NEW HAVEN, Conn., March 30, 2020 /PRNewswire/ -- Biohaven
Pharmaceutical Holding Company Ltd. (NYSE: BHVN) today announced
positive topline results in its randomized, placebo-controlled
pivotal clinical trial (NCT03732638) evaluating the efficacy and
safety of oral rimegepant 75 mg for the preventive treatment of
migraine in both episodic and chronic migraine patients. The study
met the primary endpoint, demonstrating a statistically significant
reduction from baseline in monthly migraine days in patients
treated with rimegepant compared with placebo. Those receiving
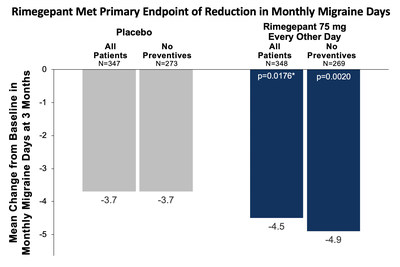
rimegepant 75 mg every other day (n=348) experienced a
statistically significant 4.5 day reduction from baseline in
monthly migraine days, compared to a 3.7 day reduction in the
placebo group (n=347; p=0.0176). Among study participants not
taking concomitant preventive treatment, there was a 4.9 day
reduction in monthly migraine days in the rimegepant group (n=273)
compared with a 3.7 day reduction in the placebo group (n=269;
nominal p=0.0020). A total of 22% of the study participants
were taking a concurrent preventive treatment, including topiramate
and amitriptyline. Importantly, 48% of the rimegepant group had at
least a 50% reduction from baseline in the mean number of moderate
to severe migraine days per month compared to 41% in the placebo
group.
Vlad Coric, M.D., Chief Executive
Officer of Biohaven commented, "Rimegepant is the first CGRP
targeting therapy to complete pivotal trials that demonstrate
efficacy in both the acute and preventive treatment of migraine.
The magnitude of effect in the rimegepant treated arm of a 4.5 day
reduction at 3 months in this study was on the higher end of what
has been demonstrated by other approved preventive drugs for
migraine patients with similar baselines. We believe rimegepant has
the potential to change the paradigm of migraine treatment,
offering patients the potential for dual-therapy action – acute and
preventive treatment — in one simple dose and convenient
formulation." Dr. Coric added, "We believe that, for prevention,
the ability to take an oral medication every other day rather than
recurring injection/intravenous therapy provides patients with a
simple, less invasive method of treatment and may allow patients to
feel more in control of their migraine. If successful in expanding
our label for NURTEC ODT to include prevention, patients for the
first time will have the convenience of one oral medication to
treat their migraine across the spectrum instead of having to mix
different acute and preventive medications."
Richard B. Lipton, M.D.,
Professor and Vice Chair of Neurology at the Albert Einstein
College of Medicine and Montefiore Health System, Director of the
Montefiore Headache Center commented, "I see many patients who
are discouraged by the limited current preventive treatment options
and continue to look for a better way to prevent disabling migraine
attacks. This is the first time that patients may be able to use a
single drug for both acute and preventive treatment. Particularly
impressive is the fact that the primary outcome measure of
reduction in monthly migraine days was achieved with every other
day dosing. I believe that rimegepant will fulfill significant
unmet needs for my patients as an oral agent that provides acute
and preventive treatment benefits."
This pivotal study enrolled patients with both episodic and
chronic migraine. The study evaluated the efficacy and safety of
rimegepant 75 mg (n=370) dosed every other day for the preventive
treatment of migraine versus placebo (n=371) in patients who had
migraine for at least one year and 4 to 18 moderate to severe
migraine attacks per month over three months prior to enrollment.
During the one-month observation period, patients experienced on
average 10.7 migraine days during the 4 week month, with 7.4
migraine days of moderate to severe pain intensity migraine during
the same period.
Robert Croop, M.D., Chief
Development Officer – Neurology at Biohaven added, "These data
demonstrate rimegepant's broad range of clinical activity to
potentially provide a new oral preventive treatment option for
people with migraine. The magnitude of effect in the rimegepant
treated arm with favorable safety and tolerability suggest that
rimegepant could be a best-in class oral therapy for both
preventive and acute treatment of migraine. We look forward to
sharing detailed results from the study at upcoming medical
conferences and continuing to advance this innovative oral
treatment for the preventive treatment of migraine."
The safety profile seen in the 370 patients who received
rimegepant 75 mg every other day was consistent with prior clinical
trial experience. There were no cases of ALT or AST > 3 x ULN
and bilirubin > 2 x ULN. An independent liver monitoring panel
did not determine any ALT/AST elevations to be in the categories of
probably or definitely related to study drug. Discontinuation rates
were higher in the placebo arm, largely driven by a greater number
of placebo patients (6%) withdrawing consent compared to rimegepant
treated participants (3%). Additional efficacy and safety study
results will be shared at medical conferences later this year.
Biohaven Conference Call Information
Biohaven is
hosting a conference call and webcast on Monday, March 30, 2020, at 8:30 a.m. ET. Participants are invited to
join the conference by dialing 877-407-9120 (toll-free) or
412-902-1009 (international). To access the audio webcast with
slides, please visit the "Events & Presentations" page in the
Investors section of the Company's website.
