As filed with the Securities and Exchange Commission on June 22, 2022
Registration No. 333-239661
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
POST-EFFECTIVE AMENDMENT No. 3
to
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
PANBELA THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
|
Delaware
(State or other jurisdiction of
incorporation or organization)
|
|
2834
(Primary Standard Industrial
Classification Code Number)
|
|
88-2805017
(I.R.S. Employer
Identification No.)
|
712 Vista Blvd, Suite 305
Waconia, Minnesota 55387
(952) 479-1196
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
Susan Horvath
Chief Financial Officer
712 Vista Blvd, Suite 305
Waconia, Minnesota 55387
(952) 479-1196
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Copies to:
W. Morgan Burns & Joshua L. Colburn
Faegre Drinker Biddle & Reath LLP
90 South Seventh Street
2200 Wells Fargo Center
Minneapolis, Minnesota 55402-3901
Telephone: (612) 766-7000
Approximate date of commencement of proposed sale to the public: From time to time after the effective date of this Registration Statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, as amended, check the following box. ☑
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer ☐
|
Accelerated filer ☐
|
|
Non-accelerated filer ☑
|
Smaller reporting company ☑
|
| |
Emerging growth company ☐
|
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided to Section 7(a)(2)(B) of the Securities Act. ☐
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the registration statement shall become effective on such date as the Commission, acting pursuant to said Section 8(a), may determine.
Explanatory Note
On July 2, 2020, Panbela Research, Inc. (formerly known as Panbela Therapeutics, Inc., the “Predecessor”) filed a registration statement with the Securities and Exchange Commission (the “SEC”) on Form S-1 (Registration No. 333-239661) (as amended, the “Registration Statement”). The Registration Statement was initially declared effective by the SEC on August 27, 2020, was amended by post-effective amendments filed with, and declared effective by, the SEC on March 26, 2021 and March 28, 2022.
Effective June 15, 2022, Panbela Research completed a corporate reorganization (the “Holding Company Reorganization”) pursuant to which the Predecessor became a direct, wholly-owned subsidiary of a new public holding company, Panbela Therapeutics, Inc. (formerly known as Canary Merger Holdings, Inc., “Panbela”). The Holding Company Reorganization was effected by a merger conducted pursuant to 251(g) without a vote of the stockholders of the constituent corporations. The merger resulting in the Holding Company Reorganization was completed pursuant to the terms of an agreement and plan of merger, dated as of February 21, 2022 (the “Merger Agreement”), by and among Panbela, Cancer Prevention Pharmaceuticals, Inc. (“Cancer Prevention”) and the Predecessor, among others. Panbela became a successor issuer to the Predecessor by operation of Rule 12g-3(a) promulgated under the Securities Exchange Act of 1934, as amended.
Panbela is filing this Post-Effective Amendment No. 3 (this “Amendment”) to the Registration Statement pursuant to Rule 414 promulgated under the Securities Act of 1933, as amended (the “Securities Act”), to update the Registration Statement as a result of the Holding Company Reorganization. In accordance with Rule 414(d) under the Securities Act, except as modified by this Amendment, Panbela, as successor issuer to the Predecessor pursuant to Rule 12g-3, hereby expressly adopts the Registration Statement as its own registration statement for all purposes of the Securities Act and the Exchange Act, as updated by subsequent amendment and other filings under the Exchange Act.
The Registration Statement, as amended by this Post-Effective Amendment No. 3 (this “Amendment”), pertains solely to the issuance by Panbela of the remaining 2,306,516 shares of common stock, $0.001 par value per share, underlying warrants (collectively, the “Investor Warrants”) previously issued by Panbela Research to investors in a public offering and 127,273 shares of common stock underlying warrants previously issued to the underwriters (the “Underwriter Warrants” and, together with the Investor Warrants, the “Warrants”). The shares of common stock issuable upon exercise of the Warrants were initially registered on the Registration Statement.
For the convenience of the reader, this Amendment sets forth the Registration Statement in its entirety, as amended. Panbela is filing this Amendment to update the financial statements and other information, including pursuant to the undertakings in Item 17 of the Registration Statement, and to update and supplement the information contained in the Registration Statement and the Prospectus contained therein.
No additional securities are being registered under this Amendment. All applicable registration fees were paid at the time of the original filing of the Registration Statement.
The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell securities, and we are not soliciting offers to buy these securities, in any state where the offer or sale is not permitted.
|
PRELIMINARY PROSPECTUS
|
SUBJECT TO COMPLETION
|
DATED JUNE 22, 2022
|
2,433,789 Shares of Common Stock Underlying Previously Issued Warrants

This prospectus relates to the offer and sale by Panbela Therapeutics, Inc. of up to (i) 2,306,516 shares of our common stock underlying warrants previously issued by us to investors that are issuable at a price of $4.54 per share and (ii) 127,345 shares of our common stock underlying warrants previously issued by us to the underwriter in a public offering that are issuable at a price of $4.357 per share, in each case from time to time upon exercise of such warrants. We are not selling any shares of our common stock in this offering other than pursuant to the exercise of outstanding warrants. Upon exercise of the warrants we will receive proceeds from the exercise of such warrants if exercised for cash and not on a cashless basis.
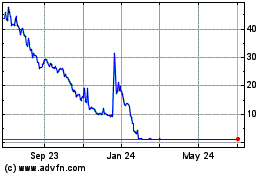
Our common stock is listed on the Nasdaq Capital Market under the symbol “PBLA.” On June 16, 2022, the last reported sale price of our common stock on the Nasdaq Capital Market was $1.33 per share. None of the warrants are listed on a national securities exchange.
Investing in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described under the heading “Risk Factors” beginning on page 9 of this prospectus, and under similar headings in any amendments or supplements to this prospectus.
See “Plan of Distribution” beginning on page 40, for more information regarding the offering. No underwriter or person has been engaged to facilitate the sale of Shares in this offering. All costs associated with the registration were borne by us.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2022
TABLE OF CONTENTS
Page
|
Prospectus Summary
|
1
|
|
The Offering
|
7
|
|
THE INITIAL CLOSING
|
8
|
|
Risk Factors
|
9
|
|
CAUTIONARY Note Regarding Forward-Looking Statements
|
20
|
|
Use of Proceeds
|
21
|
|
MARKET INFORMATION
|
22
|
|
DILUTION
|
26
|
|
Dividend Policy
|
27
|
|
Management’s Discussion and Analysis of Financial Condition and Results of Operations
|
28
|
|
FINANCIAL STATEMENTS
|
28
|
|
Business
|
29
|
|
Management
|
52
|
|
Security Ownership of Certain Beneficial Owners AND Management
|
62
|
|
DESCRIPTION OF Securities
|
63
|
|
SHARES ELIGIBLE FOR FUTURE SALE
|
67
|
|
PLAN OF DISTRIBUTION
|
68
|
|
Legal Matters
|
68
|
|
Experts
|
68
|
|
Where You Can Find More Information
|
68
|
|
INCORPORATION OF DOCUMENTS BY REFERENCE
|
69
|
|
FINANCIAL STATEMENTS
|
|
You should rely only on the information contained in this prospectus. We have not authorized anyone to provide you with any information other than that contained in this prospectus. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. This prospectus may only be used where it is legal to offer and sell our securities. The information in this prospectus is accurate only as of the date of this prospectus, regardless of the time of delivery of this prospectus or any sale of our securities. Our business, financial condition, results of operations and prospects may have changed since that date. We are not making an offer of these securities in any jurisdiction where the offer is not permitted.
For investors outside the United States: We have not done anything that would permit this offering or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. Persons outside the United States must inform themselves about, and observe any restrictions relating to, the offering of securities and the distribution of this prospectus outside the United States.
This prospectus includes statistical and other industry and market data that we obtained from industry publications and research, surveys and studies conducted by third parties. Industry publications and third-party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. We believe that the data obtained from these industry publications and third-party research, surveys and studies are reliable. We are ultimately responsible for all disclosure included in this prospectus.
You should rely only on the information contained in this prospectus, as supplemented and amended. We have not authorized anyone to provide you with information that is different. This prospectus may only be used where it is legal to sell these securities. The information in this prospectus may only be accurate on the date of this prospectus.
We urge you to read carefully this prospectus, as supplemented and amended, before deciding whether to invest in any of the securities being offered.
PROSPECTUS SUMMARY
This summary highlights information contained elsewhere in this prospectus and does not contain all of the information that you should consider in making your investment decision. Before investing in our securities, you should carefully read this entire prospectus, including our financial statements and the related notes and the information set forth under the headings “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in each case included elsewhere in this prospectus. Unless otherwise stated or the context requires otherwise, references in this prospectus to “Panbela”, the “Company”, “we”, “us”, “our” and similar references refer to Panbela Therapeutics, Inc. and its subsidiaries.
Business Overview
Panbela is a clinical stage biopharmaceutical company developing disruptive therapeutics for the treatment of patients with urgent unmet medical needs. The company’s lead assets are SBP-101 and Flynpovi. SBP-101, is a proprietary polyamine analogue designed to induce polyamine metabolic inhibition (“PMI”), a metabolic pathway of critical importance in multiple tumor types. Many tumors require greatly elevated levels of polyamines to support their growth and survival. SBP-101 has demonstrated encouraging activity against metastatic disease in a clinical trial of patients with pancreatic cancer. The efficacy and safety results demonstrated in our completed phase I clinical trial of SBP-101 in combination with gemcitabine and nab-paclitaxel in the first line treatment of metastatic pancreatic cancer provides support for the current randomized, double-blind, placebo controlled phase II/III study of SBP-101 in combination with gemcitabine and nab-paclitaxel in patients previously untreated for metastatic pancreatic cancer. We believe that SBP-101, if successfully developed, may represent a novel approach that effectively treats patients with pancreatic cancer and could become a dominant product in that market. Only three first-line treatment combinations, a single maintenance treatment for a subset (3-7%) of patients, and one second-line drug have been approved by the FDA for pancreatic cancer in the last 25 years.
On June 15, 2022 Panbela acquired Cancer Prevention Pharmaceuticals, Inc. (CPP), which added the company’s second lead asset, an investigational new drug product, Flynpovi which is a combination of the polyamine synthesis inhibitor eflornithine and the non-steroidal anti-inflammatory drug sulindac. Eflornithine is an enzyme-activated, irreversible inhibitor of the enzyme ornithine decarboxylase, the first and rate-limiting enzyme in the biosynthesis of polyamines. Sulindac facilitates the export and catabolism of polyamines. We believe the investigational drug is unique in that it is designed to treat the risk factors (e.g., polyps) that are hypothesized to lead to Familial Adenomatous Polyposis (FAP) surgeries and colon cancer and therefore may have the ability to prevent various types of colon cancer. Flynpovi has received orphan drug designation status for FAP in the United States and Europe. There are no currently approved pharmaceutical therapies for FAP.
Both of the principal active ingredients in our investigational drug candidate Flynpovi (eflornithine and sulindac) have been previously approved individually but not in combination for other uses by the U.S. Food and Drug Administration (“FDA”) and have shown limited side effects at the dosages utilized in FAP studies. Eflornithine has never been approved in an oral form, is not on the market in any systemic dosage form, and is not available in any generic form. The combination of eflornithine and sulindac is delivered in an oral form, to which we have exclusive license rights to commercialize from the Arizona Board of Regents of the University of Arizona (the “University of Arizona”). This combination showed promising results in a National Cancer Institute (“NCI”) supported randomized, placebo-controlled Phase II/III clinical trial to prevent recurrent colon adenomas, particularly high-risk pre-cancerous polyps. These results led to a Phase III program in FAP, and a Phase III program to study colon cancer risk reduction in partnership with the Southwest Oncology Group (SWOG) and the NCI.
Additional programs are evaluating eflornithine as a single agent tablet (CPP-1X) or high dose powder sachet (CPP-1X-S) for several indications including prevention of gastric cancer, treatment of neuroblastoma and recent onset Type 1 diabetes. Preclinical studies as well as Phase 1 or Phase 2 investigator-initiated trials suggest that eflornithine treatment is well tolerated and has potential activity.
Holding Company Reorganization
Effective June 15, 2022, Panbela became a successor issuer to Panbela Research, Inc. (formerly known as Panbela Therapeutics, Inc., the “Predecessor”) pursuant to a holding company reorganization pursuant to which the Predecessor became a direct, wholly-owned subsidiary of Panbela. Panbela became a successor issuer to the Predecessor by operation of Rule 12g-3(a) promulgated under the Securities Exchange Act of 1934, as amended the (“Exchange Act”).
Cancer Prevention Acquisition
On June 15, 2022, Panbela acquired Cancer Prevention Pharmaceuticals, Inc. (“Cancer Prevention”), a private clinical stage company developing therapeutics to reduce the risk and recurrence of cancer and rare diseases,, via merger for consideration consisting of (a) 6,587,576 shares of common stock, (b) 731,957 shares of common stock that remained subject to a holdback escrow (as defined in the Merger Agreement), (c) replacement options to purchase up to 1,596,754 shares of common stock at a weighted average purchase price of $0.35 per share, and (d) replacement warrants to purchase up to 338,060 shares of common stock at a weighted average purchase price of $4.145 per share, and post-closing contingent payments up to a maximum of $60 million, subject to satisfaction of milestones.
Clinical Trials
SBP-101
In August 2015, the FDA accepted our Investigational New Drug (“IND”) application for our SBP-101 product candidate. We have completed an initial clinical trial of SBP-101 in patients with previously treated locally advanced or metastatic pancreatic cancer. This was a Phase I, first-in-human, dose-escalation, safety study. Between January 2016 and September 2017, we enrolled twenty-nine patients into six cohorts, or groups, in the dose-escalation phase of our Phase I trial. Twenty-four of the patients received at least two prior chemotherapy regimens. No drug-related serious adverse events occurred during the first four cohorts. In cohort five, serious adverse events (klebsiella sepsis with metabolic acidosis in one patient, renal and hepatic toxicity in one patient, and mesenteric vein thrombosis with metabolic acidosis in one patient) were observed in three of the ten patients, two of whom exhibited progressive disease at the end of their first cycle of treatment and were determined by the Data Safety Monitoring Board (“DSMB”) to be Disease Limiting Toxicities (“DLTs”). Consistent with the study protocol, the DSMB recommended continuation of the study by expansion of cohort 4, one level below that at which DLTs were observed. Four patients were enrolled in this expansion cohort.
In addition to being evaluated for safety, 23 of the 29 patients were evaluable for preliminary signals of efficacy prior to or at the eight-week conclusion of their first cycle of treatment using the RECIST, the current standard for evaluating changes in the size of tumors. Eight of the 23 patients (35%) had Stable Disease (“SD”) and 15 of 24 (65%) had Progressive Disease (“PD”). It should be noted that of the 15 patients with PD, six came from cohorts one and two and are considered to have received less than potentially therapeutic doses of SBP-101.
By cohort, stable disease occurred in two patients in cohort 3, two patients in cohort 4 and four patients in cohort 5. The best response outcomes and best median survival were observed in the group of patients who received total cumulative doses of approximately 6 mg/kg (cohort three). Two of four patients (50%) showed SD at week eight. Median survival in this group was 5.9 months, with two patients surviving 8 and 10 months, respectively. By total cumulative dose received, five of twelve patients (42%) who received total cumulative doses between 2.5 mg/kg and 8.0 mg/kg had reductions in the CA19-9 levels, as measured at least once after the baseline assessment. Nine of these patients (67%) exceeded three months of Overall Survival (“OS”), three patients (25%) exceeded nine months of OS and two patients (17%) exceeded one year of OS and were still alive at the end of the study.
We completed enrollment of patients in our second clinical trial in December 2020. This second clinical trial was a Phase Ia/Ib study of the safety, efficacy and pharmacokinetics of SBP-101 administered in combination with two standard-of-care chemotherapy agents, gemcitabine and nab-paclitaxel. The trial included six study sites; four in Australia and two in the United States. In the Phase Ia portion of this trial, we completed enrollment during the first quarter of 2020 consisting of four cohorts with increased dosage levels of SBP-101 administered in the second and third cohorts; the fourth cohort evaluated an alternate dosing schedule. A total of 25 subjects were enrolled in four cohorts of Phase Ia. The demonstration of adequate safety in Phase Ia allowed us to immediately begin enrollment in February 2020 in the Phase Ib exploration of efficacy. By December 2020 an additional 25 subjects in Phase Ib, using the recommended dosage level and schedule determined in Phase Ia, were enrolled.
After Phase Ib enrollment was completed, some patients in the trial were noted to have complaints of serious visual adverse effects. Visual changes were not seen in the SBP-101 monotherapy study. We consulted with our DSMB and withheld the administration of SBP-101 while all other trial activities continued. In February of 2021, we also conferred with the FDA regarding our plan to withhold dosing of SBP-101. This constituted a “partial clinical hold.” In April of 2021, the FDA lifted the partial clinical hold. The Company agreed with the FDA to include in the design of all future studies the exclusion of patients with a history of retinopathy or risk of retinal detachment and scheduled ophthalmologic monitoring for all patients.
Updated, but still not final results, were presented in a poster at the American Society of Clinical Oncology - GI conference (“ASCO-GI”) in January 2022. Best response in evaluable subjects (cohorts 4 and Ib N=29) was a Complete Response (“CR”) in 1 (3%), Partial Response (“PR”) in 13 (45%), SD in 10 (34%) and PD in 5 (17%). One subject did not have post baseline scans with RECIST tumor assessments. Median Progression Free Survival (“PFS”) , now final at 6.5 months may have been negatively impacted by drug dosing interruptions to evaluate potential toxicity. Median overall survival in Cohort 4 + Phase Ib was 12.0 months when data was presented in January 2022 and is now final at 14.6 months. Two patients from cohort 2 have demonstrated long term survival. One at 30.3 months (final data) and one at 33.0 months and still alive. Seven subjects are still alive at this time, one from cohort 2 and six from cohort 4 plus Ib.
The Company announced that the ASPIRE trial, a randomized, double-blind, placebo-controlled study of SBP-101 with Gemcitabine and Nab-Paclitaxel versus Gemcitabine, Nab-Paclitaxel and placebo, was initiated in January of 2022. The trial is in patients with first-line metastatic pancreatic ductal adenocarcinoma. The trial is designed as a Phase II/III randomized trial, with a primary endpoint of overall survival. The design includes a phase II portion for which a futility analysis after 104 progression free survival events will occur. If the futility analysis is favorable, the trial will be expanded to the phase III portion, and may serve for registration. We are intending to conduct the ASPIRE trial at leading cancer centers in the United States, Europe and the Asia-Pacific Region. It is expected that there will be approximately 60 sites and we anticipate enrollment for the phase II portion to be completed in approximately 12 months after the first subject is enrolled.
If we can successfully complete all FDA recommended clinical studies, we intend to seek marketing authorization from the FDA, the European Medicines Agency (“EMA”) (European Union), Ministry of Health and Welfare (Japan) and TGA (Australia). The submission fees may be waived when SBP-101 has been designated an orphan drug in each geographic region.
Data presented at the American Association for Cancer Research (AACR) in April 2022, demonstrated in an in vitro study evaluating cancer cell lines, SBP-101 was toxic in ovarian cancer cell lines with an average IC50 of ~1.5 μM. Efficacy of SBP-101 was further assessed in the VDID8+murine ovarian cancer model (ID8+C57Bl/6 ovarian cells overexpressing both VEGF and Defensin). Mice were treated with SBP-101 at either 24 mg/kg or 6 mg/kg alternating MWF. Both doses of SBP-101 produced a statistically significant prolongation of survival (24mg/kg p=.0049, 6 mg/kg p=.0042). There was no significant difference in response between the two SBP-101 doses. The prolonged survival was correlated with a delay in the production of ascites, the indication of tumor burden in this model. Given this data, the Company intends to proceed with a clinical development program in ovarian cancer by year-end.
FLYNPOVI
In a phase III study, the efficacy and safety of the combination of eflornithine and sulindac, as compared with either drug alone, in adults with familial adenomatous polyposis was conducted. The patients were randomly assigned in a 1:1:1 ratio to receive eflornithine, sulindac, or both once daily for up to 48 months. The primary end point, assessed in a time-to-event analysis, was disease progression, defined as a composite of major surgery, endoscopic excision of advanced adenomas, diagnosis of high-grade dysplasia in the rectum or pouch, or progression of duodenal disease. A total of 171 patients underwent randomization. Disease progression occurred in 18 of 56 patients (32%) in the eflornithine–sulindac group, 22 of 58 (38%) in the sulindac group, and 23 of 57 (40%) in the eflornithine group, with a hazard ratio of 0.71 (95% confidence interval [CI], 0.39 to 1.32) for eflornithine–sulindac as compared with sulindac (P = 0.29) and 0.66 (95% CI, 0.36 to 1.23) for eflornithine–sulindac as compared with eflornithine. Adverse and serious adverse events were similar across the treatment groups. In a post-hoc analysis, none of the patients in the combination arm progressed to a need for lower gastrointestinal (LGI) surgery for up to 48 months compared with 7 (13.2%) and 8 (15.7%) patients in the sulindac and eflornithine arms. These data corresponded to risk reductions for the need for LGI surgery approaching 100% between combination and either monotherapy with HR = 0.00 (95% CI, 0.00–0.48; p = 0.005) for combination versus sulindac and HR = 0.00 (95% CI, 0.00–0.44; p = 0.003) for combination versus eflornithine. Given the statistical significance of the LGI group, a new drug application (NDA) was filed with the FDA. As the study failed to meet the primary endpoint, and the NDA was based on the results of an exploratory analysis, a complete response letter was issued. To address this deficiency concern, the Company must submit the results of one or more adequate and well-controlled clinical trials which demonstrate an effect on a clinical endpoint. As the result of an existing North American license agreement, the FAP registration trial is fully funded and is scheduled to begin in the first-half of 2023.
CPP-1X-S/CPP-1X
Additionally, there are trials evaluating CPP-1X-S in relapsed refractory neuroblastoma supported by the childrens’ oncology group (COG) and NCI (ongoing) and STK11 mutation patients with non-small cell lung cancer scheduled to begin this year. For CPP-1X-T, a phase II trial in Type I onset diabetes is scheduled to begin this year in collaboration with Indiana University and the Juvenile Diabetes Research Foundation (JDRF).
Recent Developments
As previously announced, Panbela and certain of its subsidiaries successfully completed a merger and reorganization of Cancer Prevention Pharmaceuticals, Inc. (“CPP”), a private clinical stage company developing therapeutics to reduce the risk and recurrence of cancer and rare diseases, for a combination of stock and future milestone payments. The combined entity will have an expanded pipeline; areas of initial focus include familial adenomatous polyposis (FAP), first-line metastatic pancreatic cancer, neoadjuvant pancreatic cancer, colorectal cancer prevention and ovarian cancer. The combined development programs will have a steady cadence of catalysts with programs ranging from pre-clinical to registration studies.
Through December 31, 2021, we had:
| |
●
|
secured an orphan drug designation from the FDA;
|
| |
●
|
submitted and received acceptance from the FDA for an IND application;
|
| |
●
|
received acceptance of a Clinical Trial Notification from the Australian Therapeutic Goods Administration;
|
| |
●
|
completed a Phase Ia monotherapy safety study of SBP-101in the treatment of patients with metastatic pancreatic ductal adenocarcinoma;
|
| |
●
|
received “Fast Track” designation from the FDA for SBP-101 for metastatic pancreatic cancer;
|
| |
●
|
completed enrollment and released interim results in our second trial a Phase Ia /Ib clinical study of SBP-101, a first-line study with SBP-101 given in combination with a current standard of care in patients with pancreatic ductal adenocarcinoma who were previously untreated for metastatic disease; a total of 50 subjects were enrolled in this study, 25 in the Phase Ia and 25 in the Phase Ib or expansion phase;
|
| |
●
|
secured a two year research agreement with Johns Hopkins School of Medicine led by Professor Robert Casero an internationally recognized researcher in polyamine biology;
|
| |
●
|
completed process improvement measures expected to be scalable for commercial use and received issue notification for a patent covering this new shorter synthesis of SBP-101;
|
| |
●
|
initiated a randomized, double-blind, placebo controlled study with SBP-101 given in combination with gemcitabine and nab-paclitaxel in patients with pancreatic ductal adenocarcinoma who are previously untreated for metastatic disease;
|
| |
●
|
Completed preclinical evaluation of SBP-101 for use as neoadjuvant therapy in resectable pancreatic cancer prior to surgery; and
|
| |
●
|
Obtained early, preclinical, indication of tumor growth inhibition activity in ovarian cancer.
|
Risks Associated with Our Business
Our business is subject to many significant risks, as more fully described in the section titled “Risk Factors” immediately following this prospectus summary. You should read and carefully consider these risks, together with the risks set forth under the section titled “Risk Factors” and all of the other information in this prospectus, including the financial statements and the related notes included elsewhere in this prospectus, before deciding whether to invest in our securities. If any of the risks discussed in this prospectus actually occur, our business, financial condition or operating results could be materially and adversely affected. In particular, our risks include, but are not limited to, the following:
| |
●
|
our lack of diversification and the corresponding risk of an investment in our Company;
|
| |
●
|
potential deterioration of our financial condition and results due to failure to diversify;
|
| |
●
|
our ability to successfully complete acquisitions and integrate operations for new product candidates;
|
| |
●
|
our ability to obtain additional capital, on acceptable terms or at all, required to implement our business plan;
|
| |
●
|
final results of our Phase I clinical trial;
|
| |
●
|
progress and success of our randomized Phase II/III clinical trial;
|
| |
●
|
our ability to demonstrate safety and effectiveness of our product candidates;
|
| |
●
|
our ability to obtain regulatory approvals for our product candidate in the United States, the European Union, or other international markets;
|
| |
●
|
the market acceptance and future sales of our product candidates;
|
| |
●
|
the cost and delays in product development that may result from changes in regulatory oversight applicable to our product candidates;
|
| |
●
|
the rate of progress in establishing reimbursement arrangements with third-party payors;
|
| |
●
|
the effect of competing technological and market developments;
|
| |
●
|
the costs involved in filing and prosecuting patent applications and enforcing or defending patent claims; and
|
| |
●
|
other risk factors included under the caption “Risk Factors” starting on page 9 of this prospectus.
|
Implications of Being a Smaller Reporting Company
We are a “smaller reporting company” as defined in Rule 12b-2 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and have elected to take advantage of certain of the scaled disclosure available to smaller reporting companies.
Corporate History
Panbela Therapeutics, Inc., formerly known as Sun BioPharma, Inc., was originally incorporated under the laws of the State of Delaware in September 2011. In 2015, we became a public company by completing a reverse merger transaction (the “Merger”) with a wholly owned subsidiary of Cimarron Medical, Inc., a public company organized under the laws of the State of Utah. Upon completion of the Merger and other separate but contemporaneous transactions by certain of our stockholders, our stockholders collectively owned approximately 99.0% of the post-Merger public company, which was renamed “Sun BioPharma, Inc.” In 2016, we reincorporated under the laws of the State of Delaware via a merger with our operating subsidiary, resulting in our current corporate form. The Company changed its name to Panbela Therapeutics, Inc. on December 2, 2020. On June 15, 2022, Panbela became a successor issuer pursuant to a holding company reorganization via merger by operation of Rule 12g-3(a) promulgated under the Exchange Act.
Corporate Information
Our corporate mailing address is 712 Vista Blvd, #305, Waconia, MN 55387. Our telephone number is (952) 479-1196, and our website is www.sunbiopharma.com. The information on our website is not part of this prospectus. We have included our website address as a factual reference and do not intend it to be an active link to our website. The information contained in or connected to our website is not incorporated by reference into, and should not be considered part of, this prospectus.
Trade names, trademarks, and service marks of other companies appearing in this prospectus are the property of the respective holders.
THE OFFERING
|
Common stock offered by us
|
Up to 2,433,789 shares of our common stock, consisting of (i) 2,306,516 shares issuable upon exercise of outstanding warrants with an exercise price of $4.54 per share and (ii) 127,273 shares issuable upon exercise of outstanding warrants with an exercise price of $4.537 per share. Each such warrant is exercisable at any time for the purchase of one share of our common stock and is scheduled to expire on September 1, 2025.
|
| |
|
|
Common stock outstanding as of the date of this prospectus
|
20,774,045 shares
|
| |
|
|
Common stock to be outstanding immediately after completion of this offering
|
23,207,834 shares (assuming full exercise of the warrants)
|
| |
|
|
Use of proceeds
|
We will receive approximately $10.9 million in proceeds if all of the currently outstanding warrants issued in the public offering are exercised for cash. We intend to use the net proceeds from this offering for the continued clinical development of our initial product candidate SBP-101, and for working capital and other general corporate purposes. See “Use of Proceeds” on page 20.
|
| |
|
|
Risk Factors
|
You should read the “Risk Factors” section of this prospectus beginning on page 9 for a discussion of factors to consider carefully before deciding to invest in our securities.
|
| |
|
|
Nasdaq Capital Market trading symbol
|
“PBLA”
|
| |
|
The number of shares of our common stock outstanding before and after this offering is based on 20,774,045 shares of our common stock outstanding as of June 16, 2022, and excludes:
| |
●
|
4,040,890 shares of common stock issuable upon the exercise of outstanding stock options at a weighted average exercise price of $3.62 per share;
|
| |
●
|
2,019,776 additional shares of common stock reserved and available for future issuances under our equity plans; and
|
| |
●
|
3,013,772 shares of common stock issuable upon exercise of stock purchase warrants not related to this offering at a weighted average exercise price of $4.58 per share.
|
Unless otherwise indicated, all information in this prospectus assumes no exercise of the outstanding options or warrants not relating to this offering.
THE INITIAL CLOSING
On September 1, 2020, we completed an initial closing (the “Initial Closing”) of the offering whereby we issued and sold in an underwritten public offering 2,545,454 shares of our common stock and 2,545,454 five-year warrants to purchase one share of common stock for an exercise price of $4.54 per share (collectively, the “Investor Warrants”). The securities were issued to the public at a combined price of $4.125 per share and warrant and were offered by our Company pursuant to the registration statement of which this prospectus forms a part (File No. 333-239661), which was declared effective on August 28, 2020. The aggregate net proceeds received by us from the Initial Closing were approximately $9.31 million and were used for the continued clinical development of our initial product candidate SBP-101, the repayment of approximately $0.9 million of outstanding indebtedness, and for working capital and other general corporate purposes. We also issued to the underwriter in the public offering five-year warrants to purchase 127,345 shares of common stock for an exercise price of $4.537 per share (the “Underwriter Warrant” and, together with the Investor Warrants, the “Warrants”).
