or |
| |
|
|
| |
● |
have
the product removed from the market after obtaining marketing approval. |
A
Proof-of-Concept trial was approved by the IRB at Mazumdar Shaw Medical Center, Narayana Health in Bangalore, India. The results of the
trial are described in our three peer-reviewed articles Galectin antagonist use in mild cases of SARS-CoV-2; pilot feasibility randomised,
open label, controlled trial, published in Journal of Vaccines & Vaccination on December 30, 2020, Carbohydrate ProLectin-M,
a Galectin-3 Antagonist, Blocks SARS-CoV-2 Activity published in the International Journal of Health Sciences on June 30, 2022 and
PLG-007 and Its Active Component Galactomannan-α Competitively Inhibit Enzymes That Hydrolyze Glucose Polymers published
in the International Journal of Molecular Science on July 13, 2022.
Results
from our latest Phase 2 trial on COVID-19 patients conducted at ESIS Medical College and Hospital, Sanath Nagar, Hyderabad, India were
published in in the peer-reviewed journal Virus: An Oral Galectin Antagonist in COVID-19—A Phase II Randomized Controlled Trial
on February 23, 2023, show positive results of its randomized, placebo-controlled Phase 2 clinical trial in 34 patients with mild-to-moderate
COVID-19. During the 7 days of treatment, an orally administered Galectin Antagonist in the form of a chewable tablet was administered
8 times per day on an hourly basis. The endpoint was a statistically significant reduction in viral load measured by the number of patients
reaching a below threshold PCR value (Ct value ≥ 29) by day 7. The trial met its endpoint with a 100% response rate by day 7 versus
6% in placebo, which was statistically significant (p-value = .001). Our analysis also revealed an 82% response rate by day 3, which
was statistically significant (p-value = .001). There were no drug-related serious adverse events (SAE’s) in the patient population
or viral rebounds by day 14 in the patient population.
On
December 2, 2022, India’s Central Drugs Standard Control Organisation (CDSCO) issued an IND with permission to conduct: “A
Phase 1b/2a Randomized, Blinded, placebo-controlled Study in Participants with Mild to Moderate COVID-19 to Evaluate the Safety, Efficacy,
and Pharmacokinetics of Orally Administered ProLectin-M”. The trial is planned to start on, or around, October 1, 2023.
On
August 21, 2023 the Company’s IND #153742 under the title “PROTECT: ProLectin-M, a nucleocapsid TErminal GaleCTin antagonist
for COVID-19 (PROTECT), a Randomized, Double-blinded Clinical Trial to Evaluate the Efficacy and Safety in Non-Hospitalized Adult Participants
with COVID-19” was approved by the FDA, the trial is expected to start in the first quarter of 2024, provided we obtain adequate
funding.
On
January 27, 2023, an additional IND with the CDSCO was issued for ProLectin-I for an “IV treatment of SARS-CoV-2 in hospitalized
patients with moderate Covid-19 infections and for Long Covid”, and for ProLectin-F for “treatment of lung-fibrosis as a
result of use of ventilator”.
On
April 19, 2023, the Company announced that its long awaited Acelluar Oxygen Carrier (“AOC”) BXT-25 had been successfully
tested in animals. The initial results are very encouraging because they show the non-toxicity of the experimental drug, along with the
corresponding full recovery in Swiss Albino mice, in an experiment carried out in a joint venture with NDPD Pharma, Inc. As a next step,
the Company intends to proceed with a 14-day repeated dose toxicity study using New Zealand Rabbits and Wistar Rats as funding permits.
Our
product development costs will increase if we experience delays in clinical testing or marketing approvals. We do not know whether any
of our intended preclinical studies or clinical trials will begin as planned, will need to be restructured or will be completed on schedule,
or at all. Significant preclinical or clinical trial delays also could shorten any periods during which we may have the exclusive right
to commercialize our drug candidates or allow our competitors to bring products to market before we do, potentially impairing our ability
to successfully commercialize our drug candidates and harming our business and results of operations.
A
fast track, breakthrough therapy or other designation by the FDA may not actually lead to a faster development or regulatory review or
approval process.
We
may seek fast-track, breakthrough therapy or similar designation for our drug candidates. If a drug is intended for the treatment of
a serious or life-threatening condition and the drug demonstrates the potential to address unmet medical needs for this condition, the
drug sponsor may apply for FDA fast track designation. The FDA has broad discretion whether or not to grant this designation, and even
if we believe a particular drug candidate is eligible for this designation, we cannot assure you that the FDA would decide to grant it.
Even if we do receive fast track designation, we may not experience a faster development process, review or approval compared to conventional
FDA procedures. The FDA may withdraw fast track designation if it believes that the designation is no longer supported by data from our
clinical development program.
Additionally,
we may, in the future seek a breakthrough therapy designation for some of our product candidates that reach the regulatory review process.
A breakthrough therapy is a drug candidate that is intended, alone or in combination with one or more other drugs, to treat a serious
or life-threatening disease or condition, and that, as indicated by preliminary clinical evidence, may demonstrate substantial improvement
over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical
development. Drugs designated as breakthrough therapies by the FDA are eligible for accelerated approval and increased interaction and
communication with the FDA designed to expedite the development and review process.
As
with fast-track designation, designation as a breakthrough therapy is within the discretion of the FDA. Accordingly, even if we believe
one of our product candidates meets the criteria for designation as a breakthrough therapy, the FDA may disagree and may determine not
to grant such a designation. Even if we receive a breakthrough therapy designation for any of our product candidates, the designation
may not result in a materially faster development process, review or approval compared to conventional FDA procedures. Further, obtaining
a breakthrough therapy designation does not assure or increase the likelihood of the FDA’s approval of the applicable product candidate.
In addition, even if one or more of our product candidates qualifies as a breakthrough therapy, the FDA could later determine that those
products no longer meet the conditions for the designation or determine not to shorten the time period for FDA review or approval.
We
will rely on third parties to conduct our clinical trials, and those third parties may not perform satisfactorily, including failing
to meet deadlines for the completion of such trials.
We
intend to use third-party clinical research organizations, or CROs, to conduct our planned clinical trials and do not plan to independently
conduct clinical trials of BXT-25 or any future drug candidates. We rely on third parties, such as CROs, clinical data management organizations,
medical institutions and clinical investigators, to conduct and manage our clinical trials. These agreements might terminate for a variety
of reasons, including a failure to perform by the third parties. If we need to enter into alternative arrangements, that will delay our
product development activities.
Our
reliance on these third parties for research and development activities reduces our control over these activities but does not relieve
us of our responsibilities. For example, we remain responsible for ensuring that each of our clinical trials is conducted in accordance
with the general investigational plan and protocols for the trial. Moreover, the FDA requires us to comply with regulatory standards,
commonly referred to as good clinical practices, or GCPs, for conducting, recording and reporting the results of clinical trials to assure
that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are
protected. Other countries’ regulatory agencies also have requirements for clinical trials with which we must comply. We also are
required to register ongoing clinical trials and post the results of completed clinical trials on a government-sponsored database, ClinicalTrials.gov,
within specified timeframes. Failure to do so can result in fines, adverse publicity and civil and criminal sanctions.
Furthermore,
these third parties may also have relationships with other entities, some of which may be our competitors. If these third parties do
not successfully carry out their contractual duties, meet expected deadlines or conduct our clinical trials in accordance with regulatory
requirements or our stated protocols, we will not be able to obtain, or may be delayed in obtaining, marketing approvals for our drug
candidates and will not be able to, or may be delayed in our efforts to, successfully commercialize our drug candidates.
We
also expect to rely on other third parties to store and distribute drug supplies for our clinical trials. Any performance failure on
the part of our distributors could delay clinical development or marketing approval of our drug candidates or commercialization of our
products, producing additional losses and depriving us of potential product revenue.
If
we experience delays or difficulties in the enrollment of patients in clinical trials, our receipt of necessary regulatory approvals
could be delayed or prevented.
We
may not be able to initiate or continue clinical trials for our drug candidates if we are unable to locate and enroll a sufficient number
of eligible patients to participate in these trials as required by the FDA or similar regulatory authorities outside the United States,
such as the EMA. In addition, some of our competitors have ongoing clinical trials for drug candidates that treat the same indications
as our drug candidates, and patients who would otherwise be eligible for our clinical trials may instead enroll in clinical trials of
our competitors’ drug candidates.
Patient
enrollment is affected by other factors including:
| |
● |
the
severity of the disease under investigation; |
| |
|
|
| |
● |
the
patient eligibility criteria for the study in question; |
| |
|
|
| |
● |
the
perceived risks and benefits of the drug candidate under study; |
| |
|
|
| |
● |
the
efforts to facilitate timely enrollment in clinical trials; |
| |
|
|
| |
● |
our
payments for conducting clinical trials; |
| |
|
|
| |
● |
the
patient referral practices of physicians; |
| |
|
|
| |
● |
the
ability to monitor patients adequately during and after treatment; and |
| |
|
|
| |
● |
the
proximity and availability of clinical trial sites for prospective patients. |
We
are unable to forecast with precision our ability to enroll patients. Our inability to enroll a sufficient number of patients for our
clinical trials would result in significant delays and could require us to abandon one or more clinical trials altogether. Enrollment
delays in our clinical trials may result in increased development costs for our drug candidates, which would cause the value of our Company
to decline and limit our ability to obtain additional financing.
If
serious adverse or unacceptable side effects are identified during the development of our drug candidate or we observe limited efficacy,
we may need to abandon or limit our development of some of our drug candidate.
If
our drug candidate is associated with undesirable side effects in clinical trials, have limited efficacy or have characteristics that
are unexpected, we may need to abandon their development or limit development to more narrow uses or subpopulations in which the undesirable
side effects or other characteristics are less prevalent, less severe or more acceptable from a risk-benefit perspective. Although we
have finalized a number of peer-reviewed proof-of-concept studies, we have not commenced regulated pre-clinical trials of BXT-25 and
regulated human trials ProLectin, which even if it proves successful, may later be found to cause side effects that will prevent further
development of the compounds.
Even
if our drug candidate receives marketing approval, it may fail to achieve the degree of market acceptance by physicians, patients, third-party
payers and others in the medical community necessary for commercial success.
Even
if our drug candidate receives marketing approval, it may nonetheless fail to gain sufficient market acceptance by physicians, patients,
third-party payers and others in the medical community. If our drug candidate does not achieve an adequate level of acceptance, we may
not generate significant product revenues and we may not become profitable. The degree of market acceptance of our drug candidate, if
approved for commercial sale, will depend on a number of factors, including:
| |
● |
their
efficacy, safety and other potential advantages compared to alternative treatments; |
| |
|
|
| |
● |
our
ability to offer them for sale at competitive prices; |
| |
● |
their
convenience and ease of administration compared to alternative treatments; |
| |
|
|
| |
● |
the
willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| |
|
|
| |
● |
the
strength of marketing and distribution support; |
| |
|
|
| |
● |
the
availability of third-party coverage and adequate reimbursement for our drug candidate; |
| |
|
|
| |
● |
the
prevalence and severity of their side effects; |
| |
|
|
| |
● |
any
restrictions on the use of our products together with other medications; |
| |
|
|
| |
● |
interactions
of our products with other medicines patients are taking; and |
| |
|
|
| |
● |
inability
of certain types of patients to take our products. |
If
we are unable to address and overcome these and similar concerns, our business and results of operations could be substantially harmed.
If
we are unable to establish effective sales, marketing and distribution capabilities or enter into agreements with third parties with
such capabilities, we may not be successful in commercializing our drug candidate if and when they are approved.
We
do not have a sales or marketing infrastructure and have limited experience in the sale, marketing or distribution of our products. To
achieve commercial success for any product for which we obtain marketing approval, we will need to successfully establish and maintain
relationships with third parties to perform sales and marketing functions.
Factors
that may inhibit our efforts to commercialize our products on our own include:
| |
● |
our
inability to recruit, train and retain adequate numbers of effective sales and marketing personnel; |
| |
|
|
| |
● |
the
inability of sales personnel to obtain access to or educate physicians on the benefits of our products; |
| |
|
|
| |
● |
the
lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies
with more extensive product lines; |
| |
|
|
| |
● |
unforeseen
costs and expenses associated with creating an independent sales and marketing organization; |
| |
|
|
| |
● |
inability
to obtain sufficient coverage and reimbursement from third-party payors and governmental agencies; and |
| |
|
|
| |
● |
inability
to obtain sufficient coverage and reimbursement from third-party payors and governmental agencies. |
We
will rely on third parties to sell, market and distribute our drug candidate. We may not be successful in entering into, or maintaining,
arrangements with such third parties or may be unable to do so on terms that are favorable to us. In addition, our product revenues and
our profitability, if any, may be lower if we rely on third parties for these functions than if we were to market, sell and distribute
any products that we develop ourselves. We likely will have little control over such third parties, and any of them may fail to devote
the necessary resources and attention to sell and market our products effectively. If we do not establish sales, marketing and distribution
capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our
drug candidate.
If
we are unable to convince physicians as to the benefits of our proposed products, we may incur delays or additional expense in our attempt
to establish market acceptance.
Broad
use of our proposed products may require physicians to be informed regarding our proposed products and the intended benefits. Inability
to carry out this physician education process may adversely affect market acceptance of our proposed products. We may be unable to timely
educate physicians regarding our proposed products in sufficient numbers to achieve our marketing plans or to achieve product acceptance.
Any delay in physician education may materially delay or reduce demand for our products. In addition, we may expend significant funds
toward physician education before any acceptance or demand for our proposed products is created, if at all.
We
face substantial competition, which may result in others discovering, developing or commercializing competing products before or more
successfully than we do.
The
development and commercialization of new drug products is highly competitive. We face competition with respect to BXT-25 and will face
competition with respect to any drug candidates that we may seek to develop or commercialize in the future, from major pharmaceutical
companies, specialty pharmaceutical companies and biotechnology companies worldwide. There are a number of large pharmaceutical and biotechnology
companies that currently market and sell products or are pursuing the development of products in the field of oxygen therapeutics for
the treatment of a variety of conditions and any of such products may target the stroke. Potential competitors also include academic
institutions, government agencies and other public and private organizations that conduct research, seek patent protection and establish
collaborative arrangements for research, development, manufacturing and commercialization.
A
substantial number of the companies against which we are competing or against which we may compete in the future have significantly greater
financial resources, established presence in the market and expertise in research and development, manufacturing, preclinical testing,
conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Mergers and acquisitions in the
pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors.
Smaller
and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large
and established companies. These third parties compete with us in recruiting and retaining qualified scientific, sales and marketing
and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies
complementary to, or necessary for, our programs.
Our
commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are more effective,
have fewer or less severe side effects, are more convenient or are less expensive than any products that we may develop. Our competitors
also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result
in our competitors establishing a strong market position before we are able to enter the market. In addition, our ability to compete
may be affected in many cases by insurers or other third-party payors seeking to encourage the use of generic products.
We
may be unable to compete in our target marketplaces, which could impair our ability to generate revenues, thus causing a material adverse
impact on our results of operations.
Our
success depends upon our ability to retain key executives and to attract, retain, and motivate qualified personnel, and the loss of these
persons could adversely affect our operations and results.
We
are highly dependent on the principal members of our management, scientific and clinical team, including Dr. David Platt, our Chairman,
President and Chief Executive Officer, Mike Sheikh, our Chief Communications Officer and Ola Soderquist, our Chief Financial Officer.
We do not have “key person” insurance for any of Dr. Platt, Mr. Sheikh or Mr. Soderquist and even if such policies were to
be obtained, such policies may not adequately compensate us for the loss of their services.
The
loss of the services of any of our executive officers or of any members of our scientific and medical advisory board, could impede the
achievement of our research, development and commercialization objectives and seriously harm our ability to successfully implement our
business strategy. Furthermore, replacing executive officers and key employees may be difficult and may take an extended period of time
because of the limited number of individuals in our industry with the breadth of skills and experience required to successfully develop,
gain regulatory approval of and commercialize products. Competition to hire from this limited pool is intense, and we may be unable to
hire, train, retain or motivate these key personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology
companies for similar personnel. We also experience competition for the hiring of scientific and clinical personnel from universities
and research institutions. In addition, we rely and expect to continue to rely to a significant degree on consultants and advisors, including
scientific and clinical advisors, to assist us in formulating our research and development and commercialization strategy. Our consultants
and advisors may be employed by employers other than us and may have commitments under consulting or advisory contracts with other entities
that may limit their availability to us. If we are unable to continue to attract and retain high quality personnel, our ability to pursue
our growth strategy will be limited.
Our
lack of operating experience may cause us difficulty in managing our growth which could lead to our inability to implement our business
plan.
We
have limited experience in marketing and the selling of pharmaceutical products. Any growth will require us to expand our management
and our operational and financial systems and controls. If we are unable to do so, our business and financial condition would be materially
harmed. If rapid growth occurs, it may strain our operational, managerial and financial resources.
We
will depend on third parties to manufacture and market our products and to design trial protocols, arrange for and monitor the clinical
trials, and collect and analyze data.
We
do not have, and do not now intend to develop, facilities for the manufacture of any of our products for clinical or commercial production.
In addition, we are not a party to any long-term agreement with any of our suppliers, and accordingly, we have our products manufactured
on a purchase-order basis from one of two primary suppliers. We will need to develop relationships with manufacturers and enter into
collaborative arrangements with licensees or have others manufacture our products on a contract basis. We expect to depend on such collaborators
to supply us with products manufactured in compliance with standards imposed by the FDA and foreign regulators.
Moreover,
as we develop products eligible for clinical trials, we contract with independent parties to design the trial protocols, arrange for
and monitor the clinical trials, collect data and analyze data. In addition, certain clinical trials for our products may be conducted
by government-sponsored agencies and will be dependent on governmental participation and funding. Our dependence on independent parties
and clinical sites involves risks including reduced control over the timing and other aspects of our clinical trials.
We
are exposed to product liability, pre-clinical and clinical liability risks which could place a substantial financial burden upon us,
should we be sued.
Our
business exposes us to potential product liability and other liability risks that are inherent in the testing, manufacturing and marketing
of pharmaceutical formulations and products. Such claims may be asserted against us. In addition, the use in our clinical trials of pharmaceutical
formulations and products that our potential collaborators may develop and the subsequent sale of these formulations or products by us
or our potential collaborators may cause us to bear a portion of or all product liability risks. A successful liability claims, or series
of claims brought against us could have a material adverse effect on our business, financial condition and results of operations.
Since
we do not currently have any FDA-approved products or other formulations, we do not currently have any other product liability insurance
covering commercialized products. We may not be able to obtain or maintain adequate product liability insurance, when needed, on acceptable
terms, if at all, or such insurance may not provide adequate coverage against our potential liabilities. Furthermore, our potential partners
with whom we intend to have collaborative agreements, or our future licensees may not be willing to indemnify us against these types
of liabilities and may not themselves be sufficiently insured or have sufficient liquidity to satisfy any product liability claims. Claims
or losses in excess of any product liability insurance coverage that may be obtained by us could have a material adverse effect on our
business, financial condition and results of operations.
In
addition, we may be unable to obtain or to maintain clinical trial liability insurance on acceptable terms, if at all. Any inability
to obtain and/or maintain insurance coverage on acceptable terms could prevent or limit the commercialization of any products we develop.
If
users of our proposed products are unable to obtain adequate reimbursement from third-party payers or if new restrictive legislation
is adopted, market acceptance of our proposed products may be limited, and we may not achieve revenues.
The
continuing efforts of government and insurance companies, health maintenance organizations and other payers of healthcare costs to contain
or reduce costs of health care may affect our future revenues and profitability, and the future revenues and profitability of our potential
customers, suppliers and collaborative partners and the availability of capital. For example, in certain international markets, pricing
or profitability of prescription pharmaceuticals is subject to government control. In the U.S., given recent federal and state government
initiatives directed at lowering the total cost of health care, the U.S. Congress and state legislatures will likely continue to focus
on health care reform, the cost of prescription pharmaceuticals and on the reform of the Medicare and Medicaid systems. While we cannot
predict whether any such legislative or regulatory proposals will be adopted, the announcement or adoption of such proposals could materially
harm our business, financial condition and results of operations.
Our
ability to commercialize our proposed products will depend in part on the extent to which appropriate reimbursement levels for the cost
of our proposed formulations and products and related treatments are obtained by governmental authorities, private health insurers and
other organizations, such as HMOs. Third-party payers are increasingly challenging the prices charged for medical drugs and services.
Also, the trend toward managed health care in the U.S. and the concurrent growth of organizations such as HMOs, which could control or
significantly influence the purchase of health care services and drugs, as well as legislative proposals to reform health care or reduce
government insurance programs, may all result in lower prices for or rejection of our products.
There
are risks associated with our reliance on third parties for marketing, sales and distribution infrastructure and channels.
We
intend to enter into agreements with commercial partners to engage in sales, marketing and distribution efforts around our products in
development. We may be unable to establish or maintain these third-party relationships, or establish new relationships, on a commercially
reasonable basis, if at all. In addition, these third parties may have similar or more established relationships with our competitors.
If we do not enter into or maintain relationships with third parties for the sales and marketing of our proposed products, we will need
to develop our own sales and marketing capabilities. Furthermore, even if engaged, these distributors may:
| |
● |
fail
to satisfy financial or contractual obligations to us; |
| |
|
|
| |
● |
fail
to adequately market our products; |
| |
|
|
| |
●
|
cease
operations with little or no notice to us; or |
| |
|
|
| |
●
|
offer,
design, manufacture or promote competing formulations or products. |
If
we fail to develop sales, marketing and distribution channels, we could experience delays in generating sales and incur increased costs,
which would harm our financial results.
We
will be subject to risks if we seek to develop our own sales force.
If
we choose at some point to develop our own sales and marketing capability, our experience in developing a fully integrated commercial
organization is limited. If we choose to establish a fully integrated commercial organization, we will likely incur substantial expenses
in developing, training and managing such an organization. We may be unable to build a fully integrated commercial organization on a
cost-effective basis, or at all. Any such direct marketing and sales efforts may prove to be unsuccessful. In addition, we will compete
with many other companies that currently have extensive and well-funded marketing and sales operations. Our marketing and sales efforts
may be unable to compete against these other companies. We may be unable to establish a sufficient sales and marketing organization on
a timely basis, if at all.
Risks
Related to Our Industry
We
will need regulatory approvals to commercialize our products as drugs.
In
offering BXT-25, ProLectin or any other product as a drug, we are required to obtain approval from the FDA to sell our products in the
U.S. and from foreign regulatory authorities to sell our products in other countries. The FDA’s review and approval process is
lengthy, expensive and uncertain. Extensive pre-clinical and clinical data and supporting information must be submitted to the FDA for
each indication for each product candidate to secure FDA approval. Before receiving FDA clearance to market our proposed products, we
will have to demonstrate that our products are safe and effective on the patient population and for the diseases that are to be treated.
Clinical trials, manufacturing and marketing of drugs are subject to the rigorous testing and approval process of the FDA and equivalent
foreign regulatory authorities. The Federal Food, Drug and Cosmetic Act and other federal, state and foreign statutes and regulations
govern and influence the testing, manufacture, labeling, advertising, distribution and promotion of drugs and medical devices. As a result,
regulatory approvals can take a number of years or longer to accomplish and require the expenditure of substantial financial, managerial
and other resources. The FDA could reject an application or require us to conduct additional clinical or other studies as part of the
regulatory review process. Delays in obtaining or failure to obtain FDA approvals would prevent or delay the commercialization of our
product candidates, which would prevent, defer or decrease our receipt of revenues. In addition, if we receive initial regulatory approval,
our product candidates will be subject to extensive and rigorous ongoing domestic and foreign government regulation.
Data
obtained from clinical trials are susceptible to varying interpretations, which could delay, limit or prevent regulatory clearances.
Data
we obtain from our planned pre-clinical studies and clinical trials will not necessarily predict the results that will be obtained from
later pre-clinical studies and clinical trials. Moreover, pre-clinical and clinical data is susceptible to varying interpretations, which
could delay, limit or prevent regulatory approval. A number of companies in the pharmaceutical industry have suffered significant setbacks
in advanced clinical trials, even after promising results in earlier trials. The failure to adequately demonstrate the safety and effectiveness
of a proposed formulation or product under development could delay or prevent regulatory clearance of the potential drug, resulting in
delays to commercialization, and could materially harm our business. Our clinical trials may not demonstrate sufficient levels of safety
and efficacy necessary to obtain the requisite regulatory approvals for our drugs, and thus our proposed drugs may not be approved for
marketing.
Our
competitive position depends on protection of our intellectual property.
Development
and protection of our intellectual property are critical to our business. All of our intellectual property has been invented and/or developed
or co-developed by Dr. David Platt; and other intellectual property that is important to the development of BXT-25 is in the public domain.
If we do not adequately protect our intellectual property, or if competitors develop technologies incorporating the same or similar technologies
that already are in the public domain, those competitors may be able to practice our technologies. Our success depends in part on our
ability to obtain patent protection for our products or processes in the U.S. and other countries, protect trade secrets, and prevent
others from infringing on our proprietary rights.
Since
patent applications in the U.S. are maintained in secrecy for at least portions of their pendency periods (published on U.S. patent issuance
or, if earlier, 18 months from earliest filing date for most applications) and since other publication of discoveries in the scientific
or patent literature often lags behind actual discoveries, we cannot be certain that we are or will be the first to make the inventions
to be covered by our patent applications. The patent position of biopharmaceutical firms generally is highly uncertain and involves complex
legal and factual questions. The U.S. Patent and Trademark Office (USPTO) has not established a consistent policy regarding the breadth
of claims that it will allow in biotechnology patents.
The
patent applications we file, including applications that will follow the filing of Provisionals, may not issue as patents or the claims
of any issued patents may not afford meaningful protection for our technologies or products. In addition, patents issued to us or to
any future licensors may be challenged and subsequently narrowed, invalidated or circumvented. Patent litigation is widespread in the
biotechnology industry and could harm our business. Litigation might be necessary to protect our patent position or to determine the
scope and validity of third-party proprietary rights, and we may not have the required resources to pursue such litigation or to protect
our patent rights.
Although
we will require our scientific and technical employees and consultants to enter into broad assignment of inventions agreements, and all
of our employees, consultants and corporate partners with access to proprietary information to enter into confidentiality agreements,
these agreements may not be honored. Currently, we do not have any scientific or technical employees.
Products
we develop could be subject to infringement claims asserted by others.
We
cannot assure that products based on our patents or intellectual property that we license from others will not be challenged by a third-party
claiming infringement of its proprietary rights. If we were not able to successfully defend patents that may be issued to us, that we
may acquire, or that we may license in the future, we may have to pay substantial damages, possibly including treble damages, for past
infringement.
We
face intense competition in the biotechnology and pharmaceutical industries.
The
biotechnology and pharmaceutical industries are intensely competitive. We face direct competition from U.S. and foreign companies focusing
on pharmaceutical products, which are rapidly evolving. Our competitors include major multinational pharmaceutical and chemical companies,
specialized biotechnology firms and universities and other research institutions. Many of these competitors have greater financial and
other resources, larger research and development staffs and more effective marketing and manufacturing organizations, than we do. In
addition, academic and government institutions are increasingly likely to enter into exclusive licensing agreements with commercial enterprises,
including our competitors, to market commercial products based on technology developed at such institutions. Our competitors may succeed
in developing or licensing technologies and products that are more effective or less costly than ours or succeed in obtaining FDA or
other regulatory approvals for product candidates before we do. Acquisitions of, or investments in, competing pharmaceutical or biotechnology
companies by large corporations could increase such competitors’ financial, marketing, manufacturing and other resources.
The
market for our proposed products is rapidly changing and competitive, and new drugs and new treatments which may be developed by others
could impair our ability to maintain and grow our business and remain competitive.
The
pharmaceutical and biotechnology industries are subject to rapid and substantial technological change. Developments by others may render
our proposed products noncompetitive or obsolete, or we may be unable to keep pace with technological developments or other market factors.
Technological competition from pharmaceutical and biotechnology companies, universities, governmental entities and others diversifying
into the field is intense and is expected to increase.
As
a pre-revenue company engaged in the development of drug technologies, our resources are limited, and we may experience technical challenges
inherent in such technologies. Competitors have developed or are in the process of developing technologies that are, or in the future
may be, the basis for competition. Some of these technologies may have an entirely different approach or means of accomplishing similar
therapeutic effects compared to our proposed products. Our competitors may develop drugs that are safer, more effective or less costly
than our proposed products and, therefore, present a serious competitive threat to us.
The
potential widespread acceptance of therapies that are alternatives to ours may limit market acceptance of our proposed products, even
if commercialized. Many of our targeted diseases and conditions can also be treated by other medication. These treatments may be widely
accepted in medical communities and have a longer history of use. The established use of these competitive drugs may limit the potential
for our technologies, formulations and products to receive widespread acceptance if commercialized.
Health
care cost containment initiatives and the growth of managed care may limit our returns.
Our
ability to commercialize our products successfully may be affected by the ongoing efforts of governmental and third-party payers to contain
the cost of health care. These entities are challenging prices of health care products and services, denying or limiting coverage and
reimbursement amounts for new therapeutic products, and for FDA-approved products considered experimental or investigational, or which
are used for disease indications without FDA marketing approval.
Even
if we succeed in bringing any products to the market, they may not be considered cost-effective and third-party reimbursement might not
be available or sufficient. If adequate third-party coverage is not available, we may not be able to maintain price levels sufficient
to realize an appropriate return on our investment in research and product development. In addition, legislation and regulations affecting
the pricing of pharmaceuticals may change in ways adverse to us before or after any of our proposed products are approved for marketing.
Risks
Related to Our Intellectual Property
If
we are unable to obtain and maintain patent protection for our products, or if the scope of the patent protection obtained is not sufficiently
broad, competitors could develop and commercialize products similar or identical to ours, and our ability to successfully commercialize
our products may be impaired.
Our
plan for the development of BXT-25 is based in part on a technology developed by the Biopure Corporation which separates hemoglobin from
red blood cells. Biopure filed for bankruptcy in 2009 and the technology we use from Biopure is in the public domain. We plan to apply
our proprietary chemistry to break down and augment a bovine hemoglobin molecule producing a co-polymer-based molecule we call BXT-25.
We face competitors and other entities who are engaged in the further development of some or all of that public-domain technology for
the purpose of creating products that may compete directly with our products.
Among
such competitors and other entities is Boston Therapeutics, Inc. (OTCQB: BTHE). Our chairman, Dr. Platt, was founder, and until April
1, 2015, Chief Executive Officer of Boston Therapeutics; that entity is a pharmaceutical company focused on developing, manufacturing
and commercializing novel compounds based on complex carbohydrate chemistry to address unmet medical needs in diabetes. According to
its website, products Boston Therapeutics seeks to develop an anti-necrosis glyco-protein based therapeutic agent that consists of a
stabilized glycoprotein composition containing oxygen-rechargeable iron, targeting both human and animal tissues and organ systems deprived
of oxygen and in need of metabolic support. The Boston Therapeutic development efforts are, like the efforts of the Company, based in
part on Biopure technology that is now in the public domain. While Boston Therapeutics is focused on medical conditions that are different
from the conditions that will be addressed by the Company, and while the Company’s proprietary technology is very different from
the technology under development at Boston Therapeutics at the time of Dr. Platt’s departure from that entity, a refocus of Boston
Therapeutics to treat conditions that are central to the Company’s focus may make it a direct competitor. In 2021 BTHE ceased all
existing operations after a reverse merger with Nanomix.
Currently
there are four drug candidates to treat a stroke. Abciximab, from Eli Lilly, is a platelet aggregation Antagonist. Clinical trials show
little advantage over placebos and could lead to dangerous side effects, including more bleeding in patients. Cerovive, from AstraZeneca,
is a Nitrone-based neuro protectant currently in phase III clinical trials which shows no significant benefit over placebos with respect
to changes in neurological impairment as measured by the national institute of health stroke scale. Candesartan, from AstraZeneca, is
an angiotensin receptor blocker which was used to control blood pressure. Its efficacy in stroke patients still must be proven. Ancod,
from Knoll Pharmaceuticals, is an anti-coagulant that acts by breaking down the fibrinogen. It increases the risk of hemorrhage similar
to those associated with tPA.
Our
success depends in large part on our ability to obtain and maintain patent and other intellectual property protection in the United States
and other countries with respect to our proprietary products. We seek to protect our proprietary position by filing patent applications
in the United States and abroad related to our drug candidates.
The
patent prosecution process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent
applications at a reasonable cost, in a timely manner, or in all jurisdictions. It is also possible that we will fail to identify patentable
aspects of our research and development output before it is too late to obtain patent protection.
The
patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions
and has in recent years been the subject of much litigation. In addition, the laws of foreign countries may not protect our rights to
the same extent as the laws of the United States and we may fail to seek or obtain patent protection in all major markets. For example,
European patent law restricts the patentability of methods of treatment of the human body more than United States law does. Publications
of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and
other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. Therefore, we cannot know
with certainty whether we were the first to make the inventions claimed in our owned patents or pending patent applications, or that
we were the first to file for patent protection of such inventions, nor can we know whether those from whom we license patents were the
first to make the inventions claimed or were the first to file. As a result, the issuance, scope, validity, enforceability and commercial
value of our patent rights are highly uncertain. Our pending and future patent applications may not result in patents being issued which
protect our technology or products, in whole or in part, or which effectively prevent others from commercializing competitive technologies
and products. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish
the value of our patents or narrow the scope of our patent protection.
Recent
patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement
or defense of our issued patents. On September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into
law. The Leahy-Smith Act includes a number of significant changes to United States patent law. These include provisions that affect the
way patent applications are prosecuted and may also affect patent litigation. The USPTO, recently developed new regulations and procedures
to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act,
and in particular, the first to file provisions, only became effective on March 16, 2013. Accordingly, it is not clear what, if any,
impact the Leahy-Smith Act will have on the operation of our business. However, the Leahy-Smith Act and its implementation could increase
the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents,
all of which could have a material adverse effect on our business and financial condition.
Moreover,
we may be subject to a third-party pre-issuance submission of prior art to the USPTO, or become involved in opposition, derivation, reexamination,
inter partes review, post-grant review or interference proceedings challenging our patent rights or the patent rights of others.
An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate our patent rights,
allow third parties to commercialize our technology or products and compete directly with us, without payment to us, or result in our
inability to manufacture or commercialize products without infringing third-party patent rights. In addition, if the breadth or strength
of protection provided by our patents and patent applications is threatened, it could dissuade companies from collaborating with us to
license, develop or commercialize current or future drug candidates.
Even
if our patent applications issue as patents, they may not issue in a form that will provide us with any meaningful protection, prevent
competitors from competing with us or otherwise provide us with any competitive advantage. Our competitors may be able to circumvent
our owned or licensed patents by developing similar or alternative technologies or products in a non-infringing manner.
The
issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our patents may be challenged in
the courts or patent offices in the United States and abroad. Such challenges may result in loss of exclusivity or freedom to operate
or in patent claims being narrowed, invalidated or held unenforceable, in whole or in part, which could limit our ability to stop others
from using or commercializing similar or identical products, or limit the duration of the patent protection of our products. Given the
amount of time required for the development, testing and regulatory review of new drug candidates, patents protecting such candidates
might expire before or shortly after such candidates are commercialized. As a result, our patent portfolio may not provide us with sufficient
rights to exclude others from commercializing products similar or identical to ours.
We
may become involved in lawsuits to protect or enforce our patents or other intellectual property, which could be expensive, time-consuming
and ultimately unsuccessful.
Competitors
may infringe our issued patents or other intellectual property. To counter infringement or unauthorized use, we may be required to file
infringement claims, which can be expensive and time-consuming. Any claims we assert against perceived infringers could provoke these
parties to assert counterclaims against us alleging that we infringe their intellectual property. In addition, in a patent infringement
proceeding, a court may decide that a patent of ours is invalid or unenforceable, in whole or in part, construe the patent’s claims
narrowly or refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology
in question. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated or interpreted
narrowly, which could adversely affect us.
Third
parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would
be uncertain and could have a material adverse effect on the success of our business.
Our
commercial success depends upon our ability to develop, manufacture, market and sell our drug candidates without infringing the proprietary
rights of third parties. There is considerable intellectual property litigation in the biotechnology and pharmaceutical industries. While
no such litigation has been brought against us and we have not been held by any court to have infringed a third party’s intellectual
property rights, we cannot guarantee that our products or use of our products do not infringe third-party patents. It is also possible
that we have failed to identify relevant third-party patents or applications. For example, applications filed before November 29, 2000,
and certain applications filed after that date that will not be filed outside the United States remain confidential until patents issue.
Patent applications in the United States and elsewhere are published approximately 18 months after the earliest filing, which is referred
to as the priority date. Therefore, patent applications covering our products or technology could have been filed by others without our
knowledge. Additionally, pending patent applications which have been published can, subject to certain limitations, be later amended
in a manner that could cover our technologies, our products or the use of our products.
We
may become party to, or threatened with, future adversarial proceedings or litigation regarding intellectual property rights with respect
to our products and technology, including inter parties review, interference, or derivation proceedings before the USPTO and similar
bodies in other countries. Third parties may assert infringement claims against us based on existing intellectual property rights and
intellectual property rights that may be granted in the future.
If
we are found to infringe a third party’s intellectual property rights, we could be required to obtain a license from such third
party to continue developing and marketing our products. However, we may not be able to obtain any required license on commercially reasonable
terms or at all. Even if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same
technologies licensed to us. We could be forced, including by court order, to cease commercializing the infringing technology or product.
In addition, we could be found liable for monetary damages, including treble damages and attorneys’ fees if we are found to have
willfully infringed a patent. A finding of infringement could prevent us from commercializing our drug candidates or force us to cease
some of our business operations, which could materially harm our business. Claims that we have misappropriated the confidential information
or trade secrets of third parties could have a similar negative impact on our business.
Obtaining
and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements
imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic
maintenance fees on any issued patent are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime
of the patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary,
fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured
by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result
in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction.
Noncompliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure
to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal
documents. In such an event, our competitors might be able to enter the market, which would have a material adverse effect on our business.
We
may be subject to claims by third parties asserting that our employees or we have misappropriated their intellectual property, or claiming
ownership of what we regard as our own intellectual property.
The
employees and consultants we may hire likely will have been previously employed at universities or other biotechnology or pharmaceutical
companies, including our competitors or potential competitors. Although we will try to ensure that our employees and contractors do not
use the proprietary information or know-how of others in their work for us, we may be subject to claims that these employees or we have
used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former
employer. Litigation may be necessary to defend against these claims.
In
addition, while it is our policy to require our employees and contractors who may be involved in the development of intellectual property
to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party
who in fact develops intellectual property that we regard as our own. Our and their assignment agreements may not be self-executing or
may be breached, and we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine
the ownership of what we regard as our intellectual property.
If
we fail in prosecuting or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property
rights or personnel. Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial
costs and be a distraction to management.
Intellectual
property litigation could cause us to spend substantial resources and distract our personnel from their normal responsibilities.
Even
if resolved in our favor, litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant
expenses and could distract our technical and management personnel from their normal responsibilities. In addition, there could be public
announcements of the results of hearings, motions or other interim proceedings or developments and if securities analysts or investors
perceive these results to be negative, it could have a substantial adverse effect on the price of our Common Stock. Such litigation or
proceedings could substantially increase our operating losses and reduce the resources available for development activities or any future
sales, marketing or distribution activities. We may not have sufficient financial or other resources to conduct such litigation or proceedings
adequately. Some of our competitors may be able to sustain the costs of such litigation or proceedings more effectively than we can because
of their greater financial resources. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings
could compromise our ability to compete in the marketplace.
If
we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In
addition to seeking patents for some of our technology and drug candidates, we also intend to rely on trade secrets, including unpatented
know-how, technology and other proprietary information, to maintain our competitive position. We will seek to protect these trade secrets,
in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees,
corporate collaborators, outside scientific collaborators, contract manufacturers, consultants, advisors and other third parties. We
also seek to enter into confidentiality and invention or patent assignment agreements with our employees and consultants. Despite these
efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we
may not be able to obtain adequate remedies for such breaches. Our trade secrets may also be obtained by third parties by other means,
such as breaches of our physical or computer security systems. Enforcing a claim that a party illegally disclosed or misappropriated
a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside
the United States are less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or
independently developed by a competitor, we would have no right to prevent them, or those to whom they communicate it, from using that
technology or information to compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor,
our competitive position would be harmed.
Risks
Relating to this Offering and Ownership of Our Common Stock
Prior
to this offering, we had a limited public market for our shares of Common Stock and you may not be able to resell our shares at or above
the price you paid, or at all.
On
March 30, 2022, the Company received the FINRA Clearance Letter and on June 14, 2022, OTC Market Group agreed to the removal of the Caveat
Emptor symbol. The Company was approved for quotation on the OTCQB on September 28, 2022. We cannot assure you that an active public
market for our Common Stock will develop or that the market price of our shares will not decline below the Proposed Maximum Offering
Price. The Proposed Maximum Offering Price of our shares may not be indicative of prices that will prevail in the trading market following
the offering.
Because
we are subject to the “Penny Stock” rules, the level of trading activity in our stock may be reduced.
The
SEC has adopted regulations which generally define “penny stock” to be any listed, trading equity security that has a market
price less than $5.00 per share or an exercise price of less than $5.00 per share, subject to certain exemptions. The penny stock rules
require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk
disclosure document that provides information about penny stocks and the risks in the penny stock market. The broker-dealer must also
provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson
in the transaction, and monthly account statements showing the market value of each penny stock held in the customer’s account.
In addition, the penny stock rules generally require that prior to a transaction in a penny stock, the broker-dealer make a special written
determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s written agreement to
the transaction. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market
for a stock that becomes subject to the penny stock rules which may increase the difficulty Purchasers may experience in attempting to
liquidate such securities.
