Tandem Diabetes Care, Inc. (NASDAQ: TNDM), a leading insulin
delivery and diabetes technology company, today announced the
United States commercial launch of its new Tandem Mobi, the world’s
smallest, durable automated insulin delivery system for people
living with diabetes.* The company has begun taking orders and
shipping to eligible customers in the United States.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20240213214991/en/
Tandem Mobi, the world’s smallest,
durable insulin delivery system, now shipping in the United States.
(Photo: Business Wire)
Tandem Mobi is a new way for people living with diabetes to
experience the life-changing benefits of Control-IQ technology, the
same technology that helped make Tandem the #1 recommended pump
brand.1 The Control-IQ advanced hybrid closed-loop automated
insulin delivery feature, cleared for use by people with type 1
diabetes age 6 and up, predicts and helps prevent high and low
blood sugar, and has demonstrated improved time in range throughout
the day and night in numerous controlled clinical trials and
real-world studies.2-4
“This tiny, wearable pump exceeded the expectations of users
from an early access program, and we are thrilled to begin offering
this exciting new technology to more people in the diabetes
community,” said John Sheridan, president and chief executive
officer. "With this launch, we are executing on our strategy to
offer a differentiated portfolio of durable insulin pumps,
providing choice, along with new options in wearability.”
Key Tandem Mobi System Features:
- Impressively Small – Less than half the size of the
t:slim X2 insulin pump and small enough to fit in the coin pocket
of a pair of jeans
- Wearability Like Never Before – Can be worn almost
anywhere, giving users greater discretion, comfort, and options for
how they manage their diabetes:
- Wear it on-body with a lightweight adhesive sleeve (sold
separately)
- Clip it discreetly to clothing
- Slip it into a pocket
- Infusion Set Options – Compatible with all existing
Tandem-branded infusion sets manufactured by Convatec, including a
new 5-inch tubing option made just for Tandem Mobi. Infusion sets
allow users to temporarily disconnect from their pump for
convenience and provide the flexibility of more than 30
mix-and-match infusion site and tubing length combinations.
- Tandem Mobi Mobile App – Full iOS mobile control through
a user’s compatible iPhone5
- Sensor Compatibility – Compatible with the Dexcom G6
Continuous Glucose Monitoring (CGM) System. Additional CGM sensor
integrations are planned, including Dexcom G7 in the second quarter
of 2024, followed by integration with the Abbott FreeStyle Libre 3
sensor.
- #1 Rated1 Control-IQ Technology – Uses values
from a Dexcom G6 CGM to predict glucose levels 30 minutes ahead and
automatically adjust insulin, if needed, to help prevent highs and
lows
- Clear 200-unit cartridge – Allows users to see their
remaining insulin level
- Water-Resistant – The pump is water-resistant to a depth
of 8 feet (2.4 meters) for up to 2 hours (IP28 rating)
- Flexible Pump Control – The Tandem Mobi system has a
physical button that allows users to deliver a bolus of insulin
without their iPhone.
- Advanced Technology – Uses modern wireless charging and
is capable of remote software updates6
Accessing the Tandem Mobi System
Orders will be processed on a first come, first served basis,
with initial shipments to users who meet specific eligibility
requirements, including use of the Dexcom G6 CGM to access
automated insulin delivery features. The Tandem Customer Care team
is available now to help determine eligibility and potential timing
of pump shipments for customers. People living with diabetes
interested in the Tandem Mobi system can start the order process
at: www.tandemdiabetes.com/mobi.
About Tandem Diabetes Care
Tandem Diabetes Care, a global insulin delivery and diabetes
technology company, manufactures and sells advanced automated
insulin delivery systems that reduce the burden of diabetes
management, while creating new possibilities for patients, their
loved ones, and healthcare providers. The Company’s pump portfolio
features the Tandem Mobi system and the t:slim X2 insulin pump,
both of which feature Control-IQ advanced hybrid closed-loop
technology. Tandem Diabetes Care is based in San Diego, California.
For more information, visit tandemdiabetes.com.
Follow Tandem Diabetes Care on X @tandemdiabetes; use #tslimX2
#TandemMobi and #TandemDiabetes.
Follow Tandem Diabetes Care on Facebook at
www.facebook.com/TandemDiabetes.
Follow Tandem Diabetes Care on LinkedIn at
https://www.linkedin.com/company/tandemdiabetes.
© 2024 Tandem Diabetes Care, Inc. All rights reserved. Tandem
Diabetes Care, the Tandem logo, Control-IQ, t:slim X2, and Tandem
Mobi are either registered trademarks or trademarks of Tandem
Diabetes Care, Inc. in the United States and/or other countries.
Dexcom, Dexcom G6, Dexcom G7, and any related logos and design
marks are either registered trademarks or trademarks of Dexcom,
Inc. in the United States and/or other countries. FreeStyle, Libre,
and related brand marks are marks of Abbott and used with
permission. iPhone is a trademark of Apple Inc., registered in the
U.S. and other countries. All other third-party marks are the
property of their respective owners.
Responsible Use of Control-IQ technology
Control-IQ technology does not prevent all highs and lows. Users
must still bolus for meals and actively manage their diabetes.
Visit tandemdiabetes.com/responsible-use for additional important
safety information.
*As of Feb., 2024. Data on file, Tandem Diabetes Care.
