We are subject to the reporting requirements of the Exchange Act and
file annual, quarterly and current reports, proxy statements and other information with the SEC. You can read our SEC filings, including
the registration statement of which this prospectus is a part, over the Internet at the SEC’s website at http://www.sec.gov.
We also maintain a website at http://www.citiuspharma.com, at which you may access these materials free of charge as soon as reasonably
practicable after they are electronically filed with, or furnished to, the SEC. The information contained in, or that can be accessed
through, our website is not part of this prospectus. You may also request a copy of these filings, at no cost, by writing or telephoning
us at: 11 Commerce Drive, First Floor, Cranford, New Jersey 07016, (908) 967-6677.
INCORPORATION OF DOCUMENTS BY REFERENCE
The SEC allows us to “incorporate by reference” information
that we file with them. Incorporation by reference allows us to disclose important information to you by referring you to those other
documents. The information incorporated by reference is an important part of this prospectus and any applicable accompanying prospectus,
and information that we file later with the SEC will automatically update and supersede this information. We filed a registration statement
on Form S-3 under the Securities Act with the SEC with respect to the securities being offered pursuant to this prospectus and any
applicable accompanying prospectus. This prospectus omits certain information contained in the registration statement, as permitted by
the SEC. You should refer to the registration statement, including the exhibits, for further information about us and the securities being
offered pursuant to this prospectus and any applicable accompanying prospectus. Statements in this prospectus and any applicable accompanying
prospectus regarding the provisions of certain documents filed with, or incorporated by reference in, the registration statement are not
necessarily complete, and reference is made to the actual documents for complete information. Copies of all or any part of the registration
statement, including the documents incorporated in therein by reference or the exhibits, may be obtained upon payment of the prescribed
rates at the offices of the SEC listed above in “Where You Can Find Additional Information.” The documents we are incorporating
by reference into this prospectus are:
|
|
●
|
the description of our common stock contained in our Registration Statement on Form 8-A, filed on July 28, 2017;
|
|
|
●
|
our Annual Report on Form 10-K for the fiscal year ended September 30, 2020, filed with the SEC pursuant to Section 13 of the Exchange
Act on December 16, 2020;
|
|
|
●
|
our Quarterly Report on Form 10-Q for the quarter ended December 31, 2020, filed with the SEC pursuant to Section 13 of the Exchange
Act on February 11, 2021;
|
|
|
●
|
our Current Reports on Form 8-K, filed with the SEC pursuant to Section 13 of the Exchange Act on October 9, October 26, November 30, December 8 and December 9, 2020, and January 11, January 27, February 9, February 16 (but not Item 7.01) and February 19, 2021; and
|
|
|
●
|
our definitive proxy statement on Schedule 14A for the annual meeting of stockholders held on February 9, 2021, filed with the SEC
pursuant to Section 14 of the Exchange Act on December 21, 2020.
|
In addition, all documents subsequently filed by us pursuant to Section
13(a), 13(c), 14 or 15(d) of the Exchange Act before the date any offering is terminated or completed are deemed to be incorporated by
reference into, and to be a part of, this prospectus, provided that that we are not incorporating by reference any information furnished
to, but not filed with, the SEC.
Any statement contained in this prospectus and any applicable accompanying
prospectus or in a document incorporated or deemed to be incorporated by reference into this prospectus and any applicable accompanying
prospectus will be deemed to be modified or superseded for purposes of this prospectus and any applicable accompanying prospectus to the
extent that a statement contained in this prospectus and any applicable accompanying prospectus or any other subsequently filed document
that is deemed to be incorporated by reference into this prospectus and any applicable accompanying prospectus modifies or supersedes
the statement. Any statement so modified or superseded will not be deemed, except as so modified or superseded, to constitute a part of
this prospectus and any applicable accompanying prospectus.
We will furnish without charge to you, on written or oral request,
a copy of any or all of the documents incorporated by reference in the registration statement and this prospectus, including exhibits
to these documents. You should direct any requests for documents to Citius Pharmaceuticals, Inc., Attention: Secretary, 11 Commerce Drive,
1st Floor, Cranford, New Jersey 07016, (908) 967-6677.
You should rely only on information contained in, or incorporated by
reference into, this prospectus and any applicable accompanying prospectus. We have not authorized anyone to provide you with information
different from that contained in this prospectus and any applicable accompanying prospectus or incorporated by reference in this prospectus
and any applicable accompanying prospectus. We are not making offers to sell the securities in any jurisdiction in which such an offer
or solicitation is not authorized or in which the person making such offer or solicitation is not qualified to do so or to anyone to whom
it is unlawful to make such offer or solicitation.
The information in this prospectus is not
complete and may be changed. The selling stockholders may not sell these securities or accept an offer to buy these securities until
the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these
securities, and we are not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
Subject to completion,
dated April 2, 2021
Prospectus

3,558,140 Shares of Common Stock Offered by
Selling Stockholders
This prospectus relates to the sale or other disposition from time
to time of up to 3,558,140 shares of our common stock, $0.001 par value per share, issuable upon the exercise of warrants held by the
selling stockholders named in this prospectus, including their transferees, pledgees, donees or successors. We are not selling any shares
of common stock under this prospectus and will not receive any of the proceeds from the sale of shares of common stock by the selling
stockholders.
The selling stockholders may sell or otherwise dispose of the shares
of common stock covered by this prospectus in a number of different ways and at varying prices. We provide more information about how
the selling stockholders may sell or otherwise dispose of their shares of common stock in the section entitled “Plan of Distribution”
beginning on page 15. The selling stockholders will pay all brokerage fees and commissions and similar expenses. We will pay all expenses
(except brokerage fees and commissions and similar expenses) relating to the registration of the shares with the Securities and Exchange
Commission. No underwriter or other person has been engaged to facilitate the sale of shares of our common stock in this offering.
Investing in our securities involves a high degree of risk. See
“Risk Factors” beginning on page 12 of this prospectus, in any accompanying prospectus supplement and in the documents
incorporated by reference into this prospectus or any accompanying prospectus supplement, to read about factors you should consider before
investing in our securities.
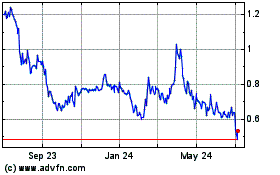
Our common stock is listed on the Nasdaq Capital Market under the symbol
“CTXR”. The last reported sale price of our common stock on March 31, 2021 was $1.78 per share. We recommend that you obtain
current market quotations for our common stock prior to making an investment decision.
Neither the Securities and Exchange Commission nor any other regulatory
body has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus. Any representation to
the contrary is a criminal offense.
The date of this prospectus is , 2021
TABLE OF CONTENTS
ABOUT THIS
PROSPECTUS
You should rely only on the information
that we have provided or incorporated by reference in this prospectus and any prospectus supplement that we may authorize to be provided
to you. We have not, and the selling stockholders have not, authorized anyone to provide you with different information. No dealer, salesperson
or other person is authorized to give any information or to represent anything not contained in this prospectus or any prospectus supplement
that we may authorize to be provided to you. If anyone provides you with different or inconsistent information, you should not rely on
it. You should assume that the information in this prospectus and any prospectus supplement or incorporated herein or therein is accurate
only as of the date on the cover of the document and that any information included in this prospectus or any prospectus supplement or
that we have incorporated by reference herein or therein is accurate only as of the date of the such document, regardless of the time
of delivery of this prospectus or any prospectus supplement or any sale of a security. Our business, financial condition, results of operations
and prospects may have changed since those dates.
We urge you to carefully read this
prospectus and any prospectus supplement, together with the information incorporated herein or therein by reference as described under
the heading “Where You Can Find Additional Information” and “Incorporation of Documents by Reference.”
Unless the context otherwise requires,
we use the terms “Citius”, “the Company”, “our company”, “we”, “us”, and “our”
in this prospectus to refer to the consolidated operations of Citius Pharmaceuticals, Inc. and its consolidated subsidiaries as a whole.
We own or have rights to various
U.S. federal trademark registrations and applications, and unregistered trademarks and servicemarks, including Mino-Lok®. All other
trade names, trademarks and service marks appearing in this prospectus are the property of their respective owners. We have assumed that
the reader understands that all such terms are source-indicating. Accordingly, such terms, when first mentioned in this prospectus, appear
with the trade name, trademark or service mark notice and then throughout the remainder of this prospectus without trade name, trademark
or service mark notices for convenience only and should not be construed as being used in a descriptive or generic sense.
SPECIAL NOTE
REGARDING FORWARD-LOOKING STATEMENTS AND INDUSTRY DATA
This prospectus contains forward-looking statements that
are based on our management’s belief and assumptions and on information currently available to our management. Although we believe
that the expectations reflected in these forward-looking statements are reasonable, these statements relate to future events or our future
financial performance, and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels
of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements
expressed or implied by these forward-looking statements. Forward-looking statements in this prospectus include, but are not limited to,
statements about:
|
|
●
|
our need for, and ability to raise, additional capital;
|
|
|
●
|
the number, designs, timing, costs and results of our pre-clinical and clinical trials;
|
|
|
●
|
the regulatory review process and any regulatory approvals that may be issued or denied by the U.S. Food and Drug Administration or
other regulatory agencies;
|
|
|
●
|
the commercial success and market acceptance of any of our product candidates that are approved for marketing in the United States
or other countries;
|
|
|
●
|
the accuracy of our estimates and of third-party estimates of the size and characteristics of the markets that may be addressed by
our product candidates;
|
|
|
●
|
our ability to manufacture sufficient amounts of our product candidates for clinical trials and, if approved, our products for commercialization
activities;
|
|
|
●
|
our need to secure collaborators to license, manufacture, market and sell any products for which we receive regulatory approval;
|
|
|
●
|
our ability to protect our intellectual property and operate our business without infringing upon the intellectual property rights
of others;
|
|
|
●
|
the medical benefits, effectiveness and safety of our product candidates;
|
|
|
●
|
the safety and efficacy of medicines or treatments introduced by competitors that are targeted to indications for which our product
candidates are being developed;
|
|
|
●
|
our current or prospective collaborators’ compliance or non-compliance with their obligations under our agreements with them;
|
|
|
●
|
the impact of the COVID-19 pandemic on our clinical trials, business and operations; and
|
|
|
●
|
other factors discussed elsewhere in this prospectus or incorporated by reference herein.
|
In some cases, you can identify forward-looking statements
by terminology such as “may”, “will”, “should”, “expects”, “intends”, “plans”,
“anticipates”, “believes”, “estimates”, “predicts”, “potential”, “continue”
or the negative of these terms or other comparable terminology. These statements are only predictions. You should not place undue reliance
on forward-looking statements because they involve known and unknown risks, uncertainties and other factors, which are, in some cases,
beyond our control and which could materially affect results. Factors that may cause actual results to differ materially from current
expectations include, among other things, those listed under “Risk Factors” and elsewhere in this prospectus or incorporated
by reference herein. Actual events or results may vary significantly from those implied or projected by the forward-looking statements.