We
do not expect to pay dividends in the foreseeable future. Any return on investment may be limited to the value of our Common Stock.
We
do not anticipate paying cash dividends on our Common Stock in the foreseeable future. The payment of dividends on our Common Stock will
depend on earnings, financial condition and other business and economic factors affecting it at such time as the Board of Directors may
consider relevant. If we do not pay dividends, our Common Stock may be less valuable because a return on your investment will occur only
if our stock price appreciates.
Provisions
in the Nevada Revised Statutes and our Bylaws could make it very difficult for an investor to bring any legal actions against our Directors
or officers for violations of their fiduciary duties or could require us to pay any amounts incurred by our Directors or officers in
any such actions.
Members
of our Board of Directors (the “Board”) and our officers will have no liability for breaches of their fiduciary duty of care
as a Director or officer, except in limited circumstances, pursuant to provisions in the Nevada Revised Statutes and our Bylaws as authorized
by the Nevada Revised Statutes. Specifically, Section 78.138 of the Nevada Revised Statutes provides that a director or officer is not
individually liable to the Company or its Shareholders or creditors for any damages as a result of any act or failure to act in his or
her capacity as a director or officer unless it is proven that (1) the director’s or officer’s act or failure to act constituted
a breach of his or her fiduciary duties as a director or officer and (2) his or her breach of those duties involved intentional misconduct,
fraud or a knowing violation of law. This provision is intended to afford directors and officers protection against and to limit their
potential liability for monetary damages resulting from suits alleging a breach of the duty of care by a director or officer. Accordingly,
you may be unable to prevail in a legal action against our Directors or officers even if they have breached their fiduciary duty of care.
In addition, our Bylaws allow us to indemnify our Directors and officers from and against any and all costs, charges and expenses resulting
from their acting in such capacities with us. This means that if you were able to enforce an action against our Directors or officers,
in all likelihood, we would be required to pay any expenses they incurred in defending the lawsuit and any judgment or settlement they
otherwise would be required to pay. Accordingly, our indemnification obligations could divert needed financial resources and may adversely
affect our business, financial condition, results of operations and cash flows, and adversely affect prevailing market prices for our
Common Stock.
Future
sales of substantial amounts of the shares of Common Stock by existing Shareholders could adversely affect the price of our Common Stock.
If
our existing Shareholders sell substantial amounts of the shares following this offering, the market price of our Common Stock could
fall. Such sales by our existing Shareholders might make it more difficult for us to issue new equity or equity-related securities in
the future at a time and place we deem appropriate. The shares of Common Stock offered in this offering will be eligible for immediate
resale in the public market without restrictions. All remaining shares, which are currently held by our existing Shareholders, may be
sold in the public market in the future subject to the lock-up agreements and the restrictions contained in Rule 144 under the Securities
Act. If any existing Shareholders sell a substantial number of shares, the prevailing market price for our shares could be adversely
affected.
The
market price of our Common Stock may be subject to fluctuation, and you could lose all or part of your investment.
The
Proposed Maximum Offering Price has been arbitrarily determined by us and may not be indicative of prices that will prevail in the trading
market. The price of our shares may decline following this offering. The stock market in general has been, and the market price of our
ordinary shares in particular will likely be, subject to fluctuation, whether due to, or irrespective of, our operating results and financial
condition. The market price of our shares may fluctuate as a result of a number of factors, some of which are beyond our control, including,
but not limited to:
| |
● |
actual
or anticipated variations in our and our competitors’ results of operations and financial condition; |
| |
|
|
| |
● |
market
acceptance of our products; |
| |
|
|
| |
● |
the
mix of products that we sell and related services that we provide; |
| |
|
|
| |
● |
changes
in earnings estimates or recommendations by securities analysts, if our shares are covered by analysts; |
| |
|
|
| |
● |
development
of technological innovations or new competitive products by others; |
| |
|
|
| |
● |
announcements
of technological innovations or new products by us; |
| |
|
|
| |
● |
failure
by us to achieve a publicly announced milestone; |
| |
|
|
| |
● |
delays
between our expenditures to develop and market new or enhanced products and the generation of sales from those products; |
| |
|
|
| |
● |
developments
concerning intellectual property rights, including our involvement in litigation; |
| |
|
|
| |
● |
regulatory
developments and the decisions of regulatory authorities as to the approval or rejection of new or modified products; |
| |
|
|
| |
● |
changes
in the amounts that we spend to develop, acquire or license new products, technologies or businesses; |
| |
|
|
| |
● |
changes
in our expenditures to promote our products; |
| |
|
|
| |
● |
our
sale or proposed sale, or the sale by our significant Shareholders, of our shares or other securities in the future; |
| |
● |
changes
in key personnel; |
| |
|
|
| |
● |
success
or failure of our research and development projects or those of our competitors; |
| |
|
|
| |
● |
the
trading volume of our Shares; and |
| |
|
|
| |
● |
general
economic and market conditions and other factors, including factors unrelated to our operating performance. |
These
factors and any corresponding price fluctuations may materially and adversely affect the market price of our shares and result in substantial
losses being incurred by our investors. In the past, following periods of market volatility, public company Shareholders have often instituted
securities class action litigation. If we were involved in securities litigation, it could impose a substantial cost upon us and divert
the resources and attention of our management from our business.
Future
sales of our shares could reduce the market price of our shares.
If
our existing Shareholders, particularly our management, officers and holders of outstanding Notes sell a substantial number of our shares
in the public market, the market price of our shares could decrease significantly. The perception in the public market that our Shareholders
might sell our shares could also depress the market price of our shares and could impair our future ability to obtain capital, especially
through an offering of equity securities. A large majority of our shares outstanding prior to this offering, including the use of an
if-converted method for all convertible instruments, options and warrants, an additional approximately eighteen million eight hundred
forty-five thousand two hundred ninety (18,845,290) of our shares will be eligible for sale in the public market, all of which will be
subject to restrictions on volume and manner of sale pursuant to Rule 144 under the Securities Act of 1933, as amended. The market price
of our shares may drop significantly when the restrictions on resale by our existing Shareholders lapse and these Shareholders are able
to sell our shares into the market. In addition, a sale by the Company of additional shares or similar securities in order to raise capital
might have a similar negative impact on our share price. A decline in the price of our shares might impede our ability to raise capital
through the issuance of additional shares of Common Stock or other equity securities and may cause you to lose part or all of your investment
in our shares.
We
have broad discretion as to the use of the net proceeds from this offering and may not use them effectively.
We
currently intend to use the net proceeds from this offering to further build our sales and marketing infrastructure, fund research and
development projects and scale up manufacturing and for other general corporate purposes. However, our management will have broad discretion
in the application of the net proceeds. Our Shareholders may not agree with the manner in which our management chooses to allocate the
net proceeds from this offering. The failure by our management to apply these funds effectively could have a material adverse effect
on our business, financial condition and results of operation. Pending their use, we may invest the net proceeds from this offering in
a manner that does not produce income.
The
financial and operational projections that we may make from time to time are subject to inherent risks.
The
projections that we provide herein or our management may provide from time to time (including, but not limited to, those relating to
potential peak sales amounts, clinical and regulatory timelines, production and supply matters, commercial launch dates, and other financial
or operational matters) reflect numerous assumptions made by management, including assumptions with respect to our specific as well as
general business, regulatory, economic, market and financial conditions and other matters, all of which are difficult to predict and
many of which are beyond our control. Accordingly, there is a risk that the assumptions made in preparing the projections, or the projections
themselves, will prove inaccurate. There may be differences between actual and projected results, and actual results may be materially
different from than those contained in the projections. The inclusion of the projections in this Prospectus should not be regarded as
an indication that we, our management, or their representatives considered or consider the projections to be a guaranteed prediction
of future events, and the projections should not be relied upon as such.
An
investment in our Company may involve tax implications, and you are encouraged to consult your own advisors as neither we nor any related
party is offering any tax assurances or guidance regarding our Company or your investment.
An
investment in our Company generally, involves complex federal, state and local income tax considerations. Neither the Internal Revenue
Service (IRS) nor any State or local taxing authority has reviewed the transactions described herein, and may take different positions
than the ones contemplated by management. You are strongly urged to consult your own tax and other advisors prior to investing, as neither
we nor any of our officers, directors or related parties is offering you tax or similar advice, nor are any such persons making any representations
and warranties regarding such matters.
Our
ability to use our net operating loss carry-forwards and certain other tax attributes may be limited.
Under
Section 382 of the Internal Revenue Code of 1986, as amended, referred to as the Internal Revenue Code, if a corporation undergoes an
“ownership change” (generally defined as a greater than 50% change (by value) in its equity ownership over a three-year period),
the corporation’s ability to use its pre-change net operating loss carry-forwards and other pre-change tax attributes (such as
research tax credits) to offset its post-change income may be limited. We may also experience ownership changes in the future as a result
of subsequent shifts in our stock ownership, including as a result of the completion of this offering when it is taken together with
other transactions we may consummate in the succeeding three-year period. As a result, if we earn net taxable income, our ability to
use our pre-change net operating loss carry-forwards to offset U.S. federal taxable income may be subject to limitations, which potentially
could result in increased future tax liability to us.
Our
Certificate of Incorporation permits “blank check” Preferred Stock, which can be designated by our Board of Directors without
stockholder approval.
We
have fifty million (50,000,000) authorized shares of Preferred Stock. The shares of our Preferred Stock may be issued from time to time
in one or more series, each of which shall have a distinctive designation or title as is determined by our Board of Directors prior to
the issuance of any shares thereof. The Preferred Stock may have such voting powers, full or limited, or no voting powers, and such preferences
and relative, participating, optional or other special rights and such qualifications, limitations or restrictions thereof as adopted
by the Board. Because the Board is able to designate the powers and preferences of the Preferred Stock without the vote of a majority
of our stockholders, stockholders will have no control over what designations and preferences our Preferred Stock will have. If Preferred
Stock is designated and issued, then depending upon the designation and preferences, the holders of the Preferred Stock may exercise
voting control over us. As a result, our stockholders will have no control over the designations and preferences of the Preferred Stock
and as a result the operations of our Company.
Our
management collectively owns a substantial majority of our Common Stock.
Collectively,
our officers and Directors own or exercise voting and investment control of approximately sixty-one and 9/10 percent (61.9%) of our outstanding
Common Stock. As a result, investors may be prevented from affecting matters involving our Company, including:
| |
● |
the
composition of our Board and, through it, any determination with respect to our business direction and policies, including the appointment
and removal of officers; |
| |
|
|
| |
● |
any
determinations with respect to mergers or other business combinations; |
| |
|
|
| |
● |
our
acquisition or disposition of assets; and |
| |
|
|
| |
● |
our
corporate financing activities. |
Furthermore,
this concentration of voting power could have the effect of delaying, deterring or preventing a change of control or other business combination
that might otherwise be beneficial to our Stockholders. This significant concentration of share ownership may also adversely affect the
trading price for our Common Stock because investors may perceive disadvantages in owning stock in a company that is controlled by a
small number of stockholders.
If
we fail to establish and maintain an effective system of internal control or disclosure controls and procedures are not effective, we
may not be able to report our financial results accurately and timely or to prevent fraud. Any inability to report and file our financial
results accurately and timely could harm our reputation and adversely impact the trading price of our Common Stock.
Effective
internal controls are necessary for us to provide reliable financial reports and effectively prevent fraud. Section 404 of the Sarbanes-Oxley
Act of 2002 requires us to evaluate and report on our internal controls over financial reporting and, depending on our future growth,
may require our independent registered public accounting firm to annually attest to our evaluation, as well as issue their own opinion
on our internal controls over financial reporting. The process of implementing and maintaining proper internal controls and complying
with Section 404 is expensive and time consuming. We cannot be certain that the measures we will undertake will ensure that we will maintain
adequate controls over our financial processes and reporting in the future. Furthermore, if we are able to rapidly grow our business,
the internal controls that we will need may become more complex, and significantly more resources will be required to ensure our internal
controls remain effective. Failure to implement required controls, or difficulties encountered in their implementation, could harm our
operating results or cause us to fail to meet our reporting obligations. If our auditors or we discover a material weakness in our internal
controls, the disclosure of that fact, even if the weakness is quickly remedied, could diminish investors’ confidence in our financial
statements and harm our stock price. In addition, non-compliance with Section 404 could subject us to a variety of administrative sanctions,
including the suspension of trading, ineligibility for future listing on one of the NASDAQ Stock Markets or national securities exchanges,
and the inability of registered broker-dealers to make a market in our Common Stock, which may reduce our stock price.
If
securities or industry analysts do not publish research or reports about us, our business or our market, or if they make and then change
their recommendations regarding our Common Stock adversely, the price of our Common Stock and trading volume could decline.
The
trading market for our Common Stock, should it develop, may be influenced by the research and reports that securities or industry analysts
may publish about us, our business, our market or our competitors. If any of the analysts who may cover us change their recommendation
regarding our Common Stock adversely, or provide more favorable relative recommendations about our competitors, the price of our Common
Stock would likely decline. If any analyst who may cover us was to cease coverage of our Company or fail to regularly publish reports
on us, we could lose visibility in the financial markets, which in turn could cause the price of our Common Stock or trading volume to
decline.
In
making your investment decision, you should understand that we have not authorized any other party to provide you with information concerning
us or this offering.
You
should carefully evaluate all of the information in this Prospectus before investing in our Company. We may receive media coverage regarding
our Company, including coverage that is not directly attributable to statements made by our officers, that incorrectly reports on statements
made by our officers or employees, or that is misleading as a result of omitting information provided by us, our officers or employees.
We have not authorized any other party to provide you with information concerning us or this offering, and you should not rely on this
information in making an investment decision.
Risks
Related to the Note Financings
Common
Stock that we issue upon conversion of the promissory notes will dilute our existing stockholders and depress the market price of our
Common Stock.
As
of the date of this Prospectus, we are obligated to issue approximately sixteen million eight hundred forty thousand one hundred and
forty-seven (16,840,147) shares of Common Stock upon conversion of the currently outstanding Notes with a limit of ownership of 4.99%.
For the holders, the share total is based upon two million one hundred eighty-nine thousand two hundred nineteen and No/100 Dollars ($2,189,219)
of currently outstanding principal and interest at September 18, 2023, and a conversion price equal to the lesser of (i) $.13 per share
or (ii) 85% of the closing price of Any Qualified Financing, which consists of any fundraising receiving gross proceeds of not less than
five hundred thousand and No/100 Dollars ($500,000).
The
total potential issuable shares increase with the inclusion of additional interest and any decrease in our stock price. As of the date
of this Prospectus, no shares have been issued pursuant to conversion of the Notes and noteholders have not elected to convert any part
of the Notes to date.
The
issuance of shares upon conversion of the notes will dilute our existing Shareholders.
The
holders of the notes convertible into our Common Stock will pay less than the then- prevailing market price for our Common Stock.
The
Notes are convertible at a price equal to the lesser of (i) $.13 per share or (ii) 85% of the closing price of Any Qualified Financing,
which consists of any fundraising receiving gross proceeds of not less than $500,000. As such, the noteholders have a financial incentive
to sell our Common Stock. If the noteholders sell shares, the price of our Common Stock will likely decrease. If our stock price decreases,
the noteholders may have a further incentive to sell the shares of our Common Stock that they hold. These sales may put further downward
pressure on our stock price and reduce the value of your Common shares.
The
price of the Common Stock we are selling under this Offering is significantly higher than the conversion price of the Notes and the price
of our Common Stock would likely drop to or below the conversion price of the Notes upon conversion by the noteholders.
In
the event that the noteholders convert the Notes into Common Stock, the conversion price is significantly lower than the price at which
we are selling our Common Stock in this offering. As a result, the sale by the noteholders of our Common Stock could drive the market
price down to the conversion price. This could result in the purchaser of our Common Stock in this offering loose a substantial portion
of his or her investment.
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This
Prospectus contains a number of “forward-looking statements”. Specifically, all statements other than statements of historical
facts included in this Prospectus regarding our financial position, business strategy and plans and objectives of management for future
operations are forward-looking statements. These forward-looking statements are based on the beliefs of management at the time these
statements were made, as well as assumptions made by and information currently available to management. When used in this Prospectus
and the documents incorporated by reference herein, the words “anticipate,” “believe,” “estimate,”
“expect,” “may,” “will,” “continue” and “intend,” and words or phrases of
similar import, as they relate to our financial position, business strategy and plans, or objectives of management, are intended to identify
forward-looking statements. These statements reflect our current view with respect to future events and are subject to risks, uncertainties
and assumptions related to various factors.
You
should understand that the following important factors, in addition to those discussed in our periodic reports to be filed with the SEC
under the Exchange Act, could affect our future results and could cause those results to differ materially from those expressed in such
forward-looking statements:
| |
● |
We
expect to incur losses for the foreseeable future and may never achieve or maintain profitability. |
| |
|
|
| |
● |
We
are a company with limited operating history which makes it difficult to evaluate our current business and future prospects. |
| |
|
|
| |
● |
We
will require additional financing to implement our business plan may not be available on favorable terms or at all, and we may have
to accept financing terms that would adversely affect our stockholders. |
| |
|
|
| |
● |
Raising
additional capital may cause dilution to our stockholders, restrict our operations or require us to relinquish rights to our drug
candidates and dietary supplements. |
| |
● |
Our
products are based on novel, unproven technologies. |
| |
|
|
| |
● |
Clinical
drug development involves a lengthy and expensive process, with an uncertain outcome. We may incur additional costs or experience
delays in completing, or ultimately be unable to complete, the development and commercialization of our drug candidates. |
| |
|
|
| |
● |
We
may be unable to commercialize our drug candidates |
| |
|
|
| |
● |
Our
success depends upon our ability to retain key executives and to attract, retain, and motivate qualified personnel and direction
and the loss of these persons could adversely affect our operations and results. |
| |
|
|
| |
● |
We
will need regulatory approvals to commercialize our products as drugs. |
| |
|
|
| |
● |
Our
competitive position depends on protection of our intellectual property. |
| |
|
|
| |
● |
The
market for our proposed products is rapidly changing and competitive, and new drugs and new treatments which may be developed by
others could impair our ability to maintain and grow our business and remain competitive. |
| |
|
|
| |
● |
We
may become involved in lawsuits to protect or enforce patents that may issue to us, that we may acquire, or may license in the future,
or other intellectual property, which could be expensive, time-consuming and ultimately unsuccessful. |
| |
|
|
| |
● |
The
market price of our Common Stock may be highly volatile, and you could lose all or part of your investment. |
| |
|
|
| |
● |
There
is no market, and no market may develop, for our Common Stock, which makes our securities very speculative. |
| |
|
|
| |
● |
You
will experience immediate and substantial dilution as a result of this offering and may experience additional dilution in the future. |
| |
|
|
| |
● |
Our
management will have broad discretion in how we use the net proceeds of this offering. |
| |
|
|
| |
● |
As
a public company, we must implement additional and expensive finance and accounting systems, procedures and controls as we grow our
business and organization to satisfy new reporting requirements, which will increase our costs and require additional management
resources. |
Although
we believe that our expectations (including those on which our forward-looking statements are based) are reasonable, we cannot assure
you that those expectations will prove to be correct. Should any one or more of these risks or uncertainties materialize, or should any
underlying assumptions prove incorrect, actual results may vary materially from those described in our forward-looking statements as
anticipated, believed, estimated, expected or intended.
Except
for our ongoing obligations to disclose material information under the federal securities laws, we undertake no obligation to publicly
update or revise any forward-looking statements, whether as a result of new information, future events or any other reason. All subsequent
forward-looking statements attributable to us or any person acting on our behalf are expressly qualified in their entirety by the cautionary
statements contained or referred to herein. In light of these risks, uncertainties and assumptions, the forward-looking events discussed
in this Prospectus and the documents incorporated by reference herein might not occur.
USE
OF PROCEEDS
We
may receive additional cash proceeds equal to the total exercise price of the Warrants to the extent any of such Warrants are exercised
for cash. The exercise price of the Warrant Shares is $0.14 per share of Common Stock. The exercise price and the number of shares of
Common Stock issuable upon exercise of the Warrants may be adjusted in certain circumstances, including stock splits, dividends or distributions,
or other similar transactions. The Warrants contain a “cashless exercise” feature that allows the holders to exercise any
of such Warrants without making a cash payment to us if there is not an effective registration statement covering the resale of the shares
issuable upon exercise of such Warrants. There can be no assurance that any of the Warrants will be exercised by the Selling Stockholder
at all. The cash received from the Offering Shares, and to the extent we receive proceeds from the cash exercise of the Warrants, is
intended to be used for working capital purposes, the conduct of our business and other general corporate purposes, which may include
acquisitions, investments in or licenses of complementary products, technologies or businesses, operating expenses and capital expenditures.
Our management will retain broad discretion in the allocation of the net proceeds from any exercise of the Warrants for cash.
DIVIDEND
POLICY
To
date, we have not declared or paid any dividends on our outstanding shares. We currently do not anticipate paying any cash dividends
in the foreseeable future on our Common Stock. Although we intend to retain our earnings to finance our operations and future growth,
our Board of Directors will have discretion to declare and pay dividends in the future. Payment of dividends in the future will depend
upon our earnings, capital requirements and other factors, which our Board of Directors may deem relevant.
LEGAL
PROCEEDINGS
The
Company may become involved in certain legal proceedings and claims which arise in the normal course of business.
On
June 5, 2020 the Supreme Court of the State of New York, County of Nassau, issued a Commencement of Action on behalf of Power Up Lending
Group, Ltd (“Power Up” or the “Claimant”). The Claimant requested, due to the default of its note, a judgment
for an amount of not less than four hundred twenty thousand seven hundred fifty Dollars ($420,750). Among other claims Power Up asserted
that the Company willfully failed to maintain the trading status, and manipulated its stock in its efforts to defraud the public and
its investors by making false press statements and the like. The Company denied any wrong-doing. On January 20, 2021 the Supreme Court
of the State of New York, County of Nassau, granted Power Up a summary judgement against the Company for breach of contract, awarding
Power Up damages in the amount of $420,750.
The
underlying convertible note was, per agreement of the parties, cancelled on June 4, 2021, with Power Up agreeing to a stipulation of
discontinuance with prejudice of the law-suite and forfeiture of earlier awarded damages.
DIRECTORS,
EXECUTIVE OFFICERS, PROMOTERS AND CONTROL PERSONS
Our
Board of Directors, executive officers and key employees are as follows:
| Name | |
Age as at September 18, 2023 | | |
Position |
| David Platt, Ph.D. | |
| 70 | | |
Chief Executive Officer, Chairman and Director |
| Ola Soderquist, MBA, CPA, CMA | |
| 62 | | |
Chief Financial Officer, Treasurer, Secretary |
| Mike Sheikh, BS | |
| 53 | | |
Chief Marketing Officer (CMO) |
| Dale H. Conaway, D.V.M. | |
| 68 | | |
Independent Director |
| Alan M. Hoberman. Ph.D. | |
| 70 | | |
Independent Director |
| Hana Chen-Walden, MD | |
| 76 | | |
Independent Director |
| Anders Utter, MBA | |
| 56 | | |
Independent Director |
David
Platt, Ph.D. is the Chief Executive Officer and Chairman of the Board. Dr. Platt is a world-renowned expert in carbohydrate chemistry
and has founded three publicly traded companies, creating nearly $1B for investors. He has raised $150M directly in public markets in
the U.S. and has led development of two drug candidates from concept through phase II clinical trials. Prior to Bioxytran, Inc. Dr. Platt
founded Boston Therapeutics Inc. in 2010 (OTC: BTHE) where he served as Chief Executive Officer from 2010 to April 1, 2015 and as a Director
from March 2015, to June 8, 2016. From 2001 to 2009, Dr. Platt was a founder, Chief Executive Officer and Chairman of the Board at Pro-Pharmaceuticals,
Inc. (OTC: PRWP and AMEX: PRW, now NASDAQ: GALT). From 1995 to 2000 Dr. Platt was the founder of International Gene Group (NASDAQ: IGGI,
GLGS now LPJC). Dr. Platt received a Ph.D. in Chemistry in 1988 from Hebrew University in Jerusalem. In 1989, Dr. Platt was a research
fellow at the Weizmann Institute of Science, Rehovot, Israel, and from 1989 to 1991, was a research fellow at the Michigan Foundation
(re-named Barbara Ann Karmanos Institute). From 1991 to 1992, Dr. Platt was a research scientist with the Department of Internal Medicine
at the University of Michigan. Dr. Platt has published peer-reviewed articles and holds many patents, primarily in the field of carbohydrate
chemistry. Our Board believes that Dr. Platt’s expertise and experience with public biotech companies, his perspective, depth and
background in chemistry and finance, the capital formation process and leadership experience in public companies provide him with the
qualifications and skills to serve on our Board.
Ola
Soderquist, MBA, CPA, CMA, CM&AA is the Chief Financial Officer, Treasurer and Secretary. He has more than 30 years of senior
international entrepreneurial management experience within technology companies. Mr. Soderquist’s managerial experience portfolio
includes; startups, private, public, venture capital and private equity ownership. He has served as CFO and in other managerial capacities
in multiple industry sectors and companies. His public company tenures include companies in the Wallenberg Sphere (1986-1996); Industrivarden
(OMX:INDU), Electrolux (OMX:ELUX), Ericsson (NASDAQ:ERIC), Swedish Match (OMX:SWMA) and SKF AB (OMX:SKF). He also held positions in Traction
(OMX:TRAC) (1996-2001) and Belden (NYSE: BDC) (2006-2011). His private company experience includes CFO and CAO positions in Proditec,
Inc. (2001-2006), LFA Corp. (2012-2014) and Faria Beede Instruments, Inc. (2014-2016). Mr. Soderquist is a multi-lingual senior finance
professional poised to work globally and cross-functionally, particularly with complex projects involving change management, business
integration, systems implementation, continuous improvement, and process excellence. He obtained a BS and an MSA rom Stockholm School
of Economics and an MBA from Babson College.
Mike
Sheikh, BS, is the Chief Communications Officer. He is a US Air Force Academy graduate and pilot. He has a Bachelor of Science
in Economics, flew KC-135 tankers and worked as a budget officer in the comptroller’s squadron. Mr. Sheikh has prior experience
as a broker and research analyst. After the brokerage industry, he was a business development officer for a variety of specialty finance
companies. Mr. Sheikh is a long-time biotech consulting expert for public and private biotech companies with disruptive technologies.
Mr. Sheikh is the founder of Falcon Strategic Research, which focuses on companies that are not covered by traditional analysts on Wall
Street. He is also the founder of an investor relations firm.
Dale
H. Conaway, D.V.M., is a Director of the Company. Dr Conaway is a Veterinary Medical Officer in federal research. From 2010 to
September 15, 2016, Dr. Conaway served as a member of the Board of Directors of Boston Therapeutics, Inc. From 2001 to 2006, Dr. Conaway
was the Deputy Regional Director (Southern Region). From 1998 to 2001, Dr. Conaway served as Manager of the Equine Drug Testing and Animal
Disease Surveillance Laboratories for the Michigan Department of Agriculture. From 1994 to 1998, he was Regulatory Affairs Manager for
the Michigan Department of Public Health Vaccine Production Division. Dr. Conaway received a D.V.M. degree from Tuskegee Institute and
an M.S. degree in pathology from the College of Veterinary Medicine at Michigan State University. Our Board believes that Dr. Conway’s
expertise and experience as a Director in a public biotech company, his perspective, depth and background in testing and the development
of biologic compounds, and his leadership in management provide him with the qualifications and skills to serve on our Board.
Alan
M. Hoberman, Ph.D. is a Director of the Company. Dr. Hoberman is president and CEO of Argus International, Inc., overseeing a
staff of scientists and other professionals who provide consulting services for industry, government agencies, law firms and other organizations,
both in the U.S. and internationally. From 2014 to September 15, 2016, Dr. Hoberman served as a member of the Board of Directors of Boston
Therapeutics, Inc. Between 1991 and 2013 he held a series of positions of increasing responsibility at Charles River Laboratories Preclinical
Services (formerly, Argus Research Laboratories, Inc.), most recently as Executive Director of Site Operations and Toxicology. He currently
works with that organization to design, supervise and evaluate reproductive and developmental toxicity, neurotoxicity, inhalation and
photobiology studies. Dr. Hoberman holds a PhD in toxicology from Pacific Western University, an MS in interdisciplinary toxicology from
the University of Arkansas and a BS in biology from Drexel University. Our Board believes that Dr. Hoberman’s expertise and experience
as a Director in a public biotech company, his perspective, depth and background in consulting and advising clients and his experience
in the testing and development of biologic compounds, and his leadership in management provide him with the qualifications and skills
to serve on our Board.
Dr.
Hana Chen-Walden, M.D. is a Director of the Company. Dr. Chen-Walden is an Endocrinologist and has specialized in regulatory
affairs in the pharmaceutical industry in the US and Europe. Dr. Chen-Walden has more than 35 years of regulatory experience with the
EMEA and in individual European countries. Since 2004, Dr. Chen-Walden has consulted for European Clinical and Regulatory Consultancy
in medical monitoring, quality assurance and regulatory input for clinical studies in the fields of oncology, cardiology, diabetes, neurology,
respiratory diseases and medical devices. Dr. Chen-Walden received her Doctor of Medicine from University of Tel Aviv, Israel. Dr. Chen-Walden
has practiced medicine in Germany and France. Our Board believes that Dr. Chen-Walden’s expertise and experience in practicing
medicine, her perspective, depth and background in medical monitoring and quality assurance, and her leadership in regulatory affairs
provide her with the qualifications and skills to serve on our Board.
Anders
N. Utter, is a Director of the Company. He has more than 25 years of finance, accounting and management experience in medical
devices, consulting and manufacturing industries in capacities as CFO, Controller and Managing Director. He had progressively increased
management experience in the European Nolato Group and later on in the Amplex Group. Mr. Utter has had a broad business exposure with
IFRS and GAAP reporting as well as with SOX compliance. He has also worked with M&A evaluations, financing and integration as well
as more hands-on manufacturing cost accounting and reporting. He is currently in charge of the finance control at one of General Cable’s
entities. Mr. Utter is and has been serving as a Director on boards in both profit as well as non-profit organizations. Mr. Utter holds
an MBA from Babson College and a BA from Uppsala University in Sweden. Our Board believes that Mr. Utter’s expertise and experience
as a chief financial officer, his perspective, depth and background in GAAP reporting and SOX compliance, and his finance, management
and accounting experience provide him with the qualifications and skills to serve on our Board.
Our
Directors are elected annually and each holds office until the annual meeting of the Shareholders of the Company and until their respective
successors are elected and qualified. Our officers, including any officers we may elect moving forward, will hold their positions at
the pleasure of the Board, absent any employment agreement. In the event, we employ any additional officers or Directors, they may receive
compensation as determined by the Company from time to time by vote of the Board. Vacancies in the Board will be filled by majority vote
of the remaining Directors or in the event that a sole remaining Director vacates his position, by our majority Shareholders. Our Directors
may be reimbursed by the Company for expenses incurred in attending meetings of the Board.
Scientific
Advisory Board
We
are establishing a scientific advisory board to advise our management regarding our clinical and regulatory development programs and
other customary matters. Our scientific advisors are experts in various areas at medicine including diabetes and other diseases. We believe
the advice of our scientific advisors is important to the research, development and clinical testing of our products. Our scientific
advisory board is comprised of the following individuals.
Prof.
Avraham Mayevsky, Ph.D. is a worldwide authority in the field of minimal invasive monitoring of tissue and organ physiology.
Dr. Mayevsky is a professor at the Faculty of Life Sciences, Bar-Ilan University, Israel. He served as Head of the Department of Life
Sciences and Dean of the Faculty of Natural Sciences at Bar-Ilan University, where he established a center of tissue physiology. He served
as Visiting professor at University of Pennsylvania and Johns Hopkins Medical School World-recognized expert in tissue physiology, especially
in brain metabolism. He Published over 150 papers and patents. He has published over 170 papers in scientific journals and is the author
of five patents. He also founded Vital Medical Ltd. Dr. Mayevsky completed his PhD from Weizmann Institute of Science, Rehovot, Israel.
Prof.
Kevin H Mayo, Ph.D. is a well-known authority in the field of structural biology and structure-based drug design and discovery.
He received degrees from Boston University (BA) and the University of Massachusetts (PhD), and was a postdoctoral associate at the Max-Planck
Institute for Biochemistry (Alexander von Humboldt Fellow with Nobel Laureate Rudolf Moessbauer) and Yale University (Chemistry). Dr.
Mayo is presently Professor of Biochemistry, Molecular Biology & Biophysics, as well as Lab Medicine & Pathology, at the University
of Minnesota (UMN), Minneapolis, USA. He is also Director of the High Field Nuclear Magnetic Resonance Center at the UMN. Over the years,
Dr. Mayo has consulted with numerous pharmaceutical companies and is co-founder of PepTx, Inc., a startup pharmaceutical company based
in Minnesota. He also currently holds Visiting Professorships at Maastricht University (The Netherlands), Ludwigs-Maximillian-University
(Munich, Germany), and Northeast Normal University (Changchun, China). Dr. Mayo has published over 250 papers in peer-reviewed scientific
journals and is the author of 28 patents.
Medical
Advisory Board
We
are establishing a Medical Advisory Board that will be comprised of Clinicians and Clinical Research professionals who are interested
in the field of hypoxia, virology or in other subjects related to our product pipeline. The board will provide leadership and expertise
to assist us in designing, executing and implementing our clinically oriented activities in a safe, efficient and professional manner.
Dr.
Leslie Ajayi, MD PhD, brings over 20 years of clinical development experience in academia and industry. He is a fully trained
physician leader with international specialty training in internal medicine, cardiovascular medicine, and clinical pharmacology. He received
his undergraduate training in Health Sciences and his MD equivalent graduating Magna Cum Lade from Obafemi Awolowo University [OAU] in
Nigeria. A few years later, he received his PhD in clinical pharmacology from the University of Glasgow. As an academic clinical pharmacologist
in Glasgow, UK, he worked with Big Pharma as an investigator for Phase 1 first in man, proof of concept, pharmacokinetics (PK), Pharmacodynamics
(PD), PK-PD, and studies in special populations such as the elderly and in pregnancy. He was also involved in all types of designs of
randomized controlled clinical trials (double blind, placebo controlled, double dummy, single blind, cross over, parallel group, Latin
squares designs). His industry exposure was relegated to big pharma clinical research monitors and clinical research organizations. He
worked on notable projects like perindopril and cilazapril (ACEI), and amlodipine. He evaluated the effects of ACEI on Type-2 Diabetes
and insulin resistance in hypertensives.
Dr.
John Mabayoje, MD, is a practicing Emergency Room doctor and Medical Director who graduated from the University of Ife /OAU in
1980. He has 6 years of residency/ fellowship training in internal medicine, family practice, geriatric medicine, substance abuse, and
emergency medicine. He also has 125 hours of sonography training. He is licensed to practice in a number of states and has 44 years’
experience in emergency medicine in the United States and internationally. He has published research work on histochemistry. He has extensive
experience with COVID-19 patients, treating over 4,800 patients on 2 continents. He is known in circles as an astute diagnostician and
innovator looking for ways to getting the best therapeutic advantages for his patients.
Dr.
Alben Sigamani, M.D. is currently Professor and Head of Clinical Research, Narayan Health, Bangalore. He has over 17 years of
experience in clinical research and in managing multi-center academic and regulatory Randomized Controlled Trials in India. He has several
publications to his credit with a citation index (h-index) of 24. Dr. Sigamani is a Medical Professional (MD) in Clinical Pharmacology
& Therapeutics with a Master’s Degree in Clinical Trials from the University of London. In 2021, Dr. Sigamani obtained “COVID-19:
Tracking the Novel Coronavirus Certificate” from the London School of Hygiene and Tropical Medicine.
Thomaskutty
Alumparambil. B.S., C.C.P has over 30 years of clinical experience that includes heart, lung and liver transplants. He is an
expert on quality control and quality assurance programs, surgical protocols, blood gas analysis and anticoagulation management.
Employment
Agreements
Our
officers have entered into employment agreements and confidentiality, non-disclosure and assignment of inventions agreements with the
Company which include, among other things, provisions which restrict any of them from selling any shares of the Company Common Stock
in the 180 days following the effective date of this registration statement. Other than provisions in the employment agreements, there
are no arrangements or plans in which we provide pension, retirement or similar benefits for our officers or Directors. Our officers
and Directors may receive stock options at the discretion of our Board in the future. We do not have any bonus or profit-sharing plans
pursuant to which cash or non-cash compensation is or may be paid to our officers or Directors, except that stock options may be granted
at the discretion of our Board from time to time.
Change
in Control and Severance Payments
Under
the terms of their employment agreements, our executive officers are entitled to receive certain payments upon the termination without
cause from their employment. The most substantial provisions include;
| |
● |
Compensation
of three (3) times the employee’s annual salary upon the Termination Date and any target bonus earned, or if termination occurs
within twelve (12) months of a change in control, then the terminated employee shall receive two (2) times the employee’s annual
salary and any target bonus earned. |
| |
● |
Continued
coverage under any health, medical, dental or vision program or policy, in which they were eligible to participate at the time of
employment termination, for twelve (12) months. |
| |
● |
Provide
outplacement services through one or more outside firms of the employee’s choosing up to an aggregate of fifty thousand Dollars
($50,000). |
There
are no other arrangements or plans in which we provide pension, retirement or similar benefits for any of Executive Officers or Directors.
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The
following table sets forth certain information as at March 30, 2023 with respect to the beneficial ownership of shares of the Company’s
Common Stock by (i) each person or group known to us, to beneficially own more than five percent (5%) of the outstanding shares of such
stock, (ii) each Director; (iii) each of our Executive Officers named in the summary compensation table under “Director and Executive
Compensation” currently serving as an Executive Officer; and (iv) the Executive Officers and Directors as a group. All persons
listed below have (i) sole voting power and investment power with respect to their shares of Common Stock (the only class of outstanding
stock), except to the extent that authority is shared by spouses under applicable law, and (ii) record and beneficial ownership with
respect to their shares of stock. The percentage of beneficial ownership is based upon one hundred forty-three million seven hundred
eighty-four thousand nine hundred eighty-five (143,784,985) shares of Common Stock outstanding as at September 18, 2023. Except as otherwise
indicated in the footnotes to the table, the persons and entities named in the table have sole voting and investment power with respect
to all shares beneficially owned, subject to community property laws, where applicable.
| Name and Address of Beneficial Owner | |
Number of Shares | | |
Percent of Class (1) | | |
Number of
Options owned (2) | |
David Platt (3)
whereof 16,505,434 indirect | |
| 59,532,708 | | |
| 41.4 | % | |
| — | |
| | |
| | | |
| | | |
| | |
| Ola Soderquist (3) | |
| 19,535,300 | | |
| 13.6 | % | |
| — | |
| | |
| | | |
| | | |
| | |
| Mike Sheikh (3) | |
| 8,000,000 | | |
| 5.6 | % | |
| — | |
| | |
| | | |
| | | |
| | |
| Dale H. Conaway (3) | |
| 510,800 | | |
| 0.4 | % | |
| — | |
| | |
| | | |
| | | |
| | |
| Alan M. Hoberman (3) | |
| 584,100 | | |
| 0.4 | % | |
| — | |
| | |
| | | |
| | | |
| | |
| Hana Chen-Walden, MD (3) | |
| 367,800 | | |
| 0.3 | % | |
| 3,000 | |
| | |
| | | |
| | | |
| | |
| Anders Utter (3) | |
| 529,900 | | |
| 0.4 | % | |
| — | |
| | |
| | | |
| | | |
| | |
| All Officers and Directors as a Group (7 persons) | |
| 89,060,608 | | |
| 61.9 | % | |
| 3,000 | |
| (1) |
The
percentage shown in the table is based on 143,784,985 shares of Common Stock outstanding on September 18, 2023. |
| (2) |
The
options have an average remaining term of 0.1 years with an average exercise price of $0.25. |
| (3) |
The
business address of these individuals is 75 2nd Ave., Suite 605, Needham, MA 02494. |
DESCRIPTION
OF BUSINESS
Overview
Bioxytran,
Inc. (“we”, “us”, or the “Company”) is a clinical stage pharmaceutical company focused on the development,
manufacture and commercialization of therapeutic drugs designed to address hypoxia (a lack of oxygen to tissues) in humans. Hypoxia,
needs to be addressed quickly, otherwise it results in necrosis, the death of cells comprising body tissue which cannot be reversed.
Our lead drug candidate, a hypoxia platform technology with code-name BXT-25, is planned to be an Acellular Oxygen Carrier (“AOC”)
consisting of bovine hemoglobin stabilized with a co-polymer with an intended application that includes the treatment of hypoxic conditions
in the brain resulting from stroke. We believe that our approach is novel when applied to hypoxic conditions in humans. Our drug development
efforts are guided by specialists who work on co-polymer chemistry and other disciplines. We intend to supplement our efforts with input
from a Scientific and a Medical Advisory Board whose members are leading physicians.
The
Company was organized on June 9, 2008, as a Nevada corporation.
Our
subsidiary, Pharmalectin Inc. (“Pharmalectin” or the “Subsidiary”), of which we currently have 85% ownership,
is focused on the development, manufacturing and commercialization of therapeutic drugs designed to address viral diseases in humans.
Pharmalectin has developed a novel method designed to reduce the viral load and modulate the immune system using a galectin antagonist.
Our lead drug candidate is a glyco-virology platform technology named ProLectin, a complex polysaccharide that binds to, and blocks the
activity of galectin-3, a type of galectin. Galectins are a member of a family of proteins in the body called lectins. Lectins interact
with carbohydrate sugars located in, on the surface of, and in between cells. This interaction causes the cells to change behavior, including
cell movement, multiplication, and other cellular functions. The interactions between lectins and their target carbohydrate sugars occur
via a carbohydrate recognition domain, or CRD, within the lectin. Galectins are a subfamily of lectins that have a CRD that bind specifically
to ß-galactoside proteins. Galectins have a broad range of functions, including regulation of cell survival and adhesion, promotion
of cell-to-cell interactions, growth of blood vessels, regulation of the immune response and inflammation. During viral infections galectins
are upregulated and downregulated based on the type of virus.
