Genprex to Receive Inaugural “License of the Year” Award from University of Pittsburgh Innovation Institute in Recognitio...
April 20 2021 - 8:30AM
Business Wire
Groundbreaking, potentially curative gene
therapy shown to transform alpha cells in the pancreas into
functional beta-like cells is potentially applicable to both Type 1
and Type 2 diabetes
Genprex, Inc. (“Genprex” or the “Company”) (NASDAQ: GNPX), a
clinical-stage gene therapy company focused on developing
life-changing therapies for patients with cancer and diabetes,
announces today that the Company has been selected to receive the
inaugural “License of the Year” award from the University of
Pittsburgh Innovation Institute (UPII) in recognition of the
advances made with its license from University of Pittsburgh toward
progressing the development of its gene therapy for diabetes.
In February 2020, Genprex signed an exclusive license agreement
with the University of Pittsburgh for an innovative gene therapy
technology developed by lead researcher, George Gittes, M.D. at the
Rangos Research Center at the University of Pittsbugh Medical
Center ("UPMC") Children’s Hospital. The diabetes gene therapy
candidate, GPX-002, which is being evaluated in preclinical
studies, reprograms alpha cells in the pancreas into functional
beta-like cells, thereby replenishing levels of insulin and
providing the potential to cure the disease. The novel infusion is
by means of an endoscopic procedure that delivers Pdx1 and MafA
genes to the pancreas. The genes express proteins that transform
alpha cells in the pancreas into functional beta-like cells, which
can produce insulin but are distinct enough from beta cells to
evade the body’s immune system.
“We thank the UPII for acknowledging the breakthrough work of
Dr. George Gittes and his team at the Rangos Research Center at
UPMC Children’s Hospital of Pittsburgh, whose efforts have produced
this highly innovative therapeutic approach to treating diabetes
that could replace the need for insulin replacement therapy and
ultimately lead to a cure of this devastating illness afflicting
tens of millions of people around the world,” said Rodney Varner,
President and Chief Executive Officer of Genprex. “We are honored
to receive this recognition, considering the great number of
cutting-edge technologies coming out of this distinguished
university and research center. We thank UPII for bestowing this
honor on our gene therapy license with Dr. Gittes and the Rangos
Research Center and applaud them for the groundbreaking gene
therapy research being conducted at this esteemed institution.”
The “Celebration of Innovation” awards ceremony will be held
virtually on April 22, 2021 on the Pitt Innovation Institute
YouTube channel at 4:30 p.m ET.
GPX-002 has been tested in vivo in mice and nonhuman primates.
In studies in non-obese diabetic mice, a model of Type 1 autoimmune
diabetes, the gene therapy approach restored normal blood glucose
levels for an extended period of time, typically around four
months. According to the researchers, the duration of restored
blood glucose levels in mice could potentially translate to decades
in humans. If successful, this gene therapy could eliminate the
need for insulin replacement therapy for diabetic patients.
According to the U.S. Center for Disease Control, 34.2 million
Americans, or approximately 10.5% of the population, have
diabetes.
About Genprex, Inc.
Genprex, Inc. is a clinical-stage gene therapy company focused
on developing life-changing therapies for patients with cancer and
diabetes. Genprex’s technologies are designed to administer
disease-fighting genes to provide new therapies for large patient
populations with cancer and diabetes who currently have limited
treatment options. Genprex works with world-class institutions and
collaborators to develop drug candidates to further its pipeline of
gene therapies in order to provide novel treatment approaches. The
Company’s lead product candidate, REQORSA™ (quaratusugene
ozeplasmid), is being evaluated as a treatment for non-small cell
lung cancer (NSCLC). REQORSA has a multimodal mechanism of action
that has been shown to interrupt cell signaling pathways that cause
replication and proliferation of cancer cells; re-establish
pathways for apoptosis, or programmed cell death, in cancer cells;
and modulate the immune response against cancer cells. REQORSA has
also been shown to block mechanisms that create drug resistance. In
January 2020, the U.S. Food and Drug Administration granted Fast
Track Designation for REQORSA for NSCLC in combination therapy with
AstraZeneca’s Tagrisso® (osimertinib) for patients with EFGR
mutations whose tumors progressed after treatment with
Tagrisso.
For more information, please visit the Company’s web site at
www.genprex.com or follow Genprex on Twitter, Facebook and
LinkedIn.
Cautionary Language Concerning Forward-Looking
Statements
Statements contained in this press release regarding matters
that are not historical facts are "forward-looking statements"
within the meaning of the Private Securities Litigation Reform Act
of 1995. These forward-looking statements are made on the basis of
the current beliefs, expectations and assumptions of management,
are not guarantees of performance and are subject to significant
risks and uncertainty. These forward-looking statements should,
therefore, be considered in light of various important factors,
including those set forth in Genprex’s reports that it files from
time to time with the Securities and Exchange Commission and which
you should review, including those statements under “Item 1A – Risk
Factors” in Genprex’s Annual Report on Form 10-K.
Because forward-looking statements are subject to risks and
uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Such
statements include, but are not limited to, statements regarding:
the timing and success of Genprex’s clinical trials and regulatory
approvals; the effect of Genprex’s product candidates, alone and in
combination with other therapies, on cancer and diabetes; Genprex’s
future growth and financial status; Genprex’s commercial and
strategic partnerships including the scale up of the manufacture of
its product candidates; and Genprex’s intellectual property and
licenses.
These forward-looking statements should not be relied upon as
predictions of future events and Genprex cannot assure you that the
events or circumstances discussed or reflected in these statements
will be achieved or will occur. If such forward-looking statements
prove to be inaccurate, the inaccuracy may be material. You should
not regard these statements as a representation or warranty by
Genprex or any other person that Genprex will achieve its
objectives and plans in any specified timeframe, or at all. You are
cautioned not to place undue reliance on these forward-looking
statements, which speak only as of the date of this press release.
Genprex disclaims any obligation to publicly update or release any
revisions to these forward-looking statements, whether as a result
of new information, future events or otherwise, after the date of
this press release or to reflect the occurrence of unanticipated
events, except as required by law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210420005110/en/
Genprex, Inc. (877) 774-GNPX (4679) Investor
Relations GNPX Investor Relations (877) 774-GNPX (4679) ext. #2
investors@genprex.com Media Contact Genprex Media Relations
Kalyn Dabbs (877) 774-GNPX (4679) ext. #3 media@genprex.com
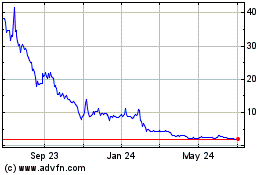
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Mar 2024 to Apr 2024
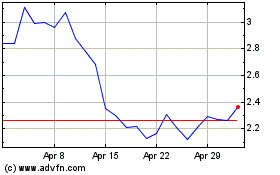
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Apr 2023 to Apr 2024