Filed Pursuant to Rule 424(b)(7)
Registration No. 333-217636
CALCULATION OF REGISTRATION FEE (1)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Title of each class of securities
to be registered
|
|
Amount to be
Registered
|
|
|
Proposed
Maximum
Offering Price
Per Share
|
|
|
Proposed
Maximum
Aggregate
Offering Price
|
|
|
Amount of
Registration Fee (2)
|
|
|
Common stock, par value $0.001 per share
|
|
|
1,448,714
|
|
|
$
|
100.69
|
|
|
$
|
145,871,012.66
|
|
|
$
|
18,160.95
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
|
The information in this Calculation of Registration Fee Table updates, with respect to the securities offered hereby, the information set forth in the Calculation of Registration Fee Table included in the
Registrant’s Registration Statement on
Form S-3
(Registration
No. 333-217636),
originally filed with the Commission on May 3, 2017
|
|
(2)
|
Estimated solely for purposes of calculating the registration fee in accordance with Rule 457(c) under the Securities Act of 1933, as amended, based on the average of the high and low reported sales prices on the New
York Stock Exchange on March 8, 2018.
|
PROSPECTUS SUPPLEMENT
(To Prospectus dated May 3, 2017)

Centene Corporation
Up to 1,448,714 Shares of Common Stock
This prospectus
supplement will be used from time to time by the selling stockholder to resell certain shares of common stock, par value $0.001 per share, of Centene Corporation, a Delaware corporation (“Centene”). The shares of our common stock that may
be offered by the selling stockholder using this prospectus supplement and the accompanying prospectus represent shares of our common stock that we issued to such selling stockholder in connection with our acquisition of Community Medical Holdings
Corp., a Delaware corporation (“CMG”), on March 12, 2018. We will not receive any proceeds from the shares of common stock sold by the selling stockholder.
Our common stock is listed on the New York Stock Exchange (the “NYSE”) under the symbol “CNC.” On March 12, 2018, the
last reported sale price of our common stock on the NYSE was $100.30 per share.
Investing in our common stock involves risks that are
described in the “
Risk Factors
” section of this prospectus supplement beginning on page
S-3
and such risk factors as may be updated from time to time in our public filings.
The selling stockholder may, from time to time, offer and sell registered shares of our common stock held by it directly or through
agents or broker-dealers on terms to be determined at the time of sale. See “Plan of Distribution” beginning on page
S-13
of this prospectus supplement for more information about how the selling
stockholder may offer and sell its shares of common stock.
The selling stockholder and any agents or broker-dealers that participate with
the selling stockholder in the distribution of registered shares may be deemed to be “underwriters” within the meaning of the Securities Act of 1933, as amended (the “Securities Act”), and any commissions received by them and any
profit on the sale of registered shares may be deemed to be underwriting commissions or discounts under the Securities Act.
Neither
the Securities and Exchange Commission (the “SEC”) nor any other regulatory body has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus supplement or the related prospectus. Any
representation to the contrary is a criminal offense.
The date of
this prospectus supplement is March 13, 2018
TABLE OF CONTENTS
Prospectus Supplement
Prospectus
You should read this document together with additional information described under the heading “Where
You Can Find More Information.” You should rely only on the information contained or incorporated by reference in this prospectus supplement and the accompanying prospectus or any free writing prospectus prepared by or on behalf of us. We have
not, and the selling stockholder has not, authorized anyone to provide you with additional or different information. We and the selling stockholder are not making an offer to sell these securities in any jurisdiction where the offer or sale is not
permitted. You should not assume that the information we have included in this prospectus supplement or the accompanying prospectus is accurate as of any date other than the date of this prospectus supplement or the accompanying prospectus or that
any information we have incorporated by reference is accurate as of any date other than the date of the document incorporated by reference. To the extent that any statement we make in this prospectus supplement is inconsistent with the statements
made in the accompanying prospectus, the statements made in the accompanying prospectus are deemed modified or superseded by the statements made in this prospectus supplement. Neither this prospectus supplement nor the accompanying prospectus
constitutes an offer, or an invitation on our behalf or on behalf of the selling stockholder, to subscribe for and purchase any of the securities and may not be used for or in connection with an offer or solicitation by anyone, in any jurisdiction
in which such an offer or solicitation is not authorized or to any person to whom it is unlawful to make such an offer or solicitation.
S-i
ABOUT THIS PROSPECTUS SUPPLEMENT
This prospectus supplement and the accompanying prospectus are part of a registration statement that we filed with the SEC utilizing a
“shelf” registration process. Under this shelf registration process, we or selling stockholders may sell the securities described in the accompanying prospectus from time to time. In this prospectus supplement, we provide you with specific
information about the shares the selling stockholder is selling in this offering and about the offering itself. Both this prospectus supplement and the accompanying prospectus include or incorporate by reference important information about us and
other information you should know before investing in the shares. This prospectus supplement also adds, updates and changes information contained or incorporated by reference in the accompanying prospectus. You should read both this prospectus
supplement and the accompanying prospectus, as well as the additional information in the documents described below under the heading “Where You Can Find More Information,” before investing in the shares.
Unless the context otherwise requires, the terms the “Company,” “we,” “us,” “our” or similar terms and
Centene” refer to Centene Corporation, together with its consolidated subsidiaries.
WHERE YOU CAN FIND
MORE INFORMATION
This prospectus supplement and the accompanying prospectus incorporate by reference information from documents filed
with the SEC, which means that we are disclosing important information to you by referring you to those documents. The information we incorporate by reference is an important part of this prospectus supplement and the accompanying prospectus, and
information we subsequently file with the SEC will automatically update and supersede that information. We incorporate by reference the documents listed below and any filings we make with the SEC under Section 13(a), 13(c), 14 or 15(d) of the
Exchange Act (other than any portion provided pursuant to Item 2.02 or Item 7.01 of Form
8-K
or other information “furnished” to the SEC), on or after the date of this prospectus supplement and
before the termination of the offering of the shares pursuant to this prospectus supplement. The documents we incorporate by reference are:
|
|
•
|
|
our Annual Report on Form
10-K
for the year ended December 31, 2017, filed with the SEC on February 20, 2018;
|
|
|
•
|
|
our Current Reports on Form
8-K,
filed with the SEC on March 24, 2016 (as amended on May 10, 2016 (excluding Item 9.01 and Exhibit 99.1 of such amendment) and as further
amended on June 9, 2016), February 5, 2018, February 9, 2018, February 27, 2018, March 13, 2018 and March 13, 2018;
|
|
|
•
|
|
our Definitive Proxy Statement on Schedule 14A, filed with the SEC on March 9, 2018 (solely to the extent incorporated by reference into Part III of our Annual Report on Form
10-K
for the year ended December 31, 2017); and
|
|
|
•
|
|
the description of our common stock contained in our registration statement on Form
8-A
filed with the SEC on October 14, 2003, as amended by our Forms
8-A/A
filed with the SEC on December 17, 2004 and April 26, 2007, including any amendments or reports filed for the purpose of updating such description.
|
The preceding list supersedes and replaces the documents listed in the accompanying prospectus under the heading “Where You Can Find More
Information.” We file annual, quarterly and current reports, proxy statements and other information with the SEC. You may read and copy any document we file at the SEC’s Public Reference Room, 100 F Street, N.E., Washington, D.C. 20549.
Please call the SEC at
1-800-SEC-0330
for further information on their public reference room. Our SEC filings are also available
to the public at the SEC’s website at http://www.sec.gov. Our common stock is listed under the symbol “CNC” and traded on the NYSE. Information about us, including our SEC filings, is also available at our Internet site at
http://www.centene.com. However, the information on our internet site is not a part of this prospectus supplement or the accompanying prospectus.
S-ii
We encourage you to read our SEC reports, as they provide additional important information about
us. For example, we filed the audited financial statements of Health Net, Inc. (“Health Net”) and its subsidiaries as of December 31, 2015 and 2014, and for each of the three years in the three-year period ended December 31,
2015, which are incorporated by reference herein from our Current Report on Form
8-K,
with the SEC on March 24, 2016 (as amended on May 10, 2016 (excluding Item 9.01 and Exhibit 99.1 of such
amendment) and as further amended on June 9, 2016) (the “Health Net Form
8-K”),
and we filed the audited financial statements of New York State Catholic Health Plan, Inc., d/b/a Fidelis Care New
York (“Fidelis Care”) as of December 31, 2016 and 2015, and for each of the years in the
two-year
period ended December 31, 2016, and the unaudited interim financial statements of Fidelis
Care as of and for the nine months ended September 30, 2017, which are incorporated by reference herein from our Current Report on Form
8-K,
with the SEC on March 13, 2018 (the “Fidelis Form
8-K”).
We will provide to each person, including any beneficial owner, to whom a prospectus supplement is delivered, a copy of any or all of the information that has been incorporated by reference in the
prospectus supplement but not delivered with the prospectus supplement, at no charge upon written or oral request by contacting us at Centene Corporation, Attn: Corporate Secretary, 7700 Forsyth Boulevard, St. Louis, Missouri 63105, telephone (314)
725-4477.
This prospectus supplement does not contain all of the information set forth in the
registration statement or in the exhibits and schedules thereto, in accordance with the rules and regulations of the SEC, and we refer you to that omitted information. The statements made in this prospectus supplement pertaining to the content of
any contract, agreement or other document that is an exhibit to the registration statement or the documents incorporated by reference in this prospectus supplement necessarily are summaries of their material provisions and we qualify those
statements in their entirety by reference to those definitive agreements and those exhibits for complete statements of their provisions. The documents incorporated by reference in this prospectus supplement and the registration statement and its
exhibits and schedules are available at the SEC’s public reference room or through its website.
S-iii
CAUTIONARY STATEMENT CONCERNING FORWARD-LOOKING STATEMENTS
Statements set forth in this prospectus supplement, the accompanying prospectus and incorporated by reference herein and therein from
documents we have filed with the SEC may contain forward-looking statements within the meaning of Section 27A of the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). We
intend such forward-looking statements to be covered by the safe-harbor provisions for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995, and we are including this statement for purposes of complying with
these safe-harbor provisions. In particular, these statements include without limitation statements about our market opportunity, our growth strategy, competition, expected activities and future acquisitions, including the proposed acquisition of
Fidelis Care (the “Proposed Fidelis Acquisition”), investments and the adequacy of our available cash resources. Readers are cautioned that matters subject to forward-looking statements involve known and unknown risks and uncertainties,
including economic, regulatory, competitive and other factors that may cause our or our industry’s actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity,
performance or achievements expressed or implied by these forward-looking statements. These statements are not guarantees of future performance and are subject to risks, uncertainties and assumptions.
All forward-looking statements included or incorporated by reference in this prospectus supplement are based on information available to us on
the date of this prospectus supplement. Except as may be otherwise required by law, we undertake no obligation to update or revise the forward-looking statements included or incorporated by reference in this prospectus supplement, whether as a
result of new information, future events or otherwise, after the date of this prospectus supplement. You should not place undue reliance on any forward-looking statements, as actual results may differ materially from projections, estimates or other
forward-looking statements due to a variety of important factors, including but not limited to:
|
|
•
|
|
our ability to accurately predict and effectively manage health benefits and other operating expenses and reserves;
|
|
|
•
|
|
membership and revenue declines or unexpected trends;
|
|
|
•
|
|
changes in healthcare practices, new technologies and advances in medicine;
|
|
|
•
|
|
increased healthcare costs;
|
|
|
•
|
|
changes in economic, political or market conditions;
|
|
|
•
|
|
changes in federal or state laws or regulations, including changes with respect to government healthcare programs as well as changes with respect to the Patient Protection and Affordable Care Act and the Healthcare and
Education Affordability Reconciliation Act and any regulations enacted thereunder that may result from changing political conditions;
|
|
|
•
|
|
rate cuts or other payment reductions or delays by governmental payors and other risks and uncertainties affecting our government business;
|
|
|
•
|
|
our ability to adequately price products on federally facilitated and state based Health Insurance Marketplaces;
|
|
|
•
|
|
disasters or major epidemics;
|
|
|
•
|
|
the outcome of legal and regulatory proceedings;
|
S-iv
|
|
•
|
|
changes in expected contract start dates;
|
|
|
•
|
|
provider, state, federal and other contract changes and timing of regulatory approval of contracts;
|
|
|
•
|
|
the expiration, suspension or termination of our or Fidelis Care’s contracts with federal or state governments (including but not limited to Medicaid, Medicare and TRICARE);
|
|
|
•
|
|
the difficulty of predicting the timing or outcome of pending or future litigation or government investigations;
|
|
|
•
|
|
challenges to our or Fidelis Care’s contract awards;
|
|
|
•
|
|
cyber-attacks or other privacy or data security incidents;
|
|
|
•
|
|
the possibility that the expected synergies and value creation from acquired businesses, including, without limitation, the acquisition (the “Health Net Acquisition”) of Health Net, and the Proposed Fidelis
Acquisition, will not be realized, or will not be realized within the expected time period, including, but not limited to, as a result of any failure to obtain any regulatory, governmental or third party consents or approvals in connection with the
Proposed Fidelis Acquisition (including any such approvals under the New York
Non-For-Profit
Corporation Law) or any conditions, terms, obligations or restrictions
imposed in connection with the receipt of such consents or approvals;
|
|
|
•
|
|
the exertion of management’s time and our resources, and other expenses incurred and business changes required in connection with complying with the undertakings in connection with any regulatory, governmental or
third party consents or approvals for the Health Net Acquisition;
|
|
|
•
|
|
disruption caused by significant completed and pending acquisitions, including the Health Net Acquisition and the Proposed Fidelis Acquisition, making it more difficult to maintain business and operational
relationships;
|
|
|
•
|
|
the risk that unexpected costs will be incurred in connection with the completion and/or integration of acquisition transactions, including among others, the Health Net Acquisition and the Proposed Fidelis Acquisition;
|
|
|
•
|
|
changes in expected closing dates, estimated purchase price and accretion for acquisitions;
|
|
|
•
|
|
the risk that acquired businesses, including Health Net and Fidelis Care, will not be integrated successfully;
|
|
|
•
|
|
the risk that the conditions to the completion of the Proposed Fidelis Acquisition may not be satisfied or completed on a timely basis, or at all;
|
|
|
•
|
|
failure to obtain or receive any required regulatory approvals, consents or clearances for the Proposed Fidelis Acquisition, and the risk that, even if so obtained or received, regulatory authorities impose conditions
on the completion of the transaction that could require the exertion of management’s time and our resources or otherwise have an adverse effect on Centene;
|
|
|
•
|
|
business uncertainties and contractual restrictions while the Proposed Fidelis Acquisition is pending, which could adversely affect our business and operations;
|
|
|
•
|
|
change of control provisions or other provisions in certain agreements to which Fidelis Care is a party, which may be triggered by the completion of the Proposed Fidelis Acquisition;
|
|
|
•
|
|
loss of management personnel and other key employees due to uncertainties associated with the Proposed Fidelis Acquisition;
|
S-v
|
|
•
|
|
the risk that, following completion of the Proposed Fidelis Acquisition, the combined company may not be able to effectively manage its expanded operations;
|
|
|
•
|
|
restrictions and limitations that may stem from the financing arrangements that the combined company will enter into in connection with the Proposed Fidelis Acquisition;
|
|
|
•
|
|
our ability to achieve improvement in the Centers for Medicare and Medicaid Services Star ratings and maintain or achieve improvement in other quality scores in each case that can impact revenue and future growth;
|
|
|
•
|
|
availability of debt and equity financing, on terms that are favorable to us;
|
|
|
•
|
|
foreign currency fluctuations.
|
This list of important factors is not intended to be
exhaustive. We discuss certain of these matters more fully, as well as certain other risk factors that may affect our business operations, financial condition and results of operations, in our filings with the SEC, including our annual reports on
Form
10-K,
quarterly reports on Form
10-Q
and current reports on Form
8-K.
All statements, other than statements of current or historical fact, contained in this filing are forward-looking statements. We have
attempted to identify these statements by terminology including “believe,” “anticipate,” “plan,” “expect,” “estimate,” “intend,” “seek,” “target,” “goal,”
“may,” “will,” “would,” “could,” “should,” “can,” “continue” and other similar words or expressions in connection with, among other things, any discussion of future operating or
financial performance. In particular, these statements include statements about our market opportunity, our growth strategy, competition, expected activities and future acquisitions, investments and the adequacy of our available cash resources.
Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such
forward looking statements. See “Risk Factors” beginning on page
S-4
of this prospectus for reference to the factors that could cause actual results to differ materially.
You should not place undue reliance on such statements, which speak only as of the date that they were made. These cautionary statements
should be considered in connection with any written or oral forward-looking statements that we may issue in the future. We do not undertake any obligation to release publicly any revisions to such forward looking statements to reflect later events
or circumstances or to reflect the occurrence of unanticipated events.
S-vi
SUMMARY
This summary highlights information contained or incorporated by reference in this prospectus supplement and the accompanying prospectus.
Because this is a summary, it may not contain all of the information that is important to you. Before making an investment decision, you should read the entire prospectus supplement, the accompanying prospectus and the documents incorporated by
reference, including the section entitled “Risk Factors” in this prospectus supplement and such risk factors as may be updated from time to time in our public filings.
Overview
Centene is a diversified,
multi-national healthcare enterprise that provides a portfolio of services to government sponsored and commercial healthcare programs, focusing on under-insured and uninsured individuals. Centene provides member-focused services through locally
based staff by assisting in accessing care, coordinating referrals to related health and social services and addressing member concerns and questions. Centene also provides education and outreach programs to inform and assist members in accessing
quality, appropriate healthcare services. Centene believes its local approach, including member and provider services, enables it to provide accessible, quality, culturally-sensitive healthcare coverage to its communities. Centene’s health
management, educational and other initiatives are designed to help members best utilize the healthcare system to ensure they receive appropriate, medically necessary services and effective management of routine, severe and chronic health problems,
resulting in better health outcomes. Centene combines its decentralized local approach for care with a centralized infrastructure of support functions such as finance, information systems and claims processing.
In September 2017, we signed a definitive agreement under which Fidelis Care (as defined herein) will become our health plan in New York
State. Under the terms of the agreement, we will acquire substantially all of the assets of Fidelis Care for $3.75 billion, subject to certain adjustments.
Centene operates in two segments: Managed Care and Specialty Services. Centene’s Managed Care segment provides health plan coverage to
individuals through government subsidized and commercial programs including Medicaid, the State Children’s Health Insurance Program (CHIP), Long-Term Services and Supports (LTSS), Medicare, Foster Care, Supplemental Security Income Program,
also known as the Aged, Blind or Disabled, or collectively ABD, and Medicare-Medicaid Plans (MMP), which cover beneficiaries who are dually eligible for Medicare and Medicaid. In addition, our commercial operations, which include our members through
the Health Insurance Marketplace, are included within the Managed Care segment. Centene’s Specialty Services segment consists of Centene’s specialty companies offering diversified healthcare services and products to state programs,
correctional facilities, healthcare organizations, employer groups, military service members and their families, and other commercial organizations, as well as to its own subsidiaries. For the year ended December 31, 2017, Centene’s
Managed Care and Specialty Services segments accounted for 95% and 5%, respectively, of Centene’s total external revenues.
