Stratasys PolyJet technology and Materialise
FDA-cleared software now most versatile 3D printing system for
hospitals and physicians to build anatomical models at the
point-of-care
Further bringing 3D printed medical models to life, Stratasys
(Nasdaq: SSYS) is expanding the suite of printers and materials
validated by its collaborator Materialise (Nasdaq: MTLS) as part of
FDA-cleared Materialise Mimics inPrint software. The end result is
the most versatile 3D printing system for point-of-care across
hospitals and physicians - advancing production of
patient-specific, life-like anatomical models for diagnostic
purposes in conjunction with other tools and expert clinical
judgement.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20181126005388/en/
Model of patient's left atrial appendage
(LAA) created with Stratasys and Materialise technology is intended
to allow surgeons to select the appropriate device and plan the
optimal approach to occlude the LAA. Image provided by
Materialise.
PolyJet multi-material and multi-color solutions validated now
include the J750 and J735 3D Printers and the high-performance
desktop Objet30 Prime 3D Printer. Materialise Mimics inPrint is the
first and currently only 3D printing software cleared by the FDA to
create anatomical models for patient care. The software is designed
to allow physicians and hospitals to leverage 3D printing at
point-of-care - building a trusted and reliable source for surgical
planning and interdisciplinary communication.
“Historically, pre-surgical planning relied on 2D imaging
requiring physicians to mentally reconstruct the patient anatomy.
But 3D printing evolves this approach by putting precise replicas
of patient anatomy directly in physician hands. Our collaboration
with Materialise is a huge step towards unlocking the potential of
this technology for patient care,” says Eyal Miller, Head of
Healthcare Business Unit, Stratasys. “Now the 3D printer that every
hospital needs to power their medical modeling comes with
additional options for an FDA-cleared software solution.”
In March of 2018, Materialise Mimics inPrint was cleared by the
FDA, becoming the only solution with 510(k) clearance as an
end-to-end 3D printing solution, as well as a fully comprehensive
3D printing solution for anatomical modeling. According to company
reports, of the top 20 U.S. hospitals as ranked by U.S. News &
World Report, 16 have implemented a medical 3D printing strategy
using Materialise Mimics technology.
“By validating Stratasys’ 3D printing technologies through our
certification process, we’re giving doctors and hospitals improved
access to high-quality anatomical models for personalized care to
patients,” said Bryan Crutchfield, Vice President and General
Manager of Materialise North America. “The addition of multi-color
and multi-material printers to the list of validated printers is
aimed to enable healthcare providers to implement a versatile
offering that can support their most complex cases across a wide
range of surgical specialties on a single printer. At Materialise,
we take a hardware-agnostic approach to software development,
offering the flexibility to partner with other leaders in the 3D
printing industry like Stratasys – a company committed to
addressing requirements of the medical community.”
The Stratasys J735/J750 3D Printers are able to develop
highly-complex models using multiple textures, while combining hard
and soft materials to mimic human tissue. The unique combination of
transparency with multiple color re-creation ensures practitioners
can differentiate anatomy, view critical structures within an organ
replica, and create realistic representations of any bone, tissue,
and organ.
The Stratasys’ Objet30 Prime 3D Printer is a cost-effective,
desktop platform providing an entry point to hospitals seeking a
point-of-care printing solution - without compromising quality,
resolution or accuracy. The versatile offering supports a range of
anatomical models and applications, including orthopedic, cardiac,
neurosurgery and other use-cases for visualization, surgical
planning, training and education.
For more information on how you can integrate Stratasys’ 3D
printing solutions and Materialise software to revolutionize
medical modeling practices and improve patient outcomes, visit
https://www.stratasys.com/medical
Visit Stratasys at RSNA 2018
Stratasys J750 and Objet30 Prime 3D Printers will be at RSNA
2018 at McCormick Place in Chicago, IL from November 25 to November
30. The company is offering exclusive hands-on demos, detailed
customer use-cases and presentations throughout the show at Booth #
1968L, South Hall.
Materialise incorporates 27 years of 3D printing
experience into a range of software solutions and 3D printing
services, which together form the backbone of the 3D printing
industry. Materialise’s open and flexible solutions enable players
in a wide variety of industries, including healthcare, automotive,
aerospace, art and design, and consumer goods, to build innovative
3D printing applications that aim to make the world a better and
healthier place. Headquartered in Belgium, with branches worldwide,
Materialise combines the largest group of software developers in
the industry with one of the largest 3D printing facilities in the
world. Further information at: www.materialise.com.
