The Ministry of Health and the Medicinal Cannabis Agency of
New Zealand grant Tilray the first approval under the new Medical
Cannabis Scheme to launch medical cannabis products across the
country
Tilray, Inc. (NASDAQ: TLRY),
a global pioneer in cannabis research, cultivation, production, and
distribution, today announced that it has received the necessary
approvals from New Zealand's Ministry of Health and the Medicinal
Cannabis Agency to launch Tilray medical cannabis products across
the country.
Tilray is one of the leading providers of medical cannabis in
Australia and New Zealand for commercial, compassionate access, and
research purposes and the first Licensed Producer to legally export
medical cannabis from North America to Australia and New Zealand
from its Good Manufacturing Practices (GMP)-certified facility in
Canada.
Brendan Kennedy, Tilray's Chief Executive Officer, said, "As
medical cannabis regulations continue to progress around the world,
we're incredibly honored to be recognized as a trusted partner in
offering the highest-quality medical cannabis products. We are
grateful to partner with New Zealand's Ministry of Health and the
Medicinal Cannabis Agency to improve access for patients in need
across the country."
George Polimenakos, General Manager, Tilray Australia, and New
Zealand, said, "We are proud to receive the first medical cannabis
product approvals according to New Zealand's new Medical Cannabis
Scheme. The rigorous approval process is a testament to the high
standards the Ministry of Health upholds for patients and the
quality of Tilray's medical cannabis products."
Tilray's product offering in New Zealand approved under the
scheme is centered around its Purified CBD products. Patients
looking to use medical cannabis have access to Tilray products by
obtaining prescriptions through their General Practitioner or
Specialist. Tilray expects to have an expanded range of
GMP-certified medical cannabis products, including cannabidiol
(CBD)-dominant, tetrahydrocannabinol (THC)-dominant and balanced
varieties available for patients by the second quarter of 2021.
In addition to supplying hospitals and pharmacies, Tilray is a
proud partner with several leading research institutions, including
the Murdoch Children's Research Institute in Australia, studying
the effectiveness of Tilray medical cannabis as a treatment for
pediatric patients with Intellectual Disabilities suffering from
Severe Behavioral Problems; a clinical trial in partnership with
The Government of New South Wales and University of Sydney Chris
O'Brien Lifehouse to develop a novel treatment for
chemotherapy-induced nausea, and a study led by the University of
Sydney examining the effects of driving under the influence of
cannabis.
Tilray looks forward to expanding its product offering further
and supplying more patients in-need with high-quality medical
cannabis products.
About Tilray®
Tilray is a global pioneer in the research, cultivation,
production, and distribution of cannabis and cannabinoids,
currently serving tens of thousands of patients and consumers in 17
countries spanning five continents.
Forward-Looking Statements
This press release contains "forward-looking statements" within
the meaning of the U.S. Private Securities Litigation Reform Act of
1995 and "forward-looking information" within the meaning of
Canadian securities laws, or collectively, forward-looking
statements. Forward-looking statements in this press release may be
identified by the use of words such as, "may", "would", "could",
"will", "likely", "expect", "anticipate", "believe, "intend",
"plan", "forecast", "project", "estimate", "outlook" and other
similar expressions. Forward-looking statements are not a guarantee
of future performance and are based upon a number of estimates and
assumptions of management in light of management's experience and
perception of trends, current conditions, and expected
developments, as well as other factors that management believes to
be relevant and reasonable in the circumstances, including
assumptions in respect of current and future European market
conditions, the current and future regulatory environment and
future approvals and permits. Actual results, performance, or
achievement could differ materially from that expressed in, or
implied by, any forward-looking statements in this press release,
and, accordingly, you should not place undue reliance on any such
forward-looking statements, and they are not guarantees of future
results. Please see the heading "Risk Factors" in Tilray's Annual
Report on Form 10-K, which was filed with the Securities and
Exchange Commission on February 19, 2021, for a discussion of the
material risk factors that could cause actual results to differ
materially from the forward-looking information. Tilray does not
undertake to update any forward-looking statements that are
included herein, except in accordance with applicable securities
laws.
Additional Information About the Transaction and Where to
Find It
This press release is not intended to and does not constitute an
offer to sell or the solicitation of an offer to subscribe for or
buy or an invitation to purchase or subscribe for any securities or
the solicitation of any vote or approval in any jurisdiction, nor
shall there be any sale, issuance or transfer of securities in any
jurisdiction in contravention of applicable law. This press release
is being made in respect of the proposed transaction involving
Aphria Inc., a corporation existing under the laws of the Province
of Ontario (“Aphria”) and Tilray pursuant to the terms of an
arrangement agreement by and among Aphria and Tilray and may be
deemed to be soliciting material relating to the proposed
transaction.
In connection with the proposed transaction, Tilray has filed a
preliminary proxy statement on Schedule 14A (which is subject to
completion and may be amended) containing important information
about the Transaction and related matters. Such preliminary proxy
statement is subject to review by the SEC before finalization. Such
preliminary proxy statement has also been made available by Aphria
and Tilray on their respective SEDAR profiles. Tilray will file a
definitive proxy statement and Aphria will file a management
information circular upon the completion of the SEC review process.
Additionally, Aphria and Tilray will file other relevant materials
in connection with the Transaction with the applicable securities
regulatory authorities. Investors and security holders of Aphria
and Tilray are urged to carefully read the entire management
information circular and definitive proxy statement (including any
amendments or supplements to such documents), respectively, before
making any voting decision with respect to the Transaction because
they contain important information about the Transaction and the
parties to the Transaction. The Aphria management information
circular and the Tilray definitive proxy statement will be mailed
to the Aphria and Tilray shareholders, respectively, as well as be
accessible on the SEDAR and EDGAR profiles of the respective
companies.
Investors and security holders of Tilray can obtain a free copy
of the preliminary proxy statement, and when available, the
definitive proxy statement, as well as other relevant filings
containing information about Tilray and the Transaction, including
materials incorporated by reference into the proxy statement,
without charge, at the SEC's website (www.sec.gov) or from Tilray
by contacting Tilray's Investor Relations at (203) 682-8253, by
email at Raphael.Gross@icrinc.com, or by going to Tilray's Investor
Relations page on its website at
https://ir.tilray.com/investor-relations and clicking on the link
titled "Financials."
Investors and security holders of Aphria will be able to obtain
a free copy of the management information circular, as well as
other relevant filings containing information about Aphria and the
Transaction, including materials incorporated by reference into the
information circular, without charge, on SEDAR at www.sedar.com or
from Aphria by contacting Aphria's investor relations at
investors@aphria.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210310005327/en/
Tilray Global: Berrin Noorata news@tilray.com
Investors Raphael Gross 203-682-8253 Raphael.Gross@icrinc.com
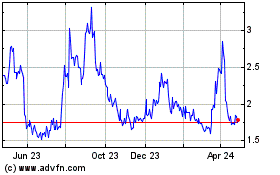
Tilray Brands (NASDAQ:TLRY)
Historical Stock Chart
From Mar 2024 to Apr 2024
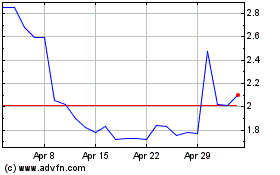
Tilray Brands (NASDAQ:TLRY)
Historical Stock Chart
From Apr 2023 to Apr 2024