|
|
|
|
|
|
|
|
|
|
|
Per Share
|
|
Total
|
|
|
Public offering price
|
|
$
|
3.50
|
|
$
|
21,700,000
|
|
|
Underwriting discounts and commissions(1)
|
|
$
|
0.245
|
|
$
|
1,519,000
|
|
|
Proceeds to Invuity, Inc., before expenses
|
|
$
|
3.255
|
|
$
|
20,181,000
|
|
-
(1)
-
See
"Underwriting" beginning on page S-20 for additional information regarding underwriting compensation.
The underwriter has a 30-day option to purchase up to 600,000 additional shares of common stock from us.
Neither the Securities and Exchange Commission nor any state securities commission has approved of anyone's investment in these securities or determined if this
prospectus supplement or the accompanying prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The underwriter expects to deliver the shares of common stock on or about March 19, 2018.
William Blair
The date of this prospectus supplement is March 15, 2018.
Table of Contents
TABLE OF CONTENTS
PROSPECTUS
We
have not, and the underwriter has not, authorized anyone to provide any information or to make any representations other than those contained in this prospectus supplement, the
accompanying prospectus or in any free writing prospectuses we have prepared and take no responsibility for, and can provide no assurance as to the reliability of, any other information that others
may give you. You should rely only on the information contained in or incorporated by reference in this prospectus supplement, the accompanying prospectus and in any free writing prospectus that we
have authorized for use in connection with this offering. We have not, and the underwriter has not, authorized anyone to provide you with different information. If anyone provides you with different
or inconsistent information, you should not rely on it. This prospectus supplement and the accompanying prospectus is an offer to sell only the shares offered hereby, but only under circumstances and
in jurisdictions where it is lawful to do so. The information contained in this prospectus supplement and the accompanying prospectus is current only as of its date.
S-i
Table of Contents
ABOUT THIS PROSPECTUS SUPPLEMENT
This document has two parts, a prospectus supplement and an accompanying prospectus dated July 21, 2016. This prospectus supplement and
the accompanying prospectus are part of a registration statement that we filed with the Securities and Exchange Commission, or the SEC, utilizing the SEC's "shelf" registration process. This
prospectus supplement, which describes certain matters relating to us and the specific terms of this offering of shares of common stock, adds to and updates information contained in the accompanying
prospectus and the documents incorporated by reference herein. Generally, when we refer to this document or prospectus, we are referring to both parts of this document combined. Both this prospectus
supplement and the accompanying prospectus include important information about us, our common stock and other information you should know before investing in our common stock. The accompanying
prospectus gives more general information, some of which may not apply to the shares of common stock offered by this prospectus supplement. To
the extent the information contained in this prospectus supplement differs or varies from the information contained in the accompanying prospectus, you should rely on the information contained in this
prospectus supplement. If the information contained in this prospectus supplement differs or varies from the information contained in a document we have incorporated by reference, you should rely on
the information in the more recent document. If you invest in our common stock, you should read the registration statement of which this document forms a part and this document, including the
documents incorporated by reference herein that are described under the heading "Incorporation by Reference." Distribution of this prospectus supplement and the accompanying prospectus and the
offering of the common stock in certain jurisdictions may be restricted by law. Neither we nor the underwriter is making an offer of the common stock in any jurisdiction where the offer is not
permitted. Persons who come into possession of this prospectus supplement and the accompanying prospectus should inform themselves about and observe any such restrictions. This prospectus supplement
and the accompanying prospectus do not constitute, and may not be used in connection with, an offer or solicitation by anyone in any jurisdiction in which such offer or solicitation is not authorized
or in which the person making such offer or solicitation is not qualified to do so or to any person to whom it is unlawful to make such offer or solicitation. You should assume that the information
appearing in this prospectus supplement, the accompanying prospectus, the documents incorporated by reference in this prospectus supplement and the accompanying prospectus, and in any free writing
prospectus that we have authorized for use in connection with this offering, is accurate only as of the date of those respective documents. Our business, financial condition, results of operations and
prospects may have changed since those dates. You should not consider any information in this prospectus supplement or the accompanying prospectus to be investment, legal or tax advice. You should
consult your own counsel, accountant and other advisors for legal, tax, business, financial and related advice regarding the purchase of the common stock. Neither we nor the underwriter is making any
representation to you regarding the legality of an investment in the common stock by you under applicable investment or similar laws.
Unless
otherwise indicated, information contained in this prospectus supplement, the accompanying prospectus or the documents incorporated by reference, concerning our industry and the
markets in which we operate, including our general expectations and market position, market opportunity and market share, is based on information from our own management estimates and research, as
well as from industry and general publications, and research, surveys and studies conducted by third parties. Management estimates are derived from publicly available information, our knowledge of our
industry and assumptions based on such information and knowledge, which we believe to be reasonable. In addition, assumptions and estimates of our and our industry's future performance are necessarily
subject to a high degree of uncertainty and risk due to a variety of important factors, including those described in "Risk Factors" in this prospectus supplement, the accompanying prospectus, in our
Annual Report on Form 10-K for the year ended December 31, 2017, which is incorporated by reference into this prospectus supplement. These and other important factors could
S-ii
Table of Contents
cause
our future performance to differ materially from our assumptions and estimates. See "Special Note Regarding Forward-Looking Statements."
Invuity, Inc.
and our logo, as well as Intelligent Photonics, are our trademarks and are used in this prospectus supplement and the accompanying prospectus. This prospectus
supplement and the accompanying prospectus also include trademarks, tradenames and service marks that are the property of other organizations.
S-iii
Table of Contents
PROSPECTUS SUPPLEMENT SUMMARY
This summary highlights information contained elsewhere or incorporated by reference in this prospectus supplement and
the accompanying prospectus and does not contain all of the information you should consider before investing in shares of our common stock. You should read this entire prospectus supplement and the
accompanying prospectus carefully, including the information incorporated by reference herein, the information included under the section entitled "Risk Factors" and the financial statements and the
related notes thereto incorporated by reference in this prospectus supplement and the accompanying prospectus, before you decide to invest in shares of our common stock.
In this prospectus supplement, unless the context otherwise requires, references to "Invuity," "we," "our," "us" and "the Company" refer to Invuity, Inc.,
a Delaware corporation.
Overview of Our Company
We are a leading medical technology company focused on developing and marketing advanced surgical devices to improve the ability of physicians
to perform minimal access surgery through smaller and hidden incisions. Our patented Intelligent Photonics® technology delivers enhanced visualization, which facilitates surgical
precision, efficiency and safety. In addition, we utilize comprehensive strategic marketing programs to create stronger institutional partnerships. Clinical applications include women's health,
encompassing breast cancer and breast reconstruction surgery, gynecology and thyroid surgery. Additional applications include procedures for electrophysiology, spine, orthopedic, cardiothoracic, and
general surgery.
We
channel our development through two broad categories of innovation. First, we integrate our Intelligent Photonics® technology platform into our single-use and reusable
advanced surgical devices to address some of the critical intracavity illumination and visualization challenges facing surgeons today. We utilize this proprietary technology to develop optical
waveguides that direct and shape thermally cool, brilliant light into broad, uniform and volumetric illumination of the surgical target. We believe that improving a surgeon's ability to see critical
anatomical structures can lead to better clinical and aesthetic outcomes, improved patient safety and reduced surgical time and healthcare costs.
Our
second broad category of innovations for minimally invasive and minimal access surgical procedures is in the development and commercialization of a novel advanced energy platform. In
September 2016, we received U.S. Food and Drug Administration, or FDA, 510(k) clearance of PhotonBlade®, a dynamic precision illuminator with enhanced energy delivery.
PhotonBlade® is a first-of-its-kind device, delivering directed, thermally cool illumination at the precise point of surgical treatment in conjunction with a novel advanced energy platform
allowing for precise tissue cutting and coagulation with minimal tissue damage. As such, PhotonBlade® represents a new category of Intelligent Photonics® and strategically
expands our current product portfolio. After a preliminary market launch in March 2017, we fully launched the PhotonBlade® in September 2017.
Finally,
in the third quarter of 2017, through a distribution arrangement with our manufacturing partner, Fluoptics Imaging Inc., we began a limited market launch of a
fluorescence imaging system, called PhotonVue, which is used for the visual assessment of blood flow in adults as an adjunctive method for the evaluation of tissue perfusion
We
sold our devices to approximately 870 hospitals in the fourth quarter of 2017, as compared to approximately 745 hospitals in the fourth quarter of 2016. Based on the number of
single-use units we have shipped as of December 31, 2017, we estimate that our devices have been used in approximately 367,000 surgical procedures. We are also using our advanced photonics
technology to develop new devices and modalities to broaden the application and adoption of minimal access procedures and enable new advanced surgical techniques.
S-1
Table of Contents
We
earn revenue from the sale of our devices primarily through our direct sales force as complemented by our independent sales agents. Our focus has been on increasing the number of
direct sales representatives and sales territories and reducing our reliance on our independent sales agents. In the three months ended December 31, 2017, 82% of our revenue was generated from
the sale of single-use devices to our customers. For the years ended December 31, 2017, 2016 and 2015, our total revenues were $39.6 million, $32.5 million and
$21.0 million, respectively. For the years ended December 31, 2017, 2016 and 2015, we had net losses of $39.9 million, $40.6 million and $37.6 million, respectively.
We expect
to continue to incur losses in the near term. Through December 31, 2017, we had an accumulated deficit of $186.1 million.
Our
advanced photonics technology has allowed us to design multiple variations of Photonguides in order to target different illumination patterns for different shapes of surgical
cavities. Because we can mold our solid core optical-grade polymer into different shapes, we are able to design Photonguides that either direct the light narrowly for deep cavities, or broadly for
larger blade cavities. Photonguides also come in narrow or wide configurations to accommodate various retractor widths that are designed for varying patient anatomies. Our versatile design and
manufacturing capabilities allow us to develop Photonguides with a variety of extraction patterns. For example, our current retractor based Photonguides utilize a complex geometry of refractive
microstructures and microlenses, whereas as our handheld illuminated aspiration devices have integrated microlens arrays. Using advanced ray-trace software modeling programs, we are able to perform
three-dimensional optical performance modeling of Photonguides, as well as an entire assembly including the retractor. We are capable of analyzing the entire optical performance of the assembly as we
monitor various characteristics such as extracted light direction, uniformity on the target, glare to the user, as well as thermal profile. This ray-trace modeling process helps us develop illuminated
surgical devices that are designed to provide optimal intracavity illumination.
Our
marketing efforts are focused on developing a strong reputation with major teaching institutions and hospitals, as well as surgeons that we have identified as key opinion leaders
based on their knowledge of our devices, clinical expertise and reputation. We developed the Invuity Hidden Scar
TM
Surgery program to train and certify surgeons on minimal access
surgical approaches and designate Centers of Excellence in Hidden Scar Surgery at hospitals and medical centers. Since launching the program, we achieved an average quarter-on-quarter increase in the
number of Certified Surgeons and the number of Centers of Excellence of 32% and 56%, respectively. As of Q4 2017, we have 318 Certified Surgeons and 139 Centers of Excellence. Breast cancer surgery is
our initial focus with the Hidden Scar Breast Surgery program and we are expanding into other specialties to include Hidden Scar Breast Reconstruction and Hidden Scar Hysterectomy to form a
broader women's health initiative.
Recent Developments
On February 28, 2018, Philip Sawyer stepped down from his position as President and Chief Executive Officer of the Company and resigned
as a member of our board of directors. The board of directors appointed Scott Flora, a member of our board of directors, to serve as the Company's Interim President and Chief Executive Officer.
Implications of Being an Emerging Growth Company
As a company with less than $1.07 billion in revenue during our last fiscal year, we qualify as an "emerging growth company" as defined
in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. An emerging growth company may take advantage of specified reduced reporting
S-2
Table of Contents
requirements
and is relieved of certain other significant requirements that are otherwise generally applicable to public companies. As an emerging growth company, among other
things:
-
•
-
we are exempt from the requirement to obtain an attestation and report from our auditors on the assessment of our internal control over
financial reporting pursuant to the Sarbanes-Oxley Act of 2002, as amended, or the Sarbanes-Oxley Act;
-
•
-
we are permitted to provide less extensive disclosure about our executive compensation arrangements; and
-
•
-
we are not required to give our stockholders non-binding advisory votes on executive compensation or golden parachute arrangements.
We
may take advantage of these provisions until the last day of the fiscal year following the fifth anniversary of the completion of our initial public offering, or December 31,
2020. However, if certain events occur prior to the end of such five-year period, including (i) if we become a "large accelerated filer" as defined in Rule 12b-2 under the Securities
Exchange Act of 1934, as amended, or the Exchange Act, (ii) our annual gross revenue equals or exceeds $1.07 billion or (iii) we issue more than $1.0 billion of
non-convertible debt in any three-year period, we will cease to be an emerging growth company prior to the end of such five-year period.
We
may choose to take advantage of some or all of these reduced burdens. We have taken advantage of reduced reporting requirements in the documents incorporated by reference in this
prospectus supplement and the accompanying prospectus. Accordingly, the information contained herein may be different from the information you receive from our competitors that are public companies,
or other public companies in which you have made an investment.
In
addition, the JOBS Act provides that an emerging growth company may take advantage of an extended transition period for complying with new or revised accounting standards. We have
irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth
companies.
Corporate History and Information
Invuity, Inc. was incorporated in California in 2004 as Spotlight Surgical, Inc. We changed our name to Invuity, Inc. in
2007. We reincorporated in Delaware in May 2015. Our principal executive office is located at 444 De Haro Street Francisco, California 94107 and our telephone number is (415) 665-2100. Our
website address is www.invuity.com.
S-3
Table of Contents
THE OFFERING
|
|
|
|
|
Common stock offered by us
|
|
6,200,000 shares.
|
|
Common stock to be outstanding immediately after this offering
|
|
23,379,258 shares (or 23,979,258 shares if the underwriter exercises in full its option to purchase additional
shares).
|
|
Underwriter's option to purchase additional shares of common stock from us
|
|
We have granted the underwriter a 30-day option from the date of this prospectus supplement to purchase up to
600,000 additional shares of our common stock at the public offering price, less underwriting discounts and commissions.
|
|
Use of proceeds
|
|
We currently intend to use the net proceeds we receive from this offering for (i) the continued expansion of our sales
and marketing activities and (ii) working capital and other general corporate purposes. See "Use of Proceeds."
|
|
Risk factors
|
|
An investment in our common stock involves substantial risks. See "Risk Factors" beginning on page S-6 of this
prospectus supplement and in the documents incorporated by reference into this prospectus supplement for a discussion of factors that you should read and consider before investing in our common stock.
|
|
NASDAQ symbol
|
|
"IVTY."
|
The
number of shares of our common stock to be outstanding immediately after this offering is based on 17,179,258 shares of common stock outstanding as of December 31, 2017 and
excludes:
-
•
-
3,232,692 shares of common stock issuable upon exercise of options outstanding as of December 31, 2017 issued under our equity incentive
plans, at a weighted-average exercise price of $7.01 per share;
-
•
-
235,415 shares of common stock issuable upon exercise of warrants outstanding as of December 31, 2017 at a weighted-average exercise
price of $10.80 per share;
-
•
-
268,260 shares of common stock issuable upon the vesting of restricted stock units issued under our equity incentive plans that were
outstanding but unvested as of December 31, 2017;
-
•
-
929,940 shares of common stock reserved for future issuance under our 2015 Equity Incentive Plan as of December 31, 2017;
-
•
-
302,100 shares of common stock issuable upon exercise of options issued from January 1, 2018 to March 14, 2018 under our equity
incentive plans, at a weighted-average exercise price of $4.14 per share; and
-
•
-
177,985 shares of common stock issuable upon the vesting of restricted stock units issued from January 1, 2018 to March 14, 2018
under our equity incentive plans.
Unless
otherwise indicated, this prospectus supplement assumes:
-
•
-
no exercise of the underwriter's option to purchase additional shares; and
-
•
-
no exercise or cancellation of the outstanding options and warrants and no vesting or cancellation of the restricted stock units described
above.
