false000177902000017790202023-08-082023-08-08
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
|
Date of Report (Date of earliest event reported): August 08, 2023 |
DANIMER SCIENTIFIC, INC.
(Exact name of Registrant as Specified in Its Charter)
|
|
|
|
|
Delaware |
001-39280 |
84-1924518 |
(State or Other Jurisdiction
of Incorporation) |
(Commission File Number) |
(IRS Employer
Identification No.) |
|
|
|
|
|
140 Industrial Boulevard |
|
Bainbridge, Georgia |
|
39817 |
(Address of Principal Executive Offices) |
|
(Zip Code) |
|
Registrant’s Telephone Number, Including Area Code: 229 243-7075 |
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class
|
|
Trading
Symbol(s) |
|
Name of each exchange on which registered
|
Class A Common stock, $0.0001 par value per share |
|
DNMR |
|
The New York Stock Exchange |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02 Results of Operations and Financial Condition.
On August 8, 2023, Danimer Scientific, Inc. (“Danimer”) issued a press release announcing its financial results for the second quarter ended June 30, 2023. A copy of this press release is attached hereto as Exhibit 99.1 and is incorporated herein by reference.
The press release contains the following non-GAAP measures: “Adjusted EBITDA”, “Adjusted Gross Profit”, and “Adjusted Gross Margin”. Danimer believes that each of Adjusted EBITDA, Adjusted Gross Profit, and Adjusted Gross Margin are useful to investors in evaluating Danimer’s performance because each measure considers the performance of Danimer’s operations, excluding decisions made with respect to capital investment, financing and other non-recurring charges. Danimer believes these non-GAAP metrics offer additional financial information that, when coupled with the GAAP results and the reconciliation to GAAP results, provides a more complete understanding of its results of operations and the factors and trends affecting its business. The non-GAAP measures are reconciled to comparable GAAP financial measures within the press release. Danimer cautions that non-GAAP measures should be considered in addition to, but not as a substitute for, Danimer’s reported GAAP results. Additionally, Danimer notes that there can be no assurance that the above referenced non-GAAP financial measures are comparable to similarly titled financial measures used by other publicly traded companies.
The information in this Item 2.02 of this Current Report on Form 8-K and Exhibit 99.1 attached hereto shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that section, and shall not be incorporated by reference into any filing of Danimer, whether made before or after the date hereof, regardless of any general incorporation language in such filing, unless expressly incorporated by specific reference to such filing.
Item 7.01 Regulation FD Disclosure
Attached as Exhibit 99.2 to this Current Report on Form 8-K and incorporated herein by reference is the form of presentation to be used by Danimer in presentations for certain of its stockholders and other persons. The information in this Item 7.01 of this Current Report on Form 8-K and Exhibit 99.2 attached hereto shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that section, and shall not be incorporated by reference into any filing of Danimer, whether made before or after the date hereof, regardless of any general incorporation language in such filing, unless expressly incorporated by specific reference to such filing.
Item 9.01 Financial Statements and Exhibits.
d) Exhibits.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
|
|
|
|
|
|
|
Danimer Scientific, Inc |
|
|
|
|
Date: |
August 8, 2023 |
By: |
/s/ Michael A. Hajost |
|
|
|
Michael A. Hajost
Chief Financial Officer |
Danimer Scientific Announces Second Quarter 2023 Results
-- Issues Separate Release Announcing Accepted Part 2 Application for DoE Loan Guarantee --
-- Expands Portfolio with Resins for Protective Films, Shrink Wrap & Produce Bags --
BAINBRIDGE, GA – August 8, 2023 – Danimer Scientific, Inc. (NYSE: DNMR) (“Danimer” or the “Company”), a leading next-generation bioplastics company focused on the development and production of highly engineered biodegradable materials, today announced financial results for its second quarter, ended June 30, 2023.
Danimer announced, in a separate release also issued after the close of market today, the acceptance of its Part II Application by the U.S. Department of Energy (DOE) under the Title XVII Loan Guarantee. The Company has been invited into the confirmatory due diligence and term sheet negotiation process that, upon successful completion could lead to funding from the U.S. Department of the Treasury’s Federal Financing for construction of its greenfield manufacturing facility in Bainbridge, Georgia.
Stephen E. Croskrey, Chairman and Chief Executive Officer of Danimer, commented, “During the second quarter, we made progress in a number of areas. We are very pleased to have moved into the next phase of the financing process with the Department of Energy. We also are excited to have launched new programs that grow and diversify our portfolio of customers and end-use applications. We expect to close this year with momentum in our business and a renewed ability to keep pace with a range of future opportunities for growth. We believe we will enter next year having demonstrated that we are the clear leader in the fast-growing market for responsible alternatives to petroleum-based materials.”
Mr. Croskrey continued, “We are very pleased, after more than three years of careful development work on three new end-use applications, to now have commercialized protective films, shrink wrap and produce bags. We are also excited to play a key role in the rapid commercial launch of compostable coffee-pods. This is a very large category currently dependent on petroleum-based materials which pending EU legislation, if passed, would prohibit from use.”
Mr. Croskrey concluded, “We continue to push forward on other meaningful commercial initiatives. As we await final selections for straw and cutlery programs for customers in the quick service restaurant space, we are making excellent progress in the development of aqueous and extruded coatings for paper cups and for thermoformed cup lids. We believe that our unique ability to offer customers this combination of four key end-use applications will make for a powerful, comprehensive and extremely valuable solution that addresses single-use plastic waste for customers across the food service industry.”
Second Quarter 2023 Financial Highlights:
•Revenues grew to $12.9 million in the second quarter of 2023 both compared to $12.7 million in the year-ago quarter of 2022 and compared to first quarter 2023 revenues of $11.9 million.
Growth was entirely attributable to product revenue, which was $12.2 million in the second quarter as compared to $11.6 million in the same period of last year, driven by stronger sales of PHA-based resins, which increased 10% compared to prior year and 69% sequentially, partially offset by a modest decline in PLA-based resin sales.
Service revenue were $0.7 million in the second quarter as compared to $1.1 million in the second quarter of last year, an expected decline that reflects the successful completion of development work for certain customers, that has now progressed to commercialization.
•Gross profit was $(6.6) million compared to $(2.2) million in the second quarter of 2022. Adjusted gross profit was $(1.6) million compared to $(0.5) million in the second quarter of
2022. The reduction in adjusted gross profit primarily reflects increased fixed production costs associated with greater capacity.
•Net loss for the second quarter was $(39.2) million compared with $(30.4) million in the prior year period.
•Adjusted EBITDA in the second quarter of 2023 improved to $(10.2) million as compared to $(12.9) million in the second quarter of 2022. As in recent previous quarters, this improvement was attributable to a comprehensive program of expense control measures implemented across many areas of the business.
