Illumina Grows Clinical IVD Portfolio with Introduction of NextSeq™ 550Dx System and Expanded Use of MiSeq™Dx System
November 15 2017 - 9:00AM
Business Wire
Systems, reagents and software to cover broad
range of clinical applications
Illumina, Inc. (NASDAQ: ILMN) announced today the introduction
of its NextSeq™ 550Dx instrument, the company’s second
FDA-regulated and CE-IVD marked next-generation sequencing (NGS)
system. The company also announced that the intended use for the
MiSeq™Dx instrument has been updated to include the use of DNA
libraries generated from formalin-fixed paraffin embedded (FFPE)
tissues, paving the way for clinical labs to use FFPE samples when
developing clinical tests for new applications. With the NextSeq
550Dx and the MiSeqDx, Illumina now has a highly robust diagnostic
NGS product portfolio capable of covering a broad range of clinical
applications to serve almost any clinical lab need for NGS-based in
vitro diagnostic (IVD) products.
This press release features multimedia. View
the full release here:
http://www.businesswire.com/news/home/20171115005389/en/
Illumina NextSeq 550Dx (Photo: Business
Wire)
NextSeq 550Dx
Illumina introduces the NextSeq 550Dx instrument, which is the
second FDA regulated CE-IVD market platform to deliver the power of
high-throughput NGS to the clinical laboratory. With dual boot
functionality, the NextSeq 550Dx platform includes a diagnostic
mode (Dx mode) and a research mode, which provides the flexibility
to perform both clinical research and develop a range of IVD assays
on a single instrument. For large clinical laboratories, the
NextSeq 550Dx instrument offers a validated high-throughput
platform and will soon provide access to an expanding pipeline of
clinical applications in oncology and reproductive health.
Assays that are run on the NextSeq 550Dx instrument follow a
simple process including sequencing and data analysis. The setup of
workflows for different applications are fully integrated in
NextSeq 550Dx software. The instrument also offers fully-integrated
onboard analysis software with modular software architecture to
support current and future assays.
“NGS is beginning to play a significant role in the clinic,”
said Garret Hampton, Executive Vice President of Clinical Genomics
at Illumina. “Illumina is working to provide clinical solutions
that support the diagnosis and management of complex diseases, as
well as expanding the IVD menu.”
For additional information on the NextSeq 550Dx, please visit
www.illumina.com/nextseq550dx.
MiSeqDx
The expanded intended use of the MiSeqDx instrument, which was
the first FDA regulated NGS sequencer, enables the development of
additional advanced diagnostic products and services at clinical
labs by giving clinicians access to information generated on
Illumina’s NGS technology. In addition to the updated intended use
for the MiSeqDx system, Illumina also offers the regulated MiSeqDx
Reagent Kit v3, the latest version of Illumina sequencing chemistry
available to MiSeqDx customers, and the TruSeq™ Custom Amplicon Kit
Dx for library preparation. These new products replace the MiSeqDx
Universal Kit 1.0, which was regulated by the FDA in 2013 and
allowed clinical labs to conduct DNA analysis using DNA obtained
from human whole blood.
“The expansion and launch of these regulated platforms represent
regulatory milestones important to the clinical community,” said
Kathy Davy, Vice President of Marketing, Clinical Genomics at
Illumina. “The most relevant genetic information that can help
cancer patients is often contained in FFPE tissues removed during
surgery. Information gleaned from these samples can inform
clinicians about tumor biology and help guide therapeutic
selection.”
Illumina has updated the existing intended use for the MiSeqDx
system to include the use of FFPE sample inputs for assays
performed on the instrument. This update will support the launch of
the following new reagent kits:
- TruSeq Custom Amplicon Kit Dx
- TruSeq Custom Amplicon Dx – FFPE QC
Kit
- MiSeqDx Reagent Kit v3 (2x300)
The MiSeqDx now has the following IVD menu:
- Cystic Fibrosis 139-Variant Assay
- Cystic Fibrosis Sequencing Assay
- Extended RAS Panel
- Germline Variant Module
- Somatic Variant Module
Illumina and its IVD development partners are now developing
assays that use FFPE as a starting point on the MiSeqDx instrument
and are working to bring additional IVD tests to the MiSeqDx. For
more information on the expanded use of the MiSeqDx system, visit
http://www.illumina.com/miseqdx.
About Illumina
Illumina is improving human health by unlocking the power
of the genome. Our focus on innovation has established us as the
global leader in DNA sequencing and array-based technologies,
serving customers in the research, clinical, and applied
markets. Our products are used for applications in the life
sciences, oncology, reproductive health, agriculture, and other
emerging segments. To learn more,
visit www.illumina.com and follow @illumina.
Use of forward-looking statements
This release contains forward-looking statements that involve
risks and uncertainties. These forward-looking statements are based
on our expectations as of the date of this release and may differ
materially from actual future events or results. Among the
important factors that could cause actual results to differ
materially from those in any forward-looking statements are (i) our
ability to further develop and commercialize our instruments and
consumables, and to deploy new products such as the NextSeq 550Dx
instrument, services and applications, and expand the markets for
our technology platforms; (ii) our ability to manufacture robust
instrumentation and consumables; (iii) our ability to successfully
identify and integrate acquired technologies, products or
businesses; (iv) the future conduct and growth of the business and
the markets in which we operate; and (v) challenges inherent in
developing, manufacturing, and launching new products and services,
together with other factors detailed in our filings with
the Securities and Exchange Commission, including our most
recent filings on Forms 10-K and 10-Q, or in information disclosed
in public conference calls, the date and time of which are released
beforehand. We undertake no obligation, and do not intend, to
update these forward-looking statements, to review or confirm
analysts’ expectations, or to provide interim reports or updates on
the progress of the current quarter.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20171115005389/en/
Illumina, Inc.Investors:Jacquie Ross,
CFA858-255-5243IR@illumina.comorMedia:Tina
Amirkiai858-882-6822PR@illumina.com
Illumina (NASDAQ:ILMN)
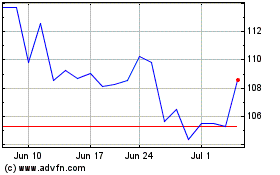
Historical Stock Chart
From Mar 2024 to Apr 2024
Illumina (NASDAQ:ILMN)
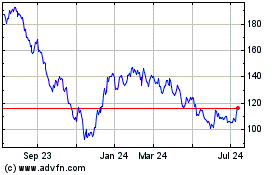
Historical Stock Chart
From Apr 2023 to Apr 2024