In
the past, similar types of carbohydrate substances have been used as a fibrosis drug and a cancer drug, but are currently being reformulated
to treat viral infections. We believe that we have a novel approach in treating viral infections in humans. Our drug development efforts
are guided by specialists on carbohydrate chemistry and other disciplines, and we intend to supplement our efforts with input from the
Scientific and the Medical Advisory Boards.
We
plan to file a pre-investigational new drug application for ProLectin for the treatment of mild to moderate Covid-19 patients. However,
we cannot provide any assurance that we will successfully initiate or complete those planned trials and be able to initiate any other
clinical trials for ProLectin or any of our future drug candidates.
Pharmalectin
was organized on October 5, 2017, as a Delaware corporation with its principal place of business in Needham, MA.
Our
subsidiary, Pharmalectin (BVI), Inc. (“Pharmalectin (BVI)”) is the owner and custodian of the Company’s Copyrights,
Trademarks and Patents. Pharmalectin (BVI) was organized on March 17, 2021 as a British Virgin Islands (BVI) Business Corporation with
its principal place of business in Road Town, BVI.
Our
subsidiary, Pharmalectin India Pvt Ltd. (“Pharmalectin India”) is managing the Company’s local clinical research and
trials, and holds the local commercialization rights. Pharmalectin India was organized on August 30, 2022, as an Indian Business Corporation
with its principal place of business in Hyderabad, Telangana, India.
Company
Overview
We
are a clinical stage pharmaceutical company founded on June 9, 2008 as America’s Driving Ranges, Inc.. On September 21, 2018, the
Company was reorganized into Bioxytran through a reverse merger to focus on the development, manufacturing and commercialization of therapeutic
drugs designed to address hypoxia in humans. Our initial focus is the treatment of hypoxic conditions in the brain resulting from stroke
and through our subsidiary, Pharmalectin, in the treatment of viral diseases, notably Covid-19.
Currently,
the Company’s lead pharmaceutical drug candidate, BXT-25 is planned to be an Acellular Oxygen Carrier (“AOC”) consisting
of bovine hemoglobin stabilized with a co-polymer. This modified hemoglobin will be designed to be an injectable intravenous drug and
we plan to begin pre-clinical studies and apply to the Food and Drug Administration (FDA) for approval to use BXT-25 to prevent necrosis
by carrying oxygen to human tissue with blood flow to the brain. If we successfully complete Phase I testing with the FDA, we plan to
explore the use of additional drug candidates using chemical structures that are a sub-class of BXT-25 and share the same physical properties
to treat wound healing due to hypoxia, cardiovascular ischemia, anemia, cancer conditions and trauma, subject to FDA approval. However,
we will need to raise additional funds in excess of ten million Dollars ($10,000,000) in order to expand the use of BXT-25.
BXT-25
is a novel unproven technology. Although we have not conducted research applying our co-polymer technology and related chemistry to the
treatment of hypoxic conditions, we know from Dr. Platt’s prior research that our technology enables the creation of molecules
that are 5,000 times smaller than human red blood cells and we believe that our proprietary technology will enable these molecules to
carry oxygen for delivery to tissue through the bloodstream. We also believe that the small size of these molecules will more effectively
enable their delivery to hypoxic tissues which red blood cells cannot reach under the clinical conditions we intend to address. We may
be unsuccessful in developing these technologies into drugs which the FDA ultimately will approve.
Stroke
Stroke,
also known as cerebrovascular accident (CVA), or brain attack, occurs when poor blood-flow to the brain results in necrosis and cell
death. Strokes can be classified into two major categories: ischemic and hemorrhagic. Ischemic strokes are caused by interruption of
the blood supply to the brain. Hemorrhagic strokes result from the rupture of a blood vessel or an abnormal vascular structure. According
to the Center for Disease Control (CDC), approximately 87% of all strokes are ischemic. An ischemic stroke may be thrombotic, which occurs
when diseased or damaged cerebral arteries become blocked by the formation of a blood clot within the brain, or embolic, which occurs
when a clot formed originally somewhere in the body outside the brain - typically in the heart - travels in a cerebral artery. Whether
thrombotic or embolic, an ischemic stroke restricts the flow of blood to the brain and results in near-immediate physical and neurological
deficits.
According
to the CDC, there are about 795,000 cases of stroke in the United States each year, of which 610,000 are new and 185,000 recurrent cases.
130,000 Americans are killed by stroke each year, or one every four minutes. Stroke is a leading cause of serious long-term disability
and costs the United States an estimated $34 Billion each year, according to the CDC, a figure which includes the cost of health care
services, medications to treat the stroke, and missed days of work.
Hemoglobin
and Complex Co-Polymer Science
Oxygen
therapeutics describe generally a class of agents that will be administered intravenously to enhance the oxygen delivery capability of
blood. These oxygen transporting agents may be perfluorocarbon (PFC) emulsions or modified hemoglobin solutions. Our technology involves
the development of hemoglobin-based oxygen carriers. To produce BXT-25, we will take red blood cells (RBCs) from bovine sources, isolate
hemoglobin from the RBCs and, by applying our proprietary co-polymer chemistry, stabilize and modify the hemoglobin. Our novel, complex
co-polymer molecules can be produced at specific molecular weights and with other pharmaceutical properties for various hypoxic diseases;
and in the production of BXT-25.
The
BXT-25 co-polymer hemoglobin molecule will be designed to be 5,000 times smaller than an RBC, which we believe will enable that small
molecule to reach hypoxic tissue more effectively than RBCs. BXT-25 will be designed to be administered as an injectable IV drug that
will circulate in the blood collecting oxygen from the lungs and releasing the oxygen molecules where tissue has developed ischemia,
or lack of oxygen. BXT-25 will be designed to have oxygen affinity that mimics RBCs, minimize adverse effects, and be compatible with
all blood types. BXT will be designed to have a shelf life of two years at room temperature.
We
believe that the differences between a BXT-25 molecule and a red blood cell will not be limited to differences in size. Surfaces of red
blood cells include different antigens which determine the blood type as A, B, AB or O. We believe that BXT-25 will be found to be compatible
with all blood types because it is a single, modified hemoglobin molecule stabilized with a co-polymer which, unlike a red blood cell,
has neither antigens nor a Rh factor.
Certain
regulatory issues relating to our use of bovine hemoglobin as a raw material
Our
products include a commercially available raw material, bovine hemoglobin, that has been purified, chemically modified and cross-linked
for stability. It is sourced from controlled herds of U.S. cattle raised for beef production. Those herds are subject to and meet the
requirements of a herd management program that assures the origin, health, feed and quality of the cattle used as a raw material source.
Our suppliers will contract to maintain traceable records on animal origin, health, feed and care as part of our effort to assure the
use of known, healthy animals in compliance with applicable laws and regulations.
Prior
to the collection of blood, the animals undergo live inspection. Bovine whole blood will be collected in individual pre-sanitized containers.
The containers will be shipped to a separation facility. Then, following blood collection, the animal carcass undergoes U.S. Department
of Agriculture (USDA) inspection for use as beef for human consumption. If an animal carcass is retained for further inspection for final
disposition by the USDA veterinarian, we reject the corresponding container of whole blood. We have validated and tested the processes
described below for removal of potential pathogens in our raw material. Potential pathogens include bacteria, viruses such as those leading
to hepatitis and AIDS, and the transmissible spongiform encephalopathies that cause rare neurological disorders such as “mad cow
disease” and its human equivalent. The validation of a process means that it has been tested and documented and that it performs
adequately. Health and regulatory authorities have given guidance directed at three factors to control these diseases: source of animals,
the nature of tissue used and manufacturing process. We will comply with, and believe we will exceed, all current guidelines regarding
such risks for human pharmaceutical products.
There
will be four major steps in the manufacture of BXT-25: (1) hemoglobin separation; (2) hemoglobin purification; (3) polymerization/size
selection and (4) synthesizing with our co-polymer. More specifically, bovine blood will be collected in an aseptic fashion and processed
to first remove plasma and then to remove at high concentration the hemoglobin protein from red blood cells. The hemoglobin will be purified
of other red cell proteins by anion exchange chromatography. The purified hemoglobin will be stabilized by the addition of a cross-linking
agent to form hemoglobin polymers. There is an additional sizing step to remove the higher hemoglobin molecules. The final step, co-polymer
synthesis, will take place on the stabilized hemoglobin. The combination polymers will be filled with a solution suitable for infusion.
The product will be run through sterilizing filters into sterile product bags.
Pharmalectin
The
Subsidiary was organized on October 5, 2017, as a Delaware corporation under the name of Bioxytran “Bioxytran (DE)”. On April
29, 2021, the name was changed to Pharmalectin. Through the Subsidiary, we are not a party to any long-term agreement with any of our
suppliers and, accordingly, we have our products manufactured on a purchase-order basis from one of two primary well-known and established
pharmaceutical suppliers that meet FDA requirements. Due to an overwhelming amount of research on galectins we do not plan on conducting
any further research into new molecules. Instead, we intend to apply our knowledge of galectin science and drug development to create
new therapies for the treatment of viruses.
Covid-19
We
are currently working on an end-to-end solution for Covid-19 mild to severe cases and treatment for organ damage caused by the virus
or by commonly used treatment methods.
| |
● |
ProLectin-M,
a chewable polysaccharide tablet for mild to moderate cases of Covid-19. |
| |
● |
ProLectin-I,
a polysaccharide IV treatment for more severe cases of Covid-19. |
| |
● |
ProLectin-F,
a polysaccharide IV treatment of lung-fibrosis as a result of the use of ventilators used for treatment of Covid-19. |
| |
● |
ProLectin-A,
a polysaccharide and Hemoglobin IV treatment of ARDS as a result of Covid-19. |
Using
our issued patents and proprietary technology coupled with the scientific knowledge and expertise of Dr. Platt, we intend to develop
and manufacture ProLectin-M (oral) for treatment of mild cases and ProLectin-I (intravenous) for treatment of more severe cases of Covid-19.
These treatments may also be used for the treatment of other types of viral infections, such as influenza.
A
significant problem related to the Covid-19 pandemic is that an increasing number of patients are developing life-threatening complications,
such as ARDS, shock (i.e., a potentially fatal drop in blood pressure), kidney failure, acute cardiac injury and secondary bacterial
infections. The underlying cause for these complications is often a cytokine storm that results in a massive, systemic inflammatory response,
leading to the damage of vital organs such as the lungs, heart, and kidneys, and ultimately multiple organ failure and death in many
cases. For this purpose, we are developing ProLectin-A that aims to deliver oxygen to damaged organs and at the same time fight infection.
The
fourth drug in this series, ProLectin-F, is being developed to treat patients developing lung fibrosis as a result of the use of ventilator
in Covid-19 treatment. Increasing evidence from experimental and clinical studies suggests that mechanical ventilation, which is necessary
for life support in patients with acute respiratory distress syndrome, can cause lung fibrosis, which may significantly contribute to
morbidity and mortality. According to a review of medical records of 22,350 admissions showed that the cost of treating patients who
were put on a ventilator was four times higher than for those treated without a ventilator and also that the death rate of pulmonary
fibrosis patients who were put on a hospital ventilator was seven times higher than those treated without a ventilator.
Strategic
Objectives
It
is our intention to develop the drug to the point whereby the Company would be in a position to license the drug to large pharmaceuticals
capable of conducting clinical trials and managing the distribution of the product. The Company does not plan to create a sales and marketing
staff to commercialize the pharmaceutical products it produces. The Company would be dependent on third parties such as licensees, collaborators,
joint venture partners or independent distributors to market and sell those products.
The
FDC Act and other federal and state statutes and regulations govern the testing, manufacture, safety, effectiveness, labeling, storage,
record keeping, approval, advertising and promotion of our products. As a result of these laws and regulations, product development and
product approval processes are very expensive and time-consuming. Our goal is to advance our leading drug candidate, BXT-25, and our
Subsidiary’s leading drug candidate, ProLectin, through regulatory submissions for Investigational New Drug (IND) status in the
United States, is subject to expensive and time-consuming approval processes.
Management
Our
management team and advisors include our CEO and Chairman, David Platt, Ph.D., who has played a leading role in the development of complex
co-polymer therapeutics for a variety of applications to address a variety of unmet medical needs. Our CFO Ola Soderquist, CPA, CMA is
a seasoned financial officer with more than 30 years of senior international entrepreneurial management experience within many industries
in both in public and private companies. Our Chief Communications Officer (“CCO”) Mike Sheikh is a US Air Force Academy graduate
and a long-time biotech consultant with expertise in public and private biotech companies with disruptive technologies.
Dr.
Platt, Mr. Sheikh and Mr. Soderquist are our only employees and each of them is committed on a full-time basis. Dr. Platt and Mr. Soderquist
currently have a monthly salary of thirty-five thousand Dollars ($35,000) and Mike Sheik a monthly salary of twenty-six thousand two
hundred fifty Dollars ($26,250), along with a twenty-five percent (25%) 401(k) Safe Harbor coverage up to the federal limit, currently
$66,000 per year plus potential catchup, currently $7,500, as well as reimbursement of a gold-level healthcare plan.
Our
Executive Officers and Directors may also receive stock or stock options at the discretion of our Board in the according to the approved
2021 Stock Plan, or any subsequent Stock Plan.
Business
Development
BXT-25
Bioxytran
intends to develop and, through third party contracts, manufacture oxygen therapeutics. Our oxygen therapeutics are a new class of pharmaceuticals
that are administered intravenously to transport oxygen to the body’s tissues. Currently there are four drug candidates to treat
a stroke. Abciximab, from Eli Lilly, is a platelet aggregation antagonist. Clinical trials show little advantage over placebos and could
lead to dangerous side effects, including more bleeding in patients. Cerovive, from AstraZeneca, is a Nitrone-based neuro protectant
currently in phase III clinical trials which shows no significant benefit over placebos with respect to changes in neurological impairment
as measured by the national institute of health stroke scale. Candesartan, from AstraZeneca, is an angiotensin receptor blocker which
was used to control blood pressure. Its efficacy in stroke patients still must be proven. Ancod, from Knoll Pharmaceuticals, is an anti-coagulant
that acts by breaking down the fibrinogen. It increases the risk of hemorrhage similar to those associated with tPA.
Using
our proprietary technology, we will develop and manufacture BXT-25 and similar drugs for applications including treatment of stroke conditions.
Bioxytran has an exclusive license for an FDA approved technology monitoring NADH (MDX Viewer), the control marker in the body’s
conversion of Oxygen to Energy, or the energy generating chain. The technology provides a clinical end-point for measuring oxygen supply
to the brain in real-time. MDX Viewer, developed by MDX LifeSciences, Inc., provides us with the potential to develop new molecules that
could potentially address unmet medical needs in disease indications resulting from hypoxia. MDX LifeSciences has licensed a patent (Tissue
Metabolic Score for Patient Monitoring - US20210153816A1) to Bioxytran for clinical monitoring of oxygen delivery through oxygen
carriers. MDX Lifesciences is an Affiliate of the Company.
On
April 19, 2023, the Company announced that its long awaited Acelluar Oxygen Carrier (“AOC”) BXT-25 had been successfully
tested in animals. The initial results are very encouraging because they show the non-toxicity of the experimental drug, along with the
corresponding full recovery in Swiss Albino mice, in an experiment carried out in a joint venture with NDPD Pharma, Inc. As a next step,
the Company intends to proceed with a 14-day repeated dose toxicity study using New Zealand Rabbits and Wistar Rats as funding permits.
ProLectin
The
Subsidiary is focusing on the development, manufacturing and commercialization of therapeutic drugs designed to address viral diseases
in humans. The Company has developed a novel method designed to reduce the viral load and modulate the immune system using a galectin
Antagonist.
Currently,
the Subsidiary’s lead drug candidate is a glyco-virology platform technology named ProLectin, a complex galectin antagonist that
binds to, and blocks the activity of galectin-3, a type of galectin. During viral infections galectins are upregulated and downregulated
based on the type of virus.
To
our knowledge, Pharmalectin, Inc. is the only company planning to develop a viable end-to-end solution for Covid-19. We are also the
only company attempting to use a Galectin Antagonist to combat the virus, SARS-CoV-2. The technology is built on the life-time work by
the founder of the Company, Dr. Platt, who discovered, and named, the Human Galectin-3 protein coded by a single gene, LGALS3, located
on chromosome 14, and published in his groundbreaking article, Structure-Function Relationship of a Recombinant Human Galactoside-Binding
Protein, Biochemistry 1993. Galectin Antagonists block the binding of galectins to carbohydrate structures, present in numerous diseases,
reducing their capability to replicate. Over the years, Dr. Platt has used this knowledge to create a significant number of sustainable
therapeutic solutions.
Using
our issued patents and proprietary technology, we intend to develop and manufacture ProLectin-RX and similar drugs for applications including
treatment of virological conditions. Our patent position consists of 2 parts: a patent a method for treating SARS-CoV-2 by administering
an effective amount of complex polysaccharides to a subject issued in 2022 by the International Bureau of the Patent Cooperation Treaty
(PCT) expiring in February 2041 (Polysaccharides for IV Administration that Treat Sars-Cov-2 Infections - WO2022/099061)
and assigned to us outright by Dr. Platt, as well as a provisional patent (Lectin-Binding Carbohydrates for Treating Viral Infections
- US 63/320544). Dr. Platt did not receive any compensation from the Company in consideration of his assignment of the patent.
Pharmalectin,
Inc. has an exclusive license issued by NDPD Pharma (Polysaccharides for Use in Treating Sars-Cov-2 Infections - WO2022/099052)
to Pharmalectin for use of treatment of SARS-CoV-2. NDPD Pharma is an Affiliate of the Company.
Further,
Pharmalectin has received an international trademark for ProLectin (WO0000001646681).
The
Company is capitalizing on 30 years of research in Galectins and recent peer reviewed articles on Galectins and Covid-19. Dr. Platt also
has an impressive body of patents in this field which gives him an advantage with respect to filing new patents based on his prior art.
We will rely on a combination of patent applications, patent, trade secrets, proprietary know-how and trademarks to protect our proprietary
rights. We believe that to have a competitive advantage, we must develop and maintain the proprietary aspects of our technologies.
The
results of the ProLectin-M trials are from our Proof-of-Concept trial approved by the IRB at Mazumdar Shaw Medical Center, Narayana Health
in Bangalore, India and the IRB phase 2 study at ESIS Medical College and Hospital, Sanath Nagar, Hyderabad, India. The results of the
trial are described in our four peer-reviewed articles Galectin antagonist use in mild cases of SARS-CoV-2; pilot feasibility randomised,
open label, controlled trial, published in Journal of Vaccines & Vaccination on December 30, 2020, Carbohydrate ProLectin-M,
a Galectin-3 Antagonist, Blocks SARS-CoV-2 Activity published in the International Journal of Health Sciences on June 30, 2022, PLG-007
and Its Active Component Galactomannan-α Competitively Inhibit Enzymes That Hydrolyze Glucose Polymers published in the International
Journal of Molecular Science on July 13, 2022 and An Oral Galectin Antagonist in COVID-19—A Phase II Randomized Controlled Trial
published in the journal Virus on February 23, 2023.
On
December 2, 2022, India’s Central Drugs Standard Control Organisation (CDSCO) issued an IND with permission to conduct: “A
Phase 1b/2a Randomized, Blinded, placebo-controlled Study in Participants with Mild to Moderate COVID-19 to Evaluate the Safety, Efficacy,
and Pharmacokinetics of Orally Administered ProLectin-M”. The trial is planned to start on, or around, October 1, 2023.
On
August 21, 2023 the Company’s IND #153742 under the title “PROTECT: ProLectin-M, a nucleocapsid TErminal GaleCTin antagonist
for COVID-19 (PROTECT), a Randomized, Double-blinded Clinical Trial to Evaluate the Efficacy and Safety in Non-Hospitalized Adult Participants
with COVID-19” was approved by the FDA, the trial is expected to start in the first quarter of 2024, provided we obtain adequate
funding.
On
January 27, 2023, an additional IND with the CDSCO was issued for ProLectin-I for an “IV treatment of SARS-CoV-2 in hospitalized
patients with moderate Covid-19 infections and for Long Covid”, and for ProLectin-F for “treatment of lung-fibrosis as a
result of use of ventilator”.
FDA
Approval Process
In
the United States, pharmaceutical products, including biologics like BXT-25, are subject to extensive regulation by the FDA. The FDC
Act and other federal and state statutes and regulations, govern, among other things, the research, development, testing, manufacture,
storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling,
and import and export of pharmaceutical products. Failure to comply with applicable U.S. requirements may subject a company to a variety
of administrative or judicial sanctions, such as FDA refusal to approve pending new drug applications, or NDAs, warning letters, product
recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties, and criminal
prosecution.
Pharmaceutical
product development in the United States typically involves preclinical laboratory and animal tests, the submission to the FDA/EMA of
an IND application, which must become effective before clinical testing may commence, and adequate and well-controlled clinical trials
to establish the safety and effectiveness of the drug or biologic for each indication for which FDA/EMA approval is sought. Satisfaction
of FDA/EMA pre-market approval requirements typically take many years (typically between 5-7 years post an IND submission) and the actual
time required may vary substantially based upon the type, complexity and novelty of the product or disease.
Preclinical
tests include laboratory evaluation as well as animal trials to assess the characteristics and potential pharmacology and toxicity of
the product. The conduct of the preclinical tests must comply with federal regulations and requirements including good laboratory practices.
The results of preclinical testing are submitted to the FDA as part of an IND along with other information, including information about
product chemistry, manufacturing and controls, and a proposed clinical trial protocol. Long term preclinical tests, such as animal tests
of reproductive toxicity and carcinogenicity, may continue after the IND is submitted.
A
30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA
has not objected to the IND within this 30-day period, the clinical trial proposed in the IND may begin.
Clinical
trials involve the administration of the investigational drug to healthy volunteers or patients under the supervision of a qualified
investigator. Clinical trials must be conducted in compliance with federal regulations and good clinical practices, or GCP, as well as
under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety and the effectiveness criteria
to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as
part of the IND.
The
FDA may order the temporary or permanent discontinuation of a clinical trial at any time or impose other sanctions if it believes that
the clinical trial is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial
patients. The clinical trial protocol and informed consent information for patients in clinical trials must also be submitted to an institutional
review board, or IRB, for approval. An IRB may also require the clinical trial at the site to be halted, either temporarily or permanently,
for failure to comply with the IRB’s requirements, or may impose other conditions.
Clinical
trials to support New Drug Applications (NDAs) are typically conducted in three sequential Phases, but the Phases may overlap. In Phase
1, the initial introduction of the investigational drug candidate into healthy human subjects or patients, the investigational drug is
tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses and, if possible,
early evidence on effectiveness. Phase 2 usually involves trials in a limited patient population, to determine the effectiveness of the
investigational drug for a particular indication or indications, dosage tolerance and optimum dosage, and identify common adverse effects
and safety risks. In the case of product candidates for severe or life-threatening diseases such as pneumonia, the initial human testing
is often conducted in patients rather than in healthy volunteers.
If
an investigational drug demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 clinical
trials are undertaken to obtain additional information about clinical efficacy and safety in a larger number of patients, typically at
geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the investigational
drug and to provide adequate information for its labeling.
After
completion of the required clinical testing, an NDA, is prepared and submitted to the FDA. FDA approval of the marketing application
is required before marketing of the product may begin in the United States. The marketing application must include the results of all
preclinical, clinical and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture,
and controls.
The
FDA has 60 days from its receipt of an NDA to determine whether the application will be accepted for filing based on the agency’s
threshold determination that it is sufficiently complete to permit substantive review. Once the submission is accepted for filing, the
FDA begins an in-depth review. The FDA has agreed to certain performance goals in the review of marketing applications. Most such applications
for non-priority drug products are reviewed within ten months. The review process may be extended by the FDA for three additional months
to consider new information submitted during the review or clarification regarding information already provided in the submission. The
FDA may also refer applications for novel drug products or drug products that present difficult questions of safety or efficacy to an
advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether
the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such
recommendations. Before approving a marketing application, the FDA will typically inspect one or more clinical sites to assure compliance
with GCP.
Additionally,
the FDA will inspect the facility or the facilities at which the drug product is manufactured. The FDA will not approve the NDA unless
compliance with cGMP is satisfactory, and the marketing application contains data that provide substantial evidence that the product
is safe and effective in the indication studied. Manufacturers of biologics also must comply with FDA’s general biological product
standards.
After
the FDA evaluates the NDA and the manufacturing facilities, it issues an approval letter or a complete response letter. A complete response
letter outlines the deficiencies in the submission and may require substantial additional testing or information in order for the FDA
to reconsider the application. If and when those deficiencies have been addressed in a resubmission of the marketing application, the
FDA will re-initiate review. If the FDA is satisfied that the deficiencies have been addressed, the agency will issue an approval letter.
The FDA has committed to reviewing such resubmissions in two or six months depending on the type of information included. It is not unusual
for the FDA to issue a complete response letter because it believes that the drug product is not safe enough or effective enough or because
it does not believe that the data submitted are reliable or conclusive.
An
approval letter authorizes commercial marketing of the drug product with specific prescribing information for specific indications. As
a condition of approval of the marketing application, the FDA may require substantial post-approval testing and surveillance to monitor
the drug product’s safety or efficacy and may impose other conditions, including labeling restrictions, which can materially affect
the product’s potential market and profitability. Once granted, product approvals may be withdrawn if compliance with regulatory
standards is not maintained, or problems are identified following initial marketing.
Once
an NDA is approved, a product will be subject to certain post-approval requirements. For instance, the FDA closely regulates the post-approval
marketing and promotion of therapeutic products, including standards and regulations for direct-to-consumer advertising, off-label promotion,
industry-sponsored scientific and educational activities and promotional activities involving the internet.
BXT-25
The
only FDA approved treatment for ischemic strokes is tissue plasminogen activator tPA, also known as IV rtPA, given through an IV in the
arm. tPA works by dissolving the clot and improving blood flow to the part of the brain being deprived of blood flow. If administered
within 3 hours and up to 4.5 hours in certain eligible patients, tPA may improve the chances of recovering from a stroke. Another treatment
option is an endovascular procedure called mechanical thrombectomy in which a blood clot is removed by threading a wired-caged device
called a stent retriever through an artery in the groin up to the blocked artery in the brain. The stent opens and grabs the clot, enabling
the removal of the stent with the trapped clot.
Hypoxia
is a condition in which cells lack sufficient oxygen supply to support metabolic function. The BXT-25 co-polymer hemoglobin molecule
will be designed to contain an oxygen rechargeable iron which picks up oxygen in the lungs, is expected to be 5,000 times smaller than
an RBC, and we believe can reach hypoxic tissue more effectively than RBCs. Products similar to BXT-25 are stable at room temperature
and have no blood type matching requirement. We plan to introduce BXT-25 in clinical trials for hypoxic medical conditions such as stroke.
For
the production of BXT-25, we intend to utilize third party manufacturing facilities that we believe are fully compliant with Good Manufacturing
Practices (GMP) only, as required by the regulatory authorities in Europe or the United States, in order to produce a sufficient quantity
of BXT-25 for animal toxicity and pre-clinical trials for animals. We have not conducted any clinical trials on animals or humans to
confirm the efficacy of, or filed any applications with the FDA with respect to, BXT-25. The Company has developed a proof-of-concept
production line and successfully manufactured the initial batch for use in pre-clinical trials in the first quarter of 2023, provided
we obtain adequate funding.
This
product is being developed and as an early intervention in an out-of-hospital setting for the treatment of patients with ischemia of
the brain resulting from a stroke or the blockage of the blood vessels to the brain. We plan to initially conduct pre-clinical trials
and to seek approval of BXT-25 for the treatment of adults at early stages of stroke.
ProLectin
There
is an unmet medical need in Covid-19 to find a therapeutic that reduces the mortality of the disease. There are no FDA approved treatments
for Covid-19, only repurposed therapeutics. If given early enough in the disease, we believe that ProLectin will block viral entry and
act as an antiviral by eliminating the virus from the blood stream after a couple of treatments. At a later stage in the disease pathology,
we believe ProLectin could potentially restore adaptive immune function and assist in reducing or possibly eliminating the virus from
the body. Our upcoming clinical trials aim to demonstrate if the safety and efficacy of our drug candidates in humans can be relied upon.
The
cytokine storm is a severe immune reaction in which the body overproduces too many pro-inflammatory cytokines into the blood leading
to a surge of more immune cells to the site of infection. This translates into an inflammatory cycle that is not easily brought back
to homeostasis. Cytokines play an important role in normal immune responses, but having a large amount of them released in the body all
at once can be harmful. A cytokine storm can occur as a result of an infection, autoimmune condition, or other disease. It may also occur
after treatment with some type of immunotherapies. Signs and symptoms include high fever, inflammation (redness and swelling), and severe
fatigue and nausea. Sometimes, a cytokine storm may be severe or life threatening and lead to acute respiratory distress syndrome (ARDS),
and multiple organ failure.
For
the production of ProLectin, we intend to utilize third party manufacturing facilities that are fully compliant with Good Manufacturing
Practices (GMP) only, as required by the regulatory authorities in Europe or the United States, in order to produce a sufficient quantity
of ProLectin for our upcoming human trials with the CDSCO in India. Prior to this we have conducted clinical trials on animals and humans
to confirm the non-toxicity and efficacy. We also expect to file an IND application with the FDA in the third quarter of 2023, provided
we obtain adequate funding.
The
oral product is being developed as a treatment for mild to moderate Covid-19 patients, while the intravenous drugs are developed for
in moderate (Hospitalized patients) Covid-19 infections (ProLectin-I), Long Covid, and of treatment of lung-fibrosis (ProLectin-F).
European
Directorate for the Quality of Medicines Certification (EDQM)
Certification
from the European Directorate for the Quality of Medicines (EDQM) is required for all new and approved human and veterinary medicinal
products that are manufactured from materials taken from cattle and marketed in the European Union. As part of the certification process,
we will be required to provide technical information on the manufacturing process, the origin of the raw material and type of tissue
used, the cattle traceability, beginning at their country of birth, and auditing, and a risk analysis from an independent expert.
We
intend to establish and implement clinical development programs that add value to our business in the shortest period of time possible
and to seek strategic partners when a program becomes advanced and requires additional resources. We intend to continue focusing our
expertise and resources to develop novel formulations, and to leverage development partnerships to apply our complex co-polymer chemistry
designs in other medical indications. We may seek to enter into licensing, co-marketing, or co-development agreements across different
geographic regions, in order to avail ourselves of the marketing expertise of one or more seasoned marketing and/or pharmaceutical companies.
We plan to further develop new and proprietary drug candidates by using novel development pathways specific to each drug candidate.
A
core part of our strategy relies upon creating safe and efficacious drug formulations that can be administered as standalone therapies
or in combination with existing medications. We believe we utilize a novel approach that is expected to create drug formulations that
can be combined with existing therapies and potentially deliver valuable products in areas of high unmet medical needs. We will assemble
a scientific advisory board consisting of scientists with both academic and corporate research and development experience that will provide
leadership and counsel in the scientific, technological and regulatory aspects of our current and future projects. In addition, we will
assemble a medical advisory board consisting of leading physicians and key opinion leaders who have participated in relevant clinical
studies and who will guide us through ongoing clinical trial programs. Our scientific and medical advisory boards consist of some of
leading scientists, medical doctors and professionals in the co-polymer and ischemic brain injury field.
We
believe that our drug development leadership team provides us with a significant competitive advantage in designing highly efficient
clinical programs to deliver valuable products in areas of high unmet medical needs.
Project
Costs
ProLectin
Pharmalectin
is a single purpose entity aiming to develop pharmaceutical cures for Covid-19 (collectively referred to as “ProLectin”)
and bring the drugs through FDA acceptance and thereafter license out the product(s). The total cost of the project is estimated to cost
$40 million of which $3.6 million has been invested so far.
As
of December 31, 2022, Good Manufacturing Practice (GMP), pre-clinical and two clinical Phase I/II study have been completed for the initial
drug, ProLectin-M, which is an oral formulation against mild to moderate symptoms of the disease and GMP has been completed for ProLectin-I,
and -F.
On
December 2, 2022, India’s Central Drugs Standard Control Organisation (CDSCO) issued an IND with permission to conduct: “A
Phase 1b/2a Randomized, Blinded, placebo-controlled Study in Participants with Mild to Moderate COVID-19 to Evaluate the Safety, Efficacy,
and Pharmacokinetics of Orally Administered ProLectin-M”. The trial is planned to start on, or around, October 1, 2023.
On
August 21, 2023 the Company’s IND #153742 under the title “PROTECT: ProLectin-M, a nucleocapsid TErminal GaleCTin antagonist
for COVID-19 (PROTECT), a Randomized, Double-blinded Clinical Trial to Evaluate the Efficacy and Safety in Non-Hospitalized Adult Participants
with COVID-19” was approved by the FDA, the trial is expected to start in the first quarter of 2024, provided we obtain adequate
funding.
On
January 27, 2023, an additional IND with the CDSCO was issued for ProLectin-I for an “IV treatment of SARS-CoV-2 in hospitalized
patients with moderate Covid-19 infections and for Long Covid”, and for ProLectin-F for “treatment of lung-fibrosis as a
result of use of ventilator”.
In
addition to the approximately $3.6 million currently invested in the project, we believe we will be required to spend an additional $0.2
million for submission of Investigational New Drug application (IND), approximately $2.5 million in Phase I/II (dosage and pharmacokinetics)
and Phase III clinical trials and approximately $1 million for General and Administrative and general working capital purposes. Further,
we will be required to spend an additional $2.6 million in order to submit an IND with the FDA for ProLectin-M, -I and, as well as a
proof of concept for ProLectin-F.
An
additional spending in the range of $8 to 10 million will be required in order to complete the Phase IIb/III testing with the FDA and
EMA of ProLectin-I and -F.
ProLectin-A
In
order to develop ProLectin-A, the Company will need an additional $10 million, approximately $3.15 million of proceeds will be used for
preparation for scale up and manufacturing (Good Laboratory Practice (GLP) Good Manufacturing Practices (GMP)), approximately $1.5 million
will be used for toxicity testing in animals for Investigational New Drug application (IND), approximately $3.5 million for Phase I (safety)
and Phase II (proof of concept) clinical trials. We expect that obtaining a CE from the European Directorate for the Quality of Medicines
will require an additional $0.5 million in funds. G&A is expected to be $1.35 million.
BXT-25
In
order to start the development BXT-25, the Company will need an additional $10 million, approximately $3.15 million of proceeds will
be used for preparation for scale up and manufacturing (Good Laboratory Practice (GLP) Good Manufacturing Practices (GMP)), approximately
$1.5 million will be used for toxicity testing in animals for Investigational New Drug application (IND), approximately $3.5 million
for Phase I (safety) and Phase II (proof of concept) clinical trials. We expect that obtaining a CE from the European Directorate for
the Quality of Medicines will require an additional $0.5 million in funds. G&A is expected to be $1.35 million.
In
aggregate, we believe we will require an additional $30-35 million in order to complete the II/a trials with the FDA for ProLectin-A
and BXT-25 and the Phase II/b/III trials for ProLectin-I and -F. There are no guarantees the Company will be able to obtain additional
capital funder, whether through debt and/or equity financing, or will be able to raise funds on terms acceptable to the Company.
Market
Opportunity
Stroke
Our
injectable drug candidate, BXT-25, will potentially compete with existing therapies for the treatment for stroke, hypoxia and anti-necrosis
that according to Global Industry Analysts, Inc. has a global market opportunity of $50 billion. The standard therapy for acute anemia
resulting from blood loss is infusion of red blood cells mainly from supplies of donated blood. For prophylactic or long-term treatment
of anticipated or chronic anemia, medications that stimulate the creation of new red blood cells are frequently used.
Presently,
the standard therapy for reversing hypoxia is blood infusion, RBCs or hyperbaric oxygen. Hyperbaric medicine or hyperbaric oxygen therapy
(HBOT) is a medical term for using oxygen at a level higher than atmospheric pressure. The HBOT treatment can only be done at a medical
facility and each session can cost from $1,000 to more than $3,000. For decades, oxygen carriers have been developed for perfusion and
oxygenation of ischemic tissue; none have yet succeeded in becoming a proven oxygen therapeutics for stroke and wound healing. These
products were either blood-derived elements, synthetic perfluorocarbons, or red blood cell modifiers. According to the Fact Sheet No.
279 published June 7, 2014, by the World Health Organization, there is a global shortage of transfusion suitable blood of 110 million
units, and the need for blood is rising 6- 7% annually. We will design BXT-25 and any new drug candidates to enhance HBOT treatment and
reduce the demand on blood transfusions, subject to testing as required by the FDA.
Covid-19
There
is an unmet medical need in Covid-19 to find a therapeutic that reduces the mortality of the disease. There are no FDA approved treatments
for Covid-19, only repurposed therapeutics. If given early enough in the disease, we believe that ProLectin will block viral entry and
act as an antiviral by eliminating the virus from the blood stream after a couple of treatments. At a later stage in the disease pathology,
we believe ProLectin could potentially restore adaptive immune function and assist in reducing or possibly eliminating the virus from
the body. Our upcoming clinical trials aim to demonstrate if the safety and efficacy of our drug candidates in humans can be relied upon.
Key
Strengths
We
believe that our key differentiating elements include:
| |
● |
Focus
on novel therapeutic opportunities provided by co-polymer: We are focused on development of co-polymer compounds to stabilize
the modified hemoglobin molecule. The Co-polymer method of chemical stabilization has not received as much scientific attention as
nucleic acids and proteins, but the Company believes that it is a viable alternative to these other materials. |
| |
|
|
| |
●
|
Experienced
management |
| |
|
|
| |
●
|
Our
President, Chief Executive Officer and Chairman, David Platt, Ph.D., is a chemical engineer, a pioneer in designing drugs made from
co-polymers, and has more than 30 years of experience in the development of therapeutic drugs. We are the fourth biotechnology company
founded by Dr. Platt. The prior company is Boston Therapeutics Inc. (OTC: BTHE). The first two are International Gene Group, which
later became Prospect Therapeutics, and is now known as La Jolla Pharmaceuticals (Nasdaq: LJPC), and Pro-Pharmaceuticals (now Galectin
Therapeutics) (Nasdaq: GALT). Their core technologies were either developed or co-developed by Dr. Platt. |
| |
●
|
Our
CFO Ola Soderquist has more than 30 years of senior international entrepreneurial management experience within technology companies.
Ola’s managerial experience portfolio includes; Start-ups, Private, Public, Venture Capital and Private Equity ownership. He
has served in CFO and other managerial capacities in multiple industry sectors and companies. Mr. Soderquist is a multi-lingual senior
finance professional poised to work globally and cross-functionally, particularly with complex projects involving change management,
business integration, systems implementation, continuous improvement, and process excellence. He obtained a BS and an MS in Accounting
from Stockholm School of Economics and an MBA from Babson College. |
| |
|
|
| |
● |
Our
CCO Mike Sheikh, is a US Air Force Academy graduate and pilot. He has a Bachelor of Science in Economics, and flew KC-135 tankers
and worked as a budget Officer in the comptroller’s squadron. He worked for Dean Witter and National Securities as a broker
and eventually research analyst. After the brokerage industry, he was a business development officer for a variety of specialty finance
companies that did factoring and purchase order financing. He is a long-time biotech consultant expert for public and private biotech
companies with disruptive technologies. Mr. Sheikh the founder of Falcon Strategic Research, which focuses on small-cap and micro-cap
companies that are not covered by traditional analysts on Wall Street. He is also the founder of an investor relations firm. |
| |
|
|
| |
●
|
We
have assembled a Scientific and a Medical Advisory Board consisting of leading physicians and key opinion leaders who have participated
in relevant clinical studies and who will guide us through ongoing clinical trial programs. Our scientific and medical advisory boards
consist of some of the leading scientists, medical doctors and professionals in the ischemia or hypoxia fields. |
| |
|
|
| |
● |
Products
are differentiated and address significant unmet needs: Our lead product candidates, BXT-25, ProLectin, and any additional products
will be designed to address significant unmet medical needs. Oxygen therapy management, including stroke, other hypoxia management
and treatment of diseases and medical conditions associate with hypoxia, remain a critical area of unmet need. Increasingly, patients,
physicians and the media are highlighting the deficiencies of current oxygen therapy related therapies and the growing population
of individuals adversely affected by ischemia, unhealed wounds, or traumatic brain injury. |
| |
|
|
| |
● |
Efficient
development strategy: We believe that our regulatory development pathway is a standard generic pathway approval for a drug. |
Corporate
Information
We
are a clinical stage pharmaceutical company founded on June 9, 2008, as America’s Driving Ranges, Inc. On September 21, 2018, the
Company was reorganized into Bioxytran through a reverse merger to focus on the development, manufacturing and commercialization of therapeutic
drugs designed to address hypoxia in humans.
Our
principal executive offices are located at 75 2nd Ave., Suite 605, Needham, MA 02494.
Emerging
Growth Company Status
The
Company meets the emerging growth company requirements. The Company will report its results in this S-1 in accordance with the emerging
growth company requirements and in its reports filed with the SEC.
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF
FINANCIAL
CONDITION AND RESULTS OF OPERATIONS
The
following discussion and analysis is based on, and should be read in conjunction with the audited financial statements and the notes
thereto for the years ended December 31, 2022, and 2021, included elsewhere in this Prospectus. This discussion contains forward-looking
statements. These statements are often identified by the use of words such as “may,” “will,” “expect,”
“believe,” “anticipate,” “intend,” “could,” “estimate,” or “continue,”
and similar expressions or variations. Such forward looking statements are subject to risks, uncertainties and other factors that could
cause actual results and the timing of certain events to differ materially from future results expressed or implied by such forward-looking
statements. The forward-looking statements in this Prospectus represent our views as of the date of this Prospectus. We anticipate that
subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements
at some point in the future, we have no current intention of doing so except to the extent required by applicable law. You should, therefore,
not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this Prospectus.