Centene’s membership totaled 12.2 million as of December 31, 2017. For the year ended December 31, 2017, Centene’s
total revenues and net earnings from continuing operations attributable to Centene were $48.4 billion and $828 million, respectively, and its total cash flow from operations was $1.5 billion.
Our initial health plan commenced operations in Wisconsin in 1984. We were organized in Wisconsin in 1993 as a holding company for our initial
health plan and reincorporated in Delaware in 2001.
The selling stockholder acquired the shares of our common stock which are the subject
of this prospectus supplement in connection with our acquisition of CMG on March 12, 2018. See “Selling Stockholder” below.
The principal executive offices are located at 7700 Forsyth Boulevard, St. Louis, Missouri 63105, and our telephone number is (314)
725-4477.
Centene’s website address is www.centene.com. We do not incorporate the information contained on our website herein, and you should not consider it part of this prospectus supplement or the
accompanying prospectus.
Our common stock is publicly traded on the NYSE under the ticker symbol “CNC.”
S-1
THE OFFERING
The summary below contains basic information about this offering. It does not contain all of the information you should consider in making
your investment decision. You should read the entire prospectus supplement and accompanying prospectus and the information included or incorporated and deemed to be incorporated by reference herein and therein before making an investment decision.
As used in this section, except where otherwise indicated, the terms “us,” “we” and “our” refer to Centene Corporation and not to any of its subsidiaries.
|
|
|
|
|
Issuer
|
|
Centene Corporation, a Delaware corporation.
|
|
|
|
|
Securities offered by selling stockholder
|
|
1,448,714 shares of common stock, par value $0.001 per share.
|
|
|
|
|
Number of shares to be outstanding after this offering
|
|
175,011,676 shares (based on 175,011,676 shares of our common stock outstanding as of March 12, 2018).
|
|
|
|
|
Use of Proceeds
|
|
We will not receive any proceeds from the sale of shares of common stock by the selling stockholder in this offering.
|
|
|
|
|
NYSE Symbol
|
|
“CNC.”
|
|
|
|
|
Transfer agent and registrar
|
|
The transfer agent and registrar for our common stock is Broadridge Corporate Issuer Solutions, Inc.
|
S-2
RISK FACTORS
An investment in our common stock involves a number of risks. You should carefully consider all the information set forth in this
prospectus supplement and the accompanying prospectus and incorporated by reference herein and therein before deciding to invest in the common stock. In particular, we urge you to consider carefully the factors set forth below and such risk factors
as may be updated from time to time in our public filings. Any of these risks could materially and adversely affect our business, financial condition and results of operations and the actual outcome of matters as to which forward-looking statements
are made in this prospectus supplement and the accompanying prospectus. While we believe we have identified and discussed below, in the accompanying prospectus and in the documents incorporated by reference herein and therein, the material risks
affecting our business, there may be additional risks and uncertainties that we do not presently know or that we do not currently believe to be material that may adversely affect such business, financial condition and results of operations in the
future. Unless the context otherwise requires, the terms the “Company,” “we,” “us,” “our” or similar terms and “Centene” refer to Centene Corporation, together with its consolidated subsidiaries.
Risks Related to Ownership of Our Common Stock
Our stock price has fluctuated in the past and may fluctuate in the future. Accordingly, you may not be able to resell your shares at or
above the price at which you purchased them.
The trading price of our common stock has fluctuated in the past. The trading price
of our common stock could fluctuate significantly in the future and could be negatively affected in response to various factors including:
|
|
•
|
|
conditions in the broader stock market in general;
|
|
|
•
|
|
our ability to make investments with attractive risk-adjusted returns;
|
|
|
•
|
|
market perception of our current and projected financial condition, potential growth, future earnings and future cash dividends;
|
|
|
•
|
|
announcements we make regarding dividends;
|
|
|
•
|
|
actual or anticipated fluctuations in our quarterly financial and operating results;
|
|
|
•
|
|
additional offerings of our common stock or equity-linked securities;
|
|
|
•
|
|
actions by rating agencies;
|
|
|
•
|
|
short sales of our common stock;
|
|
|
•
|
|
any decision to pursue a distribution or disposition of a meaningful portion of our assets;
|
|
|
•
|
|
issuance of new or changed securities analysts’ reports or recommendations;
|
|
|
•
|
|
market perception or media coverage of us, other similar companies or the outlook of the markets and industries in which we compete;
|
|
|
•
|
|
major reductions in trading volumes on the exchanges on which we operate;
|
|
|
•
|
|
legislative or regulatory developments, including changes in the status of our regulatory approvals or licenses; and
|
|
|
•
|
|
litigation and governmental investigations.
|
S-3
These and other factors may cause the market price and demand for our common stock to fluctuate
substantially, which may negatively affect the price or liquidity of our common stock. In addition, the market price of our common stock may fluctuate significantly following consummation of the Proposed Fidelis Acquisition if, among other things,
the combined company is unable to achieve the expected growth in earnings, or if the operational cost savings estimates in connection with the integration of our and Fidelis Care’s businesses are not realized, or if the transaction costs
relating to the Proposed Fidelis Acquisition are greater than expected, or if the financing relating to the transaction is on unfavorable terms. The market price also may decline if the combined company does not achieve the perceived benefits of the
Proposed Fidelis Acquisition as rapidly or to the extent anticipated by financial or industry analysts or if the effect of the Proposed Fidelis Acquisition on the combined company’s financial position, results of operations or cash flows is not
consistent with the expectations of financial or industry analysts. In addition, our business differs from that of Fidelis Care, and accordingly, the results of operations of the combined company and the market price of our common stock after the
completion of the Proposed Fidelis Acquisition may be affected by factors different from those currently affecting the independent results of operations of each of our and Fidelis Care’s businesses.
When the market price of a stock has been volatile or has decreased significantly in the past, holders of that stock have, at times,
instituted securities class action litigation against the company that issued the stock. If any of our stockholders brought a lawsuit against us, we could incur substantial costs defending, settling or paying any resulting judgments related to the
lawsuit. Such a lawsuit could also divert the time and attention of our management from our business and hurt our share price.
Shares eligible for future sale may adversely affect our common stock price.
Sales of our common stock or other securities in the public or private market, or the perception that these sales may occur, could cause the
market price of our common stock to decline. This could also impair our ability to raise additional capital through the sale of our equity securities. Under our certificate of incorporation, we are authorized to issue up to 410,000,000 shares of
capital stock, consisting of 400,000,000 shares of common stock, par value $0.001 per share, and 10,000,000 shares of preferred stock, par value $0.001 per share. We cannot predict the size of future issuances of our common stock or other securities
or the effect, if any, that future sales and issuances of our common stock and other securities would have on the market price of our common stock.
Future issuances and sales of additional shares of preferred or common stock, including shares issued in connection with the Proposed
Fidelis Acquisition, could reduce the market price of our shares of common stock.
Subject to market conditions, we intend to fund
the purchase price for the Proposed Fidelis Acquisition with $2.3 billion of new equity, including shares paid as consideration. Any such issuances and sales of our preferred or common stock could have the effect of depressing the market price
for our common stock. Further, any sale of shares to finance a portion of the purchase price for the Proposed Fidelis Acquisition will be subject to market conditions and could be negatively impacted by a decline in the market price for our common
stock. In addition, in the future we may issue additional securities to raise capital or in connection with acquisitions. We often acquire interests in other companies by using a combination of cash and our common stock or just our common stock.
Further, shares of preferred stock may be issued from time to time in one or more series as our Board may from time to time determine each such series to be distinctively designated. The issuance of any such preferred stock could materially
adversely affect the rights of holders of our common stock. Any of these events may dilute your ownership interest in our company and have an adverse impact on the price of our common stock.
Our shares of common stock will rank junior to all of our consolidated liabilities.
In the event of a bankruptcy, liquidation, dissolution or winding up, our assets will be available to pay obligations on the common stock only
after all of our consolidated liabilities have been paid. In the event of a bankruptcy, liquidation, dissolution or winding up, there may not be sufficient assets remaining, after paying our and our subsidiaries’ liabilities, to pay any amounts
with respect to the common stock then outstanding. We have a significant amount of debt, which amounted to $4.7 billion as of December 31, 2017, with $1,500 million of availability under our existing revolving credit facility and
access to an additional $500 million of availability subject
S-4
to lender commitments. We expect to incur additional debt to fund the Proposed Fidelis Acquisition. On a pro forma basis to give effect to the Proposed Fidelis Acquisition and the other events
described under “Unaudited Pro Forma Condensed Combined Financial Information” herein, we would have had $6.2 billion of outstanding debt on a consolidated basis as of September 30, 2017, with $805 million of availability
under our revolving credit facility, and may also take on additional long-term debt and working capital lines of credit to meet future financing needs, subject to certain restrictions under the terms of our existing debt.
Our corporate documents and provisions of Delaware law may prevent a change in control or management that stockholders may consider
desirable.
Section 203 of the Delaware General Corporation Law, laws of states in which we operate, and our certificate of
incorporation and
by-laws
contain provisions that might enable our management to resist a takeover of our company. These provisions could have the effect of delaying, deferring, or preventing a change in
control of Centene or a change in our management that stockholders may consider favorable or beneficial. These provisions could also discourage proxy contests and make it more difficult for you and other stockholders to elect directors and take
other corporate actions. These provisions could also limit the price that investors might be willing to pay in the future for shares of our common stock.
S-5
USE OF PROCEEDS
We will not receive any proceeds from the shares of common stock sold by the selling stockholder.
S-6
SELLING STOCKHOLDER
In connection with our acquisition of CMG on March 12, 2018, we issued 1,448,714 shares of our common stock as a portion of the
consideration in a private placement to the selling stockholder. We are registering these shares of common stock in order to permit the selling stockholder to offer the shares for resale from time to time.
The following table sets forth the information about the selling stockholder, including the number of shares of our common stock owned by or
attributable to the selling stockholder immediately prior to this registration (including the shares offered by this prospectus supplement), the number of shares offered hereby and registered by the registration statement of which this prospectus
supplement is a part, and the number of shares of our common stock to be owned by the selling stockholder after this offering. The number of shares to be owned after this offering assumes that all shares covered by this prospectus supplement will be
sold by the selling stockholder and that no additional shares of our common stock are subsequently bought or sold by the selling stockholder.
|
|
|
|
|
|
|
|
|
|
|
|
|
Name
|
|
Ownership Prior to
the Registration
|
|
Shares
Covered by
this
Registration
Statement
|
|
Ownership After the
Shares are Sold
|
|
|
|
Number of
Shares
|
|
Percent
|
|
|
Number
of
Shares
|
|
Percent
|
|
CMG Holding Company, LLC
(1)
|
|
1,448,714
|
|
*
|
|
1,448,714
|
|
—
|
|
*
|
|
*
|
Represents less than 1% of the total outstanding shares of our common stock.
|
|
(1)
|
The shares are held directly by CMG Holding Company, LLC, a Delaware limited liability company (“CMG Holding”). CMG Holding is managed through a board of
directors. Through their power to appoint representatives holding a majority of the voting power of the CMG Holding board of directors, Nautic Partners VII, L.P. and Nautic Partners
VII-A,
L.P., each a
Delaware limited partnership (and collectively, the “Nautic VII Funds”), may be deemed to have the shared power to vote or direct the vote of (and the shared power to dispose or direct the disposition of) the shares held by CMG Holding.
Nautic Capital VII, L.P. (the “Nautic GP”) is the general partner of each of the Nautic VII Funds, and Nautic Associates VII, LLC (the “Nautic Ultimate GP”) is the general partner of the Nautic GP. By virtue of their respective
positions as members of the investment committee of the Nautic GP, Bernard V. Buonanno, III, Christopher F. Corey, Christopher J. Crosby, Habib Y. Gorgi, Scott F. Hilinski, Douglas C. Hill and Christopher A. Pierce may be deemed to share beneficial
ownership, together with the Nautic GP, the Nautic Ultimate GP and each of the Nautic VII Funds, of the shares held by CMG Holding. Each such person disclaims beneficial ownership of the shares held by CMG Holding except to the extent of such
person’s pecuniary interest in such shares.
|
S-7
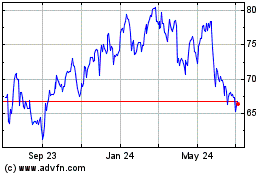
PRICE RANGE OF OUR COMMON STOCK AND DIVIDENDS
Our common stock is listed and traded on the NYSE under the symbol “CNC.” The following table sets forth, for the periods indicated,
the high and low
intra-day
sale prices of our common stock as reported on the NYSE:
|
|
|
|
|
|
|
|
|
|
|
2016
|
|
High
|
|
|
Low
|
|
|
First Quarter
|
|
$
|
68.42
|
|
|
$
|
47.36
|
|
|
Second Quarter
|
|
$
|
71.53
|
|
|
$
|
55.60
|
|
|
Third Quarter
|
|
$
|
75.57
|
|
|
$
|
63.37
|
|
|
Fourth Quarter
|
|
$
|
67.41
|
|
|
$
|
50.00
|
|
|
|
|
|
|
2017
|
|
High
|
|
|
Low
|
|
|
First Quarter
|
|
$
|
73.23
|
|
|
$
|
56.00
|
|
|
Second Quarter
|
|
$
|
85.80
|
|
|
$
|
69.20
|
|
|
Third Quarter
|
|
$
|
98.72
|
|
|
$
|
79.06
|
|
|
Fourth Quarter
|
|
$
|
104.65
|
|
|
$
|
83.56
|
|
|
|
|
|
|
2018
|
|
High
|
|
|
Low
|
|
|
First Quarter (through March 12, 2018)
|
|
$
|
112.42
|
|
|
$
|
97.61
|
|
The closing sale price of our common stock as reported on the NYSE on March 12, 2018 was $100.30 per
share. For a description of our common stock, see “Description of Capital Stock” in the accompanying prospectus and our certificate of incorporation, which is incorporated herein by reference.
Dividend Policy
We have never declared
any cash dividends on our shares of common stock and currently anticipate that we will retain any future earnings for the development, operation and expansion of our business.
S-8
UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION OF
CENTENE AND FIDELIS CARE
The unaudited pro forma condensed combined statements of operations for the nine months ended September 30, 2017, combine the historical
consolidated statements of operations of Centene and Fidelis Care, giving effect to the Acquisition Financing (as defined in Note 1 herein) and the Proposed Fidelis Acquisition as if they each had occurred on January 1, 2017. The unaudited pro
forma condensed combined balance sheet as of September 30, 2017, combines the historical consolidated balance sheets of the Company and Fidelis Care, giving effect to the Acquisition Financing and the Proposed Fidelis Acquisition as if they
each had occurred on September 30, 2017. The historical consolidated financial information has been adjusted in the unaudited pro forma condensed combined financial statements to give effect to pro forma events that are (i) directly
attributable to the Proposed Fidelis Acquisition, (ii) factually supportable and (iii) with respect to the statements of operations, expected to have a continuing impact on the combined results. The unaudited pro forma condensed combined
financial information should be read in conjunction with the accompanying notes to the unaudited pro forma condensed combined financial statements. In addition, the unaudited pro forma condensed combined financial information was derived from and
should be read in conjunction with the following historical consolidated financial statements and accompanying notes:
|
|
•
|
|
separate historical unaudited interim financial statements of the Company as of and for the nine months ended September 30, 2017, and the related notes included in the Company’s Quarterly Report on Form
10-Q
for the quarterly period ended September 30, 2017; and
|
|
|
•
|
|
separate historical unaudited interim financial statements of Fidelis Care as of and for the nine months ended September 30, 2017, and the related notes, filed on the Fidelis Form 8-K as Exhibit 99.2.
|
The unaudited pro forma condensed combined financial information has been prepared by us using the acquisition method of
accounting in accordance with GAAP. We have been treated as the acquirer in the Proposed Fidelis Acquisition for accounting purposes. The acquisition accounting is dependent upon certain valuation and other studies that have yet to commence or
progress to a stage where there is sufficient information for a definitive measurement. The consummation of the Proposed Fidelis Acquisition remains subject to the satisfaction of customary closing conditions, including the receipt of regulatory
approvals, and there can be no assurance that the Proposed Fidelis Acquisition will occur on or before a certain time, on the terms described herein, or at all. The Proposed Fidelis Acquisition or any other financing transaction are not conditioned
upon each other. In addition, under certain relevant laws and regulations, before completion of the Proposed Fidelis Acquisition, there are certain limitations regarding what we can learn about Fidelis Care. Until the Proposed Fidelis Acquisition is
completed, we will not have complete access to all relevant information. The assets and liabilities of Fidelis Care have been measured based on various preliminary estimates using assumptions that we believe are reasonable based on information that
is currently available. Differences between these preliminary estimates and the final acquisition accounting may occur, and those differences could have a material impact on the accompanying unaudited pro forma condensed combined financial
statements and the combined company’s future results of operations and financial position. The pro forma adjustments are preliminary and have been made solely for informational purposes.
We intend to commence the necessary valuation and other studies required to complete the acquisition accounting promptly upon completion of
the Proposed Fidelis Acquisition and will finalize the acquisition accounting as soon as practicable within the required measurement period in accordance with ASC 805, but in no event later than one year following completion of the Proposed Fidelis
Acquisition.
S-9
The unaudited pro forma adjustments are based upon available information and certain assumptions
that our management believes are reasonable. The unaudited pro forma condensed combined financial information has been presented for informational purposes only and is based on assumptions and estimates considered appropriate by our management;
however, it is not necessarily indicative of our financial position or results of operations that would have been achieved had the pro forma events taken place on the dates indicated, or of the future consolidated results of operations or of the
financial position of the combined company. You should not place undue reliance on the summary unaudited pro forma condensed combined financial information in deciding whether or not to purchase our common stock.
Management expects that the strategic and financial benefits of the Proposed Fidelis Acquisition will result in certain cost savings
opportunities. However, given the preliminary nature of those cost savings, they have not been reflected in the accompanying unaudited pro forma condensed combined statements of operations for either period. For a discussion of risks related to
anticipated cost savings, see “Risk Factors—Factors that may affect Future Results and the Trading Price of Our Common Stock—The combined company may be unable to successfully integrate our business with the assets acquired in the
Proposed Fidelis Acquisition and realize the anticipated benefits of the Proposed Fidelis Acquisition” in Item 1A. of Part I of our Annual Report on
Form 10-K
for the year ended
December 31, 2017.
