Processa Pharmaceuticals Announces the Appointment of Dr. Khalid Islam to Its Board of Directors
November 05 2020 - 9:15AM

Processa Pharmaceuticals, Inc. (NASDAQ: PCSA), (“Processa” or the
“Company”), a clinical-stage biopharmaceutical company developing
products to improve the survival and/or quality of life for
patients who have a high unmet medical need, is pleased to announce
the appointment of Dr. Khalid Islam to its board of directors.
“I’d like to personally welcome Dr. Islam to the
board,” commented Dr. David Young, Chairman and CEO of Processa
Pharmaceuticals. “Dr. Islam’s extensive experience and insight
within the pharmaceutical industry and his diverse perspectives
will be a great addition to the board and the Company.”
Dr. Islam was the chairman and CEO of Gentium
S.p.A. (a Nasdaq-listed company; 2009-2014) where he led its
transition to a profitable company that subsequently sold for US$1
billion. He is an advisor to the venture group Kurma Biofund
(Paris). He is currently the Chairman of the Board of Directors of
Fennec Pharmaceuticals Inc. (Nasdaq: FENC), Gain Therapeutics Inc.,
Minoryx Therapeutics SL and currently serves on the board of
Immunomedics Inc. (NASDAQ: IMMU) which recently maximized
shareholder value through a $21 billion transaction with Gilead
Sciences.
Dr. Islam has served in a variety of roles of
increasing responsibility from entrepreneurial start-ups to Fortune
100 companies and has served in both advisory and board capacities
for several public and private health-care related companies. Dr.
Islam holds a Ph.D from Imperial College, University of London. He
also holds several patents and has published over 80 articles in
leading journals.
Dr. Islam said, “I have followed Dr. Young and
his team’s successes in effectively and efficiently developing
drugs through clinical development for conditions which need
therapeutic alternatives. I am confident that Processa’s approach
will accelerate the development of all the drugs in the Processa
pipeline given their past experience and successful interactions
with the FDA. I am excited to join the Processa team.”
About Processa Pharmaceuticals, Inc.
The mission of Processa is to develop products
for which existing clinical evidence of efficacy has already been
established for unmet or under-served medical conditions for which
patients need treatment options that improve survival and/or
quality of life. The Company used these criteria to develop our
pipeline programs in order to achieve high value milestones
effectively and efficiently using the Processa Regulatory Science
approach. The members of the Processa development team have been
involved with more than 30 drug approvals by the FDA (including
drug products targeted to orphan disease conditions) and more than
100 FDA meetings throughout their careers.
For more information, visit the company’s website
at www.ProcessaPharma.com.
Forward-Looking Statements
This release contains forward-looking
statements. The statements in this press release that are not
purely historical are forward-looking statements which involve
risks and uncertainties. Actual future performance outcomes and
results may differ materially from those expressed in
forward-looking statements. Please refer to the registration
statement relating to the securities being sold in this offering,
which identifies important risk factors which could cause actual
results to differ from those contained in the forward-looking
statements.
For More Information: Michael Floyd mfloyd@processapharma.com
301-651-4256
Corporate Communications:InvestorBrandNetwork
(IBN) Los Angeles, California www.InvestorBrandNetwork.com
310.299.1717 Office Editor@InvestorBrandNetwork.com
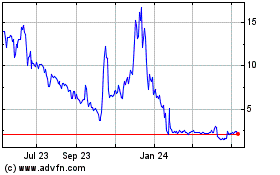
Processa Pharmaceuticals (NASDAQ:PCSA)
Historical Stock Chart
From Apr 2024 to May 2024
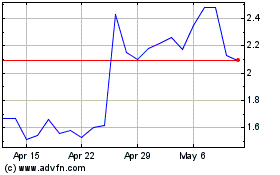
Processa Pharmaceuticals (NASDAQ:PCSA)
Historical Stock Chart
From May 2023 to May 2024