Overview
We
do not currently have sufficient capital resources to fund operations. To stay in business and to continue the development of our products,
we will need to raise additional capital through public or private sales of our securities, debt financing or short-term bank loans,
or a combination of the foregoing. We believe that if we can raise three million seven hundred thousand Dollars ($3,700,000), we will
have sufficient working capital to develop our business over the next approximately fifteen (15) months. At funding raised that is significantly
less than $3,700,000, we can likely continue to develop our business over the same 15-month period, but funding at that level will delay
the development of our technology and business.
Bioxytran,
Inc. is headquartered in Needham, Massachusetts. The Company’s initial product pipeline is focused on developing and commercializing
therapeutic molecules for stroke. BXT-25 will be designed to be an injectable anti-necrosis drug specifically designed to treat a person
immediately after that person suffers an ischemic stroke. The drug is designed to be injected intravenously to travel to the lungs to
pick up oxygen molecules to carry to the brain. Like a red blood cell, the drug will cross the blood brain barrier, which is a protective
semi-permeable membrane allowing some material to cross but preventing others from crossing. BXT-25 will be designed to diffuse oxygen
into the brain tissues. We expect the BXT-25 molecule to be 5,000 times smaller than a red blood cell.
On
December 2, 2022, India’s Central Drugs Standard Control Organisation (CDSCO) issued an IND with permission to conduct: “A
Phase 1b/2a Randomized, Blinded, placebo-controlled Study in Participants with Mild to Moderate COVID-19 to Evaluate the Safety, Efficacy,
and Pharmacokinetics of Orally Administered ProLectin-M”. The trial is planned to start on, or around, October 1, 2023.
On
August 21, 2023 the Company’s IND #153742 under the title “PROTECT: ProLectin-M, a nucleocapsid TErminal GaleCTin antagonist
for COVID-19 (PROTECT), a Randomized, Double-blinded Clinical Trial to Evaluate the Efficacy and Safety in Non-Hospitalized Adult Participants
with COVID-19” was approved by the FDA, the trial is expected to start in the first quarter of 2024, provided we obtain adequate
funding.
On
January 27, 2023, an additional IND with the CDSCO was issued for ProLectin-I for an “IV treatment of SARS-CoV-2 in hospitalized
patients with moderate Covid-19 infections and for Long Covid”, and for ProLectin-F for “treatment of lung-fibrosis as a
result of use of ventilator”.
On
April 19, 2023, the Company announced that its long awaited Acelluar Oxygen Carrier (“AOC”) BXT-25 has been successfully
tested in animals. The initial results are very encouraging because they show the non-toxicity of the experimental drug, along with the
corresponding full recovery in Swiss Albino mice, in an experiment carried out in a joint venture with NDPD Pharma, Inc. As a next step,
the Company intends to proceed with a 14-day repeated dose toxicity study using New Zealand Rabbits and Wistar Rats as funding permits.
The
accompanying consolidated financial statements have been prepared assuming the Company will continue as a going concern. The Company
has limited resources and operating history. The Company currently has convertible loans outstanding at a total face value of one million
nine hundred thousand Dollars ($1,900,000). As shown in the accompanying consolidated financial statements, the Company had an accumulated
deficit of thirteen million four hundred thirteen thousand nine hundred one dollars ($13,413,901) as at June 30, 2023. The accumulated
deficit as at December 31, 2022 was eleven million two hundred seventeen thousand six hundred Dollars ($11,217,600).
The
future of the Company is dependent upon its ability to obtain financing to develop its new business opportunities and support the cost
of the drug development including clinical trials and regulatory submission to the FDA.
Management
plans to seek additional capital through private placements and public offerings of its Common Stock. There can be no assurance that
the Company will be successful in accomplishing its objectives. Without such additional capital or the establishment of strategic relationships
with established pharmaceutical companies, the Company may be required to cease operations. These conditions raise substantial doubt
about the Company’s ability to continue as a going concern. The financial statements do not include any adjustments relating to
the recoverability and classification of recorded assets, or the amounts of and classification of liabilities that might be necessary
in the event the Company cannot continue operations.
RESULTS
OF OPERATIONS FOR THE YEARS ENDED DECEMBER 31, 2022, AND 2021
We
are a clinical stage company. Historically, Bioxytran was engaged in formation, fund raising and identifying and consulting with the
scientific community regarding the development, formulation and testing of its products. We are actively engaged in research and development
activities through our Subsidiary, Pharmalectin, Inc., developing ProLectin.
Research
and Development
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Research and development: | |
| | | |
| | |
| Process development | |
$ | — | | |
$ | 339,000 | |
| Product development | |
| 123,580 | | |
| 305,743 | |
| Regulatory | |
| 231,078 | | |
| 177,074 | |
| Clinical trials | |
| 583,750 | | |
| 1,016,765 | |
| Project management | |
| 39,360 | | |
| 175,180 | |
| Total research and development | |
$ | 977,768 | | |
$ | 2,013,762 | |
| |
During
the twelve months ended December 31, 2022, the Company recorded $977,768 in R&D expenses. During the twelve months ended December
31, 2021, the Company recorded $2,013,762. The significant difference is due to a lack of funding as we’re waiting to start
our clinical trials. |
General
and Administrative
| |
|
December
31, 2022 |
|
|
December
31, 2021 |
|
| General
and administrative expenses: |
|
|
|
|
|
|
|
|
| Payroll
and related expenses |
|
$ |
343,167 |
|
|
$ |
1,391,431 |
|
| Costs
for legal, accounting and other professional services |
|
|
78,925 |
|
|
|
84,056 |
|
| Costs
for legal, accounting and other professional services related party |
|
|
44,220 |
|
|
|
5,125 |
|
| Promotional
expenses |
|
|
339,251 |
|
|
|
5,500 |
|
| Miscellaneous
expenses |
|
|
172,399 |
|
|
|
131,698 |
|
| Total
general and administrative |
|
$ |
977,962 |
|
|
$ |
1,617,810 |
|
| |
The
significant increase in Payroll and related expenses for the twelve months ended December 31, 2022, were due to the retro-active
roll-out of market-based salaries for the Company’s management starting January 1, 2022. On August 1, 2022, the Company’s
Officers forfeited of the majority of their accrued salaries and benefits for a total value of $1,273,000. |
| |
|
| |
The
Costs for legal, accounting and other professional services for the twelve months ended December 31, 2022, decreased due to
reduced legal fees. |
| |
|
| |
The
Costs for legal, accounting and other professional services related party for two License Agreements with two affiliated companies.
Bioxytran reimbursed the affiliates for incurred administrative costs in making the licenses, and their maintenance. |
| |
|
| |
Promotional
expenses for the twelve months ended December 31, 2022, were $339,251, as compared to $5,500 for the twelve months ended December
31, 2021. The increase costs stock promotion incurred by the Company’s return to being listed on OTCQB. |
| |
|
| |
Miscellaneous
G&A expenses during the twelve months ended December 31, 2022, was $172,399 and $131,698, respectively. The increase is based
on the Company’s costs to return to being listed on OTCQB. |
Stock-based
Compensation
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Compensation expense to BoD and Management | |
$ | 61,578 | | |
$ | 493,578 | |
| Compensation expense to consultants | |
| 116,804 | | |
| 89,284 | |
| Total compensation expense | |
$ | 178,382 | | |
$ | 582,862 | |
| |
Stock-based
compensation mounted to $178,382 for the twelve months ended December 31, 2022. The stock-based compensation for the twelve months
ended December 31, 2021, was $582,862 and is explained by the liquidation of the 2010 Stock Plan in 2021. |
Other
expenses
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Other expenses: | |
| | | |
| | |
| Interest expense | |
$ | 207,117 | | |
$ | 236,577 | |
| Debt discount amortization | |
| 128,859 | | |
| 77,031 | |
| Amortization of warrants | |
| 190,335 | | |
| — | |
| Forfeiture of warrants | |
| (6,763 | ) | |
| — | |
| Amortization of IP | |
| 3,644 | | |
| — | |
| Total other income (expenses) | |
$ | 523,192 | | |
$ | 313,608 | |
| |
During
the twelve months ended December 31, 2022, the Company recorded $128,859 in amortization of debt discount and the interest
expense was $207,117, $3,644 was amortized from the Company’s IP at net of $183,572 in amortization of warrants. During
the twelve months ended December 31, 2021, the Company recorded $77,031 in amortization of debt discount while the interest expense
was $171,627. The increase is due to the Company’s fund-raising activities. |
Non-Controlling
Interest
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Net loss attributable to the non-controlling interest | |
$ | 193,732 | | |
$ | 496,297 | |
| |
For
the twelve months ended December 31, 2022, and 2021 there was a non-controlling interest attribution of $193,732 and $496,297 respectively.
The significant difference is due to a significant reduction in the R&D activities in the current year due to lack of capital. |
| | |
# of shares | | |
# of options | | |
December 31, 2022 | | |
December 31, 2021 | |
| Minority owners cash investment | |
| 4,650,000 | | |
| | | |
$ | 160,485 | | |
$ | 160,485 | |
| Bioxytran non-dilutive equity | |
| 15,000,000 | | |
| | | |
| 1,500 | | |
| 1,500 | |
| Issued stock options @ $0.33 | |
| | | |
| 4,500,000 | | |
| 450 | | |
| 450 | |
| Total outstanding | |
| 19,650,000 | | |
| 4,500,000 | | |
$ | 162,435 | | |
$ | 162,435 | |
| |
There
are currently 30,000,000 issued and 19,650,000 outstanding shares; 15,000,000 Common shares (76%) are held by Bioxytran and 4,650,000
Common shares (24%) are held by an affiliate. An additional 4,500,000 options are also held by an affiliate. The option agreement
includes provisions for dilutive issuance and cash-less exercise. The beneficial ownership of the affiliate includes Mike Sheikh,
Ola Soderquist and David Platt. |
Net
Loss
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Net loss attributable to Bioxytran | |
$ | (2,463,932 | ) | |
$ | (4,031,745 | ) |
| | |
| | | |
| | |
| Loss per common share, basic and diluted | |
$ | (0.02 | ) | |
$ | (0.04 | ) |
| | |
| | | |
| | |
| Weighted average number of common shares outstanding, basic and diluted | |
| 115,361,105 | | |
| 106,252,116 | |
| |
The
Company generated a net loss for the twelve months ended December 31, 2022, of $2,463,932. In comparison, for the twelve months ended
December 31, 2021, the Company generated a net loss of $4,031,745. The significant difference is due to a significant reduction in
the R&D activities in the current year due to lack of capital. |
CASH-FLOWS
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Net cash used in operating activities | |
$ | (1,805,670 | ) | |
$ | (1,697,399 | ) |
| | |
| | | |
| | |
| Net cash used in investing activities | |
| (32,247 | ) | |
| (36,931 | ) |
| | |
| | | |
| | |
| Net cash provided by financing activities | |
| 2,060,960 | | |
| 1,765,000 | |
| | |
| | | |
| | |
| Net increase in cash | |
| 223,043 | | |
| 30,670 | |
| Cash, beginning of period | |
| 72,358 | | |
| 41,688 | |
| Cash, end of period | |
$ | 295,401 | | |
$ | 72,358 | |
| |
Net
cash used in operating activities was $1,805,670 and $1,697,399 for the twelve months ended December 31, 2022, and 2021, respectively.
The decrease was due to a reduction of the research and development activities due to lack of funding. |
| |
|
| |
In
the twelve months ended December 31, 2022, the Company is in the process of filing a patent, and $32,247 was spent in legal fees.
In the twelve months ended December 31, 2021 the amount was $36,931. |
| |
|
| |
Cash
flows from financing activities were $2,060,960 and $1,765,000 for the twelve months ended December 31, 2022, and 2021, respectively.
|
| |
|
| |
The
available cash was $295,401 and $72,358 in the end of the twelve months ended December 31, 2022, and 2021, respectively. |
LIQUIDITY
AND CAPITAL RESOURCES
Current
Assets
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Current assets: | |
| | | |
| | |
| Cash | |
$ | 295,401 | | |
$ | 72,358 | |
| Total current assets | |
$ | 295,401 | | |
$ | 72,358 | |
| |
As
of December 31, 2022, our current assets consisted of $295,401 in cash at December 31, 2021 we had $72,358 in cash. |
Current
Liabilities
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable and accrued expenses | |
$ | 749,395 | | |
$ | 624,316 | |
| Accounts payable related party | |
| 709,727 | | |
| 531,000 | |
| Un-issued shares liability | |
| 960 | | |
| — | |
| Un-issued shares liability related party | |
| 38,400 | | |
| — | |
| Convertible notes payable, net of discount | |
| 2,165,000 | | |
| 2,122,181 | |
| Total current liabilities | |
| 3,663,482 | | |
| 3,277,497 | |
| |
At
December 31, 2022 we had total liabilities of $3,663,482, which consisted of $1,459,121 in accounts payable and accrued expenses
(of which $709,727 was payable to related parties), $39,360 in un-issued shares (of which $38,400 was payable to related parties),
and $2,165,000 in four convertible loans. At December 31, 2021 total liabilities were $3,277,497, consisting of $1,155,316 in accounts
payable and accrued expenses (of which $531,000 was payable to related parties), and $2,122,181 in the form of four convertible loans
net of discount. On August 1, 2022, Management forfeited all accrued salaries prior to May 2022. |
Net
Working Capital and Accumulated Deficit
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Net working capital | |
$ | (3,368,080 | ) | |
$ | (3,205,139 | ) |
| | |
| | | |
| | |
| Accumulated deficit | |
$ | (11,217,600 | ) | |
$ | (8,753,668 | ) |
| |
At
December 31, 2022, the net working capital was negative $3,368,080 and the accumulated deficit of $11,217,600. Comparatively, on
December 31, 2021, we had net working capital of negative $3,205,139 and an accumulated deficit of $8,753,668. We believe that we
must raise not less than $3,700,000 to be able to continue our business operations for the next 15 months. |
Cash
Proceeds from Financing Activities
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Cash proceeds from financing activities | |
| | | |
| | |
| Proceeds from Subsidiary stock transactions | |
$ | — | | |
$ | 600,000 | |
| Proceeds from stock transactions | |
| 680,000 | | |
| | |
| Proceeds from issuance of convertible notes payable | |
| 1,380,460 | | |
| 1,165,000 | |
| Net cash provided by financing activities | |
$ | 2,060,960 | | |
$ | 1,765,000 | |
| |
During
the twelve months ending December 31, 2022, the Company had raised $1,467,000 through an 8-month convertible notes at 6% interest,
with net cash proceeds of $1,380,460, as well as 680,000 in net cash for private placements. During the twelve months ending December
31, 2021, the Company had raised $600,000 in cash proceeds from the issuance of Common Stock in our Subsidiary and $1,165,000 cash
generating 1-year convertible notes at 6% interest, extended through April 30, 2024, with net cash proceeds of $1,045,150. The Company
is aware that its current cash on hand will not be sufficient to fund its projected operating requirements through the month of May
2023. |
RESULTS
OF OPERATIONS FOR THE THREE AND SIX MONTHS ENDED JUNE 30, 2023
We
are a clinical stage company. Historically, Bioxytran was engaged in formation, fund raising and identifying and consulting with the
scientific community regarding the development, formulation and testing of its products as of the fourth quarter of 2021 the Company
has engaged in research and development activities through its Subsidiary, Pharmalectin, Inc., developing the Company’s anti-viral
therapeutic ProLectin. In the second quarter of the company also started the development of our hypoxia platform technology Acellular
Oxygen Carrier (AOC).
Research
and Development
| | |
Three months ended | | |
Six months ended | |
| | |
June 30,
2023 | | |
June 30,
2022 | | |
June 30,
2023 | | |
June 30,
2022 | |
| Research and development: | |
| | | |
| | | |
| | | |
| | |
| Process development | |
$ | 125,439 | | |
$ | — | | |
$ | 125,439 | | |
$ | — | |
| Product development | |
| 19,939 | | |
| 40,834 | | |
| 19,939 | | |
| 99,723 | |
| Regulatory | |
| 12,510 | | |
| (15,000 | ) | |
| 59,514 | | |
| 33,850 | |
| Clinical trials | |
| (12,250 | ) | |
| 70,692 | | |
| 61,750 | | |
| 147,283 | |
| Project management | |
| 4,000 | | |
| (53,385 | ) | |
| 22,000 | | |
| 2,410 | |
| Total research and development | |
$ | 149,638 | | |
$ | 43,141 | | |
$ | 288,642 | | |
$ | 283,266 | |
| |
During
the three months ended June 30, 2023, the Company recorded $149,638 in R&D expenses. During the three months ended June 30, 2023,
the Company recorded $43,141 in R&D expenses after a receiving a $300,000 refund from a Contract Research Organization (CRO).
During the six months ended June 30, 2023, the Company recorded $288,642 in R&D expenses. During the six months ended June 30,
2023, the Company recorded $283,266 in R&D expenses. During the 6 months ending in June, 2023, $98,265 was invested in, ProLectin,
while 190,377 was invested in the AOC. All prior development was focused on ProLectin, only. |
General
and Administrative
| | |
Three months ended | | |
Six months ended | |
| | |
June 30,
2023 | | |
June 30,
2022 | | |
June 30,
2023 | | |
June 30,
2022 | |
| General and administrative expenses: | |
| | | |
| | | |
| | | |
| | |
| Payroll and related expenses | |
$ | 393,529 | | |
$ | 339,995 | | |
$ | 752,670 | | |
$ | 717,110 | |
| Costs for legal, accounting and other professional services | |
| 77,058 | | |
| 25,995 | | |
| 120,171 | | |
| 48,320 | |
| Promotional expenses | |
| 363,698 | | |
| 31,500 | | |
| 528,949 | | |
| 148,700 | |
| Miscellaneous expenses | |
| 32,647 | | |
| 49,870 | | |
| 62,780 | | |
| 89,811 | |
| Total general and administrative | |
$ | 866,932 | | |
$ | 447,360 | | |
$ | 1,464,570 | | |
$ | 1,003,941 | |
| |
Payroll
and related expenses for the three months ended June 30, 2022 were $393,529 for the 3 months ended June 30, 2023 and $752,670
for the 6 months ended June 30, 2023. For the same periods in 2022 the amount was $339,995 and $717,110, respectively. |
| |
|
| |
The
Costs for legal, accounting and other professional services for the three and six months ended June 30, 2023 were $77,058
and $120,171 respectively, as compared to $25,995 and $48,320 for the three and six months ended June 30, 2022. The increased costs
are for contracted investments services for an amount of $75,000 in the second quarter of 2023. |
| |
|
| |
Promotional
expenses for the three and six months ended June 30, 2023 were $363,698 and $528,949 respectively, as compared to $31,500 and
$148,700 for the three and six months ended June 30, 2022. The increase costs stock promotion incurred by the Company’s return
to being listed on OTCQB. The Company has currently a non-reimbursable advance paid Public Relations Agreement running through July
31, 2024. |
| |
|
| |
Miscellaneous
G&A expenses during the three and six months ended June 30, 2023 was $32,647 and $62,780, respectively. During the three
and six months ended June 30, 2022 was $49,870 and $89,811. |
Stock-based
Compensation
| | |
Three months ended | | |
Six months ended | |
| | |
June 30,
2023 | | |
June 30,
2022 | | |
June 30,
2023 | | |
June 30,
2022 | |
| Compensation expense to BoD and Management | |
$ | 4,750 | | |
$ | 49,840 | | |
$ | 17,500 | | |
$ | 56,240 | |
| Compensation expense to consultants | |
| 360 | | |
| (3,117 | ) | |
| 1,210 | | |
| 12,883 | |
| Total compensation expense | |
$ | 5,110 | | |
$ | 46,723 | | |
$ | 18,710 | | |
$ | 69,123 | |
| |
Stock-based
compensation amounted to $5,110 for the three months ended June, 2023. The stock-based compensation for the three months ended June
30, 2022 was $46,723. Stock-based compensation amounted to $18,710 for the six months ended June, 2023. Stock-based compensation
amounted to $69,123 for the six months ended June, 2022. |
Other
expenses
| | |
Three months ended | | |
Six months ended | |
| | |
June 30,
2023 | | |
June 30,
2022 | | |
June 30,
2023 | | |
June 30,
2022 | |
| Other (expenses): | |
| | | |
| | | |
| | | |
| | |
| Interest expense | |
$ | 39,477 | | |
$ | 54,480 | | |
$ | 106,698 | | |
$ | 106,515 | |
| Debt discount amortization | |
| — | | |
| 41,425 | | |
| — | | |
| 90,509 | |
| Amortization of warrants | |
| 348,637 | | |
| — | | |
| 348,637 | | |
| 42,250 | |
| Amortization of IP | |
| 2,188 | | |
| 911 | | |
| 2,702 | | |
| 1,822 | |
| Total other income (expenses) | |
$ | 390,302 | | |
$ | 96,816 | | |
$ | 458,037 | | |
$ | 241,096 | |
| |
During
the three months ended June 30, 2023, the Company recorded $348,637 in amortization warrants
and the interest expense was $39,477, $2,188 was amortized from the Company’s
IP. During the three months ended June 30, 2022, the Company recorded $41,425 in amortization
of debt discount while the interest expense was $54,480, $911 was amortized from the Company’s
IP.
During
the six months ended June 30, 2023, the Company amortized $2,702 from the Company’s IP and $348,637 in amortization of warrants,
as compared to, $1,822 in IP amortization and $42,250 of warrant amortization of for the six months ended June 30, 2022. The interest
for the six months ended June 30, 2023 for the convertible notes amounted to $106,698, as compared to $106,515 for the six months
ended June 30, 2022. |
Non-Controlling
Interest
| | |
Three months ended | | |
Six months ended | |
| | |
June 30,
2023 | | |
June 30,
2022 | | |
June 30,
2023 | | |
June 30,
2022 | |
| Net loss attributable to the non-controlling interest | |
$ | 764 | | |
$ | 11,691 | | |
$ | 33,658 | | |
$ | 62,807 | |
| |
For
the three months ended June 30, 2023 and 2022 there was a non-controlling interest attribution of $764 and 11,691 respectively. For
the six months ended June 30, 2023 and 2022 there was a non-controlling interest attribution of $33,658 and $62,807 respectively.
The significant difference is directly related to the Company’s R&D activities due to lack of capital. |
| | |
# of shares | | |
# of options * | | |
June 30, 2023 | | |
December 31, 2022 | |
| Minority owners cash investment | |
| 4,650,000 | | |
| | | |
$ | 160,485 | | |
$ | 160,485 | |
| Bioxytran non-dilutive equity | |
| 15,000,000 | | |
| | | |
| 1,500 | | |
| 1,500 | |
| Issued stock options @ $0.33 | |
| | | |
| 4,500,000 | | |
| 450 | | |
| 450 | |
| Total outstanding | |
| 19,650,000 | | |
| 4,500,000 | | |
$ | 162,435 | | |
$ | 162,435 | |
| |
As
at June 30, 2023 and at December 31, 2022 there are 30,000,000 issued and 19,650,000 outstanding
shares; 15,000,000 Common shares (76%) are held by Bioxytran and 4,650,000 Common shares
are held by an affiliate. Further, an additional 4,500,000 options to purchase Common shares
exercisable at $0.33 are held by an affiliate.
*
The option agreements are held by an affiliate and include provisions for dilutive issuance and cash-less exercise. If exercised
at June 30, 2023 the provisions would result in an issuance of 11,423,077 shares at an average conversion-price of $0.13. The beneficial
ownership of the affiliate includes the Company’s management. |
Net
Loss
| | |
Three months ended | | |
Six months ended | |
| | |
June 30,
2023 | | |
June 30,
2022 | | |
June 30,
2023 | | |
June 30,
2022 | |
| Net loss attributable to Bioxytran | |
$ | (1,411,218 | ) | |
$ | (622,349 | ) | |
$ | (2,196,301 | ) | |
$ | (1,534,619 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Loss per Common share, basic and diluted | |
$ | (0.01 | ) | |
$ | (0.01 | ) | |
$ | (0.02 | ) | |
$ | (0.01 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Weighted average number of Common shares outstanding, basic | |
| 128,804,789 | | |
| 110,840,998 | | |
| 126,224,323 | | |
| 110,840,998 | |
| |
The
Company generated a net loss for the three months ended June 30, 2023 of $1,411,218. In comparison, for the three months ended June
30, 2022, the Company generated a net loss of $622,349. The Company generated a net loss for the six months ended June 30, 2023 of
$2,196,301. In comparison, for the six months ended June 30, 2022, the Company generated a net loss of $1,534,619. The significant
difference is directly related to the Company’s R&D activities due to lack of capital in 2022 |
CASH-FLOWS
| | |
Six months ended | |
| | |
June 30, 2023 | | |
June 30, 2022 | |
| Net cash used in operating activities | |
$ | (340,173 | ) | |
$ | (930,203 | ) |
| | |
| | | |
| | |
| Net cash used in investing activities | |
| (25,047 | ) | |
| (22,438 | ) |
| | |
| | | |
| | |
| Net cash provided by financing activities | |
| 115,000 | | |
| 1,380,960 | |
| | |
| | | |
| | |
| Net increase (decrease) in cash | |
$ | (250,220 | ) | |
$ | 428,319 | |
| Cash, beginning of period | |
| 295,401 | | |
| 72,358 | |
| Cash, end of period | |
| 45,181 | | |
| 500,667 | |
| |
Net
cash used in operating activities was $340,173 and $930,203 for the Six months ended June 30, 2023 and 2022, respectively. The
decrease was due to a reduction of the research and development activities due to lack of funding. |
| |
|
| |
In
the Six months ended June 30, 2023 the Company is in the process of filing a patent, and $25,047 was spent in legal fees. In the
Six months ended June 30, 2022 the amount was $22,438. |
| |
|
| |
During
the Six months ending June 30, 2023, the Company had raised $115,000 through issuance of common shares. In the period ended June
30,2022 the company entered agreements for thirty-eight (38) convertible notes at 6% interest, with net cash proceeds of $1,380,460.
The convertible notes have, since then, been converted to Common Stock. |
| |
|
| |
The
available cash was $45,181 and $500,667 in the end of the Six months ended June 30, 2023 and 2022, respectively. |
LIQUIDITY
AND CAPITAL RESOURCES
Cash
and Cash Equivalents
| |
|
June
30, 2023 |
|
|
December
31, 2022 |
|
| Current
assets: |
|
|
|
|
|
|
|
|
| Cash |
|
$ |
45,181 |
|
|
$ |
295,401 |
|
| Total
current assets |
|
$ |
45,181 |
|
|
$ |
295,401 |
|
As
of June 30, 2023, our current assets consisted of $45,181 of cash at December 31, 2022 we had $295,401 of cash.
Current
Liabilities
| |
|
June
30, 2023 |
|
|
December
31, 2022 |
|
| Current
liabilities: |
|
|
|
|
|
|
|
|
| Accounts
payable and accrued expenses |
|
$ |
562,662 |
|
|
$ |
749,395 |
|
| Accounts
payable related party |
|
|
200,500 |
|
|
|
709,727 |
|
| Un-issued
shares liability |
|
|
380 |
|
|
|
960 |
|
| Un-issued
shares liability related party |
|
|
5,700 |
|
|
|
38,400 |
|
| Convertible
notes payable, net of discount |
|
|
2,000,000 |
|
|
|
2,165,000 |
|
| Total
current liabilities |
|
|
2,769,242 |
|
|
|
3,663,482 |
|
At
June 30, 2023 we had total liabilities of $3,269,243, which consisted of $763,163 in accounts payable and accrued expenses (of which
$200,502 was payable to related parties), $6,080 in un-issued shares (of which $5,700 was payable to related parties and $2,000,000 in
two convertible loans. At December 31, 2022 total liabilities were $3,663,482, consisting of $1,459,121 in accounts payable and accrued
expenses (of which $709,727 was payable to related parties), $39,360 in un-issued shares (of which $38,400 was payable to related parties)
and $2,165,000 in the form of four convertible loans net of discount.
Net
Working Capital and Accumulated Deficit
| | |
June 30, 2023 | | |
December 31, 2022 | |
| Net working capital | |
$ | (2,724,061 | ) | |
$ | (3,368,081 | ) |
| | |
| | | |
| | |
| Accumulated deficit | |
$ | (13,413,901 | ) | |
$ | (11,217,600 | ) |
At
June 30, 2023, the net working capital was negative $2,724,061 and the accumulated deficit of $13,413,901. Comparatively, on December
31, 2022, we had net working capital of negative $3,368,081 and an accumulated deficit of $11,217,600. We believe that we must raise
an additional $3,700,000 to be able to continue our business operations for the next 15 months.
Upcoming
Financing Activities
On
March 13, 2023 the Company filed a Form D with the SEC, wherein we show the intention to make a capital raise in the order of $5.0 million
during the second half of 2023.
There
can be no assurance that these funds will be available on terms acceptable to the Company, or will be sufficient to enable the Company
to fully complete its development activities or sustain operations. If the Company is unable to raise sufficient additional funds, it
will have to develop and implement a plan to further extend payables, reduce overhead, or scale back its current business plan until
sufficient additional capital is raised to support further operations. There can be no assurance that such a plan will be successful.
Commitments
We
have no current commitment from our officers and Directors or any of our Shareholders, to supplement our operations or provide us with
financing in the future. If we are unable to raise additional capital from conventional sources and/or additional sales of stock in the
future, we may be forced to curtail or cease our operations. Even if we are able to continue our operations, the failure to obtain financing
could have a substantial adverse effect on our business and financial results. In the future, we may be required to seek additional capital
by selling debt or equity securities, selling assets, or otherwise be required to bring cash flows in balance when we approach a condition
of cash insufficiency. The sale of additional equity or debt securities, if accomplished, may result in dilution to our then Shareholders.
We provide no assurance that financing will be available in amounts or on terms acceptable to us, or at all.
Contractual
Obligations
| | |
June 30, 2023 | | |
December 31, 2022 | |
| Interest on notes payable | |
$ | 149,377 | | |
$ | 134,581 | |
| Convertible notes payable | |
| 2,165,000 | | |
| 2,165,000 | |
| Total | |
$ | 2,314,377 | | |
$ | 2,299,581 | |
| |
As
at June 30, 2023, our contractual obligations include four convertible notes, for a total of $2,165,000 and of accrued interest for
these notes mounting to $149,377, as at December 31, 2022 there were four convertible notes, for a total of $2,165,000 and of accrued
interest for these notes mounting to $134,581. |
Our
Executive Officers have entered into employment contracts and confidentiality, non-disclosure and assignment of invention agreements.
The most substantial provisions include;
| |
● |
Compensation
of three (3) times the employee’s annual salary upon the Termination Date and any target bonus earned, or if termination occurs
within 12 months of a change in control, then the terminated employee shall receive two (2) times the employee’s annual salary
and any target bonus earned. |
| |
● |
Continued
coverage under any health, medical, dental or vision program or policy, in which they were eligible to participate at the time of
employment termination, for 12 months. |
| |
● |
Provide
outplacement services through one or more outside firms of the employee’s choosing up to an aggregate of $50,000. |
Off-Balance
Sheet Arrangements
We
do not have any off-balance sheet arrangements that have, or are reasonably likely to have, a current or future material effect on our
consolidated financial condition, results of operations, liquidity, capital expenditures or capital resources.
CRITICAL
ACCOUNTING POLICIES
In
presenting our financial statements in conformity with generally accepted accounting principles, we are required to make estimates and
assumptions that affect the amounts reported therein. Several of the estimates and assumptions we are required to make relate to matters
that are inherently uncertain as they pertain to future events. However, events that are outside of our control cannot be predicted and,
as such, they cannot be contemplated in evaluating such estimates and assumptions. If there is a significant unfavorable change to current
conditions, it could result in a material adverse impact to our results of operations, financial position and liquidity. We believe that
the estimates and assumptions we used when preparing our financial statements were the most appropriate at that time. Presented below
are those accounting policies that we believe require subjective and complex judgments that could potentially affect reported results.
However, the majority of our businesses operate in environments where we pay a fee for a service performed, and therefore the results
of the majority of our recurring operations are recorded in our financial statements using accounting policies that are not particularly
subjective, nor complex.
Stock
Based Compensation
The
Company has share-based compensation plans under which non-employees, consultants and suppliers may be granted restricted stock, as well
as options to purchase shares of Company Common Stock at the fair market value at the time of grant. Stock-based compensation cost is
measured by the Company at the grant date, based on the fair value of the award over the requisite service period.
The
Company applies ASC 718 for options, Common Stock and other equity-based grants to its employees and Directors. ASC 718 requires measurement
of all employee equity-based payment awards using a fair-value method and recording of such expense in the consolidated financial statements
over the requisite service period. The fair value concepts have not changed significantly in ASC 718; however, in adopting this standard,
companies must choose among alternative valuation models and amortization assumptions. After assessing alternative valuation models and
amortization assumptions, the Company will continue using both the Black-Scholes valuation model and straight-line amortization of compensation
expense over the requisite service period for each separately vesting portion of the grant.
Recent
Accounting Standards
In
August 2020, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) 2020-06,
Debt — Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging — Contracts in Entity’s
Own Equity (Subtopic 815-40) (“ASU 2020-06”) to simplify accounting for certain financial instruments. ASU 2020-06 eliminates
the current models that require separation of beneficial conversion and cash conversion features from convertible instruments and simplifies
the derivative scope exception guidance pertaining to equity classification of contracts in an entity’s own equity. The new standard
also introduces additional disclosures for convertible debt and freestanding instruments that are indexed to and settled in an entity’s
own equity. ASU 2020-06 amends the diluted earnings per share guidance, including the requirement to use the if-converted method for
all convertible instruments. ASU 2020-06 is effective January 1, 2022, and should be applied on a full or modified retrospective basis,
with early adoption permitted beginning on January 1, 2021. The Company adopted ASU 2020-06 effective January 1, 2021. The adoption of
AASU 2020-06 did not have an impact on the Company’s financial statements.
DESCRIPTION
OF PROPERTY
We
do not currently own any real property. We lease access to shared office space at 75, Second Ave., Suite 605, Needham, MA 02494 on a
month-to-month basis for $163 per month. We believe this facility is adequate for our current needs. As we receive funding and our operations
expand, we anticipate that we will seek to lease additional office space.
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS
From
the date of the Company’s Merger on September 21, 2018, we have not entered into any material transactions or series of transactions
that would be considered material in which any officer, Director or beneficial owner of 5% or more of any class of our capital stock,
or any immediate family member of any of the preceding persons, had a direct or indirect material interest, and there are no transactions
presently proposed, except as follows:
The
Company hold License Agreements (the “License/s” or “Agreement/s”) for a medical device (license obtained in
2019) and a compound (license obtained in 2021), with two affiliated companies where in the officers of the Company hold a majority interest.
The products were developed prior to the establishment of Bioxytran. The yearly maintenance fee for each license is five thousand Dollars
($5,000). For more detailed information, see Exhibits 10.22 and 10.60 in the below. As at June 30, 2023, there are $5,000 owed to an
affiliate.
As
at June 30, 2023, the Company has accrued a total amount of seventy-one thousand Dollars ($71,000)
to David Platt, seventy thousand Dollars ($70,000) to Ola Soderquist and fifty-four thousand
five hundred Dollars ($54,500) to Mike Sheikh in salary and expenses.
DIRECTOR
AND EXECUTIVE COMPENSATION
The
following table sets forth information concerning all cash all cash and non-cash compensation awarded to, earned by or paid to the Company’s
Chief Executive Officer (“CEO”), Chief Financial Officer (“CFO”) and the Chief Communications Officer (“CCO”),
regardless of compensation level. The Company’s CEO, CFO and the CCO are the only Officers of the Company for whom compensation
disclosure is required pursuant to instruction 1 to Item 402(m)(2) of Regulation S-K.
Summary
Compensation Table
| Name and Principal Position | |
Year | | |
Salary | | |
Bonus | | |
Stock
Awards | | |
Total Compensation | |
| David Platt, Chairman of the Board, | |
| 2022 | | |
$ | 108,900 | | |
$ | — | | |
$ | — | | |
$ | 108,900 | |
| CEO and President | |
| 2021 | | |
$ | 214,000 | | |
$ | — | | |
$ | — | | |
$ | 214,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Ola Soderquist, CFO | |
| 2022 | | |
$ | 108,900 | | |
$ | — | | |
$ | — | | |
$ | 108,900 | |
| | |
| 2021 | | |
$ | 214,000 | | |
$ | — | | |
$ | — | | |
$ | 214,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Mike Sheikh, CCO | |
| 2022 | | |
$ | 43,427 | | |
$ | — | | |
$ | — | | |
$ | 43,427 | |
| | |
| 2021 | | |
$ | 105,000 | | |
$ | — | | |
$ | — | | |
$ | 105,000 | |
Grants
of Plan-Based Awards
There
were no equity awards to the Company’s Executive Officers during the years ended at December 31, 2022, and 2021.
Outstanding
Equity Awards at December 31, 2022; Option exercises and vested
There
were no outstanding options or equity awards held by the Company’s Executive Officers at December 31, 2022.
Director
Compensation
All
compensation paid to our employee Directors is set forth in the table summarizing Executive Officer compensation above. Our non-employee
Directors currently are entitled to receive ten thousand (10,000) shares of our Common Stock for each board and/or committee meeting
that they attend per quarter in arrears. There were two hundred eighty thousand (280,000) shares, at a fair market value of seventy-eight
thousand four hundred four Dollars ($78,404), issued as compensation to the board in 2022. There were one million two hundred ninety-one
thousand two hundred (1,291,200) shares, at a fair market value of two hundred thirty-eight thousand eight hundred eight Dollars ($238,808),
issued in 2021. Except for the foregoing, there are currently no agreements in effect entitling them to compensation.
Employment
Contracts
Our
Executive Officers have entered into employment contracts and confidentiality, non-disclosure and assignment of invention agreements.
The most substantial provisions include;
| |
● |
Compensation
of three (3) times the employee’s annual salary upon the Termination Date and any target bonus earned, or if termination occurs
within twelve (12) months of a change in control, then the terminated employee shall receive two (2) times the employee’s annual
salary and any target bonus earned. |
| |
● |
Continued
coverage under any health, medical, dental or vision program or policy, in which they were eligible to participate at the time of
employment termination, for twelve (12) months. |
| |
● |
Provide
outplacement services through one or more outside firms of the employee’s choosing up to an aggregate of fifty thousand Dollars
($50,000). |
There
are no other arrangements or plans in which we provide pension, retirement or similar benefits for any of Executive Officers or Directors.
The
Board has set the monthly salary for David Platt and Ola Soderquist to thirty-five thousand Dollars ($35,000) and for Mike Sheikh of
Twenty-six thousand two hundred Dollars ($26,250). Additionally, along with a 25% 401(k) Safe Harbor coverage up to the federal limit,
currently $66,000 per year plus potential catchup, currently $7,500, as well as reimbursement of a gold-level healthcare plan.
Our
Executive Officers and Directors may also receive stock or stock options at the discretion of our Board according to the approved 2021
Stock Plan, or any subsequent Stock Plan.
Compensation
Risk Assessment
We
have formed a Compensation Committee. In setting compensation, the Compensation Committee will consider the risks to the Company’s
Stockholders and to achievement of its goals that may be inherent in its compensation programs. The Compensation Committee will review
and discuss its assessment with management and outside legal counsel to confirm that the Company’s compensation programs are and
will be within industry standards and designed with the appropriate balance of risk and reward to align employees’ interests with
those of the Company without incenting employees to take unnecessary or excessive risks. We believe our compensation plans will be appropriately
structured consistent with the Company’s status as a pre-revenue start-up enterprise and will not be reasonably likely to result
in a material adverse effect on the Company.
Securities
Authorized for Issuance under Equity Compensation Plans
Securities
Authorized for Issuance under Equity Compensation Plans
On
January 18, 2020, the 2010 Employee, Director and Consultant Stock Plan (the “2010 Plan”) expired and on January
19, 2021, the Company established a 2021 Employee, Director and Consultant Stock Plan (the “2021 Plan”). The
2021 Plan was approved by the Company’s Board and by the majority of the Shareholders. The material features of the 2021
Plan are described below and are identical to the expired 2010 Plan.
Administration
A
designated administrator, or in the absence of such, our Boards’ Compensation Committee or both, in the sole discretion of our
Board, administers the 2021 Plan, which was approved by the Company’s Board on January 19, 2021. The Board, subject to the provisions
of the 2021 Plan, has the authority to determine and designate officers, employees, Directors and consultants to whom awards shall be
made and the terms, conditions and restrictions applicable to each award (including, but not limited to, the option price, any restriction
or limitation, any vesting schedule or acceleration thereof, and any forfeiture restrictions). The Board may, in its sole discretion,
accelerate the vesting of awards. The Board of Directors must approve all grants of Options and Stock Awards issued to our officers or
Directors.
Types
of Awards
The
2021 Plan is designed to enable us to offer certain officers, employees, Directors and consultants of us and our subsidiaries equity
interests in us and other incentive awards in order to attract, retain and reward such individuals and to strengthen the mutuality of
interests between such individuals and our stockholders. In furtherance of this purpose, the 2021 Plan contains
provisions for granting incentive and non-statutory stock options, stock wards and stock appreciation rights.
Stock
Options. A “stock option” is a contractual right to purchase a number of shares of Common Stock at a price determined
on the date the option is granted. The option price per share of Common Stock purchasable upon exercise of a stock option and the time
or times at which such options shall be exercisable shall be determined by the Board at the time of grant. Such option price shall not
be less than 100% of the fair market value of the Common Stock on the date of grant. The option price must be paid in cash, money order,
check or Common Stock of the Company. The Options may also contain at the time of grant, at the discretion of the Board,
certain other cashless exercise provisions.