S-10
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(in millions, except per share data in dollars and shares in thousands)
|
|
Nine Months Ended September 30, 2017
|
|
|
|
Centene
|
|
|
Fidelis Care
|
|
|
Pro Forma
Adjustments
(Note 6)
|
|
|
Pro
Forma
Combined
|
|
|
Unaudited Pro Forma Condensed Combined Statement of Operations
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Revenues:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Premium
|
|
$
|
32,393
|
|
|
$
|
7,263
|
|
|
$
|
15
|
(a)
|
|
$
|
39,671
|
|
|
Service
|
|
|
1,634
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,634
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Premium and service revenues
|
|
|
34,027
|
|
|
|
7,263
|
|
|
|
15
|
|
|
|
41,305
|
|
|
Premium tax
|
|
|
1,549
|
|
|
|
—
|
|
|
|
122
|
(a)
|
|
|
1,671
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total revenues
|
|
|
35,576
|
|
|
|
7,263
|
|
|
|
137
|
|
|
|
42,976
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Medical costs
|
|
|
28,278
|
|
|
|
6,546
|
|
|
|
—
|
|
|
|
34,824
|
|
|
Cost of services
|
|
|
1,334
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,334
|
|
|
Selling, general and administrative expenses
|
|
|
3,186
|
|
|
|
417
|
|
|
|
—
|
|
|
|
3,603
|
|
|
Amortization of acquired intangible assets
|
|
|
117
|
|
|
|
—
|
|
|
|
58
|
(b)
|
|
|
175
|
|
|
Premium tax expense
|
|
|
1,643
|
|
|
|
—
|
|
|
|
137
|
(a)
|
|
|
1,780
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total operating expenses
|
|
|
34,558
|
|
|
|
6,963
|
|
|
|
195
|
|
|
|
41,716
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Earnings from operations
|
|
|
1,018
|
|
|
|
300
|
|
|
|
(58
|
)
|
|
|
1,260
|
|
|
Other income (expense):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Investment and other income
|
|
|
137
|
|
|
|
71
|
|
|
|
(15
|
)(c)
|
|
|
193
|
|
|
Interest expense
|
|
|
(189
|
)
|
|
|
(2
|
)
|
|
|
(61
|
)(d)
|
|
|
(252
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Earnings before income tax expense
|
|
|
966
|
|
|
|
369
|
|
|
|
(134
|
)
|
|
|
1,201
|
|
|
Income tax expense
|
|
|
381
|
|
|
|
—
|
|
|
|
91
|
(e)
|
|
|
472
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net earnings
|
|
|
585
|
|
|
|
369
|
|
|
|
(225
|
)
|
|
|
729
|
|
|
Loss attributable to noncontrolling interests
|
|
|
13
|
|
|
|
—
|
|
|
|
—
|
|
|
|
13
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net earnings attributable to common stockholders
|
|
$
|
598
|
|
|
$
|
369
|
|
|
$
|
(225
|
)
|
|
$
|
742
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net earnings per common share:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic earnings per common share
|
|
$
|
3.47
|
|
|
|
|
|
|
|
|
|
|
$
|
3.80
|
|
|
Diluted earnings per common share
|
|
$
|
3.39
|
|
|
|
|
|
|
|
|
|
|
$
|
3.73
|
|
|
Weighted average number of common shares outstanding:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic
|
|
|
172,314
|
|
|
|
|
|
|
|
22,766
|
(f)
|
|
|
195,080
|
|
|
Diluted
|
|
|
176,414
|
|
|
|
|
|
|
|
22,766
|
(f)
|
|
|
199,180
|
|
See the accompanying notes to the unaudited pro forma condensed combined financial statements, which are an
integral part of this statement. The pro forma adjustments are explained in Note 6.
Income Statement Pro Forma Adjustments
.
S-11
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(in millions, except shares in thousands)
|
|
As of September 30, 2017
|
|
|
|
Centene
|
|
|
Fidelis Care
|
|
|
Pro Forma
Adjustments
(Note 7)
|
|
|
Pro Forma
Combined
|
|
|
Unaudited Pro Forma Condensed Combined Balance Sheet
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
ASSETS
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
4,281
|
|
|
$
|
1,721
|
|
|
$
|
(578
|
)(a)
|
|
$
|
5,424
|
|
|
Premium and related receivables
|
|
|
3,955
|
|
|
|
194
|
|
|
|
—
|
|
|
|
4,149
|
|
|
Short-term investments
|
|
|
595
|
|
|
|
1,316
|
|
|
|
(100
|
)(a)
|
|
|
1,811
|
|
|
Other current assets
|
|
|
829
|
|
|
|
213
|
|
|
|
31
|
(f)
|
|
|
1,073
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
9,660
|
|
|
|
3,444
|
|
|
|
(647
|
)
|
|
|
12,457
|
|
|
Long-term investments
|
|
|
4,927
|
|
|
|
—
|
|
|
|
—
|
|
|
|
4,927
|
|
|
Restricted deposits
|
|
|
138
|
|
|
|
450
|
|
|
|
—
|
|
|
|
588
|
|
|
Property, software and equipment, net
|
|
|
1,003
|
|
|
|
197
|
|
|
|
(139
|
)(a)
|
|
|
1,061
|
|
|
Goodwill
|
|
|
4,712
|
|
|
|
16
|
|
|
|
1,337
|
(b)
|
|
|
6,065
|
|
|
Intangible assets, net
|
|
|
1,428
|
|
|
|
—
|
|
|
|
1,000
|
(c)
|
|
|
2,428
|
|
|
Other long-term assets
|
|
|
132
|
|
|
|
—
|
|
|
|
—
|
|
|
|
132
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total assets
|
|
$
|
22,000
|
|
|
$
|
4,107
|
|
|
$
|
1,551
|
|
|
$
|
27,658
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES, REDEEMABLE NONCONTROLLING INTERESTS, STOCKHOLDERS’ EQUITY AND NET
ASSETS
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Medical claims liability
|
|
$
|
4,333
|
|
|
$
|
1,325
|
|
|
$
|
—
|
|
|
$
|
5,658
|
|
|
Accounts payable and accrued expenses
|
|
|
4,804
|
|
|
|
448
|
|
|
|
117
|
(a),(c),(f)
|
|
|
5,369
|
|
|
Unearned revenue
|
|
|
568
|
|
|
|
106
|
|
|
|
—
|
|
|
|
674
|
|
|
Current portion of long-term debt
|
|
|
4
|
|
|
|
14
|
|
|
|
(14
|
)(a)
|
|
|
4
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
9,709
|
|
|
|
1,893
|
|
|
|
103
|
|
|
|
11,705
|
|
|
Long-term debt
|
|
|
4,717
|
|
|
|
75
|
|
|
|
1,407
|
(a),(d)
|
|
|
6,199
|
|
|
Other long-term liabilities
|
|
|
901
|
|
|
|
—
|
|
|
|
—
|
|
|
|
901
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities
|
|
|
15,327
|
|
|
|
1,968
|
|
|
|
1,510
|
|
|
|
18,805
|
|
|
Commitments and contingencies:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Redeemable noncontrolling interests
|
|
|
20
|
|
|
|
—
|
|
|
|
—
|
|
|
|
20
|
|
|
Stockholders’ equity:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Preferred stock
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Common stock
(1)
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Additional
paid-in
capital
|
|
|
4,310
|
|
|
|
—
|
|
|
|
2,234
|
(e)
|
|
|
6,544
|
|
|
Accumulated other comprehensive earnings
|
|
|
9
|
|
|
|
—
|
|
|
|
—
|
|
|
|
9
|
|
|
Retained earnings
|
|
|
2,518
|
|
|
|
—
|
|
|
|
(54
|
)(f)
|
|
|
2,464
|
|
|
Treasury stock, at cost
|
|
|
(197
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(197
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total stockholders’ equity attributable to common stockholders
|
|
|
6,640
|
|
|
|
—
|
|
|
|
2,180
|
|
|
|
8,820
|
|
|
Noncontrolling interest
|
|
|
13
|
|
|
|
—
|
|
|
|
—
|
|
|
|
13
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total stockholders’ equity
|
|
|
6,653
|
|
|
|
—
|
|
|
|
2,180
|
|
|
|
8,833
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net assets
|
|
$
|
—
|
|
|
$
|
2,139
|
|
|
$
|
(2,139
|
)(g)
|
|
$
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities, redeemable noncontrolling interests, stockholders’ equity and net
assets
|
|
$
|
22,000
|
|
|
$
|
4,107
|
|
|
$
|
1,551
|
|
|
$
|
27,658
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
|
On a historical basis, share information of the Company is as follows: 400,000 shares authorized; 172,566 shares issued and outstanding. On a pro forma combined basis, share information is as follows: 400,000
shares authorized; 195,332 shares issued and outstanding.
|
See the accompanying notes to the unaudited pro forma condensed
combined financial statements, which are an integral part of this statement. The pro forma adjustments are explained in Note 7.
Balance Sheet Pro Forma Adjustments.
S-12
NOTES TO THE PRO FORMA CONDENSED COMBINED FINANCIAL STATEMENTS
(DOLLARS IN MILLIONS, EXCEPT SHARE DATA)
(UNAUDITED)
1. Description of
Transaction
On September 12, 2017, we entered into an Asset Purchase Agreement (the “Asset Purchase Agreement”) with
Fidelis Care to acquire substantially all of the assets (the “Acquired Assets”) and assume certain liabilities of Fidelis Care. The Asset Purchase Agreement provides, among other things, that upon the terms and subject to the conditions
set forth therein, a newly formed New York health maintenance organization and a wholly owned subsidiary of the Company (the “Buyer”) will purchase the Acquired Assets for a total of $3.75 billion, subject to certain closing and
post-closing adjustments, as described in the Asset Purchase Agreement (the “Acquisition Consideration”). The composition of the Acquisition Consideration, between Cash Consideration (as defined below) and Share Consideration (as defined
below), if any, is variable under the Asset Purchase Agreement, subject to the Share Consideration Election Amount (as defined below).
Under the terms of the Asset Purchase Agreement, no later than five business days prior to the closing of the Proposed Fidelis Acquisition, we
may elect to deliver Acquisition Consideration that consists of (1) solely Cash Consideration or (2) a combination of Cash Consideration and Share Consideration. Subject to certain limitations set forth in the Asset Purchase Agreement, we have the
option to fund up to $500.0 million of the Acquisition Consideration in our common stock, of which (i) up to $125.0 million may be used to fund the Acquisition Consideration payable at the closing of the Proposed Fidelis Acquisition (such
amount, if any, the “Share Consideration Election Amount” and such shares, the “Share Consideration”) and (ii) up to $375.0 million may be used to fund the Escrow Fund (as defined below) (such amount, the “Escrow Share
Amount” and such shares, the “Escrow Shares”). The number of shares of our common stock comprising the Share Consideration Election Amount, if any, and/or the Escrow Share Amount, if any, will be determined by dividing (i) such amount
by (ii) the average price of our common stock over the five consecutive trading days preceding the date that is two trading days prior to the closing of the Proposed Fidelis Acquisition. The amount of Acquisition Consideration that Fidelis Care will
receive in cash (the “Cash Consideration”) is equal to (a) $3.75 billion minus (b) the Share Consideration Election Amount, subject to a working capital adjustment.
The amount of Cash Consideration that Fidelis Care will receive at the closing of the Proposed Fidelis Acquisition will be further reduced by,
among other things, a $375.0 million escrow which will be used to satisfy any of our post-closing indemnification claims (the “Escrow Fund”). No later than five business days prior to the closing of the Proposed Fidelis Acquisition,
we may elect the composition of the Escrow Fund, being any combination of cash and Escrow Shares up to $375.0 million. Any Escrow Shares included in the Escrow Fund will be liquidated prior to final settlement of the Escrow Fund’s proceeds
and will not be delivered to Fidelis Care. Fidelis Care will receive only cash proceeds from the Escrow Fund. To the extent that the value of the Escrow Fund exceeds $375.0 million at final settlement, we will be entitled to retain any such
surplus. Likewise, to the extent that the value of the Escrow Fund is less than $375.0 million at final settlement or the amount due to be delivered to Fidelis Care taking into account post-closing indemnification claims, as applicable, the
Company will be required to fund any such deficit.
For pro forma purposes only, it is assumed that the Acquisition Consideration will
consist solely of Cash Consideration and the Escrow Fund will consist solely of cash. The final value of the Acquisition Consideration for accounting purposes will ultimately be based on our election of Share Consideration and Escrow Shares.
Accordingly, the Acquisition Consideration and its related allocation to the underlying net assets of Fidelis Care could change materially.
We cannot assure you that we will be able to consummate the Proposed Fidelis Acquisition on a timely basis or at all. See “Risk
Factors—Factors that may affect Future Results and the Trading Price of Our Common Stock” in Item 1A. of Part I of our Annual Report on
Form 10-K
for the year ended December 31,
2017.
2. Basis of Presentation
The
unaudited pro forma condensed combined financial information was prepared using the acquisition method of accounting and was based on the historical financial statements of the Company and Fidelis Care. The acquisition method of accounting is based
on ASC 805 and uses the fair value concepts defined in ASC 820, Fair Value Measurements.
S-13
ASC 805 requires, among other things, that most assets acquired and liabilities assumed be
recognized at their fair values as of the acquisition date. In addition, ASC 805 requires that the consideration transferred be measured at the date the Proposed Fidelis Acquisition is completed at the then-current market price. This requirement
will likely result in a per share equity component that is different from the amount assumed in these unaudited pro forma condensed combined financial statements.
ASC 820 defines the term “fair value” and sets forth the valuation requirements for any asset or liability measured at fair value,
expands related disclosure requirements and specifies a hierarchy of valuation techniques based on the nature of the inputs used to develop the fair value measures. Fair value is defined in ASC 820 as “the price that would be received to sell
an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date.” This is an exit price concept for the valuation of the asset or liability. In addition, market participants are assumed to
be buyers and sellers in the principal (or the most advantageous) market for the asset or liability. Fair value measurements for an asset assume the highest and best use by these market participants. As a result of these standards, the Company may
be required to record the fair value of assets which are not intended to be used or sold and/or to value assets at fair value measures that do not reflect the Company’s intended use of those assets. Many of these fair value measurements can be
highly subjective, and it is possible that other professionals, applying reasonable judgment to the same facts and circumstances, could develop and support a range of alternative estimated amounts.
Under the acquisition method of accounting, the assets acquired and liabilities assumed will be recorded, as of completion of the Proposed
Fidelis Acquisition, primarily at their respective fair values and added to those of the Company. Financial statements and reported results of operations of the Company issued after completion of the Proposed Fidelis Acquisition will reflect these
values, but will not be retroactively restated to reflect the historical financial position or results of operations of Fidelis Care.
Under ASC 805, acquisition-related transaction costs (e.g., advisory, legal, accounting, valuation and other professional fees) are not
included as a component of consideration transferred but are accounted for as expenses in the periods in which the costs are incurred. Total acquisition-related transaction costs expected to be incurred by the Company are estimated to be
approximately $85 million, of which $7 million had been incurred as of September 30, 2017. Acquisition-related transaction costs expected to be incurred by the Company include estimated fees related to a bridge financing commitment
agreement and other advisory fees associated with the transaction. Those costs are reflected in the unaudited pro forma condensed combined balance sheet as an increase to accrued expenses and other current liabilities, with the related tax benefits
reflected as an increase in other current assets and the after tax impact presented as a decrease to retained earnings.
The unaudited pro
forma condensed combined financial statements do not reflect the projected realization of any anticipated cost savings following completion of the Proposed Fidelis Acquisition. These potential cost savings opportunities are expected to arise from
network and medical management savings, as well as administrative cost savings. The unaudited pro forma condensed combined financial statements do not reflect projected pretax restructuring and integration charges associated with the projected cost
savings, which are projected to be approximately $40 million to $50 million over a period of two years following completion of the Proposed Fidelis Acquisition. Such restructuring and integration charges will be expensed in the appropriate
accounting periods after completion of the Proposed Fidelis Acquisition.
3. Accounting Policies
At completion of the Proposed Fidelis Acquisition, the Company will review Fidelis Care’s accounting policies to determine if differences
in accounting policies require restatement or reclassification of results of operations or reclassification of assets or liabilities to conform to the Company’s accounting policies and classifications. As a result of that review, the Company
may identify differences between the accounting policies of the two companies that, when conformed, could have a material impact on the combined financial statements. At this time, the Company is not aware of any differences that would have a
material impact on the combined financial statements. The unaudited pro forma condensed combined financial statements assume there are no material differences in accounting policies.
S-14
4. Estimate of Consideration Expected to be Transferred
For pro forma purposes only, it is assumed that the Acquisition Consideration will consist solely of Cash Consideration and the Escrow Fund
will consist solely of cash. The final value of the Acquisition Consideration for accounting purposes will ultimately be based on our election of Share Consideration and Escrow Shares. Accordingly, the Acquisition Consideration and its related
allocation to the underlying net assets of Fidelis Care could change materially.
The following is a preliminary estimate of consideration
expected to be transferred to effect the acquisition of Fidelis Care:
|
|
|
|
|
|
|
|
|
Estimated
Fair Value
|
|
|
Consideration Transferred:
|
|
|
|
|
|
Cash
|
|
$
|
3,750
|
|
|
Centene Common Stock
|
|
|
—
|
|
|
|
|
|
|
|
|
Estimate of Total Consideration Expected to be Transferred
(a)
|
|
$
|
3,750
|
|
|
|
|
|
|
|
|
(a)
|
The estimated consideration expected to be transferred reflected in these unaudited pro forma condensed combined financial statements does not purport to represent the actual consideration that will be transferred when
the Proposed Fidelis Acquisition is completed. In accordance with ASC 805, the fair value of equity securities issued as part of the consideration transferred will be measured on the date the Proposed Fidelis Acquisition is completed at the
then-current market price.
|
5. Estimate of Assets to be Acquired and Liabilities to be Assumed
The following is a preliminary estimate of the assets to be acquired and the liabilities to be assumed by the Company in the Proposed Fidelis
Acquisition, reconciled to the estimate of total consideration expected to be transferred:
|
|
|
|
|
|
|
|
|
As of September 30, 2017
|
|
|
Assets Acquired and Liabilities Assumed:
|
|
|
|
|
|
Net book value of net assets acquired
|
|
$
|
1,413
|
|
|
Less historical goodwill
|
|
|
(16
|
)
|
|
|
|
|
|
|
|
Adjusted book value of net assets acquired
|
|
$
|
1,397
|
|
|
Goodwill
(a)
|
|
|
1,353
|
|
|
Identified intangible assets
(b)
|
|
|
1,000
|
|
|
Property and equipment
(c)
|
|
|
—
|
|
|
|
|
|
|
|
|
Consideration transferred
|
|
$
|
3,750
|
|
|
|
|
|
|
|
|
(a)
|
Goodwill is calculated as the difference between the acquisition date fair value of the total consideration expected to be transferred and the aggregate values assigned to the assets acquired and liabilities assumed.
