Highly Statistically Significant Results (P<0.001)
Demonstrated on Primary Composite Endpoint and All Secondary
Endpoints
DUBLIN, Nov. 17, 2016 /CNW/ -- Endo
International plc (NASDAQ / TSX: ENDP) announced today positive
results from its Phase 2b study of collagenase clostridium
histolyticum (or "CCH") for the treatment of edematous
fibrosclerotic panniculopathy ("EFP"), commonly known as cellulite.
CCH is known in its currently approved indications in the U.S. as
XIAFLEX® for adult Dupuytren's contracture and Peyronie's disease.
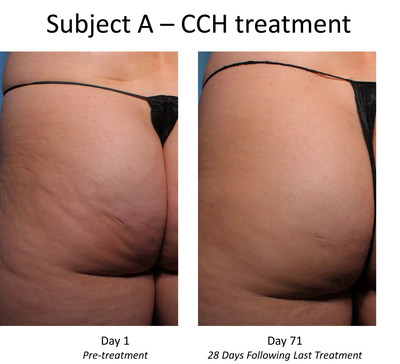
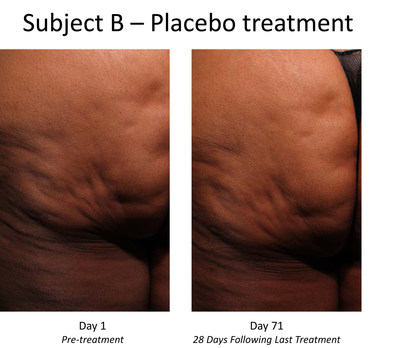
Trial subjects receiving CCH showed statistically significant
levels of improvement in the appearance of cellulite with
treatment, as measured by the trial's primary endpoint
(p<0.001), compared to those subjects receiving placebo. CCH was
well-tolerated in the actively treated subjects with most adverse
events (AEs) being mild to moderate in severity, and primarily
limited to the local injection area.
"We continue to be encouraged and very excited by the promising
results of our CCH program, especially in the cellulite indication
– a condition which affects many millions of people," said Dr.
Susan Hall, Executive Vice
President, Chief Scientific Officer and Global Head of R&D at
Endo. "We believe these data further reinforce our belief that CCH
could be a potential treatment option for those with cellulite and
we look forward to working with FDA to efficiently and effectively
advance our development of the program into Phase 3."
The Phase 2b trial enrolled 375 women with moderate or severe
cellulite aged 18 years or older in the
United States. Each subject received up to three treatment
sessions of CCH (0.84 mg / session) or placebo with each treatment
session occurring approximately 21 days apart. Twelve injections
were administered into cellulite dimples during each session across
an entire treatment quadrant – left or right buttock or left or
right posteriolateral thigh. At both the outset and conclusion of
the study period (28 days after the last treatment), cellulite
severity was assessed by each patient and clinician using two
photonumeric cellulite severity scales developed by Endo and
third-party experts. The scales – the Photonumeric Cellulite
Severity Scale (PCSS) – are 5-point scales ranging from 0 (no
cellulite) to 4 (severe cellulite) that measure improvement in the
appearance of cellulite.
The Phase 2b trial's primary endpoint was the proportion of
composite responders at Day 71 defined as subjects with a 2-point
improvement in severity from baseline in the clinician-reported
(CR) PCSS and a 2-point improvement in the patient-reported (PR)
PCSS. Additional endpoints include a composite of 1-point
responders, the percentage of responders with 1-point and 2-point
improvements on the CR-PCSS and PR-PCSS, assessment of improvement
by patient and clinician using the Global Aesthetic Improvement
Scale (GAIS); subject satisfaction, and change in the Hexsel
cellulite severity scale.
"I believe these positive results demonstrate that CCH has the
potential to be a treatment option for cellulite, a condition for
which there are very limited safe and proven effective treatments,"
said Mitchel P. Goldman, MD, Medical
Director, Cosmetic Laser Dermatology. "With so many millions of
women affected by cellulite, there is a substantial need for new
and innovative therapeutic options."
