Kyowa Kirin Co., Ltd. (Kyowa Kirin, TSE: 4151) a Japan-based global
specialty pharmaceutical company (J-GSP) discovering and developing
novel medicines utilizing the latest biotechnology, and Orchard
Therapeutics plc (Orchard Therapeutics, Nasdaq: ORTX), a global
gene therapy leader, today announced Kyowa Kirin has successfully
completed the acquisition of Orchard Therapeutics. The acquisition
of all outstanding shares of Orchard Therapeutics by way of a
Scheme of Arrangement procedure under the UK Companies Act 2006 was
completed on January 24, 2024, and Orchard Therapeutics has become
a wholly-owned subsidiary of Kyowa Kirin. The integrated business
will now increase its focus on meeting the needs of people living
with devastating genetic and other severe diseases where the burden
is immense and current treatment options are limited or do not
exist.
Orchard Therapeutics’ portfolio comprises Libmeldy®
(atidarsagene autotemcel), which is intended for the treatment of
eligible patients with early-onset metachromatic leukodystrophy
(MLD), a rare and life-threatening inherited disease of the body’s
metabolic system. In the most severe form of MLD, babies develop
normally but in late infancy start to rapidly lose the ability to
walk, talk and interact with the world around them. Libmeldy is
approved by the European Commission (EC) and UK Medicines and
Healthcare products Regulatory Agency (MHRA). Libmeldy is known as
OTL-200 in the U.S., where it is currently an investigational drug
under Priority Review by the Food and Drug Administration (FDA)
with a Prescription Drug User Fee Act (PDUFA) goal date of March
18, 2024.
Using the same hematopoietic stem cell (HSC) gene therapy
technology platform, Orchard Therapeutics is also progressing two
clinical-stage programs, OTL-203 for the treatment of
mucopolysaccharidosis type I Hurler’s syndrome (MPS-IH) and OTL-201
in development for mucopolysaccharidosis type IIIA (MPS-IIIA), also
known as Sanfilippo syndrome.
“We are truly excited about the acquisition of Orchard
Therapeutics, a leading provider of HSC gene therapy. This platform
offers significant potential to deliver more innovative treatments
and breakthrough therapies and aligns with our purpose to deliver
life-changing value for people living with rare and complex
diseases,” said Masashi Miyamoto, Ph.D., Representative Director,
President and CEO of Kyowa Kirin. “Going forward, our companies
will build on the extensive experience of Orchard’s gene therapy
platform and apply it to under-served indications and diseases
where we believe it to be scientifically and clinically
differentiated.”
“We look forward to this next chapter in Orchard Therapeutics’
evolution and are eager to partner with our new colleagues at Kyowa
Kirin to unlock the full potential of our HSC gene therapy
approach,” said Bobby Gaspar, co-founder and chief executive
officer of Orchard Therapeutics. “The next 12 months have the
potential to provide several breakout opportunities that we believe
would cement our leadership position in the field, including the
potential approval and launch of OTL-200 in the U.S., the
acceleration of Libmeldy growth in Europe, the progression of our
global registrational trial for OTL-203 in MPS-IH, as well as the
advancement of our next-in-line neurometabolic program in MPS-IIIA
and earlier-stage research programs.”
In the new organizational structure, Bobby Gaspar, M.D., Ph.D.,
will report to Kyowa Kirin President & CEO Masashi Miyamoto,
Ph.D., and become a member of the Kyowa Kirin's senior R&D
leadership team, helping the organization evaluate next-generation
therapeutic candidates. Members of the Orchard Therapeutics team
will continue to operate from its existing facilities in London and
Boston. Kyowa Kirin anticipates significant synergies with Orchard
Therapeutics and plans to announce its 2024 Financial Guidance in
conjunction with its 2023 earnings on 7 February.
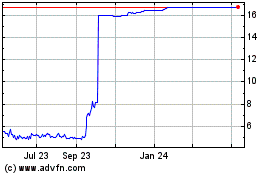
Overview of the transactionUnder the terms of
the agreement, Kyowa Kirin completed the scheme of arrangement to
acquire all outstanding shares of Orchard Therapeutics at a price
of $16.00 per American Depositary Share (ADS) in cash which
represents a premium of 144% to Orchard Therapeutics’
volume-weighted average price per ADS over the 30 days ended
October 4, 2023, the day before the transaction was announced.
In connection with the transaction, an additional contingent
value right (CVR) of $1.00 per ADS is payable to Orchard
shareholders for a total of $17.00 per ADS. The additional CVR
payments are contingent and payable only on U.S. approval of
OTL-200 in 2024 per the terms of the CVR agreement.
Overview of Orchard Therapeutics
|
(1 |
) |
Name |
Orchard Therapeutics plc |
|
(2 |
) |
Location |
245 Hammersmith Road, 3rd Floor London W6 8PW United Kingdom |
|
(3 |
) |
Job title and name of representative |
Chief Executive OfficerBobby Gaspar |
|
(4 |
) |
Description of business |
Development and commercialization of hematopoietic stem cell gene
therapy |
|
(5 |
) |
Share capital |
$29,463 thousand (as of September 30, 2023) |
|
(6 |
) |
Date of establishment |
2015 |
About Libmeldy / OTL-200Libmeldy® (atidarsagene
autotemcel), also known as OTL-200, has been approved by the
European Commission for the treatment of metachromatic
leukodystrophy (MLD) in patients characterized by biallelic
mutations in the ARSA gene leading to a reduction of the ARSA
enzymatic activity in children with i) late infantile or early
juvenile forms, without clinical manifestations of the disease, or
ii) the early juvenile form, with early clinical manifestations of
the disease, who still have the ability to walk independently and
before the onset of cognitive decline. Libmeldy is the first
therapy approved for eligible patients with early-onset MLD.