S-4
Table of Contents
SUMMARY FINANCIAL DATA
The following tables set forth a summary of our historical financial data as of and for the periods indicated. We have derived the summary
statements of operations data for the years ended December 31, 2017, 2016 and 2015 and the summary balance sheet data as of December 31, 2017 and 2016 from our audited financial
statements in our Annual Report on Form 10-K for the year ended December 31, 2017 incorporated by reference in this prospectus supplement and the accompanying prospectus. We have derived
the summary balance sheet data as of December 31, 2015 from our audited financial statements that are incorporated by reference in the accompanying prospectus. Our historical results are not
necessarily indicative of our future results. The following summary financial data should be read in conjunction with "Management's Discussion and Analysis of Financial Condition and Results of
Operations" and our financial statements and related notes incorporated herein by reference.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
|
2017
|
|
2016
|
|
2015
|
|
|
Statements of Operations Data:
|
|
|
|
|
|
|
|
|
|
|
|
Revenue
|
|
$
|
39,619
|
|
$
|
32,461
|
|
$
|
21,031
|
|
|
Cost of goods sold
|
|
|
11,741
|
|
|
8,824
|
|
|
7,733
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
27,878
|
|
|
23,637
|
|
|
13,298
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses:
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
9,018
|
|
|
9,908
|
|
|
7,869
|
|
|
Selling, general and administrative
|
|
|
54,119
|
|
|
52,409
|
|
|
40,636
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating expenses
|
|
|
63,137
|
|
|
62,317
|
|
|
48,505
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss from operations
|
|
|
(35,259
|
)
|
|
(38,680
|
)
|
|
(35,207
|
)
|
|
Interest expense
|
|
|
(2,370
|
)
|
|
(2,018
|
)
|
|
(1,881
|
)
|
|
Interest income
|
|
|
222
|
|
|
133
|
|
|
28
|
|
|
Other expense, net
|
|
|
(208
|
)
|
|
(44
|
)
|
|
(510
|
)
|
|
Loss on extinguishment of debt
|
|
|
(2,303
|
)
|
|
—
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss and comprehensive loss
|
|
$
|
(39,918
|
)
|
$
|
(40,609
|
)
|
$
|
(37,570
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per common share, basic and diluted
|
|
$
|
(2.34
|
)
|
$
|
(2.73
|
)
|
$
|
(4.94
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted-average shares used to compute net loss per common share, basic and diluted
|
|
|
17,051,037
|
|
|
14,868,501
|
|
|
7,606,172
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31,
|
|
|
|
2017
|
|
2016
|
|
2015
|
|
|
Balance Sheet Data:
|
|
|
|
|
|
|
|
|
|
|
|
Cash, cash equivalents, and short-term investments
|
|
$
|
21,002
|
|
$
|
39,037
|
|
$
|
46,296
|
|
|
Working capital
|
|
|
22,678
|
|
|
41,235
|
|
|
49,314
|
|
|
Total assets
|
|
|
45,495
|
|
|
60,335
|
|
|
66,305
|
|
|
Total long-term debt
|
|
|
29,116
|
|
|
13,261
|
|
|
14,480
|
|
|
Accumulated deficit
|
|
|
(186,134
|
)
|
|
(146,216
|
)
|
|
(105,607
|
)
|
|
Total stockholders' equity (deficit)
|
|
$
|
(862
|
)
|
$
|
34,448
|
|
$
|
42,343
|
|
S-5
Table of Contents
RISK FACTORS
Investing in our common stock involves a high degree of risk. You should consider carefully the risks and uncertainties
described below, set forth under the caption "Risk Factors" in our Annual Report on Form 10-K for the year ended December 31, 2017 which is incorporated by reference in this prospectus
supplement and the accompanying prospectus and other information contained in or incorporated by reference in this prospectus supplement and the accompanying prospectus, including our consolidated
financial statements and the related notes, before you decide whether to purchase our common stock.
Risks Related to this Offering and Ownership of Our Common Stock
We currently are required to obtain additional funds to continue as a going concern. If we are unable to
obtain sufficient funds, we may be unable to invest in our sales and marketing efforts and research and development activities in order to grow our business. Additionally, we may not be able to secure
these funds on commercially reasonable terms, or at all.
We have experienced significant operating losses, and we expect to continue to incur operating losses for the next several years as we implement
additional initiatives designed to grow our business, including, among other things, increasing sales and developing new devices. We incurred net losses of $39.9 million, $40.6 million
and $37.6 million for the years ended December 31, 2017, 2016 and 2015, respectively. As of December 31, 2017, our accumulated deficit was $186.1 million. Our prior losses,
combined with expected future losses, have had and will continue to have, for the foreseeable future, an adverse effect on our stockholders' deficit and working capital. These factors raise
substantial doubt about our ability to continue as a going concern for the one year period from the date of issuance of the financial statements, included elsewhere in our Annual Report on
Form 10-K. To date, we have financed our operations primarily through the sale of equity securities, certain debt-related financing arrangements and from sales of our approved devices. We have
devoted substantially all of our resources to research and development of our devices, sales and marketing activities and certain clinical and quality assurance initiatives. Our ability to generate
sufficient revenue from our existing devices or from any of our device candidates in development, and to transition to profitability and generate consistent positive cash flows, is uncertain. We will
need to generate significant sales to achieve profitability, and we might not be able to do so. If our revenue grows more slowly than we anticipate or if we fail to continue to grow our revenue, or if
our operating expenses are higher than we expect, we may not be able to achieve profitability as anticipated, or ever, our financial condition will suffer and our stock price could decline. Even if we
achieve profitability in the future, we may not be able to sustain profitability in subsequent periods.
Our management might apply the proceeds of this offering in ways that do not increase the value of your
investment.
Our management will have broad discretion as to the use of the net proceeds of this offering and you will be relying on the judgment of our
management regarding the application of these proceeds. We might apply the net proceeds of this offering in ways with which you do not agree, or in ways that do not yield a favorable return. If our
management applies these proceeds in a manner that does not yield a significant return, if any, on our investment of these net proceeds, it would
adversely affect the market price of our common stock. For more information on our management's planned use of proceeds, please read "Use of Proceeds" elsewhere in this prospectus supplement. Pending
their use, we may also invest the net proceeds from this offering in a manner that does not produce income or that loses value.
S-6
Table of Contents
Purchasers in this offering will experience immediate and substantial dilution in the book value of their
shares.
The public offering price of our common stock in this offering is substantially higher than the net tangible book value per share of our common
stock before giving effect to this offering. Based on the public offering price of $3.50 per share and our net tangible book value as of December 31, 2017, if you purchase shares of common
stock in this offering, you will suffer immediate and substantial dilution of $2.69 per share. Furthermore, if outstanding options or warrants are exercised, you could experience further dilution. For
a further description of the dilution that you will experience immediately after this offering, see the section in this prospectus supplement entitled "Dilution."
You may experience future dilution as a result of future equity offerings.
To the extent that we raise additional funds through the sale of equity or securities that are convertible into or exchangeable for, or that
represent the right to receive, common stock or substantially similar securities, the issuance of such securities will result in dilution to our stockholders. We may sell shares or other securities in
any other offering at a price per share that is less than the price per share paid by investors in this offering, and investors purchasing shares or other securities in the future could have rights
superior to existing stockholders.
If our operating results and financial performance do not meet the guidance that we have provided to the
public, our stock price may decline.
We provide public guidance on our expected operating and financial results. Although we believe that this guidance provides our stockholders,
investors and analysts with a better understanding of our expectations for the future, such guidance is comprised of forward-looking statements which are subject to the risks and uncertainties
described in this report and in our other public filings and public statements. Our actual results may not meet the guidance we have provided. If our operating or financial results do not meet our
guidance or the expectations of investment analysts, our stock price may decline.
A substantial number of shares of common stock may be sold in the market following this offering, which may
depress the market price for our common stock.
We maintain a shelf registration statement on Form S-3 with the SEC pursuant to which we may, from time to time, sell up to an aggregate
of $100.0 million of our common stock, preferred stock, depositary shares, warrants, debt securities or units. We have sold, and plan in the future to sell, shares of our common stock in
underwritten offerings and have established, and may in the future establish, "at-the-market" offering programs pursuant to which we may offer and sell shares of our common stock.
Sales
of a substantial number of shares of our common stock in the public market following this offering could cause the market price of our common stock to decline. These sales, or the
perception in the market that our officers, directors or the holders of a large number of shares of our common stock intend to sell shares, could reduce the market price of our common stock. A
substantial majority of the outstanding shares of our common stock are, and the shares of common stock sold in this offering upon issuance will be, freely tradable without restriction or further
registration under the Securities Act of 1933, as amended, or the Securities Act. Other than our directors and executive officers, and certain stockholders affiliated with our directors, none of our
stockholders are subject to any lock-up agreement in connection with this offering.
We
cannot predict what effect, if any, sales of our shares in the public market or the availability of shares for sale will have on the market price of our common stock. Future sales of
substantial amounts of our common stock in the public market, including shares issued upon exercise of outstanding options
S-7
Table of Contents
or
warrants, or the perception that such sales may occur, however, could adversely affect the market price of our common stock and also could adversely affect our future ability to raise capital
through the sale of our common stock or other equity-related securities of ours at times and prices we believe appropriate.
Our stock price may be volatile, and you may not be able to resell shares of our common stock at or above the
price you paid.
Our stock price may be highly volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond our
control. These factors include those discussed in the "Risk Factors" section in our Annual Report on Form 10-K for the year ended December 31, 2017, which is incorporated by reference in
this prospectus supplement and the accompanying prospectus, and others such as:
-
•
-
variance in our financial performance from the financial projects we may provide to the public, any changes in these projections or our failure
to meet these projections;
-
•
-
changes in analysts' estimates, investors' perceptions, recommendations by securities analysts or our failure to achieve analysts' estimates;
-
•
-
announcements of significant new devices or device enhancements by us or our competitors;
-
•
-
actual or anticipated quarterly variations in our or our competitors' results of operations;
-
•
-
changes in operating performance and stock market valuations of other technology companies generally, or those in the medical device industry
in particular;
-
•
-
changes in our pricing policies or the pricing policies of our competitors;
-
•
-
legislation or regulatory policies, practices or actions affecting our business;
-
•
-
lawsuits threatened or filed against us;
-
•
-
the sale of our common stock or other securities in the future by us or our stockholders, including upon expiration of market standoff or
contractual lock-up agreements;
-
•
-
developments or disputes concerning our intellectual property or other proprietary rights;
-
•
-
announcements related to patents issued to us or our competitors and to litigation;
-
•
-
recruitment or departure of key personnel, including changes in our board of directors and management;
-
•
-
changes in market valuation or earnings of our competitors;
-
•
-
the trading volume of our common stock;
-
•
-
changes in the estimation of the future size and growth rate of our markets;
-
•
-
general market conditions and other factors unrelated to our operating performance or the operating performance of our competitors; and
-
•
-
developments in our industry.
In
addition, the market prices of the stock of many new issuers in the medical device industry and of other companies with smaller market capitalizations like us have been volatile and
from time to time have experienced significant share price and trading volume changes unrelated or disproportionate to the operating performance of those companies. In the past, stockholders have
filed securities class action litigation following periods of market volatility. If we were to become involved in securities litigation, it could subject us to substantial costs, divert resources and
the attention of management from our business, and adversely affect our business, results of operations, financial condition, reputation and cash flows. These factors may materially and adversely
affect the market price of our common stock.
S-8
Table of Contents
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus supplement and the accompanying prospectus contain or incorporate by reference "forward-looking statements" within the meaning
of Section 27A of the Securities Act, Section 21E of the Exchange Act, and the Private Securities Litigation Reform Act of 1995 that reflect our current views with respect to, among
other things, our operations and financial performance. Forward-looking statements include all statements that are not historical facts. In some cases, you can identify these forward-looking
statements by the use of words such as "believe," "anticipate," "expect," "intend," "plan," "focus," "assume," "goal," "objective," "will," "may" "should," "would," "could," "estimate," "predict,"
"potential," "continue," "encouraging" or the negative version of these words or other comparable words or similar expressions. We have based these forward-looking statements largely on our current
expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business
strategy and financial needs. Forward-looking statements include, but are not limited to, statements about:
-
•
-
our expectations regarding the potential market size and widespread adoption of our devices, including applications in additional surgical
specialties;
-
•
-
our ability to demonstrate to surgeons and hospitals the merits of our devices and timely obtain approval by hospitals to sell our devices;
-
•
-
developments and projections relating to our competitors or our industry;
-
•
-
the expected growth in our business and our organization, including outside of the United States;
-
•
-
our financial performance, our estimates of our expenses, ongoing losses, future revenue, capital requirements and our needs for, or ability to
obtain, additional financing;
-
•
-
our ability to retain and recruit key personnel, including the continued development and expansion of a sales and marketing infrastructure;
-
•
-
our ability to obtain an adequate supply of components for our devices from our third-party suppliers;
-
•
-
our ability to identify and develop new and planned devices;
-
•
-
our ability to obtain and maintain intellectual property protection for our devices or avoid claims of infringement;
-
•
-
our compliance with extensive government regulation;
-
•
-
our expected uses of the net proceeds from this offering;
-
•
-
our ability to continue as a going concern;
-
•
-
the volatility of our share price; and
-
•
-
our expectations regarding the time during which we will be an emerging growth company under the JOBS Act under the federal securities laws.
These
statements involve known and unknown risks, uncertainties, and other factors that may cause our actual results of operation, financial condition, levels of activity, performance or
achievements to be materially different from the information expressed or implied by these forward-looking statements. You should refer to the sections entitled "Risk Factors" in this prospectus
supplement, in our Annual Report on Form 10-K for the year ended December 31, 2017, which are incorporated by reference into
this prospectus supplement and the accompanying prospectus for a discussion of important risks that may cause our actual results to differ materially from those expressed or implied by our
forward-looking statements. Although we believe that the expectations reflected in
S-9
Table of Contents
the
forward-looking statements contained in this prospectus are reasonable, we caution you that these statements are based on a combination of facts and factors currently known by us and our
projections of the future, about which we cannot be certain. Moreover, we operate in a competitive, challenging and rapidly changing industry in which new risks may emerge from time to time, and it is
not possible for management to predict all risks and uncertainties that could have an impact on the forward-looking statements contained or incorporated by reference in this prospectus supplement and
the accompanying prospectus. We cannot assure you that the results, events and circumstances reflected in the forward-looking statements will be achieved or occur, and actual results, events, or
circumstances could differ materially from those described in the forward-looking statements. Forward-looking statements made or incorporated by reference in this prospectus supplement and the
accompanying prospectus relate only to events as of the date on which the statements are made. We undertake no obligation to publicly update or review any forward-looking statement, whether as a
result of new information, future developments or otherwise, except as required by law. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements
and you should not place undue reliance on our forward-looking statements. Our forward-looking statements do not reflect the potential impact of any future acquisitions, mergers, dispositions, joint
ventures, investments or other strategic transactions we may make.
S-10
Table of Contents
USE OF PROCEEDS
We estimate that the net proceeds of this offering, after deducting underwriting discounts and commissions and estimated offering expenses
payable by us, will be approximately
$19.8 million, or $21.7 million if the underwriter exercises in full its option to purchase additional shares. We cannot assure you that this offering will be completed.
We
currently intend to use the net proceeds we receive from this offering for (i) the continued expansion of our sales and marketing activities and (ii) working capital and
other general corporate purposes. The amounts and timing of our expenditures will depend upon numerous factors, including the rate of adoption of our devices, the expenses we incur in selling and
marketing our devices, the scope of our research and development efforts, the timing and success of clinical trials we may commence in the future, and the timing of regulatory submissions.
Accordingly, our management will have broad discretion over the use of the net proceeds of the offerings. Pending their ultimate use, we intend to invest the net proceeds in short-term,
interest-bearing, investment-grade securities, certificates of deposit or government securities.
S-11
Table of Contents
MARKET PRICE OF COMMON STOCK
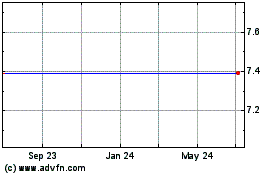
Our common stock has been listed on NASDAQ under the symbol "IVTY" since June 16, 2015. Prior to that date, there was no public trading
market for our common stock. Our common stock priced at $12.00 per share in our initial public offering on June 15, 2015. The following table sets forth for the periods indicated the high and
low intra-day sale prices per share of our common stock as reported on NASDAQ:
|
|
|
|
|
|
|
|
|
|
|
High
|
|
Low
|
|
|
2016
|
|
|
|
|
|
|
|
|
Second Quarter 2016
|
|
$
|
10.03
|
|
$
|
4.80
|
|
|
Third Quarter 2016
|
|
$
|
14.25
|
|
$
|
9.28
|
|
|
Fourth Quarter 2016
|
|
$
|
13.95
|
|
$
|
4.50
|
|
|
2017
|
|
|
|
|
|
|
|
|
First Quarter 2017
|
|
$
|
8.75
|
|
$
|
5.75
|
|
|
Second Quarter 2017
|
|
$
|
9.70
|
|
$
|
5.75
|
|
|
Third Quarter 2017
|
|
$
|
9.25
|
|
$
|
5.95
|
|
|
Fourth Quarter 2017
|
|
$
|
9.55
|
|
$
|
6.15
|
|
|
2018
|
|
|
|
|
|
|
|
|
First Quarter 2018 (through March 14, 2018)
|
|
$
|
6.30
|
|
$
|
3.30
|
|
On
March 14, 2018, the last reported sale price of our common stock on NASDAQ was $4.30 per share. On March 13, 2018, there were 17,207,944 shares of our common stock
outstanding. As of March 13, 2018, we had 36 holders of record of our common stock. The actual number of holders of common stock is greater than this number of record holders and includes
stockholders who are beneficial owners, but whose shares are held in street name by brokers and nominees. The number of holders of record also does not include stockholders whose shares may be held in
trust by other entities.