No forward-looking statement is a guarantee of future performance. You should read this prospectus and the documents that we incorporate
by reference in this prospectus and have filed with the SEC as exhibits to this prospectus completely and with the understanding that
our actual future results may be materially different from any future results expressed or implied by these forward-looking statements.
The forward-looking statements in this prospectus or incorporated herein by reference represent our views as of the date of this prospectus
or the document incorporated by reference herein. We anticipate that subsequent events and developments will cause our views to change.
However, while we may elect to update these forward-looking statements at some point in the future, we have no current intention of doing
so except to the extent required by applicable law. You should therefore not rely on these forward-looking statements as representing
our views as of any date subsequent to the date of this prospectus.
This prospectus and the documents incorporated by reference
into this prospectus contain “forward-looking statements” that involve risks and uncertainties, as well as assumptions that,
if they never materialize or prove incorrect, could cause our results to differ materially from those expressed or implied by such forward-looking
statements. The statements contained in this prospectus and the documents incorporated by reference into this prospectus that are not
purely historical are forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, or Securities
Act, and Section 21E of the Securities Exchange Act of 1934, as amended, or Exchange Act.
This prospectus, the documents incorporated by reference
into this prospectus and the documents that we have filed as exhibits to the registration statement, of which this prospectus is a part,
includes statistical and other industry and market data that we obtained from industry publications and research, surveys and studies
conducted by third parties. Industry publications and third-party research, surveys and studies generally indicate that their information
has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information.
We believe that the data obtained from these industry publications and third-party research, surveys and studies are reliable. We are
ultimately responsible for all disclosure included in this prospectus.
You should rely only on the information contained in this
prospectus, as supplemented and amended. We have not authorized anyone to provide you with information that is different. This prospectus
may only be used where it is legal to sell these securities. The information in this prospectus may only be accurate on the date of this
prospectus.
In addition, projections, assumptions, and estimates of our
future performance and the future performance of the industry in which we operate are necessarily subject to a high degree of uncertainty
and risk due to a variety of factors, including those described in “Risk Factors”. These and other factors could cause results
to differ materially from those expressed in the estimates made by the independent parties and by us.
THE
COMPANY
Overview
Citius
Pharmaceuticals, Inc., headquartered in Cranford, New Jersey, is a specialty pharmaceutical company dedicated to the development
and commercialization of critical care products targeting important medical needs with a focus on anti-infective products in adjunct
cancer care, unique prescription products and, recently, mesenchymal stem cell therapy. Our goal generally is to achieve leading
market positions by providing therapeutic products that address unmet medical needs yet have a lower development risk than usually
is associated with new chemical entities. New formulations of previously approved drugs with substantial existing safety and efficacy
data are a core focus. We seek to reduce development and clinical risks associated with drug development, yet still focus on innovative
applications. Our strategy centers on products that have intellectual property and regulatory exclusivity protection, while providing
competitive advantages over other existing therapeutic approaches.
Mino-Lok
Mino-Lok
is a patented solution containing minocycline, disodium ethylenediaminetetraacetic acid (edetate), and ethyl alcohol, all of which
act synergistically to treat and salvage infected central venous catheters (“CVCs”) in patients with catheter related
bloodstream infections (“CRBSIs”). Mino-Lok breaks down biofilm barriers formed by bacterial colonies, eradicates
the bacteria, and provides anti-clotting properties to maintain patency in CVCs.
The
administration of Mino-Lok consists of filling the lumen of the catheter with 0.8 ml to 2.0 ml of Mino-Lok solution. The catheter
is then “locked”, meaning that the solution remains in the catheter without flowing into the vein. The lock is maintained
for a dwell-time of two hours while the catheter is not in use. If the catheter has multiple lumens, all lumens may be locked
with the Mino-Lok solution either simultaneously or sequentially. If patients are receiving continuous infusion therapy, the catheters
alternate between being locked with the Mino-Lok solution and delivering therapy. The Mino-Lok therapy is two hours per day for
at least five days, usually with two additional locks in the subsequent two weeks. After locking the catheter for two hours, the
Mino-Lok solution is aspirated, and the catheter is flushed with normal saline. At that time, either the infusion will be continued,
or will be locked with the standard-of-care lock solution until further use of the catheter is required. In a clinical study conducted
by MD Anderson Cancer Center (“MDACC”), there were no serum levels of either minocycline or edetate detected in the
sera of several patients who underwent daily catheter lock solution with minocycline and edetate (“M-EDTA”) at the
concentration level proposed in the Mino-Lok treatment. Thus, it has been demonstrated that the amount of either minocycline or
edetate that leaks into the serum is very low or none at all.
Phase
2b Results
From
April 2013 to July 2014, 30 patients with CVC-related bloodstream infection were enrolled at MDACC in a prospective Phase 2b study.
Patients received Mino-Lok therapy for two hours once daily for a minimum of five days within the first week, followed by two
additional locks within the next two weeks. Patients were followed for one month post-lock therapy. Demographic information, clinical
characteristics, laboratory data, therapy, as well as adverse events and outcome were collected for each patient. Median age at
diagnosis was 56 years (range: 21-73 years). In all patients, prior to the use of lock therapy, systemic treatment with a culture-directed,
first-line intravenous antibiotic was started. Microbiological eradication was achieved at the end of therapy in all cases. None
of the patients experienced any serious adverse event related to the lock therapy.
The active
arm, which is the Mino-Lok treated group of patients, was then compared to 60 patients in a matched cohort that experienced removal and
replacement of their CVCs within the same contemporaneous timeframe. The patients were matched for cancer type, infecting organism, and
level of neutropenia. All patients were cancer patients and treated at MDACC. The efficacy of Mino-Lok therapy was 100% in salvaging
CVCs, demonstrating equal effectiveness to removing the infected CVC and replacing it with a new catheter.
The main purpose of the study was to show that Mino-Lok therapy was at least as effective as the removal and
replacement of CVCs when CRBSIs are present, and that the safety was better, that is, the complications of removing an infected catheter
and replacing with a new one could be avoided. In addition to having a 100% efficacy rate with all CVCs being salvaged, Mino-Lok therapy
had no significant adverse events (“SAEs”), compared to an 18% SAE rate in the matched cohort where patients had the infected
CVCs removed and replaced with a fresh catheter. There were no overall complication rates in the Mino-Lok arm group compared to 11 patients
with events (18)% in the control group. These events included bacterial relapse (5)% at four weeks post-intervention, and a number of
complications associated with mechanical manipulation in the removal or replacement procedure for the catheter (10)% or development of
deep-seated infections such as septic thrombophlebitis and osteomyelitis (8)%. As footnoted, six patients had more than one complication
in the control arm group.
|
|
|
Mino-Lok Arm
|
|
|
Control Arm
|
|
|
Parameter
|
|
N
|
|
|
(%)
|
|
|
N
|
|
|
(%)
|
|
|
Patients
|
|
|
30
|
|
|
|
(100
|
)%
|
|
|
60
|
|
|
|
(100
|
)%
|
|
Cancer type
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- Hematologic
|
|
|
20
|
|
|
|
(67
|
)
|
|
|
48
|
|
|
|
(80
|
)
|
|
- Solid tumor
|
|
|
10
|
|
|
|
(33
|
)
|
|
|
12
|
|
|
|
(20
|
)
|
|
ICU Admission
|
|
|
4
|
|
|
|
(13
|
)
|
|
|
4
|
|
|
|
(7
|
)
|
|
Mech. Ventilator
|
|
|
3
|
|
|
|
(10
|
)
|
|
|
0
|
|
|
|
(0
|
)
|
|
Bacteremia
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- Gram+
|
|
|
17
|
|
|
|
(57
|
)*
|
|
|
32
|
|
|
|
(53
|
)
|
|
- Gram-
|
|
|
14
|
|
|
|
(47
|
)*
|
|
|
28
|
|
|
|
(47
|
)
|
|
Neutropenia (<500)
|
|
|
19
|
|
|
|
(63
|
)
|
|
|
36
|
|
|
|
(60
|
)
|
|
Microbiologic Eradication
|
|
|
30
|
|
|
|
(100
|
)
|
|
|
60
|
|
|
|
(100
|
)
|
|
- Relapse
|
|
|
0
|
|
|
|
(0
|
)
|
|
|
3
|
|
|
|
(5
|
)
|
|
Complications
|
|
|
0
|
|
|
|
(0
|
)
|
|
|
8
|
|
|
|
(13
|
)
|
|
SAEs related to R&R
|
|
|
0
|
|
|
|
(0
|
)
|
|
|
6
|
|
|
|
(10
|
)
|
|
Overall Complication
Rate
|
|
|
0
|
|
|
|
(0
|
)%
|
|
|
11
|
**
|
|
|
(18
|
)%
|
|
|
*
|
1
polymicrobial patient had a Gram+ and a Gram- organism cultured
|
|
|
**
|
6
patients had > 1 complication
|
Source:
Dr. Issam Raad, Antimicrobial Agents and Chemotherapy, June 2016, Vol. 60 No. 6, Page 3429
Phase
3 Trial
In
November 2016, the Company initiated site recruitment for Phase 3 clinical trials. From initiation through the first quarter of
2017, the Company received input from several sites related to the control arm as being less than standard-of-care for some of
the respective institutions. The Company worked closely with the U.S. Food and Drug Administration (“FDA”) with respect
to the design of the Phase 3 trial and received feedback on August 17, 2017. The FDA stated that they recognized that there is
an unmet medical need in salvaging infected catheters and agreed that an open label, superiority design would address the Company’s
concerns and would be acceptable to meet the requirements of a new drug application. The Company amended the Phase 3 study design
to remove the saline and heparin placebo control arm and to use an active control arm that conforms with today’s current
standard-of-care. Patient enrollment commenced in February 2018.
The
Mino-Lok Phase 3 trial was originally planned to enroll 700 patients in 50 participating institutions, all located in the U.S.
There will be interim analyses at both the 50% and 75% points of the trial as measured by the number of patients treated. As of
March 15, 2021, there are 25 active sites currently enrolling patients including such academic centers as MDACC, Henry Ford Health
Center, Georgetown University Medical Center, and others. There is one additional medical center in startup mode. There are no
other remaining sites in feasibility.
