false
0001412486
0001412486
2023-11-28
2023-11-28
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
8-K
CURRENT REPORT
Pursuant
to Section 13 OR 15(d) of The Securities Exchange Act of 1934
Date
of Report (Date of earliest event reported): November 28, 2023
Cocrystal Pharma, Inc.
(Exact
name of registrant as specified in its charter)
| Delaware |
|
001-38418 |
|
35-2528215 |
(State
or other jurisdiction
of
incorporation) |
|
(Commission
File
Number) |
|
(IRS
Employer
Identification
No.) |
19805
N. Creek Parkway
Bothell,
WA |
|
98011 |
| (Address
of principal executive offices) |
|
(Zip
Code) |
Registrant’s
telephone number, including area code: (786) 459-1831
N/A
(Former
name or former address, if changed since last report.)
Check
the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions (see General Instruction A.2. below):
| ☐ |
Written
communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| |
|
| ☐ |
Soliciting
material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| |
|
| ☐ |
Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| |
|
| ☐ |
Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities
registered pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading
Symbol(s) |
|
Name
of each exchange on which registered |
| Common
Stock |
|
COCP |
|
The
Nasdaq Stock Market LLC
(The
Nasdaq Capital Market) |
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405
of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging
growth company ☐
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item
7.01 Regulation FD Disclosure.
On
November 28, 2023, Sam Lee, President and Co-Chief Executive Officer of Cocrystal Pharma, Inc. (the “Company”) is presenting
at the World Vaccine Conference. A copy of the slide presentation is being furnished as Exhibit 99.1 to this Current Report on Form 8-K.
The Company will also make available a webcast recording of the presentation on its website at www.cocrystalpharma.com beginning on November
29, 2023. In addition, on November 29, 2023 the Company issued a press release regarding the presentation and the Company’s
CC-42344 influenza A product candidate described therein, a copy of which is being furnished as Exhibit 99.2 to this Current Report on
Form 8-K.
The
information in this Item 7.01 (including Exhibit 99.1) shall not be deemed “filed” for purposes of Section 18 of the Securities
Exchange Act of 1934 (the “Exchange Act”) or otherwise subject to the liabilities under such section, and shall not be deemed
to be incorporated by reference into any filing of the Company under the Securities Act of 1933 or the Exchange Act.
Item
9.01 Financial Statements and Exhibits.
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by
the undersigned hereunto duly authorized.
Dated:
November 29, 2023
| |
Cocrystal Pharma, Inc. |
| |
|
|
| |
By: |
/s/
James Martin |
| |
Name: |
James
Martin |
| |
Title: |
Chief
Financial Officer and Co-Chief Executive Officer |
Exhibit 99.1














Exhibit
99.2

Cocrystal
Pharma Highlights its Novel Inhaled and Oral Influenza A Antiviral CC-42344 at the World Vaccine Congress West Coast
BOTHELL,
Wash. (November 29, 2023) – Cocrystal Pharma, Inc. (Nasdaq: COCP) (“Cocrystal” or the “Company”), announces
the presentation of favorable data demonstrating activity of its potent broad-spectrum PB2 inhibitor CC-42344 against pandemic and seasonal
influenza A strains at the World Vaccine Congress West Coast. Cocrystal has initiated a Phase 2a human challenge trial with oral
CC-42344 in the UK in subjects infected with influenza A, and plans to begin a Phase 1 trial with inhaled CC-42344 as a
potential influenza A treatment and prophylaxis in Australia in the first half of 2024.
