Prototyping costs reduced by 75% and
development cycles accelerated by 50%, enabling rapid
time-to-market
Video: Using Stratasys’ Objet500 Connex3
Multi-material, Color 3D Printer, NIDEK Technologies is 3D printing
engineered prototyping materials that match the performance of
metal counterparts
Stratasys Ltd. (Nasdaq:SSYS), the 3D printing and additive
manufacturing solutions company, today announced that Italian-based
NIDEK Technologies, one of the world’s largest producer of surgical
eye equipment, is using a Stratasys Objet500 Connex3
Multi-material, Color 3D Printer to accelerate clinical trial
evaluations of its ophthalmological devices (vision diagnostic
systems). The R&D division has reported a 75% reduction in
prototyping costs, while cutting lead times by 50% compared to its
previous methods.
This Smart News Release features multimedia.
View the full release here:
http://www.businesswire.com/news/home/20170530005125/en/
With Stratasys 3D printing solutions,
NIDEK Technologies is producing high-performance prototypes to
enter into clinical trials 75% cheaper and 50% faster than with
traditional methods (Photo: Business Wire)
According to Cesare Tanassi, Managing Director at NIDEK
Technologies, the adoption of multi-material 3D printing is
transforming the division’s prototyping process. The team is now
able to use advanced 3D printed materials to produce parts that
directly replicate the final components. This provides the
opportunity to quickly and comprehensively evaluate the fit, form
and function of prototype devices and speed up time-to-market.
Optimizing development cycles with 3D printingLocated in
Padova, Italy, NIDEK Technologies develops technology for vision
diagnostic systems. With products having direct contact with
patients, it is crucial that the division produces fully-functional
prototypes that precisely replicate the final product.
“Our prototyping process has become much more accurate and
streamlined since incorporating Stratasys 3D printing into our
workflow,” says Tanassi. “Previously we were constrained by the
prototyping restrictions associated with traditional methods. This
proved costly in terms of lead-times and capital, particularly with
molds and CNC machining. We found that we had limited flexibility
over our prototypes and, should iterations be required, this
resulted in escalating costs.
“3D printing overcomes these bottlenecks by permitting us to
quickly validate designs before entering our prototypes into
clinical trial validation. As you can imagine, fully verifying our
products is crucial to ensuring that premium quality is
maintained,” he continues.
Replacing metal parts with durable engineered
photopolymersThroughout its development cycle the division
exchanges expensive and cumbersome metal components with tough,
engineered 3D printed parts. These include components of a working
Gonioscope® (a system designed to observe the space between the
iris and cornea) and a micro perimeter (a device used to determine
the level of light perceived by specific areas of the retina).
According to Tanassi, the ability to incorporate 3D printed
materials that match the performance of their metal counterparts is
crucial to accelerating the development cycle for clinical trial
evaluation. He explains: “In the case of the Gonioscope®, utilizing
the toughness, flexibility and snap-fit characteristics of the
Stratasys Rigur 3D printing material, we replaced several aluminum
parts with a single 3D printed component. This saw the device pass
a year-long clinical trial where eight global medical centers
examined it.”
Fully-functional prototypes down from two months to just 24
hoursThese prototyping benefits are felt across the board as
Federico Carraro, Mechanical Division Manager at NIDEK
Technologies, highlights: “Previously we used metal fabrication
when developing the micro perimeter, which took around two months.
With our Objet500 Connex3 3D Printer, we can combine a wide range
of 3D printed materials with contrasting mechanical
characteristics. This allows us to accurately emulate final parts,
including threads, seals, rubber and transparent components. In
this case, we achieved the same functional result within 24 hours
by replacing metal parts with robust 3D printed components.”
“NIDEK Technologies is a prime example of how a growing number
of manufacturers are maximizing the full potential of 3D printing
to quickly and cost-effectively deliver fully-operational
prototypes with the precision and endurance of the final product,”
says Giuseppe Cilia, Stratasys Italian Sales Manager. “In the
fast-paced medical device industry, the ability to rapidly certify
products is crucial to improving the quality of care, and in this
case, save someone’s vision.”
To learn more about how NIDEK Technologies is using advanced
multi-material 3D printing to accelerate clinical trial evaluation
of its devices, please watch this video.
For nearly 30 years, Stratasys Ltd. (NASDAQ:SSYS)
has been a defining force and dominant player in 3D printing and
additive manufacturing – shaping the way things are made.
Headquartered in Minneapolis, Minnesota and Rehovot, Israel, the
company empowers customers across a broad range of vertical markets
by enabling new paradigms for design and manufacturing. The
company’s solutions provide customers with unmatched design freedom
and manufacturing flexibility – reducing time-to-market and
lowering development costs, while improving designs and
communications. Stratasys subsidiaries include MakerBot and
Solidscape, and the Stratasys ecosystem includes 3D printers for
prototyping and production; a wide range of 3D printing materials;
parts on-demand via Stratasys Direct Manufacturing; strategic
consulting and professional services; and the Thingiverse and
GrabCAD communities with over 2 million 3D printable files for free
designs. With more than 2,700 employees and 1,200 granted or
pending additive manufacturing patents, Stratasys has received more
than 30 technology and leadership awards. Visit us online at:
www.stratasys.com or http://blog.stratasys.com/, and follow us on
LinkedIn.
Stratasys, Stratasys signet logo, Connex and PolyJet are
trademarks or registered trademarks of Stratasys Ltd. and/or its
subsidiaries or affiliates.
Attention Editors, if you publish reader-contact information,
please use:
- USA 1-877-489-9449
- Europe/Middle East/Africa
+49-7229-7772-0
- Asia Pacific +852 3944-8888
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170530005125/en/
Stratasys Media ContactsStratasysArita Mattsoff,
+972 74 745 4000 (IL)arita@stratasys.comorJoe Hiemenz, +1
952-906-2726 (US)joe.hiemenz@stratasys.comorNorth
AmericaCraig Librett, +1
518-424-2497Craig.Librett@stratasys.comorEuropeJonathan Wake
/ Miguel Afonso, +44 1737 215200Incus
Mediastratasys@incus-media.comorAsia Pacific & Greater
ChinaStratasys APJanice Lai, +852 3944
8888Media.ap@stratasys.comorJapanStratasys JapanAya
Yoshizawa, +81 90 6473
1812aya.yoshizawa@stratasys.comorKoreaStratasys KoreaJihyun
Lee, +82 2 2046 2287Jihyun.lee@stratasys.comorMexico, Central
America, Caribe and South AmericaStratasys MexicoYair Canedo,
+52 55 4169 4181yair.canedo@stratasys.comorBrazilClezia
Martins Gomes, +55 (11) 3129 5158GPCOMclezia@gpcom.com.br
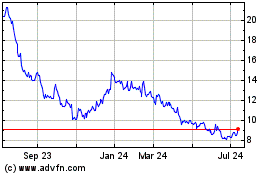
Stratasys (NASDAQ:SSYS)
Historical Stock Chart
From Apr 2024 to May 2024
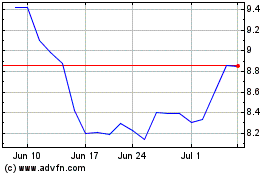
Stratasys (NASDAQ:SSYS)
Historical Stock Chart
From May 2023 to May 2024