In
September 2019, the Company announced that the FDA agreed to a new primary efficacy endpoint of “time to catheter failure”
in comparing Mino-Lok to the antibiotic lock control arm. This change in the trial design reduced the required patient sample
size of the trial from 700 subjects to approximately 144 available subjects to achieve the pre-specified 92 catheter failure events
needed to conclude the trial. Additionally, the Company submitted a response to the FDA that it will implement this change in
the primary endpoint and expected it to result in less than 150 subjects needed in its Phase 3 trial. The new primary endpoints
require that the time to catheter failure be at least 38 days for Mino-Lok versus 21 days for the standard of care antibiotic
locks.
In
October 2019, the FDA agreed that the patient sample size of approximately 144 patients was acceptable.
In
October 2019, the Company announced that the Phase 3 trial had reached the 40% completion triggering an interim futility analysis
by the data monitoring committee (the “DMC”). The DMC is an independent panel of experts that review progress regarding
the safety and efficacy of drugs in clinical trials, and to determine if the trial may be futile in achieving its endpoints or
if the trial should be modified in any way.
In
December 2019, the DMC convened and recommended that the trial continue with no changes because the analysis showed a positive
outcome, as it met the prespecified interim futility analysis criteria.
In
May 2020, we announced that we are providing free access to Mino-Lok for healthcare providers under an Expanded Access protocol
to ease the burden associated with the COVID-19 pandemic. Through the Expanded Access protocol, an infected central venous catheter
can now be treated with Mino-Lok, potentially avoiding the need for the removal and replacement procedure.
In
June 2020, we announced that we had received positive feedback from the FDA on our proposed catheter compatibility studies for
Mino-Lok. The studies, if and when successfully completed, should allow Mino-Lok to be labeled for use with all commercially available
CVCs and peripherally inserted central catheters (PICCs) on the U.S. market. It is further assumed that these studies will meet
European and world standards. The ability to be labeled without restrictions with respect to catheter type would allow Mino-Lok
unrestricted access to the full U.S. and world markets for an effective antibiotic lock therapy for central line associated blood
stream infections (“CLABSIs”).
In
September 2020, we announced that another DMC meeting was held to review the data being generated and analyzed in the Mino-Lok
Phase 3 trial based on progress to date, and to make recommendations to us as to any action that may be necessary regarding the
study. After reviewing these data, the DMC members stated that they did not find any safety signals; and they also recommended
continuing the trial without any modifications. The DMC further conducted an ad hoc meeting and agreed with the
Company that a 75% interim analysis be conducted as planned in which superior efficacy is evaluated. Due to the COVID-19 pandemic,
the interim analysis will be performed at the 65% threshold. To counter the impact of COVID-19, we have aggressively pursued outreach
programs with webinars and other remote communications, and have now been able to add randomized patients bringing us closer to
the number of events for the “superiority” analysis and to schedule a meeting with the DMC, both expected in the second
quarter of 2021.
In
September 2020 the Company announced that the three registration batches for all components of Mino Lok were manufactured and
that clinical sites were resupplied with registration product.
In
November 2020, the Company announced that the three components of Mino-Lok, minocycline, disodium edetate (“EDTA”),
and ethanol, were superior to EDTA and ethanol in their ability to eradicate resistant staphylococcal biofilms.
Fast
Track Designation
In
October 2017, the Company received official notice from the FDA that the investigational program for Mino-Lok was granted “Fast
Track” status. Fast Track is a designation that expedites FDA review to facilitate development of drugs which treat a serious
or life-threatening condition and fill an unmet medical need. A drug that receives Fast Track designation is eligible for the
following:
|
|
●
|
More
frequent meetings with the FDA to discuss the drug’s development plan and ensure collection of appropriate data needed to
support drug approval;
|
|
|
●
|
More
frequent written correspondence from the FDA about the design of the clinical trials;
|
|
|
●
|
Priority
review to shorten the FDA review process for a new drug from ten months to six months; and
|
|
|
●
|
Rolling
review, which means Citius can submit completed sections of its New Drug Application (“NDA”) for review by the FDA,
rather than waiting until every section of the application is completed before the entire application can be reviewed.
|
Mino-Lok
International Study
In
October 2017, data from an international study on Mino-Lok was presented at the Infectious Disease Conference (“ID Week”)
in San Diego, California. The 44-patient study was conducted in Brazil, Lebanon, and Japan and showed Mino-Lok therapy was an
effective intervention to salvage long-term, infected CVCs in CRBSIs in patients who had cancer with limited vascular access.
This study showed 95% effectiveness for Mino-Lok therapy in achieving microbiological eradication of the CVCs as compared to 83%
for the control. The single failure in the Mino-Lok arm was due to a patient with Burkholderia cepacia that was
resistant to all antibiotics tested.
Stability
Patent Application for Mino-Lok
In
October 2018, the U.S. Patent and Trademark Office (“USPTO”) issued U.S. Patent No. 10,086,114, entitled “Antimicrobial
Solutions with Enhanced Stability.” This invention overcomes limitations in mixing antimicrobial solutions in which components
have precipitated because of physical and/or chemical factors, thus limiting the stability of the post-mix solutions. The scientists
and technologists at MDACC have been able to improve the stability of the post-mixed solutions through adjustments of the post-mixed
pH of the solution. This may allow for longer storage time of the ready-to-use solution. Citius holds the exclusive worldwide
license which provides access to this patented technology for development and commercialization of Mino-Lok.
On
October 9, 2019, the European Patent Office (“EPO”) granted European Patent No. 3370794, entitled “Antimicrobial
Solutions with Enhanced Stability.” The grant of this European patent strengthens the intellectual property protection for
Mino-Lok through November of 2036. This invention overcomes limitations in mixing antimicrobial solutions, in which components
have precipitated because of physical and/or chemical factors, thus limiting the stability of the post-mix solutions. The scientists
and technologists at MDACC have been able to improve the stability of the post-mixed solutions through adjustments of the post-mixed
pH of the solution. This may allow for longer storage time of the ready-to-use solution.
Mino-Wrap
On
January 2, 2019, we entered into a patent and technology license agreement with the Board of Regents of the University of Texas
System on behalf of MDACC, whereby we in-licensed exclusive worldwide rights to the patented technology for any and all uses relating
to breast implants, specifically the Mino-Wrap technology. This includes rights to U.S. Patent No. 9,849,217, which was issued
on December 16, 2017. We intend to develop Mino-Wrap as a liquefying, gel-based wrap containing minocycline and rifampin for the
reduction of infections associated with breast implants following breast reconstructive surgeries. We are required to use commercially
reasonable efforts to commercialize Mino-Wrap under several regulatory scenarios and achieve milestones associated with these
regulatory options leading to an approval from the FDA. Mino-Wrap will require pre-clinical development prior to any regulatory
pathway. In July 2019, we announced that we intend to pursue the FDA’s Investigational New Drug (“IND”) regulatory
pathway for the development of Mino-Wrap. On August 4, 2020, we announced that we had submitted a briefing package to the FDA
for a pre-IND consultation on Mino-Wrap. In December 2020, we reported the FDA response to the briefing package and commented
that the FDA was in general agreement with our planned pre-clinical program and gave further guidance on our clinical plans.
Halo-Lido
Overview
Halo-Lido
is a topical formulation of halobetasol propionate, a corticosteroid and lidocaine that is intended for the treatment of hemorrhoids.
To our knowledge, there are currently no FDA-approved prescription drug products for the treatment of hemorrhoids. Some physicians
are known to prescribe topical steroids for the treatment of hemorrhoids. In addition, there are various topical combination prescription
products containing halobetasol propionate along with lidocaine or pramoxine, each a topical anesthetic, that are prescribed by
physicians for the treatment of hemorrhoids. These products contain drugs that were in use prior to the start of the Drug Efficacy
Study Implementation (“DESI”) program and are commonly referred to as DESI drugs. However, none of these single-agent
or combination prescription products have been clinically evaluated for safety and efficacy and approved by the FDA for the treatment
of hemorrhoids. Further, many hemorrhoid patients use over the counter (“OTC”) products as their first line therapy.
OTC products contain any one of several active ingredients including glycerin, phenylephrine, pramoxine, white petrolatum, shark
liver oil and/or witch hazel, for symptomatic relief.
Development
of Hemorrhoids Drugs
Hemorrhoids
are a common gastrointestinal disorder, characterized by anal itching, pain, swelling, tenderness, bleeding and difficulty defecating.
In the U.S., hemorrhoids affect nearly 5% of the population, with approximately 10 million persons annually admitting to having
symptoms of hemorrhoidal disease. Of these persons, approximately one third visit a physician for evaluation and treatment of
their hemorrhoids. The data also indicate that for both sexes a peak of prevalence occurs from age 45 to 65 years with a subsequent
decrease after age 65 years. Caucasian populations are affected significantly more frequently than African Americans, and increased
prevalence rates are associated with higher socioeconomic status in men but not women. Development of hemorrhoids before age 20
is unusual. In addition, between 50% and 90% of the general U.S., Canadian and European population will experience hemorrhoidal
disease at least once in life. Although hemorrhoids and other anorectal diseases are not life-threatening, individual patients
can suffer from agonizing symptoms which can limit social activities and have a negative impact on the quality of life.
Hemorrhoids
are defined as internal or external according to their position relative to the dentate line. Classification is important for
selecting the optimal treatment for an individual patient. Accordingly, physicians use the following grading system referred to
as the Goligher’s classification of internal hemorrhoids:
|
Grade I
|
Hemorrhoids not prolapsed
but bleeding.
|
|
Grade II
|
Hemorrhoids prolapse and reduce spontaneously
with or without bleeding.
|
|
Grade III
|
Prolapsed hemorrhoids that require reduction
manually.
|
|
Grade IV
|
Prolapsed and cannot be reduced including both
internal and external hemorrhoids that are confluent from skin tag to inner anal canal.
|
Development
Activities to Date
In
the fall of 2015, we completed dosing patients in a double-blind dose ranging placebo controlled Phase 2a study where six different
formulations containing hydrocortisone and lidocaine in various strengths were tested against the vehicle control. The objectives
of this study were to: (1) demonstrate the safety and efficacy of the formulations when applied twice daily for two weeks in subjects
with Grade I or II hemorrhoids, and (2) assess the potential contribution of lidocaine hydrochloride and hydrocortisone acetate,
alone or in combination for the treatment of symptoms of Goligher’s Classification Grade I or II hemorrhoids.
