Gamida Cell Announces Two-Part Virtual Miniseries Focused on Omidubicel to Take Place in September
September 03 2020 - 8:01AM
Business Wire
Gamida Cell Ltd. (Nasdaq: GMDA), an advanced cell therapy
company committed to cures for blood cancers and serious blood
diseases, today announced that it will host a two-part virtual
miniseries in September focused on key topics related to
omidubicel, an advanced cell therapy in Phase 3 clinical
development as a potentially life-saving treatment option for
patients in need of a bone marrow transplant. The webcasts will
take place on Friday, September 11, 2020, and Wednesday, September
23, 2020, both at 1:00 p.m. ET.
The miniseries topics are as follows:
- September 11: A discussion of the patient journey and
treatment paradigm in allogeneic bone marrow transplant featuring
Steven Devine, M.D., Chief Medical Officer, Be The Match
BioTherapies, National Marrow Donor Program®/Be The Match®, and
Associate Scientific Director, the CIBMTR® (Center for
International Blood and Marrow Transplant Research®)
- September 23: A discussion of the healthcare economics
of bone marrow transplant featuring Krishna Komanduri, M.D., Kalish
Family Chair in Stem Cell Transplantation and Chief, Division of
Transplantation and Cellular Therapy at the Sylvester Comprehensive
Cancer Center, University of Miami
In May, Gamida Cell reported that its Phase 3 study of
omidubicel met its primary endpoint, demonstrating a highly
statistically significant reduction in time to neutrophil
engraftment, a key milestone in recovery from a bone marrow
transplant. Gamida Cell expects to present the full data set,
including secondary endpoint data, at a medical meeting in the
fourth quarter of 2020. The company also expects to begin
submitting the biologics license application for omidubicel to the
U.S. Food and Drug Administration (FDA) on a rolling basis in the
fourth quarter of 2020. Omidubicel is the first bone marrow
transplant product to receive Breakthrough Therapy Designation from
the FDA and has also received Orphan Drug Designation in the U.S.
and EU.
Each webcast will be available on the “Investors & Media”
section of the Gamida Cell website at www.gamida-cell.com. A replay
of the webcast will be available about two hours after the event,
for approximately 90 days.
About Omidubicel
Omidubicel is an advanced cell therapy under development as a
potential life-saving allogeneic hematopoietic stem cell (bone
marrow) transplant solution for patients with hematologic
malignancies (blood cancers). In both Phase 1/2 and Phase 3
clinical studies (NCT01816230 and NCT02730299), omidubicel
demonstrated rapid and durable time to engraftment and was
generally well tolerated.1,2 Omidubicel is also being evaluated in
a Phase 1/2 clinical study in patients with severe aplastic anemia
(NCT03173937). The aplastic anemia investigational new drug
application is currently filed with the FDA under the brand name
CordIn®, which is the same investigational development candidate as
omidubicel. For more information on clinical trials of omidubicel,
please visit www.clinicaltrials.gov.
Omidubicel is an investigational therapy, and its safety and
efficacy has not been evaluated by the U.S. Food and Drug
Administration or any other health authority.
About Gamida Cell
Gamida Cell is an advanced cell therapy company committed to
cures for patients with blood cancers and serious blood diseases.
We harness our cell expansion platform to create therapies with the
potential to redefine standards of care in areas of serious medical
need. For additional information, please visit www.gamida-cell.com or follow Gamida Cell
on LinkedIn or Twitter at
@GamidaCellTx.
Cautionary Note Regarding Forward Looking Statements
This press release contains forward-looking statements as that
term is defined in the Private Securities Litigation Reform Act of
1995, including with respect to the anticipated timing of data
disclosures and regulatory filing submissions, which statements are
subject to a number of risks, uncertainties and assumptions,
including, but not limited to the ongoing global COVID-19 pandemic
and clinical, scientific, regulatory and technical developments. In
light of these risks and uncertainties, and other risks and
uncertainties that are described in the Risk Factors section and
other sections of Gamida Cell’s Annual Report on Form 20-F, filed
with the Securities and Exchange Commission (SEC) on February 26,
2020, and other filings that Gamida Cell makes with the SEC from
time to time (which are available at http://www.sec.gov), the
events and circumstances discussed in such forward-looking
statements may not occur, and Gamida Cell’s actual results could
differ materially and adversely from those anticipated or implied
thereby. Any forward-looking statements speak only as of the date
of this press release and are based on information available to
Gamida Cell as of the date of this release.
References
1Horwitz M.E., Wease S., Blackwell B., Valcarcel D. et al. Phase
I/II study of stem-cell transplantation using a single cord blood
unit expanded ex vivo with nicotinamide. J Clin Oncol. 2019 Feb
10;37(5):367-374. 2Gamida Cell press release, “Gamida Cell
Announces Positive Topline Data from Phase 3 Clinical Study of
Omidubicel in Patients with High-Risk Hematologic Malignancies,”
issued May 12, 2020. Last accessed August 31, 2020.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200903005080/en/
Investor Contact: Jaren Irene Madden
jaren@gamida-cell.com 1-617-286-6264
Media Inquiries: Matthew Corcoran
mcorcoran@tenbridgecommunications.com 1-617-866-7350
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Aug 2024 to Sep 2024
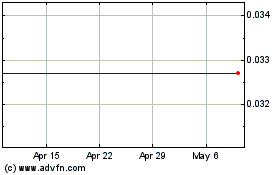
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Sep 2023 to Sep 2024