NEW HAVEN, Conn. and LEUVEN,
Belgium, Jan. 10, 2022 /PRNewswire/ -- Biohaven
Therapeutics Ltd., a subsidiary of Biohaven Pharmaceutical Holding
Company Ltd. (NYSE: BHVN), along with CD3 and LICR at KU Leuven,
announced today that they have entered into an exclusive global
license and research agreement to develop and commercialize
first-in-class TRPM3 antagonists to address the growing proportion
of people worldwide living with chronic pain disorders. The TRPM3
antagonist platform was discovered at the Centre for
Drug Design and Discovery (CD3) and the
Laboratory of Ion Channel Research (LICR) at KU Leuven.
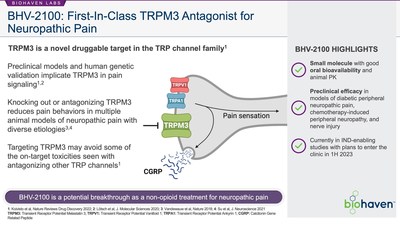
BHV-2100 is the lead TRPM3 antagonist from the platform and an
orally-bioavailable small molecule TRPM3 antagonist. TRPM3 is a
cation channel involved in pain signaling and a novel target for
the treatment of pain discovered at KU Leuven's LICR. Consistent
with the role of TRPM3 in pain, data generated at KU Leuven
demonstrated that BHV-2100 may reduce pain behaviors in animal
models without the detrimental side-effects seen with other pain
management approaches and mechanisms.
Vlad Coric, M.D., Chief Executive
Officer and Chairman of the Board of Biohaven Pharmaceutical
Holding Company Ltd., said, "We are pleased to expand our pipeline
of novel compounds targeting pain disorders, which already includes
our migraine focused CGRP franchise, through this collaboration
with CD3 and KU Leuven. There is an urgent unmet need for safe,
effective treatments for pain, and TRPM3 antagonism is a promising
approach to advance new pain therapies. Based on the extensive
characterization of the mechanism and encouraging preclinical data
generated to date by KU Leuven, we are excited to collaborate with
these academic leaders to bring BHV-2100 into clinical
development."
Thomas Voets, professor at KU
Leuven and group leader at the VIB-KU Leuven Center for Brain &
Disease Research, said "We are excited to collaborate with Biohaven
and look forward to advancing our TRPM3 program towards clinical
applications for the benefit of chronic pain sufferers."
Joris Vriens, professor at KU Leuven
added: "Our fundamental research has shown that TRPM3 is a
promising target for a novel type of analgesics, and this
partnership will give us the necessary resources to advance our
TRPM3-targeting compounds to the clinical stage."
Charlie Conway, PhD, Chief
Scientific Officer of Biohaven Pharmaceuticals, Inc. remarked,
"Professors Voets and Vriens at KU Leuven have been pioneers in
elucidating and validating the pivotal role of TRPM3 in pain
signaling. Their research provides a strong rationale for advancing
TRPM3 modulators to treat pathological pain. I'm excited to carry
BHV-2100 forward as the first candidate in a potentially
transformative class of pain therapies."
Under the agreement, Biohaven Therapeutics receives exclusive
global rights to develop, manufacture and commercialize KU Leuven's
small-molecule TRPM3 antagonists. The portfolio includes the lead
candidate, henceforth known as BHV-2100, which has demonstrated
promising efficacy in preclinical pain models and will be the first
to advance towards Phase 1 studies. Biohaven Therapeutics will
support further basic and translational research at KU Leuven on
the role of TRPM3 in pain and other disorders. KU Leuven will
receive an upfront payment and is eligible to receive additional
development, regulatory, and commercialization milestones. In
addition, KU Leuven will be eligible to receive mid-single digit
royalties on net sales of products resulting from the
collaboration.
Patrick Chaltin, Managing
Director CD3 added: "We are extremely pleased that our long lasting
and successful partnership with LICR has enabled the TRPM3
antagonist program to achieve this important milestone. Biohaven
has a proven track record of successfully developing and launching
novel therapies, and we're excited to work with Biohaven through
this global license and research collaboration agreement."