Symptom
improvement was observed based on a global score of disease severity (“GSDS”) and based on some of the individual
signs and symptoms of hemorrhoids, specifically itching and overall pain and discomfort. Within the first few days of treatment,
the combination products (containing both hydrocortisone and lidocaine) were directionally favorable versus the placebo and their
respective individual active treatment groups (e.g., hydrocortisone or lidocaine alone) in achieving ‘almost symptom free’
or ’symptom free’ status according to the GSDS scale. These differences suggest the possibility of a benefit for the
combination product formulation.
Overall,
results from adverse event reporting support the safety profile of all test articles evaluated in this study and demonstrate similar
safety profiles as compared to the vehicle. The safety findings were unremarkable. There was a low occurrence of adverse events
and a similar rate of treatment related adverse events across all treatment groups. The majority of adverse events were mild and
only one was severe. None of the adverse events were an SAE and the majority of adverse events were recovered/resolved at the
end of the study. There were only two subjects who were discontinued from the study due to adverse events.
In
addition to the safety and dose-ranging information, information was obtained relating to the use of the GSDS as an assessment
tool for measuring the effectiveness of the test articles. Individual signs and symptoms were also assessed but can vary from
patient to patient. Therefore, the goal of the GSDS was to provide an assessment tool that could be used for all patients regardless
of which signs and symptoms they are experiencing. The GSDS proved to be a more effective tool for assessing the severity of the
disease and the effectiveness of the drug when compared to the assessment of the individual signs and symptoms. Citius believes
that we can continue to develop this assessment tool as well as other patient reported outcome endpoints for use in the next trials
and in the pivotal trial.
Information
was also obtained about the formulation of the drug and the vehicle. As a result of this study, we believed that the performance
of the active arms of the study relative to the vehicle could be improved by re-formulating our topical preparation. Therefore,
we initiated work on vehicle formulation and evaluation of higher potency steroids.
In
June and July 2016, we engaged the Dominion Group, a leading provider of healthcare and pharmaceutical marketing research services.
The primary market research was conducted to better understand the symptoms that are most bothersome to patients in order to develop
meaningful endpoints for the clinical trials. We also learned about the factors that drive patients to seek medical attention
for hemorrhoids in an effort to understand the disease’s impact on quality of life. The results of this survey are able
to help us develop patient reported outcome evaluation tools. These tools can be used in clinical trials to evaluate the patients’
conditions and to assess the performance of the test articles.
In
March 2018, we announced that we had selected a higher potency corticosteroid in our steroid/anesthetic topical formulation program
for the treatment of hemorrhoids. The original topical preparation, which we referred to as Hydro-Lido or CITI-001, which was
used in the Phase 2a study, was a combination of hydrocortisone acetate and lidocaine hydrochloride. The new formulation, CITI-002,
which we refer to as Halo-Lido, combines lidocaine with the higher potency corticosteroid halobetasol propionate for symptomatic
relief of the pain and discomfort of hemorrhoids.
We
held a Type C meeting with the FDA in December 2017 to discuss the results of the Phase 2a study and to obtain the FDA’s
view on development plans to support the potential formulation change for the planned Phase 2b study. We also requested the FDA’s
feedback on our Phase 2b study design, including target patient population, inclusion/exclusion criteria, and efficacy endpoints.
The pre-clinical and clinical development programs for CITI-002 are planned to be similar to those conducted for the development
of CITI-001 to support the design for a planned Phase 3 clinical trial. We anticipate beginning a Phase 2b clinical study in the
third quarter of 2021.
NoveCite
Overview
In
October 2020, we, through our recently formed subsidiary, NoveCite, signed an exclusive agreement with Novellus Therapeutics Limited
(“Novellus”) to license iPSC-derived mesenchymal stem cells (iMSCs). Under this worldwide exclusive license, we will
be focused on developing cellular therapies. Specifically, we will seek to develop and commercialize the NoveCite mesenchymal
stem cells (“NC-iMSCs”) to treat acute respiratory conditions with a near term focus on ARDS associated with
COVID-19.
NC-iMSCs
are the next generation mesenchymal stem cell therapy. They are believed to be differentiated and superior to donor-derived MSCs.
Human donor-derived MSCs are sourced from human bone marrow, adipose tissue, placenta, umbilical tissue, etc. and have significant
challenges (e.g., variable donor and tissue sources, limited supply, low potency, inefficient and expensive manufacturing). NC-iMSCs
overcome these challenges because they:
|
|
●
|
Are
more potent and secrete exponentially higher levels of immunomodulatory proteins;
|
|
|
●
|
Have
practically unlimited supply for high doses and repeat doses;
|
|
|
●
|
Are
from a single donor and clonal so they are economically produced at scale with consistent quality and potency, as well as being
footprint free (compared to viral reprogramming methods); and
|
|
|
●
|
Have
a significantly higher expansion capability.
|
Several
cell therapy companies using donor-derived MSC therapies in treating ARDS have demonstrated that MSCs reduce inflammation, enhance
clearance of pathogens and stimulate tissue repair in the lungs. Almost all these positive results are from early clinical trials
or under the emergency authorization program.
In
December 2020, the Company announced interim data from a proof-of-concept large animal study of its NC-iMSC therapy for
acute inflammatory respiratory conditions including COVID-19 related ARDS. The available results of NC-iMSC therapy in
the study show improvement in critical parameters, such as improved oxygenation, less systemic shock, and reduced lung injury,
compared to the control group. The study was conducted in a widely accepted large animal model.
The
Company will complete additional pre-clinical studies throughout 2021 and anticipates filing an IND by the end of the second quarter
of 2022.
Corporate
History and Information
We
were founded as Citius Pharmaceuticals, LLC, a Massachusetts limited liability company, on January 23, 2007. On September 12,
2014, Citius Pharmaceuticals, LLC entered into a Share Exchange and Reorganization Agreement, with Citius Pharmaceuticals, Inc.
(formerly Trail One, Inc.), a publicly traded company incorporated under the laws of the State of Nevada. Citius Pharmaceuticals,
LLC became a wholly-owned subsidiary of Citius. On March 30, 2016, Citius acquired Leonard-Meron Biosciences, Inc. (“LMB”)
as a wholly-owned subsidiary. LMB was a pharmaceutical company focused on the development and commercialization of critical care
products with a concentration on anti-infectives. On September 11, 2020, we formed NoveCite, Inc. (“NoveCite”), a
Delaware corporation, of which we own 75% of the issued and outstanding capital stock.
Our
principal executive offices are located at 11 Commerce Drive, First Floor, Cranford, New Jersey 07016 and our telephone number
is (908) 967-6677.
THE
OFFERING
Up
to 3,558,140 Shares of Common Stock
This
prospectus relates to the resale by the selling stockholders identified in this prospectus of up to 3,558,140 shares of our common
stock issuable upon exercise of warrants issued in February 2021 to the placement agent of our registered direct offering, with
an exercise price of $1.881 per share that expire on February 16, 2026.
|
Common stock offered by
the selling stockholders
|
|
3,558,140 shares
|
|
Common stock outstanding
before the offering (1)
|
|
134,701,219 shares
|
|
Common stock to be outstanding after
the offering
|
|
138,259,359 shares
|
|
Common stock Nasdaq
Capital Market Symbol
|
|
CTXR
|
|
|
(1)
|
Based
on the number of shares outstanding as of March 31, 2021.
|
Use
of Proceeds
The
3,558,140 shares of common stock issuable upon the exercise of currently outstanding warrants that are being offered for resale
by the selling stockholders will be sold for the accounts of the selling stockholders named in this prospectus. As a result, all
proceeds from the sales of the 3,558,140 shares of common stock issuable upon the exercise of currently outstanding warrants and
offered for resale hereby will go to the selling stockholders and we will not receive any proceeds from the resale of those shares
of common stock by the selling stockholders.
We
may receive up to a total of $6,692,861 in gross proceeds if all of the warrants are exercised hereunder for cash. However, as
we are unable to predict the timing or amount of potential exercises of the warrants, we have not allocated any proceeds of such
exercises to any particular purpose. Accordingly, all such proceeds are allocated to working capital. Pursuant to conditions set
forth in the warrants, the warrants are exercisable under certain circumstances on a cashless basis, and should a selling stockholder
elect to exercise on a cashless basis we will not receive any proceeds from the sale of common stock issued upon the cashless
exercise of the warrant.
On
the termination date of the warrants, any warrant outstanding and unexercised on its termination date, and for which the exercise
price on that day is less than the then market price of our common stock, will be automatically exercised via cashless exercise
as provided in the warrants. In such event, we will not receive any cash proceeds.
We
will incur all costs associated with this registration statement and prospectus.
Dividend
Policy
We
have never paid dividends on our capital stock and do not anticipate paying any dividends for the foreseeable future.
Risk
Factors
Investing
in our common stock involves a high degree of risk. Please read the information contained under the heading “Risk Factors”
beginning on page 12 of this prospectus and in any subsequent report incorporated by reference herein.
RISK
FACTORS
Investing
in our securities involves a high degree of risk. You should consider carefully the risks and uncertainties described in “Risk
Factors” in our most recently filed Annual Report on Form 10-K filed with the SEC, in each case as these risk factors are
amended or supplemented by subsequent Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, or Current Reports on Form
8-K that have been or will be incorporated by reference in this prospectus. The risks incorporated herein by reference are those
which we believe are the material risks that we face. The occurrence of any of such risks may materially and adversely affect
our business, financial condition, results of operations and future prospects. In such an event, the market price of our common
stock could decline, and you could lose part or all of your investment.
USE
OF PROCEEDS
The
3,558,140 shares of common stock issuable upon the exercise of currently outstanding warrants and that are being offered for resale
by the selling stockholders will be sold for the accounts of the selling stockholders named in this prospectus. As a result, all
proceeds from the sales of the 3,558,140 shares of common stock issuable upon the exercise of currently outstanding warrants and
offered for resale hereby will go to the selling stockholders and we will not receive any proceeds from the resale of those shares
of common stock by the selling stockholders.
We
may receive up to a total of $6,692,861 in gross proceeds if all of the warrants are exercised hereunder for cash. However, as
we are unable to predict the timing or amount of potential exercises of the warrants, we have not allocated any proceeds of such
exercises to any particular purpose. Accordingly, all such proceeds are allocated to working capital. Pursuant to conditions set
forth in the warrants, the warrants are exercisable under certain circumstances on a cashless basis, and should a selling stockholder
elect to exercise on a cashless basis we will not receive any proceeds from the sale of common stock issued upon the cashless
exercise of the warrant.
