SYDNEY, April 24, 2018 /PRNewswire/ -- Benitec
Biopharma Limited (ASX: BLT; NASDAQ: BNTC; NASDAQ: BNTCW) today
provided an update on its two lead programs in head and neck
squamous cell carcinoma (HNSCC) and oculopharyngeal muscular
dystrophy (OPMD). This update should be read in conjunction
with the latest corporate presentation which can be found on the
Company's website.
HNSCC
Benitec is developing BB-401 as a treatment for
HNSCC. BB-401 is a recombinant DNA construct that produces an
antisense RNA with specificity against Epidermal Growth Factor
Receptor (EGFR), an oncogenic factor overexpressed in more than 90%
in lesions from patients with HNSCC. The goal of BB-401
treatment is to inhibit the expression of EGFR in the treated
lesions and thus control the progression of disease and increase
patient survival.
Manufacturing:
Manufacturing of BB-401 clinical product was completed at the end
of last year and it is now ready for clinical use.
Clinical and Regulatory:
BB-401 was originally developed through early stage studies at the
University of Pittsburgh and has been
in-licensed by Benitec. In prior clinical studies conducted
under an investigator-sponsored Investigational New Drug
Application (IND) with the US Food & Drug Administration,
BB-401 demonstrated potent activity either as a monotherapy in a
dose range study or when used in combination with cetuximab and
radiation. The goal of the current Phase 2 study is to confirm the
clinical activity of high doses of BB-401 against HNSCC
lesions.
The Company continues to make good progress with the initiation
of this Phase 2 clinical study which is designed as an open label
study to explore the safety, tolerability and efficacy of BB-401
following intratumoral injections. These are patients who are
refractory to all standard therapies such as surgery, chemotherapy
and immunotherapy. The study is expected to enrol up to 30
patients at 5-8 sites across Australia and Russia. The trial is
registered on www.clinicaltrials.gov with the identifier:
NCT03433027, where more details can be found.
The first clinical site has now been opened in Australia and the Company anticipates having
additional Australian sites open later this month. Regulatory
review with the Ministry of Health is ongoing in Russia and the Company expects approval at the
end of May.
As shown in the figure below, the primary outcome is the
objective response rate to BB-401 in the injected lesion.
Additional secondary endpoints include progression free survival,
overall survival, duration of response, disease control rate,
safety and tolerability. The study has a two-stage design
which allows for stopping of the study based on either success or
futility at the end of the first stage, after 12 patients have been
enrolled and monitored through the primary outcome measure.
This interim analysis is anticipated to occur around the end of the
2018 calendar year.
Market Expansion Opportunities:
In addition to HNSCC, EGFR overexpression has been associated with
a number of cancers, including epithelial tumors of the head and
neck, squamous cell cancers of the lungs, anal cancers, and
glioblastoma. As such, Benitec is exploring other potential
clinical indications for anti-EGFR strategies, including rare
cancers.
OPMD
BB-301 is a single vector (gene therapy
construct) system that uses DNA directed RNA interference (ddRNAi)
to silence expression of the mutant gene associated with OPMD,
while simultaneously expressing a copy of the normal, healthy
version of the same gene to restore the function of that
gene.
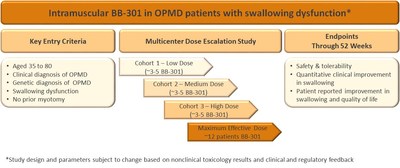
Clinical:
BB-301 is being developed as a treatment for OPMD. The first
clinical study of BB-301 will be focused on treating dysphagia in
patients with OPMD. Defined as a difficulty in swallowing, it
is the inability to restrict food intake into the stomach versus
being aspirated into the airways in dysphagia which causes the
majority of serious health problems for patients with OPMD
including death. In the initial clinical study, BB-301
will be administered via intramuscular injection into the
cricopharyngeus, a throat muscle which regulates the passage of
swallowed food into the oesophagus. The endpoints of this
study are designed to monitor safety as well as determine if
treatment results in the improvement of swallowing function and
improvement in swallowing quality of life in OPMD patients.