Goodwill is not amortized.
|
|
(b)
|
As of completion of the Proposed Fidelis Acquisition, identifiable intangible assets are required to be measured at fair value, and these acquired assets could include assets that are not intended to be used or sold or
that are intended to be used in a manner other than their highest and best use. For purposes of these unaudited pro forma condensed combined financial statements and consistent with the ASC 820 requirements for fair value measurements, it is assumed
that all assets will be used, and that all assets will be used in a manner that represents the highest and best use of those assets, but it is not assumed that any market participant synergies will be achieved.
|
S-15
The fair value of identifiable intangible assets is determined primarily using variations of the
“income approach,” which is based on the present value of the future after tax cash flows attributable to each identified intangible asset. Other valuation methods, including the market approach and cost approach, are also considered in
estimating the fair value. Under the Hart-Scott-Rodino Antitrust Improvements Act and other relevant laws and regulations, there are significant limitations on the Company’s ability to obtain specific information about the Fidelis Care
intangible assets prior to completion of the Proposed Fidelis Acquisition.
At this time, the Company does not have sufficient information
as to the amount, timing and risk of cash flows of all of Fidelis Care’s identifiable intangible assets to determine their fair value. Some of the more significant assumptions inherent in the development of intangible asset values, from the
perspective of a market participant, include: the amount and timing of projected future cash flows (including revenue and profitability); the discount rate selected to measure the risks inherent in the future cash flows; and the assessment of the
asset’s life cycle and the competitive trends impacting the asset. However, for purposes of these unaudited pro forma condensed combined financial statements and using publicly available information, such as historical revenues, Fidelis
Care’s cost structure, industry information for comparable intangible assets and certain other high-level assumptions, the fair value of Fidelis Care’s identifiable intangible assets and their weighted-average useful lives have been
estimated as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated
Fair Value
|
|
|
Estimated
Useful Life
(Years)
|
|
|
State and Federal Contracts
|
|
$
|
800
|
|
|
|
|
|
|
Trademarks/trade names
|
|
|
120
|
|
|
|
|
|
|
Provider networks
|
|
|
80
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
1,000
|
|
|
|
13
|
|
|
|
|
|
|
|
|
|
|
|
These preliminary estimates of fair value and weighted-average useful life will likely be different from the
final acquisition accounting, and the difference could have a material impact on the accompanying unaudited pro forma condensed combined financial statements. Once the Company has full access to information about Fidelis Care’s intangible
assets, additional insight will be gained that could impact (i) the estimated total value assigned to intangible assets, (ii) the estimated allocation of value between finite-lived and indefinite-lived intangible assets and/or
(iii) the estimated useful lives of intangible assets. The estimated intangible asset values and their useful lives could be impacted by a variety of factors that may become known to the Company only upon access to additional information and/or
by changes in such factors that may occur prior to completion of the Proposed Fidelis Acquisition. These factors include, but are not limited to, changes in the regulatory, legislative, legal, technological and competitive environments. Increased
knowledge about these and/or other elements could result in a change to the estimated fair value of the identifiable Fidelis Care intangible assets and/or to the estimated weighted-average useful lives from what the Company has assumed in these
unaudited pro forma condensed combined financial statements. The combined effect of any such changes could then also result in a significant increase or decrease to the Company’s estimate of associated amortization expense.
|
(c)
|
As of completion of the Proposed Fidelis Acquisition, property and equipment is required to be measured at fair value, unless those assets are classified as
held-for-sale
on the acquisition date. The acquired assets can include assets that are not intended to be used or sold, or that are intended to be used in a manner other than their highest and best use. The
Company does not have sufficient information at this time as to the specific nature, age, condition or location of Fidelis Care’s property and equipment, and the Company does not know the appropriate valuation premise,
in-use
or
in-exchange,
as the valuation premise requires a certain level of knowledge about the assets being evaluated as well as a profile of the associated market
participants. All of these elements can cause differences between fair value and net book value. Accordingly, for the purposes of these unaudited pro forma condensed combined financial statements, the Company has assumed that the current Fidelis
Care book values represent the best estimate of fair value. This estimate is preliminary and subject to change and could vary materially from the actual value on the date the Proposed Fidelis Acquisition is completed.
|
S-16
6. Income Statement Pro Forma Adjustments
This note should be read in conjunction with Note 1.
Description of Transaction
; Note 2.
Basis of Presentation
; Note 4.
Estimate of Consideration Expected to be Transferred
; and Note 5.
Estimate of Assets to be Acquired and Liabilities to be Assumed
. Adjustments included in the column under the heading “Pro Forma Adjustments” represent
the following:
|
(a)
|
Prior to the Proposed Fidelis Acquisition, Fidelis Care was a
not-for-profit
entity and not subject to the New York Franchise tax, which is
a premium tax. In accordance with actuarial standards, the premium tax expense would be included in our premium pricing. The Company’s estimates of premium tax revenue and expense are as follows:
|
|
|
|
|
|
|
|
|
|
Nine Months Ended
September 30, 2017
|
|
|
Premium revenue
|
|
$
|
15
|
|
|
Premium tax revenue
|
|
|
122
|
|
|
Premium tax expense
|
|
|
137
|
|
|
(b)
|
To record intangible amortization expense, as follows:
|
|
|
|
|
|
|
|
|
|
Nine Months Ended
September 30, 2017
|
|
|
Estimated intangible asset amortization expense (*)
|
|
$
|
58
|
|
|
(*)
|
Assumes an estimated $1.0 billion of finite-lived intangibles and a weighted average amortization period of 13 years (Refer to Note 5.
Estimate of Assets to be Acquired and Liabilities to be Assumed
).
|
|
(c)
|
The Company estimates reduced investment income to reflect lower investment balances associated with the acquired assets and lower average investment yields associated with conforming investment policies. The estimated
reduction is as follows:
|
|
|
|
|
|
|
|
|
|
Nine Months Ended
September 30, 2017
|
|
|
Estimated investment income reduction
|
|
$
|
(15
|
)
|
|
(d)
|
The Company estimates interest expense associated with debt incurred to finance the Proposed Fidelis Acquisition:
|
|
|
•
|
|
Additional interest expense of approximately $59 million in the nine months ended September 30, 2017, based on approximately $1.5 billion of long term fixed-rate indebtedness the Company expects to incur
to finance a portion of the Cash Consideration payable in connection with the Proposed Fidelis Acquisition and to pay related fees and expenses. The calculation of interest expense on the long-term indebtedness assumes maturity tranches between
eight and 10 years and an estimated weighted average annual interest rate of 5.25%. If interest rates were to increase or decrease by 0.5% from the rates assumed in estimating this pro forma adjustment to interest expense, pro forma interest expense
could increase or decrease by $6 million in the nine months ended September 30, 2017.
|
|
|
•
|
|
The Company estimates additional interest expense of approximately $2 million for the nine months ended September 30, 2017, related to the amortization of debt issuance costs associated with the approximately
$1.5 billion of long-term indebtedness the Company expects to incur to finance a portion of the Cash Consideration payable in connection with the Proposed Fidelis Acquisition and to pay related fees and expenses. Incurrence costs related to
such long-term indebtedness are assumed to be amortized over an estimated weighted average term of approximately nine years.
|
S-17
|
(e)
|
The Company assumed a blended 37% tax rate when estimating the tax impact of the acquisition, representing the federal and state tax rates. The effective tax rate of the combined company could be significantly different
depending upon post-acquisition activities of the combined company.
|
|
(f)
|
The Company estimates increased basic and diluted shares outstanding of 22,766 thousand for the nine months ended September 30, 2017, related to equity issued to finance the Proposed Fidelis Acquisition, assuming
approximately $2.3 billion is issued at $101.03 per share (closing stock price on March 6, 2018) and no additional equity is issued to fund the purchase price, including as Share Consideration.
|
The shares calculated above and those used in the unaudited pro forma condensed combined statements included herein are based on the assumed
stock price of $101.03 per share. The table below depicts a sensitivity analysis assuming a $5.00 and $10.00 increase or decrease of the closing price of Centene stock and the resulting estimated shares to be issued at the adjusted stock price as
well as the corresponding pro forma diluted earnings per share.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Sensitivity Analysis
|
|
|
Assumed stock price (in dollars)
|
|
$
|
91.03
|
|
|
$
|
96.03
|
|
|
$
|
106.03
|
|
|
$
|
111.03
|
|
|
|
|
|
|
|
|
Corresponding number of shares to be issued (in thousands)
|
|
|
25,266
|
|
|
|
23,951
|
|
|
|
21,692
|
|
|
|
20,715
|
|
|
|
|
|
|
|
|
Corresponding diluted earnings per common share (in dollars):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Diluted earnings per common share
|
|
$
|
3.68
|
|
|
$
|
3.70
|
|
|
$
|
3.75
|
|
|
$
|
3.76
|
|
7. Balance Sheet Pro Forma Adjustments
This note should be read in conjunction with Note 1.
Description of Transaction
; Note 2.
Basis of Presentation
; Note
4.
Estimate of Consideration Expected to be Transferred
; and Note 5.
Estimate of Assets to be Acquired and Liabilities to be Assumed
. Adjustments included in the column under the heading “Pro Forma Adjustments” represent
the following:
|
(a)
|
To eliminate assets not acquired and liabilities not assumed, as follows:
|
|
|
|
|
|
|
|
Assets
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
(578
|
)
|
|
Short-term investments
|
|
|
(100
|
)
|
|
Property, software and equipment
|
|
|
(139
|
)
|
|
Liabilities
|
|
|
|
|
|
Accounts payable and accrued expenses
|
|
|
(2
|
)
|
|
Short-term debt
|
|
|
(14
|
)
|
|
Long-term debt
|
|
|
(75
|
)
|
|
|
|
|
|
|
|
Total, net
|
|
$
|
(726
|
)
|
|
|
|
|
|
|
S-18
|
(b)
|
To adjust goodwill to an estimate of acquisition-date goodwill, as follows:
|
|
|
|
|
|
|
|
Eliminate Fidelis Care’s historical goodwill
|
|
$
|
(16
|
)
|
|
Estimated transaction goodwill
|
|
|
1,353
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
1,337
|
|
|
|
|
|
|
|
|
(c)
|
To record intangible assets to an estimate of fair value of $1.0 billion and to record an employee benefit liability at an estimated fair value of $34 million.
|
|
(d)
|
The Company assumes approximately $1.5 billion of long-term indebtedness is issued to finance a portion of the Cash Consideration payable in connection with the Proposed Fidelis Acquisition and to pay related fees
and expenses and incur debt incurrence costs of $24 million.
|
|
(e)
|
The Company assumes approximately $2.3 billion of equity securities is issued to finance a portion of the Cash Consideration payable in connection with the Proposed Fidelis Acquisition and to pay related fees and
expenses and incur equity issuance costs of $66 million.
|
|
(f)
|
To record estimated acquisition-related transaction costs:
|
|
|
•
|
|
Total acquisition-related transaction costs estimated to be incurred by the Company are approximately $85 million and are recorded as an increase to accrued liabilities. These acquisition-related transaction costs
are not included in the pro forma condensed combined statements of operations.
|
|
|
•
|
|
Estimated transaction current tax asset for acquisition-related transaction costs of $31 million.
|
|
|
•
|
|
Retained earnings adjustment for the
after-tax
transaction costs incurred of $54 million.
|
|
(g)
|
To eliminate Fidelis Care’s historical net assets of $2.1 billion.
|
S-19
U.S. FEDERAL INCOME TAX CONSIDERATIONS TO
NON-U.S.
HOLDERS
The following is a summary of the U.S. federal income tax considerations generally
applicable to the ownership and disposition of our common stock acquired in this offering by a
Non-U.S.
Holder (as defined below) as of the date hereof. This summary is based on the provisions of the Internal
Revenue Code of 1986, as amended, which we refer to as the “Code,” applicable U.S. Treasury regulations, judicial opinions, and administrative rulings and published positions of the Internal Revenue Service (“IRS”), each as in
effect as of the date hereof. These authorities are subject to change, possibly on a retroactive basis, and any such change could affect the accuracy of the statements and conclusions set forth in this summary.
For purposes of this summary, a
“Non-U.S.
Holder” means a beneficial owner of our common
stock that is, for U.S. federal income tax purposes, neither a partnership (or an entity or arrangement treated as a partnership for U.S. federal income tax purposes) nor any of the following:
|
|
•
|
|
an individual who is a citizen or resident of the United States;
|
|
|
•
|
|
a corporation (or any other entity treated as a corporation for U.S. federal income tax purposes) created or organized under the laws of the United States, any state thereof or the District of Columbia;
|
|
|
•
|
|
an estate the income of which is subject to U.S. federal income taxation regardless of its source; or
|
|
|
•
|
|
a trust if it (1) is subject to the primary supervision of a court within the United States and one or more U.S. persons have the authority to control all substantial decisions of the trust or (2) has a valid
election in effect under applicable U.S. Treasury regulations to be treated as a U.S. person.
|
If a partnership (including
any entity or arrangement treated as a partnership for U.S. federal income tax purposes) holds our common stock, the tax treatment of a partner in such partnership will generally depend upon the status of the partner and the activities of the
partnership. A holder of our common stock that is a partnership and partners in such a partnership are urged to consult their tax advisors about the U.S. federal income tax considerations applicable to them in their particular circumstances.
This summary does not address all aspects of U.S. federal income taxation that may be relevant to particular
Non-U.S.
Holders in light of their individual circumstances, including
Non-U.S.
Holders that are subject to special rules, such as U.S. expatriates, banks or other
financial institutions, insurance companies, brokers or dealers in securities or currencies, traders that elect to
mark-to-market
their securities, controlled foreign
corporations, passive foreign investment companies,
Non-U.S.
Holders subject to the alternative minimum tax, or
Non-U.S.
Holders who hold shares of our common stock as
part of a straddle, hedge, conversion transaction or other integrated investment. Such
Non-U.S.
Holders should consult their tax advisors to determine the U.S. federal, state, local and
non-U.S.
tax considerations applicable to the ownership and disposition of our common stock that may be relevant to them.
This summary does not discuss other U.S. federal tax consequences (e.g., estate or gift tax), any state, local, or
non-U.S.
tax considerations or the Medicare tax imposed on certain net investment income. In addition, this summary applies only to
Non-U.S.
Holders that hold our common stock
as a capital asset within the meaning of the Code (generally, property held for investment purposes).
EACH
NON-U.S.
HOLDER IS URGED TO CONSULT ITS TAX ADVISORS REGARDING THE U.S. FEDERAL, STATE, LOCAL, AND
NON-U.S.
INCOME AND OTHER TAX CONSEQUENCES TO IT OF OWNING AND DISPOSING OF
OUR COMMON STOCK.
Distributions on Common Stock
Distributions with respect to our common stock will generally be treated as dividend income to the extent such distributions are paid from our
current or accumulated earnings and profits as determined for U.S. federal income tax purposes. If a distribution exceeds our current and accumulated earnings and profits, the excess will generally be treated first as a return of capital to the
extent of the
S-20
Non-U.S.
Holder’s adjusted tax basis in our common stock (and will reduce the
Non-U.S.
Holder’s basis in
such common stock) and thereafter as capital gain from the sale or exchange of such common stock, subject to the tax treatment described below in “—Sale, Exchange, or Other Taxable Disposition of Common Stock.” Generally, the gross
amount of dividends paid to
Non-U.S.
Holders will be subject to withholding of U.S. federal income tax at a rate of 30% or at a lower rate if an applicable income tax treaty so provides and we (or our agent)
have received proper certification as to the application of that treaty.
Dividends that are “effectively connected” with a
Non-U.S.
Holder’s conduct of a trade or business within the United States (and, if required by an applicable income tax treaty, are attributable to a U.S. permanent establishment of the
Non-U.S.
Holder) are generally subject to U.S. federal income tax on a net basis at regular graduated rates, in the same manner as if the
Non-U.S.
Holder were a United States
person (as defined in the Code), and are exempt from the 30% withholding tax described above, provided that certain certification requirements are satisfied. Any such effectively connected dividends received by a
Non-U.S.
Holder that is a corporation may also, under certain circumstances, be subject to an additional “branch profits tax” at a rate of 30% or such lower rate as may be specified by an applicable
income tax treaty.
To claim the benefits of an applicable income tax treaty or an exemption from withholding because the income is
effectively connected with the conduct of a trade or business in the United States, a
Non-U.S.
Holder will generally be required to provide a properly executed Internal Revenue Service (“IRS”) Form
W-8BEN
or
W-8BEN-E
(if the holder is claiming the benefits of an income tax treaty) or IRS Form
W-8ECI
(for income effectively connected with the conduct of a trade or business in the United States) or other suitable form. A
Non-U.S.
Holder eligible for a reduced
rate of withholding tax pursuant to an income tax treaty may obtain a refund of any excess amounts withheld by filing an appropriate claim for refund with the IRS.
Non-U.S.
Holders should consult their tax
advisors regarding their entitlement to benefits under an applicable income tax treaty and the specific manner of claiming the benefits of the treaty.
Sale, Exchange, or Other Taxable Disposition of Common Stock
Subject to the discussion below on backup withholding and Foreign Account Tax Compliance Act withholding, a Non-U.S. Holder will generally not
be subject to U.S. federal income or withholding tax with respect to gain realized on the sale, exchange, or other taxable disposition of our common stock, unless:
|
|
•
|
|
in the case of a
Non-U.S.
Holder that is a
non-resident
alien individual, such
Non-U.S.
Holder is
present in the United States for 183 days or more in the taxable year of the sale, exchange, or other taxable disposition, and certain other conditions are met;
|
|
|
•
|
|
the gain is “effectively connected” with such
Non-U.S.
Holder’s conduct of a trade or business within the United States (and, if required by an applicable treaty,
the gain is attributable to a U.S. permanent establishment of such
Non-U.S.
Holder); or
|
|
|
•
|
|
we are or have been a “United States real property holding corporation” for U.S. federal income tax purposes at any time within the shorter of the five-year period ending on the date of such sale, exchange, or
other taxable disposition or the period that such
Non-U.S.
Holder held such common stock and either (a) our common stock was not treated as regularly traded on an established securities market during a
relevant period in the calendar year in which the sale, exchange, or other taxable disposition occurs, or (b) such
Non-U.S.
Holder owns or owned (actually or constructively) more than 5% of our common
stock at any time during the shorter of the two periods described above.
|
We believe that we have not been and are not, and
we do not anticipate becoming, a “United States real property holding corporation” for U.S. federal income tax purposes.
If
gain realized on the sale, exchange, or other taxable disposition of our common stock is effectively connected with the
Non-U.S.
Holder’s conduct of a trade or business within the United States (and, if
required by an applicable tax treaty, is attributable to a U.S. permanent establishment of such
Non-U.S.
Holder), such
Non-U.S.