Options
shall be exercisable at the times and subject to the conditions determined by the Board at the date of grant, but no option may be exercisable
more than ten years after the date it is granted. If the Optionee ceases to be an employee of our Company for any reason other than death,
any option granted as an Incentive Stock Option exercisable on the date of the termination of employment may be exercised for a period
of thirty days or until the expiration of the stated term of the option, whichever period is shorter. In the event of the Optionee’s
death, any granted Incentive Stock Option exercisable at the date of death may be exercised by the legal heirs of the Optionee from the
date of death until the expiration of the stated term of the option or six months from the date of death, whichever event first occurs. In
the event of disability of the Optionee, any granted Incentive Stock Options shall expire on the stated date that the Option would otherwise
have expired or 12 months from the date of disability, whichever event first occurs. The termination and other provisions
of a non-statutory stock option shall be fixed by the Board at the date of grant of each respective option.
Common
Stock Award. “Common Stock Award” is shares of Common Stock that will be issued to a recipient at the end of a restriction
period, if any, specified by the Board if he or she continues to be an employee, Director or consultant of us. If the recipient remains
an employee, Director or consultant at the end of the restriction period, the applicable restrictions will lapse and we will issue a
stock certificate representing such shares of Common Stock to the participant. If the recipient ceases to be an employee, Director or
consultant of us for any reason (including death, disability or retirement) before the end of the restriction period unless otherwise
determined by the Board, the restricted stock award will be terminated.
Eligibility
The
Company’s officers, employees, Directors and consultants of Bioxytran, Inc. are eligible to be granted stock options, and Common
Stock Awards. Eligibility shall be determined by the Board; however, all Options and Stock Awards granted to officers
and Directors must be approved by the Board.
Termination
or Amendment of the 2021 Plan
The
Board may at any time amend, discontinue, or terminate all or any part of the 2021 Plan, provided, however, that unless otherwise required
by law, the rights of a participant may not be impaired without his or her consent, and provided that we will seek the approval of our
Stockholders for any amendment if such approval is necessary to comply with any applicable federal or state securities laws or rules
or regulations.
Awards
During
the year ended December 31, 2022, three hundred two thousand (302,000) shares were issued to the Board and the Company’s Advisory
Board for their attendance in meetings during the year, while two hundred fifty thousand (250,000) shares to consultants from the 2021
Plan. Awards to consultants are made when the Company does not have sufficient cash to pay for the services provided to the Company.
During
the six months ended June 30, 2023, there were one hundred fourteen thousand (114,000) shares issued to the Company’s Directors.
There was a stock-based compensation expense of $18,710 in connection with share-based payment awards.
Shares
Subject to the 2021 Plan
Subject
to adjustment, the aggregate number of shares of Stock which may be delivered under the 2021 Plan shall not exceed a number equal to
fifteen percent (15%) of the total number of shares of Stock outstanding immediately following the Effective Time, assuming for this
purpose the conversion into Stock of all outstanding securities that are convertible by their terms (directly or indirectly) into Stock; provided,
however, that, as of January 1 of each calendar year, commencing with the year 2011, the maximum number of shares of Stock which
may be delivered under the 2021 Plan shall automatically increase by a number sufficient to cause the number of shares of Stock covered
by the 2021 Plan to equal 15% of the total number of shares of Stock then outstanding, assuming for this purpose the conversion into
Stock of all outstanding securities that are convertible by their terms (directly or indirectly) into Stock. As at June 30, 2023,
there are nineteen million eight hundred thirty-two thousand seventy-one (19,832,071) options or stock awards available for grant under
the 2021 Plan.
Federal
Tax Consequences
The
federal income tax discussion set forth below is intended for general information only. State and local income tax consequences are not
discussed, and may vary from locality to locality.
Incentive
Stock Options. Incentive stock options granted under the 2021 Plan are designed to qualify for the special tax treatment
for incentive stock options provided for in the Internal Revenue Code (the “Code”). Under the provisions of
the Code, an optionee who at all times from the date of grant until three months before the date of exercise is an employee of the Company,
and who holds the shares of Common Stock obtained upon exercise of his incentive stock option for two years after the date of grant and
one year after exercise, will recognize no taxable income on either the grant or exercise of such option and will recognize capital gain
or loss on the sale of the shares. If such shares are held by the optionee for the required holding period, the Company
will not be entitled to any tax deduction with respect to the grant or exercise of the option. If such shares are sold
by the optionee prior to the expiration of the holding periods described above, the optionee will recognize ordinary income upon such
disposition. Upon the exercise of an incentive stock option, the optionee will incur an item of tax preference equal to
the excess of the fair market value of the shares at the time of exercise over the exercise price, which may subject the optionee to
the alternative minimum tax.
Non-Qualified
Options. Under present Treasury regulations, an optionee who is granted a non-qualified option will not realize taxable income at
the time the option is granted. In general, an optionee will be subject to tax for the year of exercise on an amount of ordinary income
equal to the excess of the fair market value of the shares on the date of exercise over the option price, and the Company will receive
a corresponding deduction. Income tax withholding requirements apply upon exercise. The optionee’s basis in the shares so acquired
will be equal to the option price plus the amount of ordinary income upon which he is taxed. Upon subsequent disposition of the shares,
the optionee will realize capital gain or loss, long-term or short-term, depending upon the length of time the shares are held after
the option is exercised.
Common
Stock Awards. Recipients of shares of restricted Common Stock that are not “transferable” and are subject to “substantial
risk of forfeiture” at the time of grant will not be subject to Federal income taxes until lapse or release of the restrictions
on the shares. The recipient’s income and the Company’s deduction will be equal to the fair market value of the shares on
the date of lapse or release of such restrictions. It has been the Company’s policy to value the cost of the issuance of said unregistered
shares at the then bid price of the stock when issued.
The
issuance of any of our common or Preferred Stock is within the discretion of our Board, which has the power to issue any or all of our
authorized but unissued shares without stockholder approval.
Corporate
Governance
The
Company has established and approved charters for separate audit, compensation and nominating/governance committees of its Board of Directors.
Code
of Ethics. A code of business conduct and ethics is a written standard designed to deter wrongdoing and to promote (a) honest and ethical
conduct, (b) full, fair, accurate, timely and understandable disclosure in regulatory filings and public statements, (c) compliance with
applicable laws, rules and regulations, (d) the prompt reporting violation of the code and (e) accountability for adherence to the code.
We are not currently subject to any law, rule or regulation requiring that we adopt a code of ethics; though the Code of Ethics has been
adopted.
Audit
Committee. Our Board has established an audit committee, whose members are initially Anders Utter, as Chairman, Alan Hoberman and Dale
Conaway.
Nominating
and Governance Committee. Our Board has established a nominating and governance committee, whose initial members are Alan Hoberman, Chairman,
Anders Utter, and Dale Conaway.
Compensation
Committee. The Board has appointed Dale Conaway, Chairman, Anders Utter and Alan Hoberman to our compensation committee.
Indemnification
Agreements
Our
By-laws provide for the indemnification of Directors and Officers. See “Indemnification of Directors and Officers. As a Nevada
corporation we are generally governed by the Nevada Private Corporations Code, Title 78 of the Nevada Revised Statutes, or NRS.
Section
78.138 of the NRS provides that, unless the corporation’s articles of incorporation provide otherwise, a director or officer will
not be individually liable unless it is proven that (i) the director’s or officer’s acts or omissions constituted a breach
of his or her fiduciary duties, and (ii) such breach involved intentional misconduct, fraud, or a knowing violation of the law. Our articles
of incorporation provide the personal liability of our directors is eliminated to the fullest extent permitted under the NRS.
Section
78.7502 of the NRS permits a company to indemnify its directors and officers against expenses, judgments, fines, and amounts paid in
settlement actually and reasonably incurred in connection with a threatened, pending, or completed action, suit, or proceeding, if the
officer or director (i) is not liable pursuant to NRS 78.138, or (ii) acted in good faith and in a manner the officer or director reasonably
believed to be in or not opposed to the best interests of the corporation and, if a criminal action or proceeding, had no reasonable
cause to believe the conduct of the officer or director was unlawful. Section 78.7502 of the NRS requires a corporation to indemnify
a director or officer that has been successful on the merits or otherwise in defense of any action or suit. Section 78.7502 of the NRS
precludes indemnification by the corporation if the officer or director has been adjudged by a court of competent jurisdiction, after
exhaustion of all appeals, to be liable to the corporation or for amounts paid in settlement to the corporation, unless and only to the
extent that the court determines that in view of all the circumstances, the person is fairly and reasonably entitled to indemnity for
such expenses and requires a corporation to indemnify its officers and directors if they have been successful on the merits or otherwise
in defense of any claim, issue, or matter resulting from their service as a director or officer.
Section
78.751 of the NRS permits a Nevada company to indemnify its officers and directors against expenses incurred by them in defending a civil
or criminal action, suit, or proceeding as they are incurred and in advance of final disposition thereof, upon determination by the stockholders,
the disinterested board members, or by independent legal counsel. If so provided in the corporation’s articles of incorporation,
bylaws, or other agreement, Section 78.751 of the NRS requires a corporation to advance expenses as incurred upon receipt of an undertaking
by or on behalf of the officer or director to repay the amount if it is ultimately determined by a court of competent jurisdiction that
such officer or director is not entitled to be indemnified by the company. Section 78.751 of the NRS further permits the company to grant
its directors and officers additional rights of indemnification under its articles of incorporation, bylaws, or other agreement.
Section
78.752 of the NRS provides that a Nevada company may purchase and maintain insurance or make other financial arrangements on behalf of
any person who is or was a director, officer, employee, or agent of the company, or is or was serving at the request of the company as
a director, officer, employee, or agent of another company, partnership, joint venture, trust, or other enterprise, for any liability
asserted against him and liability and expenses incurred by him in his capacity as a director, officer, employee, or agent, or arising
out of his status as such, whether or not the company has the authority to indemnify him against such liability and expenses.
Director
Independence.
Our
Board consists of five members. We are not currently subject to any law, rule or regulation requiring that all or any portion of our
Board include “independent” directors. Four of the members of the Board, Dale H. Conaway, D.V.M., Anders Utter, Alan Hoberman
and Hana Chen-Walden are “independent” as defined in Section 4200(a)(15) of NASDAQ Stock Market Rules.
CHANGES
IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
On
March 6, 2023, the Board of Bioxytran agreed to dismiss the Company’s independent registered public accounting firm, Pinnacle Accountancy
Group of Utah (“Pinnacle”), effective as of March 06, 2023. Also on March 6, 2023, the Company engaged the accounting firm
of BF Borgers CPA PC (“BF Borgers”) as the Company’s new independent registered public accounting firm. The Board and
the Company’s Audit Committee approved of the dismissal of Pinnacle and the engagement of BF Borgers. None of the reports of Pinnacle
regarding the Company’s financial statements for the past year, since inception or any subsequent interim period contained an adverse
opinion or disclaimer of opinion, or was qualified or modified as to uncertainty, audit scope or accounting principles, except that the
Company’s audited financial statements contained in its Form 10-K for the fiscal years ended December 31, 2021 and 2020, contain
a going concern qualification.
During
Bioxytran’s most recent fiscal year, since inception and the subsequent interim periods thereto, there were no disagreements between
the Company and Pinnacle whether or not resolved, on any matter of accounting principles or practices, financial statement disclosure,
or auditing scope or procedure, which, if not resolved to Pinnacle ‘s satisfaction, would have caused it to make reference to the
subject matter of the disagreement in connection with its report on the Company’s financial statements.
DESCRIPTIONS
OF CAPITAL STOCK
We
have authorized capital stock consisting of three hundred million (300,000,000) shares of Common Stock, $.001 par value per share (“Common
Stock”) and fifty million (50,000,000) shares of Preferred Stock, $.001 par value per share (“Preferred Stock”). As
of September 18, 2023, we had one hundred forty-three million seven hundred eighty-four thousand nine hundred eighty-five (143,784,985)
shares of Common Stock issued and outstanding and no shares of Preferred Stock issued and outstanding.
COMMON
STOCK
Holders
of Common Stock are entitled to one vote for each share held on all matters submitted to a vote of Shareholders. Directors are appointed
by a plurality of the votes present at any special or annual meeting of Shareholders (by proxy or in person), and a majority of the votes
present at any special or annual meeting of Shareholders (by proxy or in person) shall determine all other matters. The holders of outstanding
shares of Common Stock are entitled to receive dividends out of assets or funds legally available for the payment of dividends at such
times and in such amounts as the Board from time to time may determine. There is no cumulative voting of the election of Directors then
standing for election. The Common Stock is not entitled to pre-emptive rights and is not subject to conversion or redemption. Upon liquidation,
dissolution or winding up of our Company, the assets legally available for distribution to Shareholders are distributable ratably among
the holders of the Common Stock after payment of liquidation preferences, if any, on any outstanding payment of other claims of creditors.
Each outstanding share of Common Stock is, and all shares of Common Stock to be outstanding upon completion of this Offering will be,
duly and validly issued, fully paid and non-assessable.
PREFERRED
STOCK
Shares
of Preferred Stock may be issued from time to time in one or more series, each of which shall have such distinctive designation or title
as shall be determined by our Board prior to the issuance of any shares thereof. Preferred Stock shall have such voting powers, full
or limited, or no voting powers, and such preferences and relative, participating, optional or other special rights and such qualifications,
limitations or restrictions thereof, as shall be stated in such resolution or resolutions providing for the issue of such class or series
of Preferred Stock as may be adopted from time to time by the Board prior to the issuance of any shares thereof. The number of authorized
shares of Preferred Stock may be increased or decreased (but not below the number of shares thereof then outstanding) by the affirmative
vote of the holders of a majority of the voting power of all the then outstanding shares of our capital stock entitled to vote generally
in the election of the Directors, voting together as a single class, without a separate vote of the holders of the Preferred Stock, or
any series thereof, unless a vote of any such holders is required pursuant to any Preferred Stock Designation.
Additionally,
while it is not possible to state the actual effect of the issuance of any shares of Preferred Stock on the rights of holders of the
Common Stock until the Board determines the specific rights of the holders of any shares of Preferred Stock, such rights may be superior
to those associated with our Common Stock, and may include:
Restricting
dividends on the Common Stock;
Rights
and preferences including dividend and dissolution rights, which are superior to our Common Stock;
Diluting
the voting power of the Common Stock;
Impairing
the liquidation rights of the Common Stock; or
Delaying
or preventing a change in control of the Company without further action by the stockholders.
REGISTRATION
RIGHTS
None.
Provisions
of the Company’s Charter or Bylaws which would delay, deter or prevent a change in control of the Company
There
are no special provisions of the Company’s Certificate of Incorporation or Bylaws which would specifically delay, deter or prevent
a change in control of the Company. Additionally, the Company has 50,000,000 shares of Preferred Stock authorized and undesignated. Shares
of Preferred Stock designated by our Board in the future may have voting powers superior to our Common Stock, and such preferences and
relative, participating, optional or other special rights and such qualifications, limitations or restrictions thereof as adopted by
the Board. Such Preferred Stock, if authorized in the future, may contain provisions (including voting rights) which could delay, deter
or prevent a change in control of the Company.
SHARES
REGISTERED FOR RESALE
Overview
We
will not receive any of the proceeds from the sale of shares of the Registered Common Stock by the Selling Stockholders. However, the
Company will receive the proceeds of the sale of the Securities pursuant to the Closing Agreement.
The
Selling Stockholders identified in this prospectus may offer the shares of the Securities from time to time through public or private
transactions at prevailing market prices or at privately negotiated prices. The Selling Stockholders can offer all, some or none of its
shares of the Securities, thus we have no way of determining the number of shares of Securities it will hold after this offering. See
“Plan of Distribution.”
The
Selling Stockholders is an “underwriter” within the meaning of Section 2(a)(11) of the Securities Act of 1933, as amended
(the “Securities Act”).
The
Shares consist of: (i) 11,000,000 Offering Shares issued as an indirect primary offering to the Selling Stockholders pursuant the Closing
Agreement with TRITON FUNDS LP; (ii) 655,905 Warrants issued to the Selling Stockholders pursuant to an Engagement Letter with WallachBeth
Capital, LLC, and (iii) 6,923,077 shares of Common Stock issuable upon the conversion of a $900,000 convertible Note with Walleye Opportunities
Master Fund Ltd., originally issued on May 3, 2021, and amended on May 5, 2023.
The
Selling Stockholders will sell its Shares at prevailing market prices, or at privately negotiated prices. The Selling Stockholder may
elect to exercise its warrant at an exercise price of $0.14 per share or as a cashless exercise, while the selling Note holder may elect
to convert its Note and accrued interest at a conversion rate of $0.13 per share.
SELLING
STOCKHOLDERS
Unless
the context otherwise requires, as used in this Prospectus, “Selling Stockholders” includes the selling stockholders listed
below and donees, pledgees, transferees or other successors-in-interest selling shares received after the date of this Prospectus from
a selling stockholder as a gift, pledge or other non-sale related transfer.
We
have prepared this Prospectus to allow the Selling Stockholders or their successors, assignees or other permitted transferees to sell
or otherwise dispose of, from time to time, up to 18,578,982 shares of our Common Stock, which will be issued pursuant to the terms of
previously issued Agreements, we will issue an aggregate of 11,000,000 shares of Common Stock to TRITON FUNDS LP pursuant the Closing
Agreement. We will issue an aggregate of 655,905 shares of Common Stock by converting the Warrants pursuant to the terms of the Engagement
Letter with WallachBeth Capital, LLC, and the 6,923,077 shares of Common Stock pursuant the terms of the Debt Modification Agreement
with Walleye Opportunities Master Fund Ltd.
The
Warrants were exercisable immediately upon issuance and expire on May 10, 2028. The Warrants are exercisable, at the option of each holder,
in whole or in part by delivering to us a duly executed exercise notice and, at any time a registration statement registering the issuance
of the common shares underlying the Warrants under the Securities Act is effective and available for the issuance of such shares, or
an exemption from registration under the Securities Act is available for the issuance of such shares, by payment in full in immediately
available funds for the number of common shares purchased upon such exercise.
The
Walleye Note will mature on April 30, 2024, and carry an interest of 10%. The underlying shares are currently eligible for resale under
Rule 144. After this period, Selling Stockholders may sell any number of shares of our Common Stock which are issuable upon conversion
of amounts due under the Selling Stockholders Notes. Because the Selling Stockholders may offer all, some or none of the shares it holds,
and because, based upon information provided to us, there are currently no agreements, arrangements or understandings with respect to
the resale of any of the shares, no definitive estimate as to the number of shares that will be held by the Selling Stockholders after
the offering can be provided. Therefore, the following table has been prepared on the assumption that the entire six million nine hundred
twenty-three thousand seventy-seven (6,923,077) shares of Common Stock being registered under this prospectus will be sold by the Selling
Stockholders to parties unaffiliated with them. Because the conversion price of the Walleye Note is the “Conversion Price”
will be the lesser of (i) $.13 per share or (ii) 85% of the of the closing price of any “qualified financing”, which consists
of any fundraising receiving gross proceeds of not less than $500,000, the number of shares of common stock that will actually be issued
upon conversion of the notes may be more or less than the number of shares of common stock being offered by this prospectus.
The
Shares were issued or will be issuable to the Selling Stockholders in reliance on the exemption from securities registration in Section
4(a)(2) under the Securities Act and Rule 506 promulgated thereunder or Regulation S, to the extent applicable.
The
shares of Common Stock to be offered by the Selling Stockholders are “restricted” securities under applicable federal and
state securities laws and are being registered under the Securities Act to give the Selling Stockholders the opportunity to sell these
shares publicly. The registration of these shares does not require that any of the shares be offered or sold by the Selling Stockholders.
Subject to these resale restrictions, the Selling Stockholders may from time to time offer and sell all or a portion of their shares
indicated below in privately negotiated transactions or on the OTCQB Market or any other market on which our Common Stock may subsequently
be listed.
The
registered shares may be sold directly or through brokers or dealers, or in a distribution by one or more underwriters on a firm commitment
or best effort basis. To the extent required, the names of any agent or broker-dealer and applicable commissions or discounts and any
other required information with respect to any particular offering will be set forth in a prospectus supplement. See the section of this
prospectus entitled “Plan of Distribution”. The Selling Stockholders and any agents or broker-dealers that participate with
the Selling Stockholders in the distribution of registered shares may be deemed to be “underwriters” within the meaning of
the Securities Act, and any commissions received by them and any profit on the resale of the registered shares may be deemed to be underwriting
commissions or discounts under the Securities Act.
No
estimate can be given as to the amount or percentage of Common Stock that will be held by the Selling Stockholders after any sales made
pursuant to this prospectus because the Selling Stockholders are not required to sell any of the Shares being registered under this prospectus.
The following table assumes that the Selling Stockholders will sell all of the Shares listed in this prospectus.
Unless
otherwise indicated in the footnotes below, no Selling Stockholder has had any material relationship with us or any of our affiliates
within the past three years other than as a Stockholder.
We
have prepared this table based on written representations and information furnished to us by or on behalf of the Selling Stockholders.
Since the date on which the Selling Stockholders provided this information, the Selling Stockholders may have sold, transferred or otherwise
disposed of all or a portion of the shares of Common Stock in a transaction exempt from the registration requirements of the Securities
Act. Unless otherwise indicated in the footnotes below, we believe that: (1) none of the Selling Stockholders are broker-dealers or affiliates
of broker-dealers, (2) no Selling Stockholder has direct or indirect agreements or understandings with any person to distribute their
Shares, and (3) the Selling Stockholders have sole voting and investment power with respect to all Shares beneficially owned, subject
to applicable community property laws. To the extent any Selling Stockholder identified below is, or is affiliated with, a broker-dealer,
it could be deemed, individually but not severally, to be an “underwriter” within the meaning of the Securities Act. Information
about the Selling Stockholders may change over time. Any changed information will be set forth in supplements to this prospectus, if
required.
The
following table sets forth information with respect to the beneficial ownership of our Common Stock held, as of March 24, 2022, by the
Selling Stockholders and the number of Shares being registered hereby and information with respect to shares to be beneficially owned
by the Selling Stockholders after completion of the offering of the shares for resale. We have determined beneficial ownership in accordance
with the rules of the SEC. The inclusion of any shares in this table does not constitute an admission of beneficial ownership by the
persons named below. The percentages in the following table reflect the shares beneficially owned by the Selling Stockholders as a percentage
of the total number of shares of Common Stock outstanding as of September 18, 2023. As of such date, 143,784,985 shares of Common Stock
were outstanding.
| | |
Shares Beneficially Owned Before this Offering (1) | | |
Maximum
Number
of Shares
of Common
Stock to
be Offered
Pursuant
to this
Prospectus | | |
Shares Beneficially Owned
After this Offering (1)(2) | |
| | |
Number | | |
% | | |
Number | | |
Number | | |
% | |
| Selling Stockholder Name | |
| | | |
| | | |
| | | |
| | | |
| | |
| TRITON FUNDS LP (3) | |
| 1,612,903 | | |
| * | | |
| 11,000,000 | | |
| 12,612,903 | | |
| 8.4 | |
| Douglas Bantum (4) | |
| 608,200 | (5) | |
| * | | |
| 229,905 | | |
| 608,200 | | |
| * | |
| Gene McNeil (4) | |
| 142,575 | (6) | |
| * | | |
| 49,000 | | |
| 142,575 | | |
| * | |
| Kenneth Bantum (4) | |
| 166,800 | (7) | |
| * | | |
| 49,000 | | |
| 166,800 | | |
| * | |
| Michael Wallach (4) | |
| 485,515 | (8) | |
| * | | |
| 164,000 | | |
| 485,515 | | |
| * | |
| David Beth (4) | |
| 485,515 | (9) | |
| * | | |
| 164,000 | | |
| 485,515 | | |
| * | |
| Walleye Opportunities Master Fund Ltd. (10) | |
| 622,000 | (11) | |
| * | | |
| 6,923,077 | | |
| 622,000 | | |
| * | |
| TOTAL | |
| 4,123,508 | | |
| 3.0 | | |
| 18,578,982 | | |
| 5,320,605 | | |
| 10.2 | |
| * |
Denotes
less than 1% |
| (1) |
Beneficial
ownership is determined in accordance with Rule 13d-3 under the Exchange Act. In computing the number of shares beneficially owned
by a person and the percentage ownership of that person, shares of Common Stock subject to the warrants, options and other convertible
securities held by that person that are currently exercisable or exercisable within 60 days (of March 24, 2022) are deemed outstanding.
Shares subject to warrants, options and other convertible securities, however, are not deemed outstanding for the purpose of computing
the percentage ownership of any other person. |
| (2) |
We
do not know when or in what amounts a Selling Stockholder may offer shares for sale. The Selling Stockholders may choose not to sell
any or all of the shares offered by this prospectus. Because the Selling Stockholders may offer all or some of the Shares pursuant
to this offering, we cannot estimate the number of the Shares that will be held by the Selling Stockholders after completion of the
offering. However, for purposes of this table, we have assumed that, after completion of the offering, all of the Shares covered
by this prospectus will be sold by the Selling Stockholders and that the Selling Stockholders do not acquire beneficial ownership
of any additional shares. |
| (3) |
The
securities are directly held by TRITON FUNDS LP, the largest independent student-run fund in United States. |
| (4) |
Douglas
Bantum, Gene McNeil, Kenneth Bantum, Michael Wallach and David Beth are employees of WallachBeth Capital, LLC a
registered broker-dealer with the Financial Industry Regulatory Authority, Inc. and acted as the placement agent for our private
placement offering in May 2023. The Selling Stockholder acquired the securities being registered hereunder in the ordinary course
of business, and at the time of the acquisition of the securities, the Selling Stockholder did not have any arrangements or understandings
with any person to distribute such securities. |
| (5) |
Consists
of (i) 226,000 shares of Common Stock held directly, (ii) 102,200 shares of Common Stock issuable upon the exercise of the Warrants
earned by prior Engagement Agreements, (iii) 229,905 shares of Common Stock issuable upon the exercise of the Warrant to Purchase
Common Stock issued on September 14, 2023 pursuant to the Engagement Letter issued on September 14, 2023. |
| (6) |
Consists
of (i) 60,675 shares of Common Stock held directly, (ii) 60,675 shares of Common Stock issuable upon the exercise of the Warrants
earned by prior Engagement Agreements, (iii) 49,000 shares of Common Stock issuable upon the exercise of the Warrant to Purchase
Common Stock issued on September 14, 2023 pursuant to the Engagement Letter issued on September 14, 2023. |
| (7) |
Consists
of (i) 84,900 shares of Common Stock held directly, (ii) 84,900 shares of Common Stock issuable upon the exercise of the Warrants
earned by prior Engagement Agreements, (iii) 49,000 shares of Common Stock issuable upon the exercise of the Warrant to Purchase
Common Stock issued on September 14, 2023 pursuant to the Engagement Letter issued on September 14, 2023. |
| (8) |
Consists
of (i) 212,500 shares of Common Stock held directly, (ii) 212,500 shares of Common Stock issuable upon the exercise of the Warrants
earned by prior Engagement Agreements, (iii) 164,000 shares of Common Stock issuable upon the exercise of the Warrant to Purchase
Common Stock issued on September 14, 2023 pursuant to the Engagement Letter issued on September 14, 2023. |
| (9) |
Consists
of (i) 212,500 shares of Common Stock held directly, (ii) 212,500 shares of Common Stock issuable upon the exercise of the Warrants
earned by prior Engagement Agreements, (iii) 164,000 shares of Common Stock issuable upon the exercise of the Warrant to Purchase
Common Stock issued on September 14, 2023 pursuant to the Engagement Letter issued on September 14, 2023. |
| (10) |
The
securities are directly held by Walleye Opportunities Master Fund Ltd managed by Walleye Capital, a multi-strategy platform hedge
fund headquartered in Minnesota. The firm manages approximately $5B of investor capital. |
| (11) |
Consists
of (i) 622,000 shares of Common Stock held directly issued on August 30, 2023 pursuant a partial conversion
of the Walleye Note. |
To
our knowledge, neither the Selling Stockholder nor its beneficial owners have ever been one of our officers or directors or an officer
or director of our predecessors or affiliates.
PLAN
OF DISTRIBUTION
We
are registering the shares of Common Stock and shares of Common Stock issuable upon exercise of the Warrants previously issued to the
Selling Stockholders to permit the resale of these shares of Common Stock by the holders of the Shares and the Warrants from time to
time after the date of this Prospectus. We will not receive any of the proceeds from the sale by the Selling Stockholders of the shares
of Common Stock. We will bear all fees and expenses incident to our obligation to register the shares of Common Stock.
The
Selling Stockholder and any of their pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their
securities covered hereby via OTCQB or any other stock exchange, market or trading facility on which the securities are traded or in
private transactions. These sales may be at fixed or negotiated prices. The Selling Stockholder may use any one or more of the following
methods when selling securities:
| |
● |
ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| |
|
|
| |
● |
block
trades in which the broker-dealer will attempt to sell the securities as agent but may position and resell a portion of the block
as principal to facilitate the transaction; |
| |
|
|
| |
● |
purchases
by a broker-dealer as principal and resale by the broker-dealer for its account; |
| |
|
|
| |
● |
an
exchange distribution in accordance with the rules of the applicable exchange; |
| |
|
|
| |
● |
privately
negotiated transactions; |
| |
|
|
| |
● |
settlement
of short sales entered into after the effective date of the registration statement of which this Prospectus is a part; |
| |
|
|
| |
● |
in
transactions through broker-dealers that agree with the Selling Shareholders to sell a specified number of such securities at a stipulated
price per security; |
| |
|
|
| |
● |
through
the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise; |
| |
|
|
| |
● |
a
combination of any such methods of sale; or |
| |
|
|
| |
● |
any
other method permitted pursuant to applicable law. |
The
Selling Stockholder may also sell securities under Rule 144 under the Securities Act of 1933, as amended (the “Securities Act”),
if available, rather than under this Prospectus.
Broker-dealers
engaged by the Selling Stockholder may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions
or discounts from the Selling Shareholders (or, if any broker-dealer acts as agent for the purchaser of securities, from the purchaser)
in amounts to be negotiated, but except as set forth in a supplement to this Prospectus, in the case of an agency transaction not in
excess of a customary brokerage commission in compliance with FINRA Rule 2440; and in the case of a principal transaction a markup or
markdown in compliance with FINRA IM-2440.
In
connection with the sale of the securities or interests therein, the Selling Stockholder may enter into hedging transactions with broker-dealers
or other financial institutions, which may in turn engage in short sales of the securities in the course of hedging the positions they
assume. The Selling Stockholder may also sell securities short and deliver these securities to close out its short positions, or loan
or pledge the securities to broker-dealers that in turn may sell these securities. The Selling Stockholder may also enter into option
or other transactions with broker-dealers or other financial institutions or create one or more derivative securities which require the
delivery to such broker-dealer or other financial institution of securities offered by this Prospectus, which securities such broker-dealer
or other financial institution may resell pursuant to this Prospectus (as supplemented or amended to reflect such transaction).
The
Selling Stockholder and any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters”
within the meaning of the Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers
or agents and any profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts
under the Securities Act. The Selling Stockholder have informed the Company that it does not have any written or oral agreement or understanding,
directly or indirectly, with any person to distribute the securities. In no event shall any broker-dealer receive fees, commissions and
markups which, in the aggregate, would exceed eight percent (8%).
The
Company is required to pay certain fees and expenses incurred by the Company incident to the registration of the securities. The Company
has agreed to indemnify the Selling Shareholders against certain losses, claims, damages and liabilities, including liabilities under
the Securities Act.
Because
Selling Stockholder may be deemed to be “underwriters” within the meaning of the Securities Act, they will be subject to
the Prospectus delivery requirements of the Securities Act including Rule 172 thereunder. In addition, any securities covered by this
Prospectus which qualify for sale pursuant to Rule 144 under the Securities Act may be sold under Rule 144 rather than under this Prospectus.
The Selling Stockholder have advised us that there is no underwriter or coordinating broker acting in connection with the proposed sale
of the resale securities by the Selling Stockholder.
We
agreed to keep this Prospectus effective until the earlier of (i) the date on which the securities may be resold by the Selling Stockholder
without registration and without regard to any volume or manner-of-sale limitations by reason of Rule 144, or (ii) all of the securities
have been sold pursuant to this Prospectus or Rule 144 under the Securities Act or any other rule of similar effect. The resale securities
will be sold only through registered or licensed brokers or dealers if required under applicable state securities laws. In addition,
in certain states, the resale securities covered hereby may not be sold unless they have been registered or qualified for sale in the
applicable state or an exemption from the registration or qualification requirement is available and is complied with.
Under
applicable rules and regulations under the Exchange Act, any person engaged in the distribution of the resale securities may not simultaneously
engage in market making activities with respect to the Common Stock for the applicable restricted period, as defined in Regulation M,
prior to the commencement of the distribution. In addition, the Selling Stockholders will be subject to applicable provisions of the
Exchange Act and the rules and regulations thereunder, including Regulation M, which may limit the timing of purchases and sales of securities
of the Common Stock by the Selling Shareholders or any other person. We will make copies of this Prospectus available to the Selling
Stockholders and have informed them of the need to deliver a copy of this Prospectus to each purchaser at or prior to the time of the
sale (including by compliance with Rule 172 under the Securities Act).
MARKET
FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
On
March 30, 2022, the Company received the FINRA Clearance Letter and on June 14, 2022, OTC Market Group agreed to the removal of the Caveat
Emptor symbol. The Company was approved for quotation on the OTCQB on September 28, 2022. We cannot assure you that an active public
market for our Common Stock will develop or that the market price of our shares will not decline below the Proposed Maximum Offering
Price. The Proposed Maximum Offering Price of our shares may not be indicative of prices that will prevail in the trading market following
the offering.
The
following tables set forth the range of high and low bid prices for our Common Stock for the each of the periods indicated as reported
by the OTCQB. These quotations reflect inter-dealer prices, without retail mark-up, mark-down or commission and may not necessarily represent
actual transactions.
| Quarter Ended | |
High | | |
Low | |
| June 30, 2023 | |
$ | 0.51 | | |
$ | 0.16 | |
| March 31, 2023 | |
| 0.53 | | |
| 0.43 | |
| Quarter Ended | |
High | | |
Low | |
| December 31, 2022 | |
$ | 0.60 | | |
$ | 0.36 | |
| September 30, 2022 | |
| 1.25 | | |
| 0.28 | |
| June 30, 2022 | |
| 0.50 | | |
| 0.15 | |
| March 31, 2022 | |
$ | 0.67 | | |
$ | 0.10 | |
| Quarter Ended | |
High | | |
Low | |
| December 31, 2021 | |
$ | 0.40 | | |
$ | 0.001 | |
| September 30, 2021 | |
| 0.01 | | |
| 0.0002 | |
| June 30, 2021 | |
| 0.18 | | |
| 0.0002 | |
| March 31, 2021 | |
$ | 0.24 | | |
$ | 0.01 | |
| Quarter Ended | |
| High | | |
| Low | |
| December 31, 2020 | |
$ | 0.29 | | |
$ | 0.03 | |
| September 30, 2020 | |
| 0.21 | | |
| 0.003 | |
On
September 18, 2023, the last reported sale price of our Common Stock as reported on the OTCQB was $0.146 per share.
Our
common shares are issued in registered form. The registrar and transfer agent for our shares is:
Securities
Transfer Corporation
2901
N. Dallas Parkway
Suite
380
Plano,
TX 75093
+1
469-633-0101
www.stctransfer.com
Penny
Stock
The
SEC has adopted rules that regulate broker-dealer practices in connection with transactions in penny stocks. Penny stocks are generally
equity securities with a market price of less than $5.00/share, other than securities registered on certain national securities exchanges
or quoted on the NASDAQ system, provided that current price and volume information with respect to transactions in such securities is
provided by the exchange or system. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock, to deliver
a standardized risk disclosure document prepared by the SEC, that: (a) contains a description of the nature and level of risk in the
market for penny stocks in both public offerings and secondary trading; (b) contains a description of the broker’s or dealer’s
duties to the customer and of the rights and remedies available to the customer with respect to a violation of such duties or other requirements
of the securities laws; (c) contains a brief, clear, narrative description of a dealer market, including bid and ask prices for penny
stocks and the significance of the spread between the bid and ask price; (d) contains a toll-free telephone number for inquiries on disciplinary
actions; (e) defines significant terms in the disclosure document or in the conduct of trading in penny stocks; and (f) contains such
other information and is in such form, including language, type size and format, as the SEC shall require by rule or regulation.
The
broker-dealer also must provide, prior to effecting any transaction in a penny stock, the customer with (a) bid and offer quotations
for the penny stock; (b) the compensation of the broker-dealer and its salesperson in the transaction; (c) the number of shares to which
such bid and ask prices apply, or other comparable information relating to the depth and liquidity of the market for such stock; and
(d) a monthly account statement showing the market value of each penny stock held in the customer’s account.
In
addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from those rules, the broker-dealer
must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s
written acknowledgment of the receipt of a risk disclosure statement, a written agreement as to transactions involving penny stocks,
and a signed and dated copy of a written suitability statement.
These
disclosure requirements may have the effect of reducing the trading activity for our Common Stock. Therefore, stockholders may have difficulty
selling our securities.
Holders
of Common Stock
As
of the date of this Prospectus, we have approximately four hundred thirty (430) holders of record of Common Stock administered by our
transfer agent, and approximately one thousand three hundred ten (1,310) holders of shares
administered by brokers and retained in custody of by the Depository Trust Company (“DTC”), totaling an estimated one thousand
seven hundred forty (1,740) holders of Common Stock. Currently, twenty-eight million six hundred thirty-six thousand two hundred
forty-five (28,636,245) (19.9%) of outstanding shares are free-trading.
Our
primary Stockholders are Dr. David Platt, Ola Soderquist and Mike Sheikh, who beneficially own 59,532,708; 19,535,300; and 8,000,000
shares, respectively, of our Common Stock, or an aggregate of eighty-seven million sixty-eight thousand eight (87,068,008) (60.6%) outstanding
shares.
Dividends
There
have been no cash dividends declared on our Common Stock since our Company was formed. Dividends are declared at the sole discretion
of our Board. Our intention is not to declare cash dividends and retain all cash for our operations.
ADDITIONAL
INFORMATION
Our
fiscal year ends on December 31. We furnish our Shareholders annual reports containing audited financial statements and other appropriate
reports, where applicable. In addition, we are a reporting company and file annual, quarterly, and current reports, and other information
with the SEC, where applicable. You may read and copy any reports, statements, or other information we file at the SEC’s public
reference room at 100 F. Street, N.E., Washington D.C. 20549. You can request copies of these documents, upon payment of a duplicating
fee by writing to the SEC. Please call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference rooms.
Our SEC filings are also available to the public on the SEC’s Internet site at httpwww.sec.gov.
INDEMNIFICATION
OF DIRECTORS AND OFFICERS
Indemnification.
Our Directors and officers are indemnified to the fullest extent permitted under Nevada law.
Insurance.
The Company may purchase and maintain insurance on behalf of any person who is or was a Director, officer or employee of the Company,
or is or was serving at the request of the Company as a Director, officer, employee or agent of another company, partnership, joint venture,
trust or other enterprise against liability asserted against him and incurred by him in any such capacity, or arising out of his status
as such, whether or not the Company would have the power to indemnify him against liability under the provisions of this section. The
Company currently not maintain such insurance.
Settlement
by the Company. The right of any person to be indemnified is subject always to the right of the Company by its Board of Directors,
in lieu of such indemnity, to settle any such claim, action, suit or proceeding at the expense of the Company by the payment of the amount
of such settlement and the costs and expenses incurred in connection therewith.
DISCLOSURE
OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES.
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to our Directors, officers and controlling
persons pursuant to the following provisions, or otherwise, we have been advised that in the opinion of the SEC such indemnification
is against public policy as expressed in the Act and is, therefore, unenforceable. In the event that a claim for indemnification
against such liabilities (other than the payment by us of expenses incurred or paid by a Director, officer or controlling person in the
successful defense of any action, suit or proceeding) is asserted by such Director, officer or controlling person in connection with
the shares being registered, we will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit
to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Act
and will be governed by the final adjudication of such issue.
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to Directors, officers or persons controlling
the Company pursuant to the foregoing provisions, or otherwise, the Company has been advised that in the opinion of the SEC, such indemnification
is against public policy as expressed in the Securities Act of 1933 and is, therefore, unenforceable.
In
the event that a claim for indemnification against such liabilities (other than the payment of expenses incurred or paid by a Director,
officer or controlling person in a successful defense of any action, suit or proceeding) is asserted by such Director, officer or controlling
person in connection with the securities being registered, we will, unless in the opinion of its counsel the matter has been settled
by controlling precedent, submit to the court of appropriate jurisdiction the question whether such indemnification by it is against
public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
At
present, there is no other pending litigation or proceeding involving any of our Directors, officers or employees as to which indemnification
is sought, nor are we aware of any threatened litigation or proceeding that may result in claims for indemnification.
LEGAL
MATTERS
Certain
legal matters with respect to the issuance of shares of Common Stock offered hereby will be passed upon by Robert J. Burnett, Witherspoon
Brajcich McPhee, PLLC, Spokane, WA
EXPERTS
The
financial statements of the Company as of and for the two years ended December 31, 2022 appearing in this Prospectus and Registration
Statement have been audited by BF Borgers CPA PC while 2021 was audited by Pinnacle Accountancy
Group of Utah (a dba of the PCAOB-registered firm Heaton & Company, PLLC), Farmington, Utah, independent registered public accounting
firms, as stated in their report appearing elsewhere herein, (which report expresses an unqualified opinion and includes an explanatory
paragraph expressing substantial doubt regarding the Company’s ability to continue as a going concern) and are included in reliance
upon such report and upon the authority of such firms as experts in accounting and auditing.