Capital Structure
At June 30, 2023, the Company reported a total debt balance of $377.8 million, which included the Company’s convertible senior notes, its recent senior secured term loan and $45.7 million dollars of low-interest New Markets Tax Credit loans that the Company expects will be forgiven beginning in 2026. The Company noted that, including $14.5 million of restricted cash, effective liquidity at the end of the second quarter was $105.2 million. The Company remains comfortable with its liquidity position and it has the strategic and operational flexibility required to execute its growth strategy.
Outlook
The Company noted that its second quarter and first half results have been consistent with its expectations, and it remains confident in its competitive position and its expectations for growth. At the same time, given a possible shift in the timing of certain significant new program awards and associated first shipment delays, the Company now believes that a bias toward the low end of its previously communicated range for 2023 full-year Adjusted EBITDA of $(23) million to $(31) million is prudent. The Company continues to anticipate full-year capital expenditures in the range of $26 million to $31 million.
Webcast & Conference Call
A webcast and conference call will be held today, August 8, 2023, at 4:30 p.m. Eastern Time to review the Company’s second quarter results, discuss recent events and conduct a question and answer session. The live webcast of the conference call can be accessed on the Investor Relations section of the Company’s website at https://ir.danimerscientific.com.
For those unable to access the webcast, the conference call will be accessible domestically or internationally, by dialing 1-888-999-3182 or 1-848-280-6330, respectively. Upon dialing in, please request to join the Danimer Scientific Second Quarter 2023 Earnings Conference Call. The archived webcast will be available for replay on the Company's website after the call.
About Danimer Scientific
Danimer is a pioneer in creating more sustainable, more natural ways to make plastic products. For more than a decade, its renewable and sustainable biopolymers have helped create plastic products that are biodegradable and compostable and return to nature instead of polluting our lands and waters. Danimer’s technology can be found in a vast array of plastic end products that people use every day. Applications for its biopolymers include additives, aqueous coatings, fibers, filaments, films and injection-molded articles, among others. Danimer holds more than 480 granted patents and pending patent applications in more than 20 countries for a range of manufacturing processes and biopolymer formulations. For more information, visit https://danimerscientific.com.
Forward‐Looking Statements
Please note that in this press release we may use words such as “appears,” “anticipates,” “believes,” “plans,” “expects,” “intends,” “future,” and similar expressions which constitute forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995.
Forward-looking statements include, without limitation, statements regarding our expectations for full year 2023 capital expenditures, Adjusted EBITDA and cash balances. Forward-looking statements are made based on our expectations and beliefs concerning future events impacting the Company and therefore involve a number of risks and uncertainties. We caution that forward-looking statements are not guarantees and that actual results could differ materially from those expressed or implied in the forward-looking statements. Potential risks and uncertainties that could cause the actual results of operations or financial condition of the Company to differ materially from those expressed or implied by forward-looking statements in this release include, but are not limited to, the overall level of consumer demand on our products; general economic conditions and other factors affecting consumer confidence, preferences, and behavior; disruption and volatility in the global currency, capital, and credit markets; the financial strength of the Company's customers; the Company's ability to implement its business strategy, including, but not limited to, its ability to expand its production facilities and plants to meet customer demand for its products and the timing thereof; risks relating to the uncertainty of the projected financial information with respect to the Company; the ability of the Company to execute and integrate acquisitions; changes in governmental regulation, legislation or public opinion relating to our products; the Company’s exposure to product liability or product warranty claims and other loss contingencies; disruptions and other impacts to the Company’s business, as a result of the COVID-19 global pandemic and government actions and restrictive measures implemented in response; stability of the Company’s manufacturing facilities and suppliers, as well as consumer demand for our products, in light of disease epidemics and health-related concerns such as the COVID-19 global pandemic; the impact on our business, operations and financial results from the ongoing conflict in Ukraine; the impact that global climate change trends may have on the Company and its suppliers and customers; the Company's ability to protect patents, trademarks and other intellectual property rights; any breaches of, or interruptions in, our information systems; the ability of our information technology systems or information security systems to operate effectively, including as a result of security breaches, viruses, hackers, malware, natural disasters, vendor business interruptions or other causes; our ability to properly maintain, protect, repair or upgrade our information technology systems or information security systems, or problems with our transitioning to upgraded or replacement systems; the impact of adverse publicity about the Company and/or its brands, including without limitation, through social media or in connection with brand damaging events and/or public perception; fluctuations in the price, availability and quality of raw materials and contracted products as well as foreign currency fluctuations; our ability to utilize potential net operating loss carryforwards; and changes in tax laws and liabilities, tariffs, legal, regulatory, political and economic risks. More information on potential factors that could affect the Company's financial results is included from time to time in the Company's public reports filed with the Securities and Exchange Commission, including the Company's Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, and Current Reports on Form 8-K. All forward-looking statements included in this press release are based upon information available to the Company as of the date of this press release, and speak only as of the date hereof. We assume no obligation to update any forward-looking statements to reflect events or circumstances after the date of this press release.
Contacts:
Investors
James Palczynski
Phone: 415-876-8429
ir@danimer.com
Media
Anthony Priwer
apriwer@daltonagency.com
Phone: 615-515-4891
Danimer Scientific, Inc.