1. dQ&A US Diabetes Connections Patient Panel Report; Net
Promoter Score; Q1 2019-Q3 2023: P.49.
2. All published clinical trials and real-world studies of
Control-IQ technology to date are based on use of Dexcom G6 CGM
with the Tandem t:slim X2 insulin pump.
3. Breton MD, Kovatchev BP. One year real-world use of the
Control-IQ advanced hybrid closed-loop technology. Diabetes Technol
Ther. 2021;23(9):601-608. Doi: 10.1089/dia.2021.0097.
4. Beck RW, Kanapka LG, Breton MD, et al. A Meta-Analysis of
Randomized Trial Outcomes for the t:slim X2 Insulin Pump with
Control-IQ Technology in Youth and Adults from Age 2 to 72.
Diabetes Technol Ther. 2023 May;25(5):329-342. doi:
10.1089/dia.2022.0558.
5. The mobile app requires a compatible iPhone model and
operating system (sold separately). Only available to pump users
who reside in the United States.
6. Future updates for all or some of Tandem’s products may not
be developed and may not be offered everywhere and would be subject
to applicable regulatory approvals. Software updates are only
available to customers who are in warranty at the time they update
their pump. Additional training may be required to access certain
software updates. Charges may apply. Tandem may discontinue select
software and features over time at its discretion.
Important Safety Information
RX ONLY. Indications for Use: Tandem Mobi system: The Tandem Mobi insulin pump
with interoperable technology (the pump) is intended for the
subcutaneous delivery of insulin, at set and variable rates, for
the management of diabetes mellitus in persons requiring insulin.
The pump is able to reliably and securely communicate with
compatible, digitally connected devices, including automated
insulin dosing software, to receive, execute, and confirm commands
from these devices. The pump is intended for single patient, home
use and requires a prescription. The pump is indicated for use in
individuals 6 years of age and greater. Control-IQ technology: Control-IQ technology is
intended for use with compatible integrated continuous glucose
monitors (iCGM, sold separately) and alternate controller enabled
(ACE) pumps to automatically increase, decrease, and suspend
delivery of basal insulin based on iCGM readings and predicted
glucose values. It can also deliver correction boluses when the
glucose value is predicted to exceed a predefined threshold.
Control-IQ technology is intended for the management of Type 1
diabetes mellitus in persons 6 years of age and greater. Control-IQ
technology is intended for single patient use. Control-IQ
technology is indicated for use with NovoLog or Humalog U-100
insulin.
WARNING: Control-IQ technology should not be used by
anyone under the age of 6 years old. It should also not be used in
patients who require less than 10 units of insulin per day or who
weigh less than 55 pounds.
Control-IQ technology is not indicated for use in pregnant
women, people on dialysis, or critically ill patients. Do not use
Control-IQ technology if using hydroxyurea. Users of a Tandem
insulin pump and Control-IQ technology must use the insulin pump,
CGM, and all other system components in accordance with their
respective instructions for use; test blood glucose levels as
recommended by their healthcare provider; demonstrate adequate
carb-counting skills; maintain sufficient diabetes self-care
skills; see healthcare provider(s) regularly; and have adequate
vision and/or hearing to recognize all functions of the pump,
including alerts, alarms, and reminders. The Tandem pump and the
CGM transmitter and sensor must be removed before MRI, CT, or
diathermy treatment. Visit tandemdiabetes.com/safetyinfo for
additional important safety information.
Forward-looking Statements
This press release contains “forward-looking statements” within
the meaning of Section 27A of the Securities Act of 1933, as
amended, and Section 21E of the Securities Exchange Act of 1934, as
amended. These forward-looking statements relate to, among other
things, the anticipated integrations of the Tandem Mobi insulin
pump with the Dexcom G7 and Abbott Freestyle Libre 3 CGM sensors.
These statements are subject to numerous risks and uncertainties,
including the risks associated with the research and development
process generally, such as the design, testing, and validation in
compliance with the applicable regulatory and legal requirements in
the markets that we serve as well as our ability to market, scale,
and maintain the systems, personnel, and infrastructure necessary
to support these integrations. These and other risks are identified
and described in greater detail under the “Risk Factors” heading of
our most recent Annual Report on Form 10-K, Quarterly Reports on
Form 10-Q, and other documents filed with the Securities and
Exchange Commission. Readers are cautioned not to place undue
reliance on these forward-looking statements, which speak only as
of the date of this release. Actual results could differ materially
from those anticipated or projected in the forward-looking
statements. Tandem undertakes no obligation to update or review any
forward-looking statement in this press release because of new
information, future events, or other factors.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240213214991/en/
Media Contact: 858-366-6900 media@tandemdiabetes.com
Investor Contact: 858-366-6900 IR@tandemdiabetes.com
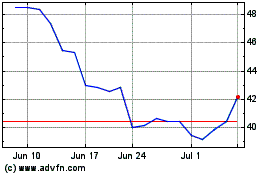
Tandem Diabetes Care (NASDAQ:TNDM)
Historical Stock Chart
From Apr 2024 to May 2024
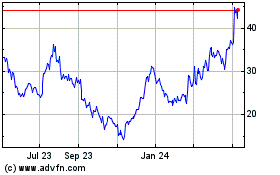
Tandem Diabetes Care (NASDAQ:TNDM)
Historical Stock Chart
From May 2023 to May 2024