On
the termination date of the warrants, any warrant outstanding and unexercised on its termination date, and for which the exercise
price on that day is less than the then market price of our common stock, will be automatically exercised via cashless exercise
as provided in the warrants. In such event, we will not receive any cash proceeds.
We
will incur all costs associated with this registration statement and prospectus.
SELLING
STOCKHOLDERS
The
following table sets forth certain information regarding the selling stockholders and the shares of common stock beneficially
owned by them, which information is available to us as of March 31, 2021. The selling stockholders may offer the shares under
this prospectus from time to time and may elect to sell under this prospectus some, all or none of the shares offered for resale
by this prospectus. However, for the purposes of the table below, we have assumed that, after completion of the offering, none
of the shares covered by this prospectus will be held by the selling stockholders. In addition, a selling stockholder may have
sold, transferred or otherwise disposed of all or a portion of that holder’s shares of common stock since the date on which
the selling stockholder provided information for this table. We have not made independent inquiries about such transfers or dispositions.
See the section entitled “Plan of Distribution” beginning on page 15.
Beneficial
ownership is determined in accordance with Rule 13d-3(d) promulgated by the SEC under the Exchange Act. The percentage of
shares beneficially owned prior to the offering is based on 134,701,219 shares of our common stock outstanding as of March 31,
2021.
|
|
|
Number
of Shares of Common Stock Beneficially Owned
|
|
|
|
|
|
Number
of Share of Common
|
|
|
Shares
of Common Stock Beneficially Owned After Sale of All Shares of Common Stock Pursuant to this Prospectus
|
|
|
Selling Stockholder
|
|
Before
Any Sale
|
|
|
%
of
Class
|
|
|
Stock
Offering
|
|
|
Number
of Shares
|
|
|
%
of
Class
|
|
|
Noam Rubinstein (1)
|
|
2,374,012
|
(2)
|
|
1.73
|
%
|
|
1,120,814
|
|
|
1,253,198
|
(2)
|
|
*
|
|
|
Michael Vasinkevich (1)
|
|
4,345,241
|
(2)
|
|
3.13
|
%
|
|
2,281,657
|
|
|
2,063,584
|
(2)
|
|
1.51
|
%
|
|
Craig Schwabe (1)
|
|
194,917
|
(2)
|
|
*
|
|
|
120,087
|
|
|
74,830
|
(2)
|
|
*
|
|
|
Charles Worthman (1)
|
|
74,494
|
(2)
|
|
*
|
|
|
35,582
|
|
|
38,912
|
(2)
|
|
*
|
|
|
TOTAL
|
|
6,988,664
|
|
|
4.93
|
%
|
|
3,558,140
|
|
|
3,430,524
|
|
|
2.48
|
%
|
|
|
*
|
Represents
beneficial ownership of less than one percent of the outstanding shares of our common
stock.
|
|
|
|
|
|
(1)
|
The
selling stockholder is an affiliate of a registered broker-dealer.
|
|
|
|
|
|
(2)
|
Consists
of warrants to purchase shares of common stock.
|
Information
about any other selling stockholders will be included in prospectus supplements or post-effective amendments, if required. Information
about the selling stockholders may change from time to time. Any changed information with respect to which we are given notice
will be included in a prospectus supplement.
PLAN
OF DISTRIBUTION
The
selling stockholders, which, as used herein, includes donees, pledgees, transferees or other successors-in-interest selling shares
of common stock or interests in shares of common stock received after the date of this prospectus from a selling stockholder as
a gift, pledge, partnership distribution or other transfer, may, from time to time, sell, transfer or otherwise dispose of any
or all of their shares of common stock or interests in shares of common stock on any stock exchange, market or trading facility
on which the shares are traded or in private transactions. These dispositions may be at fixed prices, at prevailing market prices
at the time of sale, at prices related to the prevailing market price, at varying prices determined at the time of sale, or at
negotiated prices.
The
selling stockholders may use any one or more of the following methods when disposing of shares or interests therein:
|
|
●
|
ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers;
|
|
|
●
|
block
trades in which the broker-dealer will attempt to sell the shares as agent, but may position
and resell a portion of the block as principal to facilitate the transaction;
|
|
|
●
|
purchases
by a broker-dealer as principal and resale by the broker-dealer for its account;
|
|
|
●
|
an
exchange distribution in accordance with the rules of the applicable exchange;
|
|
|
●
|
privately
negotiated transactions;
|
|
|
●
|
short
sales effected after the date the registration statement of which this prospectus is
a part is declared effective by the SEC;
|
|
|
●
|
through
the writing or settlement of options or other hedging transactions, whether through an
options exchange or otherwise;
|
|
|
●
|
broker-dealers
may agree with the selling stockholders to sell a specified number of such shares at
a stipulated price per share;
|
|
|
●
|
a
combination of any such methods of sale; and
|
|
|
●
|
any
other method permitted by applicable law.
|
The
selling stockholders may, from time to time, pledge or grant a security interest in some or all of the shares of common stock
owned by them and, if they default in the performance of their secured obligations, the pledgees or secured parties may offer
and sell the shares of common stock, from time to time, under this prospectus, or under an amendment to this prospectus under
Rule 424(b)(3) or other applicable provision of the Securities Act amending the list of selling stockholders to include the pledgee,
transferee or other successors-in-interest as selling stockholders under this prospectus. The selling stockholders also may transfer
the shares of common stock in other circumstances, in which case the transferees, pledgees or other successors-in-interest will
be the selling beneficial owners for purposes of this prospectus.
In
connection with the sale of our common stock or interests therein, the selling stockholders may enter into hedging transactions
with broker-dealers or other financial institutions, which may in turn engage in short sales of the common stock in the course
of hedging the positions they assume. The selling stockholders may also sell shares of our common stock short and deliver these
securities to close out their short positions, or loan or pledge the common stock to broker-dealers that in turn may sell these
securities. The selling stockholders may also enter into option or other transactions with broker-dealers or other financial institutions
or the creation of one or more derivative securities which require the delivery to such broker-dealer or other financial institution
of shares offered by this prospectus, which shares such broker-dealer or other financial institution may resell pursuant to this
prospectus (as supplemented or amended to reflect such transaction).
The
aggregate proceeds to the selling stockholders from the sale of the common stock offered by them will be the purchase price of
the common stock less discounts or commissions, if any. Each of the selling stockholders reserves the right to accept and, together
with their agents from time to time, to reject, in whole or in part, any proposed purchase of common stock to be made directly
or through agents. We will not receive any of the proceeds from this offering. Upon any exercise of the warrants by payment of
cash, however, we will receive the exercise price of the warrants.
The
selling stockholders also may resell all or a portion of the shares in open market transactions in reliance upon Rule 144 under
the Securities Act, provided that they meet the criteria and conform to the requirements of that rule.
The
selling stockholders and any underwriters, broker-dealers or agents that participate in the sale of the common stock or interests
therein may be “underwriters” within the meaning of Section 2(11) of the Securities Act. Any discounts, commissions,
concessions or profit they earn on any resale of the shares may be underwriting discounts and commissions under the Securities
Act. Selling stockholders who are “underwriters” within the meaning of Section 2(11) of the Securities Act will
be subject to the prospectus delivery requirements of the Securities Act.
To
the extent required, the shares of our common stock to be sold, the names of the selling stockholders, the respective purchase
prices and public offering prices, the names of any agents, dealer or underwriter, any applicable commissions or discounts with
respect to a particular offer will be set forth in an accompanying prospectus supplement or, if appropriate, a post-effective
amendment to the registration statement of which this prospectus is a part.
In
order to comply with the securities laws of some states, if applicable, the common stock may be sold in those jurisdictions only
through registered or licensed brokers or dealers. In addition, in some states the common stock may not be sold unless it has
been registered or qualified for sale or an exemption from registration or qualification requirements is available and is complied
with.
We
have advised the selling stockholders that the anti-manipulation rules of Regulation M under the Exchange Act may apply to sales
of shares in the market and to the activities of the selling stockholders and their affiliates. In addition, to the extent applicable
we will make copies of this prospectus (as it may be supplemented or amended from time to time) available to the selling stockholders
for the purpose of satisfying the prospectus delivery requirements of the Securities Act. The selling stockholders may indemnify
any broker-dealer that participates in transactions involving the sale of the shares against certain liabilities, including liabilities
arising under the Securities Act.
We
will pay all expenses of the registration of the shares of common stock, including, without limitation, SEC filing fees and expenses
of compliance with state securities or “blue sky” laws; provided, however, that each selling stockholder will pay
all underwriting discounts and selling commissions, if any, and any related legal expenses incurred by it. We will indemnify the
selling stockholders against certain liabilities, including some liabilities under the Securities Act, arising in connection with
the registration statement of which this prospectus is a part.
We
have agreed with the selling stockholders to keep the registration statement of which this prospectus is a part effective until
the time that no recipient of the placement agent warrants in the February 2021 private placement owns any such warrants or shares
of common stock issuable upon exercise of the warrants.
DESCRIPTION
OF OUR CAPITAL STOCK
The
following description summarizes the material terms of our capital stock as of the date of this prospectus. Because it is only
a summary, it does not contain all the information that may be important to you. For a complete description of our capital
stock, you should refer to our certificate of incorporation and our bylaws, and to the provisions of applicable Nevada law.
General
Our
authorized capital stock consists of 200,000,000 shares of common stock, par value $0.001, of which 134,701,219 shares were issued
and outstanding as of March 31, 2021, and 10,000,000 shares of preferred stock, none of which are issued and outstanding. We have
proposed to our stockholders that our articles of incorporation be amended to increase the authorized shares of common stock to
400,000,000 shares and this proposed amendment will be submitted to our stockholders for approval at a special meeting of our
stockholders expected to be held in May 2021.
Our
preferred stock and/or common stock may be issued from time to time without prior approval by our stockholders. Our preferred
stock and/or common stock may be issued for such consideration as may be fixed from time to time by our Board of Directors.
Common
Stock
We
are authorized to issue 200,000,000 shares of common stock, $0.001 par value. As noted above, we have proposed to our stockholders
that our articles of incorporation be amended to increase the authorized shares of common stock to 400,000,000 shares and this
proposed amendment will be submitted to our stockholders for approval at a special meeting of our stockholders expected to be
held in May 2021.
Each
share of common stock shall have one vote per share for all purposes. The holders of a majority of the shares entitled to vote,
present in person or represented by proxy shall constitute a quorum at all meetings of our stockholders. Our common stock does
not provide preemptive, subscription or conversion rights and there are no redemption or sinking fund provisions or rights. Our
common stockholders are not entitled to cumulative voting for election of the Board of Directors.