Stratasys is a global leader in additive manufacturing or
3D printing technology, and is the manufacturer of FDM® and
PolyJet™ 3D Printers. The company’s technologies are used to create
prototypes, manufacturing tools, and production parts for
industries, including aerospace, automotive, healthcare, consumer
products and education. For 30 years, Stratasys products have
helped manufacturers reduce product-development time, cost, and
time-to-market, as well as reduce or eliminate tooling costs and
improve product quality. The Stratasys 3D printing ecosystem of
solutions and expertise includes: 3D printers, materials, software,
expert services, and on-demand parts production.
Corporate Headquarters: Minneapolis, Minnesota and Rehovot,
Israel. Online at: http://www.stratasys.com/,
http://blog.stratasys.com/ and LinkedIn.
Stratasys is a registered trademark and J750, Objet30 Prime, and
Stratasys signet are trademarks of Stratasys Ltd. and/or its
subsidiaries or affiliates. All other trademarks are the property
of their respective owners.
Note Regarding Forward-Looking Statements
The statements in this press release relating to Stratasys’
beliefs regarding the benefits consumers will experience from the
Stratasys J750, Objet30 Prime 3D Printers or their validation with
Materialise, Stratasys’ expectation on the timing of shipping the
Stratasys J750, Objet30 Prime 3D Printers or their validation with
Materialise, are forward-looking statements reflecting management's
current expectations and beliefs. These forward-looking statements
are based on current information that is, by its nature, subject to
rapid and even abrupt change. Due to risks and uncertainties
associated with Stratasys' business, actual results could differ
materially from those projected or implied by these forward-looking
statements. These risks and uncertainties include, but are not
limited to: the risk that consumers will not perceive the benefits
of the Stratasys J750, Objet30 Prime 3D Printers or their
validation with Materialise to be the same as Stratasys does; the
risk that unforeseen technical difficulties will delay the shipping
of the Stratasys J750, Objet30 Prime 3D Printers or their
validation with Materialise; and other risk factors set forth under
the caption “Risk Factors” in Stratasys' most recent Annual Report
on Form 20-F, filed with the Securities and Exchange Commission
(SEC) February 28, 2018. Stratasys is under no obligation (and
expressly disclaims any obligation) to update or alter its
forward-looking statements, whether as a result of new information,
future events or otherwise, except as otherwise required by the
rules and regulations of the SEC.
Attention Editors, if you publish reader-contact information,
please use:
- USA +800-801-6491
- Europe/Middle East/Africa
+49-7229-7772-0
- Asia Pacific +852 3944-8888
View source
version on businesswire.com: https://www.businesswire.com/news/home/20181126005388/en/
Stratasys Media ContactsStratasys Corporate &
North AmericaCraig.Librett@stratasys.com+1 612-364-3208
Europe, Middle East, and AfricaJonathan Wake / Miguel
Afonso, Incus Mediastratasys@incus-media.com+44 1737 215200
Greater China, Southeast Asia, ANZ, and IndiaAlice
Chiumedia.ap@stratasys.com+86-21-33196051
Japan and KoreaAya.Yoshizawa@stratasys.com+81 3 5542
0042
Mexico, Central America, Caribe and South
AmericaErica.massini@stratasys.com+55 11 2626-9229
BrazilCaio.Ramos@GPcom.com.brNando@GPcom.com.brGP
Communications+55 (11) 3129 5158
Materialise Media Contact:Virginia GobleVice President of
Marketing
-NAMaterialise248-921-5500virginia.goble@materialise.com
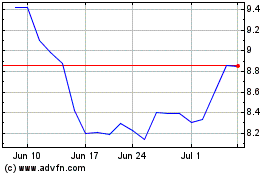
Stratasys (NASDAQ:SSYS)
Historical Stock Chart
From Mar 2024 to Apr 2024
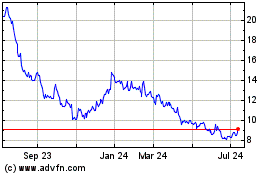
Stratasys (NASDAQ:SSYS)
Historical Stock Chart
From Apr 2023 to Apr 2024