Biocept to Host Key Opinion Leader Webinar on April 8 to Discuss its CSF Assay for the Diagnosis of Cancer Involving the Cent...
April 01 2021 - 8:05AM
Business Wire
Presenters include Dr. Amir Azadi of Barrow
Neurological Institute, Dr. Nicholas Blondin of Yale University and
Dr. Priya U. Kumthekar of Northwestern University
Biocept, Inc. (Nasdaq: BIOC), a leading provider of molecular
diagnostic assays, products and services, will host a webinar
featuring leading neuro-oncologists to discuss the use of the
Company’s proprietary cerebrospinal fluid (CSF) assay for
diagnosing and managing tumors that have metastasized to the
central nervous system (CNS), including the brain or spinal column.
Biocept’s CSF assay provides enhanced sensitivity compared with CSF
cytology, the current standard of care, and has the added advantage
of identifying actionable molecular targets for use in treatment
decisions while providing quantitative information needed for
assessing treatment response and monitoring disease
progression.
“Our goal is to establish our CSF assay as the new standard of
care for the diagnosis of patients with suspected cancer metastasis
to the CNS under National Comprehensive Cancer Network guidelines,”
said Michael Dugan, MD, Biocept’s Senior Vice President, Chief
Medical Officer and Medical Director. “We have already gained
substantial interest among neuro-oncologists and other cancer
specialists from nearly two dozen leading academic institutions
across the country. Most of these physicians have already ordered
our CSF assay with many becoming repeat users.
“The CSF assay addresses a high unmet clinical need as current
diagnostic tools for patients with brain metastases are inadequate
or imprecise for assessing therapy response; however, many
therapies are now available that offer substantial promise for
improved survival and resolution of symptoms,” Dr. Dugan added.
“Between 10% and 30% of adult patients with cancer, depending on
the type, will develop brain metastases. We estimate this market
opportunity at more than $1 billion annually.”
Webinar Details
Date:
Thursday, April 8, 2021
Time:
12:00 p.m. Eastern time (9:00
a.m. Pacific time)
Registration:
Participants can pre-register or
register at webinar start time here.
The webinar also will be available live and archived at
https://ir.biocept.com/.
Amir Azadi, MD is a medical oncologist specializing in
neuro-oncology and an Assistant Professor in the Department of
Neurology at Barrow Neurological Institute. His expertise includes
the diagnosis and treatment of brain tumors. He is board certified
in internal medicine and medical oncology by the American Board of
Internal Medicine, and is a member of the American College of
Physicians, the American Society of Clinical Oncology, the Society
for Immunotherapy of Cancer and the Society of Neuro-Oncology. Dr.
Azadi received his medical degree from Shahid Beheshti University
of Medical Sciences in Tehran, Iran. He completed his residency in
internal medicine at Banner – University Medical Center in Phoenix,
and fellowships in hematology and oncology at the University of
Louisville James Graham Brown Cancer Center and in neuro-oncology
at Barrow Neurological Institute.
Nicholas Blondin, MD is Assistant Professor of Clinical
Neurology at Yale School of Medicine. His clinical expertise is in
treating benign and malignant brain tumors, brain and spine
metastasis, and neurological symptoms of cancer such as seizures,
cognitive impairment, headaches, gait disturbance and weakness. Dr.
Blondin is an active investigator for clinical trials for brain
tumor patients through Yale Cancer Center. He received his Medical
Degree from the University of Connecticut School of Medicine and
completed his neurology residency at Yale New Haven Hospital, where
he was Chief Resident in Neurology, followed by a fellowship in
neuro-oncology at Yale New Haven Hospital.
Priya U. Kumthekar, MD is a United Counsel for Neurologic
Subspecialties (UCNS)-certified neuro-oncologist from Northwestern
University and is serving as the principal investigator for
Biocept’s Four C clinical study. She is dedicated to patient care
and moving the study of brain tumors forward primarily through her
leadership on clinical trials. Dr. Kumthekar serves in leadership
roles with the National Clinical Trials Network, particularly with
the Alliance for Clinical Trials, and was named as the Alliance’s
national Executive Officer of Neuro-Oncology in 2016. In this role,
she oversees the conception and development of clinical trials from
early phase through registration studies. Dr. Kumthekar is board
certified in neurology and is a member of the American Board of
Psychiatry and Neurology. She received her medical degree from
Northeastern Ohio University. She previously was Chief Resident at
Northwestern University, McGaw Medical Center.
About Biocept
Biocept, Inc. develops and commercializes molecular diagnostic
assays that provide physicians with clinically actionable
information for treating and monitoring patients diagnosed with a
variety of cancers. In addition to its broad portfolio of
blood-based liquid biopsy assays, Biocept is developing and
commercializing its cerebrospinal fluid (CSF) assay that detects
cancer that has metastasized to the central nervous system. The
Company’s CSF assay identifies molecular targets for clinical
decision making and provides quantitative information for
monitoring treatment response. Biocept’s patented Target Selector™
technology platform captures and analyzes tumor-associated
molecular markers in both circulating tumor cells (CTCs) and
circulating tumor DNA (ctDNA) with higher sensitivity and
specificity than most commercial assays. Additionally, Biocept is
leveraging its molecular diagnostic capabilities to offer
nationwide COVID-19 RT-PCR testing to support public health efforts
during this unprecedented pandemic. For additional information,
visit www.biocept.com. Follow Biocept on Facebook, LinkedIn and
Twitter.
Forward-Looking Statements Disclaimer Statement
This news release contains forward-looking statements that are
based upon current expectations or beliefs, as well as a number of
assumptions about future events. Although we believe that the
expectations reflected in the forward-looking statements and the
assumptions upon which they are based are reasonable, we can give
no assurance that such expectations and assumptions will prove to
be correct. Forward-looking statements are generally identifiable
by the use of words like "may," "will," "should," "could,"
"expect," "anticipate," "estimate," "believe," "intend" or
"project," or the negative of these words or other variations on
these words or comparable terminology. To the extent that
statements in this news release are not strictly historical,
including, without limitation, statements as to our ability to
establish our CSF assay as the new standard of care for the
diagnosis of patients with suspected cancer metastasis to the CNS
under National Comprehensive Cancer Network guidelines and our
ability to provide physicians with clinically actionable
information to improve the outcomes of cancer patients, such
statements are forward-looking, and are made pursuant to the safe
harbor provisions of the Private Securities Litigation Reform Act
of 1995. The reader is cautioned not to put undue reliance on these
forward-looking statements, as these statements are subject to
numerous risk factors as set forth in our Securities and Exchange
Commission (SEC) filings. The effects of such risks and
uncertainties could cause actual results to differ materially from
the forward-looking statements contained in this news release. We
do not plan to update any such forward-looking statements and
expressly disclaim any duty to update the information contained in
this press release except as required by law. Readers are advised
to review our filings with the SEC at http://www.sec.gov/.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210401005271/en/
Investor Contact LHA
Investor Relations Jody Cain Jcain@lhai.com, (310) 691-7100
Media Contact Sampson PR
Group Andrea Sampson asampson@sampsonprgroup.com, (562)
304-0301
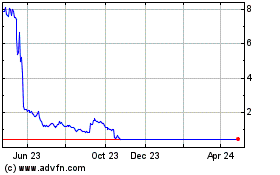
Biocept (NASDAQ:BIOC)
Historical Stock Chart
From Aug 2024 to Sep 2024
Biocept (NASDAQ:BIOC)
Historical Stock Chart
From Sep 2023 to Sep 2024