The clinical protocol has received feedback from regulatory
agencies from the US (FDA), Canada
(Health Canada) and several agencies within the EU. Further
refinement of the protocol is anticipated in a face-to-face
Clinical Advisory Board meeting with our Key Opinion Leaders
including doctors who managed treatment options for OPMD patients
as well as experts in the quantitative assessment of dysphagia.
With only palliative care and no marketed therapeutics
available, there has been a significant level of enthusiasm and
hope from the patients in the OPMD community for BB-301.
Given this and given the known geographical clusters of OPMD,
Benitec is optimistic about patient accessibility when the IND, or
other initial regulatory application, has been filed and
approved.
Manufacturing
BB-301 is being produced using baculovirus-based technology, a
highly scalable methodology that permits high yield, cost efficient
production of the BB-301 product. The Company has
successfully produced high titer and highly active material at the
50L scale which is being used for the ongoing toxicology
studies. The focus has shifted to producing supplies of
BB-301 at a 250L scale, which will support the clinical
program.
Toxicology:
Before BB-301 can be tested in human clinical trials, the necessary
safety studies (toxicology studies) must be performed in large
animal models. These IND-enabling toxicology studies are to
ensure that the delivery procedures as well as the doses of BB-301
that we intend to use do not cause any obvious safety problems.
The IND-enabling toxicology studies with BB-301 are being
conducted in sheep. This animal model was selected because
the weight of the sheep and the size of key muscles in the upper
digestive system are consistent with human subjects. These
features are key to support the route of administration of direct
injection into cricopharyngeal muscle. Given the complexity
of the intramuscular route of administration in the sheep and input
from the regulatory agencies, the number of animals being used in
these studies has been increased resulting in slightly slower
timelines. The first toxicology studies are now underway and
the Company anticipates filing the IND in 1Q 2019.
Regulatory:
As noted above, regulatory discussions have been completed with
agencies in the United States,
Canada and Europe. These are
all regions with key OPMD patient clusters and the purpose of these
meetings was to discuss the regulatory development pathway for
BB-301 as a treatment for OPMD and to ensure that Benitec's
proposed development program which includes the existing safety and
efficacy data, manufacturing plans, nonclinical plans and clinical
study design addresses the regulatory expectations of these
agencies.
Benitec's SME (Small and Medium Sized Business Entities) status
in the European Union combined with the Orphan Drug Designation in
the US and EU are invaluable as Benitec progresses BB-301 towards
commercialisation and paves the way for a potentially clear and
expeditious development pathway.
Market Expansion Opportunities:
BB-301 is initially being developed as an intramuscular injection
to treat the dysphagia associated with OPMD. Assuming
success, there is the potential to treat earlier stages of
dysphagia, systemic administration to treat proximal muscle
weakness and ptosis and prophylactic treatment to prevent the
development of muscle weakness.
In addition to the market expansion opportunities noted above
for BB-301 there are opportunities to use single vector 'silence
and replace' strategies in other disease indications, particularly
rare diseases, as a competitive advantage versus other companies
and technologies.
Commenting on the update CEO Greg
West said: "The advancement of the HNSCC program into the
clinic and the OPMD program becoming closer to 'clinic ready' shows
how our novel technology can address significant unmet clinical
needs. We are very pleased with the progress in these programs.
Positive outcomes in the clinical trials would contribute to both
the validation of our technology and improvement in the valuation
of Benitec.