Holders
of common stock are entitled to receive ratably such dividends as may be declared by the Board of Directors out of funds legally
available therefor as well as any distributions to the security holders. We have never paid cash dividends on our common stock,
and do not expect to pay such dividends in the foreseeable future.
In
the event of a liquidation, dissolution or winding up of our company, holders of common stock are entitled to share ratably in
all of our assets remaining after payment of liabilities. Holders of common stock have no preemptive or other subscription or
conversion rights.
Preferred
Stock
We
are authorized to issue 10,000,000 shares of preferred stock. Our Board of Directors is authorized to cause us to issue, from
our authorized but unissued shares of preferred stock, one or more series of preferred stock, to establish from time to time the
number of shares to be included in each such series, as well as to fix the designation and any preferences, conversion and other
rights and limitations of such series. These rights and limitations may include voting powers, limitations as to dividends, and
qualifications and terms and conditions of redemption of the shares of each such series.
Options
As
of December 31, 2020, under the Company’s 2014 Stock Incentive Plan, 2018 Omnibus Stock Incentive Plan and 2020
Omnibus Stock Incentive Plan, we had outstanding options to purchase an aggregate of 4,490,171 shares of our common stock
at a weighted average exercise price of $2.145 per share. Of these, an aggregate of 1,964,638 are exercisable. The remainder has
vesting requirements. No more grants may be made under our 2014 Stock Incentive Plan or our 2018 Omnibus Stock Incentive Plan.
Warrants
As
of December 31, 2020, we had outstanding warrants to purchase an aggregate of 26,751,656 shares of our common stock at a weighted
average price of $1.527 per share, with a weighted average remaining life of 3.30 years.
Trading
Market
The
shares of our common stock are currently listed on the Nasdaq Capital Market under the symbol “CTXR” and certain of
our warrants issued in August 2017 are currently listed on the Nasdaq Capital Market under the symbol “CTXRW”.
Transfer
Agent
The
transfer agent of our common stock is VStock Transfer. Their address is 18 Lafayette Place, Woodmere, NY 11598.
Nevada’s
Anti-Takeover Law and Provisions of Our Articles of Incorporation and Bylaws
Acquisition
of Controlling Interest Statutes. Nevada’s “acquisition of controlling interest” statutes contain provisions
governing the acquisition of a controlling interest in certain Nevada corporations. These “control share” laws provide
generally that any person that acquires a “controlling interest” in certain Nevada corporations may be denied certain
voting rights, unless a majority of the disinterested stockholders of the corporation elects to restore such voting rights. These
statutes provide that a person acquires a “controlling interest” whenever a person acquires shares of a subject corporation
that, but for the application of these provisions of the Nevada Revised Statutes, would enable that person to exercise (1) one-fifth
or more, but less than one-third, (2) one-third or more, but less than a majority or (3) a majority or more, of all of the voting
power of the corporation in the election of directors. Once an acquirer crosses one of these thresholds, shares which it acquired
in the transaction taking it over the threshold and within the 90 days immediately preceding the date when the acquiring person
acquired or offered to acquire a controlling interest become “control shares” to which the voting restrictions described
above apply. Our articles of incorporation and bylaws currently contain no provisions relating to these statutes, and unless our
articles of incorporation or bylaws in effect on the tenth day after the acquisition of a controlling interest were to provide
otherwise, these laws would apply to us if we were to (i) have 200 or more stockholders of record (at least 100 of which have
addresses in the State of Nevada appearing on our stock ledger) and (ii) do business in the State of Nevada directly or through
an affiliated corporation. As of March 31, 2020, we had 96 record stockholders and did not have 100 stockholders of record with
Nevada addresses appearing on our stock ledger. If these laws were to apply to us, they might discourage companies or persons
interested in acquiring a significant interest in or control of the Company, regardless of whether such acquisition may be in
the interest of our stockholders.
Combination
with Interested Stockholders Statutes. Nevada’s “combinations with interested stockholders” statutes prohibit
certain business “combinations” between certain Nevada corporations and any person deemed to be an “interested
stockholder” for two years after such person first becomes an “interested stockholder” unless (i) the corporation’s
Board of Directors approves the combination (or the transaction by which such person becomes an “interested stockholder”)
in advance, or (ii) the combination is approved by the Board of Directors and sixty percent of the corporation’s voting
power not beneficially owned by the interested stockholder, its affiliates and associates. Furthermore, in the absence of prior
approval, certain restrictions may apply even after such two-year period. For purposes of these statutes, an “interested
stockholder” is any person who is (x) the beneficial owner, directly or indirectly, of ten percent or more of the voting
power of the outstanding voting shares of the corporation, or (y) an affiliate or associate of the corporation and at any time
within the two previous years was the beneficial owner, directly or indirectly, of ten percent or more of the voting power of
the then outstanding shares of the corporation. The definition of the term “combination” is sufficiently broad to
cover most significant transactions between the corporation and an “interested stockholder”. Subject to certain timing
requirements set forth in the statutes, a corporation may elect not to be governed by these statutes. We have not included any
such provision in our articles of incorporation.
The
effect of these statutes may be to potentially discourage parties interested in taking control of the Company from doing so if
it cannot obtain the approval of our Board of Directors.
Articles
of Incorporation and Bylaws Provisions of our certificate of incorporation and bylaws may delay or discourage transactions
involving an actual or potential change of control or change in our management, including transactions in which stockholders might
otherwise receive a premium for their shares, or transactions that our stockholders might otherwise deem to be in their best interests.
Therefore, these provisions could adversely affect the price of our common stock. Among other things, these provisions include:
|
|
●
|
the
authorization of 10,000,000 shares of “blank check” preferred stock, the rights, preferences and privileges of which
may be established and shares of which may be issued by our Board of Directors at its discretion from time to time and without
stockholder approval;
|
|
|
●
|
limiting
the removal of directors by the stockholders;
|
|
|
●
|
allowing
for the creation of a staggered Board of Directors;
|
|
|
●
|
eliminating
the ability of stockholders to call a special meeting of stockholders; and
|
|
|
●
|
establishing
advance notice requirements for nominations for election to the Board of Directors or for proposing matters that can be acted
upon at stockholder meetings.
|
LEGAL
MATTERS
The
validity of the securities being offered hereby will be passed upon by Wyrick Robbins Yates & Ponton LLP, Raleigh, North Carolina.
EXPERTS
The
financial statements of Citius Pharmaceuticals, Inc. appearing in our Annual Report on Form 10-K for the fiscal year ended September
30, 2020, have been included herein by reference in reliance on the report of Wolf & Company, P.C., independent registered
public accounting firm, given on the authority of such firm as experts in accounting and auditing.
WHERE
YOU CAN FIND ADDITIONAL INFORMATION
We
are subject to the reporting requirements of the Exchange Act and file annual, quarterly and current reports, proxy statements
and other information with the SEC. You can read our SEC filings, including the registration statement, of which this prospectus
is a part, over the Internet at the SEC’s website at http://www.sec.gov. We also maintain a website at http://www.citiuspharma.com,
at which you may access these materials free of charge as soon as reasonably practicable after they are electronically filed with,
or furnished to, the SEC. The information contained in, or that can be accessed through, our website is not part of this prospectus.
You may also request a copy of these filings, at no cost, by writing or telephoning us at: 11 Commerce Drive, First Floor, Cranford,
New Jersey 07016, (908) 967-6677.
INCORPORATION
OF DOCUMENTS BY REFERENCE
The
SEC allows us to “incorporate by reference” information that we file with them. Incorporation by reference allows
us to disclose important information to you by referring you to those other documents. The information incorporated by reference
is an important part of this prospectus, and information that we file later with the SEC will automatically update and supersede
this information. We filed a registration statement on Form S-3 under the Securities Act with the SEC with respect to
the securities being offered pursuant to this prospectus. This prospectus omits certain information contained in the registration
statement, as permitted by the SEC. You should refer to the registration statement, including the exhibits, for further information
about us and the securities being offered pursuant to this prospectus. Statements in this prospectus regarding the provisions
of certain documents filed with, or incorporated by reference in, the registration statement are not necessarily complete, and
reference is made to the actual documents for complete information. Copies of all or any part of the registration statement, including
the documents incorporated therein by reference or the exhibits, may be obtained upon payment of the prescribed rates at the offices
of the SEC listed above in “Where You Can Find Additional Information.” The documents we are incorporating by reference
into this prospectus are:
|
|
●
|
the
description of our common stock contained in our Registration Statement on Form 8-A, filed on July 28, 2017;
|
|
|
●
|
our
Annual Report on Form 10-K for the fiscal year ended September 30, 2020, filed with the SEC pursuant to Section 13 of the Exchange
Act on December 16, 2020;
|
|
|
●
|
our
Quarterly Report on Form 10-Q for the quarter ended December 31, 2020, filed with the SEC pursuant to Section 13 of the Exchange
Act on February 11, 2021;
|
|
|
●
|
our Current Reports on Form 8-K, filed with the SEC pursuant to Section 13 of the Exchange Act on October 9, October 26, November 30, December 8 and December 9, 2020, and January 11, January 27, February 9, February 16 (but not Item 7.01) and February 19, 2021; and
|
|
|
●
|
our
definitive proxy statement on Schedule 14A for the annual meeting of stockholders held on February 9, 2021, filed with the SEC
pursuant to Section 14 of the Exchange Act on December 21, 2020.
|
In
addition, all documents subsequently filed by us pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act before
the date this offering is terminated or completed are deemed to be incorporated by reference into, and to be a part of, this prospectus,
provided that that we are not incorporating by reference any information furnished to, but not filed with, the SEC.
Any
statement contained in this prospectus or in a document incorporated or deemed to be incorporated by reference into this prospectus
will be deemed to be modified or superseded for purposes of this prospectus to the extent that a statement contained in this prospectus
or any other subsequently filed document that is deemed to be incorporated by reference into this prospectus modifies or supersedes
the statement. Any statement so modified or superseded will not be deemed, except as so modified or superseded, to constitute
a part of this prospectus.
We
will furnish without charge to you, on written or oral request, a copy of any or all of the documents incorporated by reference
in this registration statement or this prospectus, including exhibits to these documents. You should direct any requests for documents
to Citius Pharmaceuticals, Inc., Attention: Secretary, 11 Commerce Drive, 1st Floor, Cranford, New Jersey 07016, (908)
967-6677.
You
should rely only on information contained in, or incorporated by reference into, this prospectus. We have not authorized anyone
to provide you with information different from that contained in this prospectus or incorporated by reference in this prospectus.