Holder will generally be subject to U.S.
federal income tax
S-21
on the net gain from the disposition of our common stock at regular graduated rates in the same manner as if such
Non-U.S.
Holder were a United States
person as defined in the Code. In the case of a
Non-U.S.
Holder that is a foreign corporation, such gain may also be subject to an additional branch profits tax rate of 30% (or a lower applicable treaty rate).
If a
Non-U.S.
Holder is an individual that is present in the United States for 183 days or more in the taxable year of disposition and certain other conditions are met, the
Non-U.S.
Holder will generally be subject to U.S. federal income tax at a rate of 30% (unless otherwise provided by an applicable treaty) on any capital gain recognized on the disposition of our common stock,
which may be offset by certain U.S.-source capital losses.
Foreign Account Tax Compliance Act
Withholding at a rate of 30% will generally be required in certain circumstances on dividends in respect of, and, after December 31, 2018,
gross proceeds from the sale or other disposition of, shares of our common stock held by or through certain financial institutions (including investment funds), unless such institution (i) enters into, and complies with, an agreement with the
IRS to report, on an annual basis, information with respect to interests in, and accounts maintained by, the institution that are owned by certain U.S. persons and by certain
non-U.S.
entities that are wholly
or partially owned by U.S. persons and to withhold on certain payments, (ii) if required under an intergovernmental agreement between the United States and an applicable foreign country, reports such information to its local tax authority,
which will exchange such information with the U.S. authorities or (iii) qualifies for an exemption. An intergovernmental agreement between the United States and an applicable foreign country may modify these requirements. Accordingly, the
entity through which our common stock is held will affect the determination of whether such withholding is required. Similarly, dividends in respect of, and, after December 31, 2018, gross proceeds from the sale or other disposition of, our
common stock held by a
Non-U.S.
Holder that is a
non-financial
non-U.S.
entity that does not qualify under certain exceptions
generally will be subject to withholding at a rate of 30%, unless such entity either (x) certifies to the applicable withholding agent that such entity does not have any “substantial United States owners” or (y) provides certain
information regarding the entity’s “substantial United States owners,” which information the applicable withholding agent will be required in turn to provide to the U.S. Department of Treasury. Prospective investors should consult
their tax advisors regarding the possible implications of these rules on their investment in our common stock.
Information reporting and backup
withholding
Non-U.S.
Holders will generally be required to comply with certain certification
procedures to establish that they are not U.S. persons in order to avoid backup withholding with respect to dividends paid to such
Non-U.S.
Holders on our common stock or, depending on the circumstances, the
proceeds received by such
Non-U.S.
Holders on the sale, exchange, or other taxable disposition of our common stock. In addition, we are required to report annually to the IRS and to each
Non-U.S.
Holder the amount of any dividends paid to such
Non-U.S.
Holder, regardless of whether any tax was actually withheld. Copies of the information returns reporting such
dividends and the amount withheld may also be made available to the tax authorities in the country in which the
Non-U.S.
Holder resides under the provisions of an applicable income tax treaty.
Non-U.S.
Holders will generally be required to comply with certain certification procedures to establish that they are not U.S. persons in order to avoid information reporting with respect to proceeds received by
such
Non-U.S.
Holders on the sale, exchange, or other taxable disposition of our common stock within the United States or conducted through certain U.S.-related financial intermediaries.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules from a payment to a
Non-U.S.
Holder will generally be allowed as a credit against such
Non-U.S.
Holder’s U.S. federal income tax liability, which may entitle the
Non-U.S.
Holder to a refund, provided that the
Non-U.S.
Holder timely provides certain required information to the IRS.
S-22
PLAN OF DISTRIBUTION
The selling stockholder may, from time to time, offer and sell shares of common stock covered by this prospectus supplement and the
accompanying prospectus. The common stock may be sold at prices and on terms then prevailing in the market, at prices related to the then-current market price of the common stock, at a fixed price or prices, which may be changed, or at negotiated
prices, by a variety of methods including the following:
|
|
•
|
|
through the NYSE or on any other national securities exchange or quotation service on which the securities may be listed or quoted at the time of the sale;
|
|
|
•
|
|
in transactions otherwise than on these exchanges or systems or in the
over-the-counter
market;
|
|
|
•
|
|
purchases by a broker-dealer as principal and resale by that broker-dealer for its own account;
|
|
|
•
|
|
ordinary brokerage transactions and transactions in which the broker solicits purchasers;
|
|
|
•
|
|
“block” sale transactions in which the broker-dealer will attempt to sell the shares as agent but may position and resell a portion of the block as principal to facilitate the transaction;
|
|
|
•
|
|
privately negotiated transactions;
|
|
|
•
|
|
a combination of any such methods of sale; and
|
|
|
•
|
|
any other method permitted pursuant to applicable law.
|
In connection with the distributions
of the shares of common stock or otherwise, the selling stockholder may:
|
|
•
|
|
enter into hedging transactions with broker-dealers or other financial institutions, which may in turn engage in short sales of the shares in the course of hedging the positions they assume;
|
|
|
•
|
|
sell the shares short and deliver the shares to close out such short positions; or
|
|
|
•
|
|
enter into option or other transactions with broker-dealers or other financial institutions which require the delivery to them of shares offered by this prospectus and any applicable prospectus supplement or
supplements, which they may in turn resell.
|
The selling stockholder may, from time to time, pledge or grant a security
interest in some or all of the shares it owns and, if the selling stockholder defaults in the performance of its secured obligations, the pledgees or secured parties may offer and sell the shares, from time to time, under this prospectus supplement,
or under an amendment to this prospectus supplement under Rule 424(b)(3) or other applicable provision of the Securities Act amending the list of selling stockholder to include any donee, pledgee, transferee or other
successors-in-interest
as a selling stockholder under this prospectus supplement. The selling stockholder also may transfer the shares in other circumstances, in which
case the donees, transferees, pledgees or other
successors-in-interest
will be the selling beneficial owners for purposes of this prospectus supplement.
In addition, the selling stockholder may sell shares of common stock under any available exemption to the registration requirements of the
Securities Act, including under the safe harbor provisions of Rule 144 under the Securities Act, if such transactions meet the criteria and conform to the requirements of that rule, rather than pursuant to this prospectus supplement and the
accompanying prospectus. In effecting sales, broker-dealer or agents engaged by the selling stockholder may arrange for other broker-dealers to participate. Broker-dealers or agents may receive commissions, discounts or concessions from the selling
stockholder, in amounts to be negotiated immediately prior to the sale.
S-23
In offering the shares of common stock covered by this prospectus supplement and the accompanying
prospectus, the selling stockholder, and any broker-dealers and any other participating broker-dealers who execute sales for the selling stockholder, may be deemed to be “underwriters” within the meaning of the Securities Act in connection
with these sales. Any profits realized by the selling stockholder and the compensation of such broker-dealers may be deemed to be underwriting discounts and commissions under the Securities Act and the rules of the Financial Industry Regulatory
Authority, Inc. To the extent the selling stockholder or any such broker-dealer may be deemed to be an underwriter, the selling stockholder and such broker-dealer will be subject to the prospectus delivery requirements of the Securities Act and may
be subject to certain statutory liabilities of, including but not limited to, Sections 11, 12 and 17 of the Securities Act and Rule
10b-5
under the Exchange Act. In no event shall any broker-dealer receive
fees, commissions and markups which, in the aggregate, would exceed eight percent (8%) of the gross proceeds of any offering.
We have
advised the selling stockholder that the anti-manipulation rules of Regulation M under the Exchange Act may apply to sales of shares in the market and to the activities of the selling stockholder and their respective affiliates. In addition, we will
make copies of this prospectus supplement and accompanying prospectus available to the selling stockholder for the purpose of satisfying the prospectus delivery requirements of the Securities Act. The selling stockholder may indemnify any
broker-dealer that participates in transactions involving the sale of the shares of common stock against certain liabilities, including liabilities arising under the Securities Act.
We have agreed to indemnify the selling stockholder against certain liabilities, including certain liabilities under the Securities Act.
There can be no assurance that the selling stockholder will sell any or all of the shares of common stock registered pursuant to the
registration statement, of which this prospectus supplement and the accompanying prospectus forms a part.
We have agreed to keep the
registration statement of which this prospectus supplement is a part effective until such time that all shares covered by the registration statement cease to constitute registrable securities (as defined in the Registration Rights Agreement by and
between Centene Corporation and CMG Holding, dated February 21, 2018). We are required to pay all fees and expenses incurred in connection with the registration of the common stock, including all registration and filing fees and expenses;
provided, however, we will not be required to pay the expense of any brokers’ commissions, underwriters’ discount or commission, any transfer taxes relating to the registration and sale of any shares and all counsels’ fees and
expenses incurred by the selling stockholder. We will not receive any of the proceeds from the sale of the common stock offered pursuant to this prospectus supplement and the accompanying prospectus.
S-24
LEGAL MATTERS
The validity of the shares of common stock offered hereby will be passed upon by Skadden, Arps, Slate, Meagher & Flom LLP, New York,
New York.
EXPERTS
The consolidated financial statements of Centene Corporation as of December 31, 2017 and 2016, and for each of the years in the
three-year period ended December 31, 2017, and management’s assessment of the effectiveness of internal control over financial reporting as of December 31, 2017 have been incorporated by reference herein in reliance upon the reports
of KPMG LLP, an independent registered public accounting firm, incorporated by reference herein, and upon the authority of said firm as experts in accounting and auditing.
The consolidated financial statements of New York State Catholic Health Plan, Inc. (d/b/a Fidelis Care New York) and subsidiaries as of
December 31, 2016 and 2015, and for each of the two years in the period ended December 31, 2016, incorporated in this prospectus supplement by reference from the Current Report on Form
8-K
dated
March 13, 2018, have been audited by Deloitte & Touche LLP, independent auditors, as stated in their report, which is incorporated by reference herein, and are incorporated in reliance upon the report of such firm given upon their
authority as experts in accounting and auditing.
The consolidated financial statements of Health Net, Inc. and subsidiaries, incorporated
in this prospectus supplement by reference from the Health Net Form
8-K,
have been audited by Deloitte & Touche LLP, an independent registered public accounting firm, as stated in their report, which
is incorporated herein by reference from the Health Net Form
8-K.
Such consolidated financial statements have been so incorporated in reliance upon the report of such firm given upon their authority as experts
in accounting and auditing.
S-25
PROSPECTUS

Debt Securities
Common Stock
Preferred
Stock
Depositary Shares
Warrants
We may offer and sell from time
to time, one or any combination of the securities we describe in this prospectus. In addition, certain selling securityholders may offer and sell our securities from time to time, together or separately, in amounts, at prices and on terms that will
be determined at the time of any such offering. We or any selling securityholders will provide specific terms of any offering in supplements to this prospectus. The prospectus supplement will contain more specific information about the offering and
the securities being offered, including the names of any selling securityholders, if applicable. The supplements may add, update or change information contained in this prospectus. You should read this prospectus and any prospectus supplement
carefully before you invest. This prospectus may not be used to sell securities unless accompanied by a prospectus supplement.
We and/or any selling
securityholders, if applicable, may offer securities for sale directly to purchasers or through underwriters, dealers or agents to be designated at a future date. The supplements to this prospectus will provide the names of any underwriters, the
specific terms of the plan of distribution and the underwriter’s discounts and commissions.
Our common stock is listed on the New York Stock
Exchange (the “NYSE”) under the symbol “CNC.” Any common stock sold pursuant to a prospectus supplement will be listed, subject to notice of issuance, on the New York Stock Exchange. If we decide to list or seek a quotation for
any other securities, the prospectus supplement relating to those securities will disclose the exchange or market on which those securities will be listed or quoted.
Investing in our securities
involves risks. See “
Risk Factors
” beginning on page 2 of this prospectus.
Neither the Securities and Exchange
Commission nor any state securities commission has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is May 3, 2017
TABLE OF CONTENTS
You should rely only on information provided or incorporated by reference in this prospectus or any applicable prospectus
supplement. No dealer, salesperson or other person is authorized to give you any information or to represent anything not contained in this prospectus. You must not rely on any unauthorized information or representations. This prospectus is an offer
to sell only the debt securities offered hereby, but only under circumstances and in jurisdictions where it is lawful to do so. The information contained in this prospectus is current only as of its date.
ABOUT THIS PROSPECTUS
This prospectus is part of an automatic “shelf” registration statement on Form
S-3
that we filed with the
Securities and Exchange Commission, or SEC, as a “well-known seasoned issuer” as defined in Rule 405 under the Securities Act of 1933, as amended (the “Securities Act”). Under this shelf registration process, we and/or certain
selling securityholders, if applicable, may, from time to time, sell the securities described in this prospectus in one or more offerings. For further information about our business and the securities, you should refer to the registration statement
and its exhibits. The exhibits to our registration statement contain the full text of certain contracts and other important documents we have summarized in this prospectus. Since these summaries may not contain all the information that you may find
important in deciding whether to purchase the securities we and/or any selling securityholders offer, you should review the full text of these documents. The registration statement and the exhibits can be obtained from the SEC as indicated under the
heading “Where You Can Find More Information.”
This prospectus provides you with a general description of our securities. Each time we and/or
any selling securityholders offer securities, we will provide you with a prospectus supplement that will contain specific information about the terms of that offering, including the names of any selling securityholders, if applicable. When we refer
to a “prospectus supplement,” we are also referring to any free writing prospectus or other offering material authorized by us. The prospectus supplement may also add, update or change information contained in this prospectus. If there is
any inconsistency between the information in this prospectus and the applicable prospectus supplement, you should rely on the information in the prospectus supplement. You should read this prospectus and any prospectus supplement together with
additional information described under the heading “Where You Can Find More Information.”
You should rely only on the information provided in
this prospectus or in any prospectus supplement, including the information incorporated by reference. We and any selling securityholders have not authorized anyone to provide you with different information. You should not assume that the information
in this prospectus or any supplement to this prospectus, is accurate at any date other than the date indicated on the cover page of these documents or the date of the statement contained in any incorporated documents, respectively. This prospectus
is not an offer to sell or a solicitation of an offer to buy any securities other than the securities referred to in the prospectus supplement. This prospectus is not an offer to sell or a solicitation of an offer to buy such securities in any
circumstances in which such offer or solicitation is unlawful. You should not interpret the delivery of this prospectus, or any sale of securities, as an indication that there has been no change in our affairs since the date of this prospectus. You
should also be aware that information in this prospectus may change after this date. Unless the context otherwise requires, in this prospectus “Centene,” “we,” “us,” “our” and “ours” refer to Centene
Corporation and its consolidated subsidiaries.
1
RISK FACTORS
Investing in our securities involves risks. You should carefully consider the risks described under “Risk Factors” in Item 1A of Part II of our
Quarterly Report on Form
10-Q
for the period ended March 31, 2017, filed with the SEC on April 25, 2017, and under “Risk Factors” in Item 1A of Part I of our Annual Report on Form
10-K
for the year ended December 31, 2016, filed with the SEC on February 21, 2017, and in the other documents incorporated by reference into this prospectus (which risk factors are incorporated by
reference herein), as well as the additional risk factors and other information contained or incorporated by reference in this prospectus or in any prospectus supplement hereto before making a decision to invest in our securities. See “Where
You Can Find More Information.”
2
CENTENE CORPORATION
Centene is a diversified, multi-national healthcare enterprise that provides services to government sponsored and commercial healthcare programs, focusing on
under-insured and uninsured individuals. Centene provides member-focused services through locally based staff by assisting in accessing care, coordinating referrals to related health and social services and addressing member concerns and questions.
Centene also provides education and outreach programs to inform and assist members in accessing quality, appropriate healthcare services. Centene believes its local approach, including member and provider services, enables it to provide accessible,
quality, culturally-sensitive healthcare coverage to its communities. Centene’s health management, educational and other initiatives are designed to help members best utilize the healthcare system to ensure they receive appropriate, medically
necessary services and effective management of routine, severe and chronic health problems, resulting in better health outcomes. Centene combines its decentralized local approach for care with a centralized infrastructure of support functions such
as finance, information systems and claims processing.
On March 24, 2016, Centene acquired all of the issued and outstanding shares of Health Net,
Inc. (Health Net), a publicly traded managed care organization that delivers healthcare services through health plans and government-sponsored managed care plans. The transaction was valued at $6.0 billion, including the assumption of debt. The
acquisition allows the Company to offer a more comprehensive and scalable portfolio of solutions and provides opportunity for additional growth across the combined company’s markets.
Centene operates in two segments: Managed Care and Specialty Services. Centene’s Managed Care segment provides health plan coverage to individuals
through government subsidized programs, including Medicaid, that also encompasses the State Children’s Health Insurance Program (CHIP), Long Term Care (LTC), Foster Care, dual-eligible individuals (Duals) and the Supplemental Security Income
Program, also known as the Aged, Blind or Disabled Program (ABD), and Medicare. The Managed Care segment also includes commercial health care products offered to individuals and large and small employer groups, as well as products to individuals
through the Health Insurance Marketplace (HIM). Centene’s Specialty Services segment consists of its specialty companies offering diversified healthcare services and products to federal and state programs, correctional facilities, healthcare
organizations, employer groups and other commercial organizations, as well as to its own subsidiaries. For the year ended December 31, 2016, Centene’s Managed Care and Specialty Services segments accounted for 94% and 6% respectively, of
its total revenues.
Centene’s managed care membership totaled approximately 11.4 million as of December 31, 2016. For the year ended
December 31, 2016, Centene’s total revenues and net earnings from continuing operations attributable to Centene were $40.6 billion and $559 million, respectively, and its total cash flow from operations was $1.9 billion.
Centene’s initial health plan commenced operations in Wisconsin in 1984. The Company was organized in Wisconsin in 1993 as a holding company for its
initial health plan and reincorporated in Delaware in 2001.
The principal executive offices are located at 7700 Forsyth Boulevard, St. Louis, Missouri
63105, and the telephone number is (314)
725-4477.
Centene’s website address is www.centene.com. We do not incorporate the information contained on our website into this Prospectus, and you should not
consider it part of this Prospectus.
Our common stock is publicly traded on the NYSE under the ticker symbol “CNC.”
3
WHERE YOU CAN FIND MORE INFORMATION
Because we are subject to the informational requirements of the Exchange Act, we file reports, proxy statements and other information with the SEC. You may
read and copy the registration statement, including the attached exhibits, and any reports, proxy statements or other information that we file at the SEC’s public reference room in Washington, D.C. at Room 1580, 100 F Street, N.E., 20549. You
can request copies of these documents by writing to the SEC and paying a duplicating charge. Please call the SEC at
1-800-SEC-0330
for further information on the operation of its public reference rooms in other cities. The SEC makes our filings
available to the public on its Internet site (http://www.sec.gov). In addition, you may inspect such reports and other information at the offices of the New York Stock Exchange, 11 Wall Street, New York, New York 10005.