In
his presentation, “Taking a new route: Development of novel inhaled and oral influenza antiviral, CC-42344,” Cocrystal
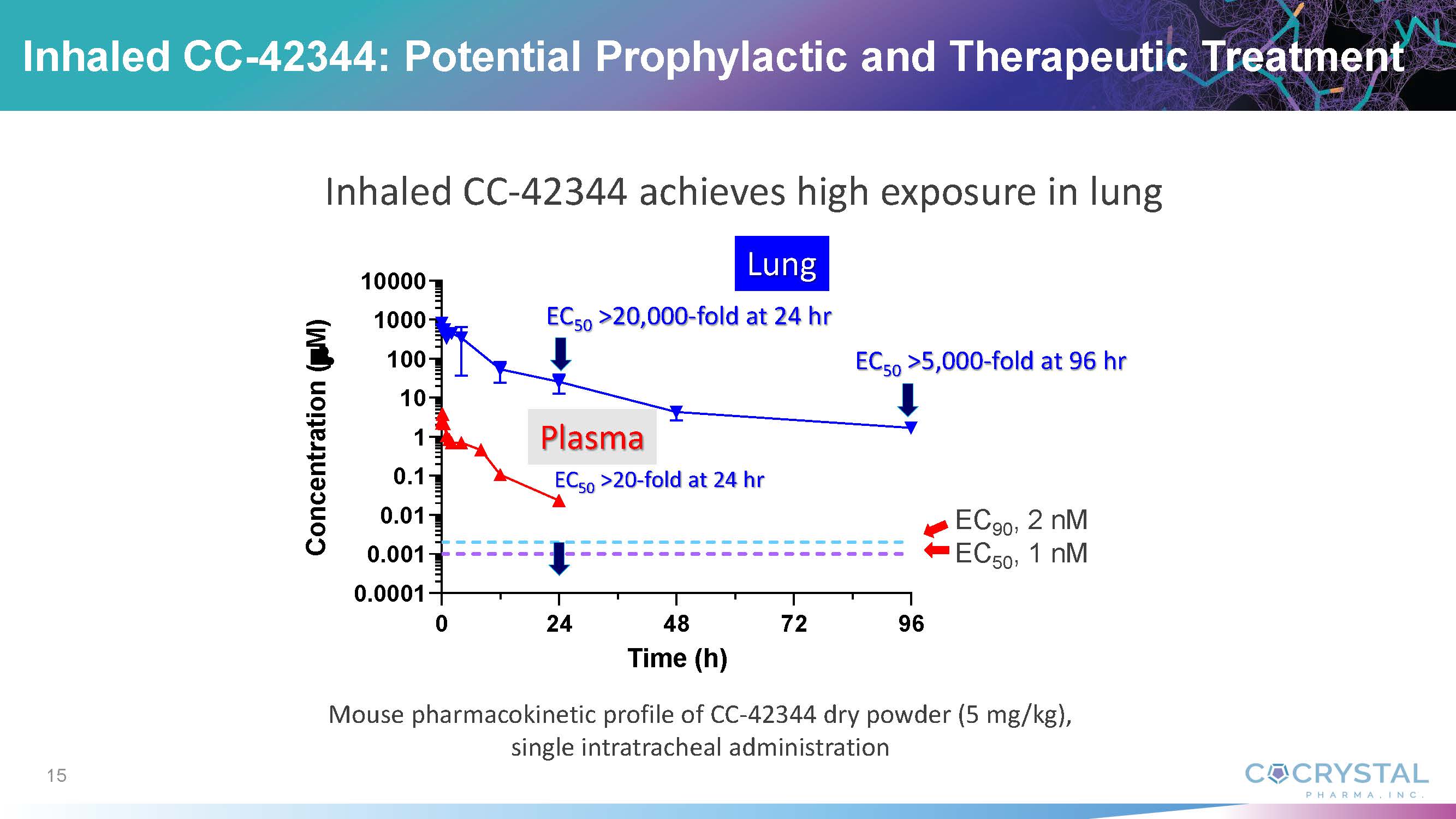
President and co-CEO Sam Lee, PhD discussed the potential prevention and therapy of influenza infection using inhaled CC-42344. Dr. Lee