As of June 16, 2022, 2,433,789 shares of our common stock remained issuable upon exercise of the Warrants.
RISK FACTORS
Any investment in our securities involves a high degree of risk. Investors should carefully consider the risks described below and all of the information contained in this prospectus before deciding whether to purchase our securities. Our business, financial condition or results of operations could be materially adversely affected by these risks if any of them actually occur. This prospectus also contains forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including the risks we face as described below and elsewhere in this prospectus.
Risks Related to Our Business and Financial Position
We are a pre-revenue company with a history of negative operating cash flow.
We have experienced negative cash flows for our operating activities since inception, primarily due to the investments required to commercialize our primary drug candidates, SBP-101 and eflornithine. Our financing cash flows historically have been positive due to proceeds from the sale of equity securities and promissory notes issuances. Our net cash used in operating activities was $6.7 million and $3.9 million for the years ended December 31, 2021 and 2020, respectively, and we had working capital of $9.6 million and $8.4 million as of the same dates, respectively. Working capital is defined as current assets less current liabilities.
Our operations are subject to all the risks, difficulties, complications and delays frequently encountered in connection with the development of new products, as well as those risks that are specific to the pharmaceutical and biotechnology industries in which we compete. Investors should evaluate us considering the delays, expenses, problems and uncertainties frequently encountered by companies developing markets for new products, services and technologies. We may never overcome these obstacles.
As a result of our current limited financial liquidity, we and our auditors have expressed substantial doubt regarding our ability to continue as a “going concern.”
As a result of our current limited financial liquidity, our auditors’ report for our 2021 financial statements, which is included as part of this report, contains a statement concerning our ability to continue as a “going concern.” Our limited liquidity could make it more difficult for us to secure additional financing or enter into strategic relationships on terms acceptable to us, if at all, and may materially and adversely affect the terms of any financing that we may obtain and our public stock price generally.
Our continuation as a “going concern” is dependent upon, among other things, achieving positive cash flow from operations and, if necessary, augmenting such cash flow using external resources to satisfy our cash needs. Our plans to achieve positive cash flow primarily include engaging in offerings of securities. Additional potential sources of funds include negotiating up-front and milestone payments on our current and potential future product candidates or royalties from sales of our products that secure regulatory approval and any milestone payments associated with such approved products. These cash sources could, potentially, be supplemented by financing or other strategic agreements. However, we may be unable to achieve these goals or obtain required funding on commercially reasonable terms, or at all, and therefore may be unable to continue as a going concern.
We may be unable to obtain the additional capital that is required to execute our business plan, which could restrict our ability to grow.
Our current capital and our other existing resources will be sufficient only to provide a limited amount of working capital and will not be sufficient to fund our expected continuing opportunities. While we project that our current capital resources are to fund our operations, including increased clinical trial costs, into the fourth quarter of 2022, we will require additional capital to continue to operate our business and complete our clinical development plans.
Future research and development, including clinical trial cost, capital expenditures and possible acquisitions, and our administrative requirements, such as salaries, insurance expenses and general overhead expenses, as well as legal compliance costs and accounting expenses, will require a substantial amount of additional capital and cash flow. There is no guarantee that we will be able to raise additional capital required to fund our ongoing business on commercially reasonable terms or at all.
We intend to pursue sources of additional capital through various financing transactions or arrangements, including collaboration arrangements, debt financing, equity financing or other means. We may not be successful in locating suitable financing transactions on commercially reasonable terms, in the time period required or at all, and we may not obtain the capital we require by other means. If we do not succeed in raising additional capital, our resources will not be sufficient to fund our operations going forward.
Any additional capital raised through the sale of equity may dilute the ownership percentage of our stockholders. This could also result in a decrease in the fair market value of our equity securities because our assets would be owned by a larger pool of outstanding equity. The terms of securities we issue in future capital transactions may be more favorable to our new investors, and may include preferences, superior voting rights and the issuance of warrants or other derivative securities which may have a further dilutive effect.
Our ability to obtain needed financing may be impaired by such factors as the capital markets, both generally and in the pharmaceutical and other drug development industries in particular, the limited diversity of our activities and/or the loss of key personnel. If the amount of capital we are able to raise from financing activities is not sufficient to satisfy our capital needs, even to the extent that we reduce our operations, we may be required to cease our operations.
We may incur substantial costs in pursuing future capital financing, including investment banking fees, legal fees, accounting fees, securities law compliance fees, printing and distribution expenses and other costs, which may adversely impact our financial condition.
Our business is subject to risks arising from epidemic diseases, such as the 2020 outbreak of the COVID-19 illness.
March of 2022 marked two years since the outbreak of COVID-19, was declared by the World Health Organization to be a pandemic. A pandemic, including COVID-19, or other public health epidemics pose the risk that we or our employees, contractors, suppliers, and other vendors may be prevented from conducting business activities for an indefinite period of time, including due to the spread of the disease within these groups or due to shutdowns that may be requested or mandated by governmental authorities. Early in the pandemic we paused enrollment in our Phase Ia/Ib clinical trial for 6 weeks to allow the health care systems involved in the trial time to focus resources on responding to the pandemic. Once enrollment was restarted in May 2020, we experienced no further delays. This delay did not have a material effect on the completion of enrollment or costs of the clinical trial. During the course of the pandemic, health care facilities have limited our ability to conduct on site patient data monitoring for our clinical trial, these visits are now successfully conducted remotely as necessary. We also experienced a delay, a short delay, early in the pandemic, in the manufacturing of our active ingredient and another minor delay in 2021 in the preparation of drug product We believe our supply of drug is adequate and these delays have not caused any disruption in the start of the new Phase II/III clinical trial which we initiated in January of 2022. This new trial has been designed to mitigate any potential effects of Covid-19 on site activations or subject enrollment.
While we have not, to date experienced any significant disruptions as a result of the pandemic, we are unable to estimate the future impact that COVID-19 could have on our operations. The recent trends in reduced infections and deaths, and increased levels of vaccination should help reduce the risk that the pandemic may slow potential enrollment of clinical trials and reduce the number of eligible patients for our clinical trials. While the pandemic could still disrupt the supply chain and the manufacture or shipment of both drug substance and finished drug product for our product candidates for preclinical testing and clinical trials, we believe that product secured in 2021 will be sufficient to complete the conduct of our new clinical trial. We often attend and present clinical updates at various medical and investor conferences throughout the year. The COVID-19 outbreak has caused, and may to continue to cause, cancellations or reduced attendance of these conferences and we may need to seek alternate methods to present clinical updates and to engage with the medical and investment communities. The COVID-19 outbreak, including new variants of the virus, and future mitigation measures may also have an adverse impact on global economic conditions which could have an adverse effect on our business and financial condition and our potential to conduct financings on terms acceptable to us, if at all. The extent to which the COVID-19 outbreak impacts our results will depend on future developments that remain uncertain and cannot be predicted, including new information that may emerge concerning the severity of the virus and the actions to contain its impact.
The markets for our product candidates are highly competitive and are subject to rapid scientific change, which could have a material adverse effect on our business, results of operations and financial condition.
The pharmaceutical and biotechnology industries in which we compete are highly competitive and characterized by rapid and significant technological change. We face intense competition from organizations such as pharmaceutical and biotechnology companies, as well as academic and research institutions and government agencies. Some of these organizations are pursuing products based on technologies similar to our technology. Other of these organizations have developed and are marketing products or are pursuing other technological approaches designed to produce products that are competitive with our product candidates in the therapeutic effect these competitive products have on the diseases targeted by our product candidates. Our competitors may discover, develop or commercialize products or other novel technologies that are more effective, safer or less costly than any that we may develop. Our competitors may also obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for our product candidates.
Many of our competitors are substantially larger than we are and have greater capital resources, research and development staffs and facilities than we have. In addition, many of our competitors are more experienced in drug discovery, development and commercialization, obtaining regulatory approvals and drug manufacturing and marketing.
We anticipate that the competition with our product candidates and technology will be based on a number of factors including product efficacy, safety, availability and price. The timing of market introduction of our planned future product candidates and competitive products will also affect competition among products. We expect the relative speed with which we can develop our product candidates, complete the required clinical trials, establish strategic partners and supply appropriate quantities of the product candidate for late stage trials, if required, to be important competitive factors. Our competitive position will also depend upon our ability to attract and retain qualified personnel, to obtain patent protection in non-U.S. markets, which we currently do not have, or otherwise develop proprietary products or processes and to secure sufficient capital resources for the period between technological conception and commercial sales or out-license to pharmaceutical partners. If we fail to develop and deploy a proposed product candidate in a successful and timely manner, we will in all likelihood not be competitive.
Our lack of diversification increases the risk of an investment in our Company and our financial condition and results of operations may deteriorate if we fail to diversify.
Our Board of Directors has centered our attention on our drug development activities, which are currently focused a limited number of product candidates. Our ability to diversify our investments will depend on our access to additional capital and financing sources and the availability and identification of suitable opportunities.
Larger companies have the ability to manage their risk by diversification. However, we lack and expect to continue to lack diversification, in terms of both the nature and geographic scope of our business. As a result, we will likely be impacted more acutely by factors affecting pharmaceutical and biotechnology industries in which we compete than we would if our business were more diversified, enhancing our risk profile. If we cannot diversify our operations, our financial condition and results of operations could deteriorate.
Our business may suffer if we do not attract and retain talented personnel.
Our success will depend in large measure on the abilities, expertise, judgment, discretion, integrity and good faith of our management and other personnel in conducting our business. We have a small management team, and the loss of a key individual or inability to attract suitably qualified staff could materially adversely impact our business.
Our success depends on the ability of our management, employees, consultants and strategic partners, if any, to interpret market data correctly and to interpret and respond to economic market and other conditions in order to locate and adopt appropriate investment opportunities, monitor such investments, and ultimately, if required, to successfully divest such investments. Further, no assurance can be given that our key personnel will continue their association or employment with us or that replacement personnel with comparable skills can be found. We will seek to ensure that management and any key employees are appropriately compensated; however, their services cannot be guaranteed. If we are unable to attract and retain key personnel, our business may be adversely affected.
We may be required to defend lawsuits or pay damages for product liability claims.
Product liability is a major risk in testing and marketing biotechnology and pharmaceutical products. We may face substantial product liability exposure in human clinical trials and in the sale of products after regulatory approval. Product liability claims, regardless of their merits, could exceed policy limits, divert management’s attention and adversely affect our reputation and the demand for our product. In any such event, your investment in our securities could be materially and adversely affected.
Risks Related to Acquisitions and Integrations
We have and expect to incur substantial costs related to the Mergers and subsequent integration efforts.
We have incurred and expect to incur a number of non-recurring costs associated with the Mergers and related transactions. These costs include legal, financial advisory, accounting, consulting and other advisory fees, retention, severance and employee benefit-related costs, regulatory fees, closing, integration and other related costs. Some of these costs are payable regardless of whether or not the Mergers are completed.
Although the Mergers have been completed, integration may be more difficult, costly, or time-consuming than expected, and we may not realize the anticipated benefits of the underlying acquisition.
The anticipated benefits of the combined company, including product candidate diversification and growth, may not be realized fully or at all or may take longer to commercialize than expected and integration may result in additional and unforeseen expenses. An inability to realize the full extent of the anticipated benefits, as well as any delays encountered in the integration process, could have an adverse effect upon our operating results.
In addition, we and CPP operated independently prior to the completion of the Mergers. It is possible that the now-active integration process could result in the loss of one or more key employees, including employees of CPP, the disruption of each company’s ongoing businesses or inconsistencies in standards, controls, procedures, and policies that adversely affect each company’s ability to maintain relationships with clients, customers, depositors, and employees or to achieve the anticipated benefits of the Mergers. Integration efforts between the companies may also divert management attention and resources. These integration matters could have an adverse effect on the Company during this transition period and for an undetermined period.
We may not have discovered certain liabilities or other matters related to CPP, which may adversely affect the future financial performance of the combined company.
In the course of the due diligence review that we conducted prior to the execution of the merger agreement, we may not have discovered, or may have been unable to properly quantify, certain liabilities of CPP or other factors that may have an adverse effect on the business, results of operations, financial condition, and cash flows of the combined company.
Our estimates and judgments related to the acquisition accounting methods used to record the purchase price allocation related to the merger may be inaccurate.
Our management will make significant accounting judgments and estimates related to the application of acquisition accounting of the Mergers under GAAP, as well as the underlying valuation models. Our business, operating results, and financial condition could be materially adversely impacted in future periods if the accounting judgments and estimates prove to be inaccurate.
Risks Related to the Development and Approval of New Drugs
Clinical trials required for our product candidate are expensive and time-consuming, and their outcome is highly uncertain. If any of our drug trials are delayed or yield unfavorable results, we will have to delay or may be unable to obtain regulatory approval for our product candidate.
We must conduct extensive testing of our product candidate before we can obtain regulatory approval to market and sell it. We need to conduct both preclinical animal testing and human clinical trials. Conducting these trials is a lengthy, time-consuming and expensive process. These tests and trials may not achieve favorable results for many reasons, including, among others, failure of the product candidate to demonstrate safety or efficacy, the development of serious or life-threatening adverse events, or side effects, caused by or connected with exposure to the product candidate, difficulty in enrolling and maintaining subjects in the clinical trial, lack of sufficient supplies of the product candidate or comparator drug, and the failure of clinical investigators, trial monitors, contractors, consultants, or trial subjects to comply with the trial protocol. A clinical trial may fail because it did not include a sufficient number of patients to detect the endpoint being measured or reach statistical significance. A clinical trial may also fail because the dose(s) of the investigational drug included in the trial were either too low or too high to determine the optimal effect of the investigational drug in the disease setting. Many clinical trials are conducted under the oversight of Independent Data Monitoring Committees (“IDMCs”) also known as DSMB’s. These independent oversight bodies are made up of external experts who review the progress of ongoing clinical trials, including available safety and efficacy data, and make recommendations concerning a trial’s continuation, modification, or termination based on interim, unblinded data. Any of our ongoing clinical trials may be discontinued or amended in response to recommendations made by responsible IDMCs based on their review of such interim trial results.
We will need to reevaluate our product candidate if it does not test favorably and either conduct new trials, which are expensive and time consuming, or abandon our drug development program. Even if we obtain positive results from preclinical or clinical trials, we may not achieve the same success in future trials. Many companies in the biopharmaceutical industry have suffered significant setbacks in clinical trials, even after promising results have been obtained in earlier trials. The failure of clinical trials to demonstrate safety and effectiveness for the desired indication could harm the development of our product candidate and our business, financial condition and results of operations may be materially harmed.
We face significant risks in our product candidate development efforts.
Our business depends on the successful development and commercialization of our product candidates. We are currently focused on developing our product candidates, SBP-101 and eflornithine, and are not permitted to market it in the United States until we receive approval of an NDA from the FDA, or in any foreign jurisdiction until we receive the requisite approvals from such jurisdiction. The process of developing new drugs and/or therapeutic products is inherently complex, unpredictable, time-consuming, expensive and uncertain. We must make long-term investments and commit significant resources before knowing whether our development programs will result in drugs that will receive regulatory approval and achieve market acceptance. A product candidate that appears to be promising at all stages of development may not reach the market for a number of reasons that may not be predictable based on results and data from the clinical program. A product candidate may be found ineffective or may cause harmful side effects during clinical trials, may take longer to progress through clinical trials than had been anticipated, may not be able to achieve the pre-defined clinical endpoints even though clinical benefit may have been achieved, may fail to receive necessary regulatory approvals, may prove impracticable to manufacture in commercial quantities at reasonable cost and with acceptable quality, or may fail to achieve market acceptance.
We cannot predict whether or when we will obtain regulatory approval to commercialize our initial product candidate and we cannot, therefore, predict the timing of any future revenues from this or other product candidates, if any. The FDA has substantial discretion in the drug approval process, including the ability to delay, limit or deny approval of a product candidate for many reasons. For example, the FDA:
| |
●
|
could determine that the information provided by us was inadequate, contained clinical deficiencies or otherwise failed to demonstrate the safety and effectiveness of any of our product candidates for any indication;
|
| |
●
|
may not find the data from clinical trials sufficient to support the submission of an NDA or to obtain marketing approval in the United States, including any findings that the clinical and other benefits of our product candidates outweigh their safety risks;
|
| |
●
|
may disagree with our trial design or our interpretation of data from preclinical studies or clinical trials or may change the requirements for approval even after it has reviewed and commented on the design for our trials;
|
| |
●
|
may identify deficiencies in the manufacturing processes or facilities of third-party manufacturers with which we enter into agreements for the manufacturing of our product candidates;
|
| |
●
|
may approve our product candidates for fewer or more limited indications than we request or may grant approval contingent on the performance of costly post-approval clinical trials;
|
| |
●
|
may change its approval policies or adopt new regulations; or
|
| |
●
|
may not approve the labeling claims that we believe are necessary or desirable for the successful commercialization of our product candidates.
|
Any failure to obtain regulatory approval of our initial product candidate or future product candidates we develop, if any, would significantly limit our ability to generate revenues, and any failure to obtain such approval for all of the indications and labeling claims we deem desirable could reduce our potential revenues.
Our product candidate is based on new formulation of an existing technology which has never been approved for the treatment of any cancer and, consequently, is inherently risky. Concerns about the safety and efficacy of our product candidate could limit our future success.
We are subject to the risks of failure inherent in the development of product candidates based on new technologies. These risks include the possibility that any product candidates we create will not be effective, that our current product candidate will be unsafe, ineffective or otherwise fail to receive the necessary regulatory approvals or that our product candidate will be hard to manufacture on a large scale or will be uneconomical to market.
Many pharmaceutical products cause multiple potential complications and side effects, not all of which can be predicted with accuracy and many of which may vary from patient to patient. Long term follow-up data may reveal additional complications associated with our product candidate. The responses of potential physicians and others to information about complications could materially affect the market acceptance of our product candidate, which in turn would materially harm our business.
Our ability to commence and complete the planned FAP registration trial depends substantially on a third-party and its resources.
In July 2021, Cancer Prevention licensed the U.S. and Canadian rights to Flynpovi to One-Two Therapeutics Assets Limited (“One-Two”), a private commercial-stage specialty pharma company focused on GI and orphan disease. Under the terms of the license, One-Two is responsible for all costs of development and approval of Flynpovi in North America. Accordingly, our ability to potentially obtain FDA approval of Flynpovi is dependent on One-Two’s ability to fund and complete the registration trial. Any failure to obtain regulatory approval of Flynpovi in this context, could significantly limit our ability to obtain milestone payments or generate revenues from Flynpovi.
Due to our reliance on third parties to conduct our clinical trials, we are unable to directly control the timing, conduct, expense and quality of our clinical trials, which could adversely affect our clinical data and results and related regulatory approvals.
We extensively outsource our clinical trial activities and expect to directly perform only a small portion of the preparatory stages for planned trials. We rely on independent third-party CROs to perform most of our clinical trials, including document preparation, site identification, screening and preparation, pre-study visits, training, program management and bio-analytical analysis. Many important aspects of the services performed for us by the CROs are out of our direct control. If there is any dispute or disruption in our relationship with our CROs, our clinical trials may be delayed. Moreover, in our regulatory submissions, we rely on the quality and validity of the clinical work performed by third-party CROs. If a CRO’s processes, methodologies or results are determined to be invalid or inadequate, our own clinical data and results and related regulatory approvals could be adversely affected or invalidated.
We rely on third-party suppliers and other third parties for production of our product candidate and our dependence on these third parties may impair the advancement of our research and development programs and the development of our product candidates.
We rely on, and expect to continue to rely on, third parties for the supply of raw materials and manufacture of drug supplies necessary to conduct our preclinical studies and clinical trials. During 2021 the Company, in collaboration with our manufacturing partner confirmed a new shorter and less expensive synthesis of the active drug substance. However, delays in production by third parties could delay our clinical trials or have an adverse impact on any commercial activities. In addition, the fact that we are dependent on third parties for the manufacture of and formulation of our product candidates means that we are subject to the risk that the products may have manufacturing defects that we have limited ability to prevent or control. Although we oversee these activities to ensure compliance with our quality standards, budgets and timelines, we have had and will continue to have less control over the manufacturing of our product candidates than potentially would be the case if we were to manufacture our product candidates. Further, the third parties we deal with could have staffing difficulties, might undergo changes in priorities or may become financially distressed, which would adversely affect the manufacturing and production of our product candidates.
The results of pre-clinical studies and completed clinical trials are not necessarily predictive of future results, and our current product candidates may not have favorable results in later studies or trials.
Pre-clinical studies and Phase I clinical trials are not primarily designed to test the efficacy of a product candidate in the general population, but rather to test initial safety, to study pharmacokinetics and pharmacodynamics, to study limited efficacy in a small number of study patients in a selected disease population, and to identify and attempt to understand the product candidate’s side effects at various doses and dosing schedules. Success in pre-clinical studies or completed clinical trials does not ensure that later studies or trials, including continuing pre-clinical studies and large-scale clinical trials, will be successful nor does it necessarily predict future results. Favorable results in early studies or trials may not be repeated in later studies or trials, and product candidates in later stage trials may fail to show acceptable safety and efficacy despite having progressed through earlier trials.
Risks Related to the Regulation of our Business
Federal and state pharmaceutical marketing compliance and reporting requirements may expose us to regulatory and legal action by state governments or other government authorities.
The Food and Drug Administration Modernization Act (the “FDMA”) established a public registry of open clinical trials involving drugs intended to treat serious or life-threatening diseases or conditions in order to promote public awareness of and access to these clinical trials. Under the FDMA, pharmaceutical manufacturers and other trial sponsors are required to post the general purpose of these trials, as well as the eligibility criteria, location and contact information of the trials. Failure to comply with any clinical trial posting requirements could expose us to negative publicity, fines and other penalties, all of which could materially harm our business.
In recent years, several states, including California, Vermont, Maine, Minnesota, New Mexico and West Virginia have enacted legislation requiring pharmaceutical companies to establish marketing compliance programs and file periodic reports on sales, marketing, pricing and other activities. Similar legislation is being considered in other states. Many of these requirements are new and uncertain, and available guidance is limited. Unless we are in full compliance with these laws, we could face enforcement actions and fines and other penalties and could receive adverse publicity, all of which could harm our business.
If the product candidate we develop becomes subject to unfavorable pricing regulations, third party reimbursement practices or healthcare reform initiatives, our ability to successfully commercialize our product candidate may be impaired.
Our future revenues, profitability and access to capital will be affected by the continuing efforts of governmental and private third-party payors to contain or reduce the costs of health care through various means. We expect several federal, state and foreign proposals to control the cost of drugs through government regulation. We are unsure of the impact recent health care reform legislation may have on our business or what actions federal, state, foreign and private payors may take in response to the recent reforms. Therefore, it is difficult to predict the effect of any implemented reform on our business. Our ability to commercialize our product candidate successfully will depend, in part, on the extent to which reimbursement for the cost of such product candidate and related treatments will be available from government health administration authorities, such as Medicare and Medicaid in the United States, private health insurers and other organizations. Significant uncertainty exists as to the reimbursement status of newly approved health care products, particularly for indications for which there is no current effective treatment or for which medical care typically is not sought. Adequate third-party coverage may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product research and development. If adequate coverage and reimbursement levels are not provided by government and third-party payors for use of our product candidates, our product candidates may fail to achieve market acceptance and our results of operations will be harmed.
Healthcare legislative reform measures may have a material adverse effect on our business and results of operations.
Legislative and regulatory actions affecting government prescription drug procurement and reimbursement programs occur relatively frequently. In the United States, the ACA was enacted in 2010 to expand healthcare coverage. Since then, numerous efforts have been made to repeal, amend or administratively limit the ACA in whole or in part. For example, the Tax Cuts and Jobs Act, signed into law by President Trump in 2017, repealed the individual health insurance mandate, which is considered a key component of the ACA. In December 2018, a Texas federal district court struck down the ACA on the grounds that the individual health insurance mandate is unconstitutional, although this ruling has been stayed pending appeal. The ongoing challenges to the ACA and new legislative proposals have resulted in uncertainty regarding the ACA's future viability and destabilization of the health insurance market. The resulting impact on our business is uncertain and could be material.
Efforts to control prescription drug prices could also have a material adverse effect on our business. For example, in 2018, President Trump and the Secretary of the U.S. Department of Health and Human Services (“HHS”) released the “American Patients First Blueprint” and have begun implementing certain portions. The initiative includes proposals to increase generic drug and biosimilar competition, enable the Medicare program to negotiate drug prices more directly and improve transparency regarding drug prices and ways to lower consumers’ out-of-pocket costs. The Trump administration also proposed to establish an “international pricing index” that would be used as a benchmark to determine the costs and potentially limit the reimbursement of drugs under Medicare Part B. Among other pharmaceutical manufacturer industry-related proposals, Congress has proposed bills to change the Medicare Part D benefit to impose an inflation-based rebate in Medicare Part D and to alter the benefit structure to increase manufacturer contributions in the catastrophic phase. The volume of drug pricing-related bills has dramatically increased under the current Congress, and the resulting impact on our business is uncertain and could be material.
In addition, many states have proposed or enacted legislation that seeks to regulate pharmaceutical drug pricing indirectly or directly, such as by requiring biopharmaceutical manufacturers to publicly report proprietary pricing information or to place a maximum price ceiling on pharmaceutical products purchased by state agencies. For example, in 2017, California’s governor signed a prescription drug price transparency state bill into law, requiring prescription drug manufacturers to provide advance notice and explanation for price increases of certain drugs that exceed a specified threshold. Both Congress and state legislatures are considering various bills that would reform drug purchasing and price negotiations, allow greater use of utilization management tools to limit Medicare Part D coverage, facilitate the import of lower-priced drugs from outside the United States and encourage the use of generic drugs. Such initiatives and legislation may cause added pricing pressures on our products.
Changes to the Medicaid program at the federal or state level could also have a material adverse effect on our business. Proposals that could impact coverage and reimbursement of our products, including giving states more flexibility to manage drugs covered under the Medicaid program and permitting the re-importation of prescription medications from Canada or other countries, could have a material adverse effect by limiting our products’ use and coverage. Furthermore, state Medicaid programs could request additional supplemental rebates on our products as a result of an increase in the federal base Medicaid rebate. To the extent that private insurers or managed care programs follow Medicaid coverage and payment developments, they could use the enactment of these increased rebates to exert pricing pressure on our products, and the adverse effects may be magnified by their adoption of lower payment schedules.
Other proposed regulatory actions affecting manufacturers could have a material adverse effect on our business. It is difficult to predict the impact, if any, of any such proposed legislative and regulatory actions or resulting state actions on the use and reimbursement of our products in the United States, but our results of operations may be adversely affected.
Risks Related to our Intellectual Property
If we are unable to obtain, maintain and enforce our proprietary rights, we may not be able to compete effectively or operate profitably.
We are party to a license agreement with UFRF. The patent underlying the licensed intellectual property and those of other biopharmaceutical companies, are generally uncertain and involve complex legal, scientific and factual questions.
Our ability to develop and commercialize drugs depends in significant part on our ability to: (i) obtain and/or develop broad, protectable intellectual property; (ii) obtain additional licenses, if required, to the proprietary rights of others on commercially reasonable terms; (iii) operate without infringing upon the proprietary rights of others; (iv) prevent others from infringing on our proprietary rights; and (v) protect our corporate know-how and trade secrets.
Patents that we may acquire and those that might be issued in the future, may be challenged, invalidated or circumvented, and the rights granted thereunder may not provide us with proprietary protection or competitive advantages against competitors with similar technology. Furthermore, our competitors may independently develop similar technologies or duplicate any technology we develop. Because of the extensive time required for development, testing and regulatory review of a potential product candidates, it is possible that, before any of our product candidates can be commercialized, any related patent may expire or remain in force for only a short period following commercialization, thus reducing any advantage of the patent.
Because patent applications in the U.S. and many foreign jurisdictions are typically not published until at least 12 months after filing, or in some cases not at all, and because publications of discoveries in the scientific literature often lag behind actual discoveries, neither we nor our licensors can be certain that either we or our licensors were the first to make the inventions claimed in issued patents or pending patent applications, or that we were the first to file for protection of the inventions set forth in these patent applications.
Additionally, UFRF previously elected to seek protection for certain elements of the licensed technology only in the United States, and the time to file for international patent protection has expired. This limits the strength of the Company’s intellectual property position in certain markets and could affect the overall value of the Company to a potential corporate partner.
Obtaining and maintaining our patent protection depends upon compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
The United States Patent and Trademark Office and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other provisions during the patent process. There are situations in which noncompliance can result in abandonment or lapse of a patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, competitors might be able to enter the market earlier than would otherwise have been the case.
We may be exposed to infringement or misappropriation claims by third parties, which, if determined adversely to us, could cause us to pay significant damage awards.
There has been substantial litigation and other proceedings regarding patent and other intellectual property rights in the pharmaceutical and biotechnology industries. We may become a party to various types of patent litigation or other proceedings regarding intellectual property rights from time to time even under circumstances where we are not using and do not intend to use any of the intellectual property involved in the proceedings.
The cost of any patent litigation or other proceeding, even if resolved in our favor, could be substantial. Some of our competitors may be able to sustain the cost of such litigation or proceedings more effectively than we will be able to because our competitors may have substantially greater financial resources. If any patent litigation or other proceeding is resolved against us, we or our collaborators may be enjoined from developing, manufacturing, selling or importing our drugs without a license from the other party and we may be held liable for significant damages. We may not be able to obtain any required license(s) on commercially acceptable terms or at all.
Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace. Patent litigation and other proceedings may also absorb significant management time.
Confidentiality agreements with employees and others may not adequately prevent disclosure of our know-how, trade secrets and other proprietary information and may not adequately protect our intellectual property, which could impede our ability to compete.