Condensed Consolidated Balance Sheets
|
|
|
|
|
|
|
|
|
|
|
June 30, |
|
|
December 31, |
|
(in thousands, except share and per share data) |
|
2023 |
|
|
2022 |
|
Assets: |
|
|
|
|
|
|
Current assets: |
|
|
|
|
|
|
Cash and cash equivalents |
|
$ |
90,771 |
|
|
$ |
62,792 |
|
Accounts receivable, net |
|
|
12,998 |
|
|
|
17,989 |
|
Other receivables, net |
|
|
1,481 |
|
|
|
1,635 |
|
Inventories, net |
|
|
29,866 |
|
|
|
32,743 |
|
Prepaid expenses and other current assets |
|
|
5,441 |
|
|
|
5,225 |
|
Contract assets, net |
|
|
4,818 |
|
|
|
4,687 |
|
Total current assets |
|
|
145,375 |
|
|
|
125,071 |
|
|
|
|
|
|
|
|
Property, plant and equipment, net |
|
|
454,136 |
|
|
|
453,949 |
|
Intangible assets, net |
|
|
79,332 |
|
|
|
80,941 |
|
Right-of-use assets |
|
|
19,153 |
|
|
|
19,028 |
|
Leverage loans receivable |
|
|
31,446 |
|
|
|
31,446 |
|
Restricted cash |
|
|
14,467 |
|
|
|
1,609 |
|
Other assets |
|
|
344 |
|
|
|
226 |
|
Total assets |
|
$ |
744,253 |
|
|
$ |
712,270 |
|
|
|
|
|
|
|
|
Liabilities and Stockholders' Equity: |
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
Accounts payable |
|
$ |
3,558 |
|
|
$ |
14,977 |
|
Accrued liabilities |
|
|
4,985 |
|
|
|
5,001 |
|
Deferred revenue |
|
|
875 |
|
|
|
- |
|
Current portion of lease liability |
|
|
3,337 |
|
|
|
3,337 |
|
Current portion of long-term debt, net |
|
|
1,847 |
|
|
|
1,972 |
|
Total current liabilities |
|
|
14,602 |
|
|
|
25,287 |
|
|
|
|
|
|
|
|
Private warrants liability |
|
|
245 |
|
|
|
212 |
|
Long-term lease liability, net |
|
|
22,001 |
|
|
|
22,114 |
|
Long-term debt, net |
|
|
375,994 |
|
|
|
286,398 |
|
Deferred income taxes |
|
|
44 |
|
|
|
200 |
|
Other long-term liabilities |
|
|
1,083 |
|
|
|
447 |
|
Total liabilities |
|
$ |
413,969 |
|
|
$ |
334,658 |
|
|
|
|
|
|
|
|
Stockholders' equity: |
|
|
|
|
|
|
Common stock, $0.0001 par value; 200,000,000 shares authorized: 101,938,376 and 101,804,454 shares issued and outstanding at June 30, 2023 and December 31, 2022, respectively |
|
$ |
10 |
|
|
$ |
10 |
|
Additional paid-in capital |
|
|
704,802 |
|
|
|
676,250 |
|
Accumulated deficit |
|
|
(374,528 |
) |
|
|
(298,648 |
) |
Total stockholders’ equity |
|
|
330,284 |
|
|
|
377,612 |
|
Total liabilities and stockholders’ equity |
|
$ |
744,253 |
|
|
$ |
712,270 |
|
Danimer Scientific, Inc.
Condensed Consolidated Statements of Operations
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended June 30, |
|
|
Six Months Ended June 30, |
|
(in thousands, except share and per share data) |
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
Revenue: |
|
|
|
|
|
|
|
|
|
|
|
|
Products |
|
$ |
12,174 |
|
|
$ |
11,575 |
|
|
$ |
23,270 |
|
|
$ |
24,791 |
|
Services |
|
|
691 |
|
|
|
1,128 |
|
|
|
1,521 |
|
|
|
2,655 |
|
Total revenue |
|
|
12,865 |
|
|
|
12,703 |
|
|
|
24,791 |
|
|
|
27,446 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Costs and expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
Cost of revenue |
|
|
19,433 |
|
|
|
14,934 |
|
|
|
37,642 |
|
|
|
30,999 |
|
Selling, general and administrative |
|
|
16,844 |
|
|
|
20,975 |
|
|
|
35,543 |
|
|
|
43,211 |
|
Research and development |
|
|
7,709 |
|
|
|
8,913 |
|
|
|
14,784 |
|
|
|
16,044 |
|
Loss on sale of assets |
|
|
- |
|
|
|
1 |
|
|
|
170 |
|
|
|
1 |
|
Total costs and expenses |
|
|
43,986 |
|
|
|
44,823 |
|
|
|
88,139 |
|
|
|
90,255 |
|
Loss from operations |
|
|
(31,121 |
) |
|
|
(32,120 |
) |
|
|
(63,348 |
) |
|
|
(62,809 |
) |
Nonoperating income (expense): |
|
|
|
|
|
|
|
|
|
|
|
|
Gain (loss) on remeasurement of private warrants |
|
|
1,083 |
|
|
|
2,012 |
|
|
|
(33 |
) |
|
|
7,007 |
|
Interest, net |
|
|
(9,162 |
) |
|
|
(652 |
) |
|
|
(12,548 |
) |
|
|
(1,644 |
) |
Loss on loan extinguishment |
|
|
(102 |
) |
|
|
- |
|
|
|
(102 |
) |
|
|
- |
|
Other, net |
|
|
- |
|
|
|
75 |
|
|
|
- |
|
|
|
84 |
|
Total nonoperating income (expense): |
|
|
(8,181 |
) |
|
|
1,435 |
|
|
|
(12,683 |
) |
|
|
5,447 |
|
Loss before income taxes |
|
|
(39,302 |
) |
|
|
(30,685 |
) |
|
|
(76,031 |
) |
|
|
(57,362 |
) |
Income taxes |
|
|
61 |
|
|
|
240 |
|
|
|
151 |
|
|
|
531 |
|
Net loss |
|
$ |
(39,241 |
) |
|
$ |
(30,445 |
) |
|
$ |
(75,880 |
) |
|
$ |
(56,831 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted net loss per share |
|
$ |
(0.38 |
) |
|
$ |
(0.30 |
) |
|
$ |
(0.74 |
) |
|
$ |
(0.56 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of shares used to compute: |
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted net loss per share |
|
|
101,938,376 |
|
|
|
101,047,650 |
|
|
|
101,917,585 |
|
|
|
100,888,185 |
|
Danimer Scientific, Inc.