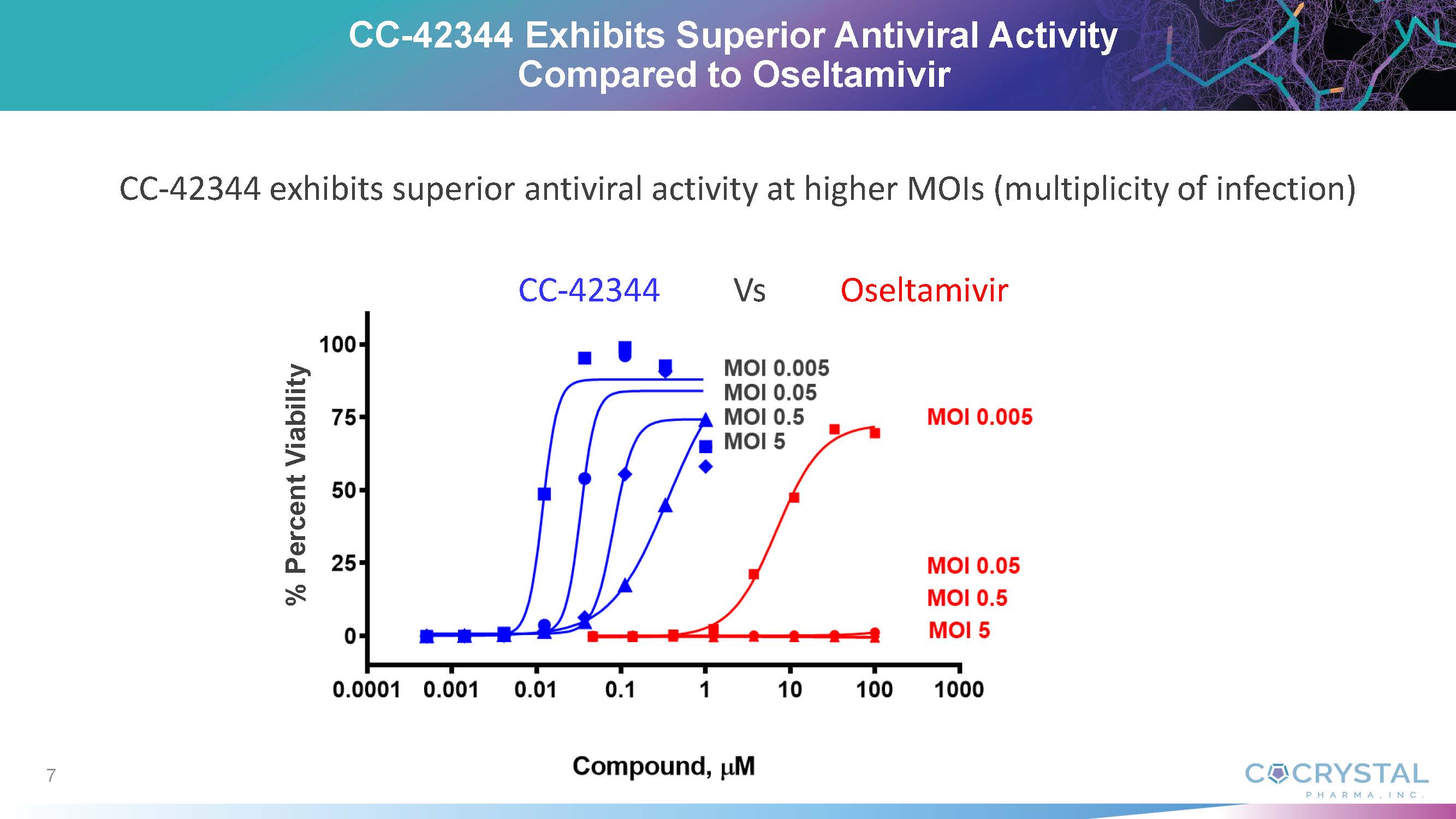
commented that CC-42344 exhibits superior antiviral activity compared with oseltamivir (Tamiflu®) and demonstrates a novel mechanism
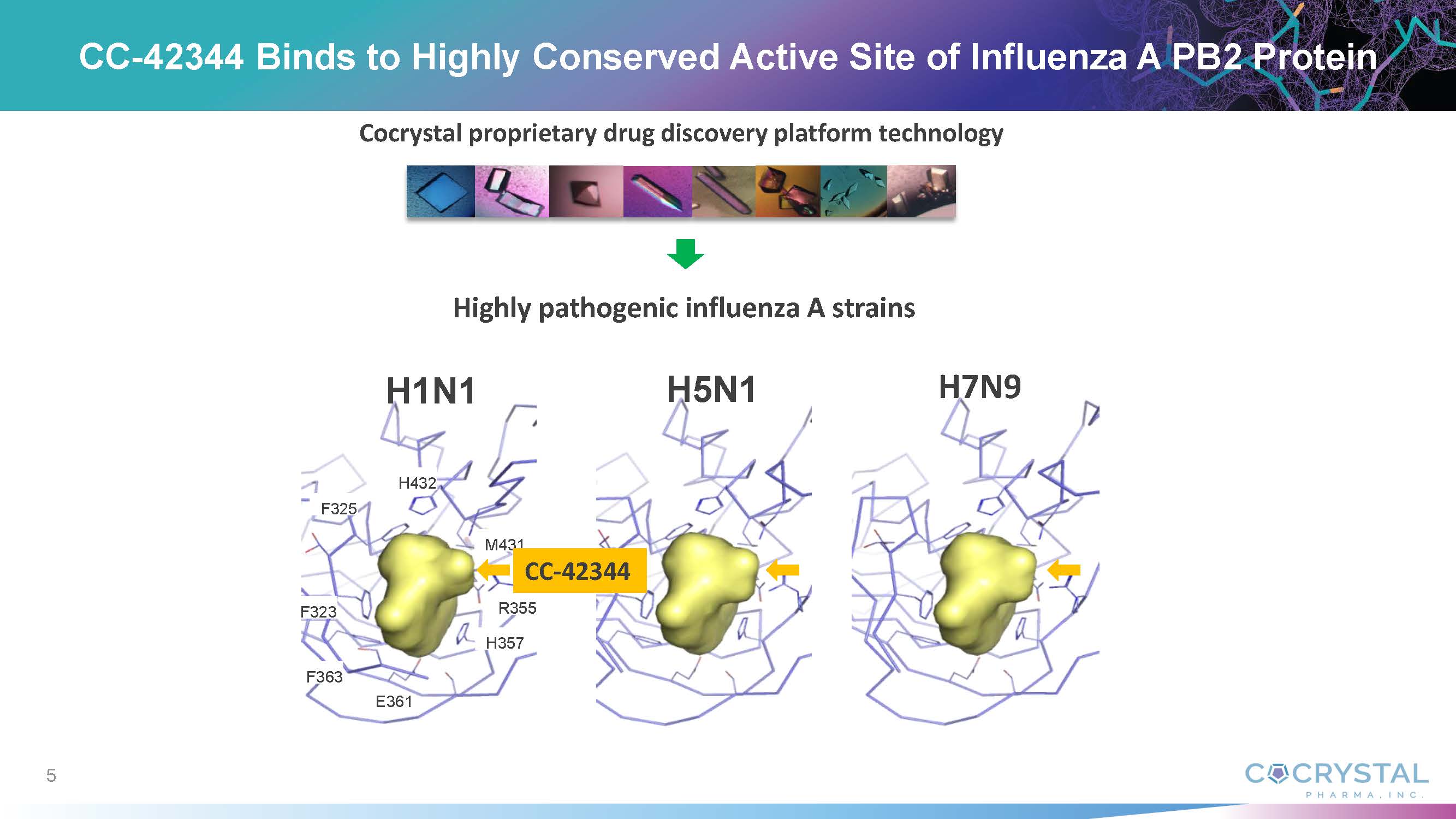
of action with high barrier of resistance. He noted that Cocrystal discovered and developed CC-42344 utilizing the Company’s proprietary
structure-based drug discovery platform technology, which is proving successful in delivering multiple broad-spectrum antiviral leads
for influenza and other viral diseases.