Because we operate in the highly technical field of medical technology development, we rely in part on trade secret protection in order to protect our proprietary trade secrets and unpatented know-how. However, trade secrets are difficult to protect, and we cannot be certain that others will not develop the same or similar technologies on their own. We have taken steps, including entering into confidentiality agreements with all of our employees, consultants and corporate partners to protect our trade secrets and unpatented know-how. These agreements generally require that the other party keep confidential and not disclose to third parties any confidential information developed by the party or made known to the party by us during the course of the party’s relationship with us. We also typically obtain agreements from these parties which provide that inventions conceived by the party in the course of rendering services to us will be our exclusive property. However, these agreements may not be honored and may not effectively assign intellectual property rights to us. Enforcing a claim that a party illegally obtained and is using our trade secrets or know-how is difficult, expensive and time consuming, and the outcome is unpredictable. In addition, courts outside the United States may be less willing to protect trade secrets or know-how. The failure to obtain or maintain trade secret protection could adversely affect our competitive position.
We may be subject to claims that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
As is common in the biotechnology industry, we employ individuals who were previously employed at other biotechnology companies, including our competitors or potential competitors. Although no claims against us are currently pending, we may be subject to claims that these employees or we have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
Risks Associated with Our Common Stock
Raising additional capital may cause dilution to our stockholders or restrict our operations.
To the extent that we raise additional capital through the sale of equity or convertible debt securities, stockholders’ ownership interest will be diluted, and the terms may include liquidation or other preferences that adversely affect their rights as stockholders. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions such as incurring additional debt, making capital expenditures or declaring dividends. Any of these events could adversely affect our ability to achieve our product development and commercialization goals and harm our business. We do not anticipate any adverse effects stemming from the lack of available credit facilities at this time.
Issuances of common stock in offerings or pursuant to the exercise of rights to purchase shares may cause the price of our common stock to decline and cause investors to lose a significant portion of their investment.
If our stockholders sell substantial amounts of our common stock in the public market or upon the expiration of any statutory holding period under Rule 144, or upon expiration of lock-up periods applicable to outstanding shares or issued upon the exercise of outstanding options or warrants, it could create a circumstance commonly referred to as an “overhang” and in anticipation of which the market price of our common stock could fall. The existence of an overhang, whether sales have occurred or are occurring, also could make our ability to raise additional financing through the sale of equity or equity-related securities in the future at a time and price that we deem reasonable or appropriate more difficult. As of December 31, 2021, we had outstanding options to purchase 2,463,636 shares of our common stock at a weighted-average exercise price of $5.83 per share with a remaining contractual life of 7.3 years and outstanding warrants to purchase 5,109,501 shares of common stock at a weighted-average exercise price of $4.59 per share and a remaining exercise period of 3.1 years.
Securities analysts may not initiate coverage or continue to cover our common stock, and this may have a negative impact on the market price of our common stock.
Common stock prices are often significantly influenced by the research and reports that securities analysts publish about companies and their business. We do not have any control over these analysts. There is no guarantee that securities analysts will cover, or continue to cover, our common stock. If securities analysts do not cover our common stock, the lack of research coverage may adversely affect the market price of our common stock. If our common stock is covered by securities analysts and our stock is downgraded, our stock price will likely decline. If one or more of these analysts ceases to cover us or fails to publish regular reports on us, we can lose visibility in the financial markets, which can cause our stock price or trading volume to decline.
Our charter documents and Delaware law may inhibit a takeover that stockholders consider favorable.
Provisions of our certificate of incorporation and bylaws and applicable provisions of Delaware law may make it more difficult for or prevent a third party from acquiring control of us without the approval of our board of directors. These provisions:
| |
●
|
set limitations on the removal of directors;
|
| |
●
|
limit who may call a special meeting of stockholders;
|
| |
●
|
establish advance notice requirements for nominations for election to our board of directors or for proposing matters that can be acted upon at stockholder meetings;
|
| |
●
|
do not permit cumulative voting in the election of our directors, which would otherwise permit less than a majority of stockholders to elect directors;
|
| |
●
|
establish a classified board of directors limiting the number of directors that are elected each year; and
|
| |
●
|
provide our board of directors the ability to designate the terms of and issue preferred stock without stockholder approval.
|
In addition, Section 203 of the Delaware General Corporation Law generally limits our ability to engage in any business combination with certain persons who own 15% or more of our outstanding voting stock or any of our associates or affiliates who at any time in the past three years have owned 15% or more of our outstanding voting stock unless our board of directors has pre-approved the acquisitions that lead to such ownership. These provisions may have the effect of entrenching our management team and may deprive stockholders of the opportunity to sell their shares to potential acquirers at a premium over prevailing prices. This potential inability to obtain a control premium could reduce the price of our common stock.
If we issue preferred stock, the rights of holders of our common stock and the value of such common stock could be adversely affected.
Our Board of Directors is authorized to issue classes or series of preferred stock, without any action on the part of the stockholders. The Board of Directors also has the power, without stockholder approval, to set the terms of any such classes or series of preferred stock, including voting rights, dividend rights and preferences over the common stock with respect to dividends or upon the liquidation, dissolution or winding-up of our business and other terms. If we issue preferred stock in the future that has a preference over the common stock with respect to the payment of dividends or upon liquidation, dissolution or winding-up, or if we issue preferred stock with voting rights that dilute the voting power of the common stock, the rights of holders of the common stock or the value of the common stock would be adversely affected.
If we fail to maintain effective internal controls over financial reporting, the price of our common stock may be adversely affected.
We are required to establish and maintain appropriate internal controls over financial reporting. Failure to establish those controls, or any failure of those controls once established, could adversely impact our public disclosures regarding our business, financial condition or results of operations. Any failure of these controls could also prevent us from maintaining accurate accounting records and discovering accounting errors and financial fraud.
Management’s assessment of internal controls over financial reporting may identify weaknesses that need to be addressed or other potential matters that may raise concerns for investors. Any actual or perceived weaknesses that need to be addressed in our internal control over financial reporting or disclosure of management’s assessment of our internal controls over financial reporting may have an adverse impact on the price of our common stock.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”).
In some cases, you can identify forward-looking statements by the following words: “anticipate,” “assume,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “ongoing,” “plan,” “potential,” “predict,” “project,” “should,” “will,” “would,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. Forward-looking statements are not a guarantee of future performance or results and will not necessarily be accurate indications of the times at, or by, which such performance or results will be achieved. Forward-looking statements are based on information available at the time the statements are made and involve known and unknown risks, uncertainties and other factors that may cause our results, levels of activity, performance or achievements to be materially different from the information expressed or implied by the forward-looking statements in this report. These factors include:
| |
●
|
our lack of diversification and the corresponding risk of an investment in our Company;
|
| |
●
|
potential deterioration of our financial condition and results due to failure to diversify;
|
| |
●
|
our ability to successfully complete acquisitions;
|
| |
●
|
our ability to integrate acquired companies and operations for new product candidates
|
| |
●
|
our ability to obtain additional capital, on acceptable terms or at all, required to implement our business plan;
|
| |
●
|
final results of our Phase I clinical trial;
|
| |
●
|
progress and success of our randomized Phase II/III clinical trial;
|
| |
●
|
our ability to demonstrate safety and effectiveness of our product candidate;
|
| |
●
|
our ability to obtain regulatory approvals for our product candidate in the United States, the European Union, or other international markets;
|
| |
●
|
the market acceptance and future sales of our product candidate;
|
| |
●
|
the cost and delays in product development that may result from changes in regulatory oversight applicable to our product candidate;
|
| |
●
|
the rate of progress in establishing reimbursement arrangements with third-party payors;
|
| |
●
|
the effect of competing technological and market developments;
|
| |
●
|
the costs involved in filing and prosecuting patent applications and enforcing or defending patent claims; and
|
| |
●
|
other risk factors included under the caption “Risk Factors” starting on page 9 of this report.
|
You should read the matters described in “Risk Factors” and the other cautionary statements made in this report as being applicable to all related forward-looking statements wherever they appear in this report. We cannot assure you that the forward-looking statements in this report will prove to be accurate and therefore you are encouraged not to place undue reliance on forward-looking statements. You should read this report completely. Other than as required by law, we undertake no obligation to update or revise these forward-looking statements, even though our situation may change in the future.
We caution readers not to place undue reliance on any forward-looking statement that speaks only as of the date made and to recognize that forward-looking statements are predictions of future results, which may not occur as anticipated. Actual results could differ materially from those anticipated in the forward-looking statements and from historical results due to the risks and uncertainties described under the heading “Risk Factors” in this prospectus, as well as others that we may consider immaterial or do not anticipate at this time. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. Our expectations reflected in our forward-looking statements can be affected by inaccurate assumptions that we might make or by known or unknown risks and uncertainties, including those described under the heading “Risk Factors” in this prospectus. The risks and uncertainties described i under the heading “Risk Factors” in this prospectus are not exclusive and further information concerning us and our business, including factors that potentially could materially affect our financial results or condition, may emerge from time to time. We assume no obligation to update forward-looking statements to reflect actual results or changes in factors or assumptions affecting such forward-looking statements. We advise stockholders and investors to consult any further disclosures we may make on related subjects in our subsequent annual reports on Form 10-K, quarterly reports on Form 10-Q, and current reports on Form 8-K that we file with or furnish to the U.S. Securities and Exchange Commission (the “SEC”).
USE OF PROCEEDS
If all of the warrants offered pursuant to this prospectus are exercised in full for cash, we will receive approximately an additional $10.9 million in cash, based on the exercise price of $4.54 per share. However, the warrants contain a cashless exercise provision that permit exercise of warrants on a cashless basis at any time where there is no effective registration statement under the Securities Act covering the issuance of the underlying shares.
We intend to use the net proceeds from this offering as follows:
| |
●
|
for the continued clinical development of our initial product candidates SBP-101 and eflornithine; and
|
| |
●
|
the remainder for working capital, business development and other general corporate purposes.
|
Our expected use of net proceeds from this offering represents our intentions based on our present plans and business conditions, which could change as our plans and business conditions evolve. The amount and timing of our actual expenditures will depend on numerous factors, including the timing and success of clinical studies or clinical studies we may commence in the future, the timing of regulatory submissions and the feedback from regulatory authorities. As a result, our management will have broad discretion over the use of the net proceeds from this offering. Pending our use of the net proceeds from this offering, we may temporarily invest the net proceeds in investment-grade, interest-bearing securities.
We anticipate that the net proceeds from this offering together with our existing cash and cash equivalents will be sufficient to enable us to fund our operating expenses and capital expenditure requirements into the first quarter of 2023. We currently estimate the funds will allow us to make significant progress in the conduct of our new Phase II/III randomized double-blind, placebo-controlled clinical trial (known as the ASPIRE trial) for the treatment of Pancreatic ductile adenocarcinoma. In addition, the funds would enable us to initiate a new development program for our product candidate SBP-101 in ovarian cancer. Expansion of the current trial, if the futility analysis is positive, into the Phase III segment will be required for FDA or other similar approvals. The cost and timing of additional clinical trials are highly dependent on the number of indications we pursue and the nature and size of the trials. However, it is estimated that the next steps in the approval process for SBP-101 in pancreatic cancer could cost between $40 and $75 million.
Predicting the cost necessary to develop product candidates can be difficult and we anticipate we will need additional funds to complete the development work generally required for obtaining regulatory approval to commercialize a drug. We have based these estimates on assumptions that may prove to be wrong, and we could use our available capital resources sooner than we currently expect.
MARKET INFORMATION
Our common stock is listed on the Nasdaq Capital Market under the symbol “PBLA.” As of June 16, 2022, there were 285 holders of record of our common stock.
Equity Compensation Plan Information
The following table presents the number of shares of common stock authorized for issuance under the Company’s equity compensation plans as of December 31, 2021:
|
Plan Category
|
|
Number of Securities to Be
Issued Upon Exercise of
Outstanding Options,
Warrants and Rights
|
|
Weighted-Average Exercise
Price of Outstanding Options,
Warrants and Rights
|
|
Number of Securities
Remaining Available for
Future Issuance under Equity
Compensation Plans
|
|
Equity compensation plans approved by security holders
|
|
2,463,636()
|
|
$5.83
|
|
1,345,863()
|
|
Equity compensation plans not approved by security holders
|
|
–
|
|
–
|
|
–
|
|
Total
|
|
2,463,636
|
|
|
|
1,345,863
|
|
(a)
|
Includes 2,239,636 shares underlying common stock options under the 2016 Plan and 224,000 shares underlying common stock options under the 2011 plan. We ceased issuing awards under the 2011 Plan upon stockholder approval of the 2016 plan in 2016.
|
|
(b)
|
The 2016 Plan provides that the number of shares of common stock available for issuance under the plan will increase on January 1 of each year beginning in 2021 and ending on January 1, 2025 in an amount equal to the lesser of (i) the number of shares necessary to increase the total option pool to 20% of the total number of fully diluted shares (as defined in the Amended 2016 Plan) as of December 31 of the immediately preceding calendar year and (ii) such lesser number of shares as may be determined by the Board of Directors or its Compensation Committee prior to January 1st of any calendar year.
|
2011 Stock Option Plan
Our 2011 Stock Option Plan (the “2011 Plan”) was adopted by our Board of Directors in 2011 and subsequently approved by our stockholders in 2012. Upon the initial stockholder approval of the 2016 Plan, our Board of Directors ceased making grants under the 2011 Plan, although awards outstanding under the 2011 Plan will remain outstanding in accordance with and pursuant to the terms thereof. Options granted under the 2011 Plan have a maximum term of ten years and generally vest over zero to two years for employees. As of June 16, 2022, options to purchase 224,000 shares of common stock remained outstanding under the 2011 Plan with a weighted average price of $2.97 per share.
2016 Omnibus Incentive Plan
Our 2016 Omnibus Incentive Plan was initially adopted by our Board of Directors in March 2016 and approved by our stockholders in May 2016. It was amended and restated by our Board of Directors in April 2020 and approved by our stockholders in May 2020 (as amended, the “2016 Plan”). The 2016 Plan permits the granting of incentive and non-statutory stock options, restricted stock, stock appreciation rights and other stock awards to eligible employees, directors and consultants. We grant options to purchase shares of common stock under the 2016 Plan at no less than the fair market value of the underlying common stock as of the date of grant. Options granted under the Plan have a maximum term of ten years. Under the 2016 Plan, a total of 2,800,000 shares of common stock were initially reserved for issuance and 1,472,992shares have been added pursuant to its annual evergreen feature. As of June 16, 2022 options to purchase 2,220,136 shares of our common stock were outstanding under the 2016 Plan with a weighted average price of $6.04 per share. A total of 2,019,776 shares of common stock remained available for future grants under the 2016 Plan as of the same date, subject to further adjustment by the evergreen provision described under “Shares Available” below.
Purpose
The purpose of the 2016 Plan is to promote the interests of our Company and our stockholders by providing key personnel of our Company and our affiliates with an opportunity to acquire a proprietary interest in the Company and thereby develop a stronger incentive to put forth maximum effort for the continued success and growth of our Company and our affiliates. In addition, the opportunity to acquire a proprietary interest in our Company will aid in attracting and retaining key personnel of outstanding ability. The 2016 Plan is also intended to provide non-employee directors of the Company with an opportunity to acquire a proprietary interest in the Company, to compensate non-employee directors for their contributions to the Company and to aid in attracting and retaining non-employee directors.
Administration
The 2016 Plan is administered by the Compensation Committee of the Board of Directors (the “Committee”). The Committee has the authority to adopt, revise and waive rules relating to the 2016 Plan and to determine the timing and identity of participants, the amount of any awards and other terms and conditions of awards. The Committee may delegate its responsibilities under the 2016 Plan to one or more of its members or to one or more directors or executive officers of the Company with respect to the selection and grants of awards to employees of the Company who are not deemed to be officers, directors or 10% stockholders of the Company under applicable federal securities laws. The Board of Directors will perform the duties and have the responsibilities of the Committee with respect to awards made to non-employee directors.
Eligibility
All employees of our Company and our affiliates, non-employee directors of our Company and any consultant or advisor who is a natural person and provides services to us or our affiliates are eligible to receive awards under the 2016 Plan at the discretion of the Committee or the Board, as applicable. No awards may be granted under the 2016 Plan in conjunction with a capital-raising transaction or the promotion or maintenance of a market for our securities. Incentive stock options under the 2016 Plan may be awarded to employees of the Company. As of June 16, 2022, there were approximately 18 total employees and non-employee directors. Such employees, directors and others who currently or may in the future provide services to us and our affiliates may be considered for the grant of awards under the 2016 Plan at the discretion of the Committee or the Board, as applicable.
Shares Available
The total number of shares of Company Common Stock available for distribution under the 2016 Plan is 2,800,000, subject to adjustment for future stock splits, stock dividends and similar changes in the capitalization of the Company. In addition, the 2016 Plan provides that the number of shares of Common Stock available for issuance under the 2016 Plan will increase on January 1 of each year beginning in 2021 and ending on January 1, 2025 in an amount equal to the lesser of (i) the number of the shares necessary to increase the total option pool to 20% of the total number of Fully Diluted Shares (as defined in the 2016 Plan) as of December 31 of the immediately preceding calendar year and (ii) such lesser number of shares as may be determined by the Board of Directors or the Committee prior to January 1st of any calendar year. The shares of our Common Stock covered by the 2016 Plan may be treasury shares or authorized but unissued shares.
Any shares subject to an award under the 2016 Plan that expires, is cancelled or forfeited or is settled for cash shall, to the extent of such expiration, cancellation, forfeiture or cash settlement, remain in the pool of shares available for grant under the 2016 Plan. The following shares will, however, continue to be charged against the foregoing maximum share limitations and will not again become available for grant: (i) shares tendered by the participant or withheld by us in payment of the purchase price of an Option, (ii) shares tendered by the participant or withheld by us to satisfy any tax withholding obligation with respect to an Option of SAR, (iii) shares subject to a SAR that are not issued in connection with the settlement of the SAR upon its exercise and (iv) shares repurchased by us with proceeds received from the exercise of a stock option issued under the 2016 Plan.
Types of Awards
The 2016 Plan allows us to grant stock options, stock appreciation rights (“SARs”), restricted stock, restricted stock units and other stock-based awards. The Committee may provide that the vesting or payment of any award will be subject to the attainment of certain performance objectives established by the Committee, in addition to completion by the plan participant of a specified period of service. The Committee may amend the terms of any award previously granted, but no amendment may materially impair the rights of any participant with respect to an outstanding award without the participant’s consent, unless such amendment is necessary to comply with applicable laws or stock exchange rules.
Stock Options
Stock options granted under the 2016 Plan may be either incentive stock options (“ISOs”), which are specifically designated as such for purposes of compliance with Section 422 of the Internal Revenue Code of 1986, as amended (the “Code”), or non-statutory stock options (“NSOs”). Options will vest as determined by the Committee, subject to statutory limitations regarding the maximum term of ISOs and the maximum value of ISOs that by vest in a single year. The exercise price of options may not be less than the fair market value of our Common Stock on the date of grant, which, if our shares or not readily tradable on an established securities market will be determined by the Committee as the result of a reasonable application of a reasonable valuation method that satisfies the requirements of Section 409A of the Code. The exercise price must be paid in full at the time of exercise and may be paid in cash or such other manner as permitted by the Committee, including by withholding shares issuable upon exercise or by delivery of shares already owned by a participant. Although not necessarily indicative of fair market value, the closing price of a share of our common stock on the Nasdaq Capital Market on June 16, 2022 was $1.33 per share.
Stock Appreciation Rights
SARs provide for payment to the participant of all or a portion of the excess of the fair market value of a specified number of shares of our Common Stock on the date of exercise over a specified exercise price, which may not be less than the fair market value of our Common Stock on the date of grant. Payment may be made in cash or shares of our Common Stock or a combination of both, as determined by the Committee.
Restricted Stock
Restricted stock awards are awards of shares of our Common Stock that are subject to vesting conditions, and the corresponding lapse or waiver of forfeiture conditions and other restrictions, based on such factors and occurring over such period of time as the Committee may determine.
Restricted Stock Units
Restricted stock units provide a participant with the right to receive, in cash or shares of our Common Stock or a combination of both, the fair market value of a specified number of shares of our Common Stock, and will be subject to such vesting and forfeiture conditions and other restrictions as the Committee determines.
Other Stock-Based Awards
The Committee may grant other awards under the 2016 Plan that are valued by reference to and/or payable in whole or in part in shares of our Common Stock.
Terms of Awards and Plan Provisions
Substitute Awards
Awards may be granted under the 2016 Plan in substitution for awards granted by another entity acquired by our company or with which our company combines. The terms and conditions of these substitute awards will be comparable to the terms of the awards replaced and may therefore differ from the terms and conditions otherwise set forth in the 2016 Plan. Shares subject to substitute awards will not count against the 2016 Plan share reserve.
Repricing of Awards
The Committee may not reduce the exercise price of stock options or SARs granted under the 2016 Plan, exchange outstanding stock options or SARs with new stock options or SARs with a lower exercise price or a new full value award, repurchase underwater stock options or SARs or take any other action that would constitute a “repricing,” unless such action is first approved by our stockholders.
Transferability of Awards
Except as noted below, during the lifetime of a person to whom an award is granted, only that person, or that person’s legal representative, may exercise an option or SAR, or receive payment with respect to performance units or any other award. No award may be sold, assigned, transferred, exchanged or otherwise encumbered other than to a successor in the event of a participant’s death or pursuant to a qualified domestic relations order. However, the Committee may provide that awards, other than incentive stock options, may be transferable to members of the participant’s immediate family or to one or more trusts for the benefit of such family members or partnerships in which such family members are the only partners, if the participant does not receive any consideration for the transfer.
Termination of Service
Unless otherwise provided in an award agreement, upon termination of a participant’s service with us, all unvested and unexercisable portions of the participant’s outstanding awards will immediately be forfeited. If a participant’s service with us terminates other than for cause (as defined in the 2016 Plan), death or disability, the vested and exercisable portions of the participant’s outstanding stock options and SARs generally will remain exercisable for 90 days after termination. If a participant’s service terminates due to death or disability, the vested and exercisable portions of the participant’s outstanding stock options and SARs generally will remain exercisable for one year after termination. Upon termination for cause, all unexercised stock options and SARs will be forfeited.
Withholding
The 2016 Plan permits us to withhold from cash awards, and to require a participant receiving Common Stock under the 2016 Plan to pay us in cash, an amount sufficient to cover any required withholding taxes. In lieu of cash, the Committee may permit a participant to cover withholding obligations through a reduction in the number of shares delivered to such participant or a surrender of shares then owned by the participant.
Change in Control
If a change in control (as defined in the 2016 Plan) that involves a corporate transaction (as defined in the 2016 Plan) occurs and any outstanding award is continued, assumed or replaced by our Company or the surviving or successor entity in connection with such change in control, and if within 12 months after the change in control a participant’s employment or other service is terminated without cause or with good reason (as defined in the 2016 Plan), then (i) each of the participant’s outstanding options and SARs will become exercisable in full, and (ii) each of the participant’s unvested full value awards will fully vest. If any outstanding award is not continued, assumed or replaced in connection with such change in control, then the same consequences as specified in the previous sentence with respect to a termination of employment or other service will occur in connection with a change in control unless and to the extent the Committee elects to terminate such award in exchange for a payment in an amount equal to the intrinsic value of the award (or, if there is no intrinsic value, the award may be terminated without payment). The Committee may, in its discretion, take such other action as it deems appropriate with respect to outstanding awards for a change in control not involving a corporate transaction or may generally provide for different circumstances upon any change in control in an individual award agreement.
Adjustment of Awards
In the event of an equity restructuring, such as a stock dividend or stock split, that affects the per share value of our Common Stock, the Committee will make appropriate adjustment to: (i) the number and kind of securities reserved for issuance under the 2016 Plan, (ii) the number and kind of securities subject to outstanding awards under the 2016 Plan, (iii) the exercise price of outstanding options and SARs, and (iv) any maximum limitations prescribed by the 2016 Plan as to grants of certain types of awards. The Committee may also make similar adjustments in the event of any other change in our company’s capitalization, including a merger, consolidation, reorganization or liquidation.
Amendment and Termination
The 2016 Plan has a term of ten years from its effective date, or the earlier termination of the 2016 Plan by our Board of Directors. Our Board may amend the 2016 Plan at any time, but no amendment may materially impair the rights of any participant with respect to outstanding awards without the participant’s consent. Stockholder approval of any amendment of the 2016 Plan will be obtained if required by applicable law or the rules of any securities exchange on which our Common Stock may then be listed. Awards that are outstanding on the 2016 Plan’s termination date will remain in effect in accordance with the terms of the 2016 Plan and the applicable award agreements.
2010 Equity Incentive Plan
We assumed Cancer Prevention’s 2010 Equity Incentive Plan (the “Assumed Plan”) in connection with our acquisition of Cancer Prevention in June 2022. As of June 16, 2022, options to purchase 1,596,754 shares of common stock remained outstanding under the Assumed Plan with a weighted average price of $0.35 per share.
DILUTION
If you purchase shares in this offering your interest will be diluted immediately to the extent of the difference between the exercise price of $4.54 per share and the as adjusted net tangible book value per share of our common stock. Net tangible book value dilution per share to new investors represents the difference between the amount per share paid by purchasers in this offering and the as adjusted net tangible book value per share of common stock.
Our net tangible book value as of March 31, 2022 was approximately $6.6 million or approximately $0.49 per share. Net tangible book value per share represents our total tangible assets less total tangible liabilities, excluding goodwill and customer relationship intangibles, divided by the number of shares of common stock outstanding as of March 31, 2022. The Company had no intangible assets as of the same date.
After giving effect to the issuance of shares of our common stock upon exercise of the Warrants at a public offering price of $4.54 per share, our as adjusted net tangible book value as of March 31, 2022, would have been $17.5 million, or $1.10 per share. This represents an immediate increase in as adjusted net tangible book value of approximately $0.61 per share to our existing stockholders, and an immediate dilution of $3.44 per share to purchasers of shares in this offering, as illustrated in the following table:
|
Offering price per share
|
|
|
|
|
|
$ |
4.54 |
|
|
Net tangible book value per share as of March 31, 2022
|
|
$ |
0.49 |
|
|
|
|
|
|
Increase per share attributable to new investors
|
|
$ |
0.61 |
|
|
|
|
|
|
As adjusted net tangible book value per share after this offering
|
|
|
|
|
|
$ |
1.10 |
|
|
Dilution per share to new investors in the offering
|
|
|
|
|
|
$ |
3.44 |
|
The number of shares of our common stock outstanding before and after this offering is based on 13,449,117 shares of our common stock outstanding as of March 31, 2022, and excludes:
| |
●
|
2,433,789 shares issuable upon the exercise of warrants sold pursuant to the Registration Statement, which included all of the Warrants;
|
| |
●
|
2,444,136 shares of common stock issuable upon the exercise of outstanding stock options at a weighted average exercise price of $5.76 per share;
|
| |
●
|
5,395 shares issuable upon the vesting of restricted units;
|
| |
●
|
2,019,776 additional shares of common stock reserved and available for future issuances under our 2016 Stock Option Plan, as amended and restated;
|
| |
●
|
2,675,712 shares of common stock issuable upon exercise of stock purchase warrants not relating to this offering at a weighted average exercise price of $4.63 per share;
|
| |
●
|
7,319,533 shares of common stock, up to 1,596,754 additional shares of common stock issuable upon the exercise of stock options at a weighted average price of $0.35 per share, and up to 338,060 additional shares of common stock issuable upon exercise of stock purchase warrants at a weighted average exercise price of $4.145 per share, in each case issued after March 31, 2022, as consideration for the acquisition of Cancer Prevention.
|
DIVIDEND POLICY
We have never declared or paid cash dividends on our capital stock. Following the completion of this offering, we intend to retain our future earnings, if any, to finance the further development and expansion of our business and do not expect to pay cash dividends in the foreseeable future. Payment of future cash dividends, if any, will be at the discretion of our Board of Directors after taking into account various factors, including our financial condition, operating results, current and anticipated cash needs, outstanding indebtedness, plans for expansion and restrictions imposed by lenders, if any.
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The disclosure appearing in Part II, Item 7, of our annual report on Form 10-K for the year ended December 31, 2021, and Part I, Item 2, of our quarterly report on Form 10-Q for the quarter ended March 31, 2022 is hereby incorporated by reference in their entirety. The Company is eligible to incorporate this information by reference pursuant to General Instruction VII of Form S-1.
FINANCIAL STATEMENTS
Our audited financial statements for the years ended December 31, 2021 and December 31, 2022, appearing in our annual report on Form 10-K for the year ended December 31, 2021, and our unaudited financial statements for the three months ended March 31, 2021 and March 31, 2022, appearing in our quarterly report on Form 10-Q for the quarter ended March 31, 2022 are hereby incorporated by reference in their entirety. The Company is eligible to incorporate this information by reference pursuant to General Instruction VII of Form S-1.
BUSINESS
As used in this report, unless specifically indicated, the terms “Panbela”,” the “Company,” “we,” “us,” “our” and similar references refer to Panbela Therapeutics, Inc. and its wholly owned subsidiaries, Panbela Therapeutics Pty Ltd., Canary Merger Holdings, Inc, Canary Merger Subsidiary I, Inc. and Canary Merger Subsidiary II, Inc. Panbela Therapeutics, Inc. was incorporated under the laws of the State of Delaware in 2011. The term “common stock” refers to our common stock, par value $0.001 per share.
Business Overview
Panbela is a clinical stage biopharmaceutical company developing disruptive therapeutics for the treatment of patients with urgent unmet medical needs. The Company’s lead assets are SBP-101 and Flynpovi. SBP-101, is a proprietary polyamine analogue designed to induce polyamine metabolic inhibition (“PMI”), a metabolic pathway of critical importance in multiple tumor types. Many tumors require greatly elevated levels of polyamines to support their growth and survival. SBP-101 has demonstrated encouraging activity against metastatic disease in a clinical trial of patients with pancreatic cancer. The efficacy and safety results demonstrated in our completed phase I clinical trial of SBP-101 in combination with gemcitabine and nab-paclitaxel in the first line treatment of metastatic pancreatic cancer provides support for the current randomized, double-blind, placebo controlled phase II/III study of SBP-101 in combination with gemcitabine and nab-paclitaxel in patients previously untreated for metastatic pancreatic cancer. We believe that SBP-101, if successfully developed, may represent a novel approach that effectively treats patients with pancreatic cancer and could become a dominant product in that market. Only three first-line treatment combinations, a single maintenance treatment for a subset (3-7%) of patients, and one second-line drug have been approved by the FDA for pancreatic cancer in the last 25 years.
