AroCell AB: AroCell signs distribution agreement with Eagle Biosciences in the United States
October 06 2016 - 5:45AM

Uppsala October 6,
2016
PRESS RELEASE
AroCell signs distribution
agreement with Eagle Biosciences in the United States
AroCell AB (publ) announced today
that a distribution agreement has been signed with the New
England-based company Eagle Biosciences, for distribution of the
AroCell TK 210 ELISA test in North America.
The TK 210 ELISA test measures TK1 protein levels
for valuable information about the speed of cell turnover. As
tumors have high cell turnover, the test may provide valuable
information for prognosis and optimization of treatment
strategy.
"We are delighted to partner with Eagle
Biosciences for the distribution of the TK 210 ELISA test", states
Jan Stålemark, AroCell CEO. "Eagle Biosciences is a leading
provider of ELISA assay kits in North America, and has primary
focus on providing innovative products that aid medical research
and clinical laboratories. This highly concurs with our strategy to
make the TK 210 ELISA test widely available for investigational use
in the US, in order to facilitate use of the product in cancer
research institutions as well as within the pharmaceutical
industry".
The TK 210 ELISA test will be commercially
available immediately through Eagle Biosciences for investigational
use only in the United States.
For additional
information:
Jan Stålemark, CEO
AroCell AB (publ)
Tel: +46(0)706 926206
info@arocell.com
www.arocell.com
About AroCell
AroCell AB (AROC) is a Swedish
company that develops standardized modern blood tests to support
the prognosis and follow up of cancer patients. AroCell's new
technology is based on patented methods to measure TK1 protein
levels, which provide valuable information about the speed of cell
turnover. A tumor has high cell turnover (speed of cell division
and cell death) and as a result TK1 can be detected in the blood
with a simple laboratory test, called TK 210 ELISA. The test
provides valuable clinical information for prognosis and
optimization of treatment strategy. The test may also be used for
monitoring disease relapse. For more information, please see
www.arocell.com. This information is information
that AroCell is obliged to make public pursuant to the EU Market
Abuse Regulation. The information was submitted for publication,
through Jan Stålemark, at 11.45 CET on 6th October
2016. Redeye is AroCell:s Certified Adviser
This
announcement is distributed by Nasdaq Corporate Solutions on behalf
of Nasdaq Corporate Solutions clients.
The issuer of this announcement warrants that they are solely
responsible for the content, accuracy and originality of the
information contained therein.
Source: AroCell AB via Globenewswire
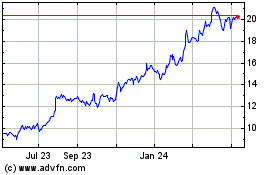
Archrock (NYSE:AROC)
Historical Stock Chart
From Mar 2024 to Apr 2024
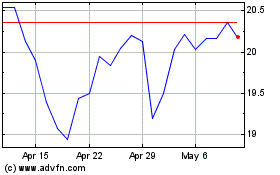
Archrock (NYSE:AROC)
Historical Stock Chart
From Apr 2023 to Apr 2024