Key Phase 2b Trial Results Include:
- Subjects receiving CCH demonstrated a highly statistically
significant improvement in the primary endpoint of composite
investigators' and patients' assessments of the appearance of
cellulite, as measured by a two-point improvement in both the
CR-PCSS and PR-PCSS scores, with a p-value of <0.001 versus
placebo
- Subjects receiving CCH demonstrated a highly statistically
significant improvement in the composite investigators' and
patients' assessments of the appearance of cellulite, as measured
by a one-point improvement in both the CR-PCSS and PR-PCSS scores,
with a p-value of <0.001 versus placebo
- A highly significant proportion of CCH subjects reported being
"Satisfied" or "Very Satisfied" with their cellulite treatment,
compared to placebo subjects, with a p-value of <0.001
- A highly significant proportion of CCH subjects were reported
as "Improved" or "Very Improved" or "Very Much Improved" in global
appearance of their cellulite area as assessed by the subjects and
investigators, compared to placebo subjects, with a p-value of
<0.001
- CCH was well-tolerated by all dose groups with most adverse
events (AEs) being mild to moderate and primarily limited to the
local injection area; 92 percent of all related AEs were mild to
moderate in the CCH group compared to 96 percent in the placebo
group; the most common AEs were expected and included injection
site bruising (approximately 75 percent) and injection site pain
(approximately 59 percent)
"These data are exciting for the medical community and
definitely warrant further investigation and development," said
Neil Sadick, MD, Clinical Professor
of Dermatology, Weill Cornell Medical College. "By successfully
advancing this program, we could offer patients currently seeking
cellulite treatment a new and promising option."
Results for an earlier Phase 2a trial of CCH for the treatment
of cellulite demonstrated that three doses of CCH (low (0.06mg),
mid (0.48mg) and high (0.84mg)) showed an improvement in the
appearance of cellulite as measured by the trial endpoints of an
investigator and a patient score on the GAIS, which was adapted for
use in cellulite. The mid and high dose groups demonstrated a
statistically significant improvement in the appearance of
cellulite, as measured by GAIS scores, with a p-value of <0.05
compared to placebo for both endpoints. In the mid and high dose
groups, 68 percent of patients reported being "Satisfied" or "Very
Satisfied" with the results of their treatment, compared to only 34
percent of patients randomized to placebo. CCH was well-tolerated
by all dose groups with most adverse events (AEs) being mild to
moderate and primarily limited to the local injection area.
About Cellulite
Cellulite is a localized
metabolic disorder of tissue under the skin that has been reported
in 85 to 98 percent of post-pubertal females and affects women of
all races and ethnicities[i] [ii]. The condition can involve the
loss of elasticity or shrinking of collagen cords, called "septae,"
that attach the skin to the muscle layers below. When fat in
cellulite prone areas swells and expands, the septae tether the
skin, which causes the surface dimpling characteristic of
cellulite[iii]. CCH is intended to target and lyse, or break, those
collagen tethers with the goal of releasing the skin dimpling and
potentially resulting in smoothing of the skin. Despite multiple
therapeutic approaches for the attempted treatment of cellulite,
there are no FDA-approved pharmacological treatments and little
scientific evidence that any current treatments are
beneficial[iv].
About XIAFLEX
XIAFLEX® (collagenase clostridium
histolyticum, or CCH) is a biologic approved in the U.S., EU,
Canada, Australia and Japan for the treatment of adult Dupuytren's
contracture (DC) patients with a palpable cord and in the U.S. and
EU for the treatment of adult men with Peyronie's disease (PD) with
a palpable plaque and penile curvature deformity of at least 30
degrees at the start of therapy. XIAFLEX® consists of a combination
of two subtypes of collagenase, derived from Clostridium
histolyticum. Together, the collagenase subtypes are thought to
work synergistically to break the bonds of the collagen structure.
XIAFLEX® has been granted Orphan status in the U.S. by the FDA for
DC and PD.
IMPORTANT SAFETY INFORMATION FOR XIAFLEX®
(DUPUYTREN'S CONTRACTURE)
INDICATION
XIAFLEX® is indicated for the treatment of adult
patients with Dupuytren's contracture with a palpable cord.
IMPORTANT SAFETY INFORMATION FOR XIAFLEX®
- XIAFLEX® is contraindicated in patients with a
history of hypersensitivity to XIAFLEX® or to
collagenase used in any other therapeutic application or
application method
- In the controlled and uncontrolled portions of clinical trials
in Dupuytren's contracture, flexor tendon ruptures occurred after
XIAFLEX® injection. Injection of XIAFLEX®
into collagen-containing structures such as tendons or ligaments of
the hand may result in damage to those structures and possible
permanent injury such as tendon rupture or ligament damage.