In addition, we make available free of charge through our Internet site (http://www.centene.com) reports we file with the SEC, including our Annual Reports on
Form
10-K,
Quarterly Reports on Form
10-Q,
Current Reports on Form
8-K
and amendments to those reports filed or furnished
pursuant to Section 13(a) or 15(d) of the Exchange Act, as soon as reasonably practical after we electronically file such material with, or furnish it to, the SEC.
The SEC allows us to “incorporate by reference” information into this prospectus, which means we can disclose important information to you by
referring you to other documents that we filed separately with the SEC. You should consider the incorporated information as if we reproduced it in this prospectus, except for any information directly superseded by information subsequently filed with
the SEC and incorporated in this prospectus.
We incorporate by reference into this prospectus the following documents (SEC File
No. 001-31826),
which contain important information about us and our business and financial results:
|
|
•
|
|
our Annual Report on Form
10-K
for the year ended December 31, 2016, filed with the SEC on February 21, 2017;
|
|
|
•
|
|
our Quarterly Report on Form
10-Q
for the quarterly period ended March 31, 2017 filed with the SEC on April 25, 2017;
|
|
|
•
|
|
our Current Reports on Form
8-K
filed with the SEC on March 24, 2016 (as amended on May 10, 2016 and as further amended on June 9, 2016), March 2, 2017 and
April 27, 2017; and
|
|
|
•
|
|
the description of our common stock contained in our registration statement on Form
8-A
filed with the SEC on October 14, 2003, as amended by our Forms
8-A/A
filed with the SEC on December 17, 2004 and April 26, 2007, including any amendments or reports filed for the purpose of updating such description.
|
We may file additional documents with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act on or after the date of this prospectus and
before the expiration of the offering. The SEC allows us to incorporate by reference into this prospectus such documents. Unless otherwise stated, information that is furnished and not filed (for example, information under Item 2.02 of our Current
Reports on Form
8-K)
is not incorporated by reference into this prospectus. You should consider any statement contained in this prospectus (or in a document incorporated into this prospectus) or any prospectus
supplement to be modified or superseded to the extent that a statement in a subsequently filed document modifies or supersedes such statement.
We
encourage you to read our SEC reports, as they provide additional information about us which prudent investors find important. We will provide to each person, including any beneficial owner, to whom a prospectus is delivered, a copy of any or all of
the information that has been incorporated by reference in the prospectus but not delivered with the prospectus at no charge upon request by contacting us at Centene Corporation, Attn: Corporate Secretary, 7700 Forsyth Boulevard, St. Louis, Missouri
63105, telephone (314)
725-4477.
USE OF PROCEEDS
Unless we specify another use in the applicable prospectus supplement, we will use the net proceeds from the sale of any securities offered by us for general
corporate purposes. Such general corporate purposes may include the repayment of indebtedness, funding for acquisitions, capital expenditures, additions to working capital and to meet statutory capital requirements in new or existing states. Pending
such use, the proceeds may be invested temporarily in short-term, interest-bearing, investment-grade securities or similar assets. Unless we specify another use in the applicable prospectus supplement, we will not receive any of the proceeds from a
sale of securities by any selling securityholders.
4
CAUTIONARY STATEMENT ON FORWARD-LOOKING STATEMENTS
Statements set forth in this prospectus and incorporated by reference from documents we have filed with the Securities and Exchange Commission may contain
forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). In particular, these statements
include without limitation statements about our market opportunity, our growth strategy, competition, expected activities and future acquisitions, investments and the adequacy of our available cash resources. Readers are cautioned that matters
subject to forward-looking statements involve known and unknown risks and uncertainties, including economic, regulatory, competitive and other factors that may cause our or our industry’s actual results, levels of activity, performance or
achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. These statements are not guarantees of future performance and are subject to
risks, uncertainties and assumptions.
All forward-looking statements included in this filing are based on information available to us on the date of this
filing. Except as may be otherwise required by law, we undertake no obligation to update or revise the forward-looking statements included in this filing, whether as a result of new information, future events or otherwise, after the date of this
filing. You should not place undue reliance on any forward looking statements, as actual results may differ materially from projections, estimates, or other forward-looking statements due to a variety of important factors, including but not limited
to:
|
|
•
|
|
our ability to accurately predict and effectively manage health benefits and other operating expenses and reserves;
|
|
|
•
|
|
membership and revenue declines or unexpected trends;
|
|
|
•
|
|
changes in healthcare practices, new technologies, and advances in medicine;
|
|
|
•
|
|
increased health care costs;
|
|
|
•
|
|
changes in economic, political or market conditions;
|
|
|
•
|
|
changes in federal or state laws or regulations, including changes with respect to government health care programs as well as changes with respect to the Patient Protection and Affordable Care Act and the Health Care
and Education Affordability Reconciliation Act and any regulations enacted thereunder that may result from changing political conditions;
|
|
|
•
|
|
rate cuts or other payment reductions or delays by governmental payors and other risks and uncertainties affecting our government businesses;
|
|
|
•
|
|
our ability to adequately price products on federally facilitated and state based Health Insurance Marketplaces;
|
|
|
•
|
|
disasters or major epidemics;
|
|
|
•
|
|
the outcome of legal and regulatory proceedings;
|
|
|
•
|
|
changes in expected contract start dates;
|
|
|
•
|
|
provider, state, federal and other contract changes and timing of regulatory approval of contracts;
|
|
|
•
|
|
the expiration, suspension, or termination of our contracts with federal or state governments (including but not limited to Medicaid, Medicare, and TRICARE);
|
|
|
•
|
|
challenges to our contract awards;
|
|
|
•
|
|
cyber-attacks or other privacy or data security incidents;
|
|
|
•
|
|
the possibility that the expected synergies and value creation from acquired businesses, including, without limitation, the acquisition of Health Net, will not be realized, or will not be realized within the expected
time period, including, but not limited to, as a result of conditions, terms, obligations or restrictions imposed by regulators in connection with their approval of, or consent to, the acquisition;
|
|
|
•
|
|
the exertion of management’s time and our resources, and other expenses incurred and business changes required in connection with complying with the undertakings in connection with certain regulatory approvals;
|
|
|
•
|
|
disruption from the acquisition making it more difficult to maintain business and operational relationships;
|
|
|
•
|
|
the risk that unexpected costs will be incurred in connection with, among other things, the acquisition and/or the integration;
|
|
|
•
|
|
changes in expected closing dates, estimated purchase price and accretion for acquisitions;
|
|
|
•
|
|
the risk that acquired businesses will not be integrated successfully;
|
|
|
•
|
|
our ability to maintain or achieve improvement in the Centers for Medicare and Medicaid Services (CMS) Star ratings and other quality scores that impact revenue;
|
|
|
•
|
|
availability of debt and equity financing, on terms that are favorable to us;
|
|
|
•
|
|
foreign currency fluctuations.
|
5
This list of important factors is not intended to be exhaustive. We discuss certain of these matters more fully,
as well as certain other risk factors that may affect our business operations, financial condition and results of operations, in our filings with the SEC, including our annual reports on Form
10-K,
quarterly
reports on Form
10-Q
and current reports on Form
8-K.
All statements,
other than statements of current or historical fact, contained in this filing are forward-looking statements. We have attempted to identify these statements by terminology including “believe,” “anticipate,” “plan,”
“expect,” “estimate,” “intend,” “seek,” “target,” “goal,” “may,” “will,” “would,” “could,” “should,” “can,” “continue”
and other similar words or expressions in connection with, among other things, any discussion of future operating or financial performance. In particular, these statements include statements about our market opportunity, our growth strategy,
competition, expected activities and future acquisitions, investments and the adequacy of our available cash resources.
Because such statements are
subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward looking statements. See “Risk Factors” beginning on page 2 of this prospectus for reference to the factors that could
cause actual results to differ materially.
You should not place undue reliance on such statements, which speak only as of the date that they were made.
These cautionary statements should be considered in connection with any written or oral forward looking statements that we may issue in the future. We do not undertake any obligation to release publicly any revisions to such forward looking
statements to reflect later events or circumstances or to reflect the occurrence of unanticipated events.
6
DESCRIPTION OF DEBT SECURITIES
The following description of the terms of our debt securities sets forth general terms that may apply to the debt securities. The particular terms of any debt
securities will be described in the prospectus supplement relating to those debt securities. For purposes of this description, the terms “we,” “our,” “ours,” and “us” refer only to Centene Corporation and not
to any of its subsidiaries.
The Indentures
The debt
securities will be issued in one or more series under an indenture (the “Indenture” or an “indenture”), to be entered into between us and The Bank of New York Mellon Trust Company, N.A., as trustee or under the Indenture, dated
April 29, 2014, among the Company and The Bank of New York Mellon Trust Company, N.A., relating to the Company’s 4.75% Senior Notes due 2022, the Indenture, dated February 11, 2016, among the Company (as successor to Centene Escrow
Corporation) and The Bank of New York Mellon Trust Company, N.A., relating to the Company’s 5.625% Senior Notes due 2021, the Indenture, dated February 11, 2016, among the Company (as successor to Centene Escrow Corporation) and The Bank
of New York Mellon Trust Company, N.A., relating to the Company’s 6.125% Senior Notes due 2024, and the Indenture, dated November 9, 2016, among the Company and The Bank of New York Mellon Trust Company, N.A., relating to the
Company’s 4.75% Senior Notes due 2025. The statements herein relating to the debt securities and the indentures are summaries and are subject to the detailed provisions of the applicable indenture. The indenture will be subject to and governed
by the Trust Indenture Act of 1939 (the “Trust Indenture Act”). The description below is a summary and does not contain all the information you may find useful. We urge you to read the indenture because it, and not this summary, defines
many of your rights as a holder of our debt securities. The form of the indenture has been filed as an exhibit to the registration statement of which this prospectus is a part. Whenever we refer to particular sections or defined terms in an
indenture, those sections and definitions are incorporated by reference.
General
The debt securities will be our general obligations. The indenture does not limit the aggregate amount of debt securities which we may issue nor does it limit
other debt we may issue. We may issue senior or subordinated debt securities under the indenture up to the aggregate principal amount authorized by our board of directors from time to time. Except as may be described in a prospectus supplement, the
indenture will not limit the amount of other secured or unsecured debt that we may incur or issue.
The senior debt securities will rank equally with all
our other unsubordinated obligations. Unless otherwise specified in the applicable prospectus supplement, the subordinated debt securities will be subordinated and junior in right of payment to all our present and future senior indebtedness to the
extent and in the manner set forth in the indenture. See “-Subordinated Debt Securities” below. The indenture will provide that the debt securities may be issued from time to time in one or more series.
The indenture will place no limitation on the amount of additional secured indebtedness that we may incur. We expect from time to time to incur additional
indebtedness constituting secured indebtedness. Our outstanding secured indebtedness would rank senior to our senior unsecured indebtedness to the extent of such security, and our outstanding short- and long-term indebtedness would rank equally with
our senior unsecured debt securities.
If this prospectus is being delivered in connection with the offering of a series of senior debt securities, the
accompanying prospectus supplement or information incorporated by reference will set forth the approximate amount of secured long-term indebtedness senior to such senior unsecured indebtedness outstanding as of a recent date.
The applicable prospectus supplement relating to the particular series of debt securities will describe specific terms of the debt securities offered thereby,
including, where applicable:
|
|
•
|
|
the title and any limit on the aggregate principal amount of the debt securities and whether the debt securities will be senior or subordinated;
|
|
|
•
|
|
the price at which we are offering the debt securities, usually expressed as a percentage of the principal amount;
|
|
|
•
|
|
the date or dates on which the debt securities of a series will be issued, and on which the principal of and any premium on such debt securities, or any installments thereof, will mature or the method of determining
such date or dates;
|
|
|
•
|
|
the rate or rates, which may be fixed or variable at which such debt securities will bear interest or the method of calculating such rate or rates, if any;
|
|
|
•
|
|
the date or dates from which any interest will accrue or the method of determining such dates;
|
|
|
•
|
|
the date or dates on which any interest will be payable and the applicable record dates;
|
|
|
•
|
|
the place or places where principal of, premium, if any, and interest, if any, on such debt securities, or installments thereof, if any, will be payable;
|
7
|
|
•
|
|
any of our obligations to redeem, repay, purchase or offer to purchase the debt securities pursuant to any mandatory redemption, sinking fund or analogous provisions or upon other conditions or at the option of the
holders of the debt securities and the periods, prices and the other terms and conditions of such redemption or repurchase, in whole or in part;
|
|
|
•
|
|
any of our rights to redeem the debt securities at our option and the periods, prices and the other terms and conditions of such redemption, in whole or in part;
|
|
|
•
|
|
if denominations other than $1,000 and any integral multiple thereof in the case of debt securities in registered form, or $1,000 and $5,000 in the case of debt securities in bearer form, the denominations in which such
debt securities will be issued;
|
|
|
•
|
|
whether the debt securities are original issue discount securities (as described below under “-Original Issue Discount Securities”) and the amount of discount;
|
|
|
•
|
|
the provisions for payment of additional amounts or tax redemptions, if any;
|
|
|
•
|
|
any addition to, or modification or deletion of, any event of default or covenant specified in the indenture with respect to such debt securities;
|
|
|
•
|
|
whether the debt securities of the series shall be issued in whole or in part in certified form;
|
|
|
•
|
|
the designation, if any, of any depositaries, trustees, paying agents, authenticating agents, security registrars or other agents with respect to the debt securities of such series;
|
|
|
•
|
|
if other than the entire principal amount, the portion of the principal amount of debt securities which becomes payable upon a declaration of acceleration of maturity or the method of determining such portion;
|
|
|
•
|
|
in the case of the subordinated debt securities, the subordination provisions pertaining to such debt securities;
|
|
|
•
|
|
material federal income tax considerations, if applicable; and
|
|
|
•
|
|
any other special terms pertaining to such debt securities.
|
Unless otherwise specified in the applicable
prospectus supplement, the debt securities will not be listed on any securities exchange or included in any market.
None of our directors, officers,
employees, incorporators, or stockholders, past, present or future, will have any liability with respect to our obligations under the indenture or debt securities.
Original Issue Discount Securities
Debt securities may
be sold at a substantial discount below their stated principal amount and may bear no interest or interest at a rate which at the time of issuance is below market rates. Important federal income tax consequences and special considerations applicable
to any such debt securities will be described in the applicable prospectus supplement.
Indexed Securities
If the amount of payments of principal of, and premium, if any, or any interest on, debt securities of any series is determined with reference to any type of
index or formula or changes in prices of particular securities or commodities, the federal income tax consequences, specific terms and other information with respect to such debt securities and such index or formula and securities or commodities
will be described in the applicable prospectus supplement.
Conversion and Exchange
If any debt securities are convertible into or exchangeable for other securities, the prospectus supplement or term sheet will explain the terms and conditions
of the conversion or exchange, including the conversion or exchange price or rate (or the calculation method), the conversion or exchange period (or how the period will be determined), if conversion or exchange will be mandatory or at the option of
the holder or us, provisions for adjusting the conversion or exchange price or rate and provisions affecting conversion or exchange in the event of the redemption of the underlying debt securities. These terms may also include provisions under which
the number or amount of other securities to be received by the holders of the debt securities upon conversion or exchange would be calculated according to the market price of the other securities as of a time stated in the prospectus supplement or
term sheet.
Payment
Unless we specify otherwise in
the applicable prospectus supplement, payments in respect of the debt securities will be made at the office or agency office or agency maintained by us in New York, New York. Payment of any installment of interest on debt securities in registered
form will be made to the person in whose name such debt security is registered at the close of business on the regular record date for such interest.
8
Registration, Transfer and Exchange
Unless we specify otherwise in the applicable prospectus supplement, a holder may transfer or exchange debt securities in accordance with the provisions of the
indenture. The registrar and the trustee may require a holder to furnish appropriate endorsements and transfer documents in connection with a transfer of debt securities. Holders will be required to pay all taxes due on transfer. We intend to
appoint the trustee under the indenture as security registrar with respect to debt securities issued under the indenture.
Consolidation, Merger,
Conveyance, Sale of Assets and Other Transfers
Unless we specify otherwise in the applicable prospectus supplement, we may not, directly or
indirectly: (1) consolidate or merge with or into another person (whether or not we are the surviving corporation) or (2) sell, assign, transfer, convey, lease or otherwise dispose of all or substantially all of our properties or assets in
one or more related transactions, to another person; unless:
(1) either:
(a) we are the surviving corporation; or
(b) the person formed by or surviving any such consolidation or merger (if other than us) or to which such sale, assignment, transfer,
conveyance or other disposition has been made (the “Surviving Entity”) is a corporation organized or existing under the laws of the United States, any state of the United States or the District of Columbia;
(2) the Surviving Entity expressly assumes pursuant to a supplemental indenture all our obligations under the debt securities and the indenture
pursuant to agreements reasonably satisfactory to the trustee;
(3) immediately after giving effect to such transaction no default or event
of default shall have occurred and be continuing; and
For purposes of this covenant, the sale, assignment, transfer, lease, conveyance or other
disposition of all or substantially all of the properties or assets of one or more of our subsidiaries, which properties or assets, if held by us instead of such subsidiaries, would constitute all or substantially all of our properties or assets on
a consolidated basis, shall be deemed to be the transfer of all or substantially all of our properties or assets.
Modification or Amendment of the
Indenture
Unless we specify otherwise in the applicable prospectus supplement, except as provided in the next two succeeding paragraphs, the indenture
or the debt securities of any series may be amended or supplemented with the consent of the holders of at least a majority in aggregate principal amount of the debt securities of each series affected thereby then outstanding (including, without
limitation, consents obtained in connection with a purchase of, or tender offer or exchange offer for, debt securities of any series), and any existing default or event of default or compliance with any provision of the indenture or the debt
securities of any series may be waived with the consent of the holders of a majority in principal amount of the then outstanding debt securities of each such series (including, without limitation, consents obtained in connection with a purchase of,
or tender offer or exchange offer for, debt securities of any series).
Unless we specify otherwise in the applicable prospectus supplement, without the
consent of each holder of debt securities affected, an amendment, supplement or waiver may not (with respect to any debt securities of any series held by a
non-consenting
holder):
(1) reduce the principal amount of debt securities whose holders must consent to an amendment, supplement or waiver;
(2) reduce the principal of or change the stated maturity of any debt security;
(3) reduce the rate of or change the time for payment of interest on any debt security;
(4) waive a default or event of default in the payment of principal of, or interest or premium, if any, on the debt securities of any series
(except a rescission of acceleration of the debt securities of any series by the holders of at least a majority in aggregate principal amount of the then outstanding debt securities of any series and a waiver of the payment default that resulted
from such acceleration);
(5) make any debt securities of any series payable in money other than that stated in the debt securities of such
series;
9
(6) in the case of subordinated debt securities of any series, modify any of the subordination
provisions or the definition of senior debt relating to such series in a manner adverse to the holders of such subordinated debt securities;
(7) make any change in the provisions (including applicable definitions) of the indenture relating to waivers of past defaults or the rights of
holders of debt securities of any series to receive payments of principal of, or interest or premium, if any, on the debt securities of such series;
(8) waive a redemption or repurchase payment with respect to any debt security of any series;
(9) make any change in the ranking of the debt securities of any series in a manner adverse to the holders of the debt securities of such
series; or
(10) make any change in the preceding amendment and waiver provisions.
