MARLBOROUGH, Mass.,
July 21, 2021 /PRNewswire/ -- Today,
Boston Scientific (NYSE: BSX) announced positive 24-month results
from the PINNACLE FLX clinical trial assessing the safety and
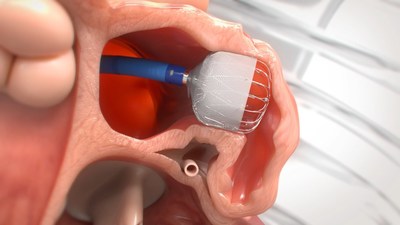
efficacy of the next-generation WATCHMAN FLX™ Left Atrial Appendage
Closure (LAAC) Device for patients with non-valvular atrial
fibrillation (NVAF). Presented as late-breaking clinical science at
TVT: The Structural Heart Summit, the study evaluated the WATCHMAN
FLX device as an alternative to long-term oral anticoagulation
therapy, including non-vitamin K antagonist oral anticoagulants
(NOACs), for stroke risk reduction in patients with NVAF.
The prospective, non-randomized PINNACLE FLX trial included 400
patients in the U.S. with NVAF who were eligible for
anti-coagulation therapy to reduce the risk of stroke but had
appropriate rationale to seek a non-pharmaceutical alternative.
Following the positive 12-month results in which the trial met
its primary safety and efficacy endpoints, the trial met its
secondary effectiveness endpoint – defined as the occurrence of
ischemic stroke or systemic embolism over 24 months – with a rate
of 3.4% compared to the performance goal of 8.7%.1
"These findings demonstrate sustained device performance over
two years and reinforce the excellent safety and efficacy profile
of the WATCHMAN FLX technology," said Saibal Kar, M.D., study co-principal
investigator and interventional cardiologist at Los Robles Regional
Medical Center and Bakersfield Heart Hospital, California. "Building upon the low
complication rates and 100% rate of effective LAA closure seen at
12 months, the 3.4% rate of ischemic stroke and systemic embolism
at 24 months is very encouraging in this complex, elderly patient
population."
In addition to the low rate of ischemic stroke, the data through
24 months also demonstrated that no patients experienced a device
embolization or pericardial effusion requiring cardiac surgery, all
of which is favorable in the context of previous clinical
studies.2
"The final results of this pivotal study underscore how design
advancements of the WATCHMAN FLX device – which allow for improved
anchoring, a faster, more effective LAA closure and compatibility
with more complex anatomies – have translated into a safe,
effective and durable option for patients with NVAF at increased
risk for stroke and systemic embolism and an appropriate rationale
to seek a non-pharmaceutical alternative," said Dr.
Ian Meredith, AM, global chief
medical officer, Boston Scientific.
The next-generation WATCHMAN FLX device received U.S. Food and
Drug Administration (FDA) approval in July
2020 and CE Mark in March
2019, and is now used in nearly all implants in the U.S. and
Europe in lieu of the
previous-generation device.
The company continues its clinical research on the WATCHMAN FLX
device for use in patients with NVAF via two large prospective,
randomized controlled trials: the OPTION trial – comparing the
WATCHMAN FLX device to oral anticoagulants in patients who also
undergo a cardiac ablation procedure; and the CHAMPION-AF clinical
trial – studying a broader anticoagulant-eligible patient
population to evaluate the device against NOACs for embolic stroke
prevention.
For more information on the WATCHMAN FLX device, visit
www.watchman.com/implanter.
About Boston Scientific
Boston Scientific transforms
lives through innovative medical solutions that improve the health
of patients around the world. As a global medical technology leader
for more than 40 years, we advance science for life by providing a
broad range of high performance solutions that address unmet
patient needs and reduce the cost of healthcare. For more
information, visit www.bostonscientific.com and connect on Twitter
and Facebook.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of Section 27A of the Securities Act of 1933 and
Section 21E of the Securities Exchange Act of 1934. Forward-looking
statements may be identified by words like "anticipate," "expect,"
"project," "believe," "plan," "estimate," "intend" and similar
words. These forward-looking statements are based on our
beliefs, assumptions and estimates using information available to
us at the time and are not intended to be guarantees of future
events or performance. These forward-looking statements include,
among other things, statements regarding our business plans,
clinical trials and product performance and impact. If our
underlying assumptions turn out to be incorrect, or if certain
risks or uncertainties materialize, actual results could vary
materially from the expectations and projections expressed or
implied by our forward-looking statements. These factors, in
some cases, have affected and in the future (together with other
factors) could affect our ability to implement our business
strategy and may cause actual results to differ materially from
those contemplated by the statements expressed in this press
release. As a result, readers are cautioned not to place undue
reliance on any of our forward-looking statements.
Factors that may cause such differences include, among other
things: future economic, competitive, reimbursement and regulatory
conditions; new product introductions; demographic trends;
intellectual property; litigation; financial market conditions; and
future business decisions made by us and our competitors. All of
these factors are difficult or impossible to predict accurately and
many of them are beyond our control. For a further list and
description of these and other important risks and uncertainties
that may affect our future operations, see Part I, Item 1A – Risk
Factors in our most recent Annual Report on Form 10-K filed with
the Securities and Exchange Commission, which we may update in Part
II, Item 1A – Risk Factors in Quarterly Reports on Form 10-Q we
have filed or will file hereafter. We disclaim any intention
or obligation to publicly update or revise any forward-looking
statements to reflect any change in our expectations or in events,
conditions or circumstances on which those expectations may be
based, or that may affect the likelihood that actual results will
differ from those contained in the forward-looking statements. This
cautionary statement is applicable to all forward-looking
statements contained in this document.
CONTACTS:
Angela Mineo
Media Relations
(763) 955-8325 (office)
Angela.mineo@bsci.com
Lauren Tengler
Investor Relations
(508) 683-4479
BSXInvestorRelations@bsci.com
1 By Kaplan-Meier estimate
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/late-breaking-trial-data-at-tvt-demonstrate-sustained-safety-and-performance-of-watchman-flx-left-atrial-appendage-closure-device-301338450.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/late-breaking-trial-data-at-tvt-demonstrate-sustained-safety-and-performance-of-watchman-flx-left-atrial-appendage-closure-device-301338450.html
SOURCE Boston Scientific Corporation