Condensed Consolidated Statements of Cash Flows
|
|
|
|
|
|
|
|
|
|
|
Six Months Ended |
|
|
|
June 30, |
|
(in thousands) |
|
2023 |
|
|
2022 |
|
Cash flows from operating activities: |
|
|
|
|
|
|
Net loss |
|
$ |
(75,880 |
) |
|
$ |
(56,831 |
) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
Stock-based compensation |
|
|
27,974 |
|
|
|
27,983 |
|
Depreciation and amortization |
|
|
14,752 |
|
|
|
8,588 |
|
Amortization of debt issuance costs |
|
|
3,485 |
|
|
|
1,152 |
|
Accounts receivable reserves |
|
|
(948 |
) |
|
|
826 |
|
Inventory reserves |
|
|
464 |
|
|
|
597 |
|
Amortization of right-of-use assets and lease liability |
|
|
(237 |
) |
|
|
(173 |
) |
Loss on disposal of assets |
|
|
170 |
|
|
|
1 |
|
Deferred income taxes |
|
|
(155 |
) |
|
|
(531 |
) |
(Gain) loss on remeasurement of private warrants |
|
|
33 |
|
|
|
(7,007 |
) |
Contract asset reserve |
|
|
- |
|
|
|
1,216 |
|
Other |
|
|
1,046 |
|
|
|
45 |
|
Changes in operating assets and liabilities |
|
|
|
|
|
|
Accounts receivable |
|
|
5,939 |
|
|
|
(2,166 |
) |
Other receivables |
|
|
38 |
|
|
|
2,692 |
|
Inventories, net |
|
|
2,383 |
|
|
|
(10,838 |
) |
Prepaid expenses and other current assets |
|
|
1,130 |
|
|
|
1,434 |
|
Contract assets |
|
|
(959 |
) |
|
|
(1,540 |
) |
Other assets |
|
|
(120 |
) |
|
|
(5 |
) |
Accounts payable |
|
|
(2,377 |
) |
|
|
(2,693 |
) |
Accrued liabilities |
|
|
600 |
|
|
|
(2,220 |
) |
Other long-term liabilities |
|
|
636 |
|
|
|
- |
|
Unearned revenue and contract liabilities |
|
|
875 |
|
|
|
2,077 |
|
Net cash used in operating activities |
|
|
(21,151 |
) |
|
|
(37,393 |
) |
Cash flows from investing activities: |
|
|
|
|
|
|
Purchases of property, plant and equipment and intangible assets |
|
|
(23,041 |
) |
|
|
(108,850 |
) |
Acquisition of Novomer, net of cash acquired |
|
|
- |
|
|
|
(14 |
) |
Proceeds from sales of property, plant and equipment |
|
|
- |
|
|
|
55 |
|
Net cash used in investing activities |
|
|
(23,041 |
) |
|
|
(108,809 |
) |
Cash flows from financing activities: |
|
|
|
|
|
|
Proceeds from long-term debt |
|
|
130,000 |
|
|
|
- |
|
Cash paid for debt issuance costs |
|
|
(33,295 |
) |
|
|
(279 |
) |
Principal payments on long-term debt |
|
|
(11,744 |
) |
|
|
(88 |
) |
Proceeds from employee stock purchase plan |
|
|
129 |
|
|
|
296 |
|
Proceeds from exercise of stock options |
|
|
- |
|
|
|
197 |
|
Employee taxes related to stock-based compensation |
|
|
(61 |
) |
|
|
- |
|
Cost related to warrants |
|
|
- |
|
|
|
(55 |
) |
Net cash provided by financing activities |
|
|
85,029 |
|
|
|
71 |
|
Net increase (decrease) in cash and cash equivalents and restricted cash |
|
|
40,837 |
|
|
|
(146,131 |
) |
Cash and cash equivalents and restricted cash-beginning of period |
|
|
64,401 |
|
|
|
286,968 |
|
Cash and cash equivalents and restricted cash-end of period |
|
$ |
105,238 |
|
|
$ |
140,837 |
|
Non-GAAP Financial Measures
This press release includes the non-GAAP financial measures “Adjusted EBITDA”, “Adjusted gross profit” and "Adjusted gross margin". Danimer management views these metrics as a useful way to look at the performance of its operations between periods and to exclude decisions on capital investment and financing that might otherwise impact the review of profitability of the business based on present market conditions.
Adjusted EBITDA is defined as net income or loss plus net interest expense, income taxes, depreciation and amortization, as adjusted to add back certain charges or gains that Danimer may record each period such as remeasurement of private warrants, stock-based compensation expense, as well as non-recurring charges such as (i) asset disposal gains or losses as well as other significant gains or losses such as debt extinguishments and impairment of goodwill; (ii) legal settlements; or (iii) other discrete non-recurring items. Danimer believes these items are not considered an indicator of ongoing performance. Adjusted EBITDA is not a measure of performance defined in accordance with GAAP. The measure is used as a supplement to GAAP results in evaluating certain aspects of Danimer’s business, as described below.
Adjusted gross profit is defined as gross profit plus depreciation, stock-based compensation and nonrecurring items.
Adjusted gross margin is defined as adjusted gross profit divided by total revenue.
Danimer believes that each of Adjusted EBITDA, Adjusted gross profit and Adjusted gross margin is useful to investors in evaluating the Company’s performance because each measure considers the performance of the Company’s operations, excluding decisions made with respect to capital investment, financing and other non-recurring charges as outlined in the preceding paragraph. Danimer believes these non-GAAP metrics offer additional financial information that, when coupled with the GAAP results and the reconciliation to GAAP results, provides a more complete understanding of its results of operations and the factors and trends affecting its business.
Adjusted EBITDA, Adjusted gross profit and Adjusted gross margin should not be considered as an alternative to net income or loss as an indicator of its performance or as alternatives to any other measure prescribed by GAAP as there are limitations to using such non-GAAP measures. Although Danimer believes that Adjusted EBITDA, Adjusted gross profit and Adjusted gross margin may enhance an evaluation of its operating performance based on recent revenue generation and product/overhead cost control because it excludes the impact of prior decisions made about capital investment, financing and other expenses, (i) other companies in Danimer’s industry may define Adjusted EBITDA, Adjusted gross profit and Adjusted gross margin differently than Danimer does and, as a result, they may not be comparable to similarly titled measures used by other companies in its industry, and (ii) Adjusted EBITDA, Adjusted gross profit and Adjusted gross margin exclude certain financial information that some may consider important in evaluating Danimer’s performance.
Danimer compensates for these limitations by providing disclosure of the differences between Adjusted EBITDA, Adjusted gross profit and Adjusted gross margin and GAAP results, including providing a reconciliation to GAAP results, to enable investors to perform their own analysis of Danimer’s operating results. Because GAAP financial measures on a forward-looking basis are not accessible, and reconciling information is not available without unreasonable effort, reconciliations to GAAP financial measures are not provided for forward-looking non-GAAP measures. For the same reasons, the Company is unable to address the probable significance of the unavailable information, which could be material to future results.
Danimer Scientific, Inc.