Notwithstanding the preceding and unless we specify otherwise in the applicable prospectus supplement, without the consent of any holder of debt securities,
we and the trustee may amend or supplement the indenture or the debt securities of one or more series:
(1) to cure any ambiguity, defect
or inconsistency;
(2) to provide for uncertificated debt securities in addition to or in place of certificated debt securities;
(3) to provide for the assumption of our obligations to holders of debt securities in the case of a merger or consolidation or sale of all or
substantially all of our assets or any other transaction that complies with the indenture;
(4) to make any change that would provide any
additional rights or benefits to the holders of debt securities or that does not adversely affect the legal rights under the indenture of any such holder;
(5) to comply with requirements of the SEC in order to effect or maintain the qualification of the indenture under the Trust Indenture Act;
(6) to allow any guarantor to execute a supplemental indenture and/or a guarantee with respect to the debt securities of any one or more
series;
(7) to provide for the issuance of and establish the form and terms and conditions of debt securities as permitted by the
indenture;
(8) to add to our covenants such further covenants, restrictions, conditions or provisions as we shall consider to be for the
protection of the holders of debt securities, and to make the occurrence, or the occurrence and continuance, of a default in any such additional covenants, restrictions, conditions or provisions an event of default permitting the enforcement of all
or any of the several remedies provided in the indenture; provided, that in respect of any such additional covenant, restriction, condition or provision such supplemental indenture may provide for a particular period of grace after default (which
period may be shorter or longer than that allowed in the case of other defaults) or may provide for an immediate enforcement upon such an event of default or may limit the remedies available to the trustee upon such an event of default or may limit
the right of the holders of a majority in aggregate principal amount of the debt securities to waive such an event of default;
(9) to
evidence and provide the acceptance of the appointment of a successor trustee under the indenture with respect to the debt securities and to add to or change any of the provisions of the indenture as shall be necessary to provide for or facilitate
the administration of the trusts hereunder by more than one trustee;
(10) to mortgage, pledge, hypothecate or grant a security interest in
favor of the trustee for the benefit of the holders of debt securities as additional security for the payment and performance of our or a guarantor’s obligations under the indenture in any property or assets;
(11) to add to, change, or eliminate any of the provisions of the indenture in respect of the debt securities, provided that any such addition,
change, or elimination (i) will neither (A) apply to any debt security created prior to the execution of such supplemental indenture and entitled to the benefit of such provision nor (B) modify the rights of the holder of any such
debt security with respect to such provision or (ii) will become effective only when there is no such debt security outstanding;
10
(12) to comply with the rules of any applicable securities depositary;
(13) to release a guarantor from its subsidiary guarantee pursuant to the terms of the indenture when permitted or required pursuant to the
terms of the indenture; or
(14) to comply with the covenant relating to mergers, consolidations and sales of assets.
Events of Default
Unless we specify otherwise in the
applicable prospectus supplement, each of the following is an event of default:
(1) default for 30 consecutive days in the payment when
due and payable of interest on the debt securities of that series;
(2) default in the payment when due and payable of the principal of or
premium, if any, on the debt securities of that series (upon maturity, redemption, required repurchase or otherwise);
(3) default in the
deposit of any sinking fund payment, when and as due in respect of any debt security of that series;
(4) failure by us or any of our
restricted subsidiaries to comply with the provisions described under the caption “-Consolidation, Merger, Conveyance, Sale of Assets and Other Transfers;”
(5) failure by us for 60 consecutive days after notice to us by the trustee or the holders of at least 25% in aggregate principal amount of the
debt securities of that series then outstanding to comply with any of its other covenants or agreements in the indenture or the debt securities of that series (other than a covenant or warranty that has been included in the indenture solely for the
benefit of debt securities of a series other than that series);
(6) certain events of bankruptcy, insolvency or reorganization described
in the indenture with respect to us.
(7) any other event of default provided with respect to debt securities, which is specified in a
board resolution, a supplemental indenture hereto or an officers’ certificate, in accordance with the terms of the indenture.
In the case of an
event of default specified in clause (6), the principal, premium, if any, and accrued and unpaid interest, if any, of all the outstanding debt securities of each such affected series shall become due and payable immediately without further action or
notice. If any other event of default occurs and is continuing, then, and in each and every such case, except for any series of debt securities the principal of which shall have already become due and payable the trustee or the holders of at least
25% in aggregate principal amount of the then outstanding debt securities of each such affected series (each such series voting as a separate class) may declare the principal, premium, if any, and accrued and unpaid interest, if any, of all the
outstanding debt securities due and payable immediately.
The holders of at least a majority in aggregate principal amount of the debt securities of all
series affected thereby, voting as a single class, by notice to the trustee may on behalf of the holders of all of the debt securities of such affected series waive any existing default or event of default and its consequences under the indenture,
except a continuing default or event of default in the payment of interest on, or the principal of, the debt securities of such series, and rescind any acceleration and its consequences with respect to the debt securities of such series.
Legal Defeasance and Covenant Defeasance
Unless we
specify otherwise in the applicable prospectus supplement, we may, at our option and at any time, elect to have all of our obligations discharged with respect to the outstanding debt securities of any series (“Legal Defeasance”) except
for:
(1) the rights of holders of outstanding debt securities of such series to receive payments in respect of the principal of, or
interest or premium, if any, on such debt securities of such series when such payments are due from the trust referred to below;
(2) our
obligations with respect to the debt securities of such series concerning issuing temporary debt securities, mutilated, destroyed, lost or stolen debt securities and the maintenance of an office or agency for payment and money for security payments
held in trust;
(3) the rights, powers, trusts, duties and immunities of the trustee, and our obligations in connection therewith; and
11
(4) the defeasance provisions of the indenture.
In addition, unless we specify otherwise in the applicable prospectus supplement, we may, at our option and at any time, elect to have our obligations
released with respect to certain covenants in the indenture as well as any additional covenants for a particular series of debt securities (“Covenant Defeasance
”
) and thereafter any omission to comply with those covenants will not
constitute a default or event of default with respect to the debt securities of such series. In the event Covenant Defeasance occurs, certain events (not including
non-payment,
bankruptcy, receivership,
rehabilitation and insolvency events) described under “-Events of Default” will no longer constitute an event of default with respect to the debt securities of a series.
In order to exercise either Legal Defeasance or Covenant Defeasance:
(1) we must irrevocably deposit with the trustee, in trust, for the benefit of the holders of the debt securities of such series, cash in U.S.
dollars,
non-callable
Government Securities, or a combination of cash in U.S. dollars and
non-callable
Government Securities, in amounts as will be sufficient, in the
opinion of a nationally recognized firm of independent public accountants, to pay the principal of (including mandatory sinking fund or analogous payments, if any), or interest and premium, if any, on the outstanding debt securities of such series
on the stated maturity or on the applicable redemption date, as the case may be, and we must specify whether the debt securities of such series are being defeased to maturity or to a particular redemption date;
(2) in the case of Legal Defeasance, we must deliver to the trustee an opinion of counsel reasonably acceptable to the trustee confirming that
(a) we have received from, or there has been published by, the Internal Revenue Service a ruling or (b) since the issue Date, there has been a change in the applicable federal income tax law, in either case to the effect that, and based
thereon such opinion of counsel will confirm that, the holders of the outstanding debt securities of such series will not recognize income, gain or loss for federal income tax purposes as a result of such Legal Defeasance and will be subject to
federal income tax on the same amounts, in the same manner and at the same times as would have been the case if such Legal Defeasance had not occurred;
(3) in the case of Covenant Defeasance, we have delivered to the trustee an opinion of counsel reasonably acceptable to the trustee confirming
that the holders of the outstanding debt securities of such series will not recognize income, gain or loss for federal income tax purposes as a result of such Covenant Defeasance and will be subject to federal income tax on the same amounts, in the
same manner and at the same times as would have been the case if such Covenant Defeasance had not occurred;
(4) no default or event of
default has occurred and is continuing with respect to the debt securities of such series on the date of such deposit (other than a default or event of default resulting from the borrowing of funds to be applied to such deposit and the grant of any
lien securing such borrowing);
(5) such Legal Defeasance or Covenant Defeasance will not result in a breach or violation of, or constitute
a default under any material agreement or instrument (other than the indenture) to which we or any of our subsidiaries is a party or by which we or any of our subsidiaries is bound;
(6) we must deliver to the trustee an officers’ certificate stating that the deposit was not made by us with the intent of preferring the
holders of such series of debt securities over our other creditors with the intent of defeating, hindering, delaying or defrauding our creditors or others; and
(7) we must deliver to the trustee an officers’ certificate and an opinion of counsel, each stating that all conditions precedent relating
to the Legal Defeasance or the Covenant Defeasance have been complied with.
Satisfaction and Discharge
Unless we specify otherwise in the applicable prospectus supplement, the indenture will be discharged and will cease to be of further effect as to all debt
securities of any series issued thereunder, when:
(1) either:
(a) all debt securities of any series that have been authenticated, except lost, stolen or destroyed debt securities that have been replaced or
paid and debt securities for whose payment money has been deposited in trust and thereafter repaid to us, have been delivered to the trustee for cancellation; or
12
(b) all debt securities of any series that have not been delivered to the trustee for
cancellation have become due and payable by reason of the mailing of a notice of redemption or otherwise or will become due and payable or redeemable within one year, and we have irrevocably deposited or caused to be deposited with the trustee as
trust funds in trust solely for the benefit of the holders, cash in U.S. dollars,
non-callable
Government Securities, or a combination of cash in U.S. dollars and
non-callable
Government Securities, in such amounts as will be sufficient without consideration of any reinvestment of interest, to pay and discharge the entire indebtedness on the debt securities of such
series not delivered to the trustee for cancellation for principal, premium, if any, and accrued interest to the date of maturity or redemption;
(2) no default or event of default has occurred and is continuing on the date of the deposit or will occur as a result of the deposit (other
than a default or event of default resulting from the borrowing of funds to be applied to such deposit) and the deposit will not result in a breach or violation of, or constitute a default under, any other instrument to which we are a party or by
which we are bound;
(3) we have paid or caused to be paid all sums payable by it under the indenture; and
(4) we have delivered irrevocable instructions to the trustee under the indenture to apply the deposited money toward the payment of the debt
securities of such series at maturity or the redemption date, as the case may be.
In addition, we must deliver an officers’ certificate and an
opinion of counsel to the trustee stating that all conditions precedent to satisfaction and discharge have been satisfied.
Selection and Notice
Unless we specify otherwise in the applicable prospectus supplement, if less than all of the debt securities of a series are to be redeemed at any
time, the trustee will select debt securities of the series to be redeemed not more than 60 days before the redemption date therefor in any manner that the trustee in its sole discretion deems fair and appropriate. Unless we specify otherwise in the
applicable prospectus supplement, notices of redemption will be sent at least 30 but not more than 60 days before the redemption date to each holder of debt securities to be redeemed at its registered address (or electronically for global notes),
except that redemption notices may be mailed more than 60 days prior to a redemption date if the notice is issued in connection with a defeasance of the debt securities or a satisfaction and discharge of the indenture.
Subordinated Debt Securities
Debt securities of a series
may be subordinated to senior indebtedness (as defined in the applicable prospectus supplement) to the extent set forth in the prospectus supplement relating thereto. The debt securities will be structurally subordinated to all indebtedness and
other liabilities (including medical claims liability, accounts payable and accrued expenses, unearned revenue and other long-term liabilities) of our subsidiaries. Any right we have to receive assets of any of our subsidiaries upon the
subsidiary’s liquidation or reorganization (and the consequent right of the holders of the debt securities to participate in those assets) will be effectively subordinated to the claims of that subsidiary’s creditors, except to the extent
that we are itself recognized as a creditor of the subsidiary, in which case our claims would still be subordinate in right of payment to any security in the assets of the subsidiary and any indebtedness of the subsidiary senior to that held by us.
Replacement of Securities
Unless we specify
otherwise in the applicable prospectus supplement, we will replace any mutilated debt security at the expense of the holder upon surrender of the mutilated debt security to the trustee in the circumstances described in the indenture. We will replace
debt securities that are destroyed, stolen or lost at the expense of the holder upon delivery to the trustee of evidence of the destruction, loss or theft of the debt securities satisfactory to us and to the trustee in the circumstances described in
the indenture. In the case of a destroyed, lost or stolen debt security, an indemnity and/or security satisfactory to the trustee and us, and payment of any taxes, governmental charges or other expenses, may be required from the holder of the debt
security before a replacement debt security will be issued.
Governing Law
The laws of the State of New York will govern each indenture and will govern the debt securities.
13
Regarding the Trustee
If the trustee becomes a creditor of us, the indenture limits its right to obtain payment of claims in certain cases, or to realize on certain property
received in respect of any such claim as security or otherwise. The trustee will be permitted to engage in other transactions; however, if it acquires any conflicting interest, it must (i) eliminate such conflict within 90 days, (ii) apply
to the SEC for permission to continue or (iii) resign.
The holders of a majority in principal amount of the then outstanding debt securities will
have the right to direct the time, method and place of conducting any proceeding for exercising any remedy available to the trustee, subject to certain exceptions. The indenture provides that in case an event of default occurs and is continuing, the
trustee will be required, in the exercise of its power, to use the degree of care of a prudent man in the conduct of his own affairs. Subject to such provisions, the trustee will be under no obligation to exercise any of its rights or powers under
the indenture at the request of any holder of debt securities, unless such holder has offered to the trustee security and indemnity satisfactory to it against any loss, liability or expense.
14
DESCRIPTION OF CAPITAL STOCK
The following is a summary of the material terms of our capital stock and the provisions of our certificate of incorporation and
by-laws.
It also summarizes some relevant provisions of the General Corporation Law of the State of Delaware, which we refer to as Delaware law or the DGCL. Since the terms of our certificate of incorporation,
by-laws,
and Delaware law are more detailed than the general information provided below, you should only rely on the actual provisions of those documents and Delaware law. If you would like to read those documents,
they are on file with the SEC as described under the heading “Where You Can Find More Information.”
General
Centene’s authorized capital stock consists of 410 million shares, of which:
|
|
•
|
|
400 million shares are designated as common stock, par value $0.001 per share, and
|
|
|
•
|
|
10 million shares are designated as preferred stock, par value $0.001 per share.
|
As of March 31,
2017, Centene had 172,271,202 shares of common stock issued and outstanding and no shares of preferred stock issued and outstanding.
Common Stock
Each share of our common stock entitles the holder to one vote on all matters submitted to a vote of stockholders, including the election of
directors. Subject to any preference rights of holders of preferred stock, the holders of common stock are entitled to receive dividends and other distributions in cash, stock or property, if any, declared from time to time by the directors out of
legally available funds. In the event of our liquidation, dissolution or winding up, the holders of common stock are entitled to share ratably in all assets remaining after the payment of liabilities, subject to any rights of holders of preferred
stock to prior distribution. In the event of a merger or consolidation, the holders of each share of common stock shall be entitled to receive the same per share consideration on a per share basis. Holders of common stock have no cumulative voting
rights. The common stock has no preemptive or conversion rights or other subscription rights. No redemption or sinking fund provisions apply to the common stock. All outstanding shares of common stock are fully paid and nonassessable, and the shares
of common stock to be issued upon the completion of this offering will be fully paid and nonassessable.
We may issue additional shares of authorized
common stock without stockholder approval, subject to applicable rules of the NYSE and Delaware law.
The transfer agent and registrar for the common
stock is Broadridge Corporate Issuer Solutions, Inc. Information about Broadridge Corporate Issuer Solutions, Inc. may be obtained at (855)
627-5087.
Our common stock is listed on the NYSE under the symbol
“CNC.”
Preferred Stock
The following is a
description of general terms and provisions of the preferred stock. All of the terms of the preferred stock are, or will be contained in our certificate of incorporation or in one or more certificates of designation relating to each series of the
preferred stock, which will be filed with the SEC at or prior to the issuance of the series of preferred stock, and will be available as described under the heading “Where You Can Find More Information.”
Our board of directors is authorized, without further stockholder approval but subject to applicable rules of the NYSE and any limitations prescribed by law,
to issue up to 10 million shares of preferred stock from time to time. Our board of directors has the discretion to provide for the issuance of all or any shares of preferred stock in one or more classes or series, and to fix for each such
class or series such voting powers, full or limited, or no voting powers, and such designations, preferences and relative, participating, optional or other special rights and such qualifications, limitations or restrictions thereof, as shall be
stated and expressed in the resolution or resolutions adopted by the board of directors providing for the issuance of such class or series, including, without limitation, the authority to provide that any such class or series may be:
|
|
•
|
|
subject to redemption at such time or times and at such price or prices,
|
|
|
•
|
|
entitled to receive dividends (which may be cumulative or
non-cumulative)
at such rates, on such conditions, and at such times, and payable in preference to, or in such relation
to, the dividends payable on any other class or classes or any other series,
|
|
|
•
|
|
entitled to such rights upon the dissolution of Centene or upon any distribution of our assets, or
|
15
|
|
•
|
|
convertible into, or exchangeable for, shares of any other class or classes of stock or of any other series of the same or any other class or classes of stock of Centene at such price or prices or at such rates of
exchange and with such adjustments as the board may determine.
|
The purpose of authorizing our board of directors to issue preferred stock
and determine its rights and preferences is to eliminate delays associated with a stockholder vote on specific issuances. The issuance of preferred stock may provide desirable flexibility in connection with possible acquisitions and other corporate
purposes, but could have the effect of making it more difficult for a third party to acquire, or could discourage a third party from acquiring, a majority of our outstanding voting stock.
Certain Effects of Authorized but Unissued Stock
We may
issue additional shares of common stock or preferred stock without stockholder approval, subject to applicable rules of the NYSE and Delaware law, for a variety of corporate purposes, including future public or private offerings to raise additional
capital, corporate acquisitions, and employee benefit plans and equity grants. The existence of unissued and unreserved common and preferred stock may enable us to issue shares to persons who are friendly to current management, which could
discourage an attempt to obtain control of Centene by means of a proxy contest, tender offer, merger or otherwise. We will not solicit approval of our stockholders for issuance of common and preferred stock unless our board of directors believes
that approval is advisable or is required by applicable rules of the NYSE or Delaware law.
