STAAR Surgical Announces Strategic Alliance Agreement with Asian Eye Institute
September 04 2019 - 7:00AM
Business Wire
World Class Eye Institute Positions ICL as
Primary and Premium Vision Correction Procedure
STAAR Surgical Company (NASDAQ: STAA), a leading developer,
manufacturer and marketer of implantable lenses and companion
delivery systems for the eye, today announced that it has entered a
strategic alliance with Asian Eye Institute, an eye care group with
nine locations in the Philippines.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20190904005347/en/
Robert T. Ang, M.D., Head of Cornea and
Refractive Surgery Services at the Asian Eye Institute. (Photo:
Business Wire)
“‘Amazing’, ‘Wow’, ‘It’s so clear’, and ‘I am so happy’ are the
words I often hear after performing the Visian® EVO ICL procedure
and I am confident and proud to partner with STAAR in offering the
EVO family of lenses to my patients,” said Dr. Robert “Bobby”
Edward Ang, M.D., senior consultant and Head of Cornea and
Refractive Surgery Services at Asian Eye Institute. “Patient
reactions to their new EVO lenses always give me a big smile. It’s
truly remarkable that EVO can deliver such outstanding results very
consistently. Even if a patient has low to moderate refractive
error and is qualified for Lasik, PRK or SMILE, I can present the
patient an option for outstanding quality vision with the ICL and
without removing tissue from the cornea and risking ectasia. Thin
corneas, suspicious topographies, and high refractive errors are
also no longer a hindrance to giving a patient great vision with
the implantable Collamer lens. Not many products can make a doctor
feel this way.”
Asian Eye Institute was founded in 2001 and has 18 medical
doctors, two specialist optometrists, and more than 30 staff
optometrists on staff across its nine locations.
“Dr. Bobby Ang and the Asian Eye Institute are renowned for
their high level of patient care and extensive research within the
field of Ophthalmology,” said Caren Mason, President and CEO of
STAAR Surgical. “Asian countries, such as the Philippines, are
among the hardest hit by the explosive growth in myopia and, in
particular, progressive high myopia that can lead to more serious
eye disease such as glaucoma and retinal detachment. The ICL is an
excellent option for these, and many other, patients with a wide
range of vision correction needs who also seek independence from
disposable contact lenses and glasses. Dr. Ang was recognized by
The Ophthalmologist on the 2018 Power List 100, which features top
innovators in the field of Ophthalmology. He has also conducted
important research relating to the ICL, having presented his work
at STAAR’s annual Experts Meetings during the European Society of
Cataract and Refractive Surgery (ESCRS) Congress. The Asian Eye
Institute and Dr. Ang have demonstrated their commitment to vision
care excellence and delighting patients for almost two decades. We
are thrilled to expand our partnership through a new Strategic
Alliance Agreement.”
Myopia (nearsightedness) is the need for distance vision
correction. EVO, also known as the Implantable Collamer Lens, is an
additive lens implant that can help correct myopia. Unlike other
procedures, the EVO procedure does not remove corneal tissue but
works in harmony with the natural eye. EVO rests behind the iris
and is biocompatible. 99.4% of patients in a Patient Registry said
they would have the ICL procedure again.1
1Patient Registry data on file.
About STAAR Surgical
STAAR, which has been dedicated solely to ophthalmic surgery for
over 30 years, designs, develops, manufactures and markets
implantable lenses for the eye with companion delivery systems.
These lenses are intended to provide visual freedom for patients,
lessening or eliminating the reliance on glasses or contact lenses.
All of these lenses are foldable, which permits the surgeon to
insert them through a small incision. STAAR’s lens used in
refractive surgery is called an Implantable Collamer® Lens or
“ICL”, which includes the EVO Visian ICL™ product line. More than
1,000,000 Visian® ICLs have been implanted to date and STAAR
markets these lenses in over 75 countries. To learn more about the
ICL go to: www.discovericl.com. Headquartered in Lake Forest, CA,
the company operates manufacturing and packaging facilities in
Aliso Viejo, CA, Monrovia, CA and Nidau, Switzerland. For more
information, please visit the Company’s website at
www.staar.com.
About Asian Eye
Institute
Asian Eye Institute is a world-class eye facility that puts the
Philippines on the map of quality eye care. Established in 2001,
the Institute is at the forefront of efforts to make breakthrough
technologies for eye care treatment available to all patients. It
serves 60,000 local and foreign patients annually. Asian Eye’s
commitment to quality care and excellent service has been
recognized by prestigious international organizations. It is ISO
9001:2008, 14001:2015 and OHSAS 18001:2007 triple- certified, and
has received Platinum-level international healthcare accreditation
by Accreditation Canada International. It is also a 6-time winner
of the Reader’s Digest Trusted Brand Award in the Eye Center
category. Dr. Robert Edward Ang is a senior consultant and Head of
Cornea and Refractive Surgery Services at the Asian Eye Institute.
Dr. Ang’s drive for ophthalmic technology advancement and
innovation has been recognized and his team strives to continue
helping people to improve their vision, so they can enjoy the
beauty of the world and lead productive lives. For more
information, please visit the institute’s website at
www.asianeyeinstitute.com
ATTENTION:
For Physicians: Please reference the Visian ICL Product
Information for a complete listing of indications, warnings, and
precautions.
For Patients: Before considering Visian ICL surgery you should
talk with your eye care professional about Visian ICL surgery,
especially the potential benefits, risks, and complications.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190904005347/en/
Investors & Media Brian Moore Sr. Director, Investor,
Media Relations and Corporate Development (626) 303-7902, Ext. 3023
bmoore@staar.com
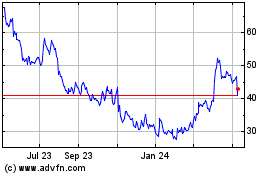
STAAR Surgical (NASDAQ:STAA)
Historical Stock Chart
From Mar 2024 to Apr 2024
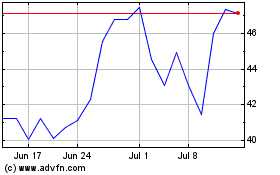
STAAR Surgical (NASDAQ:STAA)
Historical Stock Chart
From Apr 2023 to Apr 2024