INTERESTS
OF NAMED EXPERTS AND COUNSEL
No
expert or counsel named in this Prospectus as having prepared or certified any part of this Prospectus or having given an opinion upon
the validity of the securities being registered or upon other legal matters in connection with the registration or offering of the Common
Stock was employed on a contingency basis, or had, or is to receive, in connection with the offering, a substantial interest, direct
or indirect, in the registrant or any of its parents or subsidiaries. Nor was any such person connected with the registrant or any of
its parents or subsidiaries as a promoter, managing or principal underwriter, voting trustee, director, officer, or employee.
FINANCIAL
STATEMENTS
The
Financial Statements required by Article 8 of Regulation S-X are stated in U.S. dollars and are prepared in accordance with Accounting
Principles Generally Accepted in the United States of America (“US GAAP”). The following financial statements pertaining
to Bioxytran, Inc. are filed as part of this Prospectus.
BIOXYTRAN,
INC.
FINANCIAL STATEMENTS
FOR
THE YEARS ENDED DECEMBER 31, 2021 AND DECEMBER 31, 2020
TABLE
OF CONTENTS
| |
|
Page |
| |
Report of Independent Registered Public Accounting Firm (FIRM ID 6117) |
F-2 |
| |
|
|
| Financial
Statements |
|
| |
|
|
| |
Balance Sheets for the years ended December 31, 2022 and December 31, 2021 |
F-5 |
| |
|
|
| |
Statements of Operations for the years ended December 31, 2022 and December 31, 2021 |
F-6 |
| |
|
|
| |
Statement of Changes in Stockholders’ Deficit for the years ended December 31, 2022 and December 31, 2021 |
F-7 |
| |
|
|
| |
Statement of Cash Flows for the years ended December 31, 2022 and December 31, 2021 |
F-8 |
| |
|
|
| |
Notes to Financial Statements for the years ended December 31, 2022 and December 31, 2021 |
F-9
– F-21 |
| |
|
|
| Unaudited
Condensed Consolidated Financial Statements for the three and six months ended June
30, 2023 and 2022 |
|
| |
|
|
| |
Balance Sheets as of June 30, 2023 and December 31, 2022 (Unaudited) |
F-22 |
| |
|
|
| |
Statements of Operations for the three and six months ended June 30, 2023 and 2022 (Unaudited) |
F-23 |
| |
|
|
| |
Statements of Changes in Stockholders’ Deficit for the three and six months ended June 30, 2023 and 2022 (Unaudited) |
F-24 |
| |
|
|
| |
Statements of Cash Flows for the three and six months ended June 30, 2023 and 2022 (Unaudited) |
F-25 |
| |
|
|
| |
Notes to Unaudited Condensed Consolidated Financial Statements |
F-26
– F-36 |
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To
the Shareholders and the Board of Directors of Bioxytran, Inc.
Opinion
on the Financial Statements
We
have audited the accompanying consolidated balance sheet of Bioxytran, Inc. (the “Company”) as of December 31, 2022, the
related statement of operations, stockholders’ equity (deficit), and cash flows for the year then ended, and the related notes
(collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all
material respects, the financial position of the Company as of December 31, 2022, and the results of its operations and its cash flows
for the year then ended, in conformity with accounting principles generally accepted in the United States.
Substantial
Doubt about the Company’s Ability to Continue as a Going Concern
The
accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note
3 to the financial statements, the Company’s significant operating losses raise substantial doubt about its ability to continue
as a going concern. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis
for Opinion
These
financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s
financial statements based on our audit. We are a public accounting firm registered with the Public Company Accounting Oversight Board
(United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal
securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We
conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company
is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits,
we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion
on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our
audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or
fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding
the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant
estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides
a reasonable basis for our opinion.
/s/
BF Borgers CPA PC
BF
Borgers CPA PC (PCAOB ID 5041)
We
have served as the Company’s auditor since 2023
Lakewood, CO
March
31, 2023
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To
the Board of Directors and Stockholders
Bioxytran,
Inc.
Needham,
Massachusetts
Opinion
on the Financial Statements
We
have audited the accompanying consolidated balance sheet of Bioxytran, Inc. (the Company) as of December 31, 2021, and the related consolidated
statements of operations, changes in stockholders’ equity (deficit), and cash flows for the year then ended, and the related notes
(collectively referred to as the consolidated financial statements). In our opinion, the consolidated financial statements present fairly,
in all material respects, the financial position of the Company as of December 31, 2021, and the results of its operations and its cash
flows for the year then ended, in conformity with accounting principles generally accepted in the United States of America.
Going
Concern Considerations
The
accompanying financial statements have been prepared assuming that the Company will continue as a going concern. The Company has negative
working capital, has suffered losses since inception and has not achieved profitable operations, which raise substantial doubt about
its ability to continue as a going concern. Management’s plans in regard to these matters are described in Note 3. The financial
statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis
for Opinion
These
financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s
financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board
(United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities
laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We
conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company
is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audit,
we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion
on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our
audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or
fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding
the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant
estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides
a reasonable basis for our opinion.
Critical
Audit Matter
The
critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated
or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the financial
statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters
does not alter in any way our opinion on the financial statements, taken as a whole, and we are not, by communicating the critical audit
matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
Going
Concern – Disclosure
The
financial statements of the Company are prepared on a going concern basis, which assumes that the Company will continue in operation
for the foreseeable future and, accordingly, will be able to realize its assets and discharge its liabilities in the normal course of
operations. As noted in “Going Concern Considerations” above, the Company has a history of net losses, negative working capital,
a significant accumulated deficit and currently has net working capital deficit. The Company has contractual obligations, such as commitments
for repayments of accounts payable, accrued liabilities, loans payable, notes payable, and related party loans (collectively “obligations”).
Currently, management’s forecasts and related assumptions illustrate their ability to meet the obligations through management of
expenditures, implementation of planned business operations, obtaining additional debt financing, and issuance of capital stock for additional
funding to meet its operating needs. Should there be constraints on the ability to implement its planned business operations or access
financing through stock issuances, the Company will continue to manage cash outflows and meet the obligations through debt financing.
We
identified management’s assessment of the Company’s ability to continue as a going concern as a critical audit matter. Management
made judgments to conclude that it is probable that the Company’s plans will be effectively implemented and will provide the necessary
cash flows to fund the Company’s obligations as they become due. Specifically, the judgments with the highest degree of impact
and subjectivity in determining it is probable that the Company’s plans will be effectively implemented include its ability to
manage expenditures, its ability to access funding from the capital market, its ability to obtain debt financing, and the successful
implementation of its planned business operations. Auditing the judgments made by management required a high degree of auditor judgment
and an increased extent of audit effort.
Addressing
the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the financial
statements. These procedures included the following, among others: (i) evaluating the probability that the Company will be able to access
funding from the capital market; (ii) evaluating the probability that the Company will be able to manage expenditures (iii) evaluating
the probability that the Company will be able to obtain debt financing, and (iv) evaluating the implementation of its planned business
operations.
Stock-Based
Compensation
As
described in Note 8 and 9 to the consolidated financial statements, the Company recorded stock-based compensation related to the issuance
of Common Stock, stock options and warrants. Management establishes their estimates for the value of the stock-based compensation related
to Common Stock issued for services using historical stock price information. Management uses a valuation model requiring various inputs
to establish their estimates for the value of stock options and warrants.
The
principal considerations for our determination that performing procedures relating to stock-based compensation is a critical audit matter
are due to the material impact it has on the consolidated financial statements.
Addressing
the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated
financial statements. These procedures included, among others, evaluating the reasonableness of the historical stock price information
used by management for the valuation of the Common Stock along with evaluating the reasonableness of the input’s management used
in the valuation model related to the stock options and warrants to determine the stock-based compensation expense.
/s/
Pinnacle Accountancy Group of Utah
We
have served as the Company’s auditor since 2018.
(PCAOB
ID 6117)
Pinnacle
Accountancy Group of Utah
(dba of Heaton & Company, PLLC)
Farmington,
Utah
April 11, 2022
BIOXYTRAN,
INC.
CONSOLIDATED
BALANCE SHEETS
DECEMBER
31, 2022 AND DECEMBER 31, 2021
| | |
December 31, 2022 | | |
December 31, 2021 | |
| ASSETS | |
| | | |
| | |
| Current assets: | |
| | | |
| | |
| Cash | |
$ | 295,401 | | |
$ | 72,358 | |
| Total current assets | |
| 295,401 | | |
| 72,358 | |
| | |
| | | |
| | |
| Intangibles, net | |
| 75,535 | | |
| 46,932 | |
| | |
| | | |
| | |
| Total assets | |
$ | 370,936 | | |
$ | 119,290 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable and accrued expenses | |
$ | 749,395 | | |
$ | 624,316 | |
| Accounts payable related party | |
| 709,727 | | |
| 531,000 | |
| Un-issued shares liability | |
| 960 | | |
| — | |
| Un-issued shares liability related party | |
| 38,400 | | |
| — | |
| Convertible notes payable, net of premium and discount | |
| 2,165,000 | | |
| 2,122,181 | |
| Total current liabilities | |
| 3,663,482 | | |
| 3,277,497 | |
| | |
| | | |
| | |
| Total liabilities | |
| 3,663,482 | | |
| 3,277,497 | |
| | |
| | | |
| | |
| Commitments and contingencies | |
| - | | |
| - | |
| | |
| | | |
| | |
| Stockholders’ deficit: | |
| | | |
| | |
| Preferred stock, $0.001 par value; 50,000,000 shares authorized, nil issued and outstanding | |
| — | | |
| — | |
| Common stock, $0.001 par value; 300,000,000 shares authorized; 123,252,235 and 110,840,998 issued and outstanding as at December 31, 2022 and 2021, respectively | |
| 123,252 | | |
| 110,841 | |
| Additional paid-in capital | |
| 8,392,430 | | |
| 5,881,876 | |
| Non-controlling interest | |
| (590,628 | ) | |
| (397,256 | ) |
| Accumulated deficit | |
| (11,217,600 | ) | |
| (8,753,668 | ) |
| Total stockholders’ deficit | |
| (3,292,546 | ) | |
| (3,158,207 | ) |
| | |
| | | |
| | |
| Total liabilities and stockholders’ deficit | |
$ | 370,936 | | |
$ | 119,290 | |
See
the accompanying notes to these consolidated financial statements
BIOXYTRAN,
INC.
CONSOLIDATED
STATEMENTS OF OPERATIONS
FOR
THE YEARS ENDED DECEMBER 31, 2022 AND DECEMBER 31, 2021
| | |
December 31, 2022 | | |
December 31, 2021 | |
| | |
Year ended | |
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Operating expenses: | |
| | | |
| | |
| Research and development | |
$ | 977,768 | | |
$ | 2,013,762 | |
| General and administrative | |
| 933,742 | | |
| 1,612,685 | |
| General and administrative related party | |
| 44,220 | | |
| 5,125 | |
| Compensation expense | |
| 178,382 | | |
| 582,862 | |
| Total operating expenses | |
| 2,134,112 | | |
| 4,214,434 | |
| | |
| | | |
| | |
| Loss from operations | |
| (2,134,112 | ) | |
| (4,214,434 | ) |
| | |
| | | |
| | |
| Other expenses: | |
| | | |
| | |
| Interest expense | |
| (207,117 | ) | |
| (236,577 | ) |
| Amortization of Intellectual Property | |
| (3,644 | ) | |
| — | |
| Debt discount amortization | |
| (312,431 | ) | |
| (77,031 | ) |
| Debt discount amortization and issuance of warrants | |
| | | |
| | |
| Total other expenses | |
| (523,192 | ) | |
| (313,608 | ) |
| Total other income (expenses) | |
| (523,192 ) | | |
| (313,608 | |
| | |
| | | |
| | |
| Net loss before provision for income taxes | |
| (2,657,304 | ) | |
| (4,528,042 | ) |
| | |
| | | |
| | |
| Provision for income taxes | |
| — | | |
| — | |
| Net loss | |
| (2,657,304 | ) | |
| (4,528,042 | ) |
| | |
| | | |
| | |
| Net loss attributable to the non-controlling interest | |
| 193,372 | | |
| 496,297 | |
| | |
| | | |
| | |
| NET LOSS ATTRIBUTABLE TO BIOXYTRAN | |
$ | (2,463,932 | ) | |
$ | (4,031,745 | ) |
| | |
| | | |
| | |
| Loss per common share, basic and diluted | |
$ | (0.02 | ) | |
$ | (0.04 | ) |
| | |
| | | |
| | |
| Weighted average number of common shares outstanding, basic and diluted | |
| 115,139,380 | | |
| 106,252,116 | |
See
the accompanying notes to these consolidated financial statements
BIOXYTRAN,
INC.
CONSOLIDATED
STATEMENT OF CHANGES IN STOCKHOLDERS’ DEFICIT
FOR
THE YEAR ENDED DECEMBER 31, 2022 AND DECEMBER 31, 2021
| | |
| Shares | | |
| Amount | | |
| Shares | | |
| Amount | | |
| Common | | |
| Preferred | | |
| Deficit | | |
| Interest | | |
| (Deficit) | |
| | |
| Common
Stock | | |
| Preferred
Stock | | |
| Additional
Paid in
Capital | | |
| Accumulated | | |
| Non- controlling | | |
| Total Shareholder’s Equity | |
| | |
| Shares | | |
| Amount | | |
| Shares | | |
| Amount | | |
| Common | | |
| Preferred | | |
| Deficit | | |
| Interest | | |
| (Deficit) | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| January
1, 2021 | |
| 97,450,673 | | |
$ | 97,451 | | |
| — | | |
| — | | |
$ | 1,795,125 | | |
$ | — | | |
$ | (4,721,923 | ) | |
$ | 888,091 | | |
$ | (1,941,256 | ) |
| Options issued and vested –
2010/2021 Plan | |
| | | |
| | | |
| | | |
| | | |
| 14,490 | | |
| | | |
| | | |
| | | |
| 14,490 | |
| Options issued and vested | |
| | | |
| | | |
| | | |
| | | |
| 14,490 | | |
| | | |
| | | |
| | | |
| 14,490 | |
| Net of Shares issued to BoD,
Mgmnt & related party – 2010/2021 Plan | |
| 4,811,309 | | |
| 4,811 | | |
| - | | |
| - | | |
| 143,259 | | |
| - | | |
| | | |
| | | |
| 148,070 | |
| Shares issued to Consultants
– 2010/2021 Plan | |
| 2,893,600 | | |
| 2,893 | | |
| | | |
| | | |
| 406,459 | | |
| | | |
| | | |
| | | |
| 409,352 | |
| Common Stock issued for conversion
of convertible notes and accrued interest | |
| 930,864 | | |
| 931 | | |
| | | |
| | | |
| 120,111 | | |
| | | |
| | | |
| | | |
| 121,042 | |
| Forgiveness of debt by Mgmnt
and related party | |
| | | |
| | | |
| | | |
| | | |
| 2,007,187 | | |
| | | |
| | | |
| | | |
| 2,007,187 | |
| Conversion of subsidiary shares | |
| 4,754,552 | | |
| 4,755 | | |
| | | |
| | | |
| 1,395,245 | | |
| | | |
| | | |
| (1,400,000 | ) | |
| — | |
| Subsidiary shares acquired
by affiliate | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| 10,500 | | |
| 10,500 | |
| Subsidiary stock options | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| 450 | | |
| 450 | |
| Subsidiary stock transactions | |
| - | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| 600,000 | | |
| 600,000 | |
| Subsidiary stock transactions | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| 600,000 | | |
| 600,000 | |
| Net loss attributable to the
non-controlling interest | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (496,297 | ) | |
| (496,297 | ) |
| Net
loss | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (4,031,745 | ) | |
| | | |
| (4,031,745 | ) |
| December
31, 2021 | |
| 110,840,998 | | |
$ | 110,841 | | |
| — | | |
| — | | |
$ | 5,881,876 | | |
$ | — | | |
$ | (8,753,668 | ) | |
$ | (397,256 | ) | |
$ | (3,158,207 | ) |
| Balance | |
| 110,840,998 | | |
$ | 110,841 | | |
| — | | |
| — | | |
$ | 5,881,876 | | |
$ | — | | |
$ | (8,753,668 | ) | |
$ | (397,256 | ) | |
$ | (3,158,207 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net of Shares issued to BoD,
Mgmnt & related party – 2021 Plan | |
| 280,000 | | |
| 280 | | |
| - | | |
| - | | |
| 45,560 | | |
| - | | |
| | | |
| | | |
| 45,840 | |
| Net of Shares issued to BoD,
Mgmnt & related party | |
| 280,000 | | |
| 280 | | |
| | | |
| | | |
| 45,560 | | |
| | | |
| | | |
| | | |
| 45,840 | |
| Shares issued to Consultants
– 2021 Plan | |
| 354,000 | | |
| 354 | | |
| | | |
| | | |
| 92,828 | | |
| | | |
| | | |
| | | |
| 93,182 | |
| Shares issued to Consultants | |
| 354,000 | | |
| 354 | | |
| | | |
| | | |
| 92,828 | | |
| | | |
| | | |
| | | |
| 93,182 | |
| Common Stock issued for conversion
of convertible notes and accrued interest | |
| 6,081,484 | | |
| 6,081 | | |
| | | |
| | | |
| 1,514,290 | | |
| | | |
| | | |
| | | |
| 1,520,371 | |
| Issuance of warrants | |
| | | |
| | | |
| | | |
| | | |
| 190,335 | | |
| | | |
| | | |
| | | |
| 190,335 | |
| Forfeiture of warrants | |
| | | |
| | | |
| | | |
| | | |
| (6,763 | ) | |
| | | |
| | | |
| | | |
| (6,763 | ) |
| Conversion of warrants | |
| 4,139,503 | | |
| 4,140 | | |
| | | |
| | | |
| (4,140 | ) | |
| | | |
| | | |
| | | |
| — | |
| Stock transactions | |
| 1,556,250 | | |
| 1,556 | | |
| | | |
| | | |
| 648,444 | | |
| | | |
| | | |
| | | |
| 650,000 | |
| Stock subscription | |
| | | |
| | | |
| | | |
| | | |
| 30,000 | | |
| | | |
| | | |
| | | |
| 30,000 | |
| Net loss attributable to the
non-controlling interest | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (193,372 | ) | |
| (193,372 | ) |
| Net
loss | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (2,463,932 | ) | |
| | | |
| (2,463,932 | ) |
| December
31, 2022 | |
| 123,252,235 | | |
$ | 123,252 | | |
| — | | |
| — | | |
$ | 8,392,430 | | |
$ | — | | |
$ | (11,217,600 | ) | |
$ | (590,628 | ) | |
$ | (3,292,546 | ) |
| Balance | |
| 123,252,235 | | |
$ | 123,252 | | |
| — | | |
| — | | |
$ | 8,392,430 | | |
$ | — | | |
$ | (11,217,600 | ) | |
$ | (590,628 | ) | |
$ | (3,292,546 | ) |
See
the accompanying notes to these consolidated financial statements
BIOXYTRAN,
INC.
CONSOLIDATED
STATEMENT OF CASH FLOWS
FOR
THE YEAR ENDED DECEMBER 31, 2022 AND DECEMBER 31, 2021
| | |
December 31, 2022 | | |
December
31, 2021 | |
| | |
Year Ended | |
| | |
December 31, 2022 | | |
December
31, 2021 | |
| CASH FLOWS FROM OPERATING ACTIVITIES: | |
| | | |
| | |
| Net loss | |
$ | (2,657,304 | ) | |
$ | (4,528,042 | ) |
| Adjustments to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Debt discount amortization, incl. issuance of warrants | |
| | | |
| | |
| Amortization of debt discount, incl. issuance of warrants | |
| 312,431 | | |
| 77,031 | |
| Amortization of Intellectual Property | |
| 3,644 | | |
| — | |
| Stock-based compensation expense | |
| 178,382 | | |
| 582,862 | |
| Interest paid for conversion of note | |
| 53,371 | | |
| | |
| Changes in operating assets and liabilities: | |
| | | |
| | |
| Pre-paid expenses | |
| — | | |
| 274,715 | |
| Accounts payable and accrued expenses | |
| 125,079 | | |
| 1,206,088 | |
| Accounts payable related party | |
| 178,727 | | |
| 689,947 | |
| Net cash used in operating activities | |
| (1,805,670 | ) | |
| (1,697,399 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM INVESTING ACTIVITIES: | |
| | | |
| | |
| Investment in intangibles | |
| (32,247 | ) | |
| (36,931 | ) |
| Net cash used in investing activities | |
| (32,247 | ) | |
| (36,931 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | |
| | | |
| | |
| Proceeds from subsidiary stock transactions | |
| — | | |
| 600,000 | |
| Proceeds from issuance of convertible notes payable | |
| 1,380,960 | | |
| 1,165,000 | |
| Proceeds from stock transactions | |
| 680,000 | | |
| — | |
| Net cash provided by financing activities | |
| 2,060,960 | | |
| 1,765,000 | |
| | |
| | | |
| | |
| Net increase in cash | |
| 223,043 | | |
| 30,670 | |
| Cash, beginning of period | |
| 72,358 | | |
| 41,688 | |
| Cash, end of period | |
$ | 295,401 | | |
$ | 72,358 | |
| | |
| | | |
| | |
| SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION: | |
| | | |
| | |
| Interest paid | |
$ | 104,850 | | |
$ | — | |
| Income taxes paid | |
| — | | |
| — | |
| NON-CASH INVESTING & FINANCING ACTIVITIES: | |
| | | |
| | |
| Issuance of warrants | |
| 190,335 | | |
| — | |
| Forfeiture of warrants | |
| (6,763 | ) | |
| — | |
| Debt discount on convertible note | |
| 128,859 | | |
| 119,850 | |
| Common shares issued for the conversion of subsidiary shares, related party | |
| — | | |
| 1,400,000 | |
| Common shares issued for the conversion of convertible notes and accrued interest | |
| 1,520,371 | | |
| 121,042 | |
| Forgiveness of related party debt recorded to additional paid-in capital | |
$ | — | | |
$ | 2,007,187 | |
| Common shares issued for the conversion of principal and accrued interest | |
| | | |
| | |
See
the accompanying notes to these consolidated financial statements
BIOXYTRAN,
INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AS
AT DECEMBER 31, 2022 AND DECEMBER 31, 2021
NOTE
1 – BACKGROUND AND ORGANIZATION
Business
Operations
Bioxytran,
Inc. (the “Company”) is a clinical-stage pharmaceutical company focused on the development, manufacture and commercialization
of therapeutic drugs designed to address hypoxia in humans, which is a lack of oxygen to tissues, in a safe and efficient manner. If
it is not addressed, lack of oxygen to tissues, or hypoxia, results in necrosis, which is the death of cells comprising body tissue.
Necrosis cannot be reversed. Our lead drug candidate,is a hypoxia platform technology with code name BXT-25, is planned to be
an Acellular Oxygen Carrier (“AOC”) consisting of bovine hemoglobin stabilized with a co-polymer with intended applications
to include treatment of hypoxic conditions in the brain resulting from stroke, and hypoxic conditions in wounds to prevent necrosis and
to promote healing. The Company’s initial focus is the treatment of hypoxic conditions in the brain resulting from stroke, and
hypoxic conditions in wounds to prevent necrosis and to promote healing. The Company’s approach potentially will result in the
creation of safe drug alternatives to existing therapies for effectively addressing hypoxic conditions in humans. Our drug development
efforts are guided by specialists in co-polymer chemistry and other disciplines, and we intend to supplement our efforts with input from
a scientific and medical advisory board whose members are leading physicians.
Our
Subsidiary, Pharmalectin, Inc. (“Pharmalectin” or the “Subsidiary”) is pursuing their work with a candidate named,
ProLectin, a complex polysaccharide that binds to, and blocks the activity of galectin-3, a type of galectin. Galectins are a member
of a family of proteins in the body called lectins. These proteins interact with carbohydrate sugars located in, on the surface of, and
in between cells. This interaction causes the cells to change behavior, including cell movement, multiplication, and other cellular functions.
The interactions between lectins and their target carbohydrate sugars occur via a carbohydrate recognition domain, or CRD, within the
lectin. Galectins are a subfamily of lectins that have a CRD that bind specifically to se. Galectins have a broad range of functions,
including regulation of cell survival and adhesion, promotion of cell-to-cell interactions, growth of blood vessels, regulation of the
immune response and inflammation. During viral infections galectins are upregulated and downregulated based on the type of virus.
Our
Foreign Subsidiary, Pharmalectin (BVI), Inc. (“Pharmalectin BVI”) is the owner and custodian of the Company’s Copyrights,
Trade Marks and Patents.
Our
subsidiary, Pharmalectin India Pvt Ltd. (“Pharmalectin India”) is managing the Company’s local clinical research and
trials, and holds the local rights to commercialization.
Organization
Bioxytran,
Inc. was organized on October 5, 2017 as a Delaware corporation, with a taxing structure for U.S. federal and state income tax as a C-Corporation
with 95,000,000 authorized common shares with a par value of $0.0001, and 5,000,000 Preferred shares with a par value of $0.0001. On
September 21, 2018, the Company went under a reorganization in the form of a reverse merger and is currently registered as a Nevada corporation
with a taxing structure for U.S. federal and state income tax as a C-Corporation with 300,000,000 authorized common shares with a par
value of $0.001, and 50,000,000 Preferred shares with a par value of $0.001.
Basis
of Presentation
The
summary of significant accounting policies presented below is designed to assist in understanding the Company’s consolidated financial
statements. Such financial statements and accompanying notes are the representations of the Company’s management, who are responsible
for their integrity and objectivity. These accounting policies conform to accounting principles generally accepted in the United States
of America (“U.S. GAAP”) in all material respects and have been consistently applied in preparing the accompanying consolidated
financial statements. The Company has not earned any revenue from operations since inception. The Company chose December 31st
as its fiscal year end.
Principles
of Consolidation
The
accompanying consolidated financial statements include the accounts of Bioxytran, Inc. a Nevada Corporation, its wholly owned subsidiaries,
Pharmalectin (BVI), Inc of British Virgin Islands and Pharmalectin India Pvt Ltd as well as its majority owned subsidiary, Pharmalectin,
Inc. of Delaware (collectively, the “Company”) is 85% owned by the Company and the loss attributable to non-controlling interest
was $193,372 and $496,297 for the year ended December 31, 2022 and 2021. All intercompany accounts have been eliminated upon consolidation.
NOTE
2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
A
summary of the significant accounting policies applied in the preparation of the accompanying financial statements follows.
Cash
For
purposes of the Statement of Cash Flows, the Company considers all highly liquid debt instruments purchased with a maturity date of three
months or less to be cash equivalents.
Use
of Estimates
The
preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported
amounts of assets and liabilities, disclosure of contingent assets and liabilities at the date of the financial statements and the reported
amount of expenses during the reporting period. Significant estimates include the fair value of the Company’s stock, stock-based
compensation and the valuation allowance related to deferred tax assets. Actual results may differ from these estimates.
Net
Loss per Common Share, basic and diluted
The
Company computes earnings (loss) per share under Accounting Standards Codification subtopic 260-10, Earnings Per Share (“ASC 260-10”).
Net loss per common share is computed by dividing net loss by the weighted average number of shares of Common Stock outstanding during
the year. Diluted earnings per share, if presented, would include the dilution that would occur upon the exercise or conversion of all
potentially dilutive securities into Common Stock using the “treasury stock” and/or “if converted” methods as
applicable.
At
December 31, 2022, we would, based on the market price of $0.48/share, be obligated to issue approximately 17,689,085 shares of Common
Stock upon conversion of the currently outstanding convertible notes (the “New Notes”) and 492,030 shares upon exercise of
the warrants and 524,000 shares upon exercise of outstanding stock options. For the New Notes, the shares total is based on $2,299,581
of currently outstanding principal and unpaid interest. At December 31, 2021, we would, based on
the market price of $0.40/share, be obligated to issue approximately 17,312,961 shares of Common Stock upon conversion of the currently
outstanding convertible notes (the “New Notes”) and 272,000 shares upon exercise of the warrants and 668,000 shares
upon exercise of outstanding stock options. For the New Notes, the shares total is based on $2,250,685
of currently outstanding principal and unpaid interest.
The
2021 1-year notes (the “New Notes”), extended thorough May 2023, have an interest rate of 6% and are convertible at the lower
of (i) a fixed price of $0.13, or (ii) 85% of the closing price of any Qualified Financing, which consist of any fundraising receiving
gross proceeds of not less than $500,000. The New Notes are limited to converting no more than 4.99% of our issued an outstanding Common
Stock.
Stock
Based Compensation
The
Company measures the cost of services received from employees and non-employees in exchange for an award of equity instruments based
on the fair value of the award on the grant date pursuant ASC 718. Stock-based compensation expense is recorded by the Company in the
same expense classifications in the statements of operations, as if such amounts were paid in cash.
Income
Taxes
The
Company accounts for income taxes under the asset and liability method. Under this method, deferred tax assets and liabilities are recognized
for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities
and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates in effect for the year in which
those temporary differences are expected to be recovered or be settled. The effect on deferred tax assets and liabilities of a change
in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance is provided when it is more
likely than not that some portion of the gross deferred tax asset will not be realized. The Company records interest and penalties related
to income taxes as a component of provision for income taxes. The Company did not recognize any interest and penalty expense for the
years ended December 31, 2022 and 2021.
On
December 22, 2017, the Tax Cuts and Jobs Act (TCJA) was signed into law by the President of the United States. TCJA is a tax reform act
that among other things, reduced corporate tax rates to 21 percent effective January 1, 2018. FASB ASC 740, Income Taxes, requires deferred
tax assets and liabilities to be adjusted for the effect of a change in tax laws or rates in the year of enactment, which is the year
in which the change was signed into law. Accordingly, the Company adjusted its deferred tax assets and liabilities at December 31, 2017,
using the new corporate tax rate of 21 percent. See Note 10.
Research
and Development
The
Company accounts for research and development costs in accordance with Accounting Standards Codification subtopic 730-10, Research and
Development (“ASC 730-10”). Under ASC 730-10, all research and development costs must be charged to expense as incurred.
Accordingly, internal research and development costs are expensed as incurred. Third-party research and development costs are expensed
when the contracted work has been performed or as milestone results have been achieved as defined under the applicable agreement. Company-sponsored
research and development costs related to both present and future products are expensed in the period incurred. During the year ended
December 31, 2022 the Company incurred $977,768 in research and development expenses, while during the year ended December 31, 2021 the
Company incurred $2,013,762.
Intangibles
– Goodwill and Other
Valuation
of intangibles are in accordance with ASC 350. Costs associated with the application and award of patents in the U.S. and various other
countries are capitalized and amortized on a straight-line basis over the term of the patents as determined at award date, which varies
depending on the pendency period of the application, generally approximating seventeen years. Capitalized patent costs, also referred
to as patent prosecution costs, include internal legal labor, professional legal fees, government filing fees and translation fees related
to expanding the Company’s patent portfolio. Costs associated with the maintenance and annuity fees of patents are accounted for
as prepaid assets at the time of payment and amortized over the shorter of the maintenance period or remaining life of the related patent.
Accrued
Expenses
As
part of the process of preparing our consolidated financial statements, we are required to estimate accrued expenses. This process involves
identifying services that third parties have performed on our behalf and estimating the level of service performed and the associated
cost incurred on these services as at each balance sheet date in our consolidated financial statements. Examples of estimated accrued
expenses include professional service fees, such as those arising from the services of attorneys and accountants and accrued payroll
expenses. In connection with these service fees, our estimates are most affected by our understanding of the status and timing of services
provided relative to the actual services incurred by the service providers. In the event that we do not identify certain costs that have
been incurred or we under- or over-estimate the level of services or costs of such services, our reported expenses for a reporting period
could be understated or overstated. The date on which certain services commence, the level of services performed on or before a given
date, and the cost of services are often subject to our judgment. We make these judgments based upon the facts and circumstances known
to us in accordance with accounting principles generally accepted in the U.S.
Warrants
The
Company has issued Common Stock warrants in connection with the execution of certain equity and debt financings. The fair value of warrants
is determined using the Black-Scholes option-pricing model using assumptions regarding volatility of our common share price, remaining
life of the warrant, and risk-free interest rates at each period end.
Fair
Value
Accounting
Standards Codification subtopic 825-10, Financial Instruments (“ASC 825-10”) requires disclosure of the fair value of certain
financial instruments. The carrying value of cash and cash equivalents, accounts payable and accrued liabilities, and short-term borrowings,
as reflected in the balance sheets, approximate fair value because of the short-term maturity of these instruments. All other significant
financial assets, financial liabilities and equity instruments of the Company are either recognized or disclosed in the financial statements
together with other information relevant for making a reasonable assessment of future cash flows, interest rate risk and credit risk.
Where practicable the fair values of financial assets and financial liabilities have been determined and disclosed; otherwise only available
information pertinent to fair value has been disclosed.
The
Company follows Accounting Standards Codification subtopic 820-10, Fair Value Measurements and Disclosures (“ASC 820-10”)
and Accounting Standards Codification subtopic 825-10, Financial Instruments (“ASC 825-10”), which permits entities to choose
to measure many financial instruments and certain other items at fair value.
Recent
Accounting Pronouncements
There
were various updates recently issued, most of which represented technical corrections to the accounting literature or application to
specific industries and are not expected to a have a material impact on the Company’s financial position, results of operations
or cash flows.
NOTE
3 – GOING CONCERN AND MANAGEMENT’S LIQUIDITY PLANS
As
at December 31, 2022, the Company had cash of $295,401 and a negative working capital of $3,368,080. As at December 31, 2022, the Company
has not yet generated any revenues, and has incurred cumulative net losses of $11,217,600. These conditions raise substantial doubt about
the Company’s ability to continue as a going concern.
During
the year ended December 31, 2022, the Company raised $1,467,000 from issuance of convertible notes. The Company also raised $680,000
in private placements. During the same period in 2021, the Company raised $2,165,000 from issuance of convertible notes, and cleared
up the defaulted convertible loans that mounted to $2,020,323. The Company also raised $600,000 in cash proceeds from the issuance of
Common Stock in our Subsidiary. The Company is aware that its current cash on hand will not be sufficient to fund its projected operating
requirements through the month of May 2023 and is pursuing alternative opportunities to funding.
The
Company intends to raise additional capital through private placements of debt and equity securities, but there can be no assurance that
these funds will be available on terms acceptable to the Company, or will be sufficient to enable the Company to fully complete its development
activities or sustain operations. If the Company is unable to raise sufficient additional funds, it will have to develop and implement
a plan to further extend payables, reduce overhead, or scale back its current business plan until sufficient additional capital is raised
to support further operations. There can be no assurance that such a plan will be successful.
Accordingly,
the accompanying consolidated financial statements have been prepared in conformity with U.S. GAAP, which contemplates continuation of
the Company as a going concern and the realization of assets and satisfaction of liabilities in the normal course of business. The carrying
amounts of assets and liabilities presented in the financial statements do not necessarily purport to represent realizable or settlement
values. The consolidated financial statements do not include any adjustment that might result from the outcome of this uncertainty.
NOTE
4 - RELATED PARTY TRANSACTIONS
The
Company hold License Agreements (the “License/s” or “Agreement/s”) for a medical device (license obtained in
2019) and a compound (license obtained in 2021), with two affiliated companies where in the officers of the Company hold a majority interest.
The products were developed prior to the establishment of Bioxytran. The maintenance cost for each license amounted to $5,000 in 2022,
$4,500 in 2021 and $4,220 in 2020. Additionally, the Company has reimbursed the affiliates for the legal and administrative costs surrounding
the establishment of the Licenses for an amount of $12,000 per agreement and reimburse $1,500 in rental cost for storage. During the
year ended December 31, 2022 one affiliate was paid $17,000, and the other was paid $27,220. In the year ended December 31, 2021, there
was $5,125 in transactions with affiliates.
NOTE
5 - INTANGIBLES
Intangible
assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not
be recoverable. No impairment charges were recorded for the years ended December 31, 2022 and 2021.
Amortization
of capitalized patent costs associated with the application and award of patents in the U.S. and various other countries are capitalized
and amortized on a straight-line basis over the term of the patents as determined at the award date, which varies depending on the pendency
period of the application, generally approximating seventeen years. The current patent application is still in process, and is therefore
not yet amortized.
SCHEDULE
OF INTANGIBLES
| | |
Estimated Remaining
Life (years) | | |
December 31, 2022 | | |
December 31, 2021 | |
| Capitalized patent costs | |
| 18 | | |
$ | 79,179 | | |
$ | 46,932 | |
| Accumulated amortization | |
| | | |
| 3,644 | | |
| — | |
| Intangible assets, net | |
| | | |
$ | 75,535 | | |
$ | 46,932 | |
NOTE
6 – ACCOUNTS PAYABLES AND ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
On
December 31, 2022, there was $709,727 in Accounts Payables to related parties in form of payroll and advanced expenses. On December 31,
2021 there was $531,000 in Accounts Payables to related parties.
The
following table represents the major components of accounts payables and accrued expenses and other current liabilities at December 31,
2022 and 2021:
SCHEDULE
OF ACCOUNTS PAYABLES AND ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Accounts payable related party (1) | |
$ | 709,727 | | |
$ | 531,000 | |
| Professional fees | |
| 393,085 | | |
| 375,371 | |
| Interest | |
| 134,581 | | |
| 85,685 | |
| Payroll taxes | |
| 40,182 | | |
| 32,010 | |
| Pension/401K | |
| 180,557 | | |
| 131,250 | |
| Other accounts payable | |
| 990 | | |
| — | |
| Un-issued shares related party | |
| 38,400 | | |
| — | |
| Un-issued shares | |
| 960 | | |
| — | |
| Convertible note payable | |
| 2,165,000 | | |
| 2,122,181 | |
| Total | |
$ | 3,663,482 | | |
$ | 3,277,497 | |
| (1) |
$286,900 to the CEO, 269,400 to the CFO and $153,427
and the CCO for 8 months of salary for the period May through December 2022, while there was $210,000 to each the CFO and the CEO
at and $111,000 for the CCO at December 31, 2021. All earlier accrued salaries due were forfeited on August 1, 2022. |
NOTE
7 – CONVERTIBLE NOTES PAYABLE
Private
Placement, 2021 Notes
Around
April 29, 2021, we entered into nine (9) Securities Purchase Agreements (the “2021 SPA’s”), under which we agreed to
sell convertible promissory notes (the “2021 Notes”), in an aggregate principal amount of $3,266,845 with 6% interest, whereof
$1,000,000 were contributed in form of cancellation of third-party notes, while 1,101,846 were issued in compensation for accrued compensation,
$981,466 to our three officers and $120,380 to two consultants.
At
any time after the issue date of the Notes, The Holders of the Notes, (the “2021 Holders”), have the option to convert all
or any part of the outstanding and unpaid principal amount and accrued and unpaid interest of the 2021 Notes into shares of our Common
Stock at the Conversion Price. The “Conversion Price” will be the lesser of (i) $.13 per share or (ii) 85% of the closing
price of Any Qualified Financing, which consists of any fundraising whereby the Company receives gross proceeds of not less than $500,000.
The
variable conversion rate component requires that the 2021 Notes to be valued at its stock redemption value (i.e., “if-converted”
value) pursuant to ASC 480, Distinguishing Liabilities from Equity, with the excess over the undiscounted face value being deemed a premium
to be added to the principal balance and accreted to additional paid-in capital over the life of the 2021 Notes. No such recording of
a premium was required as the discounted “if-converted” rate of $0.13 per share, was identical to fair market value of the
Company’s stock on the 2021 Notes date of issuance.
The
2021 Holders are limited to holding a total of 4.99% of our issued and outstanding Common Stock at any one time.
The
Common Stock underlying the 2021 Notes, when issued, will bear a restrictive legend and have a 180-day lock-up period.
On
June 4, 2021, 8,522,125 shares of Common Stock were issued as a result of conversion of accrued interest and principal for five convertible
notes for a total of $1,101,846, or $0.13/share. To avoid dilution of the Company’s stock 7,591,261 of these shares held by
our officers were returned to treasury on November 20, 2021, while the original debt consisting of accrued salary was forfeited.
SCHEDULE
OF CONVERTIBLE CONVERSION OF ACCRUED INTEREST AND PRINCIPAL
| Name | |
| |
Principal Converted | | |
Accrued interest converted | | |
No. of shares
issued | |
| Private Placement, 2021 Notes issued to Officers | |
(1) | |
$ | 981,466 | | |
$ | 5,398 | | |
| 7,591,261 | |
| Private Placement, 2021 Notes issued to consultants | |
| |
| 120,380 | | |
| 662 | | |
| 930,864 | |
| | |
| |
$ | 1,101,846 | | |
$ | 6,060 | | |
| 8,522,125 | |
| (1) |
Net cash received for these
notes were $1,380,960, after a Debt Discount of $86,040 was paid to the sole Placement Agent: WallachBeth Capital, LLC (Member FINRA
/ SIPC). |
If
the remainder of the 2021 Notes are converted prior to us paying off such note, it would lead to substantial dilution to our Shareholders
as a result of the conversion discounted applicable to the 2021 Notes. There can be no assurance that there will be any funds available
to pay of the 2021 Notes. If we fail to obtain such additional financing on a timely basis, the 2021 Holders may convert the 2021 Notes
and sell the underlying shares, which may result in significant dilution to Shareholders due to the conversion discount, as well as a
significant decrease in our stock price.
Convertible
notes payable and interest payable consist of the following at December 31, 2022 and 2021:
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Principal balance (1), (2) | |
$ | 2,165,000 | | |
$ | 2,165,000 | |
| Interest Payable | |
| 134,581 | | |
| 85,685 | |
| Unamortized debt discount | |
| — | | |
| (42,819 | ) |
| Outstanding, net of debt discount and premium | |
$ | 2,299,581 | | |
$ | 2,207,866 | |
| (1) |
Net cash received for these
notes were $1,045,150, after a Debt Discount of $119,850 was paid to the sole Placement Agent: WallachBeth Capital, LLC (Member FINRA
/ SIPC). |
| (2) |
$2 million of principal,
accrued interest and default penalties for notes issued prior to 2021, where settled by a third party in exchange for us issuing
to them a note in the amount of $1 million. |
Private
Placement, 2022 Notes converted into Common Stock
In
January, 2022, we entered into thirty-four (34) Securities Purchase Agreements (the “2022 SPA’s”), with accredited
investors, under which we agreed to sell the Notes, in an aggregate principal amount of $1,467,000 with 6% interest (the “2022
Notes”) to the holders of the 2022 Notes (the “2022 Holders”).