On June 15, 2022 Panbela acquired Cancer Prevention Pharmaceuticals, Inc. (“Cancer Prevention”), a private clinical stage company developing therapeutics to reduce the risk and recurrence of cancer and rare diseases, which added the Company’s second lead asset, an investigational new drug product, Flynpovi which is a combination of the polyamine synthesis inhibitor eflornithine and the non-steroidal anti-inflammatory drug sulindac. Eflornithine is an enzyme-activated, irreversible inhibitor of the enzyme ornithine decarboxylase, the first and rate-limiting enzyme in the biosynthesis of polyamines. Sulindac facilitates the export and catabolism of polyamines. We believe the investigational drug is unique in that it is designed to treat the risk factors (e.g., polyps) that are hypothesized to lead to Familial Adenomatous Polyposis (FAP) surgeries and colon cancer and therefore may have the ability to prevent various types of colon cancer. Flynpovi has received orphan drug designation status for FAP in the United States and Europe. There are no currently approved pharmaceutical therapies for FAP.
Both of the principal active ingredients in our investigational drug candidate Flynpovi (eflornithine and sulindac) have been previously approved individually but not in combination for other uses by the U.S. Food and Drug Administration (“FDA”) and have shown limited side effects at the dosages utilized in FAP studies. Eflornithine has never been approved in an oral form, is not on the market in any systemic dosage form, and is not available in any generic form. The combination of eflornithine and sulindac is delivered in an oral form, to which we have exclusive license rights to commercialize from the Arizona Board of Regents of the University of Arizona (the “University of Arizona”). This combination showed promising results in a National Cancer Institute (“NCI”) supported randomized, placebo-controlled Phase II/III clinical trial to prevent recurrent colon adenomas, particularly high-risk pre-cancerous polyps. These results led to a Phase III program in FAP, and a Phase III program to study colon cancer risk reduction in partnership with the Southwest Oncology Group (SWOG) and the NCI.
Additional programs are evaluating eflornithine as a single agent tablet (CPP-1X) or high dose powder sachet (CPP-1X-S) for several indications including prevention of gastric cancer, treatment of neuroblastoma and recent onset Type 1 diabetes. Preclinical studies as well as Phase 1 or Phase 2 investigator-initiated trials suggest that eflornithine treatment is well tolerated and has potential activity.
Holding Company Reorganization
Effective June 15, 2022, Panbela became a successor issuer to Panbela Research, Inc. (formerly known as Panbela Therapeutics, Inc., the “Predecessor”) pursuant to a holding company reorganization pursuant to which the Predecessor became a direct, wholly-owned subsidiary of Panbela. Panbela became a successor issuer to the Predecessor by operation of Rule 12g-3(a) promulgated under the Securities Exchange Act of 1934, as amended the (“Exchange Act”).
Cancer Prevention Acquisition
On June 15, 2022, Panbela acquired Cancer Prevention via merger for consideration consisting of (a) 6,587,576 shares of common stock, (b) 731,957 shares of common stock that remained subject to a holdback escrow (as defined in the Merger Agreement), (c) replacement options to purchase up to 1,596,754 shares of common stock at a weighted average purchase price of $0.35 per share, and (d) replacement warrants to purchase up to 338,060 shares of common stock at a weighted average purchase price of $4.145 per share, and post-closing contingent payments up to a maximum of $60 million, subject to satisfaction of milestones.
Clinical Trials
SBP-101
In August 2015, the FDA accepted our Investigational New Drug (“IND”) application for our SBP-101 product candidate. We have completed an initial clinical trial of SBP-101 in patients with previously treated locally advanced or metastatic pancreatic cancer. This was a Phase I, first-in-human, dose-escalation, safety study. Between January 2016 and September 2017, we enrolled twenty-nine patients into six cohorts, or groups, in the dose-escalation phase of our Phase I trial. Twenty-four of the patients received at least two prior chemotherapy regimens. No drug-related serious adverse events occurred during the first four cohorts. In cohort five, serious adverse events (klebsiella sepsis with metabolic acidosis in one patient, renal and hepatic toxicity in one patient, and mesenteric vein thrombosis with metabolic acidosis in one patient) were observed in three of the ten patients, two of whom exhibited progressive disease at the end of their first cycle of treatment and were determined by the Data Safety Monitoring Board (“DSMB”) to be Disease Limiting Toxicities (“DLTs”). Consistent with the study protocol, the DSMB recommended continuation of the study by expansion of cohort 4, one level below that at which DLTs were observed. Four patients were enrolled in this expansion cohort.
In addition to being evaluated for safety, 23 of the 29 patients were evaluable for preliminary signals of efficacy prior to or at the eight-week conclusion of their first cycle of treatment using the RECIST, the current standard for evaluating changes in the size of tumors. Eight of the 23 patients (35%) had Stable Disease (“SD”) and 15 of 24 (65%) had Progressive Disease (“PD”). It should be noted that of the 15 patients with PD, six came from cohorts one and two and are considered to have received less than potentially therapeutic doses of SBP-101.
By cohort, stable disease occurred in two patients in cohort 3, two patients in cohort 4 and four patients in cohort 5. The best response outcomes and best median survival were observed in the group of patients who received total cumulative doses of approximately 6 mg/kg (cohort three). Two of four patients (50%) showed SD at week eight. Median survival in this group was 5.9 months, with two patients surviving 8 and 10 months, respectively. By total cumulative dose received, five of twelve patients (42%) who received total cumulative doses between 2.5 mg/kg and 8.0 mg/kg had reductions in the CA19-9 levels, as measured at least once after the baseline assessment. Nine of these patients (67%) exceeded three months of Overall Survival (“OS”), three patients (25%) exceeded nine months of OS and two patients (17%) exceeded one year of OS and were still alive at the end of the study.
We completed enrollment of patients in our second clinical trial in December 2020. This second clinical trial was a Phase Ia/Ib study of the safety, efficacy and pharmacokinetics of SBP-101 administered in combination with two standard-of-care chemotherapy agents, gemcitabine and nab-paclitaxel. The trial included six study sites; four in Australia and two in the United States. In the Phase Ia portion of this trial, we completed enrollment during the first quarter of 2020 consisting of four cohorts with increased dosage levels of SBP-101 administered in the second and third cohorts; the fourth cohort evaluated an alternate dosing schedule. A total of 25 subjects were enrolled in four cohorts of Phase Ia. The demonstration of adequate safety in Phase Ia allowed us to immediately begin enrollment in February 2020 in the Phase Ib exploration of efficacy. By December 2020 an additional 25 subjects in Phase Ib, using the recommended dosage level and schedule determined in Phase Ia, were enrolled.
After Phase Ib enrollment was completed, some patients in the trial were noted to have complaints of serious visual adverse effects. Visual changes were not seen in the SBP-101 monotherapy study. We consulted with our DSMB and withheld the administration of SBP-101 while all other trial activities continued. In February of 2021, we also conferred with the FDA regarding our plan to withhold dosing of SBP-101. This constituted a “partial clinical hold.” In April of 2021, the FDA lifted the partial clinical hold. The Company agreed with the FDA to include in the design of all future studies the exclusion of patients with a history of retinopathy or risk of retinal detachment and scheduled ophthalmologic monitoring for all patients.
Updated, but still not final results, were presented in a poster at the American Society of Clinical Oncology - GI conference (“ASCO-GI”) in January 2022. Best response in evaluable subjects (cohorts 4 and Ib N=29) was a Complete Response (“CR”) in 1 (3%), Partial Response (“PR”) in 13 (45%), SD in 10 (34%) and PD in 5 (17%). One subject did not have post baseline scans with RECIST tumor assessments. Median Progression Free Survival (“PFS”) , now final at 6.5 months may have been negatively impacted by drug dosing interruptions to evaluate potential toxicity. Median overall survival in Cohort 4 + Phase Ib was 12.0 months when data was presented in January 2022 and is now final at 14.6 months. Two patients from cohort 2 have demonstrated long term survival. One at 30.3 months (final data) and one at 33.0 months and still alive. Seven subjects are still alive at this time, one from cohort 2 and six from cohort 4 plus Ib.
The Company announced that the ASPIRE trial, a randomized, double-blind, placebo-controlled study of SBP-101 with Gemcitabine and Nab-Paclitaxel versus Gemcitabine, Nab-Paclitaxel and placebo, was initiated in January of 2022. The trial is in patients with first-line metastatic pancreatic ductal adenocarcinoma. The trial is designed as a Phase II/III randomized trial, with a primary endpoint of overall survival. The design includes a phase II portion for which a futility analysis after 104 progression free survival events will occur. If the futility analysis is favorable, the trial will be expanded to the phase III portion, and may serve for registration. We are intending to conduct the ASPIRE trial at leading cancer centers in the United States, Europe and the Asia-Pacific Region. It is expected that there will be approximately 60 sites and we anticipate enrollment for the phase II portion to be completed in approximately 12 months after the first subject is enrolled.
If we can successfully complete all FDA recommended clinical studies, we intend to seek marketing authorization from the FDA, the European Medicines Agency (“EMA”) (European Union), Ministry of Health and Welfare (Japan) and TGA (Australia). The submission fees may be waived when SBP-101 has been designated an orphan drug in each geographic region.
Data presented at the American Association for Cancer Research (AACR) in April 2022, demonstrated in an in vitro study evaluating cancer cell lines, SBP-101 was toxic in ovarian cancer cell lines with an average IC50 of ~1.5 μM. Efficacy of SBP-101 was further assessed in the VDID8+murine ovarian cancer model (ID8+C57Bl/6 ovarian cells overexpressing both VEGF and Defensin). Mice were treated with SBP-101 at either 24 mg/kg or 6 mg/kg alternating MWF. Both doses of SBP-101 produced a statistically significant prolongation of survival (24mg/kg p=.0049, 6 mg/kg p=.0042). There was no significant difference in response between the two SBP-101 doses. The prolonged survival was correlated with a delay in the production of ascites, the indication of tumor burden in this model. Given this data, the Company intends to proceed with a clinical development program in ovarian cancer by year-end.
FLYNPOVI
In a phase III study, the efficacy and safety of the combination of eflornithine and sulindac, as compared with either drug alone, in adults with familial adenomatous polyposis was conducted. The patients were randomly assigned in a 1:1:1 ratio to receive eflornithine, sulindac, or both once daily for up to 48 months. The primary end point, assessed in a time-to-event analysis, was disease progression, defined as a composite of major surgery, endoscopic excision of advanced adenomas, diagnosis of high-grade dysplasia in the rectum or pouch, or progression of duodenal disease. A total of 171 patients underwent randomization. Disease progression occurred in 18 of 56 patients (32%) in the eflornithine–sulindac group, 22 of 58 (38%) in the sulindac group, and 23 of 57 (40%) in the eflornithine group, with a hazard ratio of 0.71 (95% confidence interval [CI], 0.39 to 1.32) for eflornithine–sulindac as compared with sulindac (P = 0.29) and 0.66 (95% CI, 0.36 to 1.23) for eflornithine–sulindac as compared with eflornithine. Adverse and serious adverse events were similar across the treatment groups. In a post-hoc analysis, none of the patients in the combination arm progressed to a need for lower gastrointestinal (LGI) surgery for up to 48 months compared with 7 (13.2%) and 8 (15.7%) patients in the sulindac and eflornithine arms. These data corresponded to risk reductions for the need for LGI surgery approaching 100% between combination and either monotherapy with HR = 0.00 (95% CI, 0.00–0.48; p = 0.005) for combination versus sulindac and HR = 0.00 (95% CI, 0.00–0.44; p = 0.003) for combination versus eflornithine. Given the statistical significance of the LGI group, a new drug application (NDA) was filed with the FDA. As the study failed to meet the primary endpoint, and the NDA was based on the results of an exploratory analysis, a complete response letter was issued. To address this deficiency concern, the Company must submit the results of one or more adequate and well-controlled clinical trials which demonstrate an effect on a clinical endpoint. As the result of an existing North American license agreement, the FAP registration trial is fully funded and is scheduled to begin in the first-half of 2023.
CPP-1X-S/CPP-1X
Additionally, there are trials evaluating CPP-1X-S in relapsed refractory neuroblastoma supported by the childrens’ oncology group (COG) and NCI (ongoing) and STK11 mutation patients with non-small cell lung cancer scheduled to begin this year. For CPP-1X-T, a phase II trial in Type I onset diabetes is scheduled to begin this year in collaboration with Indiana University and the Juvenile Diabetes Research Foundation (JDRF).
Recent Developments
The combined entity resulting from Panbela’s acquisition of Cancer Prevention have an expanded pipeline; areas of initial focus include familial adenomatous polyposis (FAP), first-line metastatic pancreatic cancer, neoadjuvant pancreatic cancer, colorectal cancer prevention and ovarian cancer. The combined development programs have a steady cadence of catalysts with programs ranging from pre-clinical to registration studies.
Through December 31, 2021, we had:
| |
●
|
secured an orphan drug designation from the FDA;
|
| |
●
|
submitted and received acceptance from the FDA for an IND application;
|
| |
●
|
received acceptance of a Clinical Trial Notification from the Australian Therapeutic Goods Administration;
|
| |
●
|
completed a Phase Ia monotherapy safety study of SBP-101in the treatment of patients with metastatic pancreatic ductal adenocarcinoma;
|
| |
●
|
received “Fast Track” designation from the FDA for SBP-101 for metastatic pancreatic cancer;
|
| |
●
|
completed enrollment and released interim results in our second trial a Phase Ia /Ib clinical study of SBP-101, a first-line study with SBP-101 given in combination with a current standard of care in patients with pancreatic ductal adenocarcinoma who were previously untreated for metastatic disease; a total of 50 subjects were enrolled in this study, 25 in the Phase Ia and 25 in the Phase Ib or expansion phase;
|
| |
●
|
secured a two year research agreement with Johns Hopkins School of Medicine led by Professor Robert Casero an internationally recognized researcher in polyamine biology;
|
| |
●
|
completed process improvement measures expected to be scalable for commercial use and received issue notification for a patent covering this new shorter synthesis of SBP-101;
|
| |
●
|
initiated a randomized, double-blind, placebo controlled study with SBP-101 given in combination with gemcitabine and nab-paclitaxel in patients with pancreatic ductal adenocarcinoma who are previously untreated for metastatic disease;
|
| |
●
|
Completed preclinical evaluation of SBP-101 for use as neoadjuvant therapy in resectable pancreatic cancer prior to surgery; and
|
| |
●
|
Obtained early, preclinical, indication of tumor growth inhibition activity in ovarian cancer.
|
Pancreatic Cancer
Pancreatic cancer afflicts approximately 140,000 people in Europe (GLOBOCAN 2021, Global Cancer Observatory/World Health Organization), approximately 60,000 people in the United States annually (https://seer.cancer.gov/statfacts/html/pancreas.html), and 293,000 people worldwide – excluding Europe and United States (GLOBOCAN 2021). It has been identified as the fourth leading cause of death from cancer in Europe (GLOBOCAN 2021) and the third leading cause of death from cancer in the United States (SEER Cancer Statistics Factsheets 2021). On average Pancreatic Ductal Adenocarcinoma (“PDA”) represents approximately 95% of all pancreatic cancers diagnosed in given calendar year. Considering that the median overall survival for previously untreated patients with good performance status is between 8.5 months (Von Hoff 2013) and 11.1 months (Conroy 2011) with the two most commonly available treatment regimens, effective treatment for PDA has remained a major unmet medical need.
Pancreatic cancer is generally not diagnosed early because the initial clinical signs and symptoms are vague and non-specific. The most common presenting symptoms include weight loss, epigastric (upper central region of the abdomen) and/or back pain, and jaundice. The back pain is typically dull, constant, and of visceral origin radiating to the back, in contrast to the epigastric pain which is vague and intermittent. Less common symptoms include nausea, vomiting, diarrhea, anorexia, and new onset diabetes (which can be an early signal) or glucose intolerance (Hidalgo 2010).
Surgery remains the only treatment option with curative intent, although only about 20% of patients are candidates for surgical resection at the time of the diagnosis. Patients who undergo radical surgery still have a limited survival rate, averaging 23 months (Macarulla T, et al Clin Transl Oncol 2017).
For the minority of patients who present with resectable disease, surgery is the treatment of choice. Depending on the location of the tumor the operative procedures may involve cephalic pancreatoduodenectomy, referred to as a “Whipple procedure” distal pancreatectomy or total pancreatectomy. Pancreatic enzyme deficiency and diabetes are frequent complications of both the disease and these surgical procedures. Up to 70% of patients with pancreatic cancer present with biliary obstruction that can be relieved by percutaneous or endoscopic stent placement. However, even if the tumor is fully resected, the outcome in patients with pancreatic cancer has been disappointing (Hidalgo 2010, Seufferlein 2012). Post-operative administration of chemotherapy improved progression-free and overall survival in three large randomized clinical trials (Hidalgo 2010), but median post-surgical survival in patients treated in all three trials was similar, only 20-22 months. Pre-operative (neo-adjuvant) chemotherapy is of increasing interest, with the goal of improved successful resections and long-term outcomes.
For patients who present with unresectable, locally advanced or metastatic disease, which represent a majority of PDA patients, management options range from chemotherapy alone to combined forms of treatment with radiation therapy and chemotherapy. However, due to the increased toxicity of combined treatment, randomized trials of such combined regimens have had low enrollment, precluding a firm conclusion as to any advantage of adding radiation to chemotherapy (Hidalgo 2010).
Gemcitabine was the first chemotherapeutic agent approved for the treatment of patients with PDA in the modern regulatory era, providing a median survival duration of 5.65 months (Burris 1997). Gemcitabine monotherapy was the standard of care for patients with metastatic pancreatic cancer until combination therapy with gemcitabine plus erlotinib (Tarceva®) was shown to increase median survival by two weeks. This modest benefit was tempered by a significant side effect profile and high cost, limiting its adoption as a standard treatment regimen. Subsequently, the multidrug chemotherapy combination FOLFIRINOX, was shown to provide a median survival benefit of 4.3 months (OS = 11.1 months) over gemcitabine alone (6.8 months), but its significant side effect profile limits the regimen to select patients with a good performance status and often requires supplementation with WBC growth factor therapy. Nab-paclitaxel (Abraxane®) received marketing authorization for use in combination with gemcitabine (FDA approved 2013) after showing an increase in overall survival of seven weeks compared to gemcitabine alone (Von Hoff 2013). Thus, combination therapies have demonstrated a modest survival benefit compared to gemcitabine alone as summarized in the table below (Thota 2014).
Current First-Line Treatment Approaches: Survival & Toxicity Profiles Across Three Major Positive Clinical Trials
| |
|
Gemcitabine vs.
Gemcitabine/Erlotinib
Phase 3 trial
|
|
|
ACCORD 11 Trial
|
|
|
Metastatic Pancreatic
Adenocarcinoma Clinical
Trial (MPACT)
|
|
| |
|
Gemcitabine
|
|
|
Gemcitabine/
Erlotinib
|
|
|
Gemcitabine
|
|
|
FOLFIRINOX
|
|
|
Gemcitabine
|
|
|
Gemcitabine/
Nab-Paclitaxel
|
|
|
One-Year Survival
|
|
|
17% |
|
|
|
23% |
|
|
|
20.6% |
|
|
|
48.4% |
|
|
|
22% |
|
|
|
35% |
|
|
Median Overall Survival (months)
|
|
|
5.91 |
|
|
|
6.24 |
|
|
|
6.8 |
|
|
|
11.1 |
|
|
|
6.7 |
|
|
|
8.5 |
|
|
Median Progression-Free Survival (months)
|
|
|
3.55 |
|
|
|
3.75 |
|
|
|
3.3 |
|
|
|
6.4 |
|
|
|
3.7 |
|
|
|
5.5 |
|
|
Overall Response Rate
|
|
|
8% |
|
|
|
8.6% |
|
|
|
9.4% |
|
|
|
31.6% |
|
|
|
7% |
|
|
|
23% |
|
|
Toxicity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Neutropenia
|
|
|
– |
|
|
|
– |
|
|
|
21% |
|
|
|
45.7% |
|
|
|
27% |
|
|
|
38% |
|
|
Febrile neutropenia
|
|
|
– |
|
|
|
– |
|
|
|
1.2% |
|
|
|
5.4% |
|
|
|
1% |
|
|
|
3% |
|
|
Thrombocytopenia
|
|
|
– |
|
|
|
– |
|
|
|
3.6% |
|
|
|
9.1% |
|
|
|
9% |
|
|
|
13% |
|
|
Diarrhea
|
|
|
2% |
|
|
|
6% |
|
|
|
1.8% |
|
|
|
12.7% |
|
|
|
1% |
|
|
|
6% |
|
|
Sensory neuropathy
|
|
|
– |
|
|
|
– |
|
|
|
0% |
|
|
|
9% |
|
|
|
1% |
|
|
|
17% |
|
|
Fatigue
|
|
|
15% |
|
|
|
15% |
|
|
|
17.8% |
|
|
|
23.6% |
|
|
|
7% |
|
|
|
17% |
|
|
Rash
|
|
|
6% |
|
|
|
1% |
|
|
|
– |
|
|
|
– |
|
|
|
– |
|
|
|
– |
|
|
Stomatitis
|
|
|
<1% |
|
|
|
0% |
|
|
|
– |
|
|
|
– |
|
|
|
– |
|
|
|
– |
|
|
Infection
|
|
|
17 |
|
|
|
16% |
|
|
|
– |
|
|
|
– |
|
|
|
– |
|
|
|
– |
|
Source: Thota R et al., Oncology 2014; Jan 28(1):70–74
Other drugs are currently under investigation, but none have received marketing authorization as a first-line treatment of PDA since the approval of Abraxane. Lynparza®, (olaparib) was approved in December 2019 for maintenance therapy of patients with deleterious or suspected deleterious germline BRCA-mutated (gBRCAm) metastatic pancreatic adenocarcinoma whose disease has not progressed on at least 16 weeks of a first-line platinum-and chemotherapy regimen.
Ovarian Cancer
Worldwide Ovarian Cancer has annual incidence of approximately 314,000 and annual deaths of approximately 207,000 (Globocan 2020). In the United States Ovarian represents approximately 1% of all new cancer cases at approximately 21,000 and the five year survival rate for metastatic disease is approximately 29% (SEER fact sheet Ovarian 2022). According to the American Cancer Society, ovarian cancer is the fifth leading cause of cancer deaths among women, accounting for more deaths than any other cancer of the female reproductive system.
Nearly 70 % of the patients are diagnosed with advanced-stage due to the failure of screening methods for detecting early-stage disease (Giornelli. 2016; Partridge et al. 2009; Bast et al. 2007; Gohagan et al. 2000; Chudecka-Głaz 2015). Thus, most patients will relapse within the first 2 years after diagnosis, even after an optimal primary cytoreductive surgery and six cycles of the standard adjuvant chemotherapy with carboplatin/paclitaxel.
The second line chemotherapy depends mainly on the disease-free interval (“DFI”) (time between completion of first line chemotherapy and clinical relapse); or progression-free interval (“PFI”) (time between the last chemotherapy given for relapsed disease and progression). There are three classifications: Platinum-refractory/resistant with relapse during platinum treatment (refractory) or with a DFI/PFI <6 months (resistant), Platinum-sensitive relapse occurring >12 m of last platinum-based chemotherapy, or partially sensitive to platinum with disease-free survival (“DFS”)/ progression free survival (“PFS”) between 6 and 12 months from the last platinum-based chemotherapy.
According to Pignata et al. 2017, in platinum-sensitive patients, treatment with platinum-based combinations is associated with a PFS advantage compared with single agents or non-platinum combinations. For patients with partially sensitive relapse (PFI between 6 and 12 months), two options are available: platinum doublets or non-platinum therapy (single agent or combination). Last, patients with resistant or refractory relapse (PFI < 6 months) disease there are few options. For these patients, monotherapy with a non-platinum drug or participation in clinical trials is indicated.
Proprietary Technology
Function and Characteristics of Polyamines
Polyamines are metabolically distinct entities within human cells that bind to and facilitate DNA replication, RNA transcription and processing, and protein (such as pancreatic enzymes) synthesis. Human cells contain three essential and naturally occurring polyamines – putrescine, spermidine, and spermine. Polyamines perform many functions necessary for cellular proliferation, apoptosis and protein synthesis. The critical balance of polyamines within cells is maintained by several enzymes such as ornithine decarboxylase (“ODC”) and spermidine/spermine N1 acetyl transferase (“SSAT”). All of these homeostatic enzymes are short-lived, rapidly inducible intracellular proteins that serve to regulate native polyamine pools tightly and continuously. These enzymes constantly maintain polyamines within a very narrow range of concentration inside the cell.
Polyamine metabolism and cancer
Polyamines are required for cell proliferation. It is believed that many cancers, especially oncogene-driven cancers, might be sensitive to interference with polyamine metabolism. Consequently, with the loss of growth control in cancer cells, the transformed cells may be more sensitive to polyamine depletion than normal cells. Thus, the polyamine metabolic pathway is a rational target for therapeutic intervention (Casero 2018).
Polyamines are important modulators of the immune response, particularly in the tumor microenvironment where they are found in high concentrations. The inhibitory activity, most notably with spermine, of T-cells, monocytes, and macrophages, is an effective immune suppressant. This activity suggests that excess polyamines, especially spermine, may insulate the tumor microenvironment from immune cells. SBP-101 is a synthetic analogue of spermine, which is believed to reduce endogenous polyamine production. Therefore, there is a potential for SBP-101 to recondition the tumor microenvironment and act as a sensitizing agent for immune-oncology drugs such as checkpoint inhibitors.
Polyamine Analogue
Many tumors, including pancreatic cancer, display an increased uptake rate of polyamines. Polyamine analogues such as SBP-101 are structurally similar to naturally occurring polyamines and are recognized by the cell’s polyamine uptake system, allowing these compounds to gain ready entrance to the cell. We believe that pancreatic acinar cells, because of their extraordinary protein synthesis capacity, exhibit enhanced uptake of polyamines and polyamine analogues such as SBP-101. Because of this preferential uptake by pancreatic acinar cells, polyamine analogues such as SBP-101 disrupt the cell’s polyamine balance and biosynthetic network, and induce programmed cell death, or apoptosis, via processes including caspase 3 activation and poly ADP ribose polymerase (PARP) cleavage. Proof of concept has been demonstrated in multiple human pancreatic cancer models, both in vivo and in vitro, that pancreatic ductal adenocarcinoma exhibits sensitivity to SBP-101.
SBP-101
SBP-101 is a proprietary polyamine analogue, which we believe accumulates in the exocrine pancreas acinar cells due to its unique chemical structure. SBP-101 was discovered and extensively studied by Professor Raymond J. Bergeron at the University of Florida College of Pharmacy.
As laboratory studies suggest, the primary mechanism of action for SBP-101 has been demonstrated to include the enhanced uptake of the compound in the exocrine pancreas, therefore, pancreatic cancer was logical for the initial development of this compound. Sufficiently high dosing in animal models leads to correspondingly depressed levels of native polyamines, with caspase 3 activation, PARP cleavage and apoptotic destruction (programmed cell death) of the exocrine pancreatic acinar and ductal cells without an inflammatory response. Importantly, pancreatic islet cells, which secrete insulin, are structurally and functionally dissimilar to acinar cells and are not impacted by SBP-101. In animal models at two independent laboratories, SBP-101 has demonstrated significant suppression of transplanted human pancreatic cancer cells, including metastatic pancreatic cancer growth. See “Proof of Principle” below.
We believe that SBP-101 exploits the natural affinity of the exocrine pancreas, the liver and kidney, and pancreatic ductal adenocarcinoma cells while leaving the insulin-producing islet cells unharmed. Most current cancer therapies, including chemotherapy, radiation, and surgery are associated with significant side effects that further reduce the patient’s quality of life. However, based on data evaluated from clinical studies to date, we believe that the adverse effects of SBP-101 in causing bone marrow suppression or peripheral neuropathy do not overlap with or exacerbate those seen with typical chemotherapy options. The dose-limiting toxicities observed in cohort five of our first Phase I study, as noted below, were not observed at lower doses and are not expected to overlap with the adverse events of bone marrow suppression and peripheral neuropathy commonly associated with standard chemotherapy. The dose and dosing schedule evaluated in the expansion phase of the recently completed Phase Ia/Ib is below the maximum tolerable dose (‘MTD”) and at this dose level, neither the exocrine nor the endocrine human pancreas is expected to be affected by SBP-101, resulting in no treatment impact on pancreatic enzyme or insulin levels. This dose level and dosing schedule in the new ASPIRE trial will be the same as evaluated in the expansion phase of the 1a/Ib study.
Proof of Principle
SBP-101 has been tested and found effective in reducing pancreatic tumor growth in multiple separate in vivo models of human pancreatic cancer. SBP-101 was used to treat mice subcutaneously implanted with human pancreatic cancer cell line PANC-1 tumor fragments. A dose-response for efficacy was demonstrated with a 26 mg/kg daily injection resulting in near complete suppression of the transplanted tumor.
A separate orthotopic xenograft study (direct implant of human tumor cells into the pancreas of the mouse) employed a particularly aggressive human pancreatic cancer cell line, L3.6pl, that is known to metastasize from the pancreas to the liver and the peritoneum in mice. Mice implanted with L3.6pl were treated with SBP-101 and the results were compared with saline-treated control mice, with mice treated with gemcitabine alone (Gemzar®, the then current “gold standard” treatment), and the combination of both drugs. SBP-101 significantly reduced tumor volume compared to gemcitabine alone and the control group, but the combination of SBP-101 and gemcitabine was significantly better than gemcitabine alone as shown in Figure 1.
Figure 1. L3.6pl Orthotopic Xenograft Study – Mean (±SD) Tumor Volume after Treatment with SBP-101, Gemcitabine (Gemzar®) or Both
Source: Study101-Biol-101-001
The potential for SBP-101 as an effective therapy for pancreatic cancer has been demonstrated in vivo by separate investigators, in different human pancreatic cancer cell lines and in three different animal models, using SBP-101 synthesized by two different routes, confirming nearly equal, and effective, murine doses of 25 and 26 mg/kg, respectively.