We are not making offers to sell the securities in any jurisdiction in which such an offer or solicitation is not authorized or
in which the person making such offer or solicitation is not qualified to do so or to anyone to whom it is unlawful to make such
offer or solicitation.
PART
II
INFORMATION
NOT REQUIRED IN PROSPECTUS
Item
14. Other Expenses of Issuance and Distribution.
The
following table sets forth all costs and expenses paid or payable by us in connection with the sale of the common stock being
registered. None of these costs or expenses will be borne by the selling stockholders.
|
SEC registration fee
|
|
$
|
27,978
|
|
|
Legal fees and expenses
|
|
$
|
15,000
|
*
|
|
Accounting fees and expenses
|
|
$
|
8,000
|
*
|
|
Printing expenses
|
|
$
|
1,500
|
*
|
|
Miscellaneous
|
|
$
|
7,522
|
*
|
|
Total
|
|
$
|
60,000
|
*
|
|
|
*
|
Estimated, as permitted under Item 511 of Regulation S-K.
|
Item
15. Indemnification of Directors and Officers.
Neither
our Articles of Incorporation nor Bylaws prevent us from indemnifying our officers, directors and agents to the extent permitted
under the Nevada Revised Statute (“NRS”). NRS Section 78.751 provides that a corporation shall indemnify any director,
officer, employee or agent of a corporation against expenses, including attorneys’ fees, actually and reasonably incurred
by him or her in connection with any the defense to the extent that a director, officer, employee or agent of a corporation has
been successful on the merits or otherwise in defense of any action, suit or proceeding referred to in Section 78.751, or in defense
of any claim, issue or matter therein.
NRS
78.7502(1) provides that a corporation may indemnify any person who was or is a party or is threatened to be made a party to any
threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, except
an action by or in the right of the corporation, by reason of the fact that he or she is or was a director, officer, employee
or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent
of another corporation, partnership, joint venture, trust or other enterprise, against expenses, including attorneys’ fees, judgments,
fines and amounts paid in settlement actually and reasonably incurred by him or her in connection with the action, suit or proceeding
if he or she: (a) is not liable pursuant to NRS 78.138; or (b) acted in good faith and in a manner which he or she reasonably
believed to be in or not opposed to the best interests of the corporation, and, with respect to any criminal action or proceeding,
had no reasonable cause to believe his or her conduct was unlawful.
NRS
Section 78.7502(2) provides that a corporation may indemnify any person who was or is a party or is threatened to be made a party
to any threatened, pending or completed action or suit by or in the right of the corporation to procure a judgment in its favor
by reason of the fact that he or she is or was a director, officer, employee or agent of the corporation, or is or was serving
at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture,
trust or other enterprise against expenses, including amounts paid in settlement and attorneys’ fees actually and reasonably incurred
by him or her in connection with the defense or settlement of the action or suit if he or she: (a) is not liable pursuant to NRS
78.138; or (b) acted in good faith and in a manner which he or she reasonably believed to be in or not opposed to the best interests
of the corporation. Indemnification may not be made for any claim, issue or matter as to which such a person has been adjudged
by a court of competent jurisdiction, after exhaustion of all appeals therefrom, to be liable to the corporation or for amounts
paid in settlement to the corporation, unless and only to the extent that the court in which the action or suit was brought or
other court of competent jurisdiction determines upon application that in view of all the circumstances of the case, the person
is fairly and reasonably entitled to indemnity for such expenses as the court deems proper.
NRS
Section 78.747 provides that, except as otherwise specifically provided by statute or agreement, no director or officer of a corporation
is individually liable for a debt or liability of the corporation, unless the director or officer acts as the alter ego of the
corporation. The court as a matter of law must determine the question of whether a director or officer acts as the alter ego of
a corporation.
Insofar
as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons
controlling Citius pursuant to the foregoing provisions, we have been informed that, in the opinion of the SEC, such indemnification
is against public policy as expressed in the Securities Act and is therefore unenforceable. In the event that a claim
for indemnification against such liabilities (other than the payment by us of expenses incurred or paid by a director, officer
or controlling person of Citius in the successful defense of any action, suit or proceeding) is asserted by such director, officer
or controlling person in connection with the securities being registered, we will, unless in the opinion of our counsel the matter
has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification
by us is against public policy as expressed hereby in the Securities Act and we will be governed by the final adjudication
of such issue.
Item
16. Exhibits.
Exhibit
Number
|
|
Description of Document
|
|
Registrant’s
Form
|
|
Dated
|
|
Exhibit
Number
|
|
Filed
Herewith
|
|
1.1*
|
|
Form of Underwriting Agreement.
|
|
--
|
|
--
|
|
--
|
|
|
|
3.1
|
|
Amended and Restated Articles of Incorporation of Citius Pharmaceuticals, Inc.
|
|
8-K
|
|
9/18/2014
|
|
3.1
|
|
|
|
3.2
|
|
Certificate of Amendment to the Amended and Restated Articles of Incorporation of Citius Pharmaceuticals, Inc., effective September 16, 2016.
|
|
8-K
|
|
9/21/2016
|
|
3.1
|
|
|
|
3.3
|
|
Certificate of Amendment to the Amended and Restated Articles of Incorporation of Citius Pharmaceuticals, Inc., effective June 9, 2017.
|
|
8-K
|
|
6/8/2017
|
|
3.1
|
|
|
|
3.4
|
|
Amended and Restated Bylaws of Citius Pharmaceuticals, Inc.
|
|
8-K
|
|
2/9/2018
|
|
3.1
|
|
|
|
4.1
|
|
Form of Registration Rights Agreement between the Purchasers named therein and Citius Pharmaceuticals Holdings, Inc., dated September 12, 2014.
|
|
8-K
|
|
9/18/2014
|
|
10.2
|
|
|
|
4.2
|
|
Placement Agent’s Unit Warrant in favor of Merriman Capital, Inc., dated September 12, 2014.
|
|
S-1/A
|
|
12/29/2015
|
|
10.12
|
|
|
|
4.3
|
|
Form of Investor Warrant, dated September 12, 2014.
|
|
8-K
|
|
9/18/2014
|
|
10.3
|
|
|
|
4.4
|
|
Form of Common Stock Purchase Warrant, dated May 10, 2017.
|
|
10-Q
|
|
5/15/2017
|
|
10.4
|
|
|
|
4.5
|
|
Form of Representative’s Warrant, dated August 3, 2017.
|
|
8-K
|
|
8/4/2017
|
|
4.2
|
|
|
|
4.6
|
|
Form of Investor Warrant, dated December 15, 2017.
|
|
8-K
|
|
12/19/2017
|
|
4.1
|
|
|
|
4.7
|
|
Form of Placement Agent Warrant, dated December 15, 2017.
|
|
8-K
|
|
12/19/2017
|
|
4.2
|
|
|
|
4.8
|
|
Form of Investor Warrant, dated March 28, 2018.
|
|
8-K
|
|
3/29/2018
|
|
4.1
|
|
|
|
4.9
|
|
Form of Placement Agent Warrant, dated March 28, 2018.
|
|
8-K
|
|
3/29/2018
|
|
4.2
|
|
|
|
4.10
|
|
Form of Common Stock Purchase Warrant, dated August 13, 2018.
|
|
8-K
|
|
8/13/2018
|
|
4.1
|
|
|
|
4.11
|
|
Form of Pre-Funded Common Stock Purchase Warrant, dated August 13, 2018.
|
|
8-K
|
|
8/13/2018
|
|
4.2
|
|
|
|
4.12
|
|
Form of Underwriter’s Common Stock Purchase Warrant, dated August 13, 2018.
|
|
8-K
|
|
8/13/2018
|
|
4.3
|
|
|
|
4.13
|
|
Form of Investor Warrant issued April 3, 2019.
|
|
8-K
|
|
4/03/2019
|
|
4.1
|
|
|
|
4.14
|
|
Form of Placement Agent Warrant issued April 3, 2019.
|
|
8-K
|
|
4/03/2019
|
|
4.2
|
|
|
|
4.15
|
|
Form of Common Stock Purchase Warrant issued September 27, 2019.
|
|
8-K
|
|
9/27/2019
|
|
4.1
|
|
|
|
4.16
|
|
Form of Pre-Funded Common Stock Purchase Warrant issued September 27, 2019.
|
|
8-K
|
|
9/27/2019
|
|
4.2
|
|
|
|
4.17
|
|
Form of Underwriters Common Stock Purchase Warrant issued September 27, 2019.
|
|
8-K
|
|
9/27/2019
|
|
4.3
|
|
|
|
4.18
|
|
Form of Investor Warrant issued on February 19, 2020.
|
|
8-K
|
|
2/19/2020
|
|
4.1
|
|
|
|
4.19
|
|
Form of Placement Agent Warrant issued on February 19, 2020.
|
|
8-K
|
|
2/19/2020
|
|
4.2
|
|
|
|
4.20
|
|
Form of Investor Warrant issued May 18, 2020.
|
|
8-K
|
|
5/18/2020
|
|
4.1
|
|
|
|
4.21
|
|
Form of Placement Agent Warrant issued May 18, 2020.
|
|
8-K
|
|
5/18/2020
|
|
4.2
|
|
|
|
4.22
|
|
Form of Underwriter Warrant issued August 10, 2020.
|
|
8-K
|
|
8/10/2020
|
|
4.1
|
|
|
|
4.23
|
|
Form of Investor Warrant issued January 27, 2021.
|
|
8-K
|
|
1/27/2021
|
|
4.1
|
|
|
|
4.24
|
|
Form of Placement Agent Warrant issued January 27, 2021.
|
|
8-K
|
|
1/27/2021
|
|
4.2
|
|
|
|
4.25
|
|
Registration Rights Agreement, dated January 24, 2021, by and among Citius Pharmaceuticals, Incl and the investors signatory thereto.
|
|
8-K
|
|
1/27/2021
|
|
4.3
|
|
|
|
4.26
|
|
Form of Investor Warrant issued February 19, 2021.
|
|
8-K
|
|
2/19/2021
|
|
4.1
|
|
|
|
4.27
|
|
Form of Placement Agent Warrant issued February 19, 2021.
|
|
8-K
|
|
2/19/2021
|
|
4.2
|
|
|
|
4.28*
|
|
Form of Certificate of Amendment to Amended and Restated Articles of Incorporation of Citius Pharmaceuticals, Inc.
|
|
--
|
|
--
|
|
--
|
|
|
|
4.29
|
|
Form of Indenture.
|
|
|
|
|
|
|
|
X
|
|
4.30*
|
|
Form of Note.