At
any time after the issue date of the 2022 Notes the 2022 Holders have the option to convert all or any part of the outstanding and unpaid
principal amount and accrued and unpaid interest of the Notes into shares of our Common Stock at the Conversion Price. The “Conversion
Price” is set to $0.25 per share.
The
2022 Holders are limited to holding a total of 4.99% of our issued and outstanding Common Stock at any one time. The Common Stock underlying
the 2022 Notes, when issued, bear a restrictive legend and are currently eligible for resale under Rule 144.
The
notes principal and accrued interest were fully converted into 6,081,484 shares of Common Stock on August 31, 2022.
| Name | |
| |
Principal Converted | | |
Accrued interest converted | | |
No. of shares issued | |
| Private Placement, 2022 Notes | |
(1) | |
$ | 1,467,000 | | |
$ | 53,371 | | |
| 6,081,484 | |
| | |
| |
$ | 1,467,000 | | |
$ | 53,371 | | |
| 6,081,484 | |
| (1) |
Net cash received for these
notes were $1,380,960, after a Debt Discount of $86,040 was paid to the sole Placement Agent: WallachBeth Capital, LLC (Member FINRA
/ SIPC). |
NOTE
8 – STOCKHOLDERS’ EQUITY
Preferred
stock
As
at December 31, 2022 and 2021, no preferred shares have been designated or issued.
Common
stock
On
June 4, 2021, 930,864 shares of Common Stock were issued to two consultants as a result of conversion of accrued interest and principal
for two convertible notes for a total of $121,042.
On
June 4, 2021, 7,591,261 shares of Common Stock were issued to management as a result of conversion of accrued interest and principal
for three convertible notes for a total of $986,864. To avoid dilution of the Company stock, the shares were returned to treasury and
cancelled on November 20, 2021, and the original debt consisting of accrued salary was forgiven.
On
December 3, 2021 a Company affiliate converted their holdings in the Subsidiary into 4,754,552 shares of Common Stock, or $0.2945/share
in accordance with a joint venture agreement.
On
August 15, 2022 1,400,000 shares were sold in a private placement for an amount of $600,000, or $0.43/share.
On
August 31, 2022, 6,081,484 shares of Common Stock were issued against convertible notes with a principal of $1,467,000 and an accrued
interest of $53,371, or $0.25/share.
On
September 8, 2022, 4,139,503 shares of Common Stock were issued in exchange against four outstanding warrants including provisions for
dilutive issuance and cashless exercise.
On
November 28, 2022, 156,250 shares were sold in a private placement for an amount of $50,000, or $0.32/share.
On
December 29, 2022, 93,750 shares were sold in a private placement for an amount of $30,000, or $0.32/share. The investment appears under
the label Stock subscription in the Stockholders equity and are not included in the outstanding shares at December 31, 2022.
For
the year ended December 31, 2022, a net of 716,000 shares of Common Stock were awarded, at an average cost per share of $0.25, under
the 2021 Stock Plan for a total value of $178,676. For the year ended December 31, 2021, a net of 7,704,909 shares of Common Stock were
awarded, at an average cost per share of $0.07, under the 2010 and the 2021 Stock Plans for a total value of $557,422.
As
at December 31, 2022, the Company has 123,252,235 shares of Common Stock issued and outstanding. At December 31, 2021 there were 110,840,998
shares of Common Stock issued and outstanding.
Common
Stock Warrants
The
fair value of stock warrants granted for the year ended December 31, 2021 was calculated with the following assumptions:
| | |
2022 | |
|
2021 | |
| Risk-free interest rate | |
| 1.37 – 4.45 | % |
|
| 0.16 - 1.00 | % |
| Expected dividend yield | |
| 0 | % |
|
| 0 | % |
| Volatility factor (monthly) | |
| 155.52 | % |
|
| 175.34 | % |
| Expected life of warrant | |
| 5 years | |
|
| 5 years | |
For
the year ended December 31, 2022 the Company awarded 492,030 warrants, valued at $190,335, while 22,000 warrants were retired, valued
at $6,763, and 4,139,503 shares of Common Stock were issued in a cashless exercise. For the year ended December 31, 2021 the Company
did not issue any warrants.
The
following table summarizes the Company’s Common Stock warrant activity for the year ended December 31, 2022 and 2021:
| | |
Number
of Warrants * | | |
Weighted
Average Exercise Price | | |
Weighted
Average Remaining Expected Term | |
| Outstanding as at January 1, 2021 | |
| 272,000 | | |
$ | 2.00 | | |
| 3.9 | |
| Granted | |
| — | | |
| — | | |
| — | |
| Exercised | |
| — | | |
| — | | |
| — | |
| Forfeited/Cancelled | |
| — | | |
| — | | |
| — | |
| Outstanding as at December 31, 2021 | |
| 272,000 | | |
| 2.00 | | |
| 3.7 | |
| Granted | |
| 492,030 | | |
| 0.26 | | |
| 5.0 | |
| Exercised | |
| (200,000 | ) | |
| 2.00 | | |
| — | |
| Forfeited/Cancelled | |
| (22,000 | ) | |
| 2.00 | | |
| — | |
| Outstanding as at December 31, 2022 | |
| 542,030 | | |
$ | 0.42 | | |
| 4.1 | |
| * |
The
warrant agreements issued in 2019 for a total of 50,000 warrants include provisions for dilutive issuance and cash-less exercise.
If exercised at December 31, 2022 the provisions would have resulted in an issuance of 1,130,114
shares at an average conversion price of $0.09, or 1,050,114 shares in a cash-less exercise.
In order to mitigate the Company’s risk an administrative hold has been placed on one Shareholder’s stock in the event
of future exercise. |
The
following table summarizes information about stock warrants that are vested or expected to vest at December 31, 2022 with a market price
of $0.48 at December 31, 2022:
SCHEDULE
OF WARRANT OUTSTANDING AND EXERCISABLE WARRANTS
| | | |
Warrants Outstanding | | |
| | |
| | |
Exercisable Warrants | | |
| |
| Number of Warrants | | |
Weighted Average Exercise Price Per Share | | |
Weighted Average Remaining Contractual Life (Years) | | |
Aggregate Intrinsic Value | | |
Number of Warrants | | |
Weighted Average Exercise Price Per Share | | |
Weighted Average Remaining Contractual Life (Years) | | |
Aggregate Intrinsic Value | |
| | 492,030 | | |
| 0.26 | | |
| 4.3 | | |
$ | 107,014 | | |
| 492,030 | | |
| 0.26 | | |
| 4.3 | | |
$ | 107,014 | |
| | 50,000 | | |
$ | 2.00 | | |
| 1.8 | | |
$ | — | | |
| 50,000 | | |
$ | 2.07 | | |
| 1.8 | | |
$ | — | |
| | 542,030 | | |
$ | 1.14 | | |
| 4.1 | | |
$ | 107,014 | | |
| 542,030 | | |
$ | 0.42 | | |
| 4.1 | | |
$ | 107,014 | |
The
weighted-average remaining contractual life for warrants exercisable at December 31, 2022 is 4.1 years. The aggregate intrinsic value
for fully vested, exercisable warrants was $107,014 at December 31, 2022.
The
following table sets forth the status of the Company’s non-vested warrants as at December 31, 2022, there were no warrants issued
for the year ended at December 31, 2021.
SCHEDULE
OF NON-VESTED WARRANTS
| | |
Number of Warrants | | |
Weighted- Average Grant-Date Fair Value per share | |
| Non-vested as at December 31, 2021 | |
| — | | |
$ | — | |
| Granted | |
| 492,030 | | |
| 0.26 | |
| Forfeited/Cancelled | |
| — | | |
| — | |
| Vested | |
| 492,030 | | |
| 0.26 | |
| Non-vested as at December 31, 2022 | |
| — | | |
$ | — | |
Common
Stock Options
For
the year ended December 31, 2022 there were no options awarded under the 2021 Stock Plan. However, 144,000 options were forfeited. For
the year ended December 31, 2021 there were 135,000 options awarded under the 2021 Stock Option Plan. The options total fair value at
the time of award was $14,490.
NOTE
9 – STOCK OPTION PLAN AND STOCK-BASED COMPENSATION
On
January 15, 2021, the Company adopted a stock option plan entitled “The 2021 Stock Plan” (2021 Plan) under which the Company
may grant Options to Purchase Stock, Stock Awards or Stock Appreciation Rights up to 15% of the then fully diluted number of shares of
the Company’s Common Stock, automatically adjusted on January 1 each year. As at December 31, 2022, there were 668,000 outstanding
stock options valued at historic fair market value of $367,400 and 1,669,000 shares issued valued at a fair historic market value of
$43,919 at the time of award. As at December 31, 2021, there was “The 2010 Stock Plan” under this plan there were 533,000
outstanding stock options with a fair historic market value of $275,603 and 11,002,000 shares issued with a fair historic market value
of $1,075,358 at the time of award.
Under
the terms of the stock plans, the Board of Directors shall specify the exercise price and vesting period of each stock option on the
grant date. Vesting of the options is typically immediate and the options typically expire in five years. Stock Awards may be directly
issued under the Plan (without any intervening options). Stock Awards may be issued which are fully and immediately vested upon issuance.
Shares
Awarded and Issued 2010 Plan:
On
January 1, 2021 the Company granted 10,000 shares, with a fair market value of $0.24/share at the time of award, to a Medical Advisory
Board Member for her contribution in the Company’s Advisory Board, for a total of $2,400.
On
January 15, 2021 the Company granted 3,189,200 shares of Common Stock valued at $0.24/share, equally divided to 227,800 shares/each to
fourteen of the Company’s Managers, Board- and Medical Advisory Board members, as well as to indispensable Consultants currently
working on the clinical trial submissions with the FDA, for a total value of $765,408. On November 20, 2021, the Management returned
1,083,400 of these shares to the Plan in order to avoid dilution of the Company stock, the shares were cancelled upon return. The shares
market value at the time of issuance were $260,016, or $0.24/share.
Shares
Awarded and Issued 2021 Plan:
On
April 1, 2021 the Company granted 10,000 shares, with a fair market value of $0.17/share at the time of award, to a Medical Advisory
Board Member for her contribution in the Company’s Advisory Board, for a total of $1,700.
On
April 1, 2021 the Company granted 90,000 shares with a fair market value of $0.17/share to three members of the Audit Committee as compensation
for their contribution in the Audit Committee, for a total of $15,300.
On
April 22, 2021 the Company granted 150,000 shares with a fair market value of $0.17/share at the time of award, to a consultant for assistance
with the Companies PR work, for a total of $25,500.
On
June 15, 2021 the Company granted 450,000 shares with a fair market value of $0.001/share at the time of award, to a consultant for assistance
with the Companies PR work, for a total of $450.
On
July 1, 2021 the Company granted 10,000 shares to a Medical Advisory Board Member for her contribution to the Company during the second
quarter of 2021. The total fair market value at the time of the award was $10, or $0.001/share.
On
July 1, 2021 the Company granted 90,000 shares to three Board Members in reward of their attendance at Board and Committee meetings during
the second quarter of 2021. The total fair market value at the time of the award was $90, or $0.001/share.
On
August 2, 2021 the Company granted 699,000 shares to our Investment Banker as per outlined in the PPM for a total value of $699, or $0.001/share.
On
October 1, 2021 the Company granted 170,000 shares to four Board members in reward of their attendance at Board and Committee meetings
during the third quarter of 2021. The total fair market value at the time of the award was $170, or $0.001/share.
On
November 20, 2021 the Company granted 3,597,529 shares to an affiliate for their development and regulatory work with the Company’s
first indication. The total fair market value at the time of the award was $7,594, or 0.0021/share.
On
December 3, 2021 the Company granted 322,580 shares to an affiliate as compensation for Management Fee and Legal Expenses for a total
value of $95,000, or 0.2945/share as per written agreement.
On
January 10, 2022 the Company granted 40,000 shares of Common Stock to four Board Members in reward of their attendance at Board and Committee
meetings during the fourth quarter of 2021. The total fair market value at the time of the award was $6,400, or $0.16/share. The shares
were issued on August 1, 2022
On
February 18, 2022 the Company granted 100,000 shares of Common Stock to two Consultants in reward of their assistance for the product
development and our clinical trials in India. The total fair market value at the time of the award was $16,000, or $0.16/share. The shares
were issued on August 1, 2022
On
April 1, 2022 the Company granted 10,000 shares to a Medical Advisory Board Member for her contribution to the Company during the first
quarter of 2022. The total fair market value at the time of the award was $1,730, or $0.173/share. The shares were issued on August 1,
2022
On
April 1, 2022 the Company granted 70,000 shares to four Board Members in reward of their attendance at Board and Committee meetings during
the first quarter of 2022. The total fair market value at the time of the award was $12,110, or $0.173/share. The shares were issued
on August 1, 2022.
On
April 11, 2022 the Company granted 250,000 shares to three Consultants for the management of our clinical trials in India. The total
fair market value at the time of the award was $43,250, or $0.173/share. The shares were issued on August 1, 2022.
On
August 1, 2022 the Company issued 82,000 shares to four Board Members in reward of their attendance at Board and Committee meetings during
the second quarter of 2022. The total fair market value at the time of the award was $26,240, or $0.32/share.
On
October 28, 2022 the Company granted 82,000 shares to four Board Members in reward of their attendance at Board and Committee meetings
during the third quarter of 2022. The total fair market value at the time of the award was $33,292, or $0.406/share. The shares were
issued on December 19, 2022.
| | |
Number of Shares | | |
Fair Value per Share | | |
Weighted Average Market Value per Share | |
| Shares Issued as of January 1, 2021 | |
| 11,002,000 | | |
$ | 0.003 – 1.49 | | |
$ | 0.099 | |
| Shares Issued | |
| 7,704,909 | | |
| 0.001
–
0.55 | | |
| 0.072 | |
| Shares Issued as of December 31, 2021 | |
| 18,706,909 | | |
$ | 0.001 – 1.49 | | |
$ | 0.088 | |
| Shares Issued | |
| 716,000 | | |
| 0.162
–
0.48 | | |
| 0.250 | |
| Shares Issued as of December 31, 2022 | |
| 19,422,909 | | |
$ | 0.001 – 1.49 | | |
$ | 0.094 | |
For
the year ended December 31, 2022, the Company recorded stock-based compensation expense of $228,407
in connection with share-based payment awards.
For the year ended December 31, 2021, the Company recorded stock-based compensation expense of $557,422
in connection with share-based payment awards.
Stock
options granted and vested 2021 Plan:
On
February 1, 2021 the Company granted 45,000 three-year options immediately vested at an exercise price of $0.20 to an Advisory Board
Member for his contribution in the Company’s Advisory Board. The options total fair value at the time of award was $6,750.
On
May 1, 2021 the Company granted 45,000 three-year options immediately vested at an exercise price of $0.19 to a Medical Advisory Board
Member for his contribution in the Company’s Advisory Board. The options total fair value at the time of award was $7,650.
On
August 1, 2021 the Company granted 45,000 3-year options immediately vested at an exercise price of $0.001 to a Medical Advisory Board
Member for his contribution in the Company’s Advisory Board. The options total fair value at the time of award was $45.
The
fair value of stock options granted and revaluation of non-employee consultant options for the year ended December 31, 2022 and 2021
was calculated with the following assumptions:
| | |
2022 | | |
2021 | |
| Risk-free interest rate | |
| 1.02 – 4.66 | % | |
| 0.16 - 1.00 | % |
| Expected dividend yield | |
| 0 | % | |
| 0 | % |
| Volatility factor (monthly) | |
| 155.52 | % | |
| 175.34 | % |
| Expected life of option | |
| 3 years | | |
| 3 years | |
There
were no options issued in the year ended December 31, 2022. Although, 144,000 options were forfeited at the expiration date and returned
to the stock plan. For the year ended December 31, 2021, the Company recorded compensation expense of $14,445 in connection with 135,000
awarded stock options.
As
at December 31, 2022, there was no unrecognized compensation expense related to non-vested stock option awards. The following table summarizes
the Company’s stock option activity for the year ended December 31, 2022 and 2021:
SCHEDULE
OF STOCK OPTIONS ACTIVITY
| | |
Number of Options | | |
Exercise Price per Share | | |
Weighted Average Exercise Price per Share | |
| Outstanding as of January 1, 2021 | |
| 533,000 | | |
$ | 0.001 - 1.21 | | |
$ | 0.71 | |
| Granted | |
| 135,000 | | |
| 0.001 - 0.20 | | |
| 0.20 | |
| Exercised | |
| — | | |
| — | | |
| — | |
| Options forfeited/cancelled | |
| — | | |
| — | | |
| — | |
| Outstanding as of December 31, 2021 | |
| 668,000 | | |
$ | 0.001 - 1.21 | | |
$ | 0.55 | |
| Granted | |
| — | | |
| — | | |
| — | |
| Exercised | |
| — | | |
| — | | |
| — | |
| Options forfeited/cancelled | |
| (144,000 | ) | |
| 0.31 – 1.21 | | |
| 0.81 | |
| Outstanding as of December 31, 2022 | |
| 524,000 | | |
$ | 0.001 – 0.95 | | |
$ | 0.44 | |
The
following table summarizes information about stock options that are vested or expected to vest at December 31, 2022:
SCHEDULE
OF STOCK OPTION VESTED
| | | |
| | |
Options Outstanding | | |
| | |
| | |
Exercisable Options | | |
| |
| Exercise Price | | |
Number of Options | | |
Weighted Average Exercise Price Per Share | | |
Weighted Average Remaining Contractual Life (Years) | | |
Aggregate Intrinsic Value | | |
Number of Options | | |
Weighted Average Exercise Price Per Share | | |
Weighted Average Remaining Contractual Life (Years) | | |
Aggregate Intrinsic Value | |
| $ | 0.001 | | |
| 90,000 | | |
$ | 0.001 | | |
| 0.95 | | |
$ | 43,110 | | |
| 90,000 | | |
$ | 0.001 | | |
| 0.95 | | |
$ | 43,110 | |
| | 0.05 | | |
| 3,000 | | |
| 0.05 | | |
| 0.75 | | |
| 1,292 | | |
| 3,000 | | |
| 0.05 | | |
| 0.75 | | |
| 1,292 | |
| | 0.15 | | |
| 90,000 | | |
| 0.15 | | |
| 0.33 | | |
| 29,340 | | |
| 90,000 | | |
| 0.15 | | |
| 0.33 | | |
| 29,340 | |
| | 0.18 | | |
| 45,000 | | |
| 0.18 | | |
| 0.83 | | |
| 13,680 | | |
| 45,000 | | |
| 0.18 | | |
| 0.83 | | |
| 13,680 | |
| | 0.19 | | |
| 45,000 | | |
| 0.19 | | |
| 1.33 | | |
| 13,185 | | |
| 45,000 | | |
| 0.19 | | |
| 1.33 | | |
| 13,185 | |
| | 0.20 | | |
| 48,000 | | |
| 0.20 | | |
| 1.04 | | |
| 13,446 | | |
| 48,000 | | |
| 0.20 | | |
| 1.04 | | |
| 13,446 | |
| | 0.32 | | |
| 3,000 | | |
| 0.32 | | |
| 0.25 | | |
| 467 | | |
| 3,000 | | |
| 0.32 | | |
| 0.25 | | |
| 467 | |
| | 0.95 | | |
| 200,000 | | |
| 0.95 | | |
| 1.26 | | |
| — | | |
| 200,000 | | |
| 0.95 | | |
| 1.26 | | |
| — | |
| $ | 0.001-0.95 | | |
| 524,000 | | |
$ | 0.44 | | |
| 0.99 | | |
$ | 114,519 | | |
| 524,000 | | |
$ | 0.44 | | |
| 0.99 | | |
$ | 114,519 | |
The
weighted-average remaining estimated life for options exercisable at December 31, 2022 is 0.99 years.
The
aggregate intrinsic value for fully vested, exercisable options was $114,519 at December 31, 2022. The actual tax benefit realized from
stock option exercises for the year ended at December 31, 2022 and 2021 was $0 as no options were exercised.
As
at December 31, 2022 the Company has 18,729,292 options or stock awards available for grant under the 2021 Plan.
NOTE
10 – NON-CONTROLLING INTEREST
SCHEDULE
OF NON CONTROLLING INTEREST
| | |
December 31, 2022 | | |
December 31, 2021 | |
| Net loss Subsidiary | |
| (817,151 | ) | |
| (2,089,253 | ) |
| Net loss attributable to the non-controlling interest | |
| 193,372 | | |
| 496,297 | |
| Net loss affecting Bioxytran | |
| (623,780 | ) | |
| (1,592,956 | ) |
| | |
| | | |
| | |
| Accumulated losses | |
| (3,594,287 | ) | |
| (2,777,135 | ) |
| Accumulated losses attributable to the non-controlling interest | |
| 751,578 | | |
| 558,206 | |
| Accumulated losses affecting Bioxytran | |
| (2,842,709 | ) | |
| (2,218,929 | ) |
| | |
| | | |
| | |
| Net equity non-controlling interest | |
| (590,628 | ) | |
| (397,256 | ) |
As
at December 31, 2022 and 2021 there were 30,000,000 issued and 19,650,000 outstanding shares; 15,000,000 Common shares (76%) are held
by Bioxytran and 4,650,000 Common shares are held by an affiliate. Further, an additional 4,500,000 options exercisable at $0.33 are
held by an affiliate. The beneficial ownership of the
affiliate includes Mike Sheikh, Ola Soderquist and David Platt.
The
option agreements include provisions for dilutive issuance and cash-less exercise. If exercised at December 31, 2022 the provisions would
have resulted in an issuance of 16,782,189 shares at an average conversion price of $0.08849, or 15,594,189 shares in a cash-less exercise.
NOTE
11 – PROVISION FOR INCOME TAXES
Provision
for Income Taxes
During
the year ended December 31, 2022 and 2021, no provision for income taxes was recorded as the Company generated net operating losses.
The
tax effects of temporary differences that give rise to deferred tax assets are presented below:
SCHEDULE OF DEFERRED TAX ASSETS
| | |
2022 | | |
2021 | |
| Deferred Tax Assets: | |
| | | |
| | |
| Net operating loss carryforward | |
$ | 7,120,000 | | |
$ | 6,670,000 | |
| | |
| | | |
| | |
| Total deferred tax assets | |
| 1,500,000 | | |
| 1,400,000 | |
| | |
| | | |
| | |
| Valuation allowance | |
| (1,500,000 | ) | |
| (1,400,000 | ) |
| | |
| | | |
| | |
| Deferred tax asset, net of valuation allowance | |
$ | — | | |
$ | — | |
A
reconciliation of the statutory federal income tax rate to the Company’s effective tax rate is as follows:
SCHEDULE OF EFFECTIVE TAX RATE
| | |
| | | |
| | |
| Tax benefit at federal statutory rate | |
| (21.0 | )% | |
| (21.0 | )% |
| Valuation allowance | |
| 21.0 | % | |
| 21.0 | % |
| Effective Rate | |
| 0.0 | % | |
| 0.0 | % |
The
Company assesses the likelihood that deferred tax assets will be realized. To the extent that realization is not likely, a valuation
allowance is established. Based upon the Company’s history of losses since inception, management believes that it is more likely
than not that future benefits of deferred tax assets will not be realized.
At
December 31, 2022, the Company had approximately $7,120,000 of federal net operating losses that may be available to offset future
taxable income, At December 31, 2021, the Company had approximately $6,670,000 of federal net operating losses that may be available
to offset future taxable income. $2,870 of the net operating loss carry forwards (NOL), if not utilized, will expire in 2037 for federal
purposes, the remaining amount of NOL can be carried forward indefinitely. As at the fiscal year 2022, a deduction for issued warrants
and stock options and restricted shares awarded from the 2010 Stock Plan for a total of $2,200,000 has not yet been made, for
the fiscal year 2021 this total was $2,030,000. The market value less exercise price for these awards will be deducted if and when the
warrants and stock options are exercised, while the restricted shares will be deducted at market value at the date they were awarded,
once the restriction is removed.
Pursuant
to the Internal Revenue Code Section 382 (“Section 382”), certain ownership changes may subject the net operating loss carryforwards
(“carryforwards”) and research and development tax credit carryforwards to annual limitations which could reduce or defer
the carryforwards. Section 382 imposes limitations on a corporation’s ability to utilize carryforwards if it experiences an ownership
change. An ownership change may result from transactions increasing the ownership of certain stockholders in the stock of a corporation
by more than 50 percentage points over a three-year period. In the event of an ownership change, utilization of the carryforwards would
be subject to an annual limitation under Section 382 determined by multiplying the value of its stock at the time of the ownership change
by the applicable long-term tax-exempt rate. Any unused annual limitation may be carried over to later years. The imposition of this
limitation on its ability to use the carryforwards to offset future taxable income could cause the Company to pay U.S. federal income
taxes earlier than if such limitation were not in effect and could cause such carryforwards to expire unused, reducing or eliminating
the benefit of such carryforwards. The Company has not completed a Section 382 study to determine if there have been one or more ownership
changes due to the costs associated with such a study. Until a study is completed and the extent of the limitations, if any, is able
to be determined, no additional amounts have been written off or are being presented as an uncertain tax position.
On
December 22, 2017, the U.S. government enacted comprehensive tax legislation commonly referred to as the Tax Cut and Jobs Act (the “Tax
Act”). The Tax Act establishes new tax laws that affects 2019 and future years, including a reduction in the U.S. federal corporate
income tax rate to 21%, effective January 1, 2019.
The
Company applies the provisions of ASC 740-10, Income Taxes. The Company has not recognized any liability for unrecognized tax benefits
and does not believe there is any uncertainty with respect to its tax position. The Company’s policy with respect to unrecognized
tax benefits is to recognize interest accrued related to unrecognized tax benefits in interest expense and penalties in operating expenses.
The
Company files tax returns as prescribed by the tax laws of the jurisdictions in which it operates. In the normal course of business,
the Company is subject to examination by federal and state jurisdictions, where applicable. There are currently no pending income tax
examinations. Earlier years may be examined to the extent that tax credit or net operating loss carryforwards are used in future periods.
The Company’s policy is to record interest and penalties related to income taxes as part of its income tax provision.
NOTE
12 – COMMITMENTS AND CONTINGENCIES
Employment
contracts
Our
Executive Officers have entered into employment contracts and confidentiality, non-disclosure and assignment of invention agreements.
The most substantial provisions include;
| ● | Compensation
of three (3) times the employee’s annual salary upon the Termination Date and any target
bonus earned, or if termination occurs within 12 months of a change in control, then the
terminated employee shall receive two (2) times the employee’s annual salary and any
target bonus earned. |
| ● | Continued
coverage under any health, medical, dental or vision program or policy, in which they were
eligible to participate at the time of employment termination, for 12 months. |
| ● | Provide
outplacement services through one or more outside firms of the employee’s choosing
up to an aggregate of $50,000. |
There
are no other arrangements or plans in which we provide pension, retirement or similar benefits for any of Executive Officers or Directors.
Litigation
In
the normal course of business, the Company may be involved in legal proceedings, claims and assessments arising in the ordinary course
of business. Such matters are subject to many uncertainties, and outcomes are not predictable with assurance. Legal fees for such matters
are expensed as incurred and we accrue for adverse outcomes as they become probable and estimable.
On
June 5, 2020 the Supreme Court of the State of New York, County of Nassau, issued a commencement of Action based on behalf of Power Up
Lending Group, Ltd (“Power Up” or the “Claimant”). The Claimant request that due to the default of their note
requesting a judgment for an amount of not less than $420,750. On January 20, 2021 the Supreme Court of the State of New York, County
of Nassau, granted Power Up a summary judgement against the Company for Breach of Contact, awarding Power Up damages in the amount of
$420,750.
The
underlying convertible note was, per agreement of the parties, cancelled on June 4, 2021, with Power Up agreeing to a stipulation of
discontinuance with prejudice and forfeiture of on-going lawsuit and forfeiture of the mentioned awarded damages.
At
present, there is no other pending litigation or proceeding involving any of our Directors, Officers or employees as to which indemnification
is sought, nor are we aware of any threatened litigation or proceeding that may result in claims for indemnification.
NOTE
13 – SUBSEQUENT EVENTS
The
Company has evaluated events from December 31, 2022 through the date the financial statements were issued. The events requiring disclosure
for this period are as follows:
Common
stock
Reset
of 2021 Stock Plan
In
accordance with the stipulations of the 2021 Stock Plan, the Plan was reset at January 1, 2023. The Company has after the reset 19,892,071
option- or stock awards available for grant under the 2021 Plan.
Shares
issued in private placement
On
January 4, 2023 the Company issued 93,750 shares of Common Stock against $30,000, or $0.32/share, shown as stock subscription in the
December 31, 2022 stockholders’ equity statement.
On
February 10, 2023 the Company issued 156,250 shares of Common Stock against $50,000, or $0.32/share
Management
sees no further subsequent events requiring disclosure.
BIOXYTRAN,
INC.
CONDENSED
CONSOLIDATED BALANCE SHEETS
AS
OF JUNE 30, 2023 AND DECEMBER 31, 2022
(UNAUDITED)
| | |
June 30, 2023 | | |
December 31, 2022 | |
| ASSETS | |
| | | |
| | |
| Current assets: | |
| | | |
| | |
| Cash | |
$ | 45,181 | | |
$ | 295,401 | |
| Total current assets | |
| 45,181 | | |
| 295,401 | |
| | |
| | | |
| | |
| Intangibles, net | |
| 97,880 | | |
| 75,535 | |
| | |
| | | |
| | |
| Total assets | |
$ | 143,061 | | |
$ | 370,936 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable and accrued expenses | |
$ | 562,662 | | |
$ | 749,395 | |
| Accounts payable related party | |
| 200,500 | | |
| 709,727 | |
| Un-issued shares liability | |
| 380 | | |
| 960 | |
| Un-issued shares liability related party | |
| 5,700 | | |
| 38,400 | |
| Convertible notes payable, net of premium and discount | |
| 2,000,000 | | |
| 2,165,000 | |
| Total current liabilities | |
| 2,769,242 | | |
| 3,663,482 | |
| | |
| | | |
| | |
| Total liabilities | |
| 2,769,242 | | |
| 3,663,482 | |
| | |
| | | |
| | |
| Commitments and contingencies | |
| — | | |
| — | |
| | |
| | | |
| | |
| Stockholders’ deficit: | |
| | | |
| | |
| Preferred stock, $0.001 par value; 50,000,000 shares authorized, nil issued and outstanding | |
| — | | |
| — | |
| Common Stock, $0.001
par value; 300,000,000 shares authorized;
132,035,294 and 123,252,235 issued and outstanding
as at June 30, 2023, and December 31, 2022, respectively | |
| 132,035 | | |
| 123,252 | |
| Additional paid-in capital | |
| 11,279,970 | | |
| 8,392,430 | |
| Non-controlling interest | |
| (624,286 | ) | |
| (590,628 | ) |
| Accumulated deficit | |
| (13,413,901 | ) | |
| (11,217,600 | ) |
| Total stockholders’ deficit | |
| (2,626,182 | ) | |
| (3,292,546 | ) |
| | |
| | | |
| | |
| Total liabilities and stockholders’ deficit | |
$ | 143,060 | | |
$ | 370,936 | |
See
the accompanying notes to these unaudited condensed consolidated financial statements
BIOXYTRAN,
INC.
CONDENSED
CONSOLIDATED STATEMENTS OF OPERATIONS
FOR
THE THREE AND SIX MONTHS ENDED JUNE 30, 2023 AND 2022
(UNAUDITED)
| | |
June 30, 2023 | | |
June 30, 2022 | | |
June 30, 2023 | | |
June 30, 2022 | |
| | |
Three months ended | | |
Six months ended | |
| | |
June 30, 2023 | | |
June 30, 2022 | | |
June 30, 2023 | | |
June 30, 2022 | |
| Operating expenses: | |
| | | |
| | | |
| | | |
| | |
| Research and development | |
$ | 149,638 | | |
$ | 43,141 | | |
$ | 288,642 | | |
$ | 283,266 | |
| General and administrative | |
| 866,932 | | |
| 447,360 | | |
| 1,464,570 | | |
| 1,003,941 | |
| Compensation Expense | |
| 5,110 | | |
| 46,723 | | |
| 18,710 | | |
| 69,123 | |
| Total operating expenses | |
| 1,021,680 | | |
| 537,224 | | |
| 1,771,922 | | |
| 1,356,330 | |
| | |
| | | |
| | | |
| | | |
| | |
| Loss from operations | |
| (1,021,680 | ) | |
| (537,224 | ) | |
| (1,771,922 | ) | |
| (1,356,330 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Other expenses: | |
| | | |
| | | |
| | | |
| | |
| Interest expense | |
| (39,477 | ) | |
| (54,480 | ) | |
| (106,698 | ) | |
| (106,515 | ) |
| Amortization of IP | |
| (2,188 | ) | |
| (911 | ) | |
| (2,702 | ) | |
| (1,822 | ) |
| Debt discount amortization and issuance of warrants | |
| (348,637 | ) | |
| (41,425 | ) | |
| (348,637 | ) | |
| (132,759 | ) |
| Total other income (expenses) | |
| (390,302 | ) | |
| (96,816 | ) | |
| (458,037 | ) | |
| (241,096 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Net loss before provision for income taxes | |
| (1,411,982 | ) | |
| (634,040 | ) | |
| (2,229,959 | ) | |
| (1,597,426 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Provision for income taxes | |
| — | | |
| — | | |
| — | | |
| — | |
| NET LOSS | |
| (1,411,982 | ) | |
| (634,040 | ) | |
| (2,229,959 | ) | |
| (1,597,426 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Net loss attributable to the non-controlling interest | |
| 764 | | |
| 11,691 | | |
| 33,658 | | |
| 62,807 | |
| | |
| | | |
| | | |
| | | |
| | |
| NET LOSS ATTRIBUTABLE TO BIOXYTRAN | |
$ | (1,411,218 | ) | |
$ | (622,349 | ) | |
$ | (2,196,301 | ) | |
$ | (1,534,619 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Loss per Common share, basic and diluted | |
$ | (0.01 | ) | |
$ | (0.01 | ) | |
$ | (0.02 | ) | |
$ | (0.01 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Weighted average number of Common shares outstanding, basic and diluted | |
| 128,804,789 | | |
| 110,840,998 | | |
| 126,224,323 | | |
| 110,840,998 | |
See
the accompanying notes to these unaudited condensed consolidated financial statements
BIOXYTRAN,
INC.
CONDENSED
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ DEFICIT
FOR
THE SIX MONTHS ENDED JUNE 30, 2023 AND 2022
(UNAUDITED)
| | |
Shares | | |
Amount | | |
Shares | | |
Amount | | |
Common | | |
Preferred | | |
Deficit | | |
Interest | | |
Equity | |
| | |
Common Stock | | |
Preferred Stock | | |
Additional Paid in Capital | | |
Accumulated | | |
Non-controlling | | |
Total | |
| | |
Shares | | |
Amount | | |
Shares | | |
Amount | | |
Common | | |
Preferred | | |
Deficit | | |
Interest | | |
Equity | |
| January 1, 2022 | |
| 110,840,998 | | |
$ | 110,841 | | |
| — | | |
| — | | |
$ | 5,881,876 | | |
$ | — | | |
$ | (8,753,668 | ) | |
$ | (397,256 | ) | |
$ | (3,158,207 | ) |
| Issuance of Warrants | |
| | | |
| | | |
| | | |
| | | |
| 42,250 | | |
| | | |
| | | |
| | | |
| 42,250 | |
| Net loss attributable to the non-controlling interest | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (51,116 | ) | |
| (51,116 | ) |
| Net loss | |
| - | | |
| - | | |
| - | | |
| - | | |
| | | |
| - | | |
| (912,270 | ) | |
| | | |
| (912,270 | ) |
| March 31, 2022 | |
| 110,840,998 | | |
$ | 110,841 | | |
| — | | |
| — | | |
$ | 5,924,126 | | |
$ | — | | |
$ | (9,665,938 | ) | |
$ | (448,372 | ) | |
$ | (4,079,343 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net loss attributable to the non-controlling interest | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (11,691 | ) | |
| (11,691 | ) |
| Net loss | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (622,349 | ) | |
| | | |
| (622,349 | ) |
| June 30, 2023 | |
| 110,840,998 | | |
$ | 110,841 | | |
| — | | |
| — | | |
$ | 5,924,126 | | |
$ | — | | |
$ | (10,288,287 | ) | |
$ | (460,063 | ) | |
$ | (4,713,383 | ) |
| | |
Common Stock | | |
Preferred Stock | | |
Additional Paid in Capital | | |
Accumulated | | |
Non-controlling | | |
Total | |
| | |
Shares | | |
Amount | | |
Shares | | |
Amount | | |
Common | | |
Preferred | | |
Deficit | | |
Interest | | |
Equity | |
| January 1, 2023 | |
| 123,252,235 | | |
$ | 123,252 | | |
| — | | |
| — | | |
$ | 8,392,430 | | |
$ | — | | |
$ | (11,217,600 | ) | |
$ | (590,628 | ) | |
$ | (3,292,546 | ) |
| Stock transactions | |
| 250,000 | | |
| 250 | | |
| | | |
| | | |
| 79,750 | | |
| | | |
| | | |
| | | |
| 80,000 | |
| Stock subscription | |
| | | |
| | | |
| | | |
| | | |
| (30,000 | ) | |
| | | |
| | | |
| | | |
| (30,000 | ) |
| Net loss attributable to the non-controlling interest | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (32,894 | ) | |
| (32,894 | ) |
| Net loss | |
| | | |
| | | |
| - | | |
| - | | |
| | | |
| - | | |
| (785,083 | ) | |
| | | |
| (785,083 | ) |
| March 31, 2023 | |
| 123,502,235 | | |
$ | 123,502 | | |
| — | | |
| — | | |
$ | 8,442,180 | | |
$ | — | | |
$ | (12,002,683 | ) | |
$ | (623,522 | ) | |
$ | (4,060,523 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Stock transactions | |
| 192,411 | | |
| 192 | | |
| | | |
| | | |
| 64,808 | | |
| | | |
| | | |
| | | |
| 65,000 | |
| Shares issued to BoD & Mgmnt - 2021 Plan | |
| 110,000 | | |
| 110 | | |
| | | |
| | | |
| 50,090 | | |
| | | |
| | | |
| | | |
| 50,200 | |
| Shares issued to Consultants - 2021 Plan | |
| 4,000 | | |
| 4 | | |
| | | |
| | | |
| 1,786 | | |
| | | |
| | | |
| | | |
| 1,790 | |
| Shares issued to BoD & Mgmnt for conversion of debt | |
| 6,763,562 | | |
| 6,764 | | |
| | | |
| | | |
| 2,157,576 | | |
| | | |
| | | |
| | | |
| 2,164,340 | |
| Shares issued to Consultants for conversion of debt | |
| 137,656 | | |
| 138 | | |
| | | |
| | | |
| 43,912 | | |
| | | |
| | | |
| | | |
| 44,050 | |
| Conversion of debt | |
| 1,325,430 | | |
| 1,325 | | |
| | | |
| | | |
| 170,981 | | |
| | | |
| | | |
| | | |
| 172,306 | |
| Issuance of Warrants | |
| | | |
| | | |
| | | |
| | | |
| 348,637 | | |
| | | |
| | | |
| | | |
| 348,637 | |
| Net loss attributable to the non-controlling interest | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| (764 | ) | |
| (764 | ) |
| Net loss | |
| | | |
| | | |
| - | | |
| - | | |
| | | |
| - | | |
| (1,411,218 | ) | |
| | | |
| (1,411,218 | ) |
| June 30, 2023 | |
| 132,035,294 | | |
$ | 132,035 | | |
| — | | |
$ | — | | |
$ | 11,279,970 | | |
$ | — | | |
$ | (13,413,901 | ) | |
$ | (624,286 | ) | |
$ | (2,626,182 | ) |
See
the accompanying notes to these unaudited condensed consolidated financial statements
BIOXYTRAN,
INC.
CONDENSED
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR
THE SIX MONTHS ENDED JUNE 30, 2023 AND 2022
(UNAUDITED)
| | |
June 30, 2023 | | |
June 30, 2022 | |
| | |
Six Months Ended | |
| | |
June
30, 2023 | | |
June
30, 2022 | |
| CASH FLOWS FROM OPERATING ACTIVITIES: | |
| | | |
| | |
| Net loss | |
$ | (2,229,959 | ) | |
$ | (1,597,426 | ) |
| Adjustments to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Debt discount amortization, incl. issuance of warrants | |
| 348,637 | | |
| 132,759 | |
| Amortization | |
| 2,702 | | |
| 1,822 | |
| Stock-based compensation | |
| 18,710 | | |
| 69,123 | |
| Interest paid for note conversion | |
| 7,306 | | |
| — | |
| Changes in operating assets and liabilities: | |
| | | |
| | |
| Accounts payable and accrued expenses | |
| (142,682 | ) | |
| (67,481 | ) |
| Accounts payable related party | |
| 1,655,113 | | |
| 531,000 | |
| Net cash used in operating activities | |
| (340,173 | ) | |
| (930,203 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM INVESTING ACTIVITIES: | |
| | | |
| | |
| Investment in intangibles | |
| (25,047 | ) | |
| (22,438 | ) |
| Net cash used in investing activities | |
| (25,047 | ) | |
| (22,438 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | |
| | | |
| | |
| Proceeds from stock sales | |
| 115,000 | | |
| — | |
| Proceeds from issuance of convertible notes payable | |
| — | | |
| 1,380,960 | |
| Net cash provided by financing activities | |
| 115,000 | | |
| 1,380,960 | |
| | |
| | | |
| | |
| Net increase in cash | |
| (250,220 | ) | |
| 428,319 | |
| Cash, beginning of period | |
| 295,401 | | |
| 72,358 | |
| Cash, end of period | |
$ | 45,181 | | |
$ | 500,677 | |
| | |
| | | |
| | |
| SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION | |
| | | |
| | |
| Interest paid | |
$ | 52,425 | | |
$ | 69,900 | |
| Income taxes paid | |
| — | | |
| — | |
| NON-CASH INVESTING & FINANCING ACTIVITIES | |
| | | |
| | |
| Issuance of warrants | |
| 348,637 | | |
| 42,250 | |
| Debt discount on convertible note | |
| — | | |
| 90,509 | |
| Common shares issued for the conversion of principal and accrued interest | |
| 172,306 | | |
| — | |
See
the accompanying notes to these unaudited condensed consolidated financial statements
BIOXYTRAN,
INC.
