NEW HAVEN, Conn., Dec. 6, 2021 /PRNewswire/ -- Biohaven
Pharmaceutical Holding Company Ltd. (NYSE: BHVN) today announced
positive topline results from the second pivotal clinical trial
(NCT04571060) evaluating the safety and efficacy of its
investigational therapy, intranasal zavegepant, for the acute
treatment of migraine in adults. The Phase 3 study achieved its
co-primary regulatory endpoints of pain freedom and freedom from
most bothersome symptom at 2 hours and showed broad efficacy by
demonstrating statistically significant superiority to placebo
across a total of 15 prespecified primary and secondary outcome
measures. Based upon these results, combined with the prior
positive Phase 2/3 trial, Biohaven is moving forward with plans for
regulatory submissions in the United
States and other countries. Full results from this Phase 3
trial will be presented at upcoming medical conferences and/or
published in peer-reviewed journals.
Richard B. Lipton, M.D.,
Professor and Vice Chair of Neurology at the Albert Einstein
College of Medicine and Director of the Montefiore Headache Center,
said "Patients with migraine rate speed of onset as one of the most
important aspects of an effective therapy. The data from this trial
shows that intranasal zavegepant delivered impressive performance
on this metric by demonstrating statistically significant pain
relief within 15 minutes and return to normal function within 30
minutes. Additionally, non-oral treatments offer additional
benefits for patients who experience nausea, vomiting or
gastroparesis (with slow absorption). Intranasal zavegepant will be
an important new treatment option for patients who require a rapid
and non-oral option for acute treatment of their migraine
attacks."
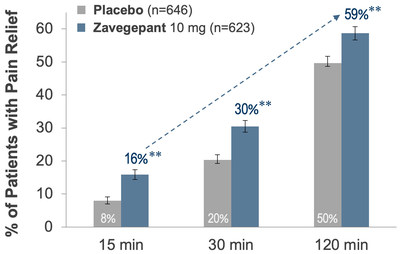
Zavegepant was statistically superior to placebo on the
co-primary endpoints of pain freedom (24% vs 15%, p < 0.0001)
and freedom from most bothersome symptom (40% vs 31%, p = 0.0012)
at 2 hours. Zavegepant was superior to placebo demonstrating pain
relief as early as 15 minutes (see Figure 1).
Patients achieved return to normal function as early as 30
minutes after dosing (p < 0.006). The efficacy benefits of
zavegepant were durable, including superiority versus placebo (p
< 0.05) on: sustained pain freedom 2 to 24 hours; sustained pain
freedom 2 to 48 hours; sustained pain relief 2 to 24 hours; and
sustained pain relief 2 to 48 hours.
Vlad Coric, M.D., Chief Executive
Officer at Biohaven stated, "Intranasal zavegepant was designed to
provide ultra-rapid pain relief and expand our CGRP
receptor-antagonist franchise by providing patients with another
important tool to combat migraine. The trial results clearly show
that the performance of this formulation exceeded expectations by
demonstrating superiority over placebo on pain relief at 15 minutes
and return to normal function by 30 minutes. The impressive
efficacy, safety and tolerability profile shown in this trial
highlights the potential of zavegepant to usher in a new era of
non-oral CGRP targeting migraine therapies that may transcend the
traditional boundaries of older legacy intranasal migraine
approaches. Biohaven is committed to delivering on its promise to
provide new treatment options for the millions of people living
with this debilitating disease and these data represent a major
milestone in that endeavor," (see Figure 2).
The Phase 3 pivotal study is a randomized, double-blind,
placebo-controlled clinical trial that randomized 1,405 adults with
at least a one-year history of migraine (with or without aura) and
migraine attacks lasting, on average, 4 to 72 hours if untreated.
Conducted at 94 sites in the United
States, the study evaluated the safety and efficacy of
zavegepant intranasal spray taken as needed in a single dose
compared to placebo for the acute treatment of a moderate to severe
migraine attack.
Zavegepant showed a favorable safety and tolerability profile
among study participants that was consistent with prior clinical
trial experience. The most common individual adverse event in the
pivotal study reported with a frequency ≥ 5% in the zavegepant
treatment arm and greater than placebo was abnormal taste (21% vs
5%). The majority of AEs were mild in intensity.
Biohaven plans to file a New Drug Application (NDA) for
zavegepant with the U.S. Food and Drug Administration (FDA) in 1Q
2022 and other countries thereafter. If ultimately approved,
zavegepant would be the first intranasal calcitonin gene-related
peptide (CGRP) receptor antagonist for the acute treatment of
migraine. Zavegepant is the second clinical candidate for Biohaven
after FDA-approved Nurtec® ODT (rimegepant) for the
acute treatment of migraine and preventive treatment of episodic
migraine in adults.
About Zavegepant
Zavegepant is a third generation,
high affinity, selective and structurally unique, small molecule
CGRP receptor antagonist from Biohaven's NOJECTION®
Migraine Platform and the only CGRP receptor antagonist in clinical
development with both intranasal and oral formulations. Previously
the efficacy and safety of intranasal zavegepant was shown in a
randomized controlled Phase 2/3 dose-ranging trial with a total of
over 1000 patients treated. In this study, zavegepant showed
statistical superiority to placebo on the coprimary endpoints of 2
hour freedom from pain and freedom from patients' most bothersome
symptom (either nausea, photophobia or phonophobia). The present
announcement represents the second zavegepant pivotal clinical
trial to meet these coprimary endpoints. For more information,
visit https://www.biohavenpharma.com.
