Theseus Gets FDA Orphan Designation for THE-630 in Advanced GIST
February 02 2022 - 8:12AM
Dow Jones News
By Colin Kellaher
Theseus Pharmaceuticals Inc. on Wednesday said the U.S. Food and
Drug Administration granted orphan-drug designation to THE-630, the
company's lead candidate for the treatment of patients with
advanced gastrointestinal stromal tumors, or GIST.
The Cambridge Mass., clinical-stage biopharmaceutical company
recently initiated a first-in-human study of THE-630 in GIST, the
most common sarcoma of the gastrointestinal tract, with an
estimated 4,000 to 6,000 new cases diagnosed in the U.S. each
year.
The FDA's orphan-drug program gives special status to drugs and
biologics for diseases and disorders that affect fewer than 200,000
people in the U.S. and provides for an extended marketing
exclusivity period against competition.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
February 02, 2022 07:57 ET (12:57 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
Theseus Pharmaceuticals (NASDAQ:THRX)
Historical Stock Chart
From Mar 2024 to Apr 2024
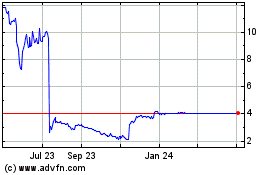
Theseus Pharmaceuticals (NASDAQ:THRX)
Historical Stock Chart
From Apr 2023 to Apr 2024