Cerevel Therapeutics Announces First Patients Dosed in all Phase 3 Trials of Tavapadon for the Treatment of Parkinson’s Dis...
October 30 2020 - 7:00AM

Cerevel Therapeutics (NASDAQ:CERE), a company dedicated to
unraveling the mysteries of the brain
to treat neuroscience diseases, today announced that
the first participants have been dosed in all three of the clinical
trials in their Phase 3 program evaluating tavapadon in patients
with Parkinson’s disease. The company is tackling neuroscience
diseases with a differentiated approach that combines expertise in
neurocircuitry with a focus on receptor selectivity. Tavapadon is
an orally-bioavailable, once-daily partial agonist that selectively
targets dopamine D1/D5 receptor subtypes. It has been rationally
designed with the goal of balancing meaningful motor control
activity while minimizing the side effects typical of drugs that
non-selectively stimulate dopamine.
“We are encouraged by the benefit-risk profile
of tavapadon based on the efficacy results observed in Phase 2
trials, as well as the tolerability profile we have seen in our
clinical program to date,” said Raymond Sanchez, M.D., chief
medical officer of Cerevel Therapeutics. “We look forward to
advancing the development of tavapadon and potentially bringing a
differentiated, cornerstone therapy to Parkinson’s patients at all
stages of the disease as supported by a robust Phase 3
program.”
The Phase 3 program includes three 27-week,
double-blind, randomized, placebo-controlled, parallel-group trials
designed to evaluate the efficacy, safety and tolerability of fixed
doses (TEMPO-1) and flexible doses (TEMPO-2) of tavapadon as a
monotherapy in patients with early-stage Parkinson’s disease or as
an adjunctive therapy to levodopa in patients with late-stage
Parkinson’s disease who are experiencing motor fluctuations
(TEMPO-3).
Approximately 1,200 patients ages 40 to 80 years
will be enrolled across all three trials. The primary endpoint of
the TEMPO-1 and TEMPO-2 trials is the change from baseline in the
Movement Disorder Society-Unified Parkinson’s Disease Rating Scale
(MDS-UPDRS) Part II and Part III combined score. The primary
endpoint of the TEMPO-3 trial is the change from baseline in total
daily “on” time without troublesome dyskinesia. A fourth 58-week,
open-label, safety extension trial will also be conducted as part
of the program.
As the company’s most advanced therapeutic
program, tavapadon has been evaluated in 272 subjects in Phase 1
and Phase 2 trials, including in both early- and late-stage
Parkinson’s patient populations, which is required for a broad
indication in Parkinson’s disease. Across Phase 1b and Phase 2
trials conducted to date, tavapadon has demonstrated motor control
benefit with the potential for an improved tolerability profile
relative to D2/D3-preferring agonists.
Initiation of the registration-directed Phase 3
program for tavapadon began in January 2020. In response to the
COVID-19 pandemic, Cerevel Therapeutics paused screening and
enrollment in all trials in March 2020 and has remained vigilant
about participant safety and data integrity. Now that the program
is resuming, the company expects preliminary data readouts in the
first half of 2023.
About
TavapadonTavapadon is a potent,
orally-bioavailable, selective partial agonist of the dopamine D1
and D5 receptors. This investigational therapeutic is being
evaluated for the once-daily symptomatic treatment of Parkinson’s
disease.
About Parkinson’s
DiseaseApproximately 10 million people worldwide are
living with Parkinson’s disease, according to the Parkinson’s
Foundation. The disease is characterized by a progressive
degeneration of dopaminergic neurons (the main source of dopamine)
leading to a loss of critical motor and non-motor functions.
Symptom severity and disease progression differ between individuals
but typically include slowness of movement (bradykinesia),
trembling in the extremities (tremors), stiffness (rigidity),
cognitive or behavioral abnormalities, sleep disturbances and
sensory dysfunction.1 There is no laboratory or blood test for
Parkinson’s disease, so a diagnosis is made based on clinical
observation,2 which may contribute to an underestimation of the
incidence of the disease.
About Cerevel
TherapeuticsCerevel Therapeutics is dedicated to
unraveling the mysteries of the brain to treat neuroscience
diseases. The company is tackling neuroscience diseases with a
differentiated approach that combines expertise in neurocircuitry
with a focus on receptor selectivity. Cerevel Therapeutics has a
diversified pipeline comprising five clinical-stage investigational
therapies and several preclinical compounds with the potential to
treat a range of neuroscience diseases, including schizophrenia,
epilepsy, Parkinson’s disease and substance use disorder.
Headquartered in Boston, Cerevel Therapeutics is advancing its
current research and development programs while exploring new
modalities through internal research efforts, external
collaborations or potential acquisitions. For more information,
visit www.cerevel.com.
Special Note Regarding Forward-Looking
StatementsThis press release contains forward-looking
statements that are based on management’s beliefs and assumptions
and on information currently available to management. In some
cases, you can identify forward-looking statements by the following
words: “may,” “will,” “could,” “would,” “should,” “expect,”
“intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,”
“project,” “potential,” “continue,” “ongoing” or the negative of
these terms or other comparable terminology, although not all
forward-looking statements contain these words. These statements
involve risks, uncertainties and other factors that may cause
actual results, levels of activity, performance or achievements to
be materially different from the information expressed or implied
by these forward-looking statements. Although we believe that we
have a reasonable basis for each forward-looking statement
contained in this press release, we caution you that these
statements are based on a combination of facts and factors
currently known by us and our projections of the future, about
which we cannot be certain. Forward-looking statements in this
press release include, but are not limited to, statements about the
potential attributes and benefits of our product candidates and the
format and timing of our product development activities and
clinical trials. We cannot assure you that the forward-looking
statements in this press release will prove to be accurate.
Furthermore, if the forward-looking statements prove to be
inaccurate, the inaccuracy may be material. In light of the
significant uncertainties in these forward-looking statements, you
should not regard these statements as a representation or warranty
by us or any other person that we will achieve our objectives and
plans in any specified time frame, or at all. The forward-looking
statements in this press release represent our views as of the date
of this press release. We anticipate that subsequent events and
developments will cause our views to change. However, while we may
elect to update these forward-looking statements at some point in
the future, we have no current intention of doing so except to the
extent required by applicable law. You should, therefore, not rely
on these forward-looking statements as representing our views as of
any date subsequent to the date of this press release.
Media Contact:Rachel EidesW2O
purereides@purecommunications.com
Investor Contact:Matthew
CalistriCerevel Therapeuticsmatthew.calistri@cerevel.com
___________________
1 J Neurol Neurosurg Psychiatry. 2008;79:368-376.
doi:10.1136/jnnp.2007.131045.2 Cold Spring Harb Perspect Med.
2012;2:a008870.
Cerevel Therapeutics (NASDAQ:CERE)
Historical Stock Chart
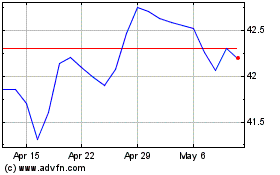
From Mar 2024 to Apr 2024
Cerevel Therapeutics (NASDAQ:CERE)
Historical Stock Chart
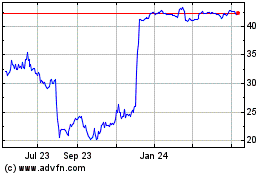
From Apr 2023 to Apr 2024