NanoViricides Signs Agreement with Viroclinics Biosciences BV for Testing of Various NanoViricides Against Specific Viruses o...
February 04 2014 - 7:00AM
Business Wire
Tuesday, February 4th, 2014 - NanoViricides, Inc. (NYSE MKT:
NNVC) (the “Company”) reported today that it has signed a
“confidential disclosure agreement” (“CDA”) with Viroclinics
Biosciences, BV (“Viroclinics”), a spinoff of the Department of
Viroscience at the Erasmus Medical Centre (“Erasmus”) in Rotterdam,
the Netherlands. The agreement will allow the scientists at
Viroclinics to develop a specific proposal for the testing of
different nanoviricides® , such as FluCide™, against viruses of
mutual interest to both organizations.
Viroclinics is a virology contract research organization serving
the biopharmaceutical community
(http://www.viroclinics.com/en-GB/). Viroclinics provides
preclinical as well as clinical development services for vaccines
and antivirals. The ongoing, intimate interaction between
scientists at Erasmus and Viroclinics staff yields a strong,
flourishing platform to create and implement assays at
the forefront of viroscience. Viroclinics participated in the
discovery and characterization of the Middle East Respiratory
Syndrome (MERS) Coronavirus in 2013 and SARS Coronavirus in 2003,
attesting to its leadership position in the field.
NanoViricides, Inc. anticipates that we will be able to test and
develop our drug candidates against influenzas, dengue, MERS
Coronavirus, as well as rabies, with the help of Viroclinics. In
particular, NanoViricides is interested in the testing of its lead
broad-spectrum anti-influenza drug candidate FluCide against
various influenza A strains including the highly lethal H5N1 and
H7N9 viruses that pose a strong global public health threat.
Testing of nanoviricides antiviral drug candidates will be
performed in a BSL2 or BSL3 facility at Viroclinics, as applicable.
These facilities are designed to contain and enable the safe
handling of organisms that can pose a significant threat to health.
Executives from NanoViricides plan to visit the Rotterdam facility
later this month to conclude the final research agreement.
About NanoViricides:
NanoViricides, Inc. (www.nanoviricides.com) is a development stage company that is creating
special purpose nanomaterials for viral therapy. The Company's
novel nanoviricide® class of drug candidates are designed to
specifically attack enveloped virus particles and to dismantle
them. The Company is developing drugs against a number of viral
diseases including H1N1 swine flu, H5N1 bird flu, seasonal
Influenza, HIV, oral and genital Herpes, viral diseases of the eye
including EKC and herpes keratitis, Hepatitis C, Rabies, Dengue
fever, and Ebola virus, among others.
This press release contains forward-looking statements that
reflect the Company's current expectation regarding future events.
Actual events could differ materially and substantially from those
projected herein and depend on a number of factors. Certain
statements in this release, and other written or oral statements
made by NanoViricides, Inc. are “forward-looking statements” within
the meaning of Section 27A of the Securities Act of 1933 and
Section 21E of the Securities Exchange Act of 1934. You should not
place undue reliance on forward-looking statements since they
involve known and unknown risks, uncertainties and other factors
which are, in some cases, beyond the Company's control and which
could, and likely will, materially affect actual results, levels of
activity, performance or achievements. The Company assumes no
obligation to publicly update or revise these forward-looking
statements for any reason, or to update the reasons actual results
could differ materially from those anticipated in these
forward-looking statements, even if new information becomes
available in the future. Important factors that could cause actual
results to differ materially from the company's expectations
include, but are not limited to, those factors that are disclosed
under the heading "Risk Factors" and elsewhere in documents filed
by the company from time to time with the United States Securities
and Exchange Commission and other regulatory authorities. Although
it is not possible to predict or identify all such factors, they
may include the following: demonstration and proof of principle in
pre-clinical trials that a nanoviricide is safe and effective;
successful development of our product candidates; our ability to
seek and obtain regulatory approvals, including with respect to the
indications we are seeking; the successful commercialization of our
product candidates; and market acceptance of our products.
NanoViricides, Inc.Amanda Schuon,
310-550-7200info@nanoviricides.com
NanoViricides (AMEX:NNVC)
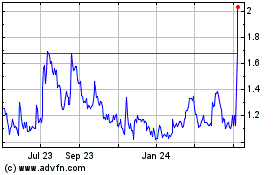
Historical Stock Chart
From Mar 2024 to Apr 2024
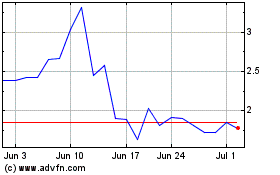
NanoViricides (AMEX:NNVC)
Historical Stock Chart
From Apr 2023 to Apr 2024