3M™ Tegaderm™ CHG I.V. Securement Dressing Only Transparent Dressing to Meet New CDC Recommendation for Reducing Catheter...
November 17 2017 - 12:17PM
Business Wire
Tegaderm CHG I.V. Securement Dressing is only
dressing fully compliant with updated CDC recommendation and INS
Infusion Therapy Standards of Practice
The Centers for Disease Control (CDC) recently updated its
recommendation on use of chlorhexidine-impregnated dressings in its
globally-recognized Guidelines for the Prevention of Intravascular
Catheter-Related Infections (2011). The new recommendation calls
for use of chlorhexidine-impregnated dressings with an FDA-cleared
label that specifies a clinical indication for reducing
catheter-related bloodstream infection (CRBSI) or
catheter-associated bloodstream infection (CABSI) to protect the
insertion site of short-term, non-tunneled central venous catheters
for patients aged 18 years and older.
This press release features multimedia. View
the full release here:
http://www.businesswire.com/news/home/20171117005655/en/
3M™ Tegaderm™ CHG Chlorhexidine Gluconate
I.V. Securement Dressing is only dressing fully compliant with
updated CDC recommendation and INS Infusion Therapy Standards of
Practice (Photo: 3M)
3M Tegaderm CHG I.V. Securement Dressing remains the only
transparent dressing indicated and proven to reduce CRBSI, aligning
with evidence-based guidelines and practice standards. This
includes the 2016 Infusion Therapy Standards of Practice
recommendation for the use of transparent dressings that permit
continuous visual inspection of the catheter site.1
“The CDC’s evidence-based recommendations elevate current best
practices in reducing life-threatening and costly bloodstream
infections. The revisions highlight the strong clinical data that
supports use of Tegaderm CHG I.V. Securement Dressing worldwide,”
said Pat Parks, MD, PhD, medical director for 3M Critical and
Chronic Care Solutions Division. “At 3M, our goal is zero
bloodstream infections. We’ll keep innovating and educating to make
that future possible.”
Tegaderm CHG I.V. Securement Dressing meets the CDC’s highest
evidence-based recommendation category, IA, meaning it is a strong
recommendation supported by high-to-moderate quality evidence
suggesting net clinical benefits or harms. This evidence includes a
randomized controlled trial of 1,879 subjects that found Tegaderm
CHG I.V. Securement Dressing reduced CRBSI by 60 percent in
patients with central and arterial lines (p=0.020).2 The Dressing
provides an integrated solution that offers infection reduction,
site visibility, consistent application and catheter securement in
one easy-to-use product to provide a more convenient and reliable
solution for I.V. care and maintenance.
For more information about the clinical evidence supporting
Tegaderm CHG I.V. Securement Dressing and how the Dressing meets
other industry standards and guidelines, please visit
3M.com/TegadermCHG.
3M and Tegaderm are trademarks of 3M Company.
About 3MAt 3M, we apply science in collaborative ways to
improve lives daily. With $30 billion in sales, our 90,000
employees connect with customers all around the world. Learn more
about 3M’s creative solutions to the world’s problems at
www.3M.com or on Twitter @3M or @3MNews.
1 Infusion Nurses Society (INS). Infusion Therapy Standards of
Practice. INS; 2016.2 Timsit JF et al. Randomized controlled trial
of chlorhexidine dressing and highly adhesive dressing for
preventing catheter-related infections in critically ill adults. Am
J Crit Care Med. 2012;186(12): 1272-1278
http://www.atsjournals.org/doi/pdf/10.1164/rccm.201206-1038OC.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20171117005655/en/
Inprela CommunicationsJackie Vos,
612-677-2022Jackie@inprela.comor3MEvan Paskach,
651-276-7641epaskach@mmm.com
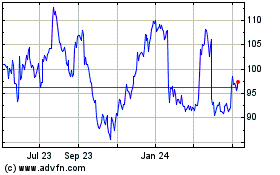
3M (NYSE:MMM)
Historical Stock Chart
From Aug 2024 to Sep 2024
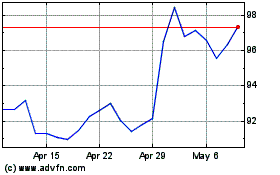
3M (NYSE:MMM)
Historical Stock Chart
From Sep 2023 to Sep 2024