Reconciliation of Adjusted EBITDA to Net Income (Unaudited)
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended June 30, |
|
|
|
2023 |
|
|
2022 |
|
(in thousands) |
|
|
|
|
|
|
Net income (loss) |
|
$ |
(39,241 |
) |
|
$ |
(30,445 |
) |
Stock-based compensation |
|
|
13,666 |
|
|
|
14,546 |
|
Interest, net |
|
|
9,162 |
|
|
|
652 |
|
Depreciation and amortization |
|
|
7,173 |
|
|
|
4,328 |
|
(Gain) loss on remeasurement of private warrants |
|
|
(1,083 |
) |
|
|
(2,012 |
) |
Loss on extinguishment of debt |
|
|
102 |
|
|
|
- |
|
Income taxes |
|
|
(61 |
) |
|
|
(240 |
) |
Litigation and other legal related |
|
|
37 |
|
|
|
801 |
|
Strategic reorganization and related |
|
|
28 |
|
|
|
- |
|
Inventory reserve |
|
|
- |
|
|
|
(520 |
) |
Loss on sale of assets |
|
|
- |
|
|
|
1 |
|
Public company transition |
|
|
- |
|
|
|
96 |
|
Other, net |
|
|
- |
|
|
|
(75 |
) |
Adjusted EBITDA |
|
$ |
(10,217 |
) |
|
$ |
(12,868 |
) |
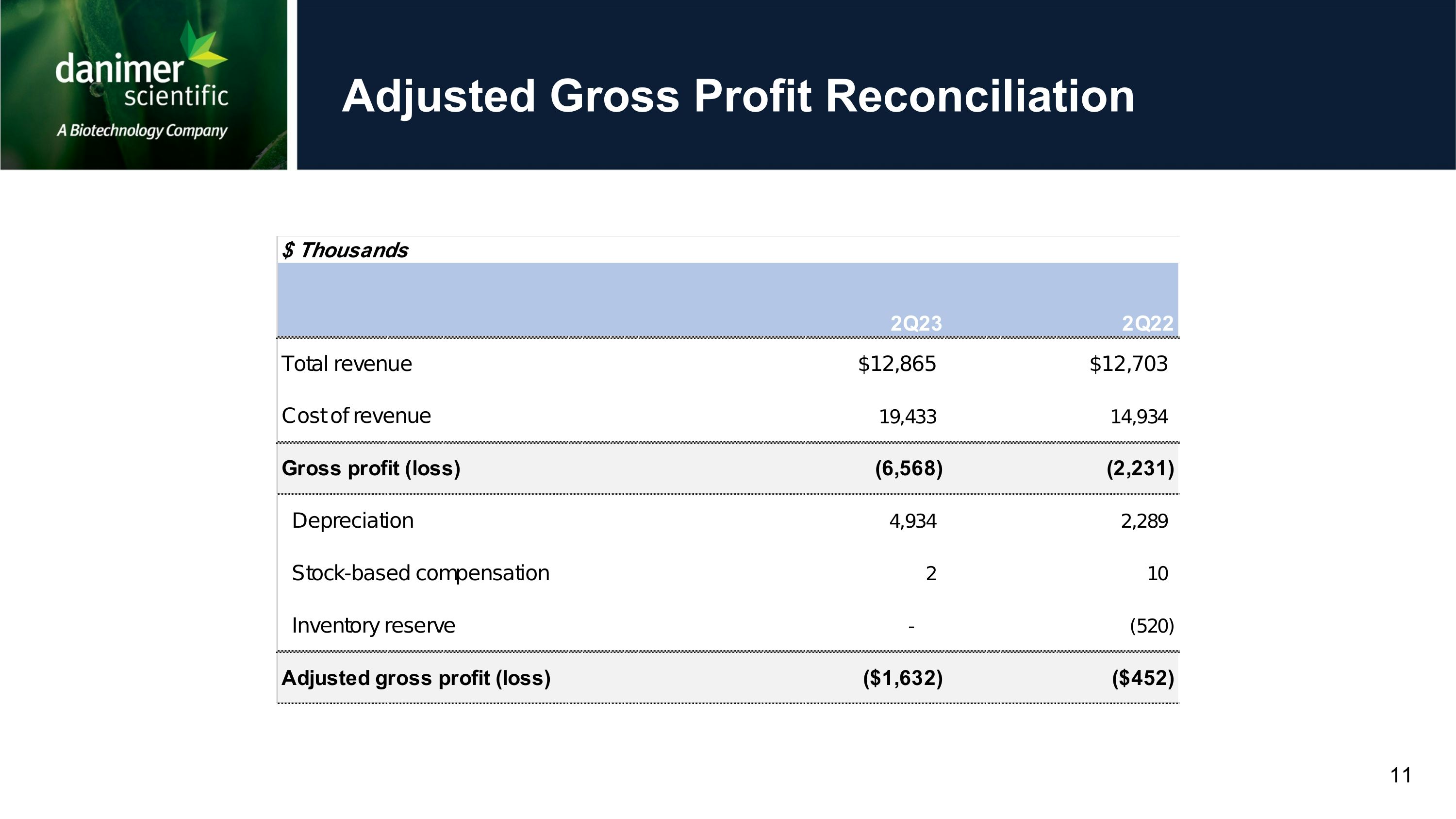
Reconciliation of Adjusted Gross Profit to Gross Profit (Unaudited)
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended June 30, |
|
|
|
2023 |
|
|
2022 |
|
(in thousands) |
|
|
|
|
|
|
Total revenue |
|
$ |
12,865 |
|
|
$ |
12,703 |
|
Cost of revenue |
|
|
19,433 |
|
|
|
14,934 |
|
Gross profit (loss) |
|
|
(6,568 |
) |
|
|
(2,231 |
) |
Depreciation |
|
|
4,934 |
|
|
|
2,289 |
|
Stock-based compensation |
|
|
2 |
|
|
|
10 |
|
Inventory reserve |
|
|
- |
|
|
|
(520 |
) |
Adjusted gross profit (loss) |
|
$ |
(1,632 |
) |
|
$ |
(452 |
) |
|
|
|
|
|
|
|
Adjusted gross margin |
|
|
-12.7 |
% |
|
|
-3.6 |
% |

Second Quarter 2023 Earnings Presentation August 8, 2023

This presentation (“Presentation”) is for informational purposes only. This Presentation shall not constitute an offer to sell, or the solicitation of an offer to buy, any securities, nor shall there be any sale of securities in any states or jurisdictions in which such offer, solicitation or sale would be unlawful. No representations or warranties, express or implied, are given in, or in respect of, this Presentation. To the fullest extent permitted by law in no circumstances will Danimer Scientific, Inc. (the “Company”) or any of its subsidiaries, stockholders, affiliates, representatives, directors, officers, employees, advisers, or agents be responsible or liable for a direct, indirect, or consequential loss or loss of profit arising from the use of this Presentation, its contents, its omissions, reliance on the information contained within it, or on opinions communicated in relation thereto or otherwise arising in connection therewith. Industry and market data used in this Presentation have been obtained from third-party industry publications and sources as well as from research reports prepared for other purposes. The Company has not independently verified the data obtained from these sources and cannot assure you of the data’s accuracy or completeness. This data is subject to change. In addition, this Presentation does not purport to be all-inclusive or to contain all of the information that may be required to make a full analysis of investigations as an investor may deem necessary. FORWARD-LOOKING STATEMENTS Please note that in this Presentation, we may use words such as “appears,” “anticipates,” “believes,” “plans,” “expects,” “intends,” “future,” and similar expressions which constitute forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include, without limitation, statements regarding our expectations for full year 2023 capital expenditures and Adjusted EBITDA. Forward-looking statements are made based on management’s expectations and beliefs concerning future events impacting the Company and therefore involve a number of risks and uncertainties. The Company cautions that forward-looking statements are not guarantees and that actual results could differ materially from those expressed or implied in the forward-looking