About Biohaven
Biohaven Pharmaceutical Holding Company
Ltd., parent company of Biohaven Therapeutics, is a
commercial-stage biopharmaceutical company with a portfolio of
innovative, best-in-class therapies to improve the lives of
patients with debilitating neurological and neuropsychiatric
diseases, including rare disorders. Biohaven's
Neuroinnovation™ portfolio includes FDA-approved
NURTEC® ODT (rimegepant) for the acute and preventive
treatment of migraine and a broad pipeline of late-stage product
candidates across three distinct mechanistic platforms: CGRP
receptor antagonism for the acute and preventive treatment of
migraine; glutamate modulation for obsessive-compulsive disorder,
Alzheimer's disease, and spinocerebellar ataxia; and
myeloperoxidase (MPO) inhibition for amyotrophic lateral sclerosis.
More information about Biohaven is available
at www.biohavenpharma.com.
About KU Leuven, CD3 and LICR
As the number one European university for innovation, KU Leuven
actively invests in launching innovative technologies in the
commercial market by creating spin-off companies, securing and
licensing intellectual property, and collaborating with industry.
KU Leuven supports researchers and students in transforming their
innovative ideas and technologies into commercial products and
services that impact people's lives across the globe. KU Leuven
Research & Development (LRD) is KU Leuven's technology transfer
office. Ever since 1972, LRD has been building bridges between
science and industry. For more information, please visit:
lrd.kuleuven.be/en
The Centre for Drug Design and Discovery (CD3) translates
innovative science into promising drug discovery programs that are
well qualified for further development by pharmaceutical or biotech
companies. CD3 brings expert drug discovery capabilities and
financial means to academic research groups and small companies in
order to discover innovative drugs. Supported by KU Leuven Research
& Development and the European Investment Fund, CD3 has entered
into several successful partnerships with pharmaceutical companies
and also integrated drug discovery programs in spin-off companies.
For more information, please visit www.cd3.be.
The Laboratory of Ion Channel Research (LICR) studies ion
channels in all their facets, from their molecular and cellular
mode of action to their role in health and disease. The central
focus is on Transient Receptor Potential (TRP) channels, a
superfamily of cation channels involved in a wide array of
physiological processes, including sensory processing and pain
signaling. Through intense collaboration with clinical researchers
and medicinal chemists, LICR aims at translating fundamental
insights on TRP channel biology towards new therapies. For more
information, please visit gbiomed.kuleuven.be/LICR and
voetslab.sites.vib.be.
Forward-looking Statement
This news release includes
forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995. The use of certain words,
including "believe", "may" and "will" and similar expressions, are
intended to identify forward-looking statements. These
forward-looking statements involve substantial risks and
uncertainties, including statements that are based on the current
expectations and assumptions of Biohaven's management about
BHV-2100 and TRPM3 as a novel target for the treatment of pain.
Biohaven may not actually achieve the plans, intentions or
expectations disclosed in the forward-looking statements and you
should not place undue reliance on Biohaven's forward-looking
statements. Various important factors could cause actual results or
events to differ materially from those that may be expressed or
implied by our forward-looking statements. Additional important
factors to be considered in connection with forward-looking
statements are described in the "Risk Factors" section of the
Company's Annual Report on Form 10-K filed with the Securities and
Exchange Commission on March 1, 2021,
and the Company's Quarterly Report on Form 10-Q filed with the
Securities and Exchange Commission on November 9, 2021. The forward-looking statements
are made as of this date and Biohaven does not undertake any
obligation to update any forward-looking statements, whether as a
result of new information, future events or otherwise, except as
required by law.
NURTEC and NURTEC ODT are registered trademarks of Biohaven
Pharmaceutical Ireland DAC.
Neuroinnovation is a trademark of
Biohaven Pharmaceutical Holding Company Ltd.
Biohaven Contact:
Dr. Vlad
Coric
Chief Executive Officer
Vlad.Coric@biohavenpharma.com
Media Contact:
Mike
Beyer
Sam Brown Inc.
mikebeyer@sambrown.com
312-961-2502
KU Leuven Contact:
Patrick Chaltin
Managing Director CD3
patrick.chaltin@kuleuven.be
+32 16 852 605
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-enters-exclusive-license-and-research-collaboration-agreement-with-ku-leuven-to-advance-first-in-class-trpm3-antagonists-for-the-treatment-of-pain-301456862.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/biohaven-enters-exclusive-license-and-research-collaboration-agreement-with-ku-leuven-to-advance-first-in-class-trpm3-antagonists-for-the-treatment-of-pain-301456862.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.