S-12
Table of Contents
DIVIDEND POLICY
We have never declared or paid any dividends on our capital stock. We currently intend to retain all of our future earnings, if any, to finance
our operations and do not anticipate paying any cash dividends on our capital stock in the foreseeable future. Future determination as to the declaration and payment of dividends, if any, will be at
the discretion of our board of directors and will depend on then existing conditions, including our operating results, financial conditions, contractual restrictions, capital requirements, business
prospects and other factors our board of directors may deem relevant. Our credit agreements with MidCap Financial Trust and affiliates contain restrictions on our ability to pay cash dividends.
S-13
Table of Contents
DILUTION
If you invest in our common stock in this offering, your ownership interest will be immediately diluted to the extent of the difference between
the public offering price per share of our common stock and the as adjusted net tangible book value per share of our common stock after this offering. As of December 31, 2017, our historical
net tangible book value was $(0.86) million, or $(0.05) per share. Our historical net tangible book value represents total tangible assets less total liabilities, all divided by the number of shares
of common stock outstanding on December 31, 2017.
After
giving effect to the sale of shares of common stock offered by us in this offering at the public offering price of $3.50 per share, and after deducting underwriting discounts and
commissions and
estimated offering expenses payable by us, our adjusted net tangible book value as of December 31, 2017 would have been approximately $18.9 million, or approximately $0.81 per share. This
represents an immediate increase in as adjusted net tangible book value of $0.86 per share to existing stockholders and an immediate dilution in as adjusted net tangible book value of approximately
$2.69 per share to new investors purchasing shares of common stock in this offering.
Dilution
per share to new investors is determined by subtracting as adjusted net tangible book value per share after this offering from the public offering price per share paid by new
investors. The following table illustrates this dilution on a per share basis:
|
|
|
|
|
|
|
|
|
|
Public offering price per share
|
|
|
|
|
$
|
3.50
|
|
|
Net tangible book value per share as of December 31, 2017
|
|
$
|
(0.05
|
)
|
|
|
|
|
Increase in net tangible book value per share attributable to this offering
|
|
$
|
0.86
|
|
|
|
|
|
As adjusted net tangible book value per share after giving effect to this offering
|
|
|
|
|
$
|
0.81
|
|
|
|
|
|
|
|
|
|
|
|
Dilution per share to new investors in this offering
|
|
|
|
|
$
|
2.69
|
|
|
|
|
|
|
|
|
|
|
The
information above assumes that the underwriter does not exercise its option to purchase additional shares. If the underwriter exercises in full its option to purchase additional
shares, our as adjusted net tangible book value per share at December 31, 2017, after giving effect to this offering, would have been approximately $0.87 per share, and the dilution in as
adjusted net tangible book value per share to investors in this offering would have been approximately $2.63 per share.
The
above discussion and table are based on 17,179,258 shares of common stock outstanding as of December 31, 2017 and excludes:
-
•
-
3,232,692 shares of common stock issuable upon exercise of options outstanding as of December 31, 2017 issued under our equity incentive
plans, at a weighted-average exercise price of $7.01 per share;
-
•
-
235,415 shares of common stock issuable upon exercise of warrants outstanding as of December 31, 2017 at a weighted-average exercise
price of $10.80 per share;
-
•
-
268,260 shares of common stock issuable upon the vesting of restricted stock units issued under our equity incentive plans that were
outstanding but unvested as of December 31, 2017;
-
•
-
929,940 shares of common stock reserved for future issuance under our 2015 Equity Incentive Plan as of December 31, 2017;
-
•
-
302,100 shares of common stock issuable upon exercise of options issued from January 1, 2018 to March 14, 2018 under our equity
incentive plans, at a weighted-average exercise price of $4.14 per share; and
-
•
-
177,985 shares of common stock issuable upon the vesting of restricted stock units issued from January 1, 2018 to March 14, 2018
under our equity incentive plans.
To
the extent any of these options or warrants are exercised or restricted stock units vest, there may be further dilution to new investors.
S-14
Table of Contents
MATERIAL UNITED STATES FEDERAL TAX CONSIDERATIONS FOR
NON-UNITED STATES HOLDERS
The following is a summary of the material United States federal income and estate tax consequences of the ownership and disposition of shares
of our common stock purchased pursuant to this offering by a non-United States holder, as we define that term below, but does not purport to be a complete analysis of all potential tax effects. This
summary is based upon current provisions of the Internal Revenue Code of 1986, as amended, or the Code, Treasury regulations promulgated thereunder, judicial opinions, administrative pronouncements
and published rulings of the United States Internal Revenue Service, or IRS, all as in effect as of the date hereof. These authorities may be changed, possibly retroactively, resulting in United
States federal tax consequences different from those set forth below or that could adversely affect a non-United States holder of our common stock. We have not sought, and will not seek, any ruling
from the IRS with respect to the statements made in the following summary, and there can be no assurance that the IRS will not take a position contrary to such statements or that any such contrary
position taken by the IRS would not be sustained.
This
summary is limited to non-United States holders who purchase shares of our common stock issued pursuant to this offering and who hold our common stock as a capital asset within the
meaning of Section 1221 of the Code (generally property held for investment) for United States federal income tax purposes. This summary does not purport to be complete and does not address the
tax considerations arising under the laws of any state, local or non-United States jurisdiction, or under United States federal estate or gift tax laws, except as specifically described below. In
addition, this summary does not address tax considerations that may be applicable to an investor's particular circumstances (including the impact of the Medicare contribution tax on net investment
income), nor does it address the special tax rules applicable to special classes of non-United States holders, including, without limitation:
-
•
-
banks, insurance companies or other financial institutions;
-
•
-
partnerships or other entities treated as partnerships for United States federal income tax purposes (and investors therein);
-
•
-
holders subject to the alternative minimum tax;
-
•
-
United States expatriates and former citizens or long-term residents of the United States;
-
•
-
tax-exempt organizations and governmental organizations;
-
•
-
tax-qualified retirement plans;
-
•
-
brokers or dealers in securities or currencies;
-
•
-
real estate investment trusts;
-
•
-
regulated investment companies;
-
•
-
mutual funds;
-
•
-
"controlled foreign corporations," "passive foreign investment companies," and corporations that accumulate earnings to avoid United States
federal income tax;
-
•
-
traders in securities that elect to use a mark-to-market method of accounting for their securities holdings;
-
•
-
persons deemed to sell our common stock under the constructive sale provisions of the Code;
-
•
-
persons that will hold common stock as a position in a hedging transaction, "straddle," "conversion," or other integrated transaction for tax
purposes; or
S-15
Table of Contents
-
•
-
persons who acquired ordinary shares through the exercise or cancellation of employee stock options or otherwise as compensation for their
services.
If
a partnership, including any entity treated as a partnership for United States federal income tax purposes, is a holder of shares of our common stock, the tax treatment of a partner
or member in the partnership or entity will generally depend upon the status of the partner, the activities of the partnership and certain determinations made at the partner level. A holder of shares
of our common stock that is a partnership, and partners in such partnership, are urged to consult their tax advisors regarding the tax consequences of the acquisition, ownership and disposition of
shares of our common stock.
For
purposes of this discussion, a "non-United States holder" is a beneficial owner of our common stock that is neither a United States person nor a partnership. A United States person
means a person who is for United States federal income tax purposes:
-
•
-
an individual who is a citizen or resident of the United States;
-
•
-
a corporation, including any entity treated as a corporation for United States federal income tax purposes, created or organized in or under
the laws of the United States, any state within the United States, or the District of Columbia;
-
•
-
an estate the income of which is subject to United States federal income taxation regardless of its source; or
-
•
-
a trust (1) if it is subject to the primary supervision of a court within the United States and one or more United States persons have
the authority to control all substantial decisions of the trust or (2) that has a valid election in effect under applicable Treasury regulations to be treated as a United States person.
An
individual is generally treated as a resident of the United States in any calendar year for United States federal income tax purposes if the individual is present in the United States
for at least 31 days in that calendar year and for an aggregate of at least 183 days during the three-year period ending on the last day of the current calendar year. For purposes of the
183-day calculation, all of the days present in the current year, one-third of the days present in the immediately preceding year and one-sixth of the days present in the second preceding year are
counted. Residents are generally taxed for United States federal income tax purposes as if they were United States citizens.
Non-United
States holders are urged to consult your tax advisor with respect to the application of the United States federal income tax laws to their particular situation as well as any
tax consequences arising under the United States federal estate or gift tax rules or under the laws of any state, local, non-United States or other taxing jurisdiction or under any applicable tax
treaty.
Distributions on Shares of Our Common Stock
As described in the section entitled "Dividend Policy," we do not anticipate declaring or paying dividends to holders of our common stock in the
foreseeable future. However, if distributions are paid on shares of our common stock, the distributions will constitute dividends for United States federal income tax purposes to the extent paid from
our current or accumulated earnings and profits, as determined under United States federal income tax principles. To the extent a distribution exceeds our current and accumulated earnings and profits,
it will constitute a return of capital that is applied against and reduces, but not below zero, the adjusted tax basis of such non-United States holder's shares in our common stock. Any remainder will
be treated as capital gain from the sale of shares of our common stock and will be treated as described below under "—Gain on Disposition of Our Common Stock." Dividends paid to a
non-United States holder generally will be subject to withholding of United States federal income tax at the rate of 30% or such lower rate as may be specified by an
S-16
Table of Contents
applicable
income tax treaty, the benefits of which a non-United States holder is eligible. However, if the dividend is effectively connected with the non-United States holder's conduct of a trade or
business in the United States and, where required by an applicable income tax treaty, is attributable to a United States permanent establishment or fixed base maintained by such non-United States
holder, the dividend will not be subject to any withholding tax, provided certain certification and disclosure requirements are met, as described below, but will be subject to United States federal
income tax imposed on net income on the same basis that applies to United States persons generally at the regular graduated rates. A corporate non-United States holder under certain circumstances also
may be subject to a branch profits tax equal to 30%, or such lower rate as may be specified by an applicable income tax treaty, the benefits of which a non-United States holder is eligible, on a
portion of its effectively connected earnings and profits for the taxable year, as adjusted for certain items. Non-United States holders are urged to consult their tax advisors regarding the potential
applicability of any income tax treaty.
To
claim the benefit of an applicable income tax treaty or to claim exemption from withholding because the income is effectively connected with the conduct of a trade or business in the
United States, a non-United States holder must provide to the applicable withholding agent a properly executed IRS Form W-8BEN or IRS Form W-8BEN-E (or other applicable documentation)
for treaty benefits or W-8ECI for effectively connected income, or such successor forms as the IRS designates, and certify under penalties of perjury that such holder is not a United States person
prior to the payment of distributions on our common stock. These forms must be periodically updated. Non-United States holders may obtain a refund or credit of any excess amounts withheld by timely
filing an appropriate claim with the IRS.
Gain on Disposition of Our Common Stock
A non-United States holder generally will not be subject to United States federal income tax, including withholding, on gain recognized on a
sale or other disposition of shares of our common stock unless any one of the following is true:
-
•
-
the gain is effectively connected with the non-United States holder's conduct of a trade or business in the United States and, where an
applicable income tax treaty applies, is attributable to a United States permanent establishment or fixed base maintained by such non-United States holder;
-
•
-
the non-United States holder is a nonresident alien individual present in the United States for 183 days or more in the taxable year of
the disposition and certain other requirements are met; or
-
•
-
our common stock constitutes a United States real property interest by reason of our status as a "United States real property holding
corporation," or USRPHC, for United States federal income tax purposes at any time during the shorter of (1) the period during which a non-United States holder holds our common stock and
(2) the five-year period ending on the date a non-United States holder disposes of our common stock.
Unless
an applicable income tax treaty provides otherwise, gain described in the first bullet point above will be subject to the United States federal income tax imposed on net income on
the same basis that applies to United States persons generally, at the regular graduated rates, but will generally not be subject to withholding. Corporate non-United States holders also may be
subject to a branch profits tax at a rate of 30% (or such lower rate specified by an applicable income tax treaty) on such gain, as adjusted for certain items. Gain described in the second bullet
point above will be subject to a flat 30% United States federal income tax (or such lower rate specified by an applicable income tax treaty), which may be offset by certain United States source
capital losses (even though the individual is not
S-17
Table of Contents
considered
a resident of the United States) provided the non-United States holder has timely filed United States federal income tax returns with respect to such losses.
With
respect to the third bullet point, we believe we are not currently, and will not become, a USRPHC for United States federal income tax purposes. However, because the determination
of whether we are a USRPHC depends on the fair market value of our United States real property interests relative to the fair market value of our other business assets, we cannot assure you that we
will not become a USRPHC in the future. If we are or were to become a USRPHC, as long as our common stock is "regularly traded," as defined by applicable Treasury regulations, on an established
securities market, it will not be treated as a United States real property interest with respect to any non-United States holder that holds, actually or constructively, no more than 5% of such
regularly traded common stock throughout the shorter of the five-year period ending on the date of the sale or other disposition or the non-United States holder's holding period for such stock, and
any gain arising from any such sale or taxable disposition will not be subject to United States federal income tax. If we are determined to be a USRPHC and the foregoing exception does not apply,
among other things, a purchaser may be required to withhold 15% of the proceeds payable to a non-United States holder from a disposition of shares of our common stock, and the non-United States holder
generally will be taxed on its net gain derived from the disposition at the graduated United States federal income tax rates applicable to United States persons.
Non-United
States holders are urged to consult any potentially applicable income tax treaties that may provide for different rules.
Legislation Involving Payments to Certain Foreign Entities
A 30% withholding tax applies to any dividends paid on shares of our common stock to a foreign financial institution or non-financial foreign
entity (including, in some cases, when such foreign financial institution or entity is acting as an intermediary), in each case as specially defined for purposes of these rules, and on the gross
proceeds of the sale or other disposition of shares of our common stock, unless (i) in the case of a foreign financial institution, such institution enters into an agreement with the United
States government to withhold on certain payments, and to collect and provide to the United States tax authorities substantial information regarding United States account holders of such institution
(which includes certain equity and debt holders of such institution, as well as certain account holders that are foreign entities with United States owners), (ii) in the case of a non-financial
foreign entity, such entity either certifies it does not have any substantial United States owners or provides the withholding agent with a certification identifying the substantial direct and
indirect United States owners of the entity or (iii) the foreign financial institution or non-financial foreign entity otherwise qualifies for an exemption from these rules. Under certain
circumstances, a
non-United States holder might be eligible for refunds or credits of such taxes. Foreign financial institutions located in jurisdictions that have an intergovernmental agreement with the United States
governing these withholding and reporting requirements may be subject to different rules.
Under
the applicable Treasury regulations and IRS guidance, these withholding and reporting requirements apply to payments of dividends on our common stock and will apply to payments of
gross proceeds from the sale or other disposition of such stock on or after January 1, 2019. Non-United States holders are urged to consult their tax advisors regarding the possible
implications of this legislation on an investment in shares of our common stock.
United States Federal Estate Taxes
Shares of our common stock owned or treated as owned by an individual who at the time of death is a non-United States holder are considered
United States situs assets and will be included in the
S-18
Table of Contents
individual's
estate for United States federal estate tax purposes, unless an applicable estate tax treaty provides otherwise.
Information Reporting and Backup Withholding
Information reporting and backup withholding (currently at a 24% rate) may apply to dividends paid with respect to, and the sale or other
disposition of, our common stock. In certain circumstances, provided the applicable withholding agent does not have knowledge or reason to know the holder is a United States person, non-United States
holders may avoid information reporting and backup withholding if they provide to the applicable withholding agent a properly executed applicable IRS Form W-8, or such successor forms as the
IRS designates, and certify under penalties of perjury as to their status as non-United States holders or otherwise establish an exemption and certain other requirements are met. Copies of information
returns may also be made available to the tax authorities in the country in which the non-United States holder resides or is established under the
provisions of an applicable income tax treaty. Non-United States holders are urged to consult their tax advisors regarding the application of the information reporting and backup withholding rules to
them.