“We
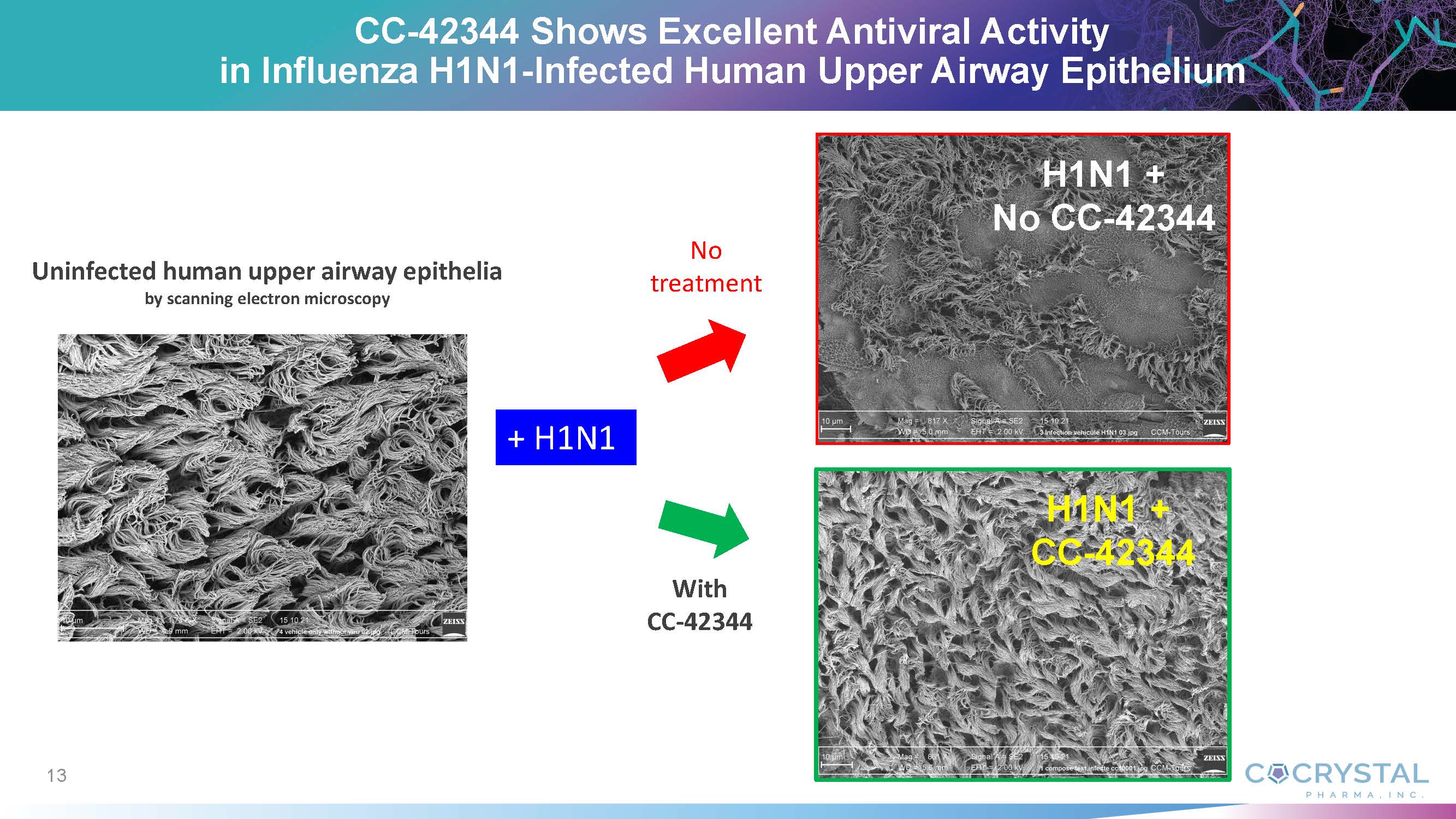
are excited to accomplish another important milestone with the influenza antiviral CC-42344. Based on our recent preclinical data, CC-42344
exhibits superior lung exposure, a favorable safety profile, and efficacy in influenza-infected human lung epithelia. We also demonstrated
highly efficient delivery of inhaled CC-42344 into the lung,” he said. “Inhaled CC-42344 could be developed for both therapeutic
and prophylactic influenza treatment. We are encouraged by this potential breakthrough influenza treatment option.”
