NRx Pharmaceuticals Gets Fast-Track Designation for NRX-101 UTI Treatment
January 16 2024 - 7:25AM
Dow Jones News
By Chris Wack
NRx Pharmaceuticals has received qualified infectious disease
product and fast-track designation from the U.S. Food and Drug
Administration for NRX-101 in the treatment of complicated urinary
tract infections and pyelonephritis.
Receipt of QIDP designation confers priority review, and five
additional years of data-exclusivity to NRX-101's intellectual
property position.
The FDA also granted NRX-101 fast-track designation for cUTI,
which additionally allows for rolling submission of the company's
New Drug Application.
NRx has composition of matter patent protection on NRX-101
through at least 2033 in all major global markets. NRx is seeking
partners with active involvement in urology, infectious disease
and/or women's health for commercialization of NRX-101.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
January 16, 2024 07:10 ET (12:10 GMT)
Copyright (c) 2024 Dow Jones & Company, Inc.
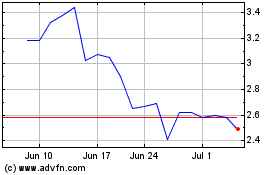
NRX Pharmaceuticals (NASDAQ:NRXP)
Historical Stock Chart
From Jun 2024 to Jul 2024
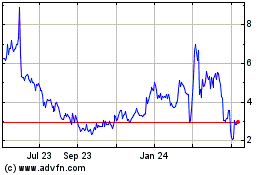
NRX Pharmaceuticals (NASDAQ:NRXP)
Historical Stock Chart
From Jul 2023 to Jul 2024