Intertek First Independent Lab in North America Accredited for End-to-End Evaluation of Implantable & Non-Implantable Medical...
August 16 2018 - 10:15AM
Business Wire
Intertek, a leading Total Quality Assurance provider to
industries worldwide, is pleased to announce that it has been
accredited by the American Association for Laboratory Accreditation
(A2LA) at its Boxborough, Massachusetts laboratory to test active
implantable medical devices (AIMDs) to ISO 14708-1 and EN 45502-1
standards. With this, Intertek becomes the first independent
laboratory in North America accredited for electrical safety and
electromagnetic compatibility (EMC) testing for both implantable
and non-implantable medical devices.
Active implantable medical devices rely on a source of power
other than what is provided by the body or gravity, and are
intended to be surgically/medically inserted into the body and
remain there after the procedure. As one of the highest risk
categories of medical devices they are subject to rigorous
regulatory standards by the U.S. Food and Drug Administration (FDA)
and other authorities around the world. To reduce risk and ensure
quality, medical device manufacturers choose to work with a
third-party laboratory to illustrate compliance with these
electrical safety, EMC and wireless standards.
Most active implantable devices consist of both the product
inserted into the body and non-implantable supporting equipment,
both of which require testing and evaluation to medical regulatory
standards, including IEC 60601-1-2 3rd Edition for non-implantable
devices and components. With the new accreditations, Intertek now
offers fully-accredited electrical safety, EMC and wireless device
testing and evaluation services for both implantable and
non-implantable products, with one all-encompassing report to be
submitted to the FDA or other notified bodies. This in turn can
help reduce costs and allow customers to get products to market
faster.
Sunny Rai, Senior Vice President at Intertek, commented: “At
Intertek, our focus has always been to provide innovative
Assurance, Testing, Inspection and Certification services to our
customers to help them bring their products to market quickly,
efficiently and safely. We are thrilled to offer the medical device
industry the evaluation they need for active implantable devices in
one location with a team of highly proficient experts who are
familiar with the necessary requirements for this growing medical
field.”
For more than 50 years Intertek has been partnering with medical
device manufacturers to deliver Total Quality Assurance solutions
for Assurance, Testing, Inspection and Certification. The Company's
experts understand the latest technologies and the regulatory
requirements that products must meet, including those for mobile
health applications and software. With engineers and technicians at
10 Centers of Excellence and 23 labs across the world dedicated to
testing medical devices equipped with state-of-the-art equipment,
Intertek helps customers launch new medical devices to market
quickly. For more information, visit www.intertek.com/medical.
ABOUT INTERTEK
Total Quality. Assured.
Intertek is a leading Total Quality Assurance provider to
industries worldwide. Our network of more than 1,000 laboratories
and offices and over 43,000 people in more than 100 countries
delivers innovative and bespoke Assurance, Testing, Inspection and
Certification solutions for our customers’ operations and supply
chains. Intertek Total Quality Assurance expertise, delivered
consistently, with precision, pace and passion, enabling our
customers to power ahead safely.
intertek.com
View source
version on businesswire.com: https://www.businesswire.com/news/home/20180816005018/en/
FOR MEDIA INFORMATION:Please contactCheryl
PrejsnarUS PR Manager+1
630-408-5538cheryl.prejsnar@intertek.com
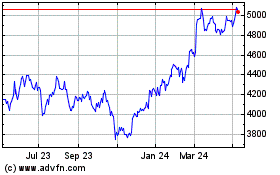
Intertek (LSE:ITRK)
Historical Stock Chart
From Mar 2024 to Apr 2024
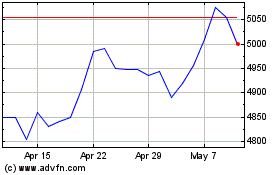
Intertek (LSE:ITRK)
Historical Stock Chart
From Apr 2023 to Apr 2024