FDA Clears Cutera’s AviClear™ Acne Device
March 25 2022 - 8:00AM
Business Wire
AviClear is the First Device Cleared for the
Treatment of Mild, Moderate and Severe Acne AviClear has been
Proven Safe and Effective Through an Extensive Clinical Trial
Provides Long-term Resolution of Acne Across All Skin Types
CUTERA, INC. (Nasdaq: CUTR) ("Cutera" or the "Company"), a
leading provider of dermatology solutions, today announced the U.S.
Food and Drug Administration’s 510(k) clearance of AviClear, the
first and only energy-based device to receive this designation for
the treatment of mild, moderate, and severe acne.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20220317006022/en/
(Photo: Business Wire)
AviClear is a laser treatment that offers a safe,
prescription-free solution for acne.1 In addition to reducing
existing acne, clinical trials show that future breakout episodes
are shorter, less intense, and more infrequent following the
AviClear procedure. Further, acne clearance results continue to
improve over time, demonstrating the long-term efficacy of this
novel treatment. Importantly, no pain mitigation was utilized or
required by any clinical study participant.
Acne vulgaris is a nearly universal skin disease, with
approximately 50 million North American teens and young adults
seeking treatment each year.2 Overproduction of sebum by the
sebaceous glands is one of the leading causes of acne.3 AviClear
resolves acne at the source by selectively targeting the sebocytes
and suppressing sebum production.
“Existing device treatments for acne are neither long-lasting
nor particularly effective,” said Jeffrey S. Dover, MD, FRCPC,
board-certified dermatologist and Cutera advisory board member.
“Topical therapies yield temporary results and oral medications
present several challenges. AviClear offers patients a safe,
well-tolerated, drug-free approach with durable results, which
significantly shifts the treatment paradigm for acne.”
"Physicians and patients have long sought a modern alternative
to the acne pills, peels and topicals that have been static for
nearly 30 years,” said David Mowry, CEO of Cutera. "Developed with
extensive physician and patient input, AviClear was created to
redefine the treatment of acne – all without a prescription.”
AviClear is expected to be made available to physicians
throughout the United States over the course of 2022. Doctors and
consumers are encouraged to visit www.AviClear.com and sign up for
updates on product availability and local treatment providers.
About Cutera, Inc. Brisbane, California-based Cutera is a
leading provider of laser and other energy-based systems for
dermatologists and aesthetic practitioners worldwide. Since 1998,
Cutera has been developing innovative, easy-to-use products that
harness the power of science and nature to enable physicians and
other qualified practitioners to offer safe and effective
treatments to their patients. For more information, call +1
415-657- 5500 or 1-888-4CUTERA or visit www.cutera.com.
References
- Data on file, FDA clearance study. Cutera, Inc.
-
https://jamanetwork.com/journals/jamadermatology/fullarticle/479093
- O’Neill AM, Gallo RL. Host-microbiome interactions and recent
progress into understanding the biology of acne vulgaris.
Microbiome 2018;6: 177.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220317006022/en/
EvolveMKD Bridget Callahan Cutera@EvolveMKD.com
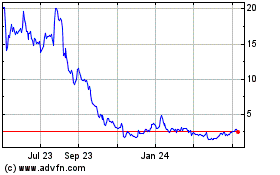
Cutera (NASDAQ:CUTR)
Historical Stock Chart
From Jun 2024 to Jul 2024
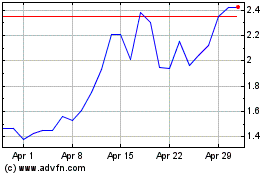
Cutera (NASDAQ:CUTR)
Historical Stock Chart
From Jul 2023 to Jul 2024