For further information regarding Benitec and its activities,
please contact the persons below, or visit the Benitec website at
www.benitec.com
|
Australia
Investor Relations
|
United States
Investor Relations
|
|
Market Eye
Orla
Keegan
Director
Tel: +61 (2) 8097
1201
Email:
orla.keegan@marketeye.com.au
|
M Group Strategic
Communications
Jay
Morakis
Managing
Director
Tel: +1
212.266.0191
Email:
jmorakis@MGroupSC.com
|
About Benitec Biopharma Limited:
Benitec
Biopharma Limited (ASX: BLT; NASDAQ: BNTC; NASDAQ: BNTCW) is a
biotechnology company developing innovative therapeutics based on
its patented gene-silencing technology called ddRNAi or 'expressed
RNAi'. Based in Sydney, Australia
with laboratories in Hayward,
California (USA), and collaborators and licensees around the
world, the company is developing ddRNAi-based therapeutics for
chronic and life-threatening human conditions including head &
neck squamous cell carcinoma, OPMD retinal based diseases such as
wet age-related macular degeneration, and hepatitis B. Benitec has
also licensed ddRNAi to other biopharmaceutical companies for
applications including HIV/AIDS, Huntington's Disease, chronic
neuropathic pain, cancer immunotherapy and retinitis
pigmentosa.
About OPMD:
OPMD is a rare inherited myopathy
characterized by dysphagia (difficulty in swallowing), the loss of
muscle strength, and weakness in multiple parts of the body.
Patients typically suffer from severe dysphagia, ptosis (eye lid
drooping), tongue atrophy, proximal lower limb weakness, dysphonia
(altered and weak voice), limitation in looking upward, as well as
facial muscle and proximal upper limb weakness. Progressing
throughout that patient's life, OPMD is not typically diagnosed
until the individuals reach their late 40s. As the dysphagia
becomes more severe, patients become malnourished, lose significant
weight, become dehydrated and suffer from repeated incidents of
aspiration pneumonia. The last two symptoms are often the cause of
death. No cure is currently available for OPMD. The
cricopharyngeal myotomy is the only treatment available to improve
swallowing in these patients, but because the root cause of the
genetic disease has not been addressed, the pharyngeal musculature
still undergoes progressive degradation leading to the previously
mentioned complications.
About Head and Neck Cancer:
Cancers that are
known as head and neck cancers usually begin in the squamous cells
that line the moist mucosal surfaces inside the head and neck, such
as inside the mouth and the throat. The global incidence of
HNSCC is expected to increase from approximately 119,000 cases in
2016 to over 136, 000 cases in 2026. Head and neck cancers
are more than twice as common among men as they are among
women. Squamous cell carcinoma of the head and neck accounts
for more than 90% of all head and neck cancers, and more than 50%
of HNSCC patients present with Stage III or higher disease (locally
advanced or metastatic), which has higher potential for progression
and recurrence. For patients with recurrent of metastatic
HNSCC the median overall survival is 7.8 months and the five year
survival rate is 3.6%. Total drug sales in the HNSCC markets
in the seven major markets (United
States, France,
Germany, Italy, Spain,
United Kingdom and Japan) are expected to increase from
$579.4 million in 2016 to just over
$4.1 billion in 2026, at a Compound
Annual Growth Rate (CAGR) of 21.6%. Reference: GlobalData Report
(March 2018): Head and Neck Squamous
Cell Carcinoma – Opportunity Analysis and Forecast to 2026
Safe Harbor Statement:
This press release
contains "forward-looking statements" within the meaning of section
27A of the US Securities Act of 1933 and section 21E of the US
Securities Exchange Act of 1934. Any forward-looking statements
that may be in this ASX/Nasdaq announcement are subject to risks
and uncertainties relating to the difficulties in Benitec's plans
to develop and commercialise its product candidates, the timing of
the initiation and completion of preclinical and clinical trials,
the timing of patient enrolment and dosing in clinical trials, the
timing of expected regulatory filings, the clinical utility and
potential attributes and benefits of ddRNAi and Benitec's product
candidates, potential future out-licenses and collaborations, the
intellectual property position and the ability to procure
additional sources of financing. Accordingly, you should not rely
on those forward-looking statements as a prediction of actual
future results.
 View original content with
multimedia:http://www.prnewswire.com/news-releases/asxnasdaq-announcement-300635074.html
View original content with
multimedia:http://www.prnewswire.com/news-releases/asxnasdaq-announcement-300635074.html
SOURCE Benitec Biopharma Limited