Backup
withholding is not an additional tax. Amounts withheld under the backup withholding rules from a payment to a non-United States holder may be refunded in instances of excess
withholding or credited against the non-United States holder's United States federal income tax liability, if any, provided an appropriate claim is timely filed with the IRS.
Each prospective investor should consult its own tax advisor regarding the particular United States federal, state and local and non-United States tax
consequences of purchasing, holding and disposing of our common stock, including the consequences of any proposed change in applicable laws.
S-19
Table of Contents
UNDERWRITING
Subject to the terms and conditions set forth in the underwriting agreement, between us and William Blair & Company, L.L.C. as the sole
underwriter and the book-running manager of this offering, we have agreed to sell to the underwriter, and the underwriter has agreed to purchase from us, the number of shares of our common stock set
forth opposite its name below.
|
|
|
|
|
Name
|
|
Number
of Shares
|
|
William Blair & Company, L.L.C.
|
|
6,200,000
|
|
Total
|
|
6,200,000
|
|
|
|
|
Subject
to the terms and conditions set forth in the underwriting agreement, the underwriter has agreed to purchase all of the shares sold under the underwriting agreement if any of
these shares are purchased.
We
have agreed to indemnify the underwriter against certain liabilities, including liabilities under the Securities Act relating to losses or claims resulting from material misstatements
in or omissions from this prospectus supplement, the accompanying base prospectus, the registration statement of which such base prospectus is a part, certain free writing prospectuses that may be
used in the offering and in any marketing materials used in connection with this offering and to contribute to payments the underwriter may be required to make in respect of those liabilities.
Certain
of our executive officers and directors have indicated an interest in purchasing shares of our common stock in this offering at the public offering price. However, because
indications of interest are not binding agreements or commitments to purchase, the underwriter could determine to sell no shares to any of these potential purchasers, and any of these potential
purchasers could determine to purchase no shares in this offering. The underwriter will receive the same underwriting discounts and commissions on any shares purchased by these parties as they will on
any other shares sold to the public in this offering.
Discounts and Commissions
The underwriter has advised us that it proposes initially to offer the shares to the public at the public offering price set forth on the cover
page of this prospectus supplement and to dealers at that price less a concession not in excess of $0.147 per share. After the public offering, the public offering price, concession or any other term
of this offering may be changed.
The
following table shows the public offering price, underwriting discounts and commissions and proceeds before expenses to us. The information assumes either no exercise or full
exercise by the underwriter of its option to purchase additional shares.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Per Share
|
|
Without Option
|
|
With Option
|
|
|
Public offering price
|
|
$
|
3.50
|
|
$
|
21,700,000
|
|
$
|
23,800,000
|
|
|
Underwriting discounts and commissions
|
|
$
|
0.245
|
|
$
|
1,519,000
|
|
$
|
1,666,000
|
|
|
Proceeds, before expenses, to us
|
|
$
|
3.255
|
|
$
|
20,181,000
|
|
$
|
22,134,000
|
|
The
underwriting agreement provides that the obligations of the underwriter to pay for and accept delivery of the shares of common stock offered by this prospectus supplement are subject
to the approval of certain legal matters by its counsel and to certain other conditions. The underwriter is obligated to take and pay for all of the shares of common stock offered by this prospectus
supplement if any such shares are taken. However, the underwriter is not required to take or pay for the shares covered by the underwriter's option to purchase additional shares described above. The
underwriter
S-20
Table of Contents
reserves
the right to withdraw, cancel or modify offers to the public and to reject orders in whole or in part.
The
estimated offering expenses payable by us, exclusive of the underwriting discounts and commissions, are approximately $0.4 million, which includes legal, accounting and
printing costs and various other fees associated with the registration and listing of our common stock. We have agreed to reimburse the underwriter for certain of its expenses in an amount up to
$145,000, which amount is included in our estimated offering expenses.
Option to Purchase Additional Shares
We have granted to the underwriter an option, exercisable for 30 days from the date of this prospectus, to purchase, from time to time, in whole
or in part, up to an aggregate of 600,000 shares from us at the public offering price set forth on the cover page of this prospectus, less underwriting discounts and commissions. If the
underwriter exercises this option, the underwriter will be obligated, subject to specified conditions, to purchase a number of additional shares as indicated in the table above.
No Sales of Similar Securities
We, our executive officers and directors (and certain stockholders affiliated with our directors) have agreed not to sell or transfer any shares
of our common stock or securities convertible into, exchangeable or exercisable for, or that represent the right to receive shares of our common stock, for 90 days after the date of the
prospectus supplement used to sell our common stock without first obtaining the written consent of William Blair & Company, L.L.C. Specifically, we and these other persons have agreed, with
certain limited exceptions, not to directly or indirectly:
-
•
-
offer, pledge, announce the intention to sell, sell or contract to sell any shares of our common stock;
-
•
-
sell any option or contract to purchase any shares of our common stock;
-
•
-
purchase any option or contract to sell any shares of our common stock;
-
•
-
grant any option, right or warrant to purchase any shares of our common stock;
-
•
-
make any short sale or otherwise transfer or dispose of any shares of our common stock;
-
•
-
enter into any swap or other agreement that transfers, in whole or in part, the economic consequences of ownership of any shares of our common
stock whether any such swap or transaction is to be settled by delivery of shares or other securities, in cash or otherwise; or
-
•
-
demand that we file a registration statement related to our common stock.
In
our case, the restrictions in the preceding paragraph do not apply to:
-
•
-
the shares of our common stock to be sold in this offering;
-
•
-
issuances of shares of common stock upon the exercise of an option or warrant or conversion of a security outstanding as of the date of the
underwriting agreement and described in the prospectus supplement (or in documents filed with the SEC and incorporated by reference into this prospectus supplement);
-
•
-
issuances of securities pursuant to our equity incentive plans in effect on the date of the underwriting agreement and described in the
prospectus supplement (or in documents filed with the SEC and incorporated by reference into this prospectus supplement);
S-21
Table of Contents
-
•
-
the filing by us of a registration statement on Form S-8 with respect to our equity incentive plans in effect on the date of the
underwriting agreement and described in the prospectus supplement (or in documents filed with the SEC and incorporated by reference into this prospectus supplement); or
-
•
-
the sale or issuance of or entry into any agreement providing for the issuance of securities in connection with any acquisition, joint venture,
commercial relationship or strategic transaction; provided that the aggregate number of shares of common stock that we may sell or issue or agree to sell or issue may not exceed 5% of the total number
of shares of common stock issued and outstanding immediately following the completion of the transactions contemplated by the underwriting agreement.
In
the case of our executive officers and directors (and certain stockholders affiliated with our directors), and subject to certain conditions, such restrictions do not apply to
transfers of securities:
-
•
-
as a bona fide gift or gifts;
-
•
-
to an immediate family member or any trust for the direct or indirect benefit of the stockholder or an immediate family member of the
stockholder;
-
•
-
if the stockholder is a corporation, partnership, limited liability company, investment fund, trust or other business entity
(i) transfers to another corporation, partnership, limited liability company, investment fund, trust or other business entity that is a direct or indirect affiliate of the stockholder or
(ii) distributions of shares of our common stock to limited partners, limited liability company members or stockholders of the stockholder, or to any investment fund or other entity that
controls or manages the stockholder;
-
•
-
if the stockholder is a trust, to the beneficiary of such trust;
-
•
-
by testate succession or intestate succession; or
-
•
-
pursuant to the underwriting agreement;
provided,
in the case of a transfer described in the first five bullets above, that such transfer does not involve a disposition for value, and each transferee agrees to be subject to the restrictions
described in the immediately preceding paragraph and that no filing by any party under Section 16(a) of the Exchange Act, shall be required or shall be made voluntarily in connection with such
transfer other than a required Form 5 filing filed within 45 days of December 31, 2018, in which case such Form 5 shall include a footnote describing the transaction being
reported.
In
addition, the transfer restrictions described above do not apply to:
-
•
-
the exercise of stock options granted pursuant to our equity plans or warrants described in this prospectus supplement (or in documents filed
with the SEC and incorporated by reference into this prospectus supplement); provided that no filing by any party under Section 16(a) of the Exchange Act shall be required or shall be made
voluntarily;
-
•
-
forfeitures to satisfy tax withholding obligations in connection with the conversion or exercise of our options or warrants; provided that if
the stockholder is required to file a report under Section 16(a) of the Exchange Act reporting a reduction in beneficial ownership of shares of common stock, the stockholder will include
a statement in such report to the effect that the purpose of the transfer was to cover tax withholding obligations;
-
•
-
transactions relating to shares of our common stock or other securities acquired in open market transactions on or after the date of this
prospectus supplement; provided that no filing by any party under Section 16(a) of the Exchange Act shall be required or shall be made voluntarily in connection with subsequent sales of our
common stock acquired in such open market transactions;
S-22
Table of Contents
-
•
-
transfers upon a termination of employment of shares of our common stock or any securities to us in connection with the repurchase of shares of
our common stock issued pursuant to an employee benefit plan disclosed in this prospectus supplement (or in documents filed with the SEC and incorporated by reference into this prospectus supplement)
or pursuant to the agreements pursuant to which such shares were issued as disclosed in this prospectus (or in documents filed with the SEC and incorporated by reference into this prospectus
supplement);
-
•
-
transfers pursuant to a "change of control" of our company;
-
•
-
transfers of shares of our common stock or other securities by operation of law to a spouse, former spouse, domestic partner, former domestic
partner, child or other dependent pursuant to a qualified domestic order or in connection with a divorce settlement; provided, that if the undersigned is required to file a report under
Section 16(a) of the Exchange Act reporting a reduction in beneficial ownership of shares of our common stock, the stockholder shall include a statement in such report to the effect that the
transfer occurred by operation of law, such as pursuant to a qualified domestic order or in connection with a divorce settlement, as applicable;
-
•
-
the establishment of any plan designed to satisfy all of the requirements under Rule 10b5-1 under the Exchange Act, provided that no
sales of the stockholder's common stock will be made under such plans for 90 days after the date of this prospectus supplement; or
-
•
-
the sale or disposal of shares of common stock pursuant to a plan that satisfies all of the requirements of Rule 10b5-1 under the
Exchange Act and that is in effect as of the date of the underwriting agreement.
Listing
Our common stock is listed on the NASDAQ Global Market under the trading symbol "IVTY".
Price Stabilization
Until the distribution of the shares is completed, SEC rules may limit underwriters and selling group members from bidding for and purchasing
shares of our common stock. However, the underwriter may engage in transactions that stabilize the price of our common stock, such as bids or purchases to peg, fix or maintain that price.
In
connection with this offering, the underwriter may purchase and sell shares of our common stock in the open market. These transactions may include short sales, purchases on the open
market to cover positions created by short sales and stabilizing transactions. Short sales involve the sale by the underwriter of a greater number of shares than they are required to purchase in this
offering. "Covered" short sales are sales made in an amount not greater than the underwriter's option to purchase additional shares described above. The underwriter may close out any covered short
position by either exercising their option to purchase additional shares or purchasing shares in the open market. In determining the source of shares to close out the covered short position, the
underwriter will consider, among other things, the price of shares available for purchase in the open market as compared to the price at which they may purchase shares through the option to purchase
additional shares. "Naked" short sales are sales in excess of the option to purchase additional shares. The underwriter must close out any naked short position by purchasing shares in the open market.
A naked short position is more likely to be created if the underwriter is concerned that there may be downward pressure on the price of our common stock in the open market after pricing that could
adversely affect investors who purchase in this offering. Stabilizing transactions consist of various bids for or purchases of shares of our common stock made by the underwriter in the open market
prior to the closing of this offering.
S-23
Table of Contents
Similar
to other purchase transactions, the underwriter's purchases to cover its short sales may have the effect of raising or maintaining the market price of our common stock or
preventing or retarding a decline in the market price of our common stock. As a result, the price of our common stock may be higher than the price that might otherwise exist in the open market. The
underwriter may conduct these transactions on the NASDAQ Global Market, in the over-the-counter market or otherwise.
Neither
we nor the underwriter make any representation or prediction as to the direction or magnitude of any effect that the transactions described above may have on the price of our
common stock. In addition, neither we nor the underwriter make any representation that the representative will engage in these transactions or that these transactions, once commenced, will not be
discontinued without notice.
Electronic Offer, Sale and Distribution of Shares
In connection with this offering, the underwriter may distribute prospectuses by electronic means, such as e-mail. In addition, the underwriter
may facilitate Internet distribution for this offering to certain of their Internet subscription customers. The underwriter may allocate a limited
number of shares for sale to its online brokerage customers. An electronic prospectus is available on the Internet websites maintained by any such underwriter. Other than the prospectus in electronic
format, the information on the website of the underwriter is not part of this prospectus.
Other Relationships
The underwriter and its affiliates are full service financial institutions engaged in various activities, which may include securities trading,
commercial and investment banking, financial advisory, investment management, investment research, principal investment, hedging, financing and brokerage activities. The underwriter and its affiliates
have engaged in, and may in the future engage in, investment banking and other commercial dealings in the ordinary course of business with us or our affiliates. They have received, or may in the
future receive, customary fees and commissions for these transactions.
In
the ordinary course of their various business activities, the underwriter and its affiliates may make or hold a broad array of investments and actively trade debt and equity
securities (or related derivative securities) and financial instruments (including bank loans) for their own account and for the accounts of their customers, and such investment and securities
activities may involve securities and/or instruments of the issuer. The underwriter and its affiliates may also make investment recommendations and/or publish or express independent research views in
respect of such securities or instruments and may at any time hold, or recommend to clients that they acquire, long and/or short positions in such securities and instruments.
Selling Restrictions
European Economic Area
In relation to each Member State of the European Economic Area that has implemented the Prospectus Directive, each referred to as a Relevant
Member State, an offer to the public of any securities which are the subject of the offering contemplated by this prospectus may not
be made in that Relevant Member State, except that an offer to the public in that Relevant Member State of any securities may be made at any time under the following exemptions under the Prospectus
Directive:
-
(a)
-
to
any legal entity which is a "qualified investor" as defined in the Prospectus Directive;
-
(b)
-
to
fewer than 150 natural or legal persons (other than qualified investors as defined in the Prospectus Directive), subject to obtaining the prior consent of the
underwriters for any such offer; or
S-24
Table of Contents
-
(c)
-
in
any other circumstances falling within Article 3(2) of the Prospectus Directive,
provided
that no such offer of securities shall require us or any of the underwriters to publish a prospectus pursuant to Article 3 of the Prospectus Directive.
For
the purposes of this provision, the expression "an offer to the public" in relation to any securities in any Relevant Member State means the communication in any form and by any
means of sufficient information on the terms of the offer and the securities to be offered so as to enable an investor to decide to purchase or subscribe to the securities, as the same may be varied
in that Relevant Member State by any measure implementing the Prospectus Directive in that Relevant Member State and the expression "Prospectus Directive" means Directive 2003/71/EC (and amendments
thereto, including by Directive 2010/73/EU) and includes any relevant implementing measure in each Relevant Member State.
United Kingdom
The underwriter has represented and agreed that:
Canada
Resale Restrictions
The distribution of shares of our common stock in Canada is being made only in the provinces of Ontario, Quebec, Alberta and British Columbia on
a private placement basis exempt from the requirement that we prepare and file a prospectus with the securities regulatory authorities in each province where trades of these securities are
made. Any resale of shares of our common stock in Canada must be made under applicable securities laws which may vary depending on the relevant jurisdiction, and which may require resales to be made
under available statutory exemptions or under a discretionary exemption granted by the applicable Canadian securities regulatory authority. Purchasers are advised to seek legal advice prior to any
resale of the securities.
S-25
Table of Contents
Representations of Canadian Purchasers
By purchasing shares of our common stock in Canada and accepting delivery of a purchase confirmation, a purchaser is representing to us and the
dealer from whom the purchase confirmation is received that:
-
•
-
the purchaser is entitled under applicable provincial securities laws to purchase shares of our common stock without the benefit of a
prospectus qualified under those securities laws as it is an "accredited investor" as defined under National Instrument 45-106—Prospectus Exemptions,
-
•
-
the purchaser is a "permitted client" as defined in National Instrument 31-103—Registration Requirements, Exemptions and
Ongoing Registrant Obligations,
-
•
-
where required by law, the purchaser is purchasing as principal and not as agent, and
-
•
-
the purchaser has reviewed the text above under Resale Restrictions.
Conflicts of Interest
Canadian purchasers are hereby notified that the underwriter is relying on the exemption set out in section 3A.3 or 3A.4, if applicable,
of National Instrument 33-105—
Underwriting Conflicts
from having to provide certain conflict of interest disclosure in this document.