|
|
--
|
|
--
|
|
--
|
|
|
|
4.31*
|
|
Form of Common Stock Warrant Agreement and Warrant Certificate.
|
|
--
|
|
--
|
|
--
|
|
|
|
4.32*
|
|
Form of Preferred Stock Agreement and Warrant Certificate.
|
|
--
|
|
--
|
|
--
|
|
|
|
4.33*
|
|
Form of Debt Securities Warrant Agreement and Warrant Certificate.
|
|
--
|
|
--
|
|
--
|
|
|
|
4.34*
|
|
Form of Unit Agreement and Unit Certificate.
|
|
--
|
|
--
|
|
--
|
|
|
|
4.35*
|
|
Form of Rights Agreement and Rights Certificate.
|
|
--
|
|
--
|
|
--
|
|
|
|
5.1
|
|
Opinion of Wyrick Robbins Yates & Ponton, LLP.
|
|
|
|
|
|
|
|
X
|
|
23.1
|
|
Consent of Wolf & Company, P.C.
|
|
|
|
|
|
|
|
X
|
|
23.2
|
|
Consent of Wyrick Robbins Yates & Ponton LLP (included in Exhibit 5.1).
|
|
|
|
|
|
|
|
X
|
|
24.1
|
|
Power of Attorney (included on signature page).
|
|
|
|
|
|
|
|
X
|
|
25.1+
|
|
Statement of Eligibility of Trustee.
|
|
|
|
|
|
|
|
|
|
|
*
|
To be filed, if necessary, by amendment to the Registration
Statement or as an exhibit to a report filed under the Exchange Act and incorporated by reference herein.
|
|
|
+
|
To be filed separately pursuant to Section 305(b)(2) of
the Trust Indenture Act of 1939, as amended.
|
Item
17. Undertakings
|
|
(a)
|
The
undersigned registrant hereby undertakes:
|
|
|
(1)
|
To
file, during any period in which offers or sales are being made, a post-effective amendment to this Registration Statement:
|
|
|
(i)
|
To
include any prospectus required by Section 10(a)(3) of the Securities Act of 1933;
|
|
|
(ii)
|
To
reflect in the prospectus any facts or events arising after the effective date of the
registration statement (or the most recent post-effective amendment thereof) which, individually
or in the aggregate, represent a fundamental change in the information set forth in the
registration statement. Notwithstanding the foregoing, any increase or decrease in volume
of securities offered (if the total dollar value of securities offered would not exceed
that which was registered) and any deviation from the low or high end of the estimated
maximum offering range may be reflected in the form of prospectus filed with the SEC
pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent
no more than a 20% change in the maximum aggregate offering price set forth in the “Calculation
of Registration Fee” table in the effective registration statement;
|
|
|
(iii)
|
To
include any material information with respect to the plan of distribution not previously
disclosed in the registration statement or any material change to such information in
the registration statement;
|
provided,
however, that paragraphs (1)(i), (1)(ii) and (1)(iii) above do not apply if the information required to be included in
a post-effective amendment by those paragraphs is contained in reports filed with or furnished to the Commission by registrant
pursuant to Section 13 and Section 15(d) of the Securities Exchange Act of 1934 that are incorporated by reference in the registration
statement, or is contained in a form of prospectus filed pursuant to Rule 424(b) that is part of the registration statement.
|
|
(2)
|
That,
for the purpose of determining any liability under the Securities Act of 1933, each
such post-effective amendment shall be deemed to be a new registration statement relating
to the securities offered therein, and the offering of such securities at that time shall
be deemed to be the initial bona fide offering thereof.
|
|
|
(3)
|
To remove
from registration by means of a post-effective amendment any of the securities being
registered which remain unsold at the termination of the offering.
|
|
|
(5)
|
That,
for the purpose of determining liability under the Securities Act of 1933 to
any purchaser:
|
|
|
(A)
|
Each
prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the
date the filed prospectus was deemed part of and included in the registration statement; and
|
|
|
(B)
|
Each
prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5) or (b)(7) as part of a registration statement in reliance on
Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii) or (x) for the purpose of providing the information
required by Section 10(a) of the Securities Act of 1933 shall be deemed to be part of and included in the registration statement
as of the earlier of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale
of securities in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and
any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement
relating to the securities in the registration statement to which that prospectus relates, and the offering of such securities
at that time shall be deemed to be the initial bona fide offering thereof. Provided, however, that no statement made in a registration
statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by
reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with
a time of contract of sale prior to such effective date, supersede or modify any statement that was made in the registration statement
or prospectus that was part of the registration statement or made in any such document immediately prior to such effective date.
|
|
|
(6)
|
That,
for the purpose of determining liability of the registrant under the Securities
Act of 1933 to any purchaser in the initial distribution of the securities:
|
The
undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this Registration
Statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or
sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser
and will be considered to offer or sell such securities to such purchaser:
|
|
(i)
|
Any
preliminary prospectus or prospectus of the undersigned registrant relating to the offering
required to be filed pursuant to Rule 424;
|
|
|
(ii)
|
Any
free writing prospectus relating to the offering prepared by or on behalf of the undersigned
registrant or used or referred to by the undersigned registrant;
|
|
|
(iii)
|
The
portion of any other free writing prospectus relating to the offering containing material
information about the undersigned registrant or its securities provided by or on behalf
of the undersigned registrant; and
|
|
|
(iv)
|
Any
other communication that is an offer in the offering made by the undersigned registrant
to the purchaser.
|
|
|
(b)
|
The
undersigned registrant hereby undertakes that, for purposes of determining any liability
under the Securities Act of 1933, each filing of the registrant’s annual report
pursuant to Section 13(a) or Section 15(d) of the Securities Exchange Act of 1934 (and,
where applicable, each filing of an employee benefit plan’s annual report pursuant
to Section 15(d) of the Securities Exchange Act of 1934) that is incorporated by reference
in the registration statement shall be deemed to be a new registration statement relating
to the securities offered therein, and the offering of such securities at that time shall
be deemed to be the initial bona fide offering thereof.
|
|
|
(h)
|
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may
be permitted to directors, officers and controlling persons of the registrant pursuant
to the foregoing provisions, or otherwise, the registrant has been advised that in the
opinion of the Securities and Exchange Commission such indemnification is against public
policy as expressed in the Act and is, therefore, unenforceable. In the event that a
claim for indemnification against such liabilities (other than the payment by the registrant
of expenses incurred or paid by a director, officer or controlling person of the registrant
in the successful defense of any action, suit or proceeding) is asserted by such director,
officer or controlling person in connection with the securities being registered, the
registrant will, unless in the opinion of its counsel the matter has been settled by
controlling precedent, submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in the Act and will
be governed by the final adjudication of such issue.
|
|
|
(i)
|
The
undersigned registrant hereby undertakes that:
|
|
|
(1)
|
For
purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus
filed as part of this Registration Statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant
pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration
statement as of the time it was declared effective; and
|
|
|
(2)
|
For
the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a
form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering
of such securities at that time shall be deemed to be the initial bona fide offering thereof.
|
|
|
(j)
|
The
undersigned registrant hereby undertakes to file an application for the purpose of determining the eligibility of the trustee
to act under subsection (a) of section 310 of the Trust Indenture Act (“Act”) in accordance with the rules and regulations
prescribed by the Commission under section 305(b)(2) of the Act.
|
SIGNATURES
Pursuant
to the requirements of the Securities Act of 1933, the Registrant certifies that it has reasonable grounds to believe that it
meets all of the requirements for filing on Form S-3 and has duly caused this Registration Statement on Form S-3 to be signed
on its behalf by the undersigned, thereunto duly authorized, in the City of Cranford, State of New Jersey, on April 2, 2021.
|
|
CITIUS
PHARMACEUTICALS, INC.
|
|
|
|
|
|
|
By:
|
/s/
Myron Holubiak
|
|
|
|
Myron Holubiak
|
|
|
|
Chief Executive Officer
|
|
|
|
(Principal Executive
Officer)
|
POWER
OF ATTORNEY
KNOW
ALL BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Myron Holubiak and Leonard Mazur
as his or her true and lawful attorneys-in-fact and agents, each with the full power of substitution, for him of her and in his
or her name, place or stead, in any and all capacities, to sign any and all amendments to this registration statement (including
post-effective amendments), and any subsequent registration statement filed by the registrant pursuant to Rule 462(b) of
the Securities Act of 1933, as amended, which relates to this registration statement, and to file the same, with exhibits
thereto and other documents in connection therewith, with the SEC, granting unto said attorneys-in-fact and agents, and each of
them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about
the premises, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all
that said attorneys-in-fact and agents, or their or his or her substitute or substitutes, may lawfully do or cause to be done
by virtue hereof.
Pursuant
to the requirements of the Securities Act, this registration statement has been signed by the following persons in the capacities
and on the dates indicated.
|
Signature
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/
Myron Holubiak
|
|
President
and Chief Executive Officer
|
|
April
2, 2021
|
|
Myron Holubiak
|
|
(Principal Executive
Officer)
|
|
|
|
|
|
|
|
|
|
/s/
Jaime Bartushak
|
|
Chief
Financial Officer and Chief Accounting Officer
|
|
April
2, 2021
|
|
Jaime Bartushak
|
|
(Principal Financial
Officer and Principal Accounting Officer)
|
|
|
|
|
|
|
|
|
|
/s/
Leonard Mazur
|
|
Executive
Chairman, Board of Directors
|
|
April
2, 2021
|
|
Leonard Mazur
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Suren Dutia
|
|
Director
|
|
April
2, 2021
|
|
Suren Dutia
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Eugene Holuka
|
|
Director
|
|
April
2, 2021
|
|
Dr.
Eugene Holuka
|
|
|
|
|
|
|
|
|
|
|
|
/s/
William Kane
|
|
Director
|
|
April 2, 2021
|
|
Dr. William Kane
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Howard Safir
|
|
Director
|
|
April
2, 2021
|
|
Howard Safir
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Carol Webb
|
|
Director
|
|
April
2, 2021
|
|
Carol Webb
|
|
|
|
|
II-6
Citius Pharmaceuticals (NASDAQ:CTXR)
Historical Stock Chart
From Mar 2024 to Apr 2024
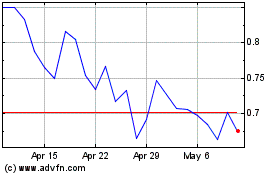
Citius Pharmaceuticals (NASDAQ:CTXR)
Historical Stock Chart
From Apr 2023 to Apr 2024