Therefore, XIAFLEX® should be injected only into the
collagen cord with a MP or PIP joint contracture, and care should
be taken to avoid injecting into tendons, nerves, blood vessels, or
other collagen-containing structures of the hand. When injecting a
cord affecting a PIP joint of the fifth finger, the needle
insertion should not be more than 2 to 3 mm in depth and avoid
injecting more than 4 mm distal to the palmar digital crease
- Other XIAFLEX®-associated serious local adverse
reactions in the controlled and uncontrolled portions of the
studies included pulley rupture, ligament injury, complex regional
pain syndrome (CRPS), sensory abnormality of the hand, and skin
laceration (tear). In a historically controlled post-marketing
trial, the incidence of skin laceration (22%) was higher for
subjects treated with two concurrent injections of
XIAFLEX® compared with subjects treated with up to three
single injections in the placebo-controlled premarketing trials
(9%). Cases of skin laceration requiring skin graft after finger
extension procedures have been reported post-marketing. Signs or
symptoms that may reflect serious injury to the injected
finger/hand should be promptly evaluated because surgical
intervention may be required
- In the controlled portions of the clinical trials in
Dupuytren's contracture, a greater proportion of
XIAFLEX®-treated patients (15%) compared to
placebo-treated patients (1%) had mild allergic reactions
(pruritus) after up to 3 injections. The incidence of
XIAFLEX®-associated pruritus increased after more
XIAFLEX® injections in patients with Dupuytren's
contracture
- Because XIAFLEX® contains foreign proteins, severe
allergic reactions to XIAFLEX® can occur. Anaphylaxis
was reported in a post-marketing clinical study in one patient who
had previous exposure to XIAFLEX® for the treatment of
Dupuytren's contracture. Healthcare providers should be prepared to
address severe allergic reactions following XIAFLEX®
injections
- In the XIAFLEX® trials in Dupuytren's contracture,
70% and 38% of XIAFLEX®-treated patients developed an
ecchymosis/contusion or an injection site hemorrhage, respectively.
Patients with abnormal coagulation (except for patients taking
low-dose aspirin, eg, up to 150 mg per day) were excluded from
participating in these studies. Therefore, the efficacy and safety
of XIAFLEX® in patients receiving anticoagulant
medications (other than low-dose aspirin, eg, up to 150 mg per day)
within 7 days prior to XIAFLEX® administration is not
known. In addition, it is recommended to avoid use of
XIAFLEX® in patients with coagulation disorders,
including patients receiving concomitant anticoagulants (except for
low-dose aspirin)
- In the XIAFLEX® clinical trials for Dupuytren's
contracture, the most common adverse reactions reported in ≥25% of
patients treated with XIAFLEX® and at an incidence
greater than placebo were edema peripheral (eg, swelling of the
injected hand), contusion, injection site hemorrhage, injection
site reaction, and pain in the injected extremity
IMPORTANT SAFETY INFORMATION FOR XIAFLEX®
(PEYRONIE'S DISEASE)
INDICATION
XIAFLEX® is
indicated for the treatment of adult men with Peyronie's disease
with a palpable plaque and curvature deformity of at least 30
degrees at the start of therapy.
IMPORTANT SAFETY INFORMATION FOR XIAFLEX®
WARNING:
CORPORAL RUPTURE (PENILE FRACTURE) OR OTHER SERIOUS PENILE INJURY
IN THE TREATMENT OF PEYRONIE'S DISEASE
Corporal rupture
(penile fracture) was reported as an adverse reaction in 5 of 1044
(0.5%) XIAFLEX®-treated patients in clinical studies. In other
XIAFLEX®-treated patients (9 of 1044; 0.9%), a combination of
penile ecchymoses or hematoma, sudden penile detumescence, and/or a
penile "popping" sound or sensation was reported, and in these
cases, a diagnosis of corporal rupture cannot be excluded. Severe
penile hematoma was also reported as an adverse reaction in 39 of
1044 (3.7%) XIAFLEX®-treated patients.
Signs or symptoms
that may reflect serious penile injury should be promptly evaluated
to assess for corporal rupture or severe penile hematoma which may
require surgical intervention.
Because of the risks of
corporal rupture or other serious penile injury, XIAFLEX® is
available for the treatment of Peyronie's disease only through a
restricted program under a Risk Evaluation and Mitigation Strategy
(REMS) called the XIAFLEX® REMS Program.