Additionally, when compared in vitro to existing therapies, SBP-101 produced superior results in suppressing growth of pancreatic cancer cells.
Development Plan for SBP-101
Development of SBP-101 for the pancreatic cancer indication has included a pre-clinical and a clinical phase. The pre-clinical phase, which was substantially completed during 2015, consisted of four primary components: chemistry, manufacturing and controls (“CMC”), preclinical (laboratory and animal) pharmacology studies, preclinical toxicology studies, and regulatory submissions in Australia and the United States. In Australia, a Human Research Ethics Committee (“HREC”) application was submitted with subsequent Clinical Trial Notification (“CTN”) to the Therapeutic Goods Administration (“TGA”). Preceding the Australian initiative, a similar, but considerably more extensive, preclinical package has been submitted to and accepted by the FDA in support of an IND application. Our initial clinical trial in previously treated patients with locally advanced or metastatic pancreatic cancer was a Phase I, first-in-human, dose-escalation, safety study conducted at clinical sites in both Australia and the United States. We engaged expert clinicians who treat pancreatic cancer at major cancer treatment centers in Melbourne and Adelaide, Australia as well as the Mayo Clinic Scottsdale and HonorHealth in Scottsdale, Arizona. These Key Opinion Leaders, with proven performance in pancreatic cancer studies, enthusiastically agreed to participate as investigators for our Phase I First-in-Human study.
Enrollment in our initial Phase I safety trial of SBP-101 in previously treated pancreatic cancer patients commenced in January 2016 and was completed in September 2017. This study was a dose-escalation study with 8-week treatment/observation cycles at each dose level. Results from this trial are discussed in Clinical Development – Pancreatic Cancer, Phase I Clinical Trial Design and Completion (SBP-101 Monotherapy) below.
We completed enrollment of patients in our second clinical trial in December 2020. This second clinical trial was a Phase Ia/Ib study of the safety, efficacy and pharmacokinetics of SBP-101 administered in combination with two standard-of-care chemotherapy agents, gemcitabine and nab-paclitaxel. A total of 25 subjects were enrolled in four cohorts of Phase Ia. Demonstration of adequate safety in Phase Ia allowed us to immediately begin enrollment in February 2020 in the Phase Ib exploration of efficacy. We completed enrollment of an additional 25 subjects in Phase Ib using the recommended dosage level and schedule determined in cohort 4 of Phase Ia. Safety and interim efficacy results from this trial are discussed in Clinical Development – Pancreatic Cancer, Phase Ia/Ib Clinical Trial Design and Interim Results (First Line Combination Therapy) below.
In January of 2022 we initiated our third clinical trial. This new trial is a randomized, double blind, placebo controlled study of safety and efficacy of SBP-101 administered in combination with two standard-of-care chemotherapy agents, gemcitabine and nab-paclitaxel. Trial design and expected timing are discussed in Clinical Development – Pancreatic Cancer, Phase II/III Clinical Trial design and anticipated timing (ASPIRE trial).
In addition, we are exploring SBP-101 for neoadjuvant treatment in appropriate pancreatic cancer patients. There is also preclinical data to suggest that SBP-101 may have potential therapeutic uses for cancers other than pancreatic. In February 2021, we entered into a research agreement with the Johns Hopkins University School of Medicine. The collaboration has focused on the further development of Panbela’s investigative agent SBP-101, including activity in cell lines outside of pancreatic cancer, biomarkers informing diagnostics and potential combination with checkpoint inhibitors. In December of 2021, the Company announced positive preclinical data supporting the activity of SBP-101 in ovarian cancer cell lines. Further data resulting from the ongoing relationship with Johns Hopkins University School of Medicine is expected throughout 2022.
Preclinical Development
To enable IND and HREC/CTN submission and as part of our pharmacology work, we conducted plasma and urine assay development and validation in animals, in vitro metabolism studies in liver microsomes and hepatocytes, in vitro interaction studies with hepatic and renal transporters, a protein binding study, animal pharmacokinetic and metabolism/mass balance studies, and human plasma and urine assay development and validation. As a part of the pharmacology evaluation, we conducted an in vitro pharmacology screen profiling assay, a study in six human pancreatic cell lines, and studies in tumor xenograft models in mice using flank transplants of human pancreatic cancer PANC-1 tumor fragments and human pancreatic cancer BxPC-3 tumor fragments as well as human pancreatic cancer cells (L3.6pl) injected orthotopically into the pancreas of nude mice.
To meet regulatory requirements and to establish the safety profile of SBP-101, we conducted, in rodents and non-rodents, toxicology dose-ranging studies, IND-enabling GLP (good laboratory practice) toxicology studies, and genetic toxicology studies, including an Ames test. Exploratory studies in mice and rats and a GLP-compliant dog toxicology study have also been completed. The relationship between dose and exposure (pharmacokinetics) has been described for three animal species. We have also completed a preclinical Human Ether-a-go-go-related Gene (hERG) assay to detect any electrocardiographic QTc interval effects (IKr potassium ion channel testing).
In anticipation of the human potential for using SBP-101 in combination therapy with gemcitabine and/or nab-paclitaxel (Abraxane®), we also conducted appropriate nonclinical studies which confirmed the potential value of such combinations, including assessing the comparative efficacy of SBP-101, gemcitabine and nab-paclitaxel in various combinations as shown in Figure 2.
Figure 2. Evaluation of SBP-101 alone and in combination with gemcitabine and nab-paclitaxel in 6 human pancreatic cancer cell lines
Source: Baker CB et al Pancreas 2015;44(8) 1350
Note that maximum percent growth inhibition (mean ± SE) at 96 hours was observed with 10 µM SBP-101 alone and in combination with gemcitabine and/or nab-paclitaxel in six human pancreatic cancer cell lines.
We have met FDA-mandated CMC requirements with a combination of in-house expertise and contractual arrangements. Preparation of anticipated metabolites, impurities and an internal standard, as a prerequisite for analytical studies, were completed through Sponsored Research Agreements with the University of Florida and with Syngene International Ltd. (“Syngene”), a contract manufacturer. We have additional Service Agreements with Syngene for the manufacture and supply of Good Manufacturing Practice (“GMP”)-compliant SBP-101 active pharmaceutical ingredient (“API”) and for the development of synthetic process improvements. As the result of efforts at Syngene to refine the synthetic process, a new shorter synthetic process has been developed on which a patent (US 11,098,005 B2) “METHODS FOR PRODUCING (65,155)-3,8,13,18-TETRAAZAICOSANE-6, 15-DIOL” issued on Aug. 24, 2021. Multiple lots of GMP-compliant API have been prepared by Syngene and released for conversion into supply dosage form. Multiple lots of investigational product (IP or clinical trial supply), together with matching placebo has been made and tested at CURIA formerly known as Albany Molecular Research Inc. (“AMRI”) in Burlington, MA.
Pancreatic Cancer IND
Our IND application package contained the following:
| |
●
|
Investigator’s Brochure;
|
| |
●
|
Statement of general investigative plans;
|
| |
●
|
Proposed Phase I pancreatic cancer study protocol;
|
| |
●
|
Data management and statistical plan;
|
| |
●
|
Pharmacology, absorption, distribution, metabolism and excretion (or “ADME”), and toxicology data.
|
Preparation of the SBP-101 IND for pancreatic cancer required collaboration by our manufacturing, preclinical toxicology, pharmacokinetic, and metabolism experts, our regulatory affairs project management, and our in-house clinical expertise. In August 2015, the FDA accepted our application and in January 2016 we commenced patient enrollment in our first Phase I clinical trial, which was a safety and tolerability study in patients with previously treated metastatic pancreatic ductal adenocarcinoma.
Clinical Development – Pancreatic Cancer
Our clinical development in Pancreatic Cancer thus far includes:
| |
●
|
a Phase I SBP-101 Monotherapy study completed in 2017, and
|
| |
●
|
a Phase Ia/Ib SBP-101 First Line Combination Therapy study which completed enrollment in late 2020, and
|
| |
●
|
a Phase II/III Randomized, Double Blind Placebo Controlled First Line Combination Therapy study was initiated in January of 2022.
|
Details of these programs follow.
Phase I Clinical Trial Design and Completion (SBP-101 Monotherapy)
Our initial Phase I study in patients with pancreatic cancer commenced the enrollment of patients in January 2016 and enrollment was completed in September 2017. This study was a dose-escalation study with 8-week cycles of treatment/observation at each dose level.
A favorable characteristic of the pancreatic action of SBP-101 is the lack of an effect on the normal insulin-producing islet cells, and no apparent effect on human exocrine pancreatic function at current or anticipated dose levels. Preservation of islet cell function predicted the absence of diabetes as a complication of SBP-101 therapy. It is important to note that diabetes is a common co-morbidity in patients with pancreatic cancer, but it has not appeared to be an adverse effect of treatment with SBP-101. No adverse effect on exocrine pancreatic enzyme production has been observed and would be mitigated by the observation that many patients with pancreatic ductal adenocarcinoma require pancreatic enzyme replacement as a feature of their underlying disease, a complication so common that pancreatic enzyme replacement with one of several commercially available products is typically covered by United States and Australian health care plans. Patients with cystic fibrosis, chronic pancreatitis and pancreatic cancer are the populations most often treated with pancreatic enzyme replacement.
Patients in our Phase I first-in-human trial underwent regular pancreatic and hepatic enzyme evaluation and obtained periodic chest and abdominal CT follow-up. Patients were also carefully monitored for clinical signs of GI adverse events, but no new onset of exocrine pancreatic insufficiency was attributed to SBP-101 therapy.
In August 2015, the FDA accepted our IND application for our SBP-101 product candidate. We have completed an initial clinical trial of SBP-101 in patients with previously treated locally advanced or metastatic pancreatic cancer. This was a Phase I, first-in-human, dose-escalation, safety study. Between January 2016 and September 2017, we enrolled twenty-nine patients into six cohorts, or groups, in the dose-escalation phase of our Phase I trial. Twenty-four of the patients received at least two prior chemotherapy regimens. No drug-related serious adverse events occurred during the first four cohorts. In cohort five, serious adverse events (klebsiella sepsis with metabolic acidosis in one patient, renal and hepatic toxicity in one patient, and mesenteric vein thrombosis with metabolic acidosis in one patient) were observed in three of the ten patients, two of whom exhibited progressive disease at the end of their first cycle of treatment and were determined by the Data Safety Monitoring Board (“DSMB”) to be DLTs. Consistent with the study protocol, the DSMB recommended continuation of the study by expansion of cohort 4, one level below that at which DLTs were observed. Four patients were enrolled in this expansion cohort. One patient developed focal pancreatitis at the site of the primary tumor after 2.3 months, but SBP-101 was considered well tolerated below dose level five. The most common drug related adverse events were nausea, vomiting, diarrhea, injection site pain and abdominal pain, which were mostly mild, grades 1 or 2, and are symptoms common in patients with pancreatic cancer. No drug-related bone marrow toxicity or peripheral neuropathy was observed at any dose level.
In addition to being evaluated for safety, 23 of the 29 patients were evaluable for preliminary signals of efficacy prior to or at the eight-week conclusion of their first cycle of treatment using the RECIST, the current standard for evaluating changes in the size of tumors. Eight of the 23 patients (35%) had SD and 15 of 24 (65%) had PD. It should be noted that of the 15 patients with PD, six came from cohorts one and two and are considered to have received less than potentially therapeutic doses of SBP-101. We also noted that 28 of the 29 patients had follow-up blood tests measuring the Tumor Marker CA 19-9 associated with pancreatic ductal adenocarcinoma. Eleven of these patients (39%) had reductions in the CA 19-9 levels, as measured at least once after the baseline assessment. Seven of the remaining 17 patients who showed no reduction in CA 19-9 came from cohorts one and two.
By cohort, stable disease occurred in two patients in cohort 3, two patients in cohort 4 and four patients in cohort 5. The best response outcomes and best median survival were observed in the group of patients who received total cumulative doses of approximately 6 mg/kg (cohort three). Two of four patients (50%) showed stable Disease (“SD”) at week eight. Median survival in this group was 5.9 months, with two patients surviving 8 and 10 months, respectively. By total cumulative dose received, five of twelve patients (42%) who received total cumulative doses between 2.5 mg/kg and 8.0 mg/kg had reductions in the CA19-9 levels, as measured at least once after the baseline assessment. Nine of these patients (67%) exceeded three months of OS, three patients (25%) exceeded nine months of OS and two patients (17%) exceeded one year of OS and were still alive at the end of the study.
Figure 3. Evaluation of SBP 101 Phase IMono-therapy Safety Trial - Median Survival by Cohort
The absence of adverse events which could potentially overlap with adverse events typically observed in the use of conventional chemotherapeutic agents, supports the case for combination of SBP-101 with conventional chemotherapeutic agents, such as gemcitabine, nab-paclitaxel, or even FOLFIRINOX.
Phase Ia/Ib Clinical Trial Design and Interim Results (First Line Combination Therapy)
Given the life-threatening nature of pancreatic ductal adenocarcinoma, the limited efficacy of current treatment options, and the long history of failures in pancreatic ductal adenocarcinoma developmental therapeutics, we have evaluated SBP-101 expeditiously as noted below.
We completed enrollment of patients in our second clinical trial in December 2020. This second clinical trial was a Phase Ia/Ib study of the safety, efficacy and pharmacokinetics of SBP-101 administered in combination with two standard-of-care chemotherapy agents, gemcitabine and nab-paclitaxel. The trial included six study sites; four in Australia and two in the United States. In the Phase Ia portion of this trial, we completed enrollment during the first quarter of 2020 of four cohorts with increased dosage levels of SBP-101 administered in the second and third cohorts; the fourth cohort evaluated an alternate dosing schedule. A total of 25 subjects were enrolled in four cohorts of Phase Ia. The demonstration of adequate safety in Phase Ia allowed us to immediately begin enrollment in February 2020 in the Phase Ib exploration of efficacy. By December 2020, an additional 25 subjects in Phase Ib using the recommended dosage level and schedule determined in Phase Ia were enrolled.
After Phase Ib enrollment was completed, some patients in the trial were noted to have complaints of serious visual adverse effects. Visual changes were not seen in the SBP-101 monotherapy study. We consulted with our DSMB and halted the administration of SBP-101 while all other trial activities continued. In February of 2021, we also conferred with the FDA regarding our plan to hold dosing of SBP-101. This withholding constituted a “partial clinical hold.” In April of 2021, the FDA lifted the partial clinical hold. The Company agreed with the FDA to include in the design of all future studies the exclusion of patients with a history of retinopathy or risk of retinal detachment and scheduled ophthalmologic monitoring for all patients.
Interim results from the full study were presented in a poster in at American Society of Clinical Oncology Gastrointestinal conference (“ASCO-GI”) in January of 2022.
Figure 4. Evaluation of SBP 101 Phase Ib First-line combo-therapy Safety Trial –
Best CA19-9 by RECIST Response
Best Response per Subject – Cohorts 4 and Ib N=29. Best response in evaluable subjects was a Complete Response (“CR”) in 1 (3%), Partial Response (“PR”) in 13 (45%), SD in 10 (34%) and PD in 5 (17%). One subjects did not have post baseline scans with RECIST tumor assessments. Source: Singhal, N., Poster Presentation, ASCO GI 2022
Figure 5 Progression-Free (PFS) and Overall (OS) Survival for Cohort 2, Cohort 4+Ib, and a historical control administered G+A
Source: Singhal, N., Poster Presentation, ASCO GI 2022
PFS and OS includes 7 subjects from Cohort 2 as these subjects received the same dose albeit on a different schedule than the 30 subject in Cohorts 4 and Ib. PFS may have been negatively impacted by drug dosing interruptions to evaluate potential toxicity. Median overall survival in Cohort 4 + Phase IB is 12.0 months and is not yet final as of the January 2022 poster date. Two subjects in Cohort 2 are still alive at 29.3+ and 30.7+ months, respectively.
Phase II/III Clinical Trial design and anticipated timing (ASPIRE trial)
The Company announced that the ASPIRE trial, a randomized study of SBP-101 with Gemcitabine and Nab-Paclitaxel versus Gemcitabine, Nab-Paclitaxel and placebo, was initiated in January of 2022. The trial is in patients with first-line metastatic pancreatic ductal adenocarcinoma. The trial is designed as a Phase II/III randomized double-blind placebo-controlled trial, with a primary endpoint of overall survival. The design includes a phase 2 portion for which a futility analysis after 104 progression free survival events will occur. If the futility analysis is favorable, the trial will be expanded to the phase 3 portion with a primary endpoint of overall survival. We are intending to conduct the ASPIRE trial at leading cancer centers in the United States, Europe and the Asia-Pacific Region. It is expected that there will be approximately 60 sites and we anticipate enrollment for the phase 2 portion to be completed in approximately 12 months.
If we can successfully complete all FDA recommended clinical studies, we intend to seek marketing authorization from the FDA, the EMA (European Union), Ministry of Health and Welfare (Japan) and TGA (Australia). The submission fees may be waived when SBP-101 has been designated an orphan drug in each geographic region, as described under “Orphan Drug Status.”
Total Development Costs
The development of SBP-101 involves a preclinical and a clinical development phase. We have completed our initial preclinical development work for pancreatic cancer as well as two Phase I clinical trials. The Phase II/III trial has just been initiated. Additional clinical trials will likely be required for FDA or other approvals in foreign jurisdictions if the results of the first-line clinical trial of our SBP-101 product candidate justify continued development. The cost and timing of additional clinical trials is highly dependent on the nature and size of the trials.
Orphan Drug Status
The Orphan Drug Act provides special status to drugs which are intended for the safe and effective treatment, diagnosis or prevention of rare diseases that affect fewer than 200,000 people in the United States, or that affect more than 200,000 persons but for which a manufacturer is not expected to recover the costs of developing and marketing such a drug. Orphan drug designation has the advantage of reducing drug development costs by: (i) streamlining the FDA’s approval process, (ii) providing tax breaks for expenses related to the drug development, (iii) allowing the orphan drug manufacturer to receive assistance from the FDA in funding the clinical testing necessary for approval of an orphan drug, and (iv) facilitating drug development efforts. More significantly, the orphan drug manufacturer’s ability to recover its investment in developing the drug is also greatly enhanced by the FDA granting the manufacturer seven years of exclusive US marketing rights upon approval. Designation of a product candidate as an orphan drug therefore may provide its sponsor with the opportunity to adopt a faster and less expensive pathway to commercializing its product. We obtained US Orphan Drug Status in 2014 and we intend to apply for Orphan Drug Status in Europe, Japan and Australia when we have additional clinical data.
Fast Track
In June 2020, we received Fast Track Designation from the FDA for development of SBP-101 for the treatment of first-line patients with metastatic PDA when administered in combination with gemcitabine and nab-paclitaxel. With the designation of Fast Track Designation, we may engage in more frequent interactions with the FDA, and the FDA may review sections of a New Drug Application (“NDA”) before the application is complete. This rolling review is available if the Company provides, and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the FDA’s time period goal for reviewing an application does not begin until the last section of the NDA is submitted. Additionally, Fast Track Designation may be withdrawn by the FDA if the FDA believes that the designation is no longer supported by data emerging in the clinical trial process.
Intellectual Property
As the result of efforts at our contract manufacturer Syngene International Ltd to refine the synthetic process, a new shorter synthetic process has been developed on which a patent (US 11,098,005 B2) “METHODS FOR PRODUCING (65,155)-3,8,13,18-TETRAAZAICOSANE-6, 15-DIOL” issued on Aug. 24, 2021 and was assigned to Panbela. The patent claims cover a novel process for the production of SBP-101 and reduces the number of synthetic steps from nineteen to six.
We are evaluating other opportunities to provide additional intellectual property.
Human Capital Management
As of December 31, 2021, we had six employees, four of which were full time. None of our employees are represented by a labor union or covered by a collective bargaining agreement. We believe our relationship with our employees is good.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, incentivizing, and integrating our existing and new employees, advisors, and consultants. The principal purposes of our equity and cash incentive plans are to attract, retain and reward personnel through the granting of stock-based and cash-based compensation awards, in order to motivate such individuals to perform to the best of their abilities and achieve our objectives and lead to the success of the Company and increase value to our stockholders.
We value diversity of backgrounds and perspectives in our workforce and our policy is that we do not discriminate based on race, religion, creed, color, national origin, ancestry, physical disability, mental disability, medical condition, genetic information, marital status, sex, gender, gender identity, gender expression, age, military and veteran status, sexual orientation or any other protected characteristic as established by federal, state or local laws.
We believe that operational responsibilities can be handled by our current employees, independent consultants and our global CRO. We have historically used the services of independent consultants and contractors to perform various professional services. We believe that this use of third-party service providers enhances our ability to minimize general and administrative expenses. We intend to periodically evaluate our staffing and talent requirements and expect to add employees if that becomes a more appropriate resourcing alternative.
Competition
The development and commercialization of new products to treat cancer is intensely competitive and subject to rapid and significant technological change. While we believe that our knowledge, experience and scientific resources provide us with competitive advantages, we face substantial competition from major pharmaceutical companies, specialty pharmaceutical companies, and biotechnology companies worldwide. Many of our competitors have significantly greater financial, technical, and human resources. Smaller and early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. As a result, our competitors may discover, develop, license or commercialize products before or more successfully than we do.
We face competition with respect to our current product candidates and will face competition with respect to future product candidates, from segments of the pharmaceutical, biotechnology and other related markets that pursue approaches to targeting molecular alterations and signaling pathways associated with cancer. Our competitors may obtain regulatory approval of their products more rapidly than we do or may obtain patent protection or other intellectual property rights that limit our ability to develop or commercialize our product candidates. Our competitors may also develop drugs that are more effective, more convenient, less costly, or possessing better safety profiles than our products, and these competitors may be more successful than us in manufacturing and marketing their products.
In addition, we may need to develop our product candidates in collaboration with diagnostic companies, and we will face competition from other companies in establishing these collaborations. Our competitors will also compete with us in recruiting and retaining qualified scientific, management and commercial personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Furthermore, we also face competition more broadly across the market for cost-effective and reimbursable cancer treatments. The most common methods of treating patients with cancer are surgery, radiation and drug therapy, including chemotherapy, immunotherapy, hormone therapy and targeted drug therapy or a combination of such methods. There are a variety of available drug therapies marketed for cancer. In many cases, these drugs are administered in combination to enhance efficacy. While our product candidates, if any are approved, may compete with these existing drug and other therapies, to the extent they are ultimately used in combination with or as an adjunct to these therapies, our product candidates may be approved as companion treatments and not be competitive with current therapies. Some of these drugs are branded and subject to patent protection, and others are available on a generic basis. Insurers and other third-party payors may also encourage the use of generic products or specific branded products. We expect that if our product candidates are approved, they will be priced at a premium over competitive generic, including branded generic, products. As a result, obtaining market acceptance of, and gaining significant share of the market for, any of our product candidates that we successfully introduce to the market will pose challenges. In addition, many companies are developing new therapeutics and we cannot predict what the standard of care will be as our product candidate progresses through clinical development.
SBP-101
Commercialization
We have not established a sales, marketing or product distribution infrastructure nor have we devoted significant management resources to planning such an infrastructure because our lead product candidate is still in early clinical development. We currently anticipate that we will partner with a larger pharmaceutical organization having the expertise and capacity to perform these functions.
Manufacturing and Suppliers
We do not own or operate, and currently have no plans to establish, any manufacturing facilities. We currently rely, and expect to continue to rely, on third parties for the manufacture of our product candidates for preclinical and clinical testing as well as for commercial manufacture of any products that we may commercialize. If needed, we intend to engage, by entering into a supply agreement or through another arrangement, third party manufacturers to provide us with additional SBP-101 clinical supply. We identified and qualified manufacturers to provide the active pharmaceutical ingredient and fill-and-finish services for our initial product candidate prior to our submission of an NDA to the FDA and expect to continue utilizing this approach for any future product candidates.
Material Agreements
The Standard Exclusive License Agreement (“License Agreement”) dated December 22, 2011, between us and UFRF grants us an exclusive license to the proprietary technology covered by issued United States Patents Nos. US 5,962,533, which expired in February 2016, and US 6,160,022 which expired in July 2020 and Know-How as defined by the License Agreement, with reservations by UFRF for academic or government uses. Under this agreement, we had agreed to pay various royalties, expenses and milestone payments to UFRF.
The License Agreement was amended in December 2016 (“First Amendment”) and again in October 2019 (“Second Amendment”).
Under the Second Amendment all minimum royalty payments and milestone payments defined in the License Agreement were eliminated. In addition, the period of payment royalties was changed to be the shorter of (i) ten (10) years from first commercial sale or (ii) the period of market exclusivity on a country-by-country basis. UFRF may also terminate this license for standard and similar causes such as material breach of the agreement, bankruptcy, failure to pay royalties and other customary conditions. The agreement allows for UFRF to terminate if the first commercial sale is not made by December 31, 2025.
The foregoing description of the material terms of the License Agreement, as amended, is qualified by the full text of the License Agreement , and the Second Amendment, both of which are incorporated herein by reference.
Government Regulation
FDA Approval Process
In the United States, pharmaceutical products are subject to extensive regulation by the FDA. The Federal Food, Drug and Cosmetic Act and other federal and state statutes and regulations govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling and import and export of pharmaceutical products. Failure to comply with applicable US requirements may subject a company to a variety of administrative or judicial sanctions, such as FDA refusal to approve pending NDAs, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties and criminal prosecution.
Pharmaceutical product development for a new product or certain changes to an approved product in the United States typically involves preclinical laboratory and animal tests, the submission to the FDA of an IND which must become effective before clinical testing may commence, and adequate and well-controlled clinical trials to establish the safety and effectiveness of the drug for each indication for which FDA approval is sought. Satisfaction of FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity and novelty of the product or disease.
Preclinical tests include laboratory evaluation of product chemistry, formulation and toxicity, as well as animal trials to assess the characteristics and potential safety and efficacy of the product. The conduct of the preclinical tests must comply with federal regulations and requirements, including good laboratory practices. The results of preclinical testing are submitted to the FDA as part of an IND along with other information, including the Investigator’s Brochure, information about product chemistry, manufacturing and controls, potential perceived side effects and risks, and a proposed clinical trial protocol. Long-term preclinical tests, such as animal tests of reproductive toxicity and carcinogenicity, may continue after the IND is submitted.
A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA has neither commented on nor questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin.
Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations; (ii) in compliance with good clinical practice (“GCP”), an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators and monitors; as well as (iii) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. Each protocol involving testing on US patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
The FDA may order the temporary, or permanent, discontinuation of a clinical trial at any time, or impose other sanctions, if it believes that the clinical trial either is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. The study protocol and informed consent information for patients in clinical trials must also be submitted to an institutional review board (“IRB”) for approval. An IRB may also require the clinical trial at the site to be halted, either temporarily or permanently, for failure to comply with the IRB’s requirements, or may impose other conditions.
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In Phase I, the initial introduction of the drug into healthy human subjects/patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses, and, if possible, early evidence of effectiveness. Phase II usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, dosage tolerance and optimum dosage, and to identify common adverse effects and safety risks. If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase II evaluations, pivotal, or Phase III trials are undertaken to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug. In many cases the FDA requires two adequate and well-controlled Phase III clinical trials to demonstrate the efficacy of the drug. A single Phase III trial with other confirmatory evidence may be sufficient in instances where the study is a large multicenter trial demonstrating internal consistency and a statistically very persuasive finding of a clinically meaningful effect on mortality, irreversible morbidity or prevention of a disease with a potentially serious outcome and confirmation of the result in a second trial would be practically or ethically impossible. After an NDA is approved, a Phase IV trial may be undertaken to evaluate safety over a long period of time, quality of life or cost effectiveness.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing of the product may begin in the United States. The NDA must include the results of all preclinical, clinical and other testing and a compilation of data relating to the product’s pharmacology, chemistry, toxicology, manufacture, controls and any proposed labeling. The cost of preparing and submitting an NDA is substantial, and the fees are typically increased annually.
The FDA has 60 days from its receipt of an NDA to determine whether the application will be accepted for filing based on the agency’s threshold determination that it is sufficiently complete to permit substantive review. Once the submission is accepted for filing, the FDA begins an in-depth review. The FDA has agreed to certain performance goals in the review of NDAs to encourage timeliness. Most applications for standard review drug products are reviewed within twelve months from submission; most applications for priority review drugs are reviewed within eight months from submission. Priority review can be applied to drugs that the FDA determines offer major advances in treatment or provide a treatment where no adequate therapy exists. If priority review is achieved, the FDA’s goal is to act on the application within six months. The review process for both standard and priority review may be extended by the FDA for three additional months to consider certain late-submitted information, or information intended to clarify information already provided in the submission. The FDA may also refer applications for novel drug products, or drug products that present difficult questions of safety or efficacy, to an outside advisory committee—typically a panel that includes clinicians and other experts—for review, evaluation and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations.
Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with current good manufacturing practice (“cGMP”), a quality system regulating manufacturing, is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the indication studied.
After the FDA evaluates the NDA and the manufacturing facilities, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the submission and may require substantial additional testing, or information, in order for the FDA to reconsider the application. If, or when, those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. The FDA has committed to reviewing such resubmissions in two or six months depending on the type of information included.
An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a risk evaluation and mitigation strategy (“REMS”) to help ensure that the benefits of the drug outweigh the potential risks. REMS can include medication guides, communication plans for healthcare professionals, and elements to assure safe use (“ETASU”). ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing.
Changes to some of the conditions established in an approved application, including changes in indications, labeling, or manufacturing processes or facilities, require submission and FDA approval of a new NDA or NDA supplement before the change can be implemented. An NDA supplement for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures and actions in reviewing NDA supplements as it does in reviewing NDAs.
Fast Track Designation and Accelerated Approval
The FDA is required to facilitate the development, and expedite the review, of drugs that are (1) intended for the treatment of a serious or life-threatening disease or (2) condition for which there is no effective treatment and which demonstrate the potential to address unmet medical needs for the condition. Under the Fast Track program, the sponsor of a new product candidate may request that the FDA designate the product candidate for a specific indication as a Fast Track drug concurrent with, or after, the filing of the IND for the product candidate. The FDA must determine if the product candidate qualifies for Fast Track Designation within 60 days of receipt of the sponsor’s request.