statements. Potential risks and uncertainties that could cause the actual results of operations or financial condition of the Company to differ materially from those expressed or implied by forward-looking statements in this Presentation include, but are not limited to, the overall level of consumer demand on its products; general economic conditions and other factors affecting consumer confidence, preferences, and behavior; disruption and volatility in the global currency, capital, and credit markets; the financial strength of the Company's customers; the Company's ability to implement its business strategy, including, but not limited to, its ability to expand its production facilities and plants to meet customer demand for its products and the timing thereof; risks relating to the uncertainty of the projected financial information with respect to the Company; the ability of the Company to execute and integrate acquisitions; changes in governmental regulation, legislation or public opinion relating to its products; the Company’s exposure to product liability or product warranty claims and other loss contingencies; disruptions and other impacts to the Company’s business, as a result of the COVID-19 global pandemic and government actions and restrictive measures implemented in response; stability of the Company’s manufacturing facilities and suppliers, as well as consumer demand for its products, in light of disease epidemics and health-related concerns such as the COVID-19 global pandemic; the impact on our business, operations and financial results from the ongoing conflict in Ukraine; the impact that global climate change trends may have on the Company and its suppliers and customers; the Company's ability to protect patents, trademarks and other intellectual property rights; any breaches of, or interruptions in, its information systems; the ability of its information technology systems or information security systems to operate effectively, including as a result of security breaches, viruses, hackers, malware, natural disasters, vendor business interruptions or other causes; its ability to properly maintain, protect, repair or upgrade its information technology systems or information security systems, or problems with its transitioning to upgraded or replacement systems; the impact of adverse publicity about the Company and/or its brands, including without limitation, through social media or in connection with brand damaging events and/or public perception; fluctuations in the price, availability and quality of raw materials and contracted products as well as foreign currency fluctuations; its ability to utilize potential net operating loss carryforwards; and changes in tax laws and liabilities, tariffs, legal, regulatory, political and economic risks. More information on potential factors that could affect the Company's financial results is included from time to time in the Company's public reports filed with the Securities and Exchange Commission, including the Company's Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, and Current Reports on Form 8-K. All forward-looking statements included in this Presentation are based upon information available to the Company as of the date of this Presentation and speak only as of the date hereof. The Company assumes no obligation to update any forward-looking statements to reflect events or circumstances after the date of this presentation. USE OF PROJECTIONS This Presentation contains projected financial information with respect to the Company. Such projected financial information constitutes forward-looking information and is for illustrative purposes only and should not be relied upon as necessarily being indicative of future results. The assumptions and estimates underlying such financial information are inherently uncertain and are subject to a wide variety of significant business, economic, competitive, and