Vaxxinity Presents Preclinical Data on Tau Vaccine Candidates at the 2022 Alzheimer's Association International Conference (AAIC)
August 01 2022 - 8:00AM

Vaxxinity, Inc. (Nasdaq: VAXX), a U.S. company pioneering the
development of a new class of immunotherapeutic vaccines, presented
new preclinical data on vaccine candidates targeting aberrant forms
of the Tau protein for Alzheimer’s disease (AD) during a poster
session at the 2022 Alzheimer's Association International
Conference (AAIC).
Tau is an intracellular protein known to misfold
and accumulate in neurons in the form of neurofibrillary tangles
(NFTs) in the brain of AD patients. The formation of NFTs
correlates with the progression of AD symptoms and may even develop
prior to the first symptoms of the disease. Thus, targeting
pathological Tau before the onset of AD symptoms, such as memory
decline, could help prevent disease and/or progression.
“Our findings show that specific forms of Tau
can be targeted by vaccines with subsequent functional impact,”
said Justin Boyd, Ph.D., Director of Translational Science at
Vaxxinity. “This preclinical research complements UB-311, our
amyloid beta-targeting investigational vaccine which has been
granted Fast Track designation by the FDA and is in clinical
development for Alzheimer’s disease. Vaccines are uniquely suited
to deliver multiple antigens in a single formulation, and thus
rather than rely on selecting just one epitope, targeting multiple
Tau epitopes may provide a more efficient way of neutralizing all
toxic Tau species within one program. We look forward to the
continued evaluation of these candidates as potential innovative
solutions for patients who have been without options for far too
long.”
Vaxxinity’s proprietary synthetic peptide-based
technology platform enables both the targeting of endogenous
proteins like Tau, and the combination of multiple epitopes/targets
within a single formulation, a unique property that may prove
critical in the prevention and treatment of neurodegenerative
diseases where multiple pathologies may underlie disease
progression. Preclinical data demonstrate that our Tau peptide lead
vaccines are highly immunogenic and induce antibodies that display
diverse binding profiles against different forms of Tau peptides
(e.g., monomeric Tau, preformed Tau fibrils, or Tau extracted from
AD brain homogenates). Binding specificities of antibodies against
diverse Tau species were confirmed by ELISA, dot blot, and
Bio-Layer Interferometry analyses. Furthermore, using cell-based
models, we show that the antibodies induced by our lead vaccines
prevent intracellular aggregation of Tau and that this effect
reflects a blockade of Tau uptake into cells rather than a direct
blockade of Tau aggregation per se.
The poster, “Characterization of Tau vaccines
identifies diverse antibody binding and efficacy profiles,” can be
found on the Events & Presentations page of the company’s
website. The poster presentation video is viewable on the AAIC
conference website to registrants through Sept. 2, 2022 and will be
available on the Vaxxinity website after Sept. 3, 2022.
About Vaxxinity
Vaxxinity, Inc. is a purpose-driven
biotechnology company committed to democratizing healthcare across
the globe. The company is pioneering a new class of synthetic,
peptide-based immunotherapeutic vaccines aimed at disrupting the
existing treatment paradigm for chronic disease, increasingly
dominated by monoclonal antibodies, which suffer from prohibitive
costs and cumbersome administration. The company’s proprietary
technology platform has enabled the innovation of novel pipeline
candidates designed to bring the efficiency of vaccines to the
treatment of chronic diseases, including Alzheimer’s, Parkinson’s,
migraine, and hypercholesterolemia. The technology is also
implemented as part of a COVID-19 vaccine program. Vaxxinity has
optimized its pipeline to achieve a potentially historic, global
impact on human health.
For more information about Vaxxinity, Inc.,
visit http://www.vaxxinity.com and follow us on social media
@vaxxinity.
Forward-looking Statement
This press release includes forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995. The use of certain words, including “expect,”
“may,” “could,” “look forward,” “potentially,” “might,” “will,”
“aim” and similar expressions, are intended to identify
forward-looking statements. These forward-looking statements
involve substantial risks and uncertainties, and are based on the
current expectations and assumptions of Vaxxinity’s management.
Forward-looking statements include statements about the development
of a new class of immunotherapeutic vaccines and the innovation and
efficacy of Vaxxinity’s product candidates. Various important
factors could cause actual results or events to differ materially
from those that may be expressed or implied by our forward-looking
statements. Additional important factors to be considered in
connection with forward-looking statements are described in the
"Risk Factors" section of the Company's Annual Report on Form 10-K
filed with the Securities and Exchange Commission on March 24, 2022
and other reports we file with the Securities and Exchange
Commission. The forward-looking statements are made as of this date
and Vaxxinity does not undertake any obligation to update any
forward-looking statements, whether as a result of new information,
future events or otherwise, except as required by law.
Investor ContactBen
Matonebenm@vaxxinity.com
Press ContactKaren
ChaseVaxxinityPR@westwicke.com
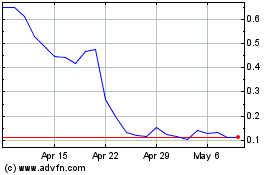
Vaxxinity (NASDAQ:VAXX)
Historical Stock Chart
From Mar 2024 to Apr 2024
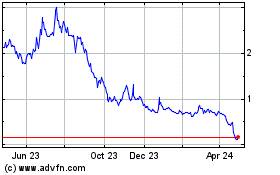
Vaxxinity (NASDAQ:VAXX)
Historical Stock Chart
From Apr 2023 to Apr 2024