Statutory Rights of Action
Securities legislation in certain provinces or territories of Canada may provide a purchaser with remedies for rescission or damages if the
offering memorandum (including any amendment thereto) such as this document contains a misrepresentation, provided that the remedies for rescission or damages are exercised by the purchaser within the
time limit prescribed by the securities legislation of the purchaser's province or territory. The purchaser of these securities in Canada should refer to any applicable provisions of the securities
legislation of the purchaser's province or territory for particulars of these rights or consult with a legal advisor.
Enforcement of Legal Rights
All of our directors and officers as well as the experts named herein may be located outside of Canada and, as a result, it may not be possible
for Canadian purchasers to effect service of process within Canada upon us or those persons. All or a substantial portion of our assets and the assets of those persons may be located outside of Canada
and, as a result, it may not be possible to satisfy a judgment against us or those persons in Canada or to enforce a judgment obtained in Canadian courts against us or those persons outside of Canada.
Taxation and Eligibility for Investment
Canadian purchasers of our shares of common stock should consult their own legal and tax advisors with respect to the tax consequences of an
investment in shares of our common stock in their particular circumstances and about the eligibility of shares of our common stock for investment by the purchaser under relevant Canadian legislation.
S-26
Table of Contents
Hong Kong
No securities have been offered or sold, and no securities may be offered or sold, in Hong Kong, by means of any document, other than to persons
whose ordinary business is to buy or sell shares or debentures, whether as principal or agent; or to "professional investors" as defined in the Securities and Futures Ordinance (Cap. 571) of Hong Kong
and any rules made under that Ordinance; or in other circumstances which do not result in the document being a "prospectus" as defined in the Companies Ordinance (Cap. 32) of Hong Kong or which
do not constitute an offer to the public within the meaning of the Companies Ordinance (Cap. 32) of Hong Kong. No document, invitation or advertisement relating to the securities has been
issued or may be issued or may be in the possession of any person for the purpose of issue (in each case whether in Hong Kong or elsewhere), which is directed at, or the contents of which are likely
to be accessed or read by, the public of Hong Kong (except if permitted under the securities laws of Hong Kong) other than with respect to securities which are or are intended to be disposed of only
to persons outside Hong Kong or only to "professional investors" as defined in the Securities and Futures Ordinance (Cap. 571) of Hong Kong and any rules made under that Ordinance.
This
prospectus has not been registered with the Registrar of Companies in Hong Kong. Accordingly, this prospectus may not be issued, circulated or distributed in Hong Kong, and the
securities may not be offered for subscription to members of the public in Hong Kong. Each person acquiring the securities will be required, and is deemed by the acquisition of the securities, to
confirm that he is aware of the restriction on offers of the securities described in this prospectus and the relevant offering documents and that he is not acquiring, and has not been offered any
securities in circumstances that contravene any such restrictions.
Singapore
This prospectus has not been registered as a prospectus with the Monetary Authority of Singapore. Accordingly, this prospectus and any other
document or material in connection with the offer or sale, or invitation for subscription or purchase, of the common shares may not be circulated or distributed, nor may the shares of common stock be
offered or sold, or be made the subject of an invitation for subscription or purchase, whether directly or indirectly, to persons in Singapore other than (i) to an institutional investor under
Section 274 of the Securities and Futures Act, Chapter 289 of Singapore, or the SFA, (ii) to a relevant person pursuant to Section 275(1), or any person pursuant to
Section 275(1A) of the SFA, and in accordance with the conditions specified in Section 275 of the SFA or (iii) otherwise pursuant to, and in accordance with the conditions of, any
other applicable provision of the SFA, in each case subject to compliance with conditions set forth in the SFA.
Where
the shares of common stock are subscribed or purchased under Section 275 of the SFA by a relevant person which is:
-
(a)
-
a
corporation (which is not an accredited investor as defined in Section 4A of the SFA) the sole business of which is to hold investments and the entire share
capital of which is owned by one or more individuals, each of whom is an accredited investor; or
-
(b)
-
a
trust (where the trustee is not an accredited investor) whose sole purpose is to hold investments and each beneficiary of the trust is an individual who is an
accredited investor,
securities
(as defined in Section 239(1) of the SFA) of that corporation or the beneficiaries' rights and interest (howsoever described) in that trust shall not be transferred for six months
after that
S-27
Table of Contents
corporation
or that trust has acquired the common shares pursuant to an offer made under Section 275 of the SFA except:
-
(a)
-
to
an institutional investor under Section 274 of the SFA or to a relevant person defined in Section 275(2) of the SFA, or to any person pursuant to an
offer referred to in Section 275(1A) or Section 276(4)(i)(B) of the SFA;
-
(b)
-
where
no consideration is or will be given for the transfer;
-
(c)
-
where
the transfer is by operation of law;
-
(d)
-
as
specified in Section 276(7) of the SFA; or
-
(e)
-
as
specified in Regulation 32 of the Securities and Futures (Offers of Investments) (Shares and Debentures) Regulations 2005 of Singapore.
Switzerland
The shares of common stock may not be publicly offered in Switzerland and will not be listed on the SIX Swiss Exchange, or SIX, or on any other
stock exchange or regulated trading facility in Switzerland. This document has been prepared without regard to the disclosure standards for issuance prospectuses under art. 652a or art. 1156 of the
Swiss Code of Obligations or the disclosure standards for listing prospectuses under art. 27 ff. of the SIX Listing Rules or the listing rules of any other stock exchange or regulated trading facility
in Switzerland. Neither this document nor any other
offering or marketing material relating to the common shares or the offering may be publicly distributed or otherwise made publicly available in Switzerland.
Neither
this document nor any other offering or marketing material relating to the offering, or the shares of common have been or will be filed with or approved by any Swiss regulatory
authority. In particular, this document will not be filed with, and the offer of shares of common stock will not be supervised by, the Swiss Financial Market Supervisory Authority FINMA, and the offer
of shares of common stock has not been and will not be authorized under the Swiss Federal Act on Collective Investment Schemes, or CISA. Accordingly, no public distribution, offering or advertising,
as defined in CISA, its implementing ordinances and notices, and no distribution to any non-qualified investor, as defined in CISA, its implementing ordinances and notices, shall be undertaken in or
from Switzerland, and the investor protection afforded to acquirers of interests in collective investment schemes under CISA does not extend to acquirers of shares of common stock.
United Arab Emirates
This offering has not been reviewed, approved or licensed by the Central Bank of the United Arab Emirates (the "UAE"), the Emirates Securities
and Commodities Authority of the UAE (the "SCA") and/or any other relevant licensing authority in the UAE including any licensing authority incorporated under the laws and regulations of any of the
free zones established and operating in the territory of the UAE (the "Free Zones"), in particular the Dubai Financial Services Authority (the "DFSA"), a regulatory authority of the Dubai
International Financial Centre the ("DIFC") or the Financial Services Regulatory Authority (the "FSRA"), a regulatory authority of Abu Dhabi Global Market ("ADGM").
This
offering is not intended to, and does not, constitute an offer, sale or delivery of shares or other securities under the laws of the UAE. The common stock has not been and will not
be registered with or licensed by the SCA or with the UAE Central Bank, the Dubai Financial Market, the Abu Dhabi Securities Exchange or with any other UAE regulatory authority or exchange.
The
issue and/or sale of the common stock has not been approved or licensed by the SCA, the UAE Central Bank or any other relevant licensing authority in the UAE, and does not constitute
a
S-28
Table of Contents
public
offer of securities in the UAE, DIFC, ADGM and/or any other Free Zone in accordance with the Commercial Companies Law, Federal Law No 2 of 2015 (as amended), the Markets Rules of the DFSA, (the
"DFSA Markets Rules"), the Markets Rules of the FSRA (the "FSRA Markets Rules") and/or Nasdaq Dubai Listing Rules or under any other law of the UAE. The common stock may not be offered to the public
in the UAE and/or any of the Free Zones.
No
marketing or promotion of the common stock has been or will be made from within the UAE and no sale of or subscription for the common stock may or will be consummated within the UAE.
It should not be assumed that Invuity, Inc., Invuity, Inc.'s advisors, their advisors or any other person is a licensed broker, dealer or investment adviser under the laws of the UAE or
that they advise as to the appropriateness of investing in or purchasing or selling securities or other financial products.
This
offering is not intended to constitute a financial promotion, an offer, sale or delivery of shares or other securities under the DIFC Markets Law (DIFC Law No. 1 of 2012, as
amended) (the "Markets Law"), the DFSA Markets Rules, the Collective Investment Law 2010 (DIFC Law No. 2 of 2010) (the "Collective Investment Law"), the ADGM Financial Services and Markets
Regulations 2015 (the "FSMR"), the FSRA Markets Rules, the Funds Rules of the FSRA ("FSRA Funds Rules"), or any other laws and regulations of the DIFC, the DFSA, ADGM or the FSRA.
This
offering and the issue or transfer of any securities related to it have not been approved or licensed by the DFSA, and do not constitute an offer of securities in the DIFC in
accordance with the Markets Law or the DFSA Markets Rules or the Collective Investment Law or any other laws and regulations of the DIFC or the DFSA. This offering and the issue or transfer of any
securities related to it have not been approved or licensed by the FSRA, and do not constitute an offer of securities in ADGM in accordance with the FSMR or the FSRA Markets Rules or the FSRA Funds
Rules or any other laws and regulations of ADGM or the FSRA.
France
This prospectus (including any amendment, supplement or replacement thereto) is not being distributed in the context of a public offering of
financial instruments (
offre au public de titres financiers
) in France within the meaning of Article L. 411-1 of the French Monetary and
Financial Code (
Code monétaire et financier
) and Articles 211-1
et seq.
of the
General Regulations of the French
Autorité des marchés financiers
(the "AMF"). The common stock have not been offered or
sold and will not be offered or sold, directly or indirectly, to the public in France.
This
prospectus and any other offering material relating to the common stock have not been, and will not be, submitted to the AMF for approval in France and, accordingly, may not and
will not be distributed or caused to be distributed, directly or indirectly, to the public in France.
Pursuant
to Article 211-3 of the AMF General Regulations, French residents are hereby informed that:
-
(a)
-
the
transaction does not require a prospectus to be submitted for approval to the AMF;
-
(b)
-
the
offer, sale and distribution of the financial instruments shall only be made in France to (i) qualified investors (
investisseurs
qualifiés
) acting for their own account, as defined in and in accordance with Articles L. 411-2-II-2° and D. 411-1, D. 411-2, D. 734-1, D.
744-1, D. 754-1 and D. 764-1 of the French Monetary and Financial Code and any implementing regulation and/or (ii) a restricted number of non-qualified investors
(
investisseurs non-qualifiés
) acting for their own account, as defined in and in accordance with Articles L.
411-2-II-2° and D. 411-4, D. 734-1, D. 744-1, D. 754-1 and D. 764-1 of the French Monetary and Financial Code and any implementing regulation; and
S-29
Table of Contents
-
(c)
-
the
financial instruments thus acquired cannot be distributed directly or indirectly to the public otherwise than in accordance with Articles L. 411-1, L.
411-2, L. 412-1 and L. 621-8 to L. 621-8-3 of the Monetary and Financial Code.
This
prospectus is not to be further distributed or reproduced (in whole or in part) in France by the recipients of this prospectus. This prospectus has been distributed on the
understanding that such recipients will only participate in the issue or sale of our common stock for their own account and undertake not to transfer, directly or indirectly, our common stock to the
public in France, other than in compliance with all applicable laws and regulations and in particular with Articles L. 411-1 and L. 411-2 of the French Monetary and Financial Code and
Articles 211-1
et seq
. of the AMF General Regulations.
Israel
The securities offered by this prospectus supplement and the accompanying prospectus have not been approved or disapproved by the Israeli
Securities Authority (the "ISA"), nor have such securities been registered for sale in Israel. The ISA has not issued permits, approvals or licenses in connection with the offering or publishing this
prospectus supplement and the accompanying prospectus; nor has it authenticated the details included herein, confirmed their reliability or completeness, or rendered an opinion as to the quality of
the securities being offered. The ordinary shares will not be offered or sold, directly or indirectly, to the public in Israel, except that the underwriter may offer and sell such shares to Israeli
investors who qualify, in accordance with the Israeli Securities Law as "qualified investors" (as defined in the First Appendix to the Israeli Securities Law) and completed and signed a questionnaire
regarding such qualification and delivered it to the underwriter. Any resale in Israel, directly or indirectly, to the public of the securities offered by this prospectus supplement and the
accompanying prospectus is subject to restrictions on transferability and must be effected only in compliance with the Israeli securities laws and regulations.
S-30
Table of Contents
LEGAL MATTERS
The validity of the shares of common stock offered hereby and certain other legal matters will be passed upon for us by Wilson Sonsini
Goodrich & Rosati, Professional Corporation, Palo Alto, California. Certain legal matters will be passed upon for the underwriter by Latham & Watkins LLP, Chicago, Illinois.
EXPERTS
The financial statements incorporated in this prospectus supplement by reference to the Annual Report on Form 10-K for the year ended
December 31, 2017 have been so incorporated in reliance on the report of PricewaterhouseCoopers LLP (which contains an explanatory paragraph relating to the Company's ability to continue
as a going concern as described in Note 1 to the financial statements), an independent registered public accounting firm, given on the authority of said firm as experts in auditing and accounting.
WHERE YOU CAN FIND MORE INFORMATION
We file annual, quarterly and current reports and proxy statements and other information with the SEC. You may read and copy any document we
file at the SEC's Public Reference Room located at 100 F Street, N.E., Washington, D.C. 20549. You may obtain information on the operation of the public reference room by calling the SEC at
1-800-SEC-0330. The SEC also maintains a web site that contains reports, proxy and information statements and other information about issuers, such as us, who file electronically with the SEC. The
address of that website is http://www.sec.gov.
This
prospectus supplement and the accompanying prospectus are part of a registration statement that we filed with the SEC. The registration statement contains more information than this
prospectus regarding us and our common stock, including certain exhibits and schedules. You can obtain a copy of the registration statement from the SEC at the address listed above or from the SEC's
Internet website.
S-31
Table of Contents
INCORPORATION BY REFERENCE
The SEC allows us to "incorporate by reference" into this prospectus supplement and the accompanying prospectus the information we file with the
SEC in other documents, which means that we can disclose important information to you by referring you to those documents that contain such information. Any statement contained or incorporated by
reference in this prospectus supplement and the accompanying prospectus shall be deemed to be modified or superseded for purposes of this prospectus supplement and the accompanying prospectus to the
extent that a statement contained herein, or in any subsequently filed document which also is incorporated by reference herein, modifies or supersedes such earlier statement. Any such statement so
modified or superseded shall not be deemed, except as so modified or superseded, to constitute a part of this prospectus.
We
incorporate by reference the documents listed below:
-
•
-
our Annual Report on Form 10-K for the year ended December 31, 2017, filed with the SEC on March 5, 2018;
-
•
-
our Current Report on Form 8-K filed with the SEC on March 6, 2018; and
-
•
-
our Registration Statement on Form 8-A filed with the SEC on June 5, 2015.
In
addition, all documents that we file with the SEC pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of this prospectus supplement and before all
of the securities offered by this prospectus supplement are sold are incorporated by reference in this prospectus supplement from the date of filing of the documents, unless we specifically provide
otherwise in each case (excluding any
information furnished and not filed with the SEC). Information that we file with the SEC will automatically update and may replace information previously filed with the SEC.
You
may obtain, without charge, a copy of any of the documents incorporated by reference in this prospectus supplement and the accompanying prospectus, other than exhibits to those
documents that are not specifically incorporated by reference into those documents, by writing or telephoning us at the following address: Invuity, Inc., 444 De Haro Street Francisco,
California 94107, phone number (415) 665-2100.
Information
contained on our website, www.invuity.com, is not a prospectus and does not constitute part of this prospectus supplement and the accompanying prospectus. You should rely
only on the information incorporated by reference or provided in this prospectus supplement and the accompanying prospectus. We have not authorized anyone else to provide you with any information. You
should not assume that the information incorporated by reference or provided in this prospectus supplement and the accompanying prospectus are accurate as of any date other than the date on the front
of each document.
S-32
Table of Contents
PROSPECTUS
$100,000,000
Invuity, Inc.
By this prospectus, Invuity may offer, from time to time:
|
|
|
|
|
|
|
|
|
•
Common stock
|
|
|
|
|
|
•
Preferred stock
|
|
|
|
|
|
•
Depositary shares
|
|
|
|
|
|
•
Warrants
|
|
|
|
|
|
•
Units
|
|
|
Invuity, Inc.,
a Delaware corporation ("Invuity"), may offer and sell from time to time, in one or more series or issuances and on terms that Invuity will determine at the time of
the offering, any combination of the securities described in this prospectus, up to an aggregate amount of $100,000,000.