other risks and uncertainties. See “Forward-Looking Statements” above. Actual results may differ materially from the results contemplated by the financial forecast information contained in this Presentation, and the inclusion of such information in this Presentation should not be regarded as a representation by any person that the results reflected in such forecasts will be achieved. FINANCIAL INFORMATION; NON-GAAP FINANCIAL MEASURES Some of the financial information and data contained in this Presentation, such as Adjusted EBITDA, and Adjusted Gross Profit has not been prepared in accordance with United States generally accepted accounting principles (“GAAP”). The Company believes these non-GAAP measures of financial results provide useful information to management and investors regarding certain financial and business trends relating to the Company’s financial condition and results of operations. The Company’s management uses these non-GAAP measures for trend analyses and for budgeting and planning purposes. A reconciliation of these non-GAAP financial measures to the closest GAAP measure is included in the Appendix to the Presentation. You should review the Company’s audited financial statements prepared in accordance with GAAP, which are included in its Annual Report on Form 10-K filed with the SEC. Forward-looking non-GAAP financial measures are presented without reconciliations to GAAP measures because the GAAP financial measures are not accessible on a forward-looking basis, and reconciling information is not available without unreasonable effort due to the inherent difficulty in forecasting and quantifying certain amounts that are necessary for such reconciliations. For the same reasons, the Company is unable to address the probable significance of the unavailable information, which could be material to future results. TRADEMARKS This Presentation contained trademarks, service marks, trade names, and copyrights of, the Company, and other companies, which are the property of their respective owners. The information contained herein is as of August 8, 2023, and does not reflect any subsequent events. Forward-Looking Statements and Non-GAAP Financial Results 2
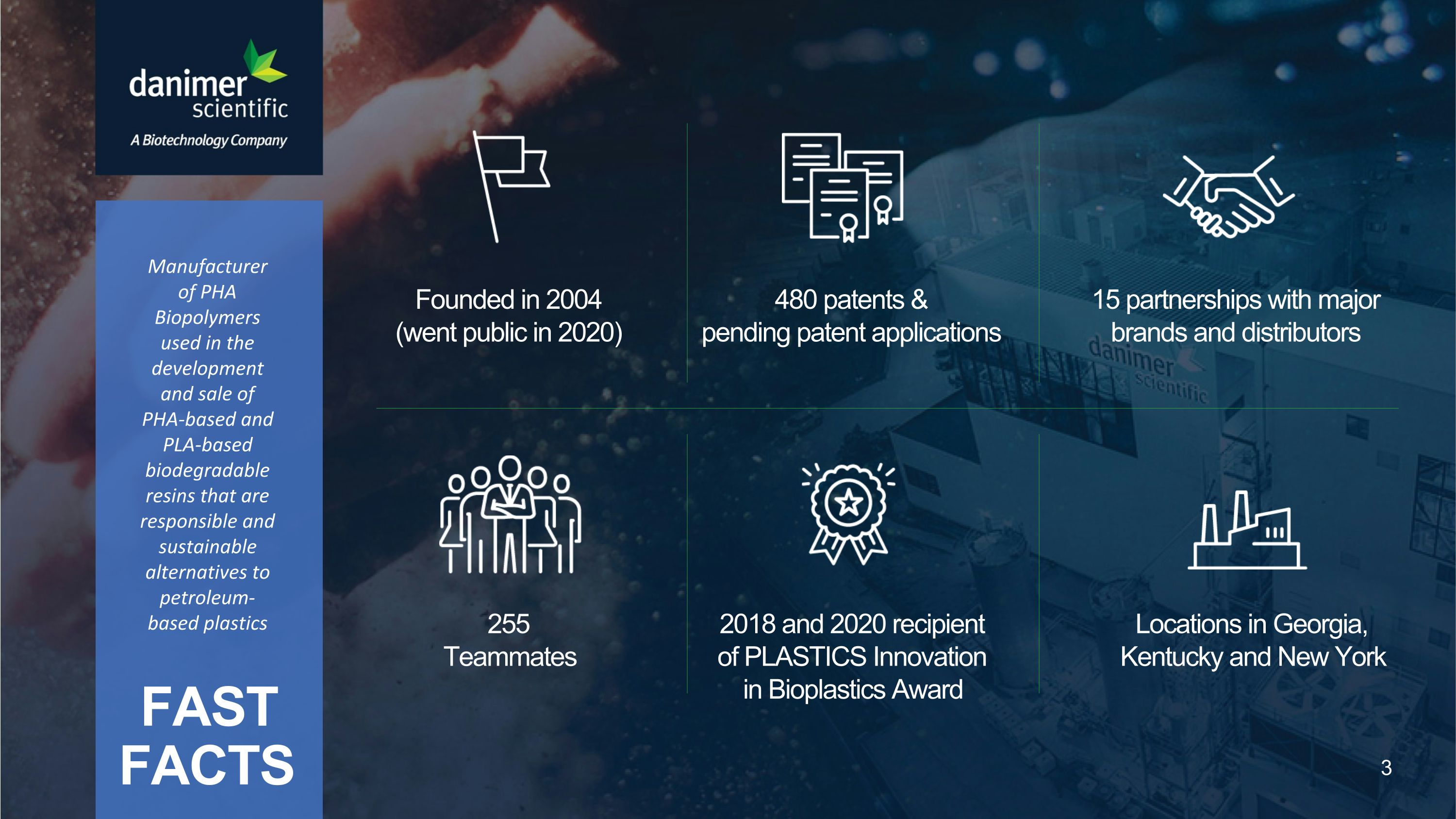
FAST FACTS Founded in 2004�(went public in 2020) 480 patents & �pending patent applications 15 partnerships with major brands and distributors 255 Teammates 2018 and 2020 recipient of PLASTICS Innovation in Bioplastics Award Locations in Georgia, Kentucky and New York Manufacturer of PHA Biopolymers used in the development and sale of PHA-based and PLA-based biodegradable resins that are responsible and sustainable alternatives to petroleum-based plastics 3

Certified biodegradable for home compost, natural soil and marine environments Highly-engineered to run on standard manufacturing equipment Indistinguishable from petroleum-based plastic to consumers at end-use Proprietary PHA-Based Resins with Disruptive Performance 1 Switch to PHA-based resin requires significant customer commitment Creates brand benefits and mitigates risk from future regulatory mandates and/or consumer focus on environment Funded joint development partnerships lead to commercial launches Adoption of Sustainable Materials is Strategic and Persistent 2 Production rate at or above current target Nameplate capacity of ~32.5 M LBs of neat PHA, ~65 M LBs of finished resin At full capacity utilization and current market pricing and mix, expect ~$190 M revenue and very strong contribution margin Kentucky Plant Capacity Serves Near-Term Demand 3 Solid liquidity position provides runway to positive EBITDA Part 2 application for DoE Loan Guarantee to fund greenfield facility accepted; due diligence and term sheet negotiation next Intellectual property contribution drives favorable partnership economics for stand-up of catalytic PHA production Kentucky utilization increase to drive