About Rimegepant
The activity of the neuropeptide CGRP
is thought to play a causal role in migraine pathophysiology.
Rimegepant is a CGRP receptor antagonist that works by
reversibly blocking CGRP receptors, thereby inhibiting the biologic
activity of the CGRP neuropeptide.
About Migraine
Nearly 40 million people in the
U.S. suffer from migraine and the World Health Organization
classifies migraine as one of the 10 most disabling medical
illnesses. Migraine is characterized by debilitating attacks
lasting four to 72 hours with multiple symptoms, including
pulsating headaches of moderate to severe pain intensity that can
be associated with nausea or vomiting, and/or sensitivity to sound
(phonophobia) and sensitivity to light (photophobia). There is a
significant unmet need for new treatments as more than 90 percent
of migraine sufferers are unable to work or function normally
during an attack.
CGRP Receptor Antagonism
Small molecule CGRP receptor
antagonists represent a novel class of drugs for the treatment of
migraine. For acute treatment, this unique mode of action
potentially offers an alternative to other agents, particularly for
patients who have contraindications to the use of triptans or who
have a poor response to triptans or are intolerant to them. Prior
studies have shown that agents such as injectable antibodies that
inhibit the CGRP pathway have clinical utility for the preventive
treatment of migraine.
About NURTEC ODT
NURTEC™ ODT (rimegepant) is
indicated for the acute treatment of migraine with or without aura
in adults. NURTEC ODT is not indicated for the preventive treatment
of migraine. NURTEC ODT should not be taken by patients with
hypersensitivity to NURTEC ODT or any of its components. If a
serious hypersensitivity reaction occurs, discontinue NURTEC ODT
and initiate appropriate therapy. Serious hypersensitivity
reactions have included dyspnea and rash, and can occur days after
administration.
In a clinical trial, the most common adverse reaction was nausea
(2% in patients who received NURTEC ODT compared to 0.4% in
patients who received placebo). Hypersensitivity, including
dyspnea and rash, occurred in less than 1% of patients treated with
NURTEC ODT. For more information about NURTEC ODT, visit
www.nurtec.com.
About Biohaven
Biohaven is a biopharmaceutical company focused on the
development and commercialization of innovative best-in-class
therapies to improve the lives of patients with debilitating
neurological and neuropsychiatric diseases. Biohaven's
neuroinnovation portfolio includes FDA-approved Nurtec™ ODT
(rimegepant) for the acute treatment of migraine and a broad
pipeline of late-stage product candidates across three distinct
mechanistic platforms: calcitonin gene-related peptide (CGRP)
receptor antagonism for the acute and preventive treatment of
migraine; glutamate modulation for obsessive-compulsive disorder,
Alzheimer's disease, and spinocerebellar ataxia; and
myeloperoxidase (MPO) inhibition for multiple system atrophy and
amyotrophic lateral sclerosis. For more information, visit
www.biohavenpharma.com.
Forward-looking Statement
This news release includes
forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995. The use of certain words,
including "believe", "continue", "may", and "will" and similar
expressions, are intended to identify forward-looking statements.
These forward-looking statements involve substantial risks and
uncertainties, including statements that are based on the current
expectations and assumptions of the Company's management about
NURTEC ODT as an acute treatment for patients with migraine.
Forward-looking statements include those related to: the Company's
ability to effectively commercialize NURTEC ODT, delays or problems
in the supply or manufacture of NURTEC ODT, complying with
applicable U.S. regulatory requirements, the expected timing,
commencement and outcomes of the Company's planned and ongoing
clinical trials, the timing of planned interactions and filings
with the FDA, the timing and outcome of expected regulatory
filings, the potential commercialization of the Company's
product candidates, the potential for the Company's product
candidates to be first in class or best in class therapies and the
effectiveness and safety of the Company's product candidates.
Various important factors could cause actual results or events to
differ materially from those that may be expressed or implied by
our forward-looking statements. Additional important factors to be
considered in connection with forward-looking statements are
described in the "Risk Factors" section of the Company's Annual
Report on Form 10-K filed with the Securities and Exchange
Commission on February 26, 2020. The
forward-looking statements are made as of this date and the Company
does not undertake any obligation to update any forward-looking
statements, whether as a result of new information, future events
or otherwise, except as required by law.
Biohaven Contact
Vlad
Coric, M.D.
Chief Executive Officer
Vlad.Coric@biohavenpharma.com
Media Contact
Mike
Beyer
Sam Brown Inc.
mikebeyer@sambrown.com
(312) 961-2502
NURTEC and NOJECTION are trademarks of Biohaven Pharmaceutical
Holding Company Ltd.
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-achieves-positive-results-in-pivotal-trial-of-oral-nurtec-dosed-every-other-day-for-the-preventive-treatment-of-migraine-301031432.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-achieves-positive-results-in-pivotal-trial-of-oral-nurtec-dosed-every-other-day-for-the-preventive-treatment-of-migraine-301031432.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.