- XIAFLEX® is contraindicated in the treatment of Peyronie's
plaques that involve the penile urethra due to potential risk to
this structure and in patients with a history of hypersensitivity
to XIAFLEX® or to collagenase used in any other therapeutic
application or application method
- Injection of XIAFLEX® into collagen-containing structures such
as the corpora cavernosa of the penis may result in damage to those
structures and possible injury such as corporal rupture (penile
fracture). Therefore, XIAFLEX® should be injected only into the
Peyronie's plaque and care should be taken to avoid injecting into
the urethra, nerves, blood vessels, corpora cavernosa or other
collagen-containing structures of the penis
- In the double-blind, placebo-controlled portions of the
clinical trials in Peyronie's disease, a greater proportion of
XIAFLEX®-treated patients (4%) compared to placebo-treated patients
(1%) had localized pruritus after up to 4 treatment cycles
(involving up to 8 XIAFLEX® injection procedures). The incidence of
XIAFLEX®-associated pruritus was similar after each injection
regardless of the number of injections administered
- Because XIAFLEX® contains foreign proteins, severe allergic
reactions to XIAFLEX® can occur. Anaphylaxis was reported in a
post-marketing clinical trial in one patient who had previous
exposure to XIAFLEX® for the treatment of Dupuytren's contracture.
Healthcare providers should be prepared to address severe allergic
reactions following XIAFLEX® injections. The safety of more than
one treatment course of XIAFLEX® is not known
- In the XIAFLEX® controlled trials in Peyronie's disease, 65.5%
of XIAFLEX®-treated patients developed penile hematoma, and 14.5%
developed penile ecchymosis. Patients with abnormal coagulation
(except for patients taking low-dose aspirin, eg, up to 150 mg per
day) were excluded from participating in these studies. Therefore,
the efficacy and safety of XIAFLEX® in patients receiving
anticoagulant medications (other than low-dose aspirin, eg, up to
150 mg per day) within 7 days prior to XIAFLEX® administration is
not known. In addition, it is recommended to avoid use of XIAFLEX®
in patients with coagulation disorders, including patients
receiving concomitant anticoagulants (except for low-dose
aspirin)
- In the XIAFLEX® clinical trials for Peyronie's disease, the
most frequently reported adverse drug reactions (≥25%) and at an
incidence greater than placebo included: penile hematoma, penile
swelling, and penile pain
Please see the full Prescribing Information,
including Boxed Warning and Medication Guide, available
at www.XIAFLEX.com.
About Endo International plc
Endo International plc
(NASDAQ / TSX: ENDP) is a global specialty pharmaceutical company
focused on improving patients' lives while creating shareholder
value. Endo develops, manufactures, markets and distributes quality
branded and generic pharmaceutical products as well as
over-the-counter medications though its operating companies. Endo
has global headquarters in Dublin,
Ireland, and U.S. headquarters in Malvern, PA. Learn more at www.endo.com.
Cautionary Note Regarding Forward-Looking
Statements
This press release contains "forward-looking
statements," including, but not limited to, the statements by Drs.
Hall, Sadick and Goldman. These statements are based on current
expectations of future events. If underlying assumptions prove
inaccurate or unknown risks or uncertainties materialize, actual
results could vary materially from Endo's expectations
and projections. Risks and uncertainties include, among other
things, general industry and market conditions; technological
advances and patents attained by competitors; challenges inherent
in the research and development and regulatory processes;
challenges related to product marketing, such as the
unpredictability of market acceptance for new products and/or the
acceptance of new indications for such products; inconsistency of
treatment results among patients; potential difficulties in
manufacturing; general economic conditions; and governmental laws
and regulations affecting domestic and foreign
operations. Endo expressly disclaims any intent or
obligation to update these forward-looking statements except as
required by law. Additional information concerning these and other
risk factors can be found in Endo's periodic reports
filed with the U.S. Securities and Exchange
Commission and in Canada on the System for
Electronic Data Analysis and Retrieval ("SEDAR"), including current
reports on Form 8-K, quarterly reports on Form 10-Q and annual
reports on Form 10-K. Additional information
about Endo is available on the World Wide Web
at www.endo.com or you can contact the Endo Investor
Relations department by calling (484) 216-0000.
[i] Avram, Cellulite: a review of its physiology and treatment,
Journal of Cosmetic Laser Therapy 2004; 6: 181–185.
[ii] Khan MH et al. Treatment of cellulite: Part I.
Pathophysiology. J Am Acad Dermatol. 2010 Mar;62(3):361-70.
[iii] Querleux, Anatomy and physiology of subcutaneous adipose
tissue by in vivo MRI and spectroscopy: Relationship with sex and
presence of cellulite, Skin Research and Technology; 8:
118-124.
[iv] Wanner M et al. An evidence-based assessment of treatments for
cellulite. J Drugs Dermatol. 2008 Apr;7(4):341-5.
Photo - http://photos.prnewswire.com/prnh/20161116/440530
Photo - http://photos.prnewswire.com/prnh/20161116/440497
To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/endo-announces-positive-data-from-phase-2b-study-of-collagenase-clostridium-histolyticum-cch-in-patients-with-cellulite-300364763.html
SOURCE Endo International plc