Gamida Cell Appoints Abigail L. Jenkins as President and Chief Executive Officer, Bringing Broad Leadership Experience in Commercializing Innovative Therapies
September 19 2022 - 7:00AM
Business Wire
Julian Adams, Ph.D., to Retire and Remain on
the Board as Planned Succession
Gamida Cell Ltd. (Nasdaq: GMDA), the global leader in the
development of NAM-enabled cell therapies for patients with
hematologic and solid cancers and other serious diseases, today
announced that Abigail “Abbey” L. Jenkins, MS, has joined as
President & CEO. Ms. Jenkins has also been appointed to Gamida
Cell’s Board of Directors. Ms. Jenkins succeeds Julian Adams,
Ph.D., who is retiring in accordance with planned succession and
will continue to serve on the company’s Board of Directors.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20220919005300/en/
“Abbey is an inspiring leader who brings to Gamida Cell an
expertise in building and scaling organizations as they mature
through commercialization alongside continued advancement of
innovations in R&D. In addition, she is skilled in corporate
strategy and is highly respected by her colleagues for her
commitments to build strong company cultures focused on patient
centric missions,” said Robert Blum, Chairman of Gamida Cell’s
Board of Directors. “On behalf of Gamida Cell’s Board, we welcome
Abbey and thank Julian for his longstanding commitment to the
company’s science and values during a pivotal time during which the
company achieved major milestones including the submission of the
BLA for omidubicel and the initiation of the clinical development
of GDA-201. We look forward to his continued service and scientific
counsel to the Board.”
Ms. Jenkins brings over 20 years of leadership experience in the
biopharmaceutical industry delivering life-enhancing therapies from
research to commercialization for patients in need. She served as
the Chief Commercial and Business Officer at Lyndra Therapeutics,
where she established and led global commercial, business
development, corporate strategy and portfolio management across
multiple therapeutic areas. Prior to Lyndra, she served as Senior
Vice President and Business Unit Head of Vaccines at Emergent
BioSolutions, where she oversaw the company’s largest therapeutic
division from discovery through commercialization. Ms. Jenkins also
served as Chief Commercial Officer and U.S. Business Head at
Aquinox Pharmaceuticals. Additionally, she has held senior
commercial and business development positions at Relypsa, Actavis,
Pfizer and Medimmune/AZ.
Ms. Jenkins holds a Master of Science in biotechnology and
biotech business enterprise from The Johns Hopkins University, a
Bachelor of Arts in psychology and biology from Indiana University,
and a certificate of achievement in General Management as a Kellogg
Executive Scholar. In September, she was recognized by PharmaVoice
as one of the top 100 Most Inspiring Leaders, Disrupter category,
for change-agents who are defining excellence in leadership in the
biopharma industry.
“I am excited to lead Gamida Cell as we work to fulfill our
mission of creating cures for blood cancers and serious hematologic
diseases. Under Julian’s leadership, the team has built a
strong pipeline of next-generation cell therapies that hold the
potential to meaningfully change the future of cancer care for
patients and healthcare providers,” said Ms. Jenkins. “Our next
goal will be to successfully deliver the first-ever allogeneic
hematopoietic stem cell therapy, omidubicel, to market if approved
and which we believe can expand access and eligibility for cancer
patients in need of a stem cell transplant as well as reduce the
overall burden on healthcare resources.”
“It has been a distinct honor and a privilege to discover and
develop novel medicines over the course of my 40-year career and to
serve this company as its CEO these past five years,” said Dr.
Adams. “I wish to thank all my Gamida Cell colleagues for their
unwavering support as well as their extraordinary efforts to bring
our science of NAM-enabled cell therapies closer to benefiting
patients with hematologic malignancies. Today, Gamida Cell is in a
position of strength, with excellent prospects for the future.”
About NAM Technology Our NAM-enabling technology is
designed to enhance the number and functionality of targeted cells,
enabling us to pursue a curative approach that moves beyond what is
possible with existing therapies. Leveraging the unique properties
of NAM (nicotinamide), we can expand and metabolically modulate
multiple cell types — including stem cells and natural killer cells
— with appropriate growth factors to maintain the cells’ active
phenotype and enhance potency. Additionally, our NAM technology
improves the metabolic fitness of cells, allowing for continued
activity throughout the expansion process.