We
will provide specific terms of any offering in a supplement to this prospectus. Any prospectus supplement may also add, update, or change information contained in this prospectus. You
should carefully read this prospectus and the applicable prospectus supplement as well as the documents incorporated or deemed to be incorporated by reference in this prospectus before you purchase
any of the securities offered hereby.
These
securities may be offered and sold in the same offering or in separate offerings; to or through underwriters, dealers, and agents; or directly to purchasers. The names of any
underwriters, dealers, or agents involved in the sale of our securities, their compensation and any over-allotment options held by them will be described in the applicable prospectus supplement. For a
more complete description of the plan of distribution of these securities, see the section entitled "Plan of Distribution" beginning on page 18 of this prospectus.
Our
common stock is listed on the NASDAQ Global Market under the symbol "IVTY." We will provide information in any applicable prospectus supplement regarding any listing of securities
other than shares of our common stock on any securities exchange.
We are an "emerging growth company" as defined under federal securities laws and are subject to reduced company reporting requirements.
INVESTING IN OUR SECURITIES INVOLVES SIGNIFICANT RISKS. SEE "
RISK FACTORS
" BEGINNING ON PAGE 6 OF THIS PROSPECTUS
AND ANY SIMILAR SECTION CONTAINED IN THE APPLICABLE PROSPECTUS SUPPLEMENT BEFORE INVESTING IN ANY SECURITIES.
NEITHER THE SECURITIES AND EXCHANGE COMMISSION NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED OF THESE SECURITIES OR PASSED UPON THE ADEQUACY OR
ACCURACY OF THIS PROSPECTUS. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENSE.
The date of this prospectus is July 21, 2016
Table of Contents
TABLE OF CONTENTS
i
Table of Contents
ABOUT THIS PROSPECTUS
This prospectus is part of a registration statement on Form S-3 that we filed with the United States Securities and Exchange Commission,
or the SEC, using a "shelf" registration process. Under this shelf process, we may, from time to time, sell any combination of the securities described in this prospectus in one or more offerings up
to a total amount of $100,000,000.
This
prospectus provides you with a general description of the securities we may offer. Each time we sell securities, we will provide a prospectus supplement that will contain specific
information about the terms of that offering. The prospectus supplement may also add to, update or change information
contained in the prospectus and, accordingly, to the extent inconsistent, information in this prospectus is superseded by the information in the prospectus supplement.
The
prospectus supplement to be attached to the front of this prospectus may describe, as applicable: the terms of the securities offered; the initial public offering price; the price
paid for the securities; estimated gross and net proceeds to us; and the other specific terms related to the offering of the securities.
You
should only rely on the information contained or incorporated by reference in this prospectus and any prospectus supplement or issuer free writing prospectus relating to a particular
offering. No person has been authorized to give any information or make any representations in connection with this offering other than those contained or incorporated by reference in this prospectus,
any accompanying prospectus supplement and any related issuer free writing prospectus in connection with the offering described herein and therein, and, if given or made, such information or
representations must not be relied upon as having been authorized by us. Neither this prospectus nor any prospectus supplement nor any related issuer free writing prospectus shall constitute an offer
to sell or a solicitation of an offer to buy offered securities in any jurisdiction in which it is unlawful for such person to make such an offering or solicitation. This prospectus does not contain
all of the information included in the registration statement. For a more complete understanding of the offering of the securities, you should refer to the registration statement of which this
prospectus is a part, including its exhibits.
You
should read the entire prospectus and any prospectus supplement and any related issuer free writing prospectus, as well as the documents incorporated by reference into this
prospectus or any prospectus supplement or any related issuer free writing prospectus, before making an investment decision. Neither the delivery of this prospectus or any prospectus supplement or any
issuer free writing prospectus nor any sale made hereunder shall under any circumstances imply that the information contained or incorporated by reference herein or in any prospectus supplement or
issuer free writing prospectus is correct as of any date subsequent to the date hereof or of such prospectus supplement or issuer free writing prospectus, as applicable. You should assume that the
information appearing in this prospectus, any prospectus supplement or any document incorporated by reference is accurate only as of the date of the applicable document, regardless of the time of
delivery of this prospectus or any sale of securities. Our business, financial condition, results of operations and prospects may have changed since that date.
The
names "Invuity" and "Intelligent Photonics" are our trademarks.
1
Table of Contents
Prospectus Summary
This summary description about us and our business highlights selected information contained elsewhere in this
prospectus or incorporated in this prospectus by reference. This summary does not contain all of the information you should consider before buying securities in this offering. You should carefully
read this entire prospectus and any applicable prospectus supplement, including each of the documents incorporated herein or therein by reference, before making an investment decision. As used in this
prospectus, "we," "us," "Invuity," "the Company" and "our" refer to Invuity, Inc., a Delaware corporation.
Invuity, Inc.
Overview
We are a commercial-stage medical technology company pioneering the use of advanced photonics to provide surgeons with improved direct
visualization of surgical cavities during minimally invasive and minimal access surgical procedures. We integrate our Intelligent Photonics technology platform into our single-use and reusable
advanced surgical devices to address some of the critical intracavity illumination and visualization challenges facing surgeons today. We utilize our proprietary Intelligent Photonics technology to
develop optical waveguides that direct and shape thermally cool, brilliant light into broad, uniform and volumetric illumination of the surgical target. We believe that improving a surgeon's ability
to see critical anatomical structures can lead to better clinical and aesthetic outcomes, improved patient safety and reduced surgical time and healthcare costs.
Photonics
is the science and technological applications of light. We have applied advanced principles of photonics to develop our Intelligent Photonics technology platform, which enables
the transmission,
management and manipulation of light in surgical procedures. Our initial application of this technology is integrated into our family of proprietary optical waveguides. Our waveguides are
sophisticated devices that rely on the principles of optics to shape and direct light. They are coupled to a modified fiber optic cable and are designed to work with the standard xenon or LED light
sources typically found and utilized in the operating room. Our optical waveguides are incorporated into surgical devices, including our customized line of illuminated surgical retractors, handheld
illuminated aspiration devices and drop-in intracavity illuminators. Our handheld illuminated aspiration devices and drop-in intracavity illuminators are single-use products. Our retractors are
reusable, but utilize a single-use optical waveguide, which we sell separately because a new waveguide must be used for each procedure.
The
fundamental attributes of our optical waveguides include a solid core optical-grade polymer, total internal reflection of light waves, light mixing and extraction by a complex
geometry of refractive microstructures or microlenses. The solid core optical-grade polymer waveguide is coupled to a fiber optic cable in order to facilitate the efficient transfer of light. This
unique coupling results in our waveguides capturing maximum light with minimal heat build-up. Our waveguides use critical angles and the properties of total internal reflection to retain and transmit
maximum light as it travels through the device. In addition, each waveguide utilizes various novel optical methods to mix light during the total internal reflection transmission process to enable more
uniform light extraction across its output surface. The output surface consists of a complex geometry of refractive microstructures or microlenses that extract, direct and shape volumetric
illumination into the surgical cavity while virtually eliminating shadows and glare. This complex geometric structure extracts and directs light at numerous different angles to enable illumination of
the surgical target, even if blood or debris accumulates on the surface of the waveguide. The uniform distribution of light extraction from the microstructures or microlenses throughout the entire
output surface of the waveguide, as well as the proprietary solid core optical-grade polymer and patented design of our waveguides, results in thermally cool illumination.
Advances
in medical technology have resulted in growing adoption of minimally invasive and minimal access surgical procedures. Minimally invasive surgery refers to surgeries performed
through
2
Table of Contents
one
or more small incisions, which offer several benefits over traditional invasive open surgery, such as fewer surgical target complications and infections, overall reduced trauma to the anatomy,
less bleeding, shorter hospitalization time, less postoperative pain, faster recovery time and improved aesthetic outcomes. Some minimally invasive procedures, such as endoscopic, laparoscopic and
arthroscopic procedures, use small tubes, tiny cameras and surgical instruments to access, visualize and perform the surgery. Other procedures also use smaller incisions than conventional open
surgery, but still provide the surgeon with direct visualization of the surgical target and the ability to use traditional surgical instruments. We refer to these procedures as open minimally invasive
and minimal access procedures. We estimate that approximately 40% of all surgical procedures in the United States are open minimally invasive and minimal access, and, based on the benefits of these
procedures over conventional open surgery, we believe this percentage will continue to grow. We have initially targeted our sales and marketing efforts to surgeons in the following specialties:
orthopedics, spine, breast, plastics, and thyroid. However, our current illuminated surgical devices have a broader indication for use and can be marketed to other specialties with limited or no
additional regulatory clearance. We intend to target other surgical specialties including gynecology; colorectal; general surgery; trauma; cardiothoracic; neurosurgery and craniomaxilliofacial
procedures. We currently estimate the annual total addressable market for our devices in these surgical specialties in the United States to be approximately $2.0 billion, based on the estimate
of our average revenue per procedure.
In
the last several years, we have transitioned from a focus on research and development to the commercialization of our device portfolio. As of December 31, 2015, we market nine
families of illuminated surgical devices, consisting of over 40 devices. Our goal is to be the global leader in providing advanced photonics systems to surgeons across a broad array of surgical
specialties, improving surgical precision and efficiency while improving patient safety. We market and sell our devices in the United States primarily through a direct sales force, which has grown to
67 as of March 31, 2016. We have plans to increase sales by further expanding this commercial organization. We believe this expansion will allow us to further penetrate and grow our market by
demonstrating the benefits of our devices to additional surgeons, surgical specialties, and hospitals and expect to expand sales to Europe and other regions, both directly and through distributors.
Corporate Information
We were incorporated in California in 2004 as Spotlight Surgical, Inc. We changed our name to Invuity, Inc. in 2007. We
reincorporated in Delaware in May 2015. Our principal executive offices are located at 444 De Haro Street, San Francisco, California, 94107, and our telephone number is (415) 655-2100. Our
website is
www.invuity.com
. Information contained on, or that can be accessed through, our website is not incorporated by reference into this
prospectus, and you should not consider information on our website to be part of this prospectus.
Implications of Being an Emerging Growth Company
As a company with less than $1.0 billion in revenue during our last fiscal year, we qualify as an "emerging growth company" as defined in
the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. An emerging growth company may take advantage of specified reduced reporting requirements and is relieved of certain other significant
requirements that are otherwise generally applicable to public companies. As an emerging growth company, among other things:
-
•
-
We are exempt from the requirement to obtain an attestation report from our independent registered public accounting firm on our internal
control over financial reporting pursuant to the Sarbanes-Oxley Act of 2002.
-
•
-
We are permitted to provide less extensive disclosure about our executive compensation arrangements in our periodic reports, proxy statements
and registration statements.
3
Table of Contents
-
•
-
We are not required to give our stockholders non-binding advisory votes on executive compensation or golden parachute arrangements.
We
may take advantage of these provisions until the last day of the fiscal year following the fifth anniversary of the completion of our initial public offering, or December 31,
2020. However, if certain events occur prior to the end of such five-year period, including (i) if we become a "large accelerated filer" as defined in Rule 12b-2 under the Securities
Exchange Act of 1934, as amended, or the Exchange Act, (ii) our annual gross revenue equals or exceeds $1.0 billion or (iii) we issue more than $1.0 billion of
non-convertible debt in any three-year period, we will cease to be an emerging growth company prior to the end of such five-year period.
We
may choose to take advantage of some or all of these reduced burdens. We have taken advantage of reduced reporting requirements in this prospectus. Accordingly, the information
contained herein may be different from the information you receive from our competitors that are public companies, or other public companies in which you have made an investment.
In
addition, the JOBS Act provides that an emerging growth company may take advantage of an extended transition period for complying with new or revised accounting standards. We have
irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth
companies.
The Securities We May Offer
We may offer up to $100,000,000 of common stock, preferred stock, depositary shares, warrants and/or units in one or more offerings and in any
combination. This prospectus provides you with a general description of the securities we may offer. A prospectus supplement, which we will provide each time we offer securities, will describe the
specific amounts, prices and terms of the securities we determine to offer.
We may offer shares of our common stock, par value $0.001 per share, either alone or underlying other registered securities convertible or
exercisable into our common stock. The holders of common stock are entitled to one vote per share on all matters submitted to a vote of our stockholders and do not have cumulative voting rights.
Accordingly, holders of a majority of the shares of common stock entitled to vote in any election of directors may elect all of the directors standing for election. Subject to preferences that may be
applicable to any preferred stock outstanding at the time,
the holders of outstanding shares of common stock are entitled to receive ratably any dividends declared by our board of directors out of assets legally available. Upon our liquidation, dissolution,
or winding up, holders of our common stock are entitled to share ratably in all assets remaining after payment of liabilities and the liquidation preference of any then outstanding shares of
convertible preferred stock. Holders of common stock have no preemptive or conversion rights or other subscription rights. There are no redemption or sinking fund provisions applicable to the common
stock.
Pursuant to our amended and restated certificate of incorporation, our board of directors has the authority, without further action by the
stockholders, to issue from time to time up to 10,000,000 shares of preferred stock in one or more series. Our board of directors may designate the rights, preferences, privileges and restrictions of
the preferred stock, including dividend rights, conversion rights, voting rights, redemption rights, liquidation preference, sinking fund terms and the number of shares constituting any series or the
designation of any series.
4
Table of Contents
We
may also issue fractional shares of preferred stock that will be represented by depositary shares and depositary receipts.
Each
series of preferred stock, depositary shares or depositary receipts, if issued, will be more fully described in the particular prospectus supplement that will accompany this
prospectus, including redemption provisions, rights in the event of our liquidation, dissolution or winding up, voting rights and rights to convert into common stock. We do not have any shares of our
preferred stock, depositary shares or depositary receipts presently outstanding.
We may issue warrants for the purchase of common stock or preferred stock. We may issue warrants independently or together with other
securities.
We may issue units comprised of one or more of the other classes of securities issued by us as described in this prospectus in any combination.
Each unit will be issued so that the holder of the unit is also the holder of each security included in the unit.
5
Table of Contents
RISK FACTORS
An investment in our securities involves a high degree of risk. The prospectus supplement applicable to each offering of
our securities will contain a discussion of the risks applicable to an investment in our securities. Prior to making a decision about investing in our securities, you should carefully consider the
specific factors discussed under the heading "Risk Factors" in the applicable prospectus supplement, together with all of the other information contained or incorporated by reference in the prospectus
supplement or appearing or incorporated by reference in this prospectus. You should also consider the risks, uncertainties and assumptions discussed under Item 1A, "Risk Factors," in our Annual
Report on Form 10-K for the fiscal year ended December 31, 2015 and any updates described in our Quarterly Reports on Form 10-Q, all of which are incorporated herein by reference,
and may be amended, supplemented or superseded from time to time by other reports we file with the SEC in the future and any prospectus supplement related to a particular offering. The risks and
uncertainties we have described are not
the only ones we face. Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also affect our operations. The occurrence of any of these known or unknown
risks might cause you to lose all or part of your investment in the offered securities
.
FORWARD-LOOKING STATEMENTS
This prospectus, each prospectus supplement and the information incorporated by reference in this prospectus and each prospectus supplement
contain certain statements that constitute "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities
Exchange Act of 1934. All statements contained in this prospectus other than statements of historical fact, including statements regarding our future results of operations and financial position, our
business strategy and plans, and our objectives for future operations, are forward-looking statements. Words such as "believe," "anticipate," "expect," "intend," "plan," "focus," "assume," "goal,"
"objective," "will," "may" "should," "would," "could," "estimate," "predict," "potential," "continue," "encouraging" or the negative of such terms or other similar expressions identify forward-looking
statements, but are not the exclusive means of identifying such statements.
Those
statements appear in this prospectus, any accompanying prospectus supplement and the documents incorporated herein and therein by reference, particularly in the sections entitled
"Prospectus Summary," "Risk Factors," "Management's Discussion and Analysis of Financial Condition and Results of Operations" and "Business," and are based on our current expectations, assumptions,
estimates and projections about our business and our industry and involve known and unknown risks, uncertainties and other factors that may cause our company's or our industry's results, levels of
activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied in, or contemplated by, the
forward-looking statements. These forward-looking statements are subject to a number of risks, uncertainties, and assumptions, including those described in "Risk Factors" above and elsewhere in this
prospectus. Moreover, we operate in a very competitive and rapidly changing environment, and new risks emerge from time to time. It is not possible for our management to predict all risks, nor can we
assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking
statements we may make. In light of these risks, uncertainties, and assumptions, the forward-looking events and circumstances discussed in this prospectus may not occur and actual results could differ
materially and adversely from those anticipated or implied in our forward-looking statements.
