Philips expands peripheral vascular disease portfolio with the QuickClear mechanical thrombectomy system for blood clot remov...
September 24 2020 - 9:00AM

Philips expands peripheral vascular disease portfolio with the
QuickClear mechanical thrombectomy system for blood clot removal
September 24, 2020
- Powerfully simple thrombectomy solution provides physicians
with an all-in-one, single-use aspiration catheter and pump
system
- Intuitive design eliminates the need for capital equipment,
with easy setup supporting faster procedures times
- Significantly smaller footprint provides the same or greater
aspiration power as currently-available thrombectomy devices
[1]
Amsterdam, the Netherlands – Royal Philips
(NYSE: PHG, AEX: PHIA), a global leader in health technology, today
announced the launch of the innovative QuickClear mechanical
thrombectomy system. The compact, single-use system provides an
all-in-one aspiration pump and catheter for the removal of blood
clots from the vessels of the peripheral arterial and venous
systems [2]. The system’s innovative all-in-one design is simple to
use while eliminating the need for capital equipment or costly
accessories, with easy setup supporting faster procedures times.
The Philips QuickClear Mechanical Thrombectomy System has received
U.S. FDA 510(k) clearance and is available for sale in the U.S.
“This novel thrombectomy system is the latest addition to
Philips’ market-leading portfolio for the diagnosis and treatment
of peripheral vascular diseases,” said Chris Landon, Senior Vice
President and General Manager Image Guided Therapy Devices at
Philips. “Its intuitive design simplifies the entire thrombectomy
procedure workflow. By taking away the high initial capital
expenditure costs associated with traditional mechanical
thrombectomy systems, QuickClear can help bring cost-effective
solutions to both the hospital and outpatient care settings.”
Simple, optimized performanceQuickClear
simplifies the entire thrombectomy procedure workflow. The small
footprint of the sterile device allows it to be placed easily and
conveniently on the table next to the patient. With the touch of a
button, the system is up and running at maximum aspiration power
within seconds. The consistency of the aspiration power during the
procedure provides physicians more control and supports faster
procedure times. The system’s range of catheters includes a large
10F aspiration catheter, providing 59% more aspiration volume than
8F aspiration catheters [1].
“QuickClear is a simple and easy to use mechanical thrombectomy
system,” said Bryan Fisher, MD, Chief of Vascular Surgery, Tristar
Centennial Medical Center, Nashville, U.S. “The system is
significantly smaller than other systems without compromising
aspiration power. The convenience of the device really shines
through with its single use and lack of capital equipment. I am
excited about the potential of this device and the impact it will
have on my practice and the patients I treat.”
Philips’ peripheral vascular portfolio already includes advanced
interventional imaging systems for precision guidance;
intravascular ultrasound (IVUS) catheters to assess the location of
the disease and lesion morphology and guide and confirm the
treatment; peripheral atherectomy devices to remove blockages; and
peripheral therapy devices, such as Philips’ Stellarex drug-coated
balloon, to treat lesions. Philips recently further extended this
portfolio with the acquisition of Intact Vascular, maker of the
Tack Endovascular System, a first-of-its-kind, minimal-metal,
dissection repair device that provides precision treatment of
peripheral arterial dissections following balloon angioplasty in
above-the-knee (ATK) and below-the-knee (BTK) therapeutic
interventions.
New Cardiovascular Horizons digital
symposiumToday, September 24th at 19:00 ET Philips will
host a symposium entitled ‘QuickClear Mechanical Thrombectomy
System: A Powerfully Simple and Cost-Effective Solution’ as part of
the New Cardiovascular Horizons (NCVH) digital education series. To
register, visit here.
The Philips QuickClear Mechanical Thrombectomy System is U.S.
FDA 510(k) cleared and available for sale in the U.S., with future
expansion of availability to other geographies planned.
[1] Testing performed in-house. Data on file.[2] The Philips
QuickClear mechanical thrombectomy system is indicated for the
removal of soft, fresh emboli and thrombi from the vessels of the
peripheral arterial and venous systems.
For further information, please contact:
Mark GrovesPhilips Global Press OfficeTel: +31 631 639 916Email:
mark.groves@philips.comTwitter: mark_groves
Fabienne van der FeerPhilips Image Guided TherapyTel: +31 622
698 001Email: fabienne.van.der.feer@philips.comTwitter: FC_Feer
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health
technology company focused on improving people's health and
enabling better outcomes across the health continuum from healthy
living and prevention, to diagnosis, treatment and home care.
Philips leverages advanced technology and deep clinical and
consumer insights to deliver integrated solutions. Headquartered in
the Netherlands, the company is a leader in diagnostic imaging,
image-guided therapy, patient monitoring and health informatics, as
well as in consumer health and home care. Philips generated 2019
sales of EUR 19.5 billion and employs approximately 81,000
employees with sales and services in more than 100 countries. News
about Philips can be found at www.philips.com/newscenter.
- Philips QuickClear mechanical thrombectomy system in hand
- Philips QuickClear mechanical thrombectomy system in use
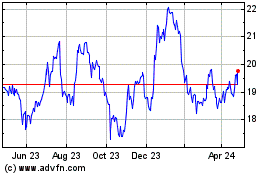
Koninklijke Philips NV (EU:PHIA)
Historical Stock Chart
From Aug 2024 to Sep 2024
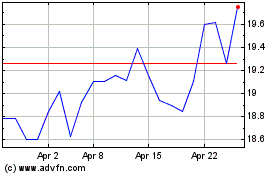
Koninklijke Philips NV (EU:PHIA)
Historical Stock Chart
From Sep 2023 to Sep 2024