NOTES
TO THE CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
FOR
THE THREE AND SIX MONTHS ENDED JUNE 30, 2023 AND 2022
(UNAUDITED)
NOTE
1 – BACKGROUND AND ORGANIZATION
Business
Operations
Bioxytran,
Inc. (the “Company”) is a clinical stage pharmaceutical company focused on the development, manufacture and commercialization
of therapeutic drugs designed to address hypoxia in humans, which is a lack of oxygen to tissues, in a safe and efficient manner.
Our
Subsidiary, Pharmalectin, Inc. (the “Subsidiary”) is a clinical stage pharmaceutical company focused on the development,
manufacture and commercialization of therapeutic drugs designed to address conditions related to Covid-19.
Our
Foreign Subsidiary, Pharmalectin (BVI), Inc. (the “Foreign Subsidiary”) is the owner and custodian of the Company’s
Copyrights, Trade Marks and Patents.
Our
subsidiary, Pharmalectin India Pvt Ltd. (“Pharmalectin India”) is managing the Company’s local clinical research and
trials, and holds the local rights to commercialization.
Organization
Bioxytran,
Inc. was organized on October 5, 2017 as a Delaware corporation, with a taxing structure for U.S. federal and state income tax as a C-Corporation
with 95,000,000 authorized common shares with a par value of $0.0001, and 5,000,000 Preferred shares with a par value of $0.0001. On
September 21, 2018, the Company went under a reorganization in the form of a reverse merger and is currently registered as a Nevada corporation
with a taxing structure for U.S. federal and state income tax as a C-Corporation with 300,000,000 authorized common shares with a par
value of $0.001, and 50,000,000 Preferred shares with a par value of $0.001.
Pharmalectin
was organized on October 5, 2017 as a Delaware corporation, with a taxing structure for U.S. federal and state income tax as a C-Corporation
with 95,000,000 authorized Common shares with a par value of $0.0001, and 5,000,000 Preferred shares with a par value of $0.0001. The
Subsidiary was founded under the name of Bioxytran “Bioxytran (DE)”. On April 29, 2020, the name was changed to Pharmalectin,
Inc. There are currently 30,000,000 issued and 19,650,000 outstanding shares; 15,000,000 Common shares are held by Bioxytran and 4,650,000
Common shares are held by an affiliate. An additional 4,500,000 options are also held by an affiliate. The option agreement includes
provisions for dilutive issuance and cash-less exercise. The beneficial ownership of the affiliate are Mike Sheikh, Ola Soderquist and
David Platt.
Pharmalectin
BVI was organized on March 17, 2021 as a British Virgin Islands (BVI) Business Corporation with a BVI corporate taxing structure with
50,000 authorized shares with a par value of $1.00. There are currently 50,000 outstanding shares held by the Company.
Pharmalectin
India Pvt Ltd. (“Pharmalectin India”) was organized on August 30, 2022 as an Indian Business Corporation with its principal
place of business in Hyderabad, Telangana, India, with
50,000 authorized shares with a par value of $0.12 (₹10). There are currently 41,020
outstanding shares whereof 41,000 (99.95%) are held by the Company.
Basis
of Presentation
The
accompanying unaudited condensed consolidated financial statements of the Company have been prepared in accordance with the rules and
regulations of the Securities and Exchange Commission (the “SEC”), including the instructions to Form 10-Q and Regulation
S-X. Certain information and note disclosures normally included in financial statements prepared in accordance with generally accepted
accounting principles in the United States of America (“U.S. GAAP”), have been condensed or omitted from these statements
pursuant to such rules and regulations and, accordingly, they do not include all the information and notes necessary for comprehensive
financial statements and should be read in conjunction with our audited consolidated financial statements.
While
the information presented in the accompanying financial statements is unaudited, it includes all adjustments which are, in the opinion
of the management, necessary to present fairly the financial position, results of operations and cash flows for the periods presented
in accordance with the accounting principles generally accepted in the United States of America (“US GAAP”). In the opinion
of management, all adjustments considered necessary for a fair presentation of the results of operations and financial position have
been included and all such adjustments are statements prepared in accordance with US GAAP have been condensed or omitted. These financial
statements should be read in conjunction with the Company’s December 31, 2022 audited financial statements and notes that can be
expected for the year ending December 31, 2022.
Principles
of Consolidation
The
accompanying unaudited condensed consolidated financial statements include the accounts of Bioxytran, Inc. a Nevada Corporation, its
majority owned subsidiary, Pharmalectin, Inc. of Delaware (collectively, the “Company”), as well as its wholly owned subsidiary,
Pharmalectin (BVI), Inc of British Virgin Islands. All intercompany accounts have been eliminated upon consolidation.
NOTE
2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
A
summary of the significant accounting policies applied in the preparation of the accompanying financial statements follows.
Cash
For
purposes of the Statement of Cash Flows, the Company considers all highly liquid debt instruments purchased with an original maturity
date of three months or less to be cash equivalents.
Use
of Estimates
The
preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported
amounts of assets and liabilities, disclosure of contingent assets and liabilities at the date of the financial statements and the reported
amount of expenses during the reporting period. Significant estimates include the fair value of the Company’s stock, stock-based
compensation, valuation of warrants, valuations in connection with convertible notes and the valuation allowance related to deferred
tax assets. Actual results may differ from these estimates.
Net
Loss per Common Share, basic and diluted
The
Company computes earnings (loss) per share under Accounting Standards Codification subtopic 260-10, Earnings Per Share (“ASC 260-10”).
Net loss per common share is computed by dividing net loss by the weighted average number of shares of Common Stock outstanding during
the year. Diluted earnings per share, if presented, would include the dilution that would occur upon the exercise or conversion of all
potentially dilutive securities into Common Stock using the “treasury stock” and/or “if converted” methods as
applicable.
At
June 30, 2023, we would, based on the market price of $0.19/share, be obligated to issue approximately 16,781,138 shares of Common Stock
upon conversion of the currently outstanding convertible notes (the “New Notes”) and 1,342,030 shares upon exercise of the
warrants and 428,000 shares upon exercise of outstanding options. For the New Notes, the shares total is based on $2,181,548 of currently
outstanding principal, and unpaid interest.
The
2021 notes (the “New Notes”), have an average interest rate of 8% and are convertible at the lower of (i) a fixed price of
$0.13, or (ii) 85% of the closing price of any Qualified Financing, which consist of any fundraising receiving gross proceeds of not
less than $500,000.
Stock
Based Compensation
The
Company measures the cost of services received from employees and non-employees in exchange for an award of equity instruments based
on the fair value of the award on the grant date pursuant ASC 718. Stock-based compensation expense is recorded by the Company over the
requisite service period, or vesting period, in the same expense classifications in the statements of operations, as if such amounts
were paid in cash.
Accounting
for subsidiary stock transactions
The
Company accounts for subsidiary stock transactions in accordance with Opinions of the Accounting Principles Board 09 (APBO No. 9). In
paragraph 28, this pronouncement excluded all adjustments from transactions in a company’s own stock “. . . from the determination
of net income or the results of operations under all circumstances.”.
Research
and Development
The
Company accounts for research and development costs in accordance with Accounting Standards Codification subtopic 730-10, Research and
Development (“ASC 730-10”). Under ASC 730-10, all research and development costs must be charged to expense as incurred.
Accordingly, internal research and development costs are expensed as incurred. Third-party research and development costs are expensed
when the contracted work has been performed or as milestone results have been achieved as defined under the applicable agreement. Company-sponsored
research and development costs related to both present and future products are expensed in the period incurred. For the six months ended
June 30, 2023 the Company incurred $288,642 in research and development expenses, while during the six months ended June 30, 2022 the
Company incurred $283,266.
Intangibles
– Goodwill and Other
Valuation
of intangibles are in accordance with ASC 350. Costs associated with the application and award of patents in the U.S. and various other
countries are capitalized and amortized on a straight-line basis over the term of the patents as determined at award date, which varies
depending on the pendency period of the application, generally approximating seventeen years. Capitalized patent costs, also referred
to as patent prosecution costs, include internal legal labor, professional legal fees, government filing fees and translation fees related
to expanding the Company’s patent portfolio. Costs associated with the maintenance and annuity fees of patents are accounted for
as prepaid assets at the time of payment and amortized over the shorter of the maintenance period or remaining life of the related patent.
Accrued
Expenses
As
part of the process of preparing our condensed consolidated financial statements, we are required to estimate accrued expenses. This
process involves identifying services that third parties have performed on our behalf and estimating the level of service performed and
the associated cost incurred on these services as at each balance sheet date in our consolidated financial statements. Examples of estimated
accrued expenses include professional service fees, such as those arising from the services of attorneys and accountants and accrued
payroll expenses. In connection with these service fees, our estimates are most affected by our understanding of the status and timing
of services provided relative to the actual services incurred by the service providers. In the event that we do not identify certain
costs that have been incurred or we under- or over-estimate the level of services or costs of such services, our reported expenses for
a reporting period could be understated or overstated. The date on which certain services commence, the level of services performed on
or before a given date, and the cost of services are often subject to our judgment. We make these judgments based upon the facts and
circumstances known to us in accordance with accounting principles generally accepted in the U.S.
Warrants
The
Company has issued Common Stock warrants in connection with the execution of certain equity and debt financings. The fair value of warrants
is determined using the Black-Scholes option-pricing model using assumptions regarding volatility of our common share price, remaining
life of the warrant, and risk-free interest rates at each period end.
Fair
Value
Accounting
Standards Codification subtopic 825-10, Financial Instruments (“ASC 825-10”) requires disclosure of the fair value of certain
financial instruments. The carrying value of cash and cash equivalents, accounts payable and accrued liabilities, and short-term borrowings,
as reflected in the balance sheets, approximate fair value because of the short-term maturity of these instruments. All other significant
financial assets, financial liabilities and equity instruments of the Company are either recognized or disclosed in the financial statements
together with other information relevant for making a reasonable assessment of future cash flows, interest rate risk and credit risk.
Where practicable the fair values of financial assets and financial liabilities have been determined and disclosed; otherwise only available
information pertinent to fair value has been disclosed.
The
Company follows Accounting Standards Codification subtopic 820-10, Fair Value Measurements and Disclosures (“ASC 820-10”)
and Accounting Standards Codification subtopic 825-10, Financial Instruments (“ASC 825-10”), which permits entities to choose
to measure many financial instruments and certain other items at fair value.
Recent
Accounting Pronouncements
In
August 2020, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) 2020-06,
Debt — Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging — Contracts in Entity’s
Own Equity (Subtopic 815-40) (“ASU 2020-06”) to simplify accounting for certain financial instruments. ASU 2020-06 eliminates
the current models that require separation of beneficial conversion and cash conversion features from convertible instruments and simplifies
the derivative scope exception guidance pertaining to equity classification of contracts in an entity’s own equity. The new standard
also introduces additional disclosures for convertible debt and freestanding instruments that are indexed to and settled in an entity’s
own equity. ASU 2020-06 amends the diluted earnings per share guidance, including the requirement to use the if-converted method for
all convertible instruments. ASU 2020-06 is effective January 1, 2022 and should be applied on a full or modified retrospective basis,
with early adoption permitted beginning on January 1, 2021. The Company adopted ASU 2020-06 effective January 1, 2021. The adoption of
ASU 2020-06 did not have an impact on the Company’s financial statements.
Management
does not believe that any recently issued, but not yet effective, accounting standards, if currently adopted, would have a material effect
on the Company’s unaudited condensed interim financial statements.
NOTE
3 – GOING CONCERN AND MANAGEMENT’S LIQUIDITY PLANS
As
at June 30, 2023, the Company had cash of $45,181 and a negative working capital of $2,724,061. The Company has not yet generated any
revenues, and has incurred cumulative net losses of $13,413,901. These conditions raise substantial doubt about the Company’s ability
to continue as a going concern.
During
the six months ended June 30, 2023, the Company raised a net of $115,000 in cash proceeds from equity. During the same period in 2022,
the Company raised a net of $1,380,960 in cash proceeds from the issuance of convertible notes.
The
Company intends to raise additional capital through private placements of debt and equity securities, but there can be no assurance that
these funds will be available on terms acceptable to the Company, or will be sufficient to enable the Company to fully complete its development
activities or sustain operations. If the Company is unable to raise sufficient additional funds, it will have to develop and implement
a plan to further extend payables, reduce overhead, or scale back its current business plan until sufficient additional capital is raised
to support further operations. There can be no assurance that such a plan will be successful.
Accordingly,
the accompanying unaudited condensed consolidated financial statements have been prepared in conformity with U.S. GAAP, which contemplates
continuation of the Company as a going concern and the realization of assets and satisfaction of liabilities in the normal course of
business. The carrying amounts of assets and liabilities presented in the unaudited condensed consolidated financial statements do not
necessarily purport to represent realizable or settlement values. The unaudited condensed consolidated financial statements do not include
any adjustment that might result from the outcome of this uncertainty.
NOTE
4 - RELATED PARTY TRANSACTIONS
The
Company hold License Agreements (the “License/s” or “Agreement/s”) for a medical device (license obtained in
2019) and a compound (license obtained in 2021), with two affiliated companies where in the officers of the Company hold a majority interest.
The products were developed prior to the establishment of Bioxytran. The yearly maintenance fee for each license amount to $5,000. During
the six months ended June 30, 2023 the affiliates were paid $5,000 each. During the same period in 2022, there was $25,720 in transactions
with affiliates as the Company also reimbursed the affiliates for the legal and administrative costs surrounding the establishment of
the Licenses.
NOTE
5 - INTANGIBLES
Intangible
assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not
be recoverable. No impairment charges were recorded for the 6 months ended June 30, 2023 and the year ended December 31, 2022.
Amortization
of capitalized patent costs associated with the application and award of patents in the U.S. and various other countries are capitalized
and amortized on a straight-line basis over the term of the patents as determined at the award date, which varies depending on the pendency
period of the application, generally approximating twenty years.
SCHEDULE
OF INTANGIBLES
| | |
Estimated Life (years) | | |
June 30, 2023 | | |
December 31, 2022 | |
| Capitalized patent costs | |
| 20 | | |
$ | 104,226 | | |
$ | 79,179 | |
| Accumulated amortization | |
| | | |
| (6,346 | ) | |
| 3,644 | |
| | |
| | | |
| | | |
| | |
| Intangible assets, net | |
| | | |
$ | 97,880 | | |
$ | 75,535 | |
NOTE
6 – ACCOUNTS PAYABLES AND ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
On
June 30, 2023, there was $200,500 in accounts payable to related parties in the form of payroll and accrued expenses and $5,700 in un-issued
shares liability related party. On December 31, 2022 there was $709,727 in accounts payable to related parties and $38,400 in un-issued
shares liability related party.
The
following table represents the major components of accounts payables and accrued expenses and other current liabilities at June 30, 2023
and at December 31, 2022:
SCHEDULE
OF ACCOUNTS PAYABLES AND ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
| | |
June 30, 2023 | | |
December 31, 2022 | |
| Accounts payable related party (1) | |
$ | 200,500 | | |
$ | 709,727 | |
| Professional fees | |
| 315,502 | | |
| 393,085 | |
| Interest | |
| 181,548 | | |
| 134,581 | |
| Payroll taxes | |
| 15,941 | | |
| 40,182 | |
| Pension/401K | |
| 48,125 | | |
| 180,557 | |
| Other | |
| 1,546 | | |
| 990 | |
| Un-issued share liability, related party (2) | |
| 5,700 | | |
| 38,400 | |
| Un-issued share liability, consultant | |
| 380 | | |
| 960 | |
| Convertible note payable | |
| 2,000,000 | | |
| 2,165,000 | |
| Total | |
$ | 2,769,242 | | |
$ | 3,663,482 | |
| (1) |
$71,000
to the CEO, $70,000 to the CFO and $54,500 and the CCO for two months of salary and expenses, there are also $5,000 owed to an affiliate.
At December 31, 2022 there were $286,900 to the CEO, $269,400 to the CFO and $153,427 and the CCO in salary and expenses. |
| (2) |
There
are currently 30,000 shares of Common Stock awarded but not issued to three Board Members in the first quarter of 2023. The total
fair market value at the time of the award was $5,700 |
NOTE
7 – CONVERTIBLE NOTES PAYABLE
Private
Placement, 2021 Notes
Around
April 29, 2021, we entered into four (4) Securities Purchase Agreements (the “2021 SPA’s”), under which we agreed to
sell convertible promissory notes (the “2021 Notes”), in an aggregate principal amount of $1,165,000 with 6% interest, whereof
$1,000,000 were contributed in form of cancellation of third-party notes.
At
any time after the issue date of the Notes, The Holders of the Notes, (the “2021 Holders”), have the option to convert all
or any part of the outstanding and unpaid principal amount and accrued and unpaid interest of the 2021 Notes into shares of our Common
Stock at the Conversion Price. The “Conversion Price” will be the lesser of (i) $.13 per share or (ii) 85% of the closing
price of Any Qualified Financing, which consists of any fundraising whereby the Company receives gross proceeds of not less than $500,000.
The
variable conversion rate component requires that the 2021 Notes to be valued at its stock redemption value (i.e., “if-converted”
value) pursuant to ASC 480, Distinguishing Liabilities from Equity, with the excess over the undiscounted face value being deemed a premium
to be added to the principal balance and accreted to additional paid-in capital over the life of the 2021 Notes. No such recording of
a premium was required as the discounted “if-converted” rate of $0.13 per share, was identical to fair market value of the
Company’s stock on the 2021 Notes date of issuance.
The
2021 Holders are limited to holding a total of 4.99% of our issued and outstanding Common Stock at any one time.
The
maturity on one note was negotiated to August 31, 2023, while the maturity of the three remaining notes were negotiated to April 30,
2024, and an increase of the interest rate to 10%. The principal and interest for two of these latter notes were converted into 1,325,430
shares of Common Stock on May 17 and on June 26, 2023 for a total value of 172,306.
SCHEDULE
OF CONVERTIBLE CONVERSION OF ACCRUED INTEREST AND PRINCIPAL
| Name |
|
Principal Converted | | |
Accrued interest converted | | |
No. of shares issued | |
| Private Placement, 2021 Notes issued to Officers (1) |
|
$ | 165,000 | | |
$ | 7,306 | | |
| 1,325,430 | |
Convertible
notes payable and interest payable consist of the following at June 30, 2023, and December 31, 2022:
SCHEDULE
OF CONVERTIBLE NOTES PAYABLE
| | |
June
30, 2023 | | |
December
31, 2022 | |
| Principal
balance (1), (2) | |
$ | 2,000,000 | | |
$ | 2,165,000 | |
| Interest
Payable | |
| 181,548 | | |
| 134,581 | |
| Outstanding,
net of debt discount and premium | |
$ | 2,181,548 | | |
$ | 2,299,581 | |
| (1) |
Net
cash received for these notes were $1,045,150, after a Debt Discount of $119,850 was paid to the sole Placement Agent: WallachBeth
Capital, LLC (Member FINRA / SIPC). $165,000 of the outstanding principal was converted into shares of Common Stock on May 17 and
on June 26, 2023. |
| (2) |
$2
million of principal, accrued interest and default penalties for notes issued prior to 2021, where settled by a third party in exchange
for us issuing to them a note in the amount of $1 million. |
There
can be no assurance that there will be any funds available to pay of the 2021 Notes. If we fail to obtain such additional financing on
a timely basis, the 2021 Holders may convert the 2021 Notes and sell the underlying shares, which may result in significant dilution
to shareholders due to the conversion discount, as well as a significant decrease in our stock price.
Private
Placement, 2022 Notes converted into Common Stock
In
January, 2022, we entered into thirty-four (34) Securities Purchase Agreements (the “2022 SPA’s”), with accredited
investors, under which we agreed to sell the Notes, in an aggregate principal amount of $1,467,000 with 6% interest (the “2022
Notes”) to the holders of the 2022 Notes (the “2022 Holders”).
At
any time after the issue date of the 2022 Notes the 2022 Holders have the option to convert all or any part of the outstanding and unpaid
principal amount and accrued and unpaid interest of the Notes into shares of our Common Stock at the Conversion Price. The “Conversion
Price” is set to $0.25 per share.
The
2022 Holders are limited to holding a total of 4.99% of our issued and outstanding Common Stock at any one time. The Common Stock underlying
the 2022 Notes, when issued, bear a restrictive legend and are currently eligible for resale under Rule 144.
The
notes principal and accrued interest were fully converted into 6,081,484 shares of Common Stock on August 31, 2022.
| Name |
|
Principal Converted | | |
Accrued interest converted | | |
No. of shares issued | |
| Private Placement, 2022 Notes (1) |
|
$ | 1,467,000 | | |
$ | 53,371 | | |
| 6,081,484 | |
| |
|
$ | 1,467,000 | | |
$ | 53,371 | | |
| 6,081,484 | |
| (1) |
Net
cash received for these notes were $1,380,960, after a Debt Discount of $86,040 was paid to the sole Placement Agent: WallachBeth
Capital, LLC (Member FINRA / SIPC). |
NOTE
8 – STOCKHOLDERS’ EQUITY
The
Company is authorized to issue 300,000,000 shares of Common Stock, and 50,000,000 shares of Preferred Stock.
Preferred
stock
As
of June 30, 2023 and at December 31, 2022, no Preferred shares have been designated or issued.
Common
Stock
There
were no shares issued in six months ended June 30, 2022.
As
at January 1, 2023 there were 123,252,235 shares of common stock issued and outstanding.
On
January 4, 2023 the Company issued 93,750 shares of Common Stock against $30,000, or $0.32/share, shown as stock subscription in the
December 31, 2022 stockholders’ equity statement.
On
February 10, 2023 the Company issued 156,250 shares of Common Stock against $50,000, or $0.32/share
On
April 14, 2023 the Company issued 137,656 shares of Common Stock were against 3rd party
supplier invoices amounting to $44,050, or $0.32/share
On
April 14, 2023 the Company issued 6,763,562 shares of Common Stock to offset an affiliate against invoices paid on behalf of the Company
and accrued salaries to our Officers, for a total value of $2,164,340., or $0.32/share
On
April 18, 2023 the Company issued 78,125 shares of Common Stock against $25,000, or $0.32/share
On
May 15, 2023 the Company issued 114,286 shares of Common Stock against $40,000, or $0.32/share
On
May 17, 2023 the Company issued 522,138 shares of Common Stock in a conversion of a note for a
value of $67,878 in principal and interest, or $0.13/share
On
June 26, 2023 the Company issued 803,292 shares of Common Stock in a conversion of a note for a
value of $104,428 in principal and interest, or $0.13/share
For
the six months ended June 30, 2022, a net of 114,000 shares of Common Stock were awarded under the 2021 Stock Plan for a total value
of $51,990, or at an average cost of $0.46 per share.
As
at June 30, 2023, the Company have 132,035,294 shares of Common Stock issued and outstanding and at December 31, 2022 the Company had
123,252,235 shares of Common Stock issued and outstanding.
Common
Stock Warrants
For
the six months ended June 30, 2023 the Company issued 800,000 5-year warrants exercisable at $0.20/share, in connection with the refinancing
of the convertible notes, valued at $0.436, based on Black and Scholes Option Pricing Model, for a total value of $348,637. For the six
months ended June 30, 2022, the Company issued 264,060 5-year warrants exercisable at $0.25/share, valued at $0.16, based on Black and
Scholes Option Pricing Model, for a total value of $42,250.
The
fair value of stock warrants granted for the 6 months ended June 30, 2022 was calculated with the following assumptions:
SCHEDULE OF STOCK WARRANTS VALUATION ASSUMPTIONS
| | |
June 30, 2023 | | |
June 30, 2022 | |
| Risk-free interest rate | |
| 3.97 | % | |
| 1.53 | % |
| Expected dividend yield | |
| 0 | % | |
| 0 | % |
| Volatility factor (monthly) | |
| 147.58 | % | |
| 169.27 | % |
| Expected life of warrant | |
| 5 years | | |
| 5 years | |
The
following table summarizes the Company’s common stock warrant activity for the 6 months ended June 30, 2023 and 2022:
SCHEDULE OF WARRANT ACTIVITY
| |
|
Number
of Warrants* |
|
|
Weighted
Average Exercise Price |
|
|
Weighted-
Average Remaining Expected Term |
|
| Outstanding
as at January 1, 2022 |
|
|
272,000 |
|
|
$ |
2.00 |
|
|
|
2.9 |
|
| Granted |
|
|
264,030 |
|
|
|
0.26 |
|
|
|
5.0 |
|
| Exercised |
|
|
— |
|
|
|
— |
|
|
|
— |
|
| Forfeited/Canceled |
|
|
— |
|
|
|
— |
|
|
|
— |
|
| Outstanding
as at June 30, 2022 |
|
|
536,030 |
|
|
|
1.14 |
|
|
|
3.7 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| Outstanding
as at January 1, 2023 |
|
|
542,030 |
|
|
$ |
0.42 |
|
|
|
4.1 |
|
| Granted |
|
|
800,000 |
|
|
|
0.20 |
|
|
|
5.0 |
|
| Exercised |
|
|
— |
|
|
|
— |
|
|
|
— |
|
| Forfeited/Canceled |
|
|
— |
|
|
|
— |
|
|
|
— |
|
| Outstanding
as at June 30, 2023 |
|
|
1,342,030 |
|
|
$ |
0.29 |
|
|
|
4.4 |
|
| * |
The
warrant agreements issued in 2019 for a total of 50,000 warrants include provisions for dilutive issuance and cash-less exercise.
If exercised at December 31, 2022 the provisions would have resulted in an issuance of 1,130,114 shares at an average conversion
price of $0.09, or 1,050,114 shares in a cash-less exercise. In order to mitigate the Company’s risk an administrative hold
has been placed on one shareholder’s stock in the event of future exercise. |
The
following table summarizes information about stock warrants that are vested or expected to vest at June 30, 2023:
SCHEDULE OF WARRANT OUTSTANDING AND EXERCISABLE WARRANTS
| | | |
Warrants Outstanding | | |
| | |
| | |
Exercisable Warrants | | |
| |
| Number of Warrants | | |
Weighted Average Exercise Price Per Share | | |
Weighted Average Remaining Contractual Life (Years) | | |
Aggregate Intrinsic Value | | |
Number of Warrants | | |
Weighted Average Exercise Price Per Share | | |
Weighted Average Remaining Contractual Life (Years) | | |
Aggregate Intrinsic Value | |
| | 800,000 | | |
$ | 0.20 | | |
| 4.9 | | |
$ | — | | |
| 800,000 | | |
$ | 0.20 | | |
| 4.9 | | |
$ | — | |
| | 492,030 | | |
| 0.26 | | |
| 3.8 | | |
| — | | |
| 492,030 | | |
| 0.26 | | |
| 4.1 | | |
| — | |
| | 50,000 | | |
| 2.00 | | |
| 1.3 | | |
| — | | |
| 50,000 | | |
| 2.00 | | |
| 1.3 | | |
| — | |
| | 1,342,030 | | |
$ | 0.29 | | |
| 4.4 | | |
$ | — | | |
| 1,342,030 | | |
$ | 0.29 | | |
| 4.4 | | |
$ | — | |
The
following table sets forth the status of the Company’s non-vested warrants as at June 30, 2023 and 2022:
SCHEDULE
OF NON-VESTED WARRANTS
| |
|
Number
of Warrants |
|
|
Weighted-Average
Grant-Date
Fair Value |
|
| Non-vested
as at January 1, 2022 |
|
|
— |
|
|
|
— |
|
| Granted |
|
|
264,030 |
|
|
|
0.25 |
|
| Forfeited |
|
|
— |
|
|
|
— |
|
| Vested |
|
|
— |
|
|
|
— |
|
| Non-vested
as at June 30, 2022 |
|
|
— |
|
|
$ |
— |
|
| |
|
|
|
|
|
|
|
|
| Non-vested
as at January 1, 2023 |
|
|
— |
|
|
$ |
— |
|
| Granted |
|
|
800,000 |
|
|
|
0.20 |
|
| Forfeited |
|
|
— |
|
|
|
— |
|
| Vested |
|
|
— |
|
|
|
— |
|
| Non-vested
as at June 30, 2023 |
|
|
— |
|
|
$ |
— |
|
NOTE
9 – STOCK OPTION PLAN AND STOCK-BASED COMPENSATION
On
January 19, 2021, the Board of Directors adopted the “2021 Stock Plan” (the “2021 Plan”) under which the Company
may grant Options to Purchase Stock, Stock Awards or Stock Appreciation Rights in an amount up to 15% of the number of issued and outstanding
shares of the Company’s Common Stock, automatically adjusted on January 1 each year. Under the terms of the 2021 Stock Plan, the
Board of Directors shall specify the exercise price and vesting period of each stock option on the grant date. Vesting of the options
is typically immediate and the options typically expire in five years. Stock Awards may be directly issued under the Plan (without any
intervening options). Stock Awards may be issued which are fully and immediately vested upon issuance. As at June 30, 2023, 90,000 options
and 700,000 shares have been awarded from the 2021 Plan.
Shares
Awarded and Issued under the 2021 Plan:
On
April 19, 2023 the Company issued 110,000 shares, with an average fair market value of $0.46/share at the time of award, to four members
of the Board of Directors as compensation for their participations of Board and Committee meetings in the fourth quarter of 2022 and
in the first quarter of 2023, for a total of $50,200.
On
April 19, 2023 the Company granted 4,000 shares with an average fair market value of $0.45/share to a Scientific Advisory Board Member
for his contribution in the fourth quarter of 2022 and in the first quarter of 2023, for a total of $1,790.
| | |
Number of Shares | | |
Fair Value per Share | | |
Weighted Average Market Value per Share | |
| Shares Issued as at January 1, 2022 | |
| 4,505,709 | | |
$ | 0.001 – 0.55 | | |
$ | 0.10 | |
| Shares Issued | |
| — | | |
| — | | |
| — | |
| Shares Issued as of June 30, 2022 | |
| 4,505,709 | | |
$ | 0.001
– 0.55 | | |
$ | 0.13 | |
| | |
| | | |
| | | |
| | |
| Shares Issued as at January 1, 2023 | |
| 5,139,709 | | |
$ | 0.001
– 0.55 | | |
$ | 0.088 | |
| Shares Issued | |
| 114,000 | | |
| 0.415
– 0.48 | | |
| 0.462 | |
| Shares Issued as of June 30, 2023 | |
| 5,263,709 | | |
$ | 0.001
– 0.55 | | |
$ | 0.096 | |
For
the six months ended June 30, 2023, the Company recorded stock-based compensation expense of $18,710 in connection with share-based payment
awards. For the six months ended June 30, 2022, the Company recorded stock-based compensation expense of $69,123.
Shares
awarded, but not yet issued, under the 2021 Stock Plan:
On
January 10, 2022 the Company granted 40,000 shares of Common Stock to four Board Members in reward of their attendance at Board and Committee
meetings during the fourth quarter of 2021. The total fair market value at the time of the award was $6,400, or $0.16/share. The shares
were issued in the third quarter of 2022.
On
February 18, 2022 the Company granted 100,000 shares of Common Stock to two Consultants in reward of their assistance for the product
development and our clinical trials in India. The total fair market value at the time of the award was $16,000, or $0.16/share. The shares
were issued in the third quarter of 2022.
On
April 1, 2022 the Company granted 10,000 shares to a Medical Advisory Board Member for her contribution to the Company during the first
quarter of 2022. The total fair market value at the time of the award was $1,730, or $0.173/share. The shares were issued in the third
quarter of 2022.
On
April 1, 2022 the Company granted 70,000 shares to four Board Members in reward of their attendance at Board and Committee meetings during
the first quarter of 2022. The total fair market value at the time of the award was $12,110, or $0.173/share. The shares were issued
in the third quarter of 2022.
On
April 11, 2022 the Company granted 250,000 shares to three Consultants for the management of our clinical trials in India. The total
fair market value at the time of the award was $43,250, or $0.173/share. The shares were issued in the third quarter of 2022.
On
June 30, 2023 the Company granted 30,000 shares of Common Stock to three Board Members in reward of their attendance at Board and Committee
meetings during the second quarter of 2023. The total fair market value at the time of the award was $5,700, or $0.19/share.
On
June 30, 2023 the Company granted 2,000 shares to a Scientific Advisory Board Member for his contribution to the Company during the second
quarter of 2023. The total fair market value at the time of the award was $380, or $0.19/share.
Stock
options granted and vested 2021 Plan:
There
were no stock options granted the six months ended June 30, 2023 and 2022. But, 96,000 stock options was forfeited in the six months
ended June 30, 2023 and 48,000 stock options was forfeited in the six months ended June 30, 2022.
The
following table summarizes the Company’s stock option activity for the six months ended June 30, 2023, and 2022:
| | |
Number of Options | | |
Exercise
Price per Share | | |
Weighted Average Exercise Price per Share | |
| Outstanding as of January 1, 2022 | |
| 668,000 | | |
$ | 0.001 – 1.21 | | |
$ | 0.55 | |
| Granted | |
| — | | |
| — | | |
| — | |
| Exercised | |
| — | | |
| — | | |
| — | |
| Options forfeited/cancelled | |
| (48,000 | ) | |
| 1.09 – 1.21 | | |
| 1.20 | |
| Outstanding as of June 30, 2022 | |
| 620,000 | | |
$ | 0.001
– 1.21 | | |
$ | 0.50 | |
| | |
| | | |
| | | |
| | |
| Outstanding as of January 1, 2023 | |
| 524,000 | | |
$ | 0.001
– 0.95 | | |
$ | 0.44 | |
| Granted | |
| — | | |
| — | | |
| — | |
| Exercised | |
| — | | |
| — | | |
| — | |
| Options forfeited/cancelled | |
| (96,000 | ) | |
| 0.001
– 0.20 | | |
| 0.01 | |
| Outstanding as of June 30, 2023 | |
| 428,000 | | |
$ | 0.001
– 0.95 | | |
$ | 0.52 | |
The
following table summarizes information about stock options that are vested or expected to vest at June 30, 2023:
SCHEDULE
OF STOCK OPTION VESTED
| |
|
|
|
|
|
Options
Outstanding |
|
|
|
|
|
|
|
|
Exercisable
Options |
|
|
|
|
| Exercise
Price |
|
|
Number
of Options |
|
|
Weighted
Average Exercise Price Per Share |
|
|
Weighted
Average Remaining Contractual Life (Years) |
|
|
Aggregate
Intrinsic Value |
|
|
Number
of Options |
|
|
Weighted
Average Exercise Price Per Share |
|
|
Weighted
Average Remaining Contractual Life (Years) |
|
|
Aggregate
Intrinsic Value |
|
| $ |
0.001 |
|
|
|
45,000 |
|
|
$ |
0.001 |
|
|
|
1.08 |
|
|
$ |
8,505 |
|
|
|
45,000 |
|
|
$ |
0.001 |
|
|
|
1.08 |
|
|
$ |
8,505 |
|
| |
0.05 |
|
|
|
3,000 |
|
|
|
0.05 |
|
|
|
0.25 |
|
|
|
420 |
|
|
|
3,000 |
|
|
|
0.05 |
|
|
|
0.25 |
|
|
|
420 |
|
| |
0.15 |
|
|
|
45,000 |
|
|
|
0.15 |
|
|
|
0.08 |
|
|
|
1,800 |
|
|
|
45,000 |
|
|
|
0.15 |
|
|
|
0.08 |
|
|
|
1,800 |
|
| |
0.18 |
|
|
|
45,000 |
|
|
|
0.18 |
|
|
|
0.33 |
|
|
|
450 |
|
|
|
45,000 |
|
|
|
0.18 |
|
|
|
0.33 |
|
|
|
450 |
|
| |
0.19 |
|
|
|
45,000 |
|
|
|
0.19 |
|
|
|
0.83 |
|
|
|
— |
|
|
|
45,000 |
|
|
|
0.19 |
|
|
|
0.83 |
|
|
|
— |
|
| |
0.20 |
|
|
|
45,000 |
|
|
|
0.20 |
|
|
|
0.59 |
|
|
|
— |
|
|
|
45,000 |
|
|
|
0.20 |
|
|
|
0.59 |
|
|
|
— |
|
| |
0.95 |
|
|
|
200,000 |
|
|
|
0.95 |
|
|
|
0.76 |
|
|
|
— |
|
|
|
200,000 |
|
|
|
0.95 |
|
|
|
0.76 |
|
|
|
— |
|
| $ |
0.001–0.95 |
|
|
|
428,000 |
|
|
$ |
0.52 |
|
|
|
0.66 |
|
|
$ |
11,175 |
|
|
|
428,000 |
|
|
$ |
0.52 |
|
|
|
0.66 |
|
|
$ |
11,175 |
|
The
weighted-average remaining estimated life for options exercisable at June 30, 2023 is 0.66 years.
The
aggregate intrinsic value for fully vested, exercisable options was $11,175 at June 30, 2023. The actual tax benefit realized from stock
option exercises for the six months ended at June 30, 2023 and 2022 was $0 as no options were exercised.
As
at June 30, 2023 the Company has 19,832,071 options or stock awards available for grant under the 2021 Plan.
NOTE
10 – NON-CONTROLLING INTEREST
SCHEDULE
OF NON CONTROLLING INTEREST
| | |
June 30, 2023 | | |
December 31, 2022 | |
| Net loss Subsidiary | |
$ | (121,063 | ) | |
$ | (817,151 | ) |
| Net loss attributable to the non-controlling interest | |
| 33,658 | | |
| 193,372 | |
| Net loss affecting Bioxytran | |
| (87,405 | ) | |
| (623,780 | ) |
| | |
| | | |
| | |
| Accumulated losses | |
| (3,574,358 | ) | |
| (3,594,287 | ) |
| Accumulated losses attributable to the non-controlling interest | |
| 785,236 | | |
| 751,578 | |
| Accumulated losses Bioxytran | |
| (2,789,122 | ) | |
| (2,842,709 | ) |
| | |
| | | |
| | |
| Net equity non-controlling interest | |
$ | (624,286 | ) | |
$ | (590,628 | ) |
As
at June 30, 2023 and at December 31, 2022 there are 30,000,000 issued and 19,650,000 outstanding shares; 15,000,000 Common shares are
held by Bioxytran and 4,650,000 Common shares are held by an affiliate. An additional 4,500,000 options are also held by an affiliate.
The option agreements include provisions for dilutive issuance and cash-less exercise.
NOTE
11 – COMMITMENTS AND CONTINGENCIES
Employment
contracts
Our
Executive Officers have entered into employment contracts and confidentiality, non-disclosure and assignment of invention agreements.
The most substantial provisions include;
| |
● |
Compensation
of three (3) times the employee’s annual salary upon the Termination Date and any target bonus earned, or if termination occurs
within 12 months of a change in control, then the terminated employee shall receive two (2) times the employee’s annual salary
and any target bonus earned. |
| |
|
|
| |
● |
Continued
coverage under any health, medical, dental or vision program or policy, in which they were eligible to participate at the time of
employment termination, for 12 months. |
| |
|
|
| |
● |
Provide
outplacement services through one or more outside firms of the employee’s choosing up to an aggregate of $50,000. |
There
are no other arrangements or plans in which we provide pension, retirement or similar benefits for any of Executive Officers or Directors.
Litigation
In
the normal course of business, the Company may be involved in legal proceedings, claims and assessments arising in the ordinary course
of business. Such matters are subject to many uncertainties, and outcomes are not predictable with assurance. Legal fees for such matters
are expensed as incurred and we accrue for adverse outcomes as they become probable and estimable.
NOTE
12 – SUBSEQUENT EVENTS
The
Company has evaluated events from June 30, 2023 through the date the financial statements were issued. and did not, other than what is
disclosed in the below, identify any further subsequent events requiring disclosure.
Stockholder’s
Equity
Conversion
of Notes into Common Stock
On
July 24, 2023, 500,000 shares of Common Stock were sold in a private placement for the amount of $100,000, or $0.20/share. These funds
should allow the company to start its Phase 2 clinical trial. The trial is intended to start during the month of August 2023 and last
for approximately 2 months.
Stock
options forfeited under the 2021 Stock Plan:
On
August 1, 2023, 45,000 options for a total value of $6,750 were forfeited through expiration and return to the stock plan.
Through
and including October 26, 2023, (the 25th day after the date of this Prospectus), all dealers effecting transactions
in the Common Stock, whether or not participating in this offering, may be required to deliver a Prospectus. This delivery requirement
is in addition to a dealer’s obligation to deliver a Prospectus when acting as an underwriter and with respect to an unsold allotment
or subscription.
18,578,982
Shares
Bioxytran,
Inc.
Common
Stock
P
R O S P E C T U S
Bioxytran (QB) (USOTC:BIXT)
Historical Stock Chart
From Mar 2024 to Apr 2024
Bioxytran (QB) (USOTC:BIXT)
Historical Stock Chart
From Apr 2023 to Apr 2024