You
should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in our forward-looking statements are reasonable,
we cannot guarantee that the future results, levels of activity, performance or events and circumstances described
in the forward-looking statements will be achieved or occur. Moreover, neither we nor any other person assumes responsibility for the accuracy and completeness of the forward-looking statements. We
6
Table of Contents
undertake
no obligation to update publicly any forward-looking statements for any reason after the date of this prospectus to conform these statements to actual results or to changes in our
expectations, except as required by law.
You
should read this prospectus, any prospectus supplement and the information incorporated by reference in this prospectus and any prospectus supplement with the understanding that our
actual future results, levels of activity, performance and events and circumstances may be materially different from what we expect. We qualify all forward-looking statements by these cautionary
statements.
7
Table of Contents
USE OF PROCEEDS
Unless otherwise indicated in the prospectus supplement, we will use the net proceeds from the sale of securities offered by this prospectus for
general corporate purposes, which may include working capital, expansion of sales and marketing activities and expansion of research and development efforts. We may also use a portion of the net
proceeds to acquire or invest in complementary products, technologies or businesses, although we have no present commitments to complete any such transaction. The amounts and timing of our
expenditures will depend upon numerous factors, including the rate of adoption of our devices, the expenses we incur in selling and marketing our devices, the scope of research and development
efforts, the timing and success of clinical trials we may commence in the future, and the timing of regulatory submissions. Accordingly, our management will have broad discretion over the use of the
net proceeds of the offerings. Pending their
ultimate use, we intend to invest the net proceeds in short-term, interest-bearing, investment-grade securities, certificates of deposit or government securities.
8
Table of Contents
DESCRIPTION OF CAPITAL STOCK
The following information describes our common stock and preferred stock, as well as certain provisions of our amended and restated certificate
of incorporation and bylaws. This summary does not purport to be complete and is qualified in its entirety by the provisions of our amended and restated certificate of incorporation and bylaws, copies
of which have been filed as exhibits to the registration statement of which this prospectus is a part.
General
Our authorized capital stock consists of 100,000,000 shares of common stock with a $0.001 par value per share, and 10,000,000 shares of
undesignated preferred stock with a $0.001 par value per share. Our board of directors may establish the rights and preferences of the preferred stock from time to time. As of March 31, 2016,
there were 13,401,671 shares of common stock issued and outstanding, held of record by 58 stockholders.
The
following is a summary of the material provisions of the common stock and preferred stock provided for in our amended and restated certificate of incorporation and bylaws. For
additional detail about our capital stock, please refer to our certificate of incorporation and bylaws, each as amended.
Common Stock
The holders of common stock are entitled to one vote per share on all matters submitted to a vote of our stockholders and do not have cumulative
voting rights. Accordingly, holders of a majority of the shares of common stock entitled to vote in any election of directors may elect all of the directors standing for election. Subject to
preferences that may be applicable to any preferred stock outstanding at the time, the holders of outstanding shares of common stock are entitled to receive ratably any dividends declared by our board
of directors out of assets legally available. Upon our liquidation, dissolution, or winding up, holders of our common stock are entitled to share ratably in all assets remaining after payment of
liabilities and the liquidation preference of any then outstanding shares of convertible preferred stock. Holders of common stock have no preemptive or conversion rights or other subscription rights.
There are no redemption or sinking fund provisions applicable to the common stock.
Our
common stock is listed on the NASDAQ Global Market under the symbol "IVTY." The transfer agent and registrar for the common stock is Computershare Trust Company, N.A. Its address is
250 Royall Street, Canton, MA 02021.
Preferred Stock
The following description of preferred stock and the description of the terms of any particular series of preferred stock that we choose to
issue hereunder and that will be set forth in the related prospectus supplement are not complete. These descriptions are qualified in their entirety by reference to our amended and restated
certificate of incorporation and the certificate of designation relating to any series of preferred stock. The rights, preferences, privileges and restrictions of the preferred stock of each series
will be fixed by the certificate of designation relating to that series. The prospectus supplement also will contain a description of certain United States federal income tax consequences relating to
the purchase and ownership of the series of preferred stock that is described in the prospectus supplement.
Pursuant
to our amended and restated certificate of incorporation, our board of directors has the authority, without further action by the stockholders, to issue from time to time up to
10,000,000 shares of preferred stock in one or more series. Our board of directors may designate the rights, preferences, privileges and restrictions of the preferred stock, including dividend rights,
conversion rights, voting
9
Table of Contents
rights,
redemption rights, liquidation preference, sinking fund terms and the number of shares constituting any series or the designation of any series. While providing flexibility in connection with
possible acquisitions, future financings and other corporate purposes, the issuance of preferred stock could have the effect of restricting dividends on the common stock, diluting the voting power of
the common stock, impairing the liquidation rights of the common stock, or delaying, deterring or preventing a change in control. Such issuance could have the effect of decreasing the market price of
the common stock. We currently have no plans to issue any shares of preferred stock.
The
prospectus supplement for a series of preferred stock will specify:
-
•
-
the maximum number of shares;
-
•
-
the designation of the shares;
-
•
-
the annual dividend rate, if any, whether the dividend rate is fixed or variable, the date or dates on which dividends will accrue, the
dividend payment dates, and whether dividends will be cumulative;
-
•
-
the price and the terms and conditions for redemption, if any, including redemption at our option or at the option of the holders, including
the time period for redemption, and any accumulated dividends or premiums;
-
•
-
the liquidation preference, if any, and any accumulated dividends upon the liquidation, dissolution or winding up of our affairs;
-
•
-
any sinking fund or similar provision, and, if so, the terms and provisions relating to the purpose and operation of the fund;
-
•
-
the terms and conditions, if any, for conversion or exchange of shares of any other class or classes of our capital stock or any series of any
other class or classes, or of any other series of the same class, or any other securities or assets, including the price or the rate of conversion or exchange and the method, if any, of adjustment;
-
•
-
the voting rights; and
-
•
-
any or all other preferences and relative, participating, optional or other special rights, privileges or qualifications, limitations or
restrictions.
Effect of Certain Provisions of our Amended and Restated Certificate of Incorporation and Bylaws and
the Delaware Anti-Takeover Statute
Our amended and restated certificate of incorporation and amended and restated bylaws contain provisions that could have the effect of delaying,
deferring, or discouraging another party from acquiring control of us. These provisions and certain provisions of Delaware law, which are summarized below, could discourage takeovers, coercive or
otherwise. These provisions are also designed, in part, to encourage persons seeking to acquire control of us to negotiate first with our board of directors. We believe that the benefits of increased
protection of our potential ability to negotiate with an unfriendly or unsolicited acquirer outweigh the disadvantages of discouraging a proposal to acquire us.
Our amended and restated certificate of incorporation and our bylaws provide for the following:
-
•
-
Undesignated Preferred Stock.
As discussed above under
"—Preferred Stock," our board of directors has the ability to designate and issue preferred stock with voting or other rights or preferences that could deter hostile takeovers or delay
changes in our control or management.
10
Table of Contents
-
•
-
Limits on Ability of Stockholders to Act by Written Consent or Call a Special
Meeting.
Our amended and restated certificate of incorporation provides that our stockholders may not act by written consent. This limit on the
ability of stockholders to act by written consent may lengthen the amount of time required to take stockholder actions. As a result, the holders of a majority of our capital stock would not be able to
amend the bylaws or remove directors without holding a meeting of stockholders called in accordance with the bylaws.
In
addition, our amended and restated certificate of incorporation and amended and restated bylaws provide that special meetings of the stockholders may be called only by the chairperson of the board,
the chief executive officer, the president (in the absence of a chief executive officer), or our board of directors. A stockholder may not call a special meeting, which may delay the ability of our
stockholders to force consideration of a proposal or for holders controlling a majority of our capital stock to take any action, including the removal of directors.
-
•
-
Requirements for Advance Notification of Stockholder Nominations and
Proposals.
Our amended and restated bylaws establish advance notice procedures with respect to stockholder proposals and the nomination of
candidates for election as directors, other than nominations made by or at the direction of our board of directors or a committee of the board of directors. These advance notice procedures may have
the effect of precluding the conduct of certain business at a meeting if the proper procedures are not followed and may also discourage or deter a potential acquirer from conducting a solicitation of
proxies to elect its own slate of directors or otherwise attempt to obtain control of our company.
-
•
-
Board Classification.
Our board of directors is divided into three classes.
The directors in each class are elected to serve for a three-year term, one class being elected each year by our stockholders. This system of electing and removing directors may tend to discourage a
third party from making a tender offer or otherwise attempting to obtain control of us, because it generally makes it more difficult for stockholders to replace a majority of the directors.
We are subject to the provisions of Section 203 of the Delaware General Corporation Law regulating corporate takeovers. In general,
Section 203 prohibits a publicly-held Delaware corporation from engaging, under certain circumstances, in a business combination with an interested stockholder for a period of three years
following the date the person became an interested stockholder unless:
-
•
-
prior to the date of the transaction, the board of directors of the corporation approved either the business combination or the transaction
which resulted in the stockholder becoming an interested stockholder;
-
•
-
upon completion of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at
least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding, but not for determining the
outstanding voting stock owned by the interested stockholder, (i) shares owned by persons who are directors and also officers, and (ii) shares owned by employee stock plans in which
employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or
-
•
-
at or subsequent to the date of the transaction, the business combination is approved by the board of directors of the corporation and
authorized at an annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66
2
/
3
% of the outstanding voting stock which is not owned by
the interested stockholder.
11
Table of Contents
Generally,
a business combination includes a merger, asset or stock sale, or other transaction resulting in a financial benefit to the interested stockholder. An interested stockholder
is a person who, together with affiliates and associates, owns or, within three years prior to the determination of interested stockholder status, did own 15% or more of a corporation's outstanding
voting stock. We expect the existence of this provision to have an anti-takeover effect with respect to transactions our board of directors does not approve in advance. We also anticipate that
Section 203 may discourage attempts that might result in a premium over the market price for the shares of common stock held by stockholders.
The
provisions of Delaware law and the provisions of our amended and restated certificate of incorporation and amended and restated bylaws could have the effect of discouraging others
from attempting hostile takeovers and as a consequence, they might also inhibit temporary fluctuations in the market price of our common stock that often result from actual or rumored hostile takeover
attempts. These provisions might also have the effect of preventing changes in our management. It is also possible that these provisions could make it more difficult to accomplish transactions that
stockholders might otherwise deem to be in their best interests.
12
Table of Contents
DESCRIPTION OF THE DEPOSITARY SHARES
General
At our option, we may elect to offer fractional shares of preferred stock, rather than full shares of preferred stock. If we do elect to offer
fractional shares of preferred stock, we will
issue receipts for depositary shares and each of these depositary shares will represent a fraction of a share of a particular series of preferred stock, as specified in the applicable prospectus
supplement. Each owner of a depositary share will be entitled, in proportion to the applicable fractional interest in shares of preferred stock underlying that depositary share, to all rights and
preferences of the preferred stock underlying that depositary share. These rights may include dividend, voting, redemption and liquidation rights.
The
shares of preferred stock underlying the depositary shares will be deposited with a bank or trust company selected by us to act as depositary, under a deposit agreement by and among
us, the depositary and the holders of the depositary receipts. The depositary will be the transfer agent, registrar and dividend disbursing agent for the depositary shares.
The
depositary shares will be evidenced by depositary receipts issued pursuant to the depositary agreement. Holders of depositary receipts agree to be bound by the deposit agreement,
which requires holders to take certain actions such as filing proof of residence and paying certain charges.
The
summary of terms of the depositary shares contained in this prospectus is not complete, and is subject to modification in any prospectus supplement for any issuance of depositary
shares. You should refer to the forms of the deposit agreement, our certificate of incorporation and the certificate of designation that are, or will be, filed with the SEC for the applicable series
of preferred stock.
Dividends
The depositary will distribute cash dividends or other cash distributions, if any, received in respect of the series of preferred stock
underlying the depositary shares to the record holders of depositary receipts in proportion to the number of depositary shares owned by those holders on the relevant record date. The relevant record
date for depositary shares will be the same date as the record date for the preferred stock.
In
the event of a distribution other than in cash, the depositary will distribute property received by it to the record holders of depositary receipts that are entitled to receive the
distribution, unless the depositary determines that it is not feasible to make the distribution. If this occurs, the depositary, with our approval, may adopt another method for the distribution,
including selling the property and distributing the net proceeds to the holders.
Liquidation preference
If a series of preferred stock underlying the depositary shares has a liquidation preference, in the event of our voluntary or involuntary
liquidation, dissolution or winding up, holders of depositary shares will be entitled to receive the fraction of the liquidation preference accorded each share of the applicable series of preferred
stock, as set forth in the applicable prospectus supplement.
Redemption
If a series of preferred stock underlying the depositary shares is subject to redemption, the depositary shares will be redeemed from the
proceeds received by the depositary resulting from the redemption, in whole or in part, of the preferred stock held by the depositary. Whenever we redeem any preferred stock held by the depositary,
the depositary will redeem, as of the same redemption date, the number of depositary shares representing the preferred stock so redeemed. The depositary will mail
13
Table of Contents
the
notice of redemption to the record holders of the depositary receipts promptly upon receiving the notice from us and not fewer than 20 or more than 60 days, unless otherwise provided in the
applicable prospectus supplement, prior to the date fixed for redemption of the preferred stock.
Voting
Upon receipt of notice of any meeting at which the holders of preferred stock are entitled to vote, the depositary will mail the information
contained in the notice of meeting to the record holders of the depositary receipts underlying the preferred stock. Each record holder of those depositary receipts on the record date will be entitled
to instruct the depositary as to the exercise of the voting rights pertaining to the amount of preferred stock underlying that holder's depositary shares. The record date for the depositary will be
the same date as the record date for the preferred stock. The depositary will, to the extent practicable, vote the preferred stock underlying the depositary shares
in accordance with these instructions. We will agree to take all action that may be deemed necessary by the depositary in order to enable the depositary to vote the preferred stock in accordance with
these instructions. The depositary will not vote the preferred stock to the extent that it does not receive specific instructions from the holders of depositary receipts.
Withdrawal of preferred stock
Owners of depositary shares will be entitled to receive upon surrender of depositary receipts at the principal office of the depositary and
payment of any unpaid amount due to the depositary, the number of whole shares of preferred stock underlying their depositary shares.
Partial
shares of preferred stock will not be issued. Holders of preferred stock will not be entitled to deposit the shares under the deposit agreement or to receive depositary receipts
evidencing depositary shares for the preferred stock.
Amendment and termination of the deposit agreement
The form of depositary receipt evidencing the depositary shares and any provision of the deposit agreement may be amended by agreement between
the depositary and us. However, any amendment which materially and adversely alters the rights of the holders of depositary shares, other than fee changes, will not be effective unless the amendment
has been approved by at least a majority of the outstanding depositary shares. The deposit agreement may be terminated by the depositary or us only if:
-
•
-
all outstanding depositary shares have been redeemed; or
-
•
-
there has been a final distribution of the preferred stock in connection with our dissolution and such distribution has been made to all the
holders of depositary shares.
Charges of depositary
We will pay all transfer and other taxes and governmental charges arising solely from the existence of the depositary arrangement. We will also
pay charges of the depositary in connection with:
-
•
-
the initial deposit of the preferred stock;
-
•
-
the initial issuance of the depositary shares;
-
•
-
any redemption of the preferred stock; and
-
•
-
all withdrawals of preferred stock by owners of depositary shares.
14
Table of Contents
Holders
of depositary receipts will pay transfer, income and other taxes and governmental charges and other specified charges as provided in the deposit agreement for their accounts. If
these charges have not been paid, the depositary may:
-
•
-
refuse to transfer depositary shares;
-
•
-
withhold dividends and distributions; and
-
•
-
sell the depositary shares evidenced by the depositary receipt.
Miscellaneous
The depositary will forward to the holders of depositary receipts all reports and communications we deliver to the depositary that we are
required to furnish to the holders of the preferred stock. In addition, the depositary will make available for inspection by holders of depositary receipts at the principal office of the depositary,
and at such other places as it may from time to time deem advisable, any reports and communications we deliver to the depositary as the holder of preferred stock.
Neither
the depositary nor we will be liable if either the depositary or we are prevented or delayed by law or any circumstance beyond the control of either the depositary or us in
performing our respective obligations under the deposit agreement. Our obligations and the depositary's obligations will be limited to the performance in good faith of our or the depositary's
respective duties under the deposit agreement. Neither the depositary nor we will be obligated to prosecute or defend any legal proceeding in respect of any depositary shares or preferred stock unless
satisfactory indemnity is furnished. The depositary and we may rely on:
-
•
-
written advice of counsel or accountants;
-
•
-
information provided by holders of depositary receipts or other persons believed in good faith to be competent to give such information; and
-
•
-
documents believed to be genuine and to have been signed or presented by the proper party or parties.
