Panbela Therapeutics, Inc. (Nasdaq: PBLA),
(“Panbela” or the “Company”), a clinical stage company developing
disruptive therapeutics for the treatment of patients with urgent
unmet medical needs, today announced the pricing of a public
offering of (i) 20,100,000 shares of its common stock (or
pre-funded warrants in lieu thereof) and (ii) warrants to purchase
up to 30,150,000 shares of its common stock (the “Public Warrants”)
at a purchase price of $0.30 per share and associated Public
Warrant. The Public Warrants will have an exercise price of $0.30
per share, are exercisable upon issuance, and will expire five
years following the date of issuance. The offering is expected to
close on or about October 4, 2022, subject to customary closing
conditions.
Roth Capital Partners, LLC is acting as lead placement agent,
Craig-Hallum Capital Group LLC and Maxim Group LLC are each acting
as co-placement agent of the offering.
Gross proceeds, before deducting placement agent fees and
commissions and offering expenses are expected to be approximately
$6 million. The Company intends to use the net proceeds from the
proposed offering for the continued clinical development of its
product candidates ivospemin (SBP-101) and eflornithine (CPP-1X),
working capital, business development and other general corporate
purposes, which may include repayment of debt.
The securities described above are being offered pursuant to a
registration statement on Form S-1 (File No. 333-267000), as
amended, that was declared effective by the U.S. Securities and
Exchange Commission (“SEC”), on September 29, 2022. Copies of the
accompanying prospectus relating to and describing the terms of the
offering may be obtained, when available, at the SEC’s website at
www.sec.gov or by contacting Roth Capital Partners, LLC, 888 San
Clemente Drive, Suite 400, Newport Beach, CA 92660 or by email at
rothecm@roth.com.
This press release does not and shall not constitute an offer to
sell or the solicitation of an offer to buy any securities, nor
shall there be any sale of these securities in any state or other
jurisdiction in which such offer, solicitation or sale would be
unlawful prior to registration or qualification under the
securities laws of any such state or other jurisdiction. Any offer,
if at all, will be made only by means of a prospectus, including a
prospectus supplement, forming a part of the effective registration
statement.
About our Pipeline
The pipeline consists of assets currently in clinical trials
with an initial focus on familial adenomatous polyposis (FAP),
first-line metastatic pancreatic cancer, neoadjuvant pancreatic
cancer, colorectal cancer prevention and ovarian cancer. The
combined development programs have a steady cadence of catalysts
with programs ranging from pre-clinical to registration
studies.
SBP-101 Ivospemin
Ivospemin is a proprietary polyamine analogue designed to induce
polyamine metabolic inhibition (PMI) by exploiting an observed high
affinity of the compound for pancreatic ductal adenocarcinoma and
other tumors. It has shown signals of tumor growth inhibition in
clinical studies of metastatic pancreatic cancer patients,
demonstrating a median overall survival (OS) of 14.6 months and an
objective response rate (ORR) of 48%, both exceeding what is
typical for the standard of care of gemcitabine + nab-paclitaxel
suggesting potential complementary activity with the existing
FDA-approved standard chemotherapy regimen. In data evaluated from
clinical studies to date, ivospemin has not shown exacerbation of
bone marrow suppression and peripheral neuropathy, which can be
chemotherapy-related adverse events. Serious visual adverse events
have been evaluated and patients with a history of retinopathy or
at risk of retinal detachment will be excluded from future SBP-101
studies. The safety data and PMI profile observed in the previous
Panbela-sponsored clinical trials provide support for continued
evaluation of ivospemin in the ASPIRE trial. For more information,
please
visit https://clinicaltrials.gov/ct2/show/NCT03412799 .
Flynpovi ™
Flynpovi is a combination of CPP-1X (eflornithine) and sulindac
with a dual mechanism inhibiting polyamine synthesis and increase
polyamine export and catabolism. In a Phase 3 clinical trial in
patients with sporadic large bowel polyps, the combination
prevented > 90% subsequent pre-cancerous sporadic adenomas
versus placebo. Focusing on FAP patients with lower
gastrointestinal tract anatomy in the recent Phase 3 trial
comparing Flynpovi to single agent eflornithine and single agent
sulindac, FAP patients with lower GI anatomy (patients with an
intact colon, retained rectum or surgical pouch), Flynpovi showed
statistically significant benefit compared to both single agents
(p≤0.02) in delaying surgical events in the lower GI for up to four
years. The safety profile for Flynpovi did not significantly differ
from the single agents and supports the continued evaluation of
Flynpovi for FAP.