The most common adverse reaction attributed to treatment with
Libmeldy was the occurrence of anti-ARSA antibodies. In addition to
the risks associated with the gene therapy, treatment with Libmeldy
is preceded by other medical interventions, namely peripheral blood
mobilization and apheresis, followed by myeloablative conditioning,
which carry their own risks. During the clinical studies of
Libmeldy, the safety profiles of these interventions were
consistent with their known safety and tolerability.
For more information about Libmeldy, please see the Summary of
Product Characteristics (SmPC) available on the EMA website.
Libmeldy is approved in the European Union, UK, Iceland,
Liechtenstein and Norway. OTL-200 is an investigational therapy in
the U.S.
Libmeldy was developed in partnership with the San
Raffaele-Telethon Institute for Gene Therapy (SR-Tiget) in Milan,
Italy.
About Kyowa KirinKyowa Kirin aims to discover
novel medicines with life-changing value. As a Japan-based Global
Specialty Pharmaceutical Company, we have invested in drug
discovery and biotechnology innovation for more than 70 years and
are currently working to engineer the next generation
of antibodies and cell and gene therapies with the
potential to help patients affected by a severe or rare
disease. A shared commitment to our values, to sustainable growth,
and to making people smile unites us across our four regions –
Japan, Asia Pacific, North America, and EMEA/International. You can
learn more about the business of Kyowa Kirin at:
https://www.kyowakirin.com.
About Orchard TherapeuticsOrchard Therapeutics,
a Kyowa Kirin company, is a global gene therapy leader focused on
ending the devastation caused by genetic and other severe diseases
by discovering, developing, and commercializing new treatments that
tap into the curative potential of hematopoietic stem cell (HSC)
gene therapy. In this approach, a patient’s own blood stem cells
are genetically modified outside of the body and then reinserted,
with the goal of correcting the underlying cause of disease with a
single treatment.
Founded in 2015, Orchard’s roots go back to some of the first
research and clinical developments involving HSC gene therapy. Our
team has played a central role in the evolution of this technology
from a promising scientific idea to a potentially life-transforming
reality. Today, Orchard is advancing a pipeline of HSC gene
therapies designed to address serious diseases where the burden is
immense for patients, families and society and current treatment
options are limited or do not exist.
For more information, please visit www.orchard-tx.com.
Availability of Other Information About
OrchardInvestors and others should note that Orchard
communicates with its investors and the public using the company
website (www.orchard-tx.com), the investor relations website
(ir.orchard-tx.com), and on social media, including but not limited
to investor presentations and investor fact sheets, U.S.
Securities and Exchange Commission filings, press releases,
public conference calls and webcasts. The information that Orchard
posts on these channels and websites could be deemed to be material
information. As a result, Orchard encourages investors, the media,
and others interested in Orchard to review the information that is
posted on these channels, including the investor relations website,
on a regular basis. This list of channels may be updated from time
to time on Orchard’s investor relations website and may include
additional social media channels. The contents of Orchard’s website
or these channels, or any other website that may be accessed from
its website or these channels, shall not be deemed incorporated by
reference in any filing under the Securities Act of 1933.
Forward-looking StatementsThis press release
contains forward-looking statements, which are made pursuant to the
safe harbor provisions of the Private Securities Litigation Reform
Act of 1995. All statements that are not statements of historical
facts are, or may be deemed to be, forward-looking statements.
These statements are neither promises nor guarantees and are
subject to a variety of risks and uncertainties, many of which are
beyond Orchard’s control, which could cause actual results to
differ materially from those contemplated in these forward-looking
statements. In particular, these risks and uncertainties include,
without limitation, the risk that prior results, including signals
of safety and efficacy, will not be replicated or will not continue
in ongoing or future studies and the risk that long-term adverse
safety findings may be discovered. Given these uncertainties, the
reader is advised not to place any undue reliance on such
forward-looking statements.Other risks and uncertainties faced by
Orchard include those identified under the heading "Risk Factors"
in Orchard’s most recent annual or quarterly report filed with the
U.S. Securities and Exchange Commission (SEC), as well as
subsequent filings and reports filed with the SEC. The
forward-looking statements contained in this press release reflect
Orchard’s views as of the date hereof, and Orchard does not assume
and specifically disclaims any obligation to publicly update or
revise any forward-looking statements, whether as a result of new
information, future events or otherwise, except as may be required
by law.
CONTACTS:
For Kyowa Kirin
Hiroki Nakamura: Global/Japan
Corporate Communications Department
media@kyowakirin.com
Lauren Walrath
Vice President, Public Affairs – North America
lauren.walrath.g4@kyowakirin.com
+1-646-526-4454
Stacey Minton: EMEA
SVP, Corporate Affairs – Kyowa Kirin International
Stacey.Minton@kyowakirin.com
For Orchard Therapeutics
Benjamin Navon
+1 857-248-9454
Benjamin.Navon@orchard-tx.com
Orchard Therapeutics (NASDAQ:ORTX)
Historical Stock Chart
From Apr 2024 to May 2024
Orchard Therapeutics (NASDAQ:ORTX)
Historical Stock Chart
From May 2023 to May 2024