Resignation and removal of depositary
The depositary may resign at any time by delivering a notice to us. We may remove the depositary at any time. Any such resignation or removal
will take effect upon the appointment of a successor depositary and its acceptance of such appointment. The successor depositary must be appointed within 60 days after delivery of the notice
for resignation or removal. The successor depositary must be a bank and trust company having its principal office in the United States of America and having a combined capital and surplus of at least
$50,000,000.
Federal income tax consequences
Owners of the depositary shares will be treated for U.S. federal income tax purposes as if they were owners of the preferred stock underlying
the depositary shares. As a result, owners will be entitled to take into account for U.S. federal income tax purposes any deductions to which they would be entitled if they were holders of such
preferred stock. No gain or loss will be recognized for U.S. federal income tax purposes upon the withdrawal of preferred stock in exchange for depositary shares. The tax basis of each share of
preferred stock to an exchanging owner of depositary shares will, upon such exchange, be the same as the aggregate tax basis of the depositary shares exchanged. The holding period for preferred stock
in the hands of an exchanging owner of depositary shares will include the period during which such person owned such depositary shares.
15
Table of Contents
DESCRIPTION OF THE WARRANTS
General
We may issue warrants for the purchase of our preferred stock or common stock, or any combination thereof. Warrants may be issued independently
or together with our preferred stock or common stock and may be attached to or separate from any offered securities. Each series of warrants will be issued under a separate warrant agreement to be
entered into between us and a bank or trust company, as warrant agent. The warrant agent will act solely as our agent in connection with the warrants. The warrant agent will not have any obligation or
relationship of agency or trust for or with any holders or beneficial owners of warrants. This summary of certain provisions of the warrants is not complete. For the terms of a particular series of
warrants, you should refer to the prospectus supplement for that series of warrants and the warrant agreement for that particular series.
Equity warrants
The prospectus supplement relating to a particular series of warrants to purchase our common stock or preferred stock will describe the terms of
the warrants, including the following:
-
•
-
the title of the warrants;
-
•
-
the offering price for the warrants, if any;
-
•
-
the aggregate number of warrants;
-
•
-
the designation and terms of the common stock or preferred stock that may be purchased upon exercise of the warrants;
-
•
-
if applicable, the designation and terms of the securities with which the warrants are issued and the number of warrants issued with each
security;
-
•
-
if applicable, the date from and after which the warrants and any securities issued with the warrants will be separately transferable;
-
•
-
the number of shares of common stock or preferred stock that may be purchased upon exercise of a warrant and the exercise price for the
warrants;
-
•
-
the dates on which the right to exercise the warrants shall commence and expire;
-
•
-
if applicable, the minimum or maximum amount of the warrants that may be exercised at any one time;
-
•
-
the currency or currency units in which the offering price, if any, and the exercise price are payable;
-
•
-
if applicable, a discussion of material U.S. federal income tax considerations;
-
•
-
the antidilution provisions of the warrants, if any;
-
•
-
the redemption or call provisions, if any, applicable to the warrants;
-
•
-
any provisions with respect to the holder's right to require us to repurchase the warrants upon a change in control or similar event; and
-
•
-
any additional terms of the warrants, including procedures, and limitations relating to the exchange, exercise and settlement of the warrants.
Holders
of equity warrants will not be entitled:
-
•
-
to vote, consent or receive dividends;
-
•
-
receive notice as stockholders with respect to any meeting of stockholders for the election of our directors or any other matter; or
-
•
-
exercise any rights as stockholders of us.
16
Table of Contents
DESCRIPTION OF THE UNITS
We may issue units comprised of one or more of the other classes of securities described in this prospectus in any combination. Each unit will
be issued so that the holder of the unit is also the holder of each security included in the unit. Thus, the holder of a unit will have the rights and obligations of a holder of each included
security. The units may be issued under unit agreements to be entered into between us and a unit agent, as detailed in the prospectus supplement relating to the units being offered. The prospectus
supplement will describe:
-
•
-
the designation and terms of the units and of the securities comprising the units, including whether and under what circumstances the
securities comprising the units may be held or transferred separately;
-
•
-
a description of the terms of any unit agreement governing the units;
-
•
-
a description of the provisions for the payment, settlement, transfer or exchange of the units;
-
•
-
a discussion of material federal income tax considerations, if applicable; and
-
•
-
whether the units if issued as a separate security will be issued in fully registered or global form.
The
descriptions of the units in this prospectus and in any prospectus supplement are summaries of the material provisions of the applicable agreements. These descriptions do not restate
those agreements in their entirety and may not contain all the information that you may find useful. We urge you to read the applicable agreements because they, and not the summaries, define your
rights as holders of the units. For more information, please review the forms of the relevant agreements, which will be filed with the SEC promptly after the offering of units and will be available as
described under the heading "Where You Can Find More Information."
17
Table of Contents
PLAN OF DISTRIBUTION
We may sell the securities offered through this prospectus (1) to or through underwriters or dealers, (2) directly to purchasers,
including our affiliates, (3) through agents, or (4) through a combination of any of these methods. The securities may be distributed at a fixed price or prices, which may be changed,
market prices prevailing at the time of sale, prices related to the prevailing market prices, or negotiated prices. The prospectus supplement will include the following
information:
-
•
-
the terms of the offering;
-
•
-
the names of any underwriters or agents;
-
•
-
the name or names of any managing underwriter or underwriters;
-
•
-
the purchase price of the securities;
-
•
-
the net proceeds from the sale of the securities;
-
•
-
any delayed delivery arrangements;
-
•
-
any underwriting discounts, commissions and other items constituting underwriters' compensation;
-
•
-
any initial public offering price;
-
•
-
any discounts or concessions allowed or reallowed or paid to dealers; and
-
•
-
any commissions paid to agents.
Sale through underwriters or dealers
If underwriters are used in the sale, the underwriters will acquire the securities for their own account, including through underwriting,
purchase, security lending or repurchase agreements with us. The underwriters may resell the securities from time to time in one or more transactions, including negotiated transactions. Underwriters
may sell the securities in order to facilitate transactions in any of our other securities (described in this prospectus or otherwise), including other public or private transactions and short sales.
Underwriters may offer securities to the public either through underwriting syndicates represented by one or more managing underwriters or directly by one or more firms acting as underwriters. Unless
otherwise indicated in the prospectus supplement, the obligations of the underwriters to purchase the securities will be subject to certain conditions, and the underwriters will be obligated to
purchase all the offered securities if they purchase any of them. The underwriters may change from time to time any initial public offering price and any discounts or concessions allowed or reallowed
or paid to dealers. The prospectus supplement will include the names of the principal underwriters the respective amount of securities underwritten, the nature of the obligation of the underwriters to
take the securities and the nature of any material relationship between an underwriter and us.
If
dealers are used in the sale of securities offered through this prospectus, we will sell the securities to them as principals. They may then resell those securities to the public at
varying prices determined by the dealers at the time of resale. The prospectus supplement will include the names of the dealers and the terms of the transaction.
Direct sales and sales through agents
We may sell the securities offered through this prospectus directly. In this case, no underwriters or agents would be involved. Such securities
may also be sold through agents designated from time to time. The prospectus supplement will name any agent involved in the offer or sale of the offered securities and will describe any commissions
payable to the agent by us. Unless otherwise indicated in
18
Table of Contents
the
prospectus supplement, any agent will agree to use its reasonable best efforts to solicit purchases for the period of its appointment.
We
may sell the securities directly to institutional investors or others who may be deemed to be underwriters within the meaning of the Securities Act with respect to any sale of those
securities. The terms of any such sales will be described in the prospectus supplement.
At-the-Market offerings
To the extent that we make sales through one or more underwriters or agents in at-the-market offerings, we will do so pursuant to the terms of a
sales agency financing agreement or other at-the-market offering arrangement between us, on the one hand, and the underwriters or agents, on the other. If we engage in at-the-market sales pursuant to
any such agreement, we will issue and sell our securities through one or more underwriters or agents, which may act on an agency basis or a principal basis. During the term of any such agreement, we
may sell securities on a daily basis in exchange transactions or otherwise as we agree with the underwriters or agents. Any such agreement
will provide that any securities sold will be sold at prices related to the then prevailing market prices for our securities. Therefore, exact figures regarding proceeds that will be raised or
commissions to be paid cannot be determined at this time. Pursuant to the terms of the agreement, we may agree to sell, and the relevant underwriters or agents may agree to solicit offers to purchase
blocks of our common stock or other securities. The terms of any such agreement will be set forth in more detail in the applicable prospectus or prospectus supplement.
Market making, stabilization and other transactions
Unless the applicable prospectus supplement states otherwise, each series of offered securities will be a new issue and will have no established
trading market. We may elect to list any series of offered securities on an exchange. Any underwriters that we use in the sale of offered securities may make a market in such securities, but may
discontinue such market making at any time without notice. Therefore, we cannot assure you that the securities will have a liquid trading market.
Any
underwriter may also engage in stabilizing transactions, syndicate covering transactions and penalty bids in accordance with Rule 104 under the Securities Exchange Act of
1934, as amended. Stabilizing transactions involve bids to purchase the underlying security in the open market for the purpose of pegging, fixing or maintaining the price of the securities. Syndicate
covering transactions involve purchases of the securities in the open market after the distribution has been completed in order to cover syndicate short positions.
Penalty
bids permit the underwriters to reclaim a selling concession from a syndicate member when the securities originally sold by the syndicate member are purchased in a syndicate
covering transaction to cover syndicate short positions. Stabilizing transactions, syndicate covering transactions and penalty bids may cause the price of the securities to be higher than it would be
in the absence of the transactions. The underwriters may, if they commence these transactions, discontinue them at any time.
Derivative transactions and hedging
We, the underwriters or other agents may engage in derivative transactions involving the securities. These derivatives may consist of short sale
transactions and other hedging activities. The underwriters or agents may acquire a long or short position in the securities, hold or resell securities acquired and purchase options or futures on the
securities and other derivative instruments with returns linked to or related to changes in the price of the securities. In order to facilitate these derivative transactions, we may enter into
security lending or repurchase agreements
with the underwriters or agents. The underwriters or agents may effect the derivative transactions through sales of the securities to the
19
Table of Contents
public,
including short sales, or by lending the securities in order to facilitate short sale transactions by others. The underwriters or agents may also use the securities purchased or borrowed from
us or others (or, in the case of derivatives, securities received from us in settlement of those derivatives) to directly or indirectly settle sales of the securities or close out any related open
borrowings of the securities.
Electronic auctions
We may also make sales through the Internet or through other electronic means. Since we may from time to time elect to offer securities directly
to the public, with or without the involvement of agents, underwriters or dealers, utilizing the Internet or other forms of electronic bidding or ordering systems for the pricing and allocation of
such securities, you should pay particular attention to the description of that system we will provide in a prospectus supplement.
Such
electronic system may allow bidders to directly participate, through electronic access to an auction site, by submitting conditional offers to buy that are subject to acceptance by
us, and which may directly affect the price or other terms and conditions at which such securities are sold. These bidding or ordering systems may present to each bidder, on a so-called "real-time"
basis, relevant information to assist in making a bid, such as the clearing spread at which the offering would be sold, based on the bids submitted, and whether a bidder's individual bids would be
accepted, prorated or rejected. For example, in the case of a debt security, the clearing spread could be indicated as a number of "basis points" above an index treasury note. Of course, many pricing
methods can and may also be used.
Upon
completion of such an electronic auction process, securities will be allocated based on prices bid, terms of bid or other factors. The final offering price at which securities would
be sold and the allocation of securities among bidders would be based in whole or in part on the results of the Internet or other electronic bidding process or auction.
General information
Agents, underwriters, and dealers may be entitled, under agreements entered into with us, to indemnification by us against certain liabilities,
including liabilities under the Securities Act.
20
Table of Contents
LEGAL MATTERS
The validity of the securities offered by this prospectus will be passed upon by Wilson Sonsini Goodrich & Rosati, Professional
Corporation, Palo Alto, California. Additional legal matters may be passed upon for us or any underwriters, dealers or agents by counsel that we will name in the applicable prospectus supplement.
EXPERTS
The financial statements incorporated in this Prospectus by reference to the Annual Report on Form 10-K for the year ended
December 31, 2015 have been so incorporated in reliance on the report of PricewaterhouseCoopers LLP, an independent registered public accounting firm, given on the authority of said firm
as experts in auditing and accounting.
WHERE YOU CAN FIND MORE INFORMATION
We file annual, quarterly and other reports, proxy statements and other information with the SEC. Our SEC filings are available to the public
over the Internet at the SEC's website at http://www.sec.gov. You may also read and copy any document we file at the SEC's Public Reference Room at 100 F Street, NE, Washington,
D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information on the Public Reference Room. Our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, and Current
Reports on Form 8-K, including any amendments to those reports, and other information that we file with or furnish to the SEC pursuant to Section 13(a) or 15(d) of the Exchange Act can
also be accessed free of charge through the Internet. These filings will be available as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC.
We
have filed with the SEC a registration statement under the Securities Act of 1933, as amended, relating to the offering of these securities. The registration statement, including the
attached exhibits, contains additional relevant information about us and the securities. This prospectus does not contain all of the information set forth in the registration statement. You can obtain
a copy of the registration statement, at prescribed rates, from the SEC at the address listed above. The registration statement and the documents referred to below under "Incorporation by Reference"
are also available on our Internet website, www.invuity.com. We have not incorporated by reference into this prospectus the information on our website, and you should not consider it to be a part of
this prospectus.
21
Table of Contents
INFORMATION INCORPORATED BY REFERENCE
The SEC allows us to incorporate by reference into this prospectus certain information we file with it, which means that we can disclose
important information by referring you to those documents. The information incorporated by reference is considered to be a part of this prospectus, and information that we file later with the SEC will
automatically update and supersede information contained in this prospectus and any accompanying prospectus supplement. We incorporate by reference the documents listed below that we have previously
filed with the SEC (excluding any portions of any Form 8-K that are not deemed "filed" pursuant to the General Instructions of Form 8-K):
-
•
-
our Annual Report on Form 10-K for the fiscal year ended December 31, 2015, filed on March 25, 2016, including the
information specifically incorporated by reference into the Annual Report on Form 10-K from our definitive proxy statement on Schedule 14A, filed on April 8, 2016;
-
•
-
our Quarterly Report on Form 10-Q for the fiscal quarter ended March 31, 2016, filed on May 5, 2016;
-
•
-
our Current Reports on Form 8-K filed February 26, 2016, March 25, 2016, May 11, 2016 and May 24, 2016; and
-
•
-
the description of our common stock contained in the Registration Statement on Form 8-A filed with the SEC on June 5, 2015
relating thereto, including any amendment or report filed for the purpose of updating such description.
We
also incorporate by reference into this prospectus additional documents that we may file with the SEC under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act prior to the
completion or termination of the offering, including all such documents we may file with the SEC after the date of the initial registration statement and prior to the effectiveness of the registration
statement, but excluding any information deemed furnished and not filed with the SEC. Any statements contained in a previously filed document incorporated by reference into this prospectus is deemed
to be modified or superseded for purposes of this prospectus to the extent that a statement contained in this prospectus, or in a subsequently filed document also incorporated by reference herein,
modifies or supersedes that statement.
This
prospectus may contain information that updates, modifies or is contrary to information in one or more of the documents incorporated by reference in this prospectus. You should rely
only on the information incorporated by reference or provided in this prospectus. We have not authorized anyone else to provide you with different information. You should not assume that the
information in this prospectus is accurate as of any date other than the date of this prospectus or the date of the documents incorporated by reference in this prospectus.
We
will provide to each person, including any beneficial owner, to whom this prospectus is delivered, upon written or oral request, at no cost to the requester, a copy of any and all of
the information that is incorporated by reference in this prospectus.
Requests
for such documents should be directed to:
Invuity, Inc.
444 De Haro Street
San Francisco, California 94107
Attn: Dan Caul
You
may also access the documents incorporated by reference in this prospectus through our website at www.invuity.com. Except for the specific incorporated documents listed above, no
information available on or through our website shall be deemed to be incorporated in this prospectus or the registration statement of which it forms a part.
22
Table of Contents
6,200,000 Shares
INVUITY, INC.
Common Stock

PROSPECTUS SUPPLEMENT
William Blair
March 15, 2018
INVUITY, INC. (NASDAQ:IVTY)
Historical Stock Chart
From Aug 2024 to Sep 2024
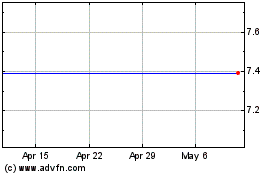
INVUITY, INC. (NASDAQ:IVTY)
Historical Stock Chart
From Sep 2023 to Sep 2024