About Omidubicel Omidubicel is a NAM-enabled cell therapy
candidate developed as a potential life-saving allogeneic
hematopoietic stem cell (bone marrow) transplant for patients with
blood cancers. Omidubicel demonstrated a statistically significant
reduction in time to neutrophil engraftment in comparison to
standard umbilical cord blood in an international, multi-center,
randomized Phase 3 study (NCT0273029) in patients with hematologic
malignancies undergoing allogeneic bone marrow transplant. The
Phase 3 study also showed reduced time to platelet engraftment,
reduced infections and fewer days of hospitalization. One-year
post-transplant data showed sustained clinical benefits with
omidubicel as demonstrated by significant reduction in infectious
complications as well as reduced non-relapse mortality and no
significant increase in relapse rates nor increases in
graft-versus-host-disease (GvHD) rates. Omidubicel is the first
stem cell transplant donor source to receive Breakthrough Therapy
Designation from the FDA and has also received Orphan Drug
Designation in the US and EU.
The BLA for omidubicel has been assigned a Prescription Drug
User Fee Act (PDUFA) target action date of January 30, 2023. If
approved, omidubicel will be the first allogeneic advanced stem
cell therapy donor source for patients with blood cancers in need
of a stem cell transplant.
Omidubicel is an investigational therapy, and its safety and
efficacy have not been established by the FDA or any other health
authority. For more information about omidubicel, please visit
https://www.gamida-cell.com.
About Gamida Cell Gamida Cell is pioneering a diverse
immunotherapy pipeline of potentially curative cell therapy
candidates for patients with solid tumor and blood cancers and
other serious blood diseases. We apply a proprietary expansion
platform leveraging the properties of NAM to allogeneic cell
sources including umbilical cord blood-derived cells and NK cells
to create therapy candidates with potential to redefine standards
of care. These include omidubicel, an investigational product with
potential as a life-saving alternative for patients in need of bone
marrow transplant, and a line of modified and unmodified
NAM-enabled NK cells targeted at solid tumor and hematological
malignancies. For additional information, please visit
www.gamida-cell.com or follow Gamida Cell on LinkedIn, Twitter,
Facebook or Instagram at @GamidaCellTx.
Cautionary Note Regarding Forward Looking Statements This
press release contains forward-looking statements as that term is
defined in the Private Securities Litigation Reform Act of 1995,
including with respect to timing of initiation and progress of, and
data reported from, the clinical trials of Gamida Cell’s product
candidates (including omidubicel), regulatory filings submitted to
the FDA (including the potential timing of the FDA’s review and
approval of the BLA for omidubicel), timing of commercialization
efforts, and the potentially life-saving or curative therapeutic
and commercial potential of Gamida Cell’s product candidates
(including omidubicel).. Any statement describing Gamida Cell’s
goals, expectations, financial or other projections, intentions or
beliefs is a forward-looking statement and should be considered an
at-risk statement. Such statements are subject to a number of
risks, uncertainties and assumptions, including those related to
the impact that the COVID-19 pandemic could have on our business,
and including the scope, progress and expansion of Gamida Cell’s
clinical trials and ramifications for the cost thereof; clinical,
scientific, regulatory and technical developments; and those
inherent in the process of developing and commercializing product
candidates that are safe and effective for use as human
therapeutics, and in the endeavor of building a business around
such product candidates. In light of these risks and uncertainties,
and other risks and uncertainties that are described in the Risk
Factors section and other sections of Gamida Cell’s Quarterly
Report on Form 10-Q, filed with the Securities and Exchange
Commission (SEC) on August 15, 2022, and other filings that Gamida
Cell makes with the SEC from time to time (which are available at
http://www.sec.gov), the events and circumstances discussed in such
forward-looking statements may not occur, and Gamida Cell’s actual
results could differ materially and adversely from those
anticipated or implied thereby. Although Gamida Cell’s
forward-looking statements reflect the good faith judgment of its
management, these statements are based only on facts and factors
currently known by Gamida Cell. As a result, you are cautioned not
to rely on these forward-looking statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220919005300/en/
For investors: Courtney Turiano Stern Investor Relations,
Inc. Courtney.Turiano@sternir.com 1-212-362-1200
For media: Heather DiVecchia Director, IR and Corporate
Communications Heather@gamida-cell.com 1-617-892-9083
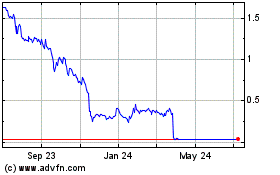
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Mar 2024 to Apr 2024
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Apr 2023 to Apr 2024