Under the Fast Track program and FDA’s accelerated approval regulations, the FDA may approve a drug for a serious or life-threatening illness that provides meaningful therapeutic benefit to patients over existing treatments based upon a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments.
In clinical trials, a surrogate endpoint is a measurement of laboratory or clinical signs of a disease or condition that substitutes for a direct measurement of how a patient feels, functions, or survives. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. A product candidate approved on this basis is subject to rigorous post-marketing compliance requirements, including the completion of Phase IV or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, will allow FDA to withdraw the drug from the market on an expedited basis. All promotional materials for product candidates approved under accelerated regulations are subject to priority review by FDA.
If a submission is granted Fast Track Designation, the sponsor may engage in more frequent interactions with the FDA, and the FDA may review sections of the NDA before the application is complete. This rolling review is available if the applicant provides, and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the FDA’s time period goal for reviewing an application does not begin until the last section of the NDA is submitted. Additionally, Fast Track Designation may be withdrawn by the FDA if the FDA believes that the designation is no longer supported by data emerging in the clinical trial process.
Breakthrough Therapy Designation
The FDA is also required to expedite the development and review of the application for approval of drugs that are intended to treat a serious or life-threatening disease or condition where preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints. Under the Breakthrough Therapy program, the sponsor of a new product candidate may request that the FDA designate the product candidate for a specific indication as a breakthrough therapy. The FDA must determine if the product candidate qualifies for Breakthrough Therapy designation within 60 days of receipt of the sponsor’s request.
Orphan Drug Designation and Exclusivity
The Orphan Drug Act provides incentives for the development of products intended to treat rare diseases or conditions. Under the Orphan Drug Act, the FDA may grant orphan designation to a drug intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or more than 200,000 individuals in the United States and for which there is no reasonable expectation that the cost of developing and making a drug available in the United States for this type of disease or condition will be recovered from sales of the product. If a sponsor demonstrates that a drug is intended to treat a rare disease or condition, the FDA will grant orphan designation for that product for the orphan disease indication, assuming that the same drug has not already been approved for the indication for which the sponsor is seeking orphan designation. If the same drug has already been approved for the indication for which the sponsor is seeking orphan designation, the sponsor must present a plausible hypothesis of clinical superiority in order to obtain orphan designation. Orphan designation must be requested before submitting an NDA. After the FDA grants orphan designation, the FDA discloses the identity of the therapeutic agent and its potential orphan use.
Orphan designation may provide manufacturers with benefits such as research grants, tax credits, Prescription Drug User Fee Act (“PDUFA”) application fee waivers and eligibility for orphan drug exclusivity. If a product that has orphan designation subsequently receives the first FDA approval of the active moiety for that disease or condition for which it has such designation, the product is entitled to orphan drug exclusivity, which for seven years prohibits the FDA from approving another product with the same active ingredient for the same indication, except in limited circumstances. Orphan drug exclusivity will not bar approval of another product under certain circumstances, including if a subsequent product with the same active ingredient for the same indication is shown to be clinically superior to the approved product on the basis of greater efficacy or safety, or providing a major contribution to patient care, or if the company with orphan drug exclusivity is not able to meet market demand. Further, the FDA may approve more than one product for the same orphan indication or disease as long as the products contain different active ingredients. Moreover, competitors may receive approval of different products for the indication for which the orphan drug has exclusivity or obtain approval for the same product but for a different indication for which the orphan drug has exclusivity.
In the European Union, orphan drug designation also entitles a party to financial incentives such as reduction of fees or fee waivers and 10 years of market exclusivity is granted following drug or biological product approval. This period may be reduced to 6 years if the orphan drug designation criteria are no longer met, including where it is shown that the product is sufficiently profitable not to justify maintenance of market exclusivity.
Orphan drug designation must be requested before submitting an application for marketing approval. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process.
Post-Approval Requirements
Once an NDA is approved, a product will be subject to certain post-approval requirements. For instance, the FDA closely regulates the post-approval marketing and promotion of drugs, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities and promotional activities involving the internet. Drugs may be marketed only for the approved indications and in accordance with the provisions of the approved labeling.
Adverse event reporting and submission of periodic reports are required following FDA approval of an NDA. The FDA also may require post-marketing testing, known as Phase IV testing, risk evaluation and mitigation strategies, or REMS, and surveillance to monitor the effects of an approved product, or FDA may place conditions on an approval that could restrict the distribution or use of the product. In addition, quality control, drug manufacture, packaging and labeling procedures must continue to conform to current good manufacturing practices, or cGMPs, after approval. Drug manufacturers and certain of their subcontractors are required to register their establishments with the FDA and certain state agencies. Registration with FDA subjects’ entities to periodic unannounced inspections by the FDA, during which the Agency inspects manufacturing facilities to assess compliance with cGMPs. Accordingly, manufacturers must continue to expend time, money and effort in the areas of production and quality-control to maintain compliance with cGMPs. Regulatory authorities may withdraw product approvals or request product recalls if a company fails to comply with regulatory standards, if it encounters problems following initial marketing, or if previously unrecognized problems are subsequently discovered.
Additional Regulations and Environmental Matters
In the United States, our activities are potentially subject to regulation by various federal, state and local authorities in addition to the FDA, including but not limited to, the Centers for Medicare and Medicaid Services, or CMS, other divisions of the U.S. Department of Health and Human Services (e.g., the Office of Inspector General), the U.S. Department of Justice, or DOJ, and individual U.S. Attorney offices within the DOJ, and state and local governments. For example, sales, marketing and scientific/educational grant programs must comply with the anti-fraud and abuse provisions of the Social Security Act, the false claims laws, and our activities may implicate the privacy provisions of the Health Insurance Portability and Accountability Act (“HIPAA”) and similar state laws, each as amended.
The federal Anti-Kickback Statute prohibits, among other things, any person or entity, from knowingly and willfully offering, paying, soliciting or receiving any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or in return for purchasing, leasing, ordering or arranging for the purchase, lease or order of any item or service reimbursable under Medicare, Medicaid or other federal healthcare programs. The term remuneration has been interpreted broadly to include anything of value. The Anti-Kickback Statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on one hand and prescribers, purchasers, and formulary managers on the other. There are statutory exceptions and regulatory safe harbors protecting some common activities from prosecution. The exceptions and safe harbors are drawn narrowly and practices that involve remuneration that may be alleged to be intended to induce prescribing, purchasing or recommending may be subject to scrutiny if they do not qualify for an exception or safe harbor. Failure to meet all the requirements of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review of all its facts and circumstances. While we reasonably believe our practices to be in compliance with the Anti-Kickback Statute, our practices may not in all cases meet all the criteria for protection under a statutory exception or regulatory safe harbor.
Additionally, the intent standard under the Anti-Kickback Statute was amended by the Affordable Care Act (“ACA”) to a stricter standard such that a person or entity no longer needs to have actual knowledge of the statute or specific intent to violate it to have committed a violation. In addition, the ACA codified case law that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal False Claims Act (as further discussed below).
The Civil Monetary Penalties statute authorizes the imposition of severe financial penalties against any person or entity who, among other things, is determined to have presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent.
The federal False Claims Act prohibits, among other things, any person or entity from knowingly presenting, or causing to be presented, a false claim for payment to, or approval by, the federal government or knowingly making, using, or causing to be made or used, a false record or statement material to a false or fraudulent claim to the federal government. As a result of a modification made by the Fraud Enforcement and Recovery Act of 2009, a claim includes “any request or demand” for money or property presented to the U.S. government. Recently, several pharmaceutical and other healthcare companies have been prosecuted under these laws for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. Other companies have been prosecuted for causing false claims to be submitted because of the companies’ marketing of the product for unapproved, and thus non-reimbursable, uses.
HIPAA created new federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud or to obtain, by means of false or fraudulent pretenses, representations or promises, any money or property owned by, or under the control or custody of, any healthcare benefit program, including private third-party payors and knowingly and willfully falsifying, concealing or covering up by trick, scheme or device, a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of, or payment for healthcare benefits, items or services.
Also, many states have similar fraud and abuse statutes or regulations that apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor.
We may be subject to data privacy and security regulations by both the federal government and the states in which we conduct our business. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act (“HITECH) and its implementing regulations, imposes requirements relating to the privacy, security and transmission of individually identifiable health information. Among other things, HITECH makes HIPAA’s privacy and security standards directly applicable to business associates, independent contractors or agents of covered entities that receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also created four new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions. In addition, state laws govern the privacy and security of health information in specified circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
Additionally, the federal Physician Payments Sunshine Act within the ACA, and its implementing regulations, require that certain manufacturers of drugs, devices, biological and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) to report information related to certain payments or other transfers of value made or distributed to physicians, other specified health care professionals and teaching hospitals, or to entities or individuals at the request of, or designated on behalf of, the physicians, other specified health care professionals and teaching hospitals and to report annually certain ownership and investment interests held by physicians and other specified health care professionals and their immediate family members. Some states have analogous laws requiring manufacturers to report certain transfers of value to covered individuals and entities. To distribute products commercially, we must comply with state laws that require the registration of manufacturers and wholesale distributors of drug and biological products in a state, including, in certain states, manufacturers and distributors who ship products into the state even if such manufacturers or distributors have no place of business within the state. Some states also impose requirements on manufacturers and distributors to establish the pedigree of product in the chain of distribution, including some states that require manufacturers and others to adopt new technology capable of tracking and tracing product as it moves through the distribution chain. Several states have enacted legislation requiring pharmaceutical and biotechnology companies to establish marketing compliance programs, file periodic reports with the state, make periodic public disclosures on sales, marketing, pricing, clinical trials and other activities, and/or register their sales representatives, as well as to prohibit pharmacies and other healthcare entities from providing certain physician prescribing data to pharmaceutical and biotechnology companies for use in sales and marketing, and to prohibit certain other sales and marketing practices. All our activities are potentially subject to federal and state consumer protection and unfair competition laws.
If our operations are found to be in violation of any of the federal and state healthcare laws described above or any other governmental regulations that apply to us, we may be subject to penalties, including without limitation, civil, criminal and/or administrative penalties, damages, fines, disgorgement, exclusion from participation in government programs, such as Medicare and Medicaid, injunctions, private “qui tam” actions brought by individual whistleblowers in the name of the government, or refusal to allow us to enter into government contracts, contractual damages, reputational harm, administrative burdens, diminished profits and future earnings, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
Coverage and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any product candidates for which we obtain regulatory approval. In the United States and markets in other countries, sales of any products for which we receive regulatory approval for commercial sale will depend, in part, on the extent to which third-party payors provide coverage and establish adequate reimbursement levels for such products. In the United States, third-party payors include federal and state healthcare programs, privately managed care providers, health insurers and other organizations. The process for determining whether a third-party payor will provide coverage for a product may be separate from the process for setting the price of a product or for establishing the reimbursement rate that such a payor will pay for the product. Third-party payors may limit coverage to specific products on an approved list, also known as a formulary, which might not include all the FDA-approved products for a particular indication. Third-party payors are increasingly challenging the price, examining the medical necessity and reviewing the cost-effectiveness of medical products, therapies and services, in addition to questioning their safety and efficacy. We may need to conduct expensive pharmaco-economic studies to demonstrate the medical necessity and cost-effectiveness of our products, in addition to the costs required to obtain the FDA approvals. Our product candidates may not be considered medically necessary or cost-effective. A payor’s decision to provide coverage for a product does not imply that an adequate reimbursement rate will be approved. Further, one payor’s determination to provide coverage for a product does not assure that other payors will also provide coverage for the product. This is also true of Medicare reimbursement, where different vendors process payments, so that coverage by one vendor does not assure that all other vendors will provide coverage. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development. In addition, the United States federal government position on matters related to drug pricing is evolving and uncertain, and any changes could have a material impact on drug pricing generally in the United States, including for our product candidates if approved.
Different pricing and reimbursement schemes exist in other countries. In the EU, governments influence the price of pharmaceutical products through their pricing and reimbursement rules and control of national health care systems that fund a large part of the cost of those products to consumers. Some jurisdictions operate positive and negative list systems under which products may only be marketed once a reimbursement price has been agreed. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials that compare the cost-effectiveness of a particular product candidate to currently available therapies. Other member states allow companies to fix their own prices for medicines but monitor and control company profits. The National Institute for Health and Care Excellence (NICE) in the United Kingdom also requires consideration of cost-benefit analysis. The downward pressure on health care costs has become very intense. As a result, increasingly high barriers are being erected to the entry of new products. In addition, in some countries, cross-border imports from low-priced markets exert a commercial pressure on pricing within a country.
The marketability of any product candidates for which we receive regulatory approval for commercial sale may suffer if the government and third-party payors fail to provide adequate coverage and reimbursement. In addition, emphasis on managed care in the United States has increased and we expect will continue to increase the pressure on healthcare pricing. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which we receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
Available Information
Our website is located at www.Panbela.com. The information contained on or connected to our website is not a part of this report. We have included our website address as a factual reference and do not intend it to be an active link to our website.
We make available, free of charge, through our website materials we file or furnish to the SEC pursuant to Section 13(a) or 15(d) of the Exchange Act, including our annual report on Form 10-K, our quarterly reports on Form 10-Q, our current reports on Form 8-K and amendments to those reports. These materials are posted to our website as soon as reasonably practical after we electronically file them with or furnish them to the SEC.
Members of the public may read and copy any materials we file with the SEC at its Public Reference Room at 450 Fifth Street, NW, Washington, DC 20549. Information on the operation of the Public Reference Room is available by calling the SEC at 1-800-SEC-0330. The SEC maintains a website that contains reports, proxy and information statements and other information about us and other issuers that file electronically at http://www.sec.gov.
Properties
Our primary business functions are conducted by our employees and independent contractors on a distributed basis. Accordingly, we do not lease or own any real property and all employees currently work from their homes. We maintain our principal mailing address at Suite 305 at 712 Vista Boulevard in Waconia, Minnesota.
Legal Proceedings
We are not currently party to any material legal proceedings. From time to time, we may be named as a defendant in legal actions arising from our normal business activities. We believe that we have obtained adequate insurance coverage or rights to indemnification in connection with potential legal proceedings that may arise.
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Properties
Our primary business functions are conducted by our employees and independent contractors on a distributed basis. Accordingly, we do not lease or own any real property and all employees currently work from their homes. We maintain our principal mailing address at Suite 305 at 712 Vista Boulevard in Waconia, Minnesota.
Legal Proceedings
We are not currently party to any material legal proceedings. From time to time, we may be named as a defendant in legal actions arising from our normal business activities. We believe that we have obtained adequate insurance coverage or rights to indemnification in connection with potential legal proceedings that may arise.
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Available Information
Our website is located at www.Panbela.com. The information contained on or connected to our website is not a part of this prospectus. We have included our website address as a factual reference and do not intend it to be an active link to our website.
We make available, free of charge, through our website at www.sunbiopharma.com, materials we file or furnish to the SEC pursuant to Section 13(a) or 15(d) of the Exchange Act, including our annual report on Form 10-K, our quarterly reports on Form 10-Q, our current reports on Form 8-K and amendments to those reports. These materials are posted to our website as soon as reasonably practicable after we electronically file them with or furnish them to the SEC.
The SEC maintains a website that contains reports, proxy and information statements and other information about us and other issuers that file electronically at www.sec.gov.
MANAGEMENT
Information about our Executive Officers
Jennifer K. Simpson, Ph.D., MSN, CRNP, age 53, has served as President and Chief Executive Officer and as a director of our Company since July 2020. Prior to joining the Company Dr. Simpson served as President and Chief Executive Officer and as a member of the board of directors of Delcath Systems, Inc. (Nasdaq: DCTH) from 2015 to June 2020. She had previously held various other leadership roles at Delcath since 2012. From 2011 to 2012, Dr. Simpson served as Vice President, Global Marketing, Oncology Brand Lead at ImClone Systems, Inc. (a wholly owned subsidiary of Eli Lilly and Company), where she was responsible for all product commercialization activities and launch preparation for one of the late-stage assets. From 2009 to 2011, Dr. Simpson served as Vice President, Product Champion and from 2008 to 2009 as the Associate Vice President, Product Champion for ImClone’s product Ramucirumab. From 2006 to 2008, Dr. Simpson served as Product Director, Oncology Therapeutics Marketing at Ortho Biotech (now Janssen Biotech), a Pennsylvania-based biotech company that focuses on innovative solutions in immunology, oncology and nephrology. Earlier in her career, Dr. Simpson spent over a decade as a hematology/oncology nurse practitioner and educator. Dr. Simpson has served on the board of directors and nominating and corporate governance committee of Eagle Pharmaceuticals, Inc. since August 2019 and on the board of Directors of CytRx Corporation since July 2021.
Susan Horvath, age 63, has served as our Vice President and Chief Financial Officer since April 2018. Ms. Horvath has held both finance and operating positions within pharmaceutical, healthcare and consumer organizations. In addition to her position with the Company, Ms. Horvath sits on the board of directors and provides financial consulting services for Photonic Pharma, LLC, a privately held company focused on efficiencies in early stage drug discovery. Prior to joining the Panbela team Ms. Horvath served as Chief Financial Officer of Eyebobs, LLC, a private company focused on eyewear for corrective vision, from 2016 to January 2018; Vice President and Chief Financial Officer of Tenacious Holdings, Inc. (d/b/a ergodyne) a privately held, safety products company, from 2014 to 2016; Chief Financial Officer and Vice President of Human Resources at Healthsense, Inc., a next generation technology (SaaS) and remote monitoring company focused on providing safety and improving quality of life while reducing overall costs of healthcare for seniors and fragile adults, from 2011 to 2014; Chief Financial Officer, Vice President of Operations & Human Resources of Hemosphere, Inc., an early commercialization stage medical device company, from 2008 to 2010; and Vice President & Team Leader International of CNS, Inc, a publicly traded consumer health care products company focused on the development and marketing of strong consumer brands, from 2004 to 2007. Ms. Horvath holds a Bachelor of Science degree in Accounting from the University of Illinois, Champaign, and is a Certified Management Accountant and Certified Public Accountant, inactive.
Information about our Board of Directors
Our business is overseen by a Board of Directors divided into three classes as nearly equal in number as possible, and directors typically are elected to a designated class for a term of three years. The following sets forth certain information regarding the current members of our Board of Directors:
Class I Directors –Terms Expiring in 2023
Daniel J. Donovan, age 58, has served as a director since June 2022. He had served as a director and Chief Business Officer, a non-employee position, of Cancer Prevention from 2011 until immediately before the completion of its acquisition by Panbela in June 2022. He has served as chief executive officer of rareLife Solutions, Inc., a private company, since he co-founded it in 2014. Before rareLife, Mr. Donovan founded Envision Pharma in 2001, serving as managing director then president until 2011. Envision Pharma was acquired by United BioSource Corporation in 2008, where Mr. Donovan served as Senior Vice President Strategy and Market Development and was a member of the leadership team. Mr. Donovan began his career at Pfizer serving in a variety of positions of increasing responsibility, ranging from sales to market research and marketing in the U.S. and internationally, culminating in his position as Director and European Team Leader. During his time at Pfizer, he played a pivotal role in the commercialization of some of the pharmaceutical industry’s most successful product launches.
Jeffrey E. Jacob, age 60, has served as a director since June 2022. He had served as Chief Executive Officer of Cancer Prevention from 2009 until immediately before the completion of its acquisition by Panbela in June 2022. He is also the principal of Tucson Pharma Ventures LLC, an Arizona-based biopharmaceutical consulting and investment firm, a role he’s held since 2004. In 2004, Mr. Jacob founded Systems Medicine Inc., a startup company applying systems biology, predictive pharmacogenomics, and clinical trial design innovations to the development of new cancer drugs and served as its chief executive officer until its sale in 2007, after which he served as a divisional chief executive officer until late 2008. Between 1987 and 2004, Mr. Jacob was employed by Research Corporation Technologies, most recently as Senior Vice President. During that time, he led the transformation of Research Corporation Technologies from a patent development and licensing organization to an early stage-technology incubation and venture deployment firm. He has served as a member of the board of directors of Research Corporation Technologies and currently serves as its chair. He is also a founding board member and previously served as the chief program officer of Critical Path Institute. Mr. Jacob holds a master’s degree in engineering and a master’s degree in Technology and Policy from the Massachusetts Institute of Technology and a bachelor’s degree in engineering from the University of Arizona.
Jennifer K. Simpson Ph.D., MSN, CRNP, has served as our President and Chief Executive Officer and as a director of our Company since July 2020. See “Information about our Executive Officers” above for further information regarding Dr. Simpson’s background and experience.
Class II Directors – Term Expiring in 2024
Michael T. Cullen, M.D., M.B.A., age 76, has served as Chairman of the Board and a non-employee director of our Company since his retirement as an employee of the Company in May 2021. Dr. Cullen had served as Executive Chairman and as a director of our Company since its co-founding in November 2011. Dr. Cullen brings 33 years of pharmaceutical experience to our Company, including expertise in working with development-stage companies in planning, designing and advancing drug candidates from preclinical through clinical development. Dr. Cullen served as our President and Chief Executive Officer between October 2018 and July 2020. He previously served as our Chief Medical Officer and President from November 2011 to June 2015. Dr. Cullen provided due diligence consulting to the pharmaceutical industry from 2009 to 2011, after one year in transition consulting to Eisai Pharmaceuticals. He developed several oncology drugs as Chief Medical Officer for MGI Pharma Inc. from 2000 to 2008, and previously at G.D. Searle, SunPharm Corporation, and as Vice President for Clinical Consulting at IBAH Inc., the world’s fifth largest contract research organization, where he provided consulting services on business strategy, creating development plans, regulatory matters and designing clinical trials for several development stage companies in the pharmaceutical industry. Dr. Cullen was also a co-founder and Chief Executive Officer of IDD Medical, a pharmaceutical start-up company. Dr. Cullen joined 3M Pharmaceuticals in 1988 and contributed to the development of cardiovascular, pulmonary, rheumatology and immune-response modification drugs. Over the course of his career Dr. Cullen has been instrumental in obtaining the approval of ten drugs, including three since 2004: Aloxi®, Dacogen® and Lusedra®. Board-certified in Internal Medicine, Dr. Cullen practiced from 1977 to 1988 at Owatonna Clinic, Owatonna, MN, where he served as president. Dr. Cullen earned his MD and BS degrees from the University of Minnesota and his MBA from the University of St. Thomas and completed his residency and Board certification in Internal Medicine through the University of North Carolina in Chapel Hill and Wilmington, NC.
D. Robert Schemel, age 67, has served as a director since September 2015. Mr. Schemel had previously served as a director of SBR since March 2012. Mr. Schemel has over 39 years’ experience in the agriculture industry. From 1973-2005, Mr. Schemel owned and operated a farming operation in Kandiyohi County, Minnesota, building a 5,000-acre operation producing corn, soybeans and sugar beets. Mr. Schemel has extensive experience in serving on boards of directors. From 1992-1996 he served as a board member for ValAdCo and then from 1996-2003 he served as the Chairman of the Board for Phenix Biocomposites. We believe that Mr. Schemel brings business insight and leadership as well as significant experience in the development and growth of early stage companies.
Class III Directors –Terms Expiring in 2022
Arthur J. Fratamico, age 56, has served as a director of our Company since December of 2019. He is a registered pharmacist with over 30 years of experience in the pharmaceutical industry and has been the Chief Executive Officer of Radiant Biotherapeutics, which is advancing a novel antibody platform that is focused on the development of Multabodies, which are multi-valent and multi-specific antibodies since May 2021. Prior to Radiant, Mr. Fratamico served as Chief Business Officer at Galera Therapeutics, Inc., a biopharmaceutical company dedicated to discovering and developing novel dismutase mimetics with the goal of transforming cancer radiotherapy, since January 2017. Prior to joining Galera, Mr. Fratamico served as Chief Business Officer of Vitae Pharmaceuticals, Inc., a Nasdaq-listed clinical-stage biotechnology company, from May 2014 until its sale to Allergan in December 2016. Prior to Vitae Pharmaceuticals, he held similar executive roles with a number of biotechnology companies leading their business development efforts, including facilitating the sales of Gemin X Pharmaceuticals, Inc. and MGI Pharma, Inc. In addition to being responsible for numerous licensing transactions and acquisitions, he also directed corporate strategy and managed external corporate communications. He also served in several senior marketing, product planning and new product development positions. Mr. Fratamico earned a bachelor’s degree in pharmacy from the Philadelphia College of Pharmacy and Science and an M.B.A. from Drexel University.
Jeffrey S. Mathiesen, age 61, has served as a director of our Company since September 2015. Mr. Mathiesen also serves as a director and audit committee chairman of NeuroOne Medical Technologies Corporation, a publicly traded medical device company. Additionally, Mr. Mathiesen serves Chief Financial Officer of Helius Medical Technologies, Inc., a publicly traded medical technology company focused on neurological wellness, where he previously served as a director and audit committee chairman. He also served as a director and audit committee chairman of eNeura, Inc., a privately held medical technology company providing therapy for both acute treatment and prevention of migraine from July 2018 to February 2020. Mr. Mathiesen has served as Advisor to the CEO of Teewinot Life Sciences, a privately held biopharmaceutical company focused on the biosynthetic production of pure pharmaceutical grade cannabinoids from October 2019 to December 2019, and as Chief Financial Officer from March 2019 to October 2019. In August 2020, Teewinot Life Sciences filed a voluntary petition under Chapter 11 of the United States Bankruptcy Code. Previously he served as Chief Financial Officer of Gemphire Therapeutics Inc., a publicly traded biopharmaceutical company from September 2015 to September 2018. From August 2015 to September 2015, he was a consultant to Gemphire. He served as Chief Financial Officer of Sunshine Heart, Inc., a publicly traded medical device company, from March 2011 to January 2015. Mr. Mathiesen has held executive positions with publicly traded companies dating back to 1993, including vice president and chief financial officer positions. Mr. Mathiesen holds a B.S. in Accounting from the University of South Dakota and is also a Certified Public Accountant.
EXECUTIVE COMPENSATION
Compensation of Named Executive Officers
The following disclosure focuses on our named executive officers. For fiscal 2021 our “named executive officers” consisted of: Dr. Simpson, Ms. Horvath, and Dr. Cullen, who retired from employment with the Company in May of 2021.
Base salaries for each of our named executive officers were initially established based on arm’s-length negotiations with the applicable executive. The Compensation Committee of our Board of Directors reviews our executive officers’ salaries annually. When negotiating or reviewing base salaries, the Compensation Committee considers market competitiveness based on the experience of its members, the executive’s expected future contribution to our success and the relative salaries and responsibilities of our other executives.
Summary Compensation Table
The following table provides information regarding the compensation earned by our named executive officers for fiscal 2021 (collectively referred to as the “Executives”):
|
Name and Principal Positions
|
|
Year
|
|
Salary
($)
|
|
|
Option Awards (a)
($)
|
|
|
Nonequity
Incentive Plan
Compensation (b)
($)
|
|
|
Total
($)
|
|
|
Jennifer K. Simpson
|
|
2021
|
|
|
476,609 |
|
|
|
537,702 |
|
|
|
182,422 |
|
|
|
1,196,733 |
|
| President and Chief Executive Officer (c) |
|
2020
|
|
|
145,587 |
|
|
|
1,529,926 |
|
|
|
78,750 |
|
|
|
1,754,263 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Susan Horvath
|
|
2021
|
|
|
302,200 |
|
|
|
155,258 |
|
|
|
99,620 |
|
|
|
557,078 |
|
| Chief Financial Officer and Vice President of Finance |
|
2020
|
|
|
226,000 |
|
|
|
98,176 |
|
|
|
90,000 |
|
|
|
414,176 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michael T. Cullen
|
|
2021
|
|
|
337,147 |
|
|
|
218,199 |
|
|
|
- |
|
|
|
555,346 |
|
| Former Executive Chairman (d) |
|
2020
|
|
|
316,000 |
|
|
|
179,900 |
|
|
|
141,750 |
|
|
|
637,650 |
|
|
(a)
|
The values of option awards in this table represent the fair value of such awards granted during the fiscal year, as computed in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718. The assumptions used to determine the valuation of the awards are discussed in Note 9 to the consolidated financial statements.
|
|
(b)
|
Represents payments made under the Company’ s 2021 and 2020 Cash Incentive Program as described further below.
|
|
(c)
|
Dr. Simpson joined the Company in July 2020.
|
|
(d)
|
Dr. Cullen served as the Company’s President and Chief Executive Officer from October 2018 until July 2020. And as Executive chairman until his retirement. After his retirement in May of 2021 Dr. Cullen received non-employee director compensation of $39,375 included in salary in this table and options and RSU’s granted after Dr. Cullen’s retirement valued at $54,409 is reflected in option awards; both amounts are also disclosed in the Director compensation table below.
|
Outstanding Equity Awards as of December 31, 2021
|
|
|
|
|
Option Awards
|
| Name |
|
Grant Date
|
|
Number of securities
underlying
unexercised options
(#) exercisable
|
|
|
Number of securities
underlying
unexercised options
(#) un-exercisable
|
|
|
Option exercise
price ($)
|
|
Option expiration
Date
|
|
Jennifer K. Simpson
|
|
7/17/2020
|
|
|
106,024 |
|
|
|
106,024 |
(a) |
|
|
9.99 |
|
7/17/2030
|
| |
|
3/30/2021
|
|
|
– |
|
|
|
170,000 |
(b) |
|
|
4.09 |
|
3/30/2031
|
| |
|
9/13/2021
|
|
|
– |
|
|
|
19,125 |
(c) |
|
|
2.26 |
|
9/13/2031
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Susan Horvath
|
|
4/17/2018
|
|
|
40,000 |
|
|
|
– |
|
|
|
5.75 |
|
4/17/2028
|
| |
|
5/21/2019
|
|
|
45,725 |
|
|
|
12,075 |
(d) |
|
|
2.95 |
|
5/21/2029
|
| |
|
9/24/2019
|
|
|
25,000 |
|
|
|
– |
|
|
|
5.00 |
|
9/24/2029
|
| |
|
6/24/2020
|
|
|
16,000 |
|
|
|
16,000 |
(e) |
|
|
4.98 |
|
6/24/2030
|
| |
|
3/30/2021
|
|
|
– |
|
|
|
40,000 |
(f) |
|
|
4.09 |
|
3/30/2031
|
| |
|
9/13/2021
|
|
|
– |
|
|
|
12,135 |
(g) |
|
|
2.26 |
|
9/13/2031
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michael T. Cullen
|
|
3/5/2015
|
|
|
80,000 |
|
|
|
– |
|
|
|
3.18 |
|
3/5/2025
|
| |
|
12/12/2016
|
|
|
15,000 |
|
|
|
– |
|
|
|
15.10 |
|
12/12/2026
|
| |
|
2/27/2018
|
|
|
100,000 |
|
|
|
– |
|
|
|
8.10 |
|
2/27/2028
|
| |
|
5/21/2019
|
|
|
127,900 |
|
|
|
28,200 |
(h) |
|
|
2.95 |
|
5/21/2029
|
| |
|
9/24/2019
|
|
|
30,000 |
|
|
|
– |
|
|
|
5.00 |
|
9/24/2029
|
| |
|
6/24/2020
|
|
|
25,000 |
|
|
|
25,000 |
(i) |
|
|
4.98 |
|
6/24/2030
|
| |
|
3/30/2021
|
|
|
– |
|
|
|
55,000 |
(j) |
|
|
4.09 |
|
3/30/2031
|
| |
|
5/25/2021
|
|
|
9,276 |
|
|
|
3,092 |
(k) |
|
|
4.17 |
|
5/25/2021
|
|
(a)
|
Scheduled to vest with respect to 53,012 on July 17th in each of 2022 and 2023.
|
|
(b)
|
Scheduled to vest with respect to 56,667 on March 30th in each of 2022, 2023 and 2024.
|
|
(c)
|
Scheduled to vest with respect to 6375 on September 13th in each of 2022, 2023 and 2024.
|
|
(d)
|
Scheduled to vest with respect to 12,075 on May 21, 2022.
|
|
(e)
|
Scheduled to vest with respect to 8,000 on June 24th in each of 2022 and 2023.
|
|
(f)
|
Scheduled to vest with respect to 13,333 on March 30th in each of 2022, 2023 and 2024.
|
|
(g)
|
Scheduled to vest with respect to 4,045 on September 13th in each of 2022, 2023 and 2024.
|
|
(h)
|
Scheduled to vest with respect to 28,200 on May 21, 2022.
|
|
(i)
|
Scheduled to vest with respect to 12,500 on June 24th in each of 2022 and 2023.
|
|
(j)
|
Scheduled to vest with respect to 18,333 on March 30th in each of 2022, 2023 and 2024.
|
|
(k)
|
Scheduled to vest with respect to 3,092 on May 25, 2022.
|
Cash Incentive Compensation
For 2020 and 2021, the Compensation Committee established performance objectives for each of the Executives based on clinical development and financial milestones. Each Executive’s potential payment upon satisfaction of the objectives was equal to the target set forth in the Executive’s employment agreement as described further below. In the first quarter of 2021, the Compensation Committee determined that all of 2020 objectives were achieved and approved payment at target for each Executive. In the first quarter of 2022, the Compensation Committee determined that Dr. Simpson’s bonus for 2021 was approved for payment at 76.55% of target and Ms. Horvath’s bonus was approved for payment at 82.41% of target. The 2021 incentive was paid in the first quarter of 2022. No cash bonus was paid or will be paid to Dr. Cullen in 2022 as the Company’s plan requires that employees are employed as of the end of the year to be eligible for a bonus.