Limitation on Liability of Directors; Indemnification
Our certificate of incorporation provides that no director shall be personally liable to Centene or any of its stockholders for monetary damages for
breach of fiduciary duty as a director, except to the extent such exemption from liability or limitation thereof is not permitted under the DGCL as the same exists or may hereafter be amended. If the DGCL is amended hereafter to authorize the
further elimination or limitation of the liability of directors, then the liability of directors shall be eliminated or limited to the fullest extent authorized by the DGCL, as so amended. Our certificate of incorporation further provides that any
repeal or modification of this limitation of liability by our stockholders shall not adversely affect any right or protection of a director of Centene existing at the time of such repeal or modification with respect to acts or omissions occurring
prior to such repeal or modification.
Our certificate of incorporation requires that we indemnify our directors and officers to the fullest extent
authorized or permitted by law, as now or hereafter in effect, and that such right to indemnification shall continue as to a person who has ceased to be a director or officer and shall inure to the benefit of his or her heirs, executors and personal
and legal representatives. Except for proceedings to enforce rights to indemnification, however, Centene shall not be obligated to indemnify in connection with a proceeding (or part thereof) if such director, officer or successor in interest
initiated such proceeding (or part thereof) unless such proceeding was authorized or consented to by the board of directors. The right to indemnification includes the right to be paid the expenses incurred in defending or otherwise participating in
any proceeding in advance of its final disposition. Any repeal or modification by the stockholders of indemnification or advancement rights shall not adversely affect any rights to indemnification and to the advancement of expenses of a director or
officer of Centene existing at the time of such repeal or modification with respect to any acts or omissions occurring prior to such repeal or modification.
The board of directors may in its discretion provide rights to indemnification and to the advancement of expenses to employees and agents of Centene similar
to those described above.
The inclusion of these provisions in our certificate of incorporation and
by-laws
may
have the effect of reducing the likelihood of derivative litigation against our directors and may discourage or deter Centene or its stockholders from bringing a lawsuit against our directors for breach of their duty of care, even though such an
action, if successful, might otherwise have benefited Centene and its stockholders.
Anti-Takeover Effects of Provisions of Delaware Law and Our
Certificate of Incorporation and
By-Laws
Some of the provisions in our certificate of incorporation and
by-laws
and Delaware law could have the following effects, among others:
|
|
•
|
|
delaying, deferring or preventing a change in control of Centene;
|
|
|
•
|
|
delaying, deferring or preventing the removal of our existing management or directors;
|
|
|
•
|
|
deterring potential acquirors from making an offer to our stockholders; and
|
|
|
•
|
|
limiting our stockholders’ opportunity to realize premiums over prevailing market prices of our common stock in connection with offers by potential acquirors.
|
16
The following is a summary of some of the provisions in our certificate of incorporation and
by-laws
that could have the effects described above. We believe that the benefits of increased protection of our potential ability to negotiate with the proponent of an unfriendly or unsolicited proposal to acquire
or restructure us outweigh the disadvantages of discouraging takeover or acquisition proposals because negotiation of these proposals could result in an improvement of their terms.
Delaware Business Combination Statute
. We must comply with Section 203 of the Delaware General Corporation Law, an anti-takeover law. In general,
Section 203 prohibits a publicly held Delaware corporation from engaging in a “business combination” with an “interested stockholder” for a period of three years following the date the person became an interested
stockholder, unless the business combination or the transaction in which the person became an interested stockholder is approved in a prescribed manner. Generally, a “business combination” includes a merger, asset or stock sale, or other
transaction resulting in a financial benefit to an interested stockholder. An “interested stockholder” includes a person who, together with affiliates and associates, owns, or did own within three years prior to the determination of
interested stockholder status, 15% or more of the corporation’s voting stock. The existence of this provision generally will have an anti-takeover effect for transactions not approved in advance by the board of directors, including discouraging
attempts that might result in a premium over the market price for the shares of common stock held by stockholders.
Other Supermajority Voting
Requirements
. In addition to the supermajority requirement for certain business combinations discussed above, Centene’s certificate of incorporation also contains other supermajority requirements, including:
|
|
•
|
|
a requirement that the vote of 75% of the outstanding shares of common stock (and any other voting shares that may be outstanding) entitled to vote generally in the election of directors is required to remove a
director, with or without cause; and
|
|
|
•
|
|
a requirement that the vote of 75% of the outstanding shares of common stock (and any other voting shares that may be outstanding) entitled to vote generally in the election of directors is required for the stockholders
to adopt, amend, alter or repeal the
by-laws;
and
|
|
|
•
|
|
a requirement that any amendment or repeal of specified provisions of Centene’s certificate of incorporation (including provisions relating to directors and amendment of our
by-laws)
must be approved by at least 75% of the outstanding shares of our common stock (and any other voting shares that may be outstanding) entitled to vote generally in the election of directors.
|
Actions at Meetings of Stockholders; Special Meetings
. Our certificate of incorporation and
by-laws
require that any action required or permitted to be taken by our stockholders must be effected at a duly called annual or special meeting of the stockholders and may not be effected by a consent in
writing. In addition, special meetings of our stockholders may be called only by the board of directors or some of our officers. These provisions may have the effect of deterring hostile takeovers or delaying or preventing changes in our control or
management.
Classified Board of Directors
. Our certificate of incorporation and
by-laws
provide that our
board of directors will be divided into three classes of directors serving staggered three-year terms. Each class, to the extent possible, will be equal in number. Each class holds office until the third annual stockholders’ meeting for
election of directors following the most recent election of such class.
Directors, and Not Stockholders, Fix the Size of the Board of Directors
.
Our certificate of incorporation and
by-laws
provide that the number of directors will be fixed from time to time exclusively pursuant to a resolution adopted by a majority of our board of directors, but in no
event will it consist of less than five nor more than eleven directors.
Board Vacancies to Be Filled by Remaining Directors and Not Stockholders
.
Under our certificate of incorporation and
by-laws,
any vacancy created by any reason prior to the expiration of the term in which the vacancy occurs will be filled by a majority of the remaining directors,
even if less than a quorum. A director elected to fill a vacancy will be elected for the unexpired term of his predecessor.
Advance Notice for
Stockholder Proposals
. Our
by-laws
contain provisions requiring that advance notice be delivered to Centene of any business to be brought by a stockholder before an annual meeting and providing for
procedures to be followed by stockholders in nominating persons for election to our board of directors. Ordinarily, the stockholder must give notice not less than 120 days nor more than 150 days prior to the anniversary date of the immediately
preceding annual meeting; provided, however, that in the event that the date of the meeting is not within 30 days before or 70 days after such date, notice by the stockholder must be received no earlier than 120 days prior to such meeting and no
later than the later of 70 days prior to the meeting or the 10th day following the day on which such public notice was first given. The notice must include a description of the proposal, the reasons for the proposal, and other specified matters. Our
board of directors may reject any proposals that have not followed these procedures or that are not a proper subject for stockholder action in accordance with the provisions of applicable law.
17
Limitation on Liability of Directors; Indemnification
Centene’s certificate of incorporation, as amended, provides that no director shall be personally liable to Centene or any of its stockholders for
monetary damages for breach of fiduciary duty as a director, except to the extent such exemption from liability or limitation thereof is not permitted under the DGCL as the same exists or may hereafter be amended. If the DGCL is amended hereafter to
authorize the further elimination or limitation of the liability of directors, then the liability of directors shall be eliminated or limited to the fullest extent authorized by the DGCL, as so amended. Centene’s certificate of incorporation,
as amended, further provides that any repeal or modification of this limitation of liability by the Centene stockholders shall not adversely affect any right or protection of a director of Centene existing at the time of such repeal or modification
with respect to acts or omissions occurring prior to such repeal or modification.
Centene’s certificate of incorporation, as amended, requires that
Centene indemnify its directors and officers to the fullest extent authorized or permitted by law, as now or hereafter in effect, and that such right to indemnification shall continue as to a person who has ceased to be a director or officer and
shall inure to the benefit of his or her heirs, executors and personal and legal representatives. Except for proceedings to enforce rights to indemnification, however, Centene shall not be obligated to indemnify in connection with a proceeding (or
part thereof) if such director, officer or successor in interest initiated such proceeding (or part thereof) unless such proceeding was authorized or consented to by the Centene Board. The right to indemnification includes the right to be paid the
expenses incurred in defending or otherwise participating in any proceeding in advance of its final disposition. Any repeal or modification by the stockholders of indemnification or advancement rights shall not adversely affect any rights to
indemnification and to the advancement of expenses of a director or officer of Centene existing at the time of such repeal or modification with respect to any acts or omissions occurring prior to such repeal or modification.
The Centene Board may in its discretion provide rights to indemnification and to the advancement of expenses to employees and agents of Centene similar to
those described above.
The inclusion of these provisions in the Centene certificate of incorporation, as amended, and amended and restated
by-laws
may have the effect of reducing the likelihood of derivative litigation against Centene’s directors and may discourage or deter Centene or its stockholders from bringing a lawsuit against Centene’s
directors for breach of their duty of care, even though such an action, if successful, might otherwise have benefited Centene and its stockholders.
General Provisions Related to Centene Preferred Stock
The following is a description of general terms and provisions of the Centene preferred stock. All of the terms of the Centene preferred stock are, or will be
contained in Centene’s certificate of incorporation, as amended, or in one or more certificates of designation relating to each series of the preferred stock, which will be filed with the SEC at or prior to the issuance of the series of
preferred stock, and will be available as described under the heading “Where You Can Find More Information.”
The Centene Board is authorized,
without further stockholder approval but subject to applicable rules of the NYSE and any limitations prescribed by law, to issue up to ten million shares of preferred stock from time to time. The Centene Board has the discretion to provide for the
issuance of all or any shares of preferred stock in one or more classes or series, and to fix for each such class or series such voting powers, full or limited, or no voting powers, and such designations, preferences and relative, participating,
optional or other special rights and such qualifications, limitations or restrictions thereof, as shall be stated and expressed in the resolution or resolutions adopted by the board of directors providing for the issuance of such class or series,
including, without limitation, the authority to provide that any such class or series may be:
|
|
•
|
|
subject to redemption at such time or times and at such price or prices;
|
|
|
•
|
|
entitled to receive dividends (which may be cumulative or
non-cumulative)
at such rates, on such conditions, and at such times, and payable in preference to, or in such relation
to, the dividends payable on any other class or classes or any other series;
|
|
|
•
|
|
entitled to such rights upon the dissolution of Centene or upon any distribution of Centene’s assets; or
|
|
|
•
|
|
convertible into, or exchangeable for, shares of any other class or classes of stock or of any other series of the same or any other class or classes of stock of Centene at such price or prices or at such rates of
exchange and with such adjustments as the board may determine.
|
The purpose of authorizing the Centene Board to issue preferred stock and
determine its rights and preferences is to eliminate delays associated with a stockholder vote on specific issuances. The issuance of preferred stock may provide desirable flexibility in connection with possible acquisitions and other corporate
purposes, but could have the effect of making it more difficult for a third party to acquire, or could discourage a third party from acquiring, a majority of Centene’s outstanding voting stock.
18
Certain Effects of Authorized but Unissued Stock
Centene may issue additional shares of common stock or preferred stock without stockholder approval, subject to applicable rules of the NYSE and Delaware law,
for a variety of corporate purposes, including future public or private offerings to raise additional capital, corporate acquisitions, and employee benefit plans and equity grants. The existence of unissued and unreserved common and preferred stock
may enable Centene to issue shares to persons who are friendly to current management, which could discourage an attempt to obtain control of Centene by means of a proxy contest, tender offer, merger or otherwise. Centene will not solicit approval of
its stockholders for issuance of common and preferred stock unless the Centene Board believes that approval is advisable or is required by applicable rules of the NYSE or Delaware law.
19
DESCRIPTION OF THE DEPOSITARY SHARES
We may elect to offer depositary shares represented by depositary receipts. If we so elect, each depositary share will represent a fractional interest in a
share of preferred stock or multiple shares of preferred stock with the amount of the preferred shares to be specified in the applicable prospectus supplement. If we issue depositary shares representing interests in shares of preferred stock, those
shares of preferred stock will be deposited with a depositary.
The shares of any series of preferred stock underlying the depositary shares will be
deposited under a separate deposit agreement between us and a bank or trust company having its principal office in the United States. The applicable prospectus supplement will set forth the name and address of the depositary and a form of deposit
agreement will be filed with the SEC as an exhibit to the registration statement by post-effective amendment or to a Current Report on Form
8-K.
Subject to the terms of the deposit agreement, each owner of a
depositary share will have a pro rata interest in all the rights and preferences of the preferred stock underlying the depositary share. Those rights include any dividend, voting, redemption, conversion, exchange and liquidation rights. In addition
to this summary, you should refer to the applicable prospectus supplement and the detailed provisions of the relevant deposit agreement for complete terms of the deposit agreement.
The depositary shares will be evidenced by depositary receipts issued under the deposit agreement. If you purchase interests in shares of the related series
of preferred stock, you will receive depositary receipts as described in the applicable prospectus supplement. While the final depositary receipts are being prepared, we may order the depositary to issue temporary depositary receipts substantially
identical to the final depositary receipts although not in final form. The holders of the temporary depositary receipts will be entitled to the same rights as if they held the depositary receipts in final form. Holders of the temporary depositary
receipts can exchange them for the final depositary receipts at our expense.
20
DESCRIPTION OF THE WARRANTS
We may issue warrants, in one or more series, for the purchase of debt securities, shares of our preferred stock or shares of our common stock. Warrants may
be issued independently or together with our debt securities, preferred stock or common stock and may be attached to or separate from any offered securities. Each series of warrants will be issued under a separate warrant agreement. In addition to
this summary, you should refer to the applicable prospectus supplement and the detailed provisions of the relevant warrant agreement for complete terms of the warrants and the warrant agreement. Unless otherwise specified in a prospectus supplement
accompanying this prospectus, each warrant agreement will be between us and a banking institution organized under the laws of the United States or a state thereof as warrant agent. In connection with an offering of our warrants, a form of warrant
agreement will be filed with the SEC as an exhibit to the registration statement by post-effective amendment or to a Current Report on Form
8-K.
Warrants will be evidenced by warrant certificates. Unless otherwise specified in the applicable prospectus supplement, the warrant certificates may be traded
separately from the debt securities, preferred stock or common stock, if any, with which the warrant certificates were issued. Warrant certificates may be exchanged for new warrant certificates of different denominations at the office of an agent
that we will appoint. Until a warrant is exercised, the holder of a warrant will not have any of the rights of a holder of our debt securities, preferred stock or common stock and will not be entitled to any payments on any debt securities,
preferred stock or common stock issuable upon exercise of the warrants.
The prospectus supplement relating to a particular series of warrants to issue
debt securities, preferred stock or common stock will describe the terms of those warrants, including the following, where applicable:
|
|
•
|
|
the title and the aggregate number of warrants;
|
|
|
•
|
|
the offering price for the warrants (if any);
|
|
|
•
|
|
the designation and terms of the securities purchasable upon exercise of the warrants;
|
|
|
•
|
|
the dates on which the right to exercise such warrants commence and expire;
|
|
|
•
|
|
the price or prices at which such warrants are exercisable;
|
|
|
•
|
|
the currency or currencies in which the offering price (if any) and the exercise price for such warrants are payable;
|
|
|
•
|
|
the periods during which and the places at which such warrants are exercisable;
|
|
|
•
|
|
the date (if any) on and after which such warrants and the securities purchasable upon exercise of such warrants will be separately transferable;
|
|
|
•
|
|
the redemption or call provisions (if any) applicable to the warrants;
|
|
|
•
|
|
the identity of the warrant agent;
|
|
|
•
|
|
the exchanges (if any) on which such warrants may be listed;
|
|
|
•
|
|
information with respect to book-entry procedures, if any;
|
|
|
•
|
|
a discussion of material U.S. federal income tax considerations; and
|
|
|
•
|
|
any other terms of or material information about such warrants, including terms, procedures and limitations relating to the exchange and exercise of the warrants.
|
21
SELLING SECURITYHOLDERS
Information about selling securityholders, where applicable, will be set forth in a prospectus supplement, in a post-effective amendment or in filings we make
with the SEC which are incorporated by reference into this prospectus.
PLAN OF DISTRIBUTION
We and/or the selling stockholders may sell any of the securities being offered by this prospectus in any one or more of the following ways from time to time:
|
|
•
|
|
through agents or dealers;
|
|
|
•
|
|
to or through underwriters;
|
|
|
•
|
|
directly by us and/or the selling stockholders to purchasers; or
|
|
|
•
|
|
a combination of any such methods of sale; and
|
|
|
•
|
|
any other method permitted pursuant to applicable law.
|
We and/or the selling stockholders will describe the
details of any such offering and the plan of distribution for any securities offering in a prospectus supplement.
In addition, to the extent this
prospectus is used by any selling securityholder to resell any securities, information with respect to the selling securityholder and the plan of distribution will be contained in a supplement to this prospectus, in a post-effective amendment or in
filings we make with the SEC under the Exchange Act that are incorporated by reference.
Underwriters, dealers and agents that participate in the
distribution of our securities may be underwriters as defined in the Securities Act and any discounts or commissions they receive from us and any profit on their resale of the securities may be treated as underwriting discounts and commissions under
the Securities Act. We will identify in the applicable prospectus supplement any underwriters, dealers or agents and will describe their compensation. We may have agreements with the underwriters, dealers and agents to indemnify them against
specified civil liabilities, including liabilities under the Securities Act. Underwriters, dealers and agents may engage in transactions with or perform services for us or our subsidiaries in the ordinary course of their businesses.
LEGAL MATTERS
The validity of the securities offered hereby will be passed upon for us by Bryan Cave LLP, St. Louis, Missouri.
EXPERTS
The
consolidated financial statements of Centene Corporation as of December 31, 2016 and 2015, and for each of the years in the three-year period ended December 31, 2016, and management’s assessment of the effectiveness of internal
control over financial reporting as of December 31, 2016 have been incorporated by reference herein in reliance upon the reports of KPMG LLP, an independent registered public accounting firm, incorporated by reference herein, and upon the
authority of said firm as experts in accounting and auditing.
The consolidated financial statements of Health Net, Inc. and subsidiaries, incorporated in
this Prospectus by reference from the Current Report on Form
8-K
of Centene Corporation filed on March 24, 2016 (as amended on May 10, 2016 and as further amended on June 9, 2016) (the
“Form
8-K”),
have been audited by Deloitte & Touche LLP, an independent registered public accounting firm, as stated in their report, which is incorporated herein by reference from the Form
8-K.
Such consolidated financial statements have been so incorporated in reliance upon the report of such firm given upon their authority as experts in accounting and auditing.
22

Centene Corporation
Up to 1,448,714 Shares of Common Stock
PROSPECTUS
SUPPLEMENT
The date of this prospectus supplement is March 13, 2018
Centene (NYSE:CNC)
Historical Stock Chart
From Aug 2024 to Sep 2024
Centene (NYSE:CNC)
Historical Stock Chart
From Sep 2023 to Sep 2024