near-term margin improvement Bainbridge greenfield plant to be more efficient; drives scale benefits with ~$400 M of new annual capacity Expect catalytic technology (DCT) to be a high-margin path to new markets, scale benefits and licensing opportunities Deeply capable executive team with cadre of veterans and strong new leaders Talented engineering, research & development, and technical sales teams Driven culture with deep commitment to helping solve global environmental and pollution issues Short, Medium & Long-Term Strategic Finance Initiatives Ongoing Opportunity for Margin Expansion World-Class Teams and a Powerful Culture 4 5 6 Key Investment Highlights 4

Received acceptance of Part 2 application to Department of Energy Loan Guarantee Program; Invited to now take part in due diligence and term sheet negotiation for potential Department of Treasury Federal Financing for construction of Bainbridge, GA greenfield facility Customer launch of Nodax®-based protective films, shrink wrap and produce bags, all new end-use categories with large market opportunities Columbia Packaging Group secures Nodax®-based straw programs with Checkers and Rally’s restaurants and AMC Theatres First of several customers for coffee pods now moving to industrial trials, final validation step to ahead of full rate production Awaiting imminent selection for new customer requiring significant annual volumes of biodegradable straw resin Confident in competitive position for new customer requiring ~15 million pounds of annual volume of Nodax®-based cutlery resin; selection expected in Q3, first shipments expected in late 4Q23 or early 1Q24 Continuous innovation effort reveals, pending validation, possible process engineering pathway to improved quality, throughput, and efficiency as well as lower input costs for neat-PHA production Recent Customer & Business Development Highlights 5
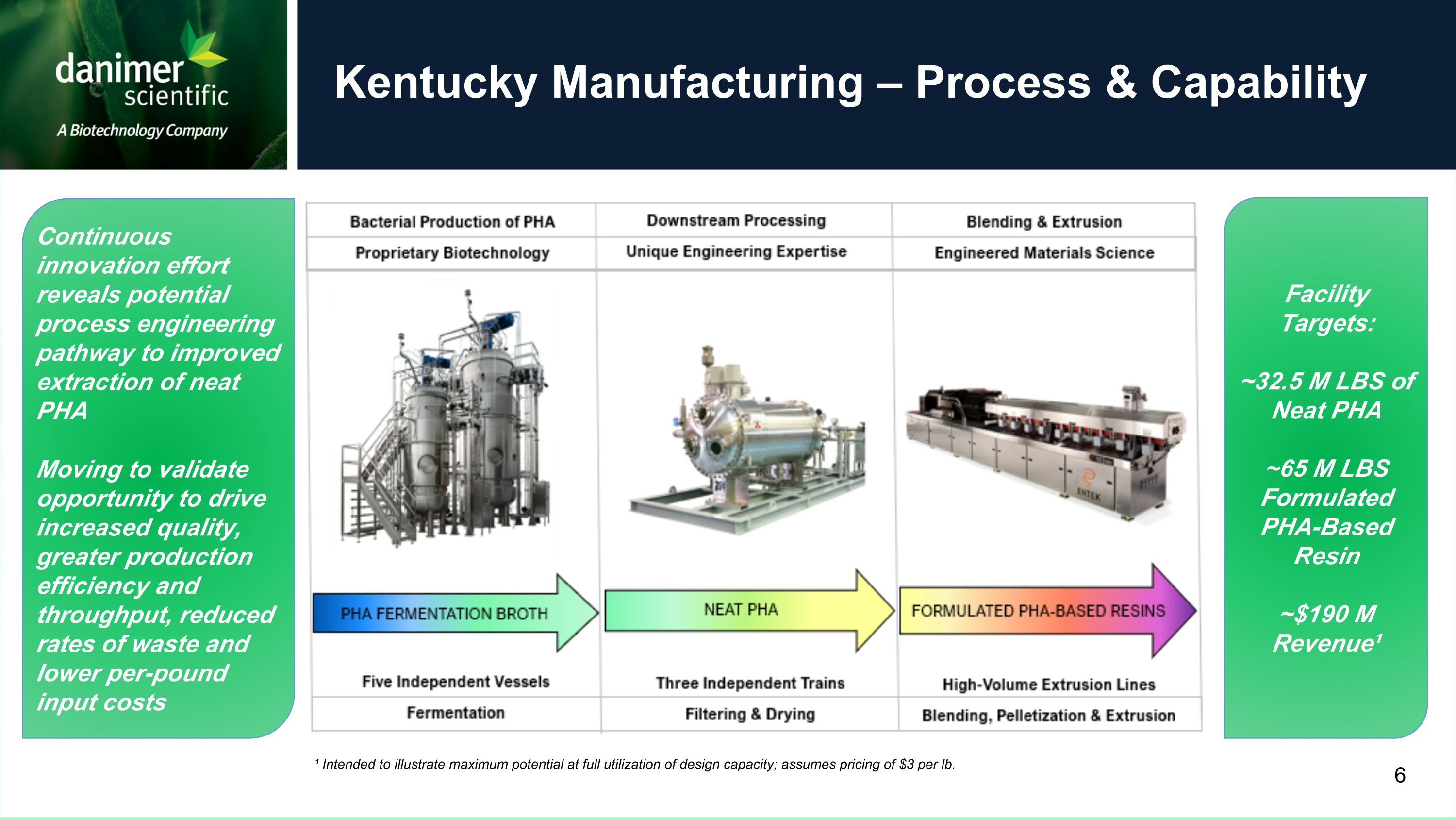
Kentucky Manufacturing – Process & Capability ¹ Intended to illustrate maximum potential at full utilization of design capacity; assumes pricing of $3 per lb. Facility Targets: ~32.5 M LBS of Neat PHA ~65 M LBS Formulated PHA-Based Resin ~$190 M Revenue¹ 6 Continuous innovation effort reveals potential process engineering pathway to improved extraction of neat PHA Moving to validate opportunity to drive increased quality, greater production efficiency and throughput, reduced rates of waste and lower per-pound input costs
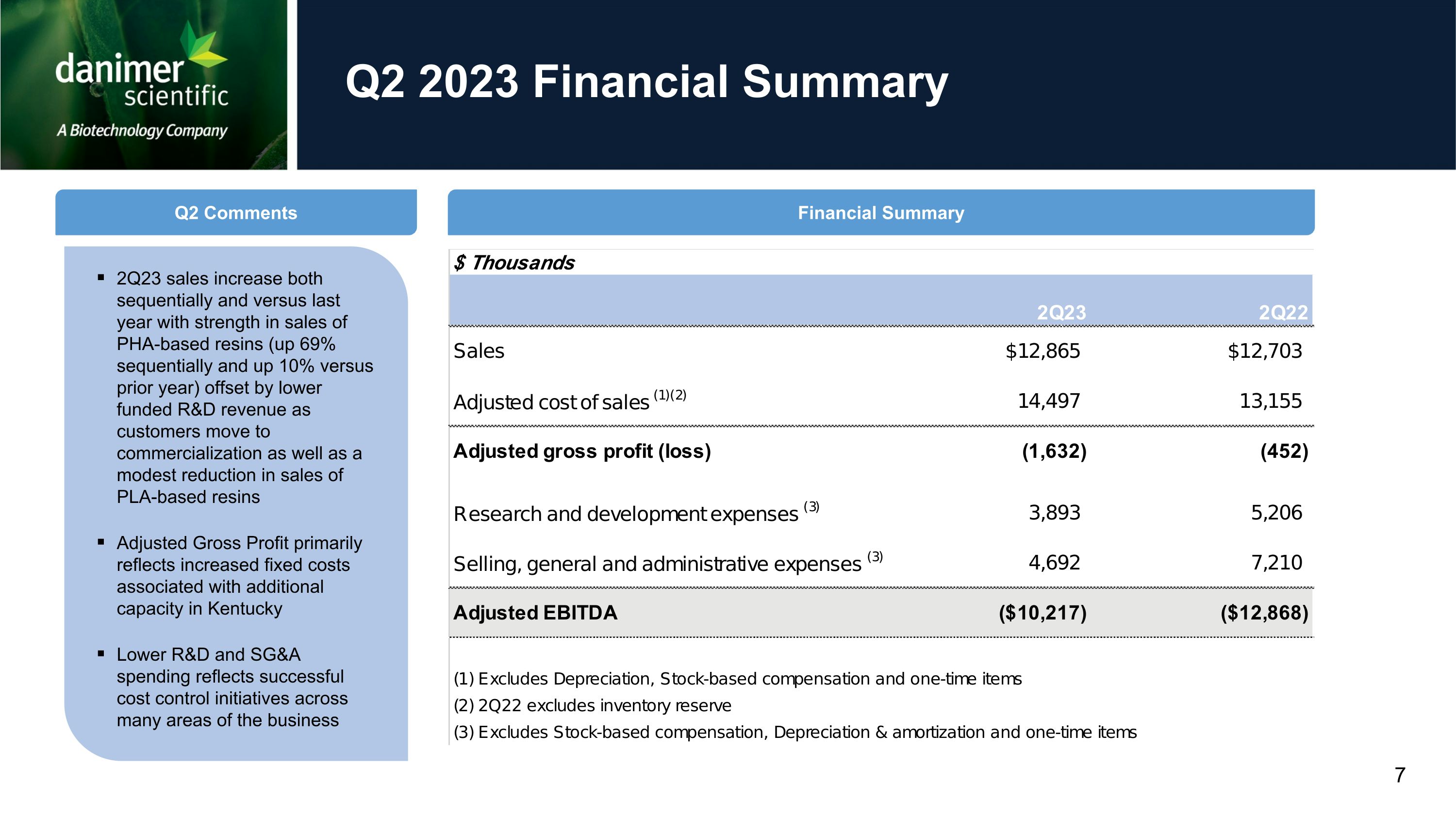
Q2 2023 Financial Summary 2Q23 sales increase both sequentially and versus last year with strength in sales of PHA-based resins (up 69% sequentially and up 10% versus prior year) offset by lower funded R&D revenue as customers move to commercialization as well as a modest reduction in sales of PLA-based resins Adjusted Gross Profit primarily reflects increased fixed costs associated with additional capacity in Kentucky Lower R&D and SG&A spending reflects successful cost control initiatives across many areas of the business Q2 Comments Financial Summary 7

1Q23 and 2Q23 results have been in-line with expectations Believe a bias toward lower end of Adj. EBITDA guidance range is prudent due to later than originally expected timing of large new program awards and increased disruption from Ukraine-Russia conflict on PLA product sales Remain confident in near-term opportunities for capture of significant new programs for several different end-use applications Continue to expect financial results to improve into year end Expectations for FY23 capital expenditures of $31 - $26 million are unchanged 8 Guidance Summary

Appendix

EBITDA Reconciliation 10
Adjusted Gross Profit Reconciliation 11

THANK YOU
v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Danimer Scientific (NYSE:DNMR)
Historical Stock Chart
From Apr 2024 to May 2024
Danimer Scientific (NYSE:DNMR)
Historical Stock Chart
From May 2023 to May 2024