Employment Agreements
During 2021, we were party to employment agreements with each of the Executives. In addition to the specific terms summarized below, each Executive is eligible to participate in the other compensation and benefit programs generally available to our employees, including our other executive officers, if any. Each such employment agreement also includes customary non-competition and non-solicitation covenants and a requirement that the Executive execute a supplemental agreement regarding confidentiality and assignment of intellectual property.
In accordance with the employment agreements, the base salary of each Executive is reviewed annually by the Compensation Committee of our Board of Directors. Pursuant to the employment agreements, the committee may authorize an increase for the applicable year but may not reduce an Executive’s base salary below its then-current level other than with the Executive’s consent or pursuant to a general wage reduction in respect of substantially all of our executive officers. As discussed above, the Compensation Committee established performance criteria for 2021 and, based upon achievement of those objectives, cash payments were approved and paid in the first quarter of 2022.
President and Chief Executive Officer
Under her employment agreement, Dr. Simpson is eligible to receive an annual performance-based cash bonus with a target amount equal to no less than 50% of her base salary. Payment of the bonus amount is subject to achievement of metrics to be established by our Board of Directors and her continued employment with the Company through the end of the applicable cash bonus period.
Chief Financial Officer
Under her employment agreement, Ms. Horvath is eligible to receive an annual performance-based cash bonus with a target amount equal to no less than 40% of her base salary. Payment of the bonus amount is subject to achievement of metrics to be established by our Board of Directors and her continued employment with the Company through the end of the applicable cash bonus period.
Former Executive Chairman
Dr. Cullen voluntarily retired from his employment with the Company in May of 2021. Under his employment agreement, Dr. Cullen had been eligible to receive an annual performance-based cash bonus with a target amount equal to no less than 45% of his base salary. Payment of the bonus amount was subject to achievement of metrics to be established by our Board of Directors and his continued employment with the Company through the end of the applicable cash bonus period.
Potential Payments Upon Termination or Change-in-Control
Under their respective employment agreements, if any of the Executive’s employment is terminated by us for any reason other than for “cause” (as defined in the applicable employment agreement) or by him or her for “good reason” (as defined in the applicable employment agreement), then he or she will be eligible to receive an amount equal to their respective annualized salary plus an amount equal to a prorated portion of their cash bonus target, if any, for the year in which the termination occurred, in addition to other amounts accrued on or before the date of termination. If any such termination occurs within six months prior or two years after a “change of control” (as defined in the applicable employment agreement), then the Executive would instead receive an amount equal to his or her respective annualized salary, plus an amount equal to his or her full cash bonus target for the year in which the termination occurred.
Director Compensation
The following table sets forth certain information regarding compensation of the persons who served as non-employee directors during the most recent completed fiscal year.
|
Name
|
|
Fees Earned or
Paid in Cash
($)
|
|
|
Stock Awards (a)
($)
|
|
|
Option Awards (b)
($)
|
|
|
Total
($)
|
|
|
Michael T. Cullen (c)
|
|
|
39,375 |
|
|
|
17,998 |
|
|
|
36,411 |
|
|
|
93,784 |
|
|
Arthur J. Fratamico (d)
|
|
|
43,752 |
|
|
|
17,998 |
|
|
|
36,411 |
|
|
|
98,161 |
|
|
Jeffrey S. Mathiesen (e)
|
|
|
73,752 |
|
|
|
17,998 |
|
|
|
36,411 |
|
|
|
128,161 |
|
|
Paul W. Schaffer (f)
|
|
|
54,996 |
|
|
|
17,998 |
|
|
|
36,411 |
|
|
|
109,405 |
|
|
D. Robert Schemel (g)
|
|
|
52,500 |
|
|
|
17,998 |
|
|
|
36,411 |
|
|
|
106,909 |
|
|
(a)
|
The values of stock awards, or restricted stock units, in this table represent the fair value of such awards granted during the fiscal year, as computed in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718. The assumptions used to determine the valuation of the awards are discussed in Note 9 to our consolidated financial statements, included in our annual report on Form 10-K for the fiscal year ended December 31, 2021.
|
|
(b)
|
The values of option awards in this table represent the fair value of such awards granted during the fiscal year, as computed in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718. The assumptions used to determine the valuation of the awards are discussed in Note 9 to our consolidated financial statements, included in our annual report on Form 10-K for the fiscal year ended December 31, 2021.
|
|
(c)
|
Dr. Cullen’s non-employee director compensation is also included in the named executive compensation table above. Dr. Cullen held unvested restricted stock units of 2,158 and options to purchase an aggregate of 498,468 shares as of December 31, 2021
|
|
(d)
|
Mr. Fratamico held unvested restricted stock units of 2,158 and options to purchase an aggregate of 57,668 shares as of December 31, 2021.
|
|
(e)
|
Mr. Mathiesen held unvested restricted stock units of 2,158 and options to purchase an aggregate of 80,668 shares as of December 31, 2021.
|
|
(f)
|
Mr. Shaffer held unvested restricted stock units of 2,158 and options to purchase an aggregate of 96,668 shares as of December 31, 2021.
|
|
(g)
|
Mr. Schemel held unvested restricted stock units of 2,158 and options to purchase an aggregate of 80,668 shares as of December 31, 2021.
|
Directors who are also our employees or who are providing consulting services receive no additional cash compensation for serving on our Board of Directors. Director, Dr. Suzanne Gagnon, was an employee of the Company until her voluntary retirement in July of 2021. At her departure, Dr. Gagnon, became a consultant of the company and as such was not entitled to any additional compensation for serving on the Board of Directors. During 2021, our Company reimbursed non-employee directors for out-of-pocket expenses incurred in connection with attending meetings of our Board of Directors and its committees.
In December 2020, the Compensation Committee approved an update to the compensation of our non-employee directors for 2021. The new program provided cash compensation based on the general responsibilities and committee memberships held by each director. The total annual amounts were paid to directors monthly. In addition, on that same date, the Compensation Committee approved the issuance of restricted stock units (RSUs) to each non-employee director. The number of RSUs granted to each director was 2,875 and they vested in 5 equivalent increments beginning January 2021 through May 2021. In May 2021, the Compensation Committee approved the issuance of RSU’s to each non-employee director. The number of RSUs granted to each director was 4,316 and they are scheduled to vest in 4 equivalent increments beginning August 2021 through May 2022. Also in May 2021, the Compensation Committee approved the issuance of stock options to each non-employee director. The number of RSUs granted to each director was 12,368 and they are scheduled to vest in 4 equivalent increments beginning August 2021 through May 2022.
In February 2022, the Compensation Committee approved an update to the cash compensation for non-employee directors. The revised annual amounts described below were effective January 1, 2022 and will be paid out monthly.
|
Annual Retainer
(all amounts in $)
|
|
General
|
|
|
Audit Committee
|
|
|
Nominating &
Governance
Committee
|
|
|
Compensation
Committee
|
|
|
Nonemployee director
|
|
|
40,000 |
|
|
‐
|
|
|
‐
|
|
|
‐
|
|
|
Chairman
|
|
|
32,500 |
(a) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Lead independent director
|
|
|
22,500 |
(b) |
|
‐
|
|
|
‐
|
|
|
‐
|
|
|
Committee chair
|
|
‐
|
|
|
|
15,000 |
|
|
|
7,500 |
|
|
|
10,000 |
|
|
Committee member
|
|
‐
|
|
|
|
7,500 |
|
|
|
4,000 |
|
|
|
5,000 |
|
|
(a)
|
Paid in addition to nonemployee director retainer.
|
|
(b)
|
Paid in addition to nonemployee director retainer.
|
Certain Relationships and Related Party Transactions
The following is a summary of transactions since January 1, 2020 to which our Company has been a party and in which the amount involved exceeded $113,000, which is approximately 1% of the average of our total assets as of the ends of our last two completed fiscal years, and in which any of our directors, executive officers, or beneficial owners of more than 10% of our capital stock had or will have a direct or indirect material interest, other than the compensation arrangements that are described under the heading “Executive Compensation: Employment Agreements” in Item 11.
On June 25, 2022, Cancer Prevention entered into a Separation and Release Agreement (the “Separation Agreement”) with Mr. Jacob, now a director of Panbela, whereby he resigned as its Chief Executive Officer, employee, and all other capacities, immediately prior to the closing under the Merger Agreement. In consideration for Mr. Jacob’s acknowledgements, representations, warranties, covenants, releases and agreements set forth in the Separation Agreement, Cancer Prevention has agreed to pay to Mr. Jacob a total of $350,000, representing one times his base salary at the time of his resignation. Such payment will become due upon the earlier of (i) Cancer Prevention or its parent completing a material financing and (ii) the two-year anniversary of the Closing Date. As further consideration, Cancer Prevention has also agreed to reimburse Mr. Jacob for the employer’s portion of the premium payments for him to continue his current medical insurance coverage for 12 months through the Consolidated Omnibus Budget Reconciliation Act (COBRA).
Dr. Suzanne Gagnon was the Company’s Chief Medical Officer until her retirement in July of 2021. Dr. Gagnon remains a member of our Board of Directors. We were party to an employment agreement with Dr. Gagnon in substantially the same form as the employment agreements with the Executives described above under the heading “Executive Compensation: Employment Agreements” in Item 11. Dr. Gagnon was eligible to participate in the other compensation and benefit programs generally available to our employees. Her employment agreement also included customary confidentiality, non-competition and non-solicitation covenants. Under the employment agreement in effect through her voluntary retirement, Dr. Gagnon was entitled to receive an annualized base salary $360,000. During 2020 and 2021, Dr. Gagnon received compensation from the Company amounting to $197,800 and $293,500, respectively. In addition, in February 2021, based on the achievement of established metrics for 2020, Dr. Gagnon received a cash bonus of $117,000. No cash bonus was paid or will be paid to Dr. Gagnon in 2022 as the Company’s plan requires that employees are employed as of the end of the year to be eligible for a bonus.
In July 2021, after approval by our Audit Committee, we entered into a consulting contract with Dr. Gagnon. The services to be provided by Dr. Gagnon include her professional support to complete the final study report for the Phase Ia/Ib clinical trial and additional support as a medical consultant for the clinical and administrative teams. The contract provides for a monthly retainer of $14,000 representing approximately eight hours per week for the first three months of the agreement; for the remainder of the term Dr. Gagnon shall be paid $400 per hour for all services provided. The contract will expire in July of 2023 but may be terminated early by either party or extended if mutually agreed upon. For the year ended December 31, 2021 Dr. Gagnon was paid approximately $54,600 in professional consulting fees.
Limitation of Liability of Directors and Officers and Indemnification
Our certificate of incorporation limits the liability of the directors to the fullest extent permitted by Delaware law.
Our bylaws provide that we will indemnify and advance expenses to the directors and officers to the fullest extent permitted by law or, if applicable, pursuant to indemnification agreements. They further provide that we may choose to indemnify other employees or agents of our Company from time to time. The Delaware General Corporation Law and the bylaws also permit us to secure insurance on behalf of any officer, director, employee or other agent for any liability arising out of his or her actions in connection with their services to our Company, regardless of whether the bylaws permit indemnification. We maintain a directors’ and officers’ liability insurance policy.
At present there is no pending litigation or proceeding involving any of the current or former directors or officers as to which indemnification is required or permitted, and we are not aware of any threatened litigation or proceeding that may result in a claim for indemnification.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the SEC this indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
Related Person Transaction Approval Policy
Our Board of Directors has adopted a written policy regarding transactions with related persons, which we refer to as our related party transaction approval policy. Our related party transaction approval policy requires that any executive officer proposing to enter into a transaction with a “related party” generally must promptly disclose to our Audit Committee the proposed transaction and all material facts with respect thereto. In reviewing a transaction, our Audit Committee will consider all relevant facts and circumstances, including (1) the commercial reasonableness of the terms, (2) the benefit and perceived benefits, or lack thereof, to us, (3) the opportunity costs of alternate transactions and (4) the materiality and character of the related party’s interest, and the actual or apparent conflict of interest of the related party.
Our Audit Committee will not approve or ratify a related party transaction unless it determines that, upon consideration of all relevant information, the transaction is beneficial to our Company and stockholders and the terms of the transaction are fair to our Company. No related party transaction will be consummated without the approval or ratification of our Audit Committee. It will be our policy that a director will recuse him- or herself from any vote relating to a proposed or actual related party transaction in which they have an interest. Under our related party transaction approval policy, a “related party” includes any of our directors, director nominees, executive officers, any beneficial owner of more than 5% of our common stock and any immediate family member of any of the foregoing. Related party transactions exempt from our policy include transactions available to all of our employees and stockholders on the same terms and transactions between us and the related party that, when aggregated with the amount of all other transactions between us and the related party or its affiliates, involved less than one percent of the average of our Company’s total assets at yearend for the last two completed fiscal years.
Director Independence
The continued listing rules of The Nasdaq Stock Market, LLC (the “Nasdaq Rules”) require that a majority of our Board of Directors be “independent directors” as that term is defined in the Nasdaq Rules. Our Board has determined that each of our non-employee directors, namely Messrs. Fratamico, Mathiesen, Schaffer, and Schemel, are “independent directors.”
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information with respect to the beneficial ownership of our outstanding common stock as of June 16, 2022 by (i) each of our named executive officers identified in the Summary Compensation Table below; (ii) each of our directors; (iii) all of our executive officers, directors and director nominees as a group; and (iv) each other beneficial owner of 5% or more of our outstanding common stock. Ownership percentages are based on 20,774,045 shares of common stock outstanding as of the close of business on the same date. Beneficial ownership is determined in accordance with the rules of the SEC. To our knowledge and subject to applicable community property laws, each of the holders of stock listed below has sole voting and investment power as to the stock owned unless otherwise noted. The table below includes the number of shares underlying rights to acquire common stock that are exercisable within 60 days from June 26, 2022. Except as otherwise noted below, the address for each director or officer listed in the table is c/o Panbela Therapeutics, Inc., 712 Vista Blvd #305, Waconia, Minnesota 55387.
|
Name
|
|
Amount and Nature of
Beneficial Ownership
|
|
|
Percentage of Outstanding
Shares*
|
|
|
Executive Officers and Directors
|
|
|
|
|
|
|
|
|
|
Jennifer K. Simpson
|
|
|
221,551 |
(a) |
|
|
1.1 |
% |
|
Susan Horvath
|
|
|
193,422 |
(b) |
|
|
* |
|
|
Michael T. Cullen
|
|
|
940,689 |
(c) |
|
|
4.4 |
% |
|
Daniel J. Donovan
|
|
|
375,036 |
(d) |
|
|
1.8 |
% |
|
Arthur J. Fratamico
|
|
|
69,707 |
(e) |
|
|
* |
|
|
Jeffrey E. Jacob
|
|
|
685,797 |
(f) |
|
|
3.2 |
% |
|
Jeffrey S. Mathiesen
|
|
|
90,859 |
(g) |
|
|
* |
|
|
D. Robert Schemel
|
|
|
471,691 |
(h) |
|
|
2.3 |
% |
|
All directors and current executive officers as a group (8 persons)
|
|
|
3,048,752 |
(i) |
|
|
13.7 |
% |
| |
|
|
|
|
|
|
|
|
|
Sucampo GmbH
|
|
|
1,722,329 |
|
|
|
8.3 |
% |
| c/o ST Shared Services |
|
|
|
|
|
|
|
|
| 675 McDonnell Boulevard |
|
|
|
|
|
|
|
|
| Hazelwood, Missouri 63042 |
|
|
|
|
|
|
|
|
|
(a)
|
Includes 1,000 shares held by spouse, 215,703 shares subject to stock options and 2,424 shares subject to warrants.
|
|
(b)
|
Includes 160,134 shares subject to stock options and 19,852 shares subject to warrants.
|
|
(c)
|
Includes 204,576 shares held by the Cullen Living Trust. 449,302 shares subject to stock options and 70,000 shares subject to warrants.
|
|
(d)
|
Includes 57,668 shares subject to stock options. Also includes 75,528 shares held by Westport Boys, LLC (“Westport”), 163,256 shares held by GDB Investments, LLP (“GDB”), and 5,183 shares subject to a warrant held by GDB Investments, LLP. Mr. Donovan is a managing member of Westport and a designated member of GDB. Mr. Donovan disclaims beneficial ownership of the securities owned by Westport and GDB except to the extent of his pecuniary interest therein.
|
|
(e)
|
Includes 57,668 shares subject to stock options and 2,424 shares subject to warrants.
|
|
(f)
|
Includes 17,771 shares held jointly with spouse and 346,547 shares subject to stock options. Also includes, 54,374 shares and 5,174 shares subject to a warrant, in each case held by the Jeffrey and Debora Jacob Family Revocable Trust.
|
|
(g)
|
Includes 80,668 shares subject to options.
|
|
(h)
|
Includes 282,654 shares held by spouse, 80,668 shares subject to stock options, and 11,750 shares held by parent’s estate over which director holds both voting and depository power but disclaims beneficial ownership.
|
|
(i)
|
Includes 1,450,224 shares subject to stock options and 105,057 shares subject to warrants.
|
DESCRIPTION OF SECURITIES
The summary of the general terms and provisions of the common stock, par value $0.001 per share (“Common Stock”), of Panbela set forth below does not purport to be complete and is subject to and qualified by reference to the Corporation’s Certificate of Incorporation, as amended (the “Certificate”), and Bylaws of the Corporation, as amended (the “Bylaws”). For additional information, please read the Certificate, Bylaws and the applicable provisions of the General Corporation Law of Delaware (the “DGCL”).
Authorized Shares
The Corporation is authorized to issue up to 110,000,000 shares of capital stock, of which 100,000,000 constitute shares of Common Stock and 10,000,000 constitute shares of preferred stock, par value $0.001 per share (“Preferred Stock”).
Common Stock
No outstanding shares of common stock is entitled to preference over any other share, and each share is equal to any other share in all respects. Holders of shares of common stock are entitled to one vote for each share held of record at each meeting of shareholders. Holders of shares of common stock are not entitled to any preemptive, subscription, conversion, redemption or sinking fund rights. The absence of preemptive rights could result in a dilution of the interest of shareholders should additional common shares be issued.
Subject to any prior rights of any Preferred Stock then outstanding, holders of common stock are entitled to receive dividends in the form of cash, property or shares of capital stock of the Corporation, when and as declared by the board of directors, provided there are sufficient net profits or surplus legally available for that purpose. In any distribution of capital assets, such as liquidation, whether voluntary or involuntary, holders of shares of common stock are entitled to receive pro rata the assets remaining after creditors have been paid in full. All of the issued and outstanding shares of common stock are non-assessable.
Anti-Takeover Provisions
The Charter Documents and the DGCL contain certain provisions that may discourage an unsolicited takeover of the Company or make an unsolicited takeover of the Company more difficult. The following are some of the more significant anti-takeover provisions that are applicable to the Company:
Delaware Anti-Takeover Law
In general, Section 203 of the DGCL prohibits a Delaware corporation with a class of voting stock listed on a national securities exchange or held of record by 2,000 or more stockholders from engaging in a Business Combination (as defined below) with an Interested Stockholder (as defined below) for a three-year period following the time that this stockholder becomes an interested stockholder, unless the Business Combination is approved in a prescribed manner. A “Business Combination” includes, among other things, a merger, asset or stock sale or other transaction resulting in a financial benefit to the Interested Stockholder. An “Interested Stockholder” is a person who, together with affiliates and associates, owns, or did own within three years prior to the determination of Interested Stockholder status, 15% or more of the corporation’s voting stock. Under Section 203, a Business Combination between a corporation and an Interested Stockholder is prohibited for three years unless it satisfies one of the following conditions:
| |
●
|
Before the stockholder became an Interested Stockholder, the board of directors approved either the Business Combination or the transaction which resulted in the stockholder becoming an Interested Stockholder;
|
| |
●
|
Upon consummation of the transaction which resulted in the stockholder becoming an Interested Stockholder, the Interested Stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding, shares owned by persons who are directors and also officers, and employee stock plans, in some instances; or
|
| |
●
|
At or after the time the stockholder became an Interested Stockholder, the Business Combination was approved by the board of directors of the corporation and authorized at an annual or special meeting of the stockholders by the affirmative vote of at least two-thirds of the outstanding voting stock which is not owned by the Interested Stockholder.
|
Requirements for Advance Notification of Stockholder Nominations and Proposals
The Bylaws establish advance-notice procedures with respect to stockholder proposals to be brought before a stockholder meeting and the nomination of candidates for election as directors, other than nominations made by or at the direction of the board of directors.
Special Meetings of Stockholders
The Certificate and Bylaws provide that a special meeting of stockholders may be called only by the board of directors, the Chairman of the Board or the Chief Executive Officer of the Corporation.
Classified Board of Directors
The Certificate provides that directors are divided into three classes and elected for staggered terms. At each annual meeting, approximately one third of the directors will be elected to serve a three-year term. Directors serving staggered terms can be removed from office only for cause and only by the affirmative vote of the holders of 75% or more of the outstanding shares of stock then entitled to vote at an election of directors.
Authority of the Board of Directors
The board of directors has the power to issue any or all of the shares of the Corporation’s capital stock, including the authority to establish one or more series of Preferred Stock and to fix the powers, preferences, rights and limitations of such class or series, without seeking stockholder approval. The board of directors has the authority to adopt and change Bylaws, subject to the right of holders of at least 66.67% of the voting power of all then-outstanding shares entitled to vote generally in the election of directors to adopt, amend or repeal Bylaws.
Preferred Stock
Our Board of Directors has the authority, without first obtaining the approval of our stockholders, to establish one or more series of preferred stock and to fix:
| |
●
|
the number of shares of such series;
|
| |
●
|
the designations, preferences and relative rights, including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences; and
|
| |
●
|
any qualifications, limitations or restrictions.
|
We believe that the ability of our Board of Directors to issue one or more series of preferred stock provides flexibility in structuring possible future financings and acquisitions, and in meeting other corporate needs that may arise. The authorized shares of preferred stock, as well as authorized and unissued shares of common stock, are available for issuance without action by the holders of common stock, unless such action is required by applicable law or the rules of any stock exchange or automated quotation system on which our securities may be listed or traded.
Our Board of Directors may authorize, without stockholder approval, the issuance of preferred stock with voting and conversion rights that could adversely affect the voting power and other rights of holders of common stock. Although our Board of Directors has no current intention of doing so, it could issue a series of preferred stock that could, depending on the terms of such series, impede the completion of a merger, tender offer or other takeover attempt of our Company. Our Board of Directors could also issue preferred stock having terms that could discourage an acquisition attempt through which an acquirer may be able to change the composition of our Board of Directors, including a tender offer or other transaction that some, or a majority, of the stockholders might believe to be in their best interests or in which stockholders might receive a premium for their stock over the then-current market price. Any issuance of preferred stock therefore could have the effect of decreasing the market price of our common stock.
Our Board of Directors will make any determination to issue such shares based on its judgment as to the best interests of our Company and stockholders. We have no current plans to issue any preferred stock.
Options
The 2016 Plan initially authorized the issuance of up to 2,800,000 shares of our common stock pursuant to awards granted thereunder and 1,472,992 shares have been added pursuant to its annual evergreen feature. As of June 16, 2022, options to purchase 2,220,136 shares of our common stock were outstanding under the 2016 Plan with a weighted average price of $6.04 per share. A total of 2,019,776, shares of common stock remained available for future grants under the 2016 Plan as of the same date.
As of June 16, 2022, options to purchase 224,000 shares of our common stock remained outstanding under the 2011 Plan with a weighted average price of $2.97 per share. We ceased making awards under the 2011 Plan upon stockholder approval of the 2016 Plan.
As of June 16, 2022, options to purchase 1,596,754 shares of our common stock remained outstanding under Cancer Prevention’s 2010 Equity Incentive Plan, all of which were assumed by us in connection with the Mergers.
Warrants Outstanding
As of June 16, 2022, we had issued and outstanding warrants to purchase 5,447,561 shares of common stock and no warrants to purchase shares of preferred stock outstanding. As of the same date, the outstanding warrants had a weighted average exercise price of $4.56 per share and an average remaining exercise period of 2.56 years.
Warrants Issued in the Offering
The following is a brief summary of certain terms and conditions of the warrants that were issued in connection with this offering and are subject in all respects to the provisions contained in the warrants.
Form
The warrants were issued in electronic book-entry form to the investors. A copy of the form of warrant, which is filed as an exhibit to the registration statement of which this prospectus forms a part, contains the terms and conditions applicable to the warrants.
Exercisability
The warrants are exercisable at any time after their original issuance, which was September 1, 2020, and at any time up to the date that is five years after their original issuance. The warrants are exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed exercise notice and, at any time a registration statement registering the issuance of the shares of common stock underlying the warrants under the Securities Act is effective and available for the issuance of such shares, or an exemption from registration under the Securities Act is available for the issuance of such shares, by payment in full in immediately available funds for the number of shares of common stock purchased upon such exercise. If a registration statement registering the issuance of the shares of common stock underlying the warrants under the Securities Act is not effective or available and an exemption from registration under the Securities Act is not available for the issuance of such shares, the holder may, in its sole discretion, elect to exercise the warrant through a cashless exercise, in which case the holder would receive upon such exercise the net number of shares of common stock determined according to the formula set forth in the warrant. No fractional shares of common stock will be issued in connection with the exercise of a warrant. In lieu of fractional shares, we will pay to the holder an amount in cash equal to the fractional amount multiplied by the exercise price or round up to the next whole share.
Exercise Limitation
A holder will not have the right to exercise any portion of the warrant if the holder (together with its affiliates) would beneficially own in excess of 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the warrants. However, any holder may increase or decrease such percentage to any other percentage not in excess of 9.99% upon at least 61 days’ prior notice from the holder to us.
Exercise Price
The exercise price per whole share of common stock purchasable upon exercise of the warrants is $4.54 per share. The exercise price is subject to appropriate adjustment in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting our common stock and also upon any distributions of assets, including cash, stock or other property to our stockholders.
Transferability
Subject to applicable laws, the warrants may be offered for sale, sold, transferred or assigned without our consent.
Fundamental Transactions
In the event of a fundamental transaction, as described in the warrants and generally including any reorganization, recapitalization or reclassification of our common stock, the sale, transfer or other disposition of all or substantially all of our properties or assets, our consolidation or merger with or into another person, the acquisition of more than 50% of our outstanding common stock, or any person or group becoming the beneficial owner of 50% of the voting power represented by our outstanding common stock, the holders of the warrants will be entitled to receive upon exercise of the warrants the kind and amount of securities, cash or other property that the holders would have received had they exercised the warrants immediately prior to such fundamental transaction. Additionally, as more fully described in the warrants, in the event of certain fundamental transactions, the holders of the warrants will be entitled to receive consideration in an amount equal to the Black Scholes value of the warrants determined according to a formula set forth in the warrants.
Warrant Agent
The warrants were issued in registered form under a warrant agency agreement between VStock Transfer, as warrant agent, and us.