CPP-1X
CPP-1X (eflornithine) is being developed as a single agent
tablet or high dose power sachet for several indications including
prevention of gastric cancer, treatment of neuroblastoma and recent
onset Type 1 diabetes. Preclinical studies as well as Phase 1 or
Phase 2 investigator-initiated trials suggest that CPP-1X treatment
may be well-tolerated and has potential activity.
About Panbela
Panbela Therapeutics, Inc. is a clinical-stage biopharmaceutical
company developing disruptive therapeutics for patients with urgent
unmet medical needs. Panbela’s lead assets are Ivospemin (SBP-101)
and Flynpovi. Further information can be found
at www.panbela.com. Panbela’s common
stock is listed on The Nasdaq Stock Market LLC under the symbol
“PBLA”.
Cautionary Statement Regarding Forward-Looking
Statements
This press release contains “forward-looking
statements,” including within the meaning of the Private Securities
Litigation Reform Act of 1995. Forward-looking statements can be
identified by words such as: “anticipate,” “design,” “expect,”
“intend,” “may,” “plan,” “scheduled,” and “will.” Examples of
forward-looking statements include statements we make regarding
results of collaborations with third parties and future studies.
All statements other than statements of historical fact are
statements that should be deemed forward-looking
statements. Forward-looking statements are neither historical
facts nor assurances of future performance. Instead, they are
based only on our current beliefs, expectations, and assumptions
regarding the future of our business, future plans and
strategies, projections, anticipated events and trends, the economy
and other future conditions. Because forward-looking
statements relate to the future, they are subject to inherent
uncertainties, risks and changes in circumstances that are
difficult to predict and many of which are outside of our
control. Our actual results and financial condition may differ
materially and adversely from the forward-looking
statements. Therefore, you should not rely on any of these
forward-looking statements. Important factors that could cause
our actual results and financial condition to differ materially
from those indicated in the forward-looking statements include,
among others, the following: (i) our ability to obtain
additional funding to execute our business and clinical development
plans; (ii) progress and success of our clinical development
program; (iii) the impact of the current COVID-19 pandemic on our
ability to conduct our clinical trials; (iv) our ability to
demonstrate the safety and effectiveness of our product candidates:
ivospemin(SBP-101) and eflornithine (CPP-1X) (v) our reliance on a
third party for the execution of the registration trial for our
product candidate Flynpovi; (vi) our ability to obtain regulatory
approvals for our product candidates, SBP-101 and CPP-1X in the
United States, the European Union or other international markets;
(vii) the market acceptance and level of future sales of our
product candidates, SBP-101 and CPP-1X; (viii) the cost and delays
in product development that may result from changes in regulatory
oversight applicable to our product candidates, SBP-101 and CPP-1X;
(ix) the rate of progress in establishing reimbursement
arrangements with third-party payors; (x) the effect of competing
technological and market developments; (xi) the costs involved in
filing and prosecuting patent applications and enforcing or
defending patent claims; and (xi) such other factors as
discussed in Part I, Item 1A under the caption “Risk Factors” in
our most recent Annual Report on Form 10-K, any additional risks
presented in our Quarterly Reports on Form 10-Q and our Current
Reports on Form 8-K. Any forward-looking statement made by us in
this press release is based on information currently available to
us and speaks only as of the date on which it is made. We
undertake no obligation to publicly update any forward-looking
statement or reasons why actual results would differ from those
anticipated in any such forward-looking statement, whether
written or oral, whether as a result of new
information, future developments or otherwise.
Contact Information:
Investors:James CarbonaraHayden IR(646)
755-7412james@haydenir.com
Media:Tammy GroenePanbela Therapeutics, Inc.(952)
479-1196IR@panbela.com
Panbela Therapeutics (NASDAQ:PBLA)
Historical Stock Chart
From Aug 2024 to Sep 2024
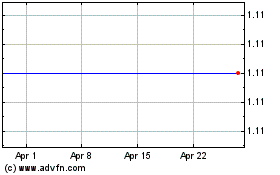
Panbela Therapeutics (NASDAQ:PBLA)
Historical Stock Chart
From Sep 2023 to Sep 2024