About Migraine
Nearly 40 million people in the
U.S. suffer from migraine and the World Health Organization
classifies migraine as one of the 10 most disabling medical
illnesses. Migraine is characterized by debilitating attacks
lasting four to 72 hours with multiple symptoms, including
pulsating headaches of moderate to severe pain intensity that can
be associated with nausea or vomiting, and/or sensitivity to sound
(phonophobia) and sensitivity to light (photophobia). There is a
significant unmet need for new treatments as more than 90 percent
of people with migraine are unable to work or function normally
during an attack.
CGRP Receptor Antagonism
Small molecule CGRP receptor
antagonists represent a novel class of drugs for the treatment of
migraine. CGRP receptor antagonists work by reversibly blocking
CGRP receptors, thereby inhibiting the biologic activity of the
CGRP neuropeptide. For acute treatment, this unique mode of action
potentially offers an alternative to other agents, particularly for
patients who have contraindications to the use of triptans or who
have a poor response to triptans or are intolerant to them. CGRP
signal-blocking therapies have not been associated with medication
overuse headache (MOH) or rebound headaches which limits the
clinical utility of other acute treatments due to increases in
migraine attacks that result from frequent use.
About Nurtec ODT
Nurtec ODT (rimegepant) is the first and only calcitonin
gene-related peptide (CGRP) receptor antagonist available in a
quick-dissolve ODT formulation that is approved by
the U.S. Food and Drug Administration (FDA) for the acute
treatment of migraine with or without aura and the preventive
treatment of episodic migraine in adults. The activity of the
neuropeptide CGRP is thought to play a causal role in migraine
pathophysiology. Nurtec ODT is a CGRP receptor antagonist that
works by reversibly blocking CGRP receptors, thereby inhibiting the
biologic activity of the CGRP neuropeptide. The recommended dose of
Nurtec ODT is 75 mg, taken as needed, up to once daily to treat or
every other day to help prevent migraine attacks. For more
information about Nurtec ODT, visit nurtec.com. The most common
adverse reaction was nausea and abdominal pain/indigestion. Avoid
concomitant administration of Nurtec ODT with strong inhibitors of
CYP3A4, strong or moderate inducers of CYP3A or inhibitors of P-gp
or BCRP. Avoid another dose of Nurtec ODT within 48 hours when it
is administered with moderate inhibitors of CYP3A4. Please click
here for full Nurtec ODT Prescribing Information and Patient
Information.
About Biohaven
Biohaven is a commercial-stage biopharmaceutical company with a
portfolio of innovative, best-in-class therapies to improve the
lives of patients with debilitating neurological and
neuropsychiatric diseases, including rare disorders. Biohaven's
Neuroinnovation™ portfolio includes FDA-approved Nurtec ODT
(rimegepant) for the acute and preventive treatment of migraine and
a broad pipeline of late-stage product candidates across three
distinct mechanistic platforms: CGRP receptor antagonism for the
acute and preventive treatment of migraine; glutamate modulation
for obsessive-compulsive disorder, Alzheimer's disease, and
spinocerebellar ataxia; and MPO inhibition for amyotrophic lateral
sclerosis. More information about Biohaven is available
at www.biohavenpharma.com.
Forward-Looking Statements
This news release includes
forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995. These forward-looking
statements involve substantial risks and uncertainties, including
statements regarding the future development, timing and potential
marketing approval and commercialization of Nurtec ODT
(rimegepant), rimegepant or zavegepant. Various important factors
could cause actual results or events to differ materially from
those that may be expressed or implied by our forward-looking
statements. Additional important factors to be considered in
connection with forward-looking statements are described in the
"Risk Factors" section of Biohaven's Annual Report on Form 10-K for
the year ended December 31, 2020,
filed with the Securities and Exchange Commission on March 1, 2021, and Biohaven's subsequent filings
with the Securities and Exchange Commission. The forward-looking
statements are made as of this date and Biohaven does not undertake
any obligation to update any forward-looking statements, whether as
a result of new information, future events or otherwise, except as
required by law.
Nurtec, Nurtec ODT and NOJECTION are registered trademarks of
Biohaven Pharmaceutical Ireland DAC.
Neuroinnovation is a trademark of Biohaven Pharmaceutical Holding
Company Ltd.
Biohaven Contact:
Vlad Coric, M.D.
Chief Executive Officer
Vlad.Coric@biohavenpharma.com
Media Contact:
Mike Beyer
Sam Brown Inc.
mikebeyer@sambrown.com
312-961-2502
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-reports-positive-topline-results-from-pivotal-migraine-trial-of-intranasal-zavegepant-demonstrating-ultra-rapid-pain-relief-by-15-minutes-prepares-for-submission-of-new-drug-application-301437767.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-reports-positive-topline-results-from-pivotal-migraine-trial-of-intranasal-zavegepant-demonstrating-ultra-rapid-pain-relief-by-15-minutes-prepares-for-submission-of-new-drug-application-301437767.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.