Rights as a Stockholder
Except as otherwise provided in the warrants or by virtue of such holder’s ownership of shares of our common stock, the holder of a warrant does not have the rights or privileges of a holder of our common stock, including any voting rights, until the holder exercises the warrant.
Underwriter's Warrants
We issued to the underwriter at the Initial Closing of the offering of common stock and warrants originally covered by the registration statement of which this prospectus forms a part, to purchase up to 127,273 shares of common stock, which was equal to 5.0% of the number of shares sold in the Initial Closing. The Underwriter’s Warrants are exercisable at any time and from time to time, in whole or in part, during the five year period commencing on September 1, 2020 at an exercise price of $4.537 per share. The Underwriter’s Warrants were deemed compensation by the Financial Industry Regulatory Authority, or FINRA, and were therefore subject to a 180-day lock-up that has since expired.
Transfer Agent and Registrar
The transfer agent and registrar for our common stock is VStock Transfer, which can be contacted at 18 Lafayette Place, Woodmere, New York, 11598, info@vstocktransfer.com, or +1 (212) 828-8436.
Electronic Distribution
This prospectus in electronic format may be made available on websites or through other online services maintained by us or our affiliates. Other than this prospectus in electronic format, the information on such websites and any information contained in any other website maintained by us is not part of this prospectus or the registration statement of which this prospectus forms a part, has not been approved and/or endorsed by us, and should not be relied upon by investors.
SHARES ELIGIBLE FOR FUTURE SALE
Future sales of substantial amounts of our common stock in the public market, including shares issued upon exercise of outstanding options and warrants, or the anticipation of these sales, could adversely affect prevailing market prices from time to time and could impair our ability to raise equity capital in the future.
Upon the completion of this offering, we will have a total of 23,207,834 shares of our common stock outstanding assuming the Warrants are exercised in full, based on the 20,774,045 shares of our common stock outstanding as of June 16, 2022. Of these outstanding shares, all of the shares sold in the offering will be freely tradable, except that any shares purchased in this offering by our affiliates, as that term is defined in Rule 144 under the Securities Act, would only be able to be sold in compliance with the limitations described below. In addition, we expect that the warrants and the shares issued upon exercise of the warrants issued in this offering will be freely tradeable except for any such warrants or shares issued to our affiliates, which would also only be able to be sold in compliance with Rule 144.
Rule 144
In general, under Rule 144 as currently in effect, once we have been subject to public company reporting requirements for at least 90 days, a person who is not deemed to have been one of our affiliates for purposes of the Securities Act at any time during the 90 days preceding a sale and who has beneficially owned the shares proposed to be sold for at least six months, including the holding period of any prior owner other than our affiliates, is entitled to sell those shares without complying with the manner of sale, volume limitation or notice provisions of Rule 144, subject to compliance with the public information requirements of Rule 144. If such a person has beneficially owned the shares proposed to be sold for at least one year, including the holding period of any prior owner other than our affiliates, then that person would be entitled to sell those shares upon expiration of the lock-up agreements described below, without complying with any of the requirements of Rule 144.
In general, under Rule 144, as currently in effect, our affiliates or persons selling shares on behalf of our affiliates are entitled to sell upon expiration of the lock-up agreements described below, within any three-month period, a number of shares that does not exceed the greater of:
| |
●
|
1% of the number of shares of our common stock then outstanding, which equaled at least 207,440 shares as of June 16, 2022; or
|
| |
●
|
the average weekly trading volume of our common stock on the Nasdaq Capital Market during the four calendar weeks preceding the filing of a notice on Form 144 with respect to the sale.
|
Sales under Rule 144 by our affiliates or persons selling shares on behalf of our affiliates are also subject to certain manner of sale provisions and notice requirements and to the availability of current public information about us.
Rule 701
Rule 701 generally allows a stockholder who purchased shares of our common stock pursuant to a written compensatory plan or contract and who is not deemed to have been an affiliate of our company during the immediately preceding 90 days to sell these shares in reliance upon Rule 144, but without being required to comply with the public information or holding period provisions of Rule 144. Rule 701 also permits affiliates of our company to sell their Rule 701 shares under Rule 144 without complying with the holding period requirements of Rule 144. All holders of Rule 701 shares, however, are required by that rule to wait until 90 days after the date of this prospectus before selling those shares pursuant to Rule 701, subject to the market standoff agreements and lock-up agreements described below.
Stock Options and Warrants
As of June 16, 2022, options to purchase a total of 4,040,890 shares of common stock were outstanding with a weighted-average exercise price of $3.63 per share and a remaining contractual life of 7.59 years. Such options were vested and exercisable with respect to 3,539,307 shares.
As of June 16, 2022, warrants to purchase a total of 5,447,561 shares of common stock at a weighted-average exercise price of $4.56 per share were outstanding. Excluding shares underlying the Warrants that are covered by this prospectus, none of the shares issuable upon exercise of our outstanding warrants are currently registered for resale.
PLAN OF DISTRIBUTION
The prices at which the shares of common stock covered by this prospectus may actually be disposed of may be at fixed prices, at prevailing market prices at the time of sale, at prices related to the prevailing market price, at varying prices determined at the time of sale or at negotiated prices.
Pursuant to the terms of the Warrants, the shares of common stock will be distributed to those holders who deliver a duly executed exercise notice and payment in full in immediately available funds for the number of shares of common stock purchased upon such exercise.
Upon receipt of proper notice by any of the holders of the Warrants that such holder desires to exercise a Warrant, we will, within the time allotted by the agreement governing the Warrant, issue instructions to our transfer agent to issue to the holder shares of common stock, free of a restrictive legend. Shares of common stock issued to affiliates upon exercise of the Warrants will be issued free of legend but will be deemed control securities.
A holder will not have the right to exercise any portion of the Warrant if the holder (together with its affiliates) would beneficially own in excess of 4.99% of the number of shares of our common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the Warrants. However, any holder may increase or decrease such percentage to any other percentage not in excess of 9.99% upon at least 61 days’ prior notice from the holder to us.
LEGAL MATTERS
The validity of the shares of common stock being offered by this prospectus has been passed upon for us by Faegre Drinker Biddle & Reath LLP.
EXPERTS
The financial statements of Panbela as of December 31, 2021 and 2020 and for the two years in the period ended December 31, 2021 incorporated in this prospectus by reference from the Company's Annual Report on Form 10-K have been audited by Cherry Bekaert LLP, an independent registered public accounting firm, as stated in their report, which is incorporated herein by reference. Such financial statements have been so incorporated in reliance upon the report of such firm given upon their authority as experts in accounting and auditing.
The financial statements of Cancer Prevention as of December 31, 2021 and 2020 and for the two years in the period ended December 31, 2021, incorporated in this prospectus by reference from the Company’s Current Report on Form 8-K have been audited by Mayer Hoffman McCann P.C., an independent public accounting firm, as stated in their report, which is incorporated herein by reference. Such financial statements have been so incorporated in reliance upon the report of such firm given upon their authority as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC a registration statement on Form S-1 under the Securities Act that registers the distribution of the securities offered under this prospectus. The registration statement, including the attached exhibits and schedules, contains additional relevant information about us and the securities. The rules and regulations of the SEC allow us to omit from this prospectus certain information included in the registration statement.
We are subject to the informational requirements of the Securities Exchange Act and are required to file annual, quarterly and current reports, proxy statements and other information with the SEC. Any information we file with the SEC, including the documents incorporated by reference into this prospectus, is also available on the SEC’s website at www.sec.gov. We also make these documents publicly available, free of charge, on our website at www.panbela.com as soon as reasonably practicable after filing such documents with the SEC. The information contained in, or that can be accessed through, our website is not part of this prospectus.
INCORPORATION OF DOCUMENTS BY REFERENCE
We have elected to incorporate by reference certain information in this prospectus pursuant to General Instruction VII of Form S-1. We have previously filed the following documents with the SEC and are incorporating them by reference into this prospectus, except for information furnished under Item 2.02 or Item 7.01 of Form 8-K, and any exhibits relating to such information, which is neither deemed filed nor incorporated by reference herein:
| |
●
|
Description of our common stock contained in Exhibit 4.1 to the Annual Report on Form 10-K for the year ended December 31, 2021, filed with the SEC on March 24, 2022.
|
As a smaller reporting company, we also are incorporating by reference any future information filed (rather than furnished) by us with the SEC under Section 13(a), 13(c), 14 or 15(d) of the Securities Exchange Act of 1934, as amended, after the date of the initial filing of the registration statement of which this prospectus is a part and before the effective date of the registration statement and after the date of this prospectus until the termination of the offering. Any statements contained in a previously filed document incorporated by reference into this prospectus is deemed to be modified or superseded for purposes of this prospectus to the extent that a statement contained in this prospectus, or in a subsequently filed document also incorporated by reference herein, modifies or supersedes that statement
Our internet address is www.panbela.com. We make available free of charge, on or through the investor relations section of our website, annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. You may access the documents incorporated by reference in this prospectus through our website at www.neurometrix.com. Except for the specific incorporated documents listed above, no information available on or through our website shall be deemed to be incorporated by reference in this prospectus or the registration statement of which it forms a part.
We hereby undertake to provide without charge to each person, including any beneficial owner, to whom a prospectus is delivered, upon written or oral request of any such person, a copy of any and all of the information that has been incorporated by reference in this prospectus, but not delivered with the prospectus. Requests for such copies should be sent to us at the following address:
Panbela Therapeutics, Inc.
712 Vista Blvd #305
Waconia, MN 55387
Attention: Investor Relations
(952) 479-1196
2,433,789 Shares of Common Stock
Panbela Therapeutics, Inc.
PROSPECTUS
, 2022
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution.
The following table sets forth the costs and expenses, other than any estimated underwriting discounts and commissions, payable by Panbela Therapeutics, Inc. (the “Company”) in connection with the offering and sale of the common stock being registered. All amounts shown are estimates, except the Securities and Exchange Commission (the “Commission”) registration fee.
|
U.S. Securities and Exchange Commission registration fee
|
|
$ |
3,441 |
|
|
Accounting fees and expenses
|
|
$ |
20,000 |
|
|
Legal fees and expenses
|
|
$ |
50,000 |
|
|
Transfer agent and registrar fees
|
|
$ |
10,000 |
|
|
Printing expenses
|
|
$ |
10,000 |
|
|
Miscellaneous
|
|
$ |
6,559 |
|
|
Total
|
|
$ |
100,000 |
|
Item 14. Indemnification of Directors and Officers.
Section 145 of the Delaware General Corporation Law provides that a corporation may indemnify any person made a party to an action by reason of the fact that he or she was a director, executive officer, employee or agent of the corporation or is or was serving at the request of the corporation against expenses (including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by him or her in connection with such action if he or she acted in good faith and in a manner he or she reasonably believed to be in, or not opposed to, the best interests of the corporation and, with respect to any criminal action or proceeding, had no reasonable cause to believe his or her conduct was unlawful, except that, in the case of an action by or in right of the corporation, no indemnification may generally be made in respect of any claim as to which such person is adjudged to be liable to the corporation.
The Company’s certificate of incorporation and amended and restated bylaws limit the liability of its directors to the fullest extent permitted by Delaware law. Delaware law provides that directors of a corporation will not be personally liable for monetary damages for breach of their fiduciary duties as directors, except for liability for any:
| |
●
|
breach of their duty of loyalty to the Company or its stockholders;
|
| |
●
|
act or omission not in good faith or that involves intentional misconduct or a knowing violation of law;
|
| |
●
|
unlawful payment of dividends or redemption of shares as provided in Section 174 of the Delaware General Corporation Law; or
|
| |
●
|
transaction from which the directors derived an improper personal benefit.
|
These limitations of liability do not apply to liabilities arising under federal securities laws and do not affect the availability of equitable remedies such as injunctive relief or rescission. The Company’s amended and restated bylaws provide that it will indemnify its directors and executive officers, and may indemnify other officers, employees and other agents, to the fullest extent permitted by law.
As permitted by the Delaware General Corporation Law, the Company has entered into indemnification agreements with each of the Company’s directors and executive officers that require the Company to indemnify such persons against expenses, judgments, penalties, fines, settlements and other amounts actually and reasonably incurred, including expenses of a derivative action, in connection with an actual or threatened proceeding if any of the Company’s directors or executive officers may be made a party because he or she is or was one of the Company’s directors. The Company will be obligated to pay such amounts only if the director acted in good faith and in a manner that he or she reasonably believed to be in or not opposed to the Company’s best interests. With respect to any criminal proceeding, the Company will be obligated to pay such amounts only if the director had no reasonable cause to believe his or her conduct was unlawful. The indemnification agreements also set forth certain procedures that will apply in the event of a claim for indemnification.
Section 145(g) of the Delaware General Corporation Law permits a corporation to purchase and maintain insurance on behalf of any person who is or was a director, officer, employee, or agent of the corporation arising out of his or her actions in connection with their services to the Company, regardless of whether its amended and restated bylaws permit indemnification. The Company has purchased and intends to maintain insurance on behalf of any person who is or was a director or officer against any loss arising from any claim asserted against him or her and incurred by him or her in any such capacity, subject to certain exclusions.
Item 15. Recent Sales of Unregistered Securities.
On June 30, 2019, the Company issued 651,758 shares of common stock upon the conversion of unsecured convertible promissory notes totaling approximately $2.3 million principal and accrued interest at a conversion rate of $3.50 per share per the terms of the notes. The notes provided for a mandatory conversion into common stock on the earlier of (1) June 30, 2019 or (2) the date the Company received gross proceeds of at least $6.0 million from the sale of equity securities (subject to certain exclusions) and bore interest at a rate of 10.0% per year. The shares were issued in reliance on the exemption from registration set forth in Section 3(a)(9) of the Securities Act as securities exchanged by an issuer with existing security holders where no commission or other remuneration is paid or given directly or indirectly by the issuer for soliciting such exchange.
On August 23, August 30, September 20 and October 10, 2019, the Company entered into securities purchase agreements and sold an aggregate of (i) 909,209 shares of its common stock (the “Shares”) and (ii) warrants to purchase up to 909,209 additional shares of common stock for total gross proceeds of approximately $3.2 million. The warrants are exercisable for a period of five years from the date of issuance at an initial exercise price of $4.00 per share.
On February 21, 2020, the Company issued to the underwriter in the public offering a five-year warrant to purchase 75,000 shares at an exercise price of $6.49 per share.
In closings on May 22, June 5, June 15, and June 22, 2020, the Company sold an aggregate of 437,000 shares of its common stock and warrants to purchase up to 437,000 additional shares of common stock for aggregate gross proceeds of approximately $1.7 million, of which approximately $90,000 was received from officers and directors of the Company. The warrants are exercisable for a period of five years from the date of issuance at an initial exercise price of $6.00 per share.
On September 1, 2020, the Company issued 35,665 shares of common stock as a result of the exercise of outstanding warrants that were set to expire as a result of the public offering. All of the warrants were exercised at $1.875 per share. Of the shares issued, 27,500 were issued for approximately $52,000 cash. One warrant to purchase 15,000 shares of common stock was exercised on a net, cashless basis, resulting in the issuance of the remaining 8,165 shares.
During the three months ended March 31, 2021, the Company issued 193,607 shares of common stock as a result of exercises of outstanding warrants. Of the shares of common stock issued, 188,607 shares were issued pursuant to net, cashless, exercises of warrants to purchase 531,140 shares and the remaining 5,000 shares were issued for $25,000 cash.
The net cash proceeds for each of the foregoing sales of securities were used for the continued clinical development of our initial product candidate SBP-101 and for working capital and other general corporate purposes.
On June 14, 2022, pursuant to the Merger Agreement, Panbela sold and issued the following securities to the holders of Cancer Prevention securities pursuant to the Second Merger: (a) 6,587,576 shares of Panbela Common Stock, (b) 731,957 shares of Panbela Common Stock that remained subject to the Holdback Escrow (as defined in the Merger Agreement), (iv) replacement options to purchase up to 1,596,754 shares of Panbela Common Stock at a weighted average purchase price of $0.35 per share, and (v) replacement warrants to purchase up to 338,060 shares of Panbela Common Stock at a weighted average purchase price of $4.145 per share.
Unless otherwise indicated, for all of the foregoing transactions, we relied on exemptions from registration set forth in Section 4(a)(2) of the Securities Act, without the use of any general solicitations or advertising to market or otherwise offer the securities for sale and all participants were “accredited investors,” as defined in Rule 501 of Regulation D as promulgated by the SEC under the Securities Act.
Item 16. Exhibits and Financial Statement Schedules.
(a) Exhibits
|
Exhibit
No.
|
|
Description
|
|
1.1
|
|
Underwriting Agreement dated August 28, 2020 (incorporated by reference to Exhibit 1.1 to current report on Form 8-K filed September 1, 2020)
|
|
2.1
|
|
Agreement and Plan of Merger, dated February 21, 2022, by and among Panbela Therapeutics, Inc., Canary Merger Holdings, Inc., Canary Merger Subsidiary I, Inc., Canary Merger Subsidiary II, Inc., Cancer Prevention Pharmaceuticals, Inc., and Fortis Advisors LLC, as Stockholder Representative (incorporated by reference to Exhibit 2.1 to current report on Form 8-K filed February 22, 2022)
|
|
3.1
|
|
Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to current report on Form 8-K filed June 16, 2022)
|
|
3.2
|
|
Bylaws (incorporated by reference to Exhibit 3.1 to current report on Form 8-K filed June 16, 2022)
|
|
Exhibit
No.
|
|
Description |
|
4.1
|
|
Description of Securities (incorporated by reference to Exhibit 4.1 to annual report on Form 10-K for fiscal year ended December 31, 2020)
|
|
4.2
|
|
Form of Common Stock Warrant issued December 2018 and January 2019 (incorporated by reference to Exhibit 10.3 to current report on Form 8-K filed December 28, 2018)
|
|
4.3
|
|
Common Stock Warrant issued April 2, 2019 (incorporated by reference to Exhibit 10.3 to quarterly report on Form 10-Q for quarter ended March 31, 2019)
|
|
4.4
|
|
Form of Common Stock Warrant issued August through October 2019 (incorporated by reference to Exhibit 10.2 to current report on Form 8-K filed August 29, 2019)
|
|
4.5
|
|
Form of Warrants issued May 22, June 5, June 15, and June 22, 2020 (incorporated by reference to Exhibit 10.2 to current report on Form 8-K filed June 11, 2020)
|
|
4.6++
|
|
Form of Common Stock Purchase Warrant
|
|
4.7++
|
|
Form of Underwriter Common Stock Purchase Warrant
|
|
4.8
|
|
Warrant Agency Agreement with VStock Transfer, LLC dated September 1, 2020 (incorporated by reference to Exhibit 4.1 to current report on Form 8-K filed September 1, 2020)
|
|
4.9
|
|
Form of Common Stock Purchase Warrant (included in Exhibit 4.8)
|
|
5.1++
|
|
Opinion of Faegre Drinker Biddle & Reath LLP
|
|
10.1*
|
|
2011 Stock Option Plan, as amended through January 1, 2015 (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed September 11, 2015)
|
|
10.2*
|
|
Form of Incentive Stock Option Agreement for awards under 2011 Plan (incorporated by reference to Exhibit 10.2 to current report on Form 8-K filed September 11, 2015)
|
|
10.3*
|
|
Form of Non-Qualified Stock Option Agreement for awards under 2011 Plan (incorporated by reference to Exhibit 10.3 to current report on Form 8-K filed September 11, 2015)
|
|
10.4*
|
|
Sun BioPharma, Inc. 2016 Omnibus Incentive Plan as amended and restated through April 9, 2020 (incorporated by reference to Exhibit 99.1 to current report on Form 8-K filed May 26, 2020)
|
|
10.5*
|
|
Form of Incentive Stock Option Agreement for awards under 2016 Plan (incorporated by reference to Exhibit 10.4 to quarterly report on Form 10-Q for quarter ended June 30, 2016)
|
|
10.6*
|
|
Form of Non-Qualified Stock Option Agreement for awards under 2016 Omnibus Incentive Plan (incorporated by reference to Exhibit 10.5 to quarterly report on Form 10-Q for quarter ended June 30, 2016)
|
|
10.7*
|
|
Form of Performance-Based Stock Option Agreement for awards under 2016 Omnibus Incentive Plan (incorporated by reference to Exhibit 10.7 to annual report on Form 10-K for fiscal year ended December 31, 2016)
|
|
10.8*
|
|
Form of Indemnification Agreement with non-employee directors (incorporated by reference to Exhibit 10.4 to current report on Form 8-K filed September 11, 2015)
|
|
10.9**
|
|
Standard Exclusive License Agreement with University of Florida Research Foundation, Inc., dated December 22, 2011 (incorporated by reference to Exhibit 10.5 to current report on Form 8-K filed September 11, 2015)
|
|
10.10
|
|
Form of First Amendment to License Agreement with University of Florida Research Foundation, Inc. dated December 12, 2016 (incorporated by reference to Exhibit 10.10 to annual report on Form 10-K for fiscal year ended December 31, 2019)
|
|
10.11
|
|
Second Amendment to License Agreement with University of Florida Research Foundation, Inc., dated October 3, 2019 (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed October 9, 2019)
|
|
10.12*
|
|
Employment Agreement with Michael T. Cullen, dated December 2, 2015 (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed December 4, 2015)
|
|
10.13*
|
|
First Amendment to Employment Agreement with Michael T. Cullen, dated September 12, 2016 (incorporated by reference to Exhibit 10.17 to registration statement on Form S-1filed September 16, 2016)
|
|
10.14*
|
|
Second Amendment to Employment Agreement with Michael T. Cullen, dated October 1, 2017 (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed October 13, 2017)
|
|
10.15*
|
|
Waiver and Third Amendment to Employment Agreement with Michael T. Cullen, effective as of February 27, 2018 (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed March 5, 2018)
|
|
10.16*
|
|
Employment Agreement with Suzanne Gagnon, dated December 2, 2015 (incorporated by reference to Exhibit 10.9 to annual report on Form 10-K for fiscal year ended December 31, 2015)
|
|
10.17*
|
|
First Amendment to Employment Agreement with Suzanne Gagnon, dated September 12, 2016 (incorporated by reference to Exhibit 10.20 to registration statement on Form S-1 filed September 16, 2016)
|
|
10.18*
|
|
Second Amendment to Employment Agreement with Suzanne Gagnon, dated October 1, 2017 (incorporated by reference to Exhibit 10.4 to current report on Form 8-K filed October 13, 2017)
|
|
10.19*
|
|
Waiver and Third Amendment to Employment Agreement with Suzanne Gagnon, effective as of February 27, 2018 (incorporated by reference to Exhibit 10.4 to current report on Form 8-K filed March 5, 2018)
|
|
10.20*
|
|
Employment Agreement with Susan Horvath, dated April 17, 2018 (incorporated by reference to Exhibit 10.4 to quarterly report on Form 10-Q for quarter ended March 31, 2018)
|
|
10.21
|
|
Employment agreement with Jennifer K Simpson dated July 15, 2020 (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed July 16, 2020)
|
|
Exhibit
No.
|
|
Description |
|
10.22
|
|
Seed Capital Accelerator Loan Agreement and Seed Capital Loan Note dated October 26, 2012 (incorporated by reference to Exhibit 10.22 to annual report on Form 10-K for fiscal year ended December 31, 2019)
|
|
10.23
|
|
First Amendment to Seed Capital Accelerator Loan Agreement and Seed Capital Loan Note dated October 13, 2017 (incorporated by reference to Exhibit 10.1 to quarterly report on Form 10-Q for quarter ended March 31, 2019)
|
|
10.24
|
|
Second Amendment to Seed Capital Accelerator Loan Agreement and Seed Capital Loan Note dated April 5, 2019 (incorporated by reference to Exhibit 10.2 to quarterly report on Form 10-Q for quarter ended March 31, 2019
|
|
10.25
|
|
Third Amendment to Seed Capital Accelerator Loan Agreement and Seed Capital Loan Noted dated December 31,2019 (incorporated by reference to Exhibit 10.25 to annual report on Form 10-K for fiscal year ended December 31, 2019)
|
|
10.26
|
|
Form of Securities Purchase Agreement, dated December 21 and 31, 2018, January 14, 25, and 31, 2019 (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed December 28, 2018)
|
|
10.27
|
|
Healthcare Professional Services Agreement with Suzanne Gagnon dated July 19, 2021 (incorporated by reference to Exhibit 10.27 to annual report on Form 10-K for fiscal year ended December 31, 2021)
|
|
10.28*
|
|
Cancer Prevention Pharmaceuticals, Inc. 2010 Equity Incentive Plan, as amended (incorporated by reference to Exhibit 10.1 to current report on Form 8-K filed June 16, 2022)
|
|
10.29*
|
|
Form of Stock Option Assumption Notice (incorporated by reference to Exhibit 10.2 to current report on Form 8-K filed June 16, 2022)
|
|
10.30
|
|
Form of Replacement Warrant (incorporated by reference to Exhibit 10.3 to current report on Form 8-K filed June 16, 2022)
|
|
10.31
|
|
Convertible Promissory Note in favor of Sucampo GmbH (f/k/a Sucampo AG), dated as of September 6, 2017, as amended through April 7, 2022 (incorporated by reference to Exhibit 10.4 to current report on Form 8-K filed June 16, 2022)
|
|
10.32
|
|
Guaranty in favor of Sucampo GmbH (f/k/a Sucampo AG), dated June 15, 2022 (incorporated by reference to Exhibit 10.5 to current report on Form 8-K filed June 16, 2022)
|
|
10.33
|
|
Separation and Release Agreement with Jeffrey E. Jacobs, dated June 15, 2022 (incorporated by reference to Exhibit 10.6 to current report on Form 8-K filed June 16, 2022)
|
|
21.1+
|
|
List of Subsidiaries
|
|
23.1+
|
|
Consent of Independent Registered Public Accounting Firm
|
|
23.2+
|
|
Consent of Independent Public Accounting Firm
|
|
23.3++
|
|
Consent of Faegre Drinker Biddle & Reath LLP (included in Exhibit 5.1)
|
|
24.1++
|
|
Powers of Attorney
|
|
101++
|
|
Financial statements of the Company for the years ended December 31, 2020 and 2021, formatted in inline XBRL: (i) the Consolidated Balance Sheets, (ii) Consolidated Statements of Comprehensive Loss, (iii) the Consolidated Statements of Stockholders’ Equity, (iv) the Consolidated Statements of Cash Flows, and (v) the Notes to Consolidated Financial Statements.
|
+ Filed herewith
++ Previously filed.
* Management compensatory plan or arrangement required to be filed as an exhibit to this prospectus.
** Portions of exhibit omitted pursuant to order granting confidential treatment issued by the Securities and Exchange Commission.
(b) Financial Statement Schedules.
All schedules are omitted as the required information is inapplicable or the information is presented in the financial statements or related notes.
Schedule II. Valuation and Qualifying Accounts
All other schedules are omitted as the required information is inapplicable or the information is presented in the financial statements or related notes.
Item 17. Undertakings.
The registrant hereby undertakes:
| |
(1)
|
To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
|
| |
(i)
|
To include any prospectus required by Section 10(a)(3) of the Securities Act;
|
| |
(ii)
|
To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and
|
| |
(iii)
|
To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in this registration statement.
|
| |
(2)
|
That, for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered herein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
|
| |
(3)
|
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
|
| |
(4)
|
That, for the purpose of determining liability under the Securities Act to any purchaser: each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness; provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
|
| |
(5)
|
That, for the purpose of determining liability under the Securities Act to any purchaser the information omitted from the form of prospectus filed as part of this Registration Statement in reliance upon Rule 430A and contained in a form of prospectus filed by the Registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this Registration Statement as of the time it was declared effective; and for the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
|
| |
(6)
|
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that, in the opinion of the Securities and Exchange Commission, such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
|
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, as amended, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Minneapolis, State of Minnesota on June 22, 2022.
|
|
PANBELA THERAPEUTICS, INC.
|
|
|
|
|
|
|
|
|
By:
|
/s/ Jennifer K. Simpson
|
|
|
|
|
Jennifer K. Simpson
|
|
|
|
|
President and Chief Executive Officer
|
|
Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities and on the dates indicated.
|
Signature
|
|
Title
|
|
Date
|
| |
|
|
|
|
|
/s/ Jennifer K. Simpson
|
|
President and Chief Executive Officer
|
|
June 22, 2022
|
|
Jennifer K. Simpson
|
|
(Principal Executive Officer), and Director
|
|
|
| |
|
|
|
|
|
/s/ Susan Horvath
|
|
Vice President of Finance, Chief Financial Officer, Treasurer
|
|
June 22, 2022
|
|
Susan Horvath
|
|
and Secretary (Principal Financial and Accounting Officer)
|
|
|
| |
|
|
|
|
|
*
|
|
Chair of the Board and Director
|
|
June 22, 2022
|
|
Michael T. Cullen
|
|
|
|
|
| |
|
|
|
|
|
/s/ Daniel J. Donovan
|
|
Director
|
|
June 22, 2022
|
|
Daniel J. Donovan
|
|
|
|
|
| |
|
|
|
|
|
*
|
|
Director
|
|
June 22, 2022
|
|
Arthur J. Fratamico
|
|
|
|
|
| |
|
|
|
|
|
/s/ Jeffrey E. Jacob
|
|
Director
|
|
June 22, 2022
|
|
Jeffrey E. Jacob
|
|
|
|
|
| |
|
|
|
|
|
*
|
|
Director
|
|
June 22, 2022
|
|
Jeffrey S. Mathiesen
|
|
|
|
|
| |
|
|
|
|
|
*
|
|
Director
|
|
June 22, 2022
|
|
D. Robert Schemel
|
|
|
|
|
|
*
|
Susan Horvath, by signing her name hereto, does hereby sign this document on behalf of each of the above-named directors of the registrant pursuant to powers of attorney duly executed by such persons.
|
| |
By:
|
/s/ Susan Horvath
|
|
| |
|
Susan Horvath
|
|
| |
|
Attorney-in-Fact
|
|
Panbela Therapeutics (NASDAQ:PBLA)
Historical Stock Chart
From Mar 2024 to Apr 2024
Panbela Therapeutics (NASDAQ:PBLA)
Historical Stock Chart
From Apr 2023 to Apr 2024