Slides
from the presentation are available on the Company’s website.
About
CC-42344
CC-42344
is specifically designed to be effective against all significant pandemic and seasonal influenza A strains and to have a high barrier
to resistance due to the way the virus’ replication machinery is targeted. CC-42344 targets the influenza polymerase, an essential
replication enzyme with several highly essential regions common to multiple influenza strains. In vitro testing showed CC-42344’s
excellent antiviral activity against influenza A strains, including pandemic and seasonal strains, as well as against strains resistant
to certain approved influenza antivirals, while also demonstrating favorable pharmacokinetic and safety profiles.
About
Seasonal Influenza
Each
year there are approximately 1 billion cases of seasonal influenza worldwide, 3-5 million severe illnesses and up to 650,000 deaths,
according to the World Health Organization. On average
about 8% of the U.S. population contracts influenza each season. Influenza is responsible
for approximately $10.4 billion in direct costs for hospitalizations and outpatient visits for adults in the U.S. annually.
About
Cocrystal Pharma, Inc.
Cocrystal
Pharma, Inc. is a clinical-stage biotechnology company discovering and developing novel antiviral therapeutics that target the replication
process of influenza viruses, coronaviruses (including SARS-CoV-2) noroviruses and hepatitis C viruses. Cocrystal employs unique structure-based
technologies and Nobel Prize-winning expertise to create first- and best-in-class antiviral drugs. For further information about Cocrystal,
please visit www.cocrystalpharma.com.
Cautionary
Note Regarding Forward-Looking Statements
This
press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including
statements regarding the Company’s ongoing Phase 2a human challenge trial for CC-42344 as a product candidate for oral treatment
of influenza A, and the planned initiation of a Phase 1 clinical trial in the first half of 2024 for CC-42344 as a product candidate
for inhaled treatment of influenza A, and the potential efficacy and clinical benefits of, and market for, such product candidate. The
words “believe,” “may,” “estimate,” “continue,” “anticipate,” “intend,”
“should,” “plan,” “could,” “target,” “potential,” “is likely,”
“will,” “expect” and similar expressions, as they relate to us, are intended to identify forward-looking statements.
We have based these forward-looking statements largely on our current expectations and projections about future events. Some or all of
the events anticipated by these forward-looking statements may not occur. Important factors that could cause actual results to differ
from those in the forward-looking statements include, but are not limited to, risks relating to our
ability to proceed with the Phase 2a and Phase 1 studies referred to above including recruiting volunteers for and procuring or manufacturing
materials for such studies by our clinical research organizations and vendors, and the results of such studies. Further information
on our risk factors is contained in our filings with the SEC, including our Annual Report on Form 10-K for the year ended December 31,
2022. Any forward-looking statement made by us herein speaks only as of the date on which it is made. Factors or events that could cause
our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We undertake no obligation
to publicly update any forward-looking statement, whether as a result of new information, future developments or otherwise, except as
may be required by law.
Investor
Contact:
LHA
Investor Relations
Jody
Cain
310-691-7100
jcain@lhai.com
Media
Contact:
JQA
Partners
Jules
Abraham
917-885-7378
Jabraham@jqapartners.com
#
# #
v3.23.3
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Cocrystal Pharma (NASDAQ:COCP)
Historical Stock Chart
From Jun 2024 to Jul 2024
Cocrystal Pharma (NASDAQ:COCP